Association of Macronutrients Composition, Physical Activity and Serum Androgen Concentration in Young Women with Polycystic Ovary Syndrome
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Population
2.2. Medical and Biochemical Evaluation
2.3. Evaluation of Macronutrients Intake and PA
2.4. Statistical Analysis
3. Results
4. Discussion
4.1. Relationship between Fat Intake and Androgen Status
4.2. Relationship between Protein, Carbohydrates and Fiber Intake and Androgene Status
4.3. Relationship between PA and Androgen Status
5. Strengths and Limitations
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Teede, H.J.; Misso, M.L.; Costello, M.F.; Dokras, A.; Laven, J.; Moran, L.; Piltonen, T.; Norman, R.J. International PCOS Network. Recomendations from the international evidence-based guideline for the assessment and management of polycystic ovary syndrome. Hum. Reprod. 2018, 33, 1602–1618. [Google Scholar] [CrossRef]
- Teede, H.; Deeks, A.; Moran, L. Polycystic ovary syndrome: A complex condition with psychological, reproductive and metabolic manifestations that impacts on health across the lifespan. BMC Med. 2010, 8, 41. [Google Scholar] [CrossRef] [PubMed]
- Azziz, R.; Carmina, E.; Chen, Z.; Dunaif, A.; Laven, J.S.; Legro, R.S.; Lizneva, D.; Natterson-Horowtiz, B.; Teede, H.J.; Yildiz, B.O. Polycystic ovary syndrome. Nat. Rev. Dis. Primers 2016, 2, 16057. [Google Scholar] [CrossRef]
- Naz, M.S.G.; Tehrani, F.R.; Majd, H.A.; Ahmadi, F.; Ozgoli, G.; Fakari, F.R.; Ghasemi, V. The prevalence of polycystic ovary syndrome in adolescents: A systematic review and meta-analysis. Int. J. Reprod. Biomed. 2019, 17, 533–542. [Google Scholar] [PubMed]
- Barrea, L.; Frias-Toral, E.; Verde, L.; Ceriani, F.; Cucalón, G.; Garcia-Velasquez, E.; Moretti, D.; Savastano, S.; Colao, A.; Muscogiuri, G. PCOS and nutritional approaches: Differences between lean and obese phenotype. Metab. Open 2021, 12, 100123. [Google Scholar] [CrossRef]
- Lizneva, D.; Suturina, L.; Walker, W.; Brakta, S.; Gavrilova-Jordan, L.; Azziz, R. Criteria, prevalence, and phenotypes of polycystic ovary syndrome. Fertil. Steril. 2016, 106, 6–15. [Google Scholar] [CrossRef]
- Anagnostis, P.; Tarlatzis, B.C.; Kauffman, R.P. Polycystic ovarian syndrome (PCOS): Long-term metabolic consequences. Metabolism 2018, 86, 33–43. [Google Scholar] [CrossRef] [PubMed]
- Sirmans, S.M.; Pate, K.A. Epidemiology, diagnosis, and management of polycystic ovary syndrome. Clin. Epidemiol. 2013, 6, 1–13. [Google Scholar] [CrossRef]
- Teede, H.J.; Joham, A.E.; Paul, E.; Moran, L.J.; Loxton, D.; Jolley, D.; Lombard, C. Longitudinal weight gain in women identified with polycystic ovary syndrome: Results of an observational study in young women. Obesity 2013, 21, 1526–1532. [Google Scholar] [CrossRef]
- Greenwood, E.A.; Kao, C.-N.; Cedars, M.I.; Huddleston, H.G. On your feet: Is sitting time linked to adverse metabolic profiles in polycystic ovary syndrome, independent of exercise? Fertil. Steril. 2017, 107, e40–e41. [Google Scholar] [CrossRef][Green Version]
- Turner-Mcgrievy, G.; Davidson, C.R.; Billings, D.L. Dietary intake, eating behaviors, and quality of life in women with polycystic ovary syndrome who are trying to conceive. Hum. Fertil. 2014, 18, 16–21. [Google Scholar] [CrossRef] [PubMed]
- Goodman, N.F.; Cobin, R.H.; Futterweit, W.; Glueck, J.S.; Legro, R.S.; Carmina, E. Guide to the Best Practices in the Evaluation and Treatment of Polycystic Ovary Syndrome: Part 1. Endocr Pract. 2015, 21, 1291–1300. [Google Scholar] [CrossRef] [PubMed]
- Sepilian, V.P.; Crochet, J.R.; Nagamani, M. Serum soluble leptin receptor levels and free leptin index in women with polycystic ovary syndrome: Relationship to insulin resistance and androgens. Fertil. Steril. 2006, 85, 1441–1447. [Google Scholar] [CrossRef] [PubMed]
- Peña, A.S.; Witchel, S.F.; Hoeger, K.M.; Oberfield, S.E.; Vogiatzi, M.G.; Misso, M.; Garad, R.; Dabadghao, P.; Teede, H. Adolescent polycystic ovary syndrome according to the international evidence-based guideline. BMC Med. 2020, 18, 72. [Google Scholar] [CrossRef]
- Tay, C.T.; Hart, R.J.; Hickey, M.; Moran, L.J.; Earnest, A.; Doherty, D.A.; Teede, H.J.; Joham, A.E. Updated adolescent diagnostic criteria for polycystic ovary syndrome: Impact on prevalence and longitudinal body mass index trajectories from birth to adulthood. BMC Med. 2020, 18, 389. [Google Scholar] [CrossRef]
- Witchel, S.F.; Oberfield, S.E.; Peña, A.S. Polycystic Ovary Syndrome: Pathophysiology, Presentation, and Treatment With Emphasis on Adolescent Girls. J. Endocr. Soc. 2019, 3, 1545–1573. [Google Scholar] [CrossRef]
- Vryonidou, A.; Paschou, S.A.; Muscogiuri, G.; Orio, F.; Goulis, D.G. Mechanisms in endocrinology: Metabolic syndrome through the female life cycle. Eur. J. Endocrinol. 2015, 173, R153–R163. [Google Scholar] [CrossRef]
- Satyaraddi, A.; Cherian, K.; Kapoor, N.; Kunjummen, A.; Kamath, M.; Thomas, N. Body composition, metabolic characteristics, and insulin resistance in obese and nonobese women with polycystic ovary syndrome. J. Hum. Reprod. Sci. 2019, 12, 78–84. [Google Scholar]
- Lorenz, L.B.; Wild, R.A. Polycystic ovarian syndrome: An evidence-based approach to evaluation and management of diabetes and cardiovascular risks for today’s clinician. Clin. Obstet. Gynecol. 2007, 50, 226–243. [Google Scholar] [CrossRef]
- Norman, R.J.; Davies, M.J.; Lord, J.; Moran, L.J. The role of lifestyle modification in polycystic ovary syndrome. Trends Endocrinol. Metab. 2002, 13, 251–257. [Google Scholar] [CrossRef]
- Álvarez-Blasco, F.; Luque-Ramírez, M.; Escobar-Morreale, H.F. Diet composition and physical activity in overweight and obese premenopausal women with or without polycystic ovary syndrome. Gynecol. Endocrinol. 2011, 27, 978–981. [Google Scholar] [CrossRef] [PubMed]
- Perelman, D.; Coghlan, N.; Lamendola, C.; Carter, S.; Abbasi, F.; McLaughlin, T. Substituting poly- and mono-unsaturated fat for dietary carbohydrate reduces hyperinsulinemia in women with polycystic ovary syndrome. Gynecol. Endocrinol. 2016, 33, 324–327. [Google Scholar] [CrossRef] [PubMed]
- Ciebiera, M.; Esfandyari, S.; Siblini, H.; Prince, L.; Elkafas, H.; Wojtyła, C.; Al-Hendy, A.; Ali, M. Nutrition in gynecological diseases: Current perspectives. Nutrients 2021, 13, 1178. [Google Scholar] [CrossRef] [PubMed]
- Afrin, S.; Alashqar, A.; El Sabeh, M.; Reschke, L.; Brennan, J.T.; Fader, A.; Borahay, M. Diet and nutrition in gynecological disorders: A focus on clinical studies. Nutrients 2021, 13, 1747. [Google Scholar] [CrossRef]
- Moran, L.J.; Ko, H.; Misso, M.; Marsh, K.; Noakes, M.; Talbot, M.; Frearson, M.; Thondan, M.; Stepto, N.; Teede, H. Dietary Composition in the Treatment of Polycystic Ovary Syndrome: A Systematic Review to Inform Evidence-Based Guidelines. J. Acad. Nutr. Diet. 2013, 113, 520–545. [Google Scholar] [CrossRef] [PubMed]
- Phelan, N.; O’Connor, A.; Tun, T.K.; Correia, N.; Boran, G.; Roche, H.M.; Gibney, J. Hormonal and metabolic effects of polyunsaturated fatty acids in young women with polycystic ovary syndrome: Results from a cross-sectional analysis and a randomized, placebo-controlled, crossover trial. Am. J. Clin. Nutr. 2011, 93, 652–662. [Google Scholar] [CrossRef]
- Eleftheriadou, M.; Stefanidis, K.; Lykeridou, K.; Iliadis, I.; Michala, L. Dietary habits in adolescent girls with polycystic ovarian syndrome. Gynecol. Endocrinol. 2014, 31, 269–271. [Google Scholar] [CrossRef]
- Eleftheriadou, M.; Michala, L.; Stefanidis, K.; Iliadis, I.; Lykeridou, A.; Antsaklis, A. Exercise and Sedentary Habits Among Adolescents with PCOS. J. Pediatric Adolesc. Gynecol. 2012, 25, 172–174. [Google Scholar] [CrossRef]
- Cui, X.; Rosner, B.; Willett, W.C.; Hankinson, S.E. Dietary Fat, Fiber, and Carbohydrate Intake and Endogenous Hormone Levels in Premenopausal Women. Horm. Cancer 2010, 1, 265–276. [Google Scholar] [CrossRef]
- Rotterdam ESHRE/ASRM-Sponsored PCOS Consensus Workshop Group. Revised 2003 consensus on diagnostic criteria and long-term health risks related to polycystic ovary syndrome. Hum. Reprod. 2004, 19, 41–47. [Google Scholar] [CrossRef]
- Mizgier, M.; Jarząbek-Bielecka, G.; Formanowicz, D.; Jodłowska-Siewert, E.; Mruczyk, K.; Cisek-Woźniak, A.; Cisek-Woźniak, A.; Kędzia, W.; Opydo-Szymaczek, J. Dietary and Physical Activity Habits in Adolescent Girls with Polycystic Ovary Syndrome (PCOS)-HAstudy. J. Clin. Med. 2021, 10, 3469. [Google Scholar] [CrossRef]
- Mizgier, M.; Jarząbek-Bielecka, G.; Opydo-Szymaczek, J.; Wendland, N.; Więckowska, B.; Kędzia, W. Risk Factors of Overweight and Obesity Related to Diet and Disordered Eating Attitudes in Adolescent Girls with Clinical Features of Polycystic Ovary Syndrome. J. Clin. Med. 2020, 9, 3041. [Google Scholar] [CrossRef] [PubMed]
- Wendland, N.; Opydo-Szymaczek, J.; Formanowicz, D.; Blacha, A.; Jarząbek-Bielecka, G.; Mizgier, M. Association between metabolic and hormonal profile, proinflammatory cytokines in saliva and gingival health in adolescent females with polycystic ovary syndrome. BMC Oral Health 2021, 21, 193. [Google Scholar] [CrossRef] [PubMed]
- Wendland, N.; Opydo-Szymaczek, J.; Mizgier, M.; Jarząbek-Bielecka, G. Subgingival microflora in adolescent females with polycystic ovary syndrome and its association with oral hygiene, gingivitis, and selected metabolic and hormonal parameters. Clin. Oral Investig. 2021, 25, 1485–1496. [Google Scholar] [CrossRef]
- Mizgier, M.; Jarząbek-Bielecka, G.; Wendland, N.; Jodłowska-Siewert, E.; Nowicki, M.; Brożek, A.; Kędzia, W.; Formanowicz, D.; Opydo-Szymaczek, J. Relation between Inflammation, Oxidative Stress, and Macronutrient Intakes in Normal and Excessive Body Weight Adolescent Girls with Clinical Features of Polycystic Ovary Syndrome. Nutrients 2021, 13, 896. [Google Scholar] [CrossRef] [PubMed]
- Kowalkowska, J.; Wadolowska, L.; Czarnocinska, J.; Czlapka-Matyasik, M.; Galinski, G.; Jezewska-Zychowicz, M.; Bronkowska, M.; Dlugosz, A.; Loboda, D.; Wyka, J. Reproducibility of a questionnaire for dietary habits, lifestyle and nutrition knowledge assessment (KomPAN) in Polish adolescents and adults. Nutrients 2018, 10, 1845. [Google Scholar] [CrossRef]
- Ballerini, M.G.; Gaido, V.; Rodríguez, M.E.; Chiesa, A.; Ropelato, M.G. Prospective and Descriptive Study on Serum Androstenedione Concentration in Healthy Children from Birth until 18 Years of Age and Its Associated Factors. Dis. Markers 2017, 2017, 9238304. [Google Scholar] [CrossRef]
- Forrester-Dumont, K.; Galescu, O.; Kolesnikov, A.; RAissouni, N.; Bhangoo, A.; Ten, S.; Suss, A. Hyperandrogenism does not influence metabolic parameters in adolescent girls with PCOS. Int. J. Endocrinol. 2012, 2012, 434830. [Google Scholar] [CrossRef]
- Orio, F.; Muscogiuri, G.; Ascione, A.; Marciano, F.; Volpe, A.; La Sala, G.; Colao, A.; Palomba, S. Effects of physical exercise on the female reproductive system. Minerva Endocrinol. 2013, 38, 305–319. [Google Scholar]
- Whittaker, J.; Wu, K. Low-fat diets and testosterone in men: Systematic review and meta-analysis of intervention studies. J. Steroid Biochem. Mol. Biol. 2021, 210, 105878. [Google Scholar] [CrossRef]
- Wang, C.; Catlin, D.H.; Starcevic, B.; Heber, D.; Ambler, C.; Berman, N.; Lucas, G.; Leung, A.; Schramm, K.; Lee, P.W.N.; et al. Low-fat high-fiber diet decreased serum and urine androgens in men. J. Clin. Endocrinol. Metab. 2005, 90, 3550–3559. [Google Scholar] [CrossRef] [PubMed]
- Dorgan, J.F.; Judd, J.T.; Longcope, C.; Brown, C.; Schatzkin, A.; Clevidence, B.A.; Campbell, W.S.; Nair, P.P.; Franz, C.; Kahle, L.; et al. Effects of dietary fat and fiber on plasma and urine androgens and estrogens in men: A controlled feeding study. Am. J. Clin. Nutr. 1996, 64, 850–855. [Google Scholar] [CrossRef] [PubMed]
- Mai, K.; Bobbert, T.; Kullmann, V.; Andres, J.; Rochlitz, H.; Osterhoff, M.; Weickert, M.O.; Bähr, V.; Mohlig, M.; Pfeiffer, A.F.H.; et al. Free fatty acids increase androgen precursors in vivo. J. Clin. Endocrinol. Metab. 2006, 91, 1501–1507. [Google Scholar] [CrossRef] [PubMed]
- Mai, K.; Bobbert, T.; Reinecke, F.; Andres, J.; Bähr, V.; Maser-Gluth, C.; Schulte, H.; Diederich, S.; Pfeiffer, A.F.; Spranger, J. Free fatty acids increase androgen precursors in vivo in young healthy women. Exp. Clin. Endocrinol. Diabetes 2007, 115, P02–P42. [Google Scholar] [CrossRef]
- Faghfoori, Z.; Fazelian, S.; Shadnoush, M.; Goodarzi, R. Nutritional management in women with polycystic ovary syndrome: A review study. Diabetes Metab. Syndr. Clin. Res. Rev. 2017, 11, S429–S432. [Google Scholar] [CrossRef]
- Parker, D.R.; Weiss, S.T.; Troisi, R.; Cassano, P.A.; Vokonas, P.S.; Landsberg, L. Relationship of dietary saturated fatty acids and body habitus to serum insulin concentrations: The normative aging study. Am. J. Clin. Nutr. 1993, 58, 129–136. [Google Scholar] [CrossRef]
- Katcher, H.I.; Kunselman, A.R.; Dmitrovic, R.; Demers, L.M.; Gnatuk, C.L.; Kris-Etherton, P.M.; Legro, R.S. Comparison of hormonal and metabolic markers after a high-fat, Western meal versus a low-fat, high-fiber meal in women with polycystic ovary syndrome. Fertil. Steril. 2009, 91, 1175–1182. [Google Scholar] [CrossRef]
- Kalgaonkar, S.; Almario, R.U.; Gurusinghe, D.; Garamendi, E.M.; Buchan, W.; Kim, K.; Karakas, S.E. Differential effects of walnuts vs almonds on improving metabolic and endocrine parameters in PCOS. Eur. J. Clin. Nutr. 2011, 65, 386–393. [Google Scholar] [CrossRef]
- Yuan, J.; Wen, X.; Jia, M. Efficacy of omega-3 polyunsaturated fatty acids on hormones, oxidative stress, and inflammatory parameters among polycystic ovary syndrome: A systematic review and meta-analysis. Ann. Palliat. Med. 2021, 10, 8991–9001. [Google Scholar] [CrossRef]
- Barrea, L.; Arnone, A.; Annunziata, G.; Muscogiuri, G.; Laudisio, D.; Salzano, C.; Pugliese, G.; Colao, A.; Savastano, S. Adherence to the mediterranean diet, dietary patterns and body composition in women with polycystic ovary syndrome (PCOS). Nutrients 2019, 11, 2278. [Google Scholar] [CrossRef]
- Kasim-Karakas, S.E.; Almario, R.U.; Gregory, L.; Wong, R.; Todd, H.; Lasley, B.L. Metabolic and Endocrine Effects of a Polyunsaturated Fatty Acid-Rich Diet in Polycystic Ovary Syndrome. J. Clin. Endocrinol. Metab. 2004, 89, 615–620. [Google Scholar] [CrossRef] [PubMed]
- Hajishafiee, M.; Askari, G.; Iranj, B.; Ghiasvand, R.; Bellissimo, N.; De Zepetnek, J.T.; Salehi-Abargouei, A. The Effect of n-3 Polyunsaturated Fatty Acid Supplementation on Androgen Status in Patients with Polycystic Ovary Syndrome: A Systematic Review and Meta-Analysis of Clinical Trials. Horm. Metab. Res. 2016, 48, 281–289. [Google Scholar] [CrossRef] [PubMed]
- Gower, B.A.; Chandler-Laney, P.C.; Ovalle, F.; Goree, L.L.; Azziz, R.; Desmond, R.A.; Granger, W.M.; Goss, A.M.; Bates, G.W. Favourable metabolic effects of a eucaloric lower-carbohydrate diet in women with PCOS. Clin. Endocrinol. 2013, 79, 550–557. [Google Scholar] [CrossRef]
- Douglas, C.C.; Gower, B.A.; Darnell, B.E.; Ovalle, F.; Oster, R.A.; Azziz, R. Role of diet in the treatment of polycystic ovary syndrome. Fertil. Steril. 2006, 85, 679–688. [Google Scholar] [CrossRef] [PubMed]
- Mavropoulos, J.C.; Yancy, W.S.; Hepburn, J.; Westman, E.C. The effects of a low-carbohydrate, ketogenic diet on the polycystic ovary syndrome: A pilot study. Nutr. Metab. 2005, 2, 35. [Google Scholar] [CrossRef]
- Neves, L.P.P.; Marcondes, R.R.; Maffazioli, G.D.N.; Simões, R.S.; Maciel, G.A.R.; Soares, J.M.; Baracat, E.C. Nutritional and dietary aspects in polycystic ovary syndrome: Insights into the biology of nutritional interventions. Gynecol. Endocrinol. 2020, 36, 1047–1050. [Google Scholar] [CrossRef]
- Connolly, A.; Leblanc, S.; Baillargeon, J.P. Role of lipotoxicity and contribution of the renin-angiotensin system in the development of polycystic ovary syndrome. Int. J. Endocrinol. 2018, 2018, 4315413. [Google Scholar] [CrossRef]
- Mehrabani, H.H.; Salehpour, S.; Meyer, B.J.; Tahbaz, F. Beneficial effects of a high-protein, low-glycemic-load hypocaloric diet in overweight and obese women with polycystic ovary syndrome: A randomized controlled intervention study. J. Am. Coll. Nutr. 2012, 31, 117–125. [Google Scholar] [CrossRef]
- Cutler, D.A.; Pride, S.M.; Cheung, A.P. Low intakes of dietary fiber and magnesium are associated with insulin resistance and hyperandrogenism in polycystic ovary syndrome: A cohort study. Food Sci. Nutr. 2019, 7, 1426–1437. [Google Scholar] [CrossRef]
- Björck, I.; Elmståhl, H.L. The glycaemic index: Importance of dietary fibre and other food properties. Proc. Nutr. Soc. 2003, 62, 201–206. [Google Scholar] [CrossRef]
- Moran, L.J.; Noakes, M.; Clifton, P.M.; Tomlinson, L.; Norman, R.J. Dietary composition in restoring reproductive and metabolic physiology in overweight women with polycystic ovary syndrome. J. Clin. Endocrinol. Metab. 2003, 88, 812–819. [Google Scholar] [CrossRef] [PubMed]
- Lin, A.W.; Siscovick, D.; Sternfeld, B.; Schreiner, P.; Lewis, C.E.; Wang, E.T.; Merkin, S.S.; Wellons, M.; Steffen, L.; Calderon-Margalit, R.; et al. Associations of diet, physical activity and polycystic ovary syndrome in the Coronary Artery Risk Development in Young Adults Women’s Study. BMC Public Health 2021, 21, 35. [Google Scholar] [CrossRef]
- Shele, G.; Genkil, J.; Speelman, D. A systematic review of the effects of exercise on hormones in women with polycystic ovary syndrome. J. Funct. Morphol. Kinesiol. 2020, 5, 35. [Google Scholar] [CrossRef] [PubMed]
- Abdolahian, S.; Tehrani, F.R.; Amiri, M.; Ghodsi, D.; Yarandi, R.B.; Jafari, M.; Majd, H.A.; Nahidi, F. Effect of lifestyle modifications on anthropometric, clinical, and biochemical parameters in adolescent girls with polycystic ovary syndrome: A systematic review and meta-analysis. BMC Endocr. Disord. 2020, 20, 71. [Google Scholar] [CrossRef] [PubMed]
- Lombardi, G.; Ziemann, E.; Banfi, G.; Corbetta, S. Physical activity-dependent regulation of parathyroid hormone and calcium-phosphorous metabolism. Int. J. Mol. Sci. 2020, 21, 5388. [Google Scholar] [CrossRef] [PubMed]
- Lane, A.R.; Hackney, A.C. Relationship between salivary and serum testosterone levels in response to different exercise intensities. Hormones 2015, 14, 258–264. [Google Scholar]
- Hayes, L.D.; Grace, F.M.; Baker, J.S.; Sculthorpe, N. Exercise-Induced Responses in Salivary Testosterone, Cortisol, and Their Ratios in Men: A Meta-Analysis. Sports Med. 2015, 45, 713–726. [Google Scholar] [CrossRef]
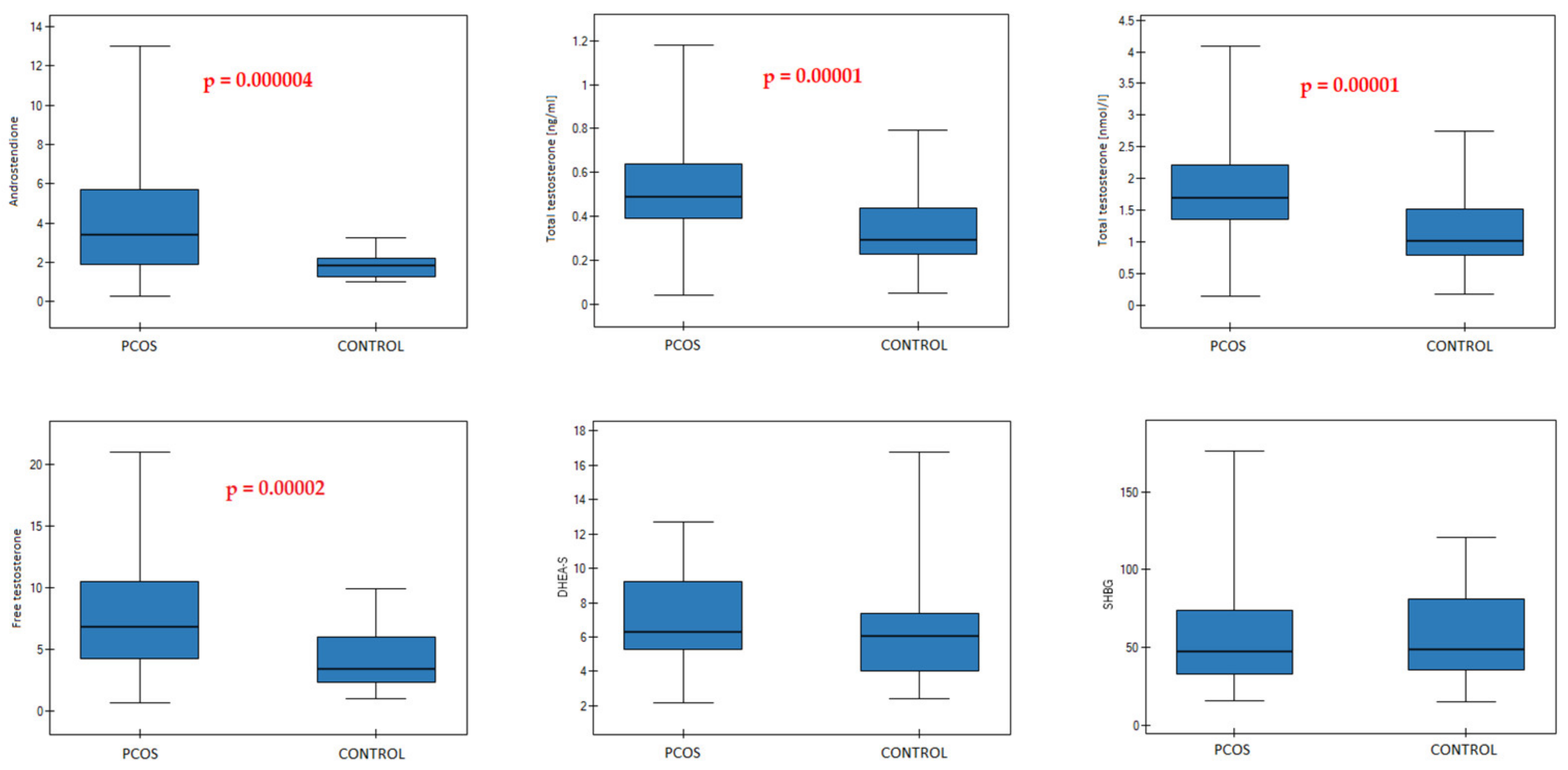
| PCOS n = 61 | CONTROL n = 35 | p-Value | |
|---|---|---|---|
| Androstenedione (ng/mL) | 0.000004 | ||
| 3.4 (1.9–5.7) | 1.85 (1.26–2.21) | ||
| Total T (ng/mL) | 0.00001 | ||
| 0.49 (0.39–0.64) | 0.29 (0.23–0.44) | ||
| Total T (nmol/L) | 0.00001 | ||
| 1.7 (1.35–2.22) | 1.01 (0.78–1.51) | ||
| Free T (ng/L) | 0.00002 | ||
| 6.8 (4.2–10.5) | 3.38 (2.34–6) | ||
| DHEA-S (μmol/L) | 0.052 | ||
| 6.32 (5.31–9.25) | 6.05 (4.05–7.39) | ||
| SHBG (nmol/L) | 0.648 | ||
| 47.55 (32.96–73.8) | 48.96 (35.29–81.38) |
| Variables | Total Protein [g/100 kcal] | Total Fat [g/100 kcal] | Carbohydrates [g/100 kcal] | Fiber [g/100 kcal] | Plant Protein [g/100 kcal] | SFA [g/100 kcal] | MUFA [g/100 kcal] | PUFA [g/100 kcal] | Cholesterol [mg/100 kcal] | PA (Work/School) | PA (Leisure Time) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| A p-value | 0.024 | 0.013 | 0.118 | 0.002 | 0.009 | 0.324 | 0.017 | 0.051 | 0.403 | 0.0004 | 0.0003 |
| r | −0.231 | 0.253 | −0.160 | −0.313 | −0.266 | 0.102 | 0.244 | 0.200 | −0.086 | −0.359 | −0.365 |
| Total T p-value | 0.002 | 0.015 | 0.194 | 0.009 | 0.042 | 0.233 | 0.138 | 0.128 | 0.263 | 0.001 | 0.015 |
| r | −0.319 | 0.248 | −0.134 | −0.266 | −0.208 | 0.123 | 0.152 | 0.156 | −0.115 | −0.333 | −0.248 |
| Free T | |||||||||||
| p-value | 0.111 | 0.004 | 0.039 | 0.004 | 0.024 | 0.143 | 0.020 | 0.045 | 0.925 | 0.001 | 0.0002 |
| r | −0.164 | 0.293 | −0.211 | −0.288 | −0.230 | 0.150 | 0.238 | 0.205 | 0.010 | −0.344 | −0.375 |
| DHEA-S p-value | 0.126 | 0.569 | 0.974 | 0.868 | 0.505 | 0.820 | 0852 | 0.382 | 0.745 | 0.173 | 0.381 |
| r | −0.157 | 0.059 | −0.003 | −0.017 | −0.069 | −0.024 | 0.019 | 0.090 | −0.034 | −0.143 | −0.090 |
| SHBG p-value | 0.100 | 0.680 | 0.830 | 0.245 | 0.539 | 0.558 | 0.243 | 0.129 | 0.403 | 0.094 | 0.0005 |
| r | 0.169 | −0.043 | −0.022 | 0.120 | 0.063 | 0.060 | −0.120 | −0.156 | 0.086 | 0.175 | 0.351 |
| Variables | Total Protein [g/100 kcal] | Total Fat [g/100 kcal] | Carbohydrates [g/100 kcal] | Fiber [g/100 kcal] | Plant Protein [g/100 kcal] | SFA [g/100 kcal] | MUFA [g/100 kcal] | PUFA [g/100 kcal] | Cholesterol [mg/100 kcal] | PA (Work/School) | PA (Leisure Time) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| A p-value | 0.425 | 0.359 | 0.547 | 0.069 | 0.141 | 0.874 | 0.164 | 0.605 | 0.643 | 0.029 | 0.001 |
| r | −0.104 | 0.120 | −0.079 | −0.235 | −0.191 | −0.021 | 0.180 | 0.068 | −0.062 | −0.282 | −0.404 |
| Total T p-value | 0.415 | 0.497 | 0.599 | 0.064 | 0.079 | 0.700 | 0.703 | 0.404 | 0.515 | 0.183 | 0.132 |
| r | −0.106 | 0.089 | −0.069 | −0.239 | −0.227 | 0.050 | 0.050 | −0.109 | −0.085 | −0.174 | −0.195 |
| Free T p-value | 0.436 | 0.387 | 0.556 | 0.059 | 0.100 | 0.943 | 0.257 | 0.862 | 0.660 | 0.045 | 0.003 |
| r | −0.102 | 0.113 | −0.077 | −0.243 | −0.213 | −0.009 | 0.147 | 0.023 | −0.057 | −0.260 | −0.374 |
| DHEA-S p-value | 0.660 | 0.932 | 0.871 | 0.938 | 0.225 | 0.782 | 0.797 | 0.615 | 0.638 | 0.899 | 0.899 |
| r | −0.058 | 0.011 | 0.021 | −0.010 | −0.148 | −0.036 | −0.034 | −0.066 | 0.061 | 0.017 | −0.017 |
| SHBG p-value | 0.734 | 0.229 | 0.372 | 0.132 | 0.264 | 0.880 | 0.062 | 0.196 | 0.892 | 0.018 | 0.0002 |
| r | 0.044 | −0.156 | 0.116 | 0.195 | 0.145 | 0.020 | −0.240 | −0.168 | 0.018 | 0.304 | 0.457 |
| Variables | Total Protein [g/100 kcal] | Total Fat [g/100 kcal] | Total Carbohydrates [g/100 kcal] | Fiber [g/100 kcal] | Plant Protein [g/100 kcal] | SFA [g/100 kcal] | MUFA [g/100 kcal] | PUFA [g/100 kcal] | Cholesterol [mg/100 kcal] | PA (Work/School) | PA (Leisure Time) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| A p-value | 0.123 | 0.074 | 0.458 | 0.546 | 0.791 | 0.708 | 0.534 | 0.404 | 0.676 | 0.895 | 0.193 |
| r | −0.265 | 0.305 | −0.130 | −0.106 | −0.047 | 0.066 * | 0.109 | 0.146 | 0.073 | 0.024 | 0.225 |
| Total T p-value | 0.007 | 0.467 | 0.612 | 0.835 | 0.527 | 0.708 | 0.748 | 0.223 | 0.896 | 0.319 | 0.997 |
| r | −0.448 * | 0.127 * | 0.089 * | −0.037 | 0.111 * | 0.066 * | 0.056 | 0.211 | −0.023 | −0.179 | 0.001 |
| Free T p-value | 0.544 | 0.026 | 0.135 | 0.491 | 0.794 | 0.013 | 0.452 | 0.403 | 0.116 | 0.665 | 0.651 |
| r | −0.106 | 0.375 | −0.258 | −0.120 | 0.046 | 0.417 | 0.131 | 0.146 | 0.270 | −0.078 | −0.079 |
| DHEA-S p-value | 0.284 | 0.607 | 0.322 | 0.235 | 0.173 | 0.707 | 0.117 | 0.331 | 0.512 | 0.190 | 0.686 |
| r | −0.186 | −0.090 | 0.173 | 0.206 | 0.236 | −0.066 | −0.270 | 0.169 | −0.115 | −0.234 | −0.071 |
| SHBG p-value | 0.022 | 0.196 | 0.025 | 0.607 | 0.244 | 0.294 | 0.152 | 0.844 | 0.240 | 0.579 | 0.394 |
| r | 0.387 | 0.224 | −0.379 | −0.090 | −0.202 | 0.182 | 0.247 | −0.034 | 0.204 | −0.100 | 0.149 |
| PA (Leisure) | A (ng/mL) | p-Value | Dunn–Bonferroni | Low | Moderate | High |
|---|---|---|---|---|---|---|
| low | 5.20 (3.40–8.65) | 0.007 | low | 0.221 | 0.005 | |
| moderate | 3.60 (1.85–5.65) | moderate | 0.221 | 0.269 | ||
| high | 2.01 (1.66–3.07) | high | 0.005 | 0.269 | ||
| PA (leisure) | Free T (ng/mL) | p-value | Dunn–Bonferroni | Low | moderate | high |
| low | 8.35 (7.05–13.03) | 0.015 | low | 0.298 | 0.011 | |
| moderate | 6.60 (4.15–9.45) | moderate | 0.298 | 0.339 | ||
| high | 4.35 (3.93–6.45) | high | 0.011 | 0.339 | ||
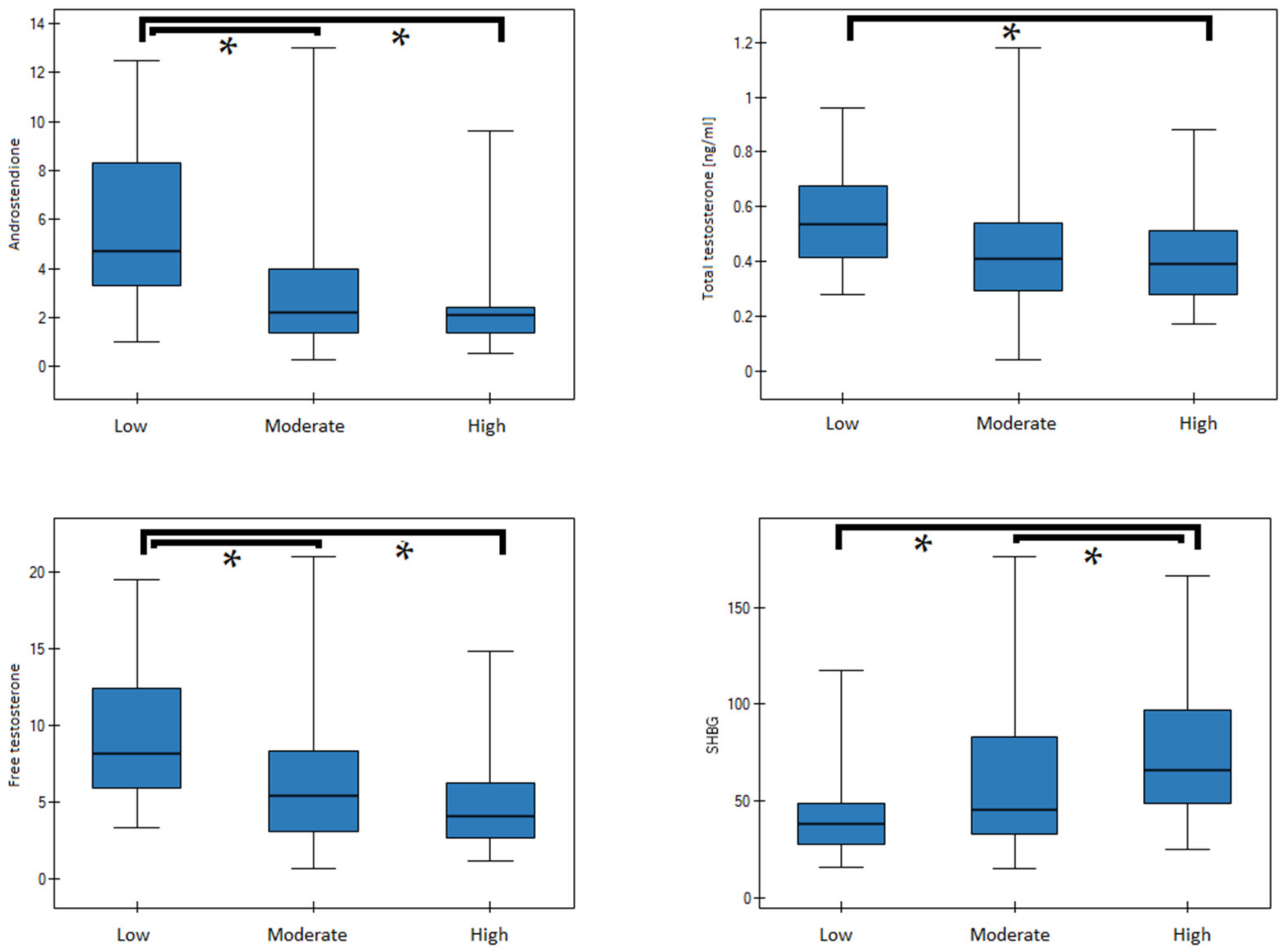
| PA (leisure) | SHBG (nmol/L) | p-value | Dunn–Bonferroni | Low | moderate | high |
| low | 34.83 (27.01–45.95) | 0.002 | low | 0.137 | 0.001 | |
| moderate | 47.55 (35.32–85.92) | moderate | 0.137 | 0.154 | ||
| high | 67.86 (57.15–107.15) | high | 0.001 | 0.154 |
| PA (Work/School) | A | p-Value | Dunn–Bonferroni | Low | Moderate | High |
|---|---|---|---|---|---|---|
| low | 4.10 (2.51–8.70) | 0.001 | low | 0.004 | 0.006 | |
| moderate | 2.10 (1.39–3.40) | moderate | 0.004 | 1.000 | ||
| high | 2.14 (1.22–2.34) | high | 0.006 | 1.000 | ||
| PA (work/school) | Total T | p-value | Dunn–Bonferroni | Low | moderate | high |
| low | 0.51 (0.43–0.64) | 0.005 | low | 0.036 | 0.008 | |
| moderate | 0.42 (0.27–0.53) | moderate | 0.036 | 0.729 | ||
| high | 0.36 (0.24–0.46) | high | 0.008 | 0.729 | ||
| PA (work/school) | Free T | p-value | Dunn–Bonferroni | Low | moderate | high |
| low | 8.00 (5.70–12.70) | 0.002 | low | 0.006 | 0.009 | |
| moderate | 4.50 (3.09–7.08) | moderate | 0.006 | 1 | ||
| high | 4.00 (2.93–6.80) | high | 0.009 | 1 | ||
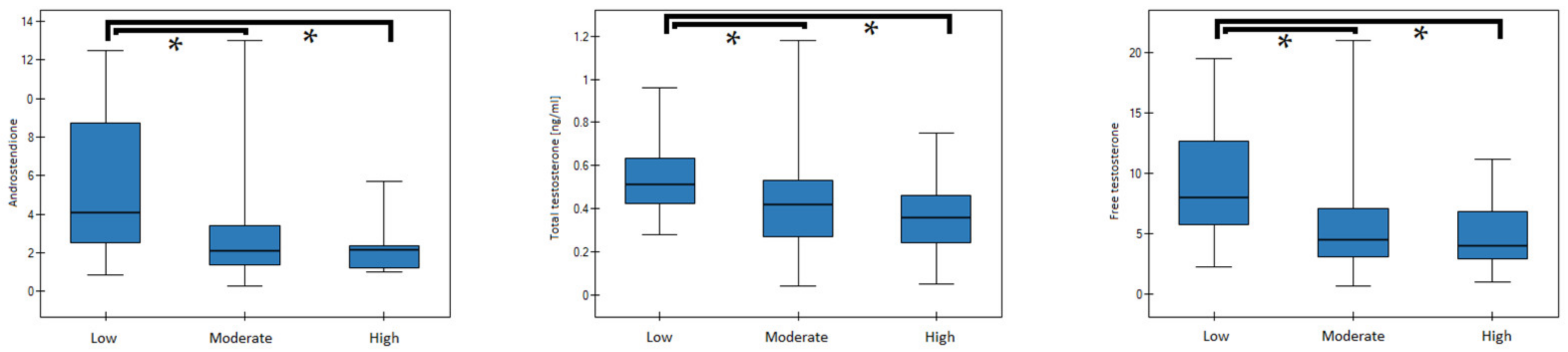
| PA (leisure) | A | p-value | Dunn–Bonferroni | Low | moderate | high |
| low | 4.70 (3.33–8.30) | 0.0009 | low | 0.014 | 0.001 | |
| moderate | 2.19 (1.36–4.00) | moderate | 0.014 | 0.610 | ||
| high | 2.09 (1.39–2.44) | high | 0.001 | 0.610 | ||
| PA (leisure) | Total T | p-value | Dunn–Bonferroni | low | moderate | high |
| low | 0.54 (0.41–0.68) | 0.03 | low | 0.087 | 0.033 | |
| moderate | 0.41 (0.29–0.54) | moderate | 0.087 | 1 | ||
| high | 0.39 (0.28–0.51) | high | 0.033 | 1 | ||
| PA (leisure) | Free T | p-value | Dunn–Bonferroni | Low | moderate | high |
| low | 8.15 (5.93–12.38) | 0.0009 | low | 0.027 | 0.001 | |
| moderate | 5.40 (3.10–8.30) | moderate | 0.027 | 0.339 | ||
| high | 4.10 (2.68–6.25) | high | 0.001 | 0.339 | ||
| PA (leisure) | SHBG | p-value | Dunn–Bonferroni | Low | moderate | high |
| low | 38.07 (27.52–48.39) | 0.003 | low | 0.448 | 0.002 | |
| moderate | 45.28 (33.13–83.17) | moderate | 0.448 | 0.049 | ||
| high | 66.11 (48.96–96.68) | high | 0.002 | 0.049 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mizgier, M.; Watrowski, R.; Opydo-Szymaczek, J.; Jodłowska-Siewert, E.; Lombardi, G.; Kędzia, W.; Jarząbek-Bielecka, G. Association of Macronutrients Composition, Physical Activity and Serum Androgen Concentration in Young Women with Polycystic Ovary Syndrome. Nutrients 2022, 14, 73. https://doi.org/10.3390/nu14010073
Mizgier M, Watrowski R, Opydo-Szymaczek J, Jodłowska-Siewert E, Lombardi G, Kędzia W, Jarząbek-Bielecka G. Association of Macronutrients Composition, Physical Activity and Serum Androgen Concentration in Young Women with Polycystic Ovary Syndrome. Nutrients. 2022; 14(1):73. https://doi.org/10.3390/nu14010073
Chicago/Turabian StyleMizgier, Małgorzata, Rafał Watrowski, Justyna Opydo-Szymaczek, Elżbieta Jodłowska-Siewert, Giovanni Lombardi, Witold Kędzia, and Grażyna Jarząbek-Bielecka. 2022. "Association of Macronutrients Composition, Physical Activity and Serum Androgen Concentration in Young Women with Polycystic Ovary Syndrome" Nutrients 14, no. 1: 73. https://doi.org/10.3390/nu14010073
APA StyleMizgier, M., Watrowski, R., Opydo-Szymaczek, J., Jodłowska-Siewert, E., Lombardi, G., Kędzia, W., & Jarząbek-Bielecka, G. (2022). Association of Macronutrients Composition, Physical Activity and Serum Androgen Concentration in Young Women with Polycystic Ovary Syndrome. Nutrients, 14(1), 73. https://doi.org/10.3390/nu14010073









