Cocoa Polyphenols and Gut Microbiota Interplay: Bioavailability, Prebiotic Effect, and Impact on Human Health
Abstract
1. Introduction
2. Cocoa Composition and Processing
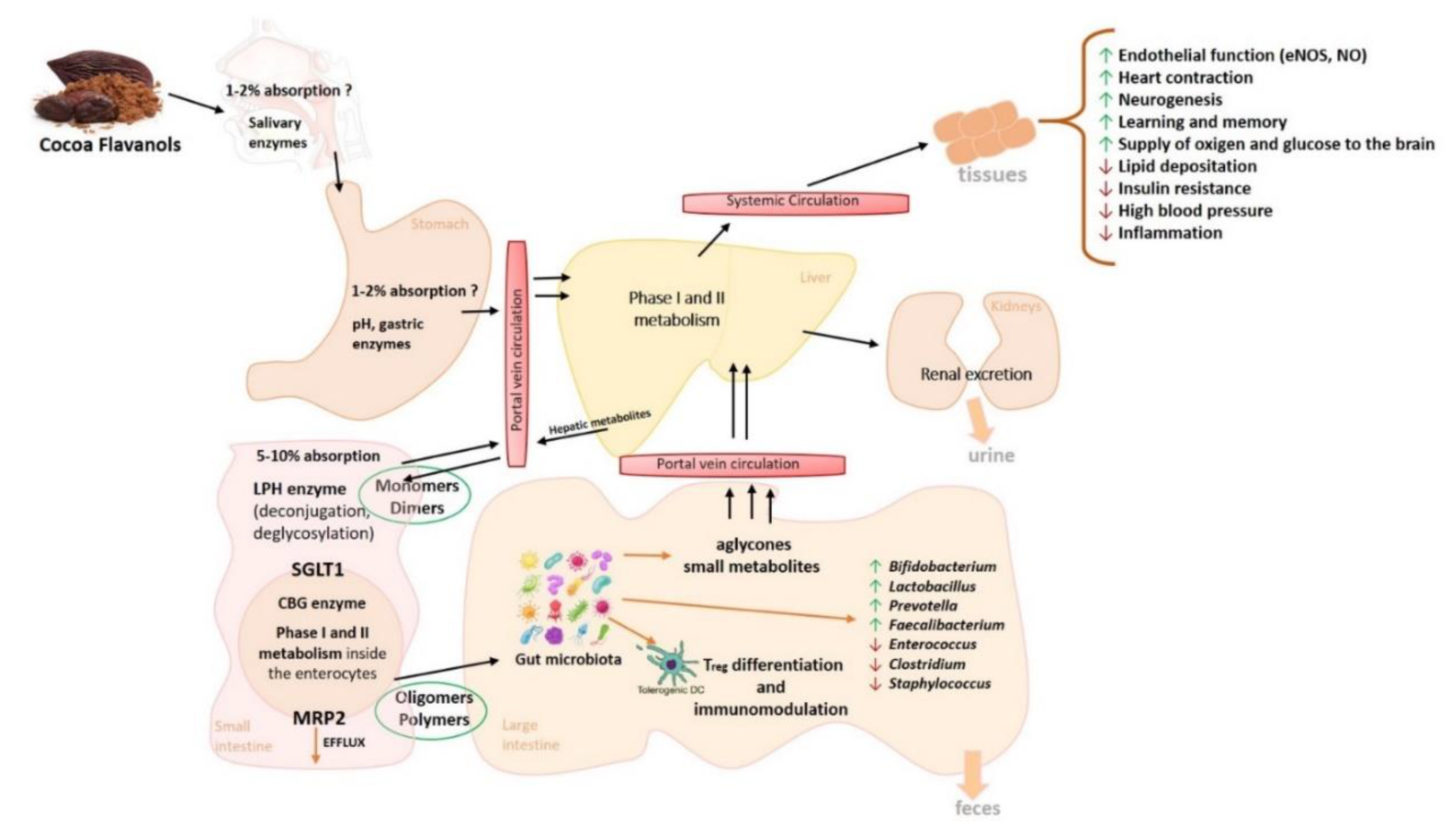
3. Cocoa Polyphenols Metabolism and Bioavailability
4. Cocoa Polyphenols and Gut Microbiota
4.1. In Vitro and In Vivo Studies
4.2. Clinical Studies
5. Conclusions and Future Perspectives
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Andújar, I.; Recio, M.C.; Giner, R.M.; Ríos, J.L. Cocoa Polyphenols and Their Potential Benefits for Human Health. Oxidative Med. Cell. Longev. 2012, 2012, 906252. [Google Scholar]
- Fraga, C.G.; Litterio, M.C.; Prince, P.D.; Calabró, V.; Piotrkowski, B.; Galleano, M. Cocoa flavanols: Effects on Vascular Nitric Oxide and Blood Pressure. J. Clin. Biochem. Nutr. 2011, 48, 63–67. [Google Scholar] [CrossRef] [PubMed]
- Schroeter, H.; Heiss, C.; Balzer, J.; Kleinbongard, P.; Keen, C.L.; Hollenberg, N.K.; Sies, H.; Kwik-Uribe, C.; Schmitz, H.H.; Kelm, M. (-)-Epicatechin mediates beneficial effects of flavanol-rich cocoa on vascular function in humans. Proc Natl Acad Sci USA 2006, 103, 1024–1029. [Google Scholar] [CrossRef]
- Taubert, D.; Roesen, R.; Lehmann, C.; Jung, N.; Schömig, E. Effects of low habitual cocoa intake on blood pressure and bioactive nitric oxide: A randomized controlled trial. JAMA 2007, 298, 49–60. [Google Scholar] [CrossRef]
- Magrone, T.; Russo, M.A.; Jirillo, E. Cocoa and dark chocolate polyphenols: From biology to clinical Applications. Front. Immunol. 2017, 8, 677. [Google Scholar] [CrossRef]
- Scapagnini, G.; Davinelli, S.; Di Renzo, L.; De Lorenzo, A.; Olarte, H.H.; Micali, G.; Cicero, A.F.; Gonzalez, S. Cocoa bioactive compounds: Significance and potential for the maintenance of skin health. Nutrients 2014, 6, 3202–3213. [Google Scholar] [CrossRef]
- Davinelli, S.; Corbi, G.; Righetti, S.; Sears, B.; Olarte, H.H.; Grassi, D.; Scapagnini, G. Cardioprotection by cocoa polyphenols and ω-3 fatty acids: A disease-prevention perspective on aging-associated cardiovascular risk. J. Med. Food 2018, 21, 1060–1069. [Google Scholar] [CrossRef]
- Calzavara-Pinton, P.; Calzavara-Pinton, I.; Arisi, M.; Rossi, M.T.; Scapagnini, G.; Davinelli, S.; Venturini, M. Cutaneous Photoprotective Activity of a Short-term Ingestion of High-Flavanol Cocoa: A Nutritional Intervention Study. Photochem. Photobiol. 2019, 95, 1029–1034. [Google Scholar] [CrossRef]
- Ali, F.; Ismail, A.; Kersten, S. Molecular mechanisms underlying the potential antiobesity-related diseases effect of cocoa polyphenols. Mol. Nutr. Food Res. 2014, 58, 33–48. [Google Scholar] [CrossRef] [PubMed]
- Davinelli, S.; Corbi, G.; Zarrelli, A.; Arisi, M.; Calzavara-Pinton, P.; Grassi, D.; De Vivo, I.; Scapagnini, G. Short-term supplementation with flavanol-rich cocoa improves lipid profile, antioxidant status and positively influences the AA/EPA ratio in healthy subjects. J. Nutr. Biochem. 2018, 61, 33–39. [Google Scholar] [CrossRef] [PubMed]
- Nehlig, A. The neuroprotective effects of cocoa flavanol and its influence on cognitive performance. Br. J. Clin. Pharmacol. 2013, 75, 716–727. [Google Scholar] [CrossRef] [PubMed]
- Socci, V.; Tempesta, D.; Desideri, G.; De Gennaro, L.; Ferrara, M. Enhancing Human Cognition with Cocoa Flavonoids. Front. Nutr. 2017, 4, 19. [Google Scholar] [CrossRef] [PubMed]
- Sokolov, A.N.; Pavlova, M.A.; Klosterhalfen, S.; Enck, P. Chocolate and the brain: Neurobiological impact of cocoa flavanols on cognition and behavior. Neurosci. Biobehav. Rev. 2013, 37, 2445–2453. [Google Scholar] [CrossRef] [PubMed]
- Mastroiacovo, D.; Kwik-Uribe, C.; Grassi, D.; Necozione, S.; Raffaele, A.; Pistacchio, L.; Righetti, R.; Bocale, R.; Lechiara, M.C.; Marini, C.; et al. Cocoa flavanol consumption improves cognitive function, blood pressure control, and metabolic profile in elderly subjects: The Cocoa, Cognition, and Aging (CoCoA) Study—A randomized controlled trial. Am. J. Clin. Nutr. 2015, 101, 538–548. [Google Scholar] [CrossRef] [PubMed]
- Guinane, C.M.; Cotter, P.D. Role of the gut microbiota in health and chronic gastrointestinal disease: Understanding a hidden metabolic organ. Ther. Adv. Gastroenterol. 2013, 6, 295–308. [Google Scholar] [CrossRef] [PubMed]
- David, L.A.; Maurice, C.F.; Rachel, N. Carmody,1 David B. Gootenberg,1 Julie E. Button,1 Benjamin E. Wolfe,1 Alisha V. Ling,3 A. Sloan Devlin,4 Yug Varma,4 Michael A. Fischbach,4 Sudha B. Biddinger,3 Rachel J. Dutton,1 and Peter J. Turnbaugh1,*Diet rapidly and reproducibly alters the human gut microbiome. Nature 2014, 505, 559–563. [Google Scholar]
- Daien, C.I.; Pinget, G.V.; Tan, J.K.; Macia, L. Detrimental impact of microbiota-accessible carbohydrate-deprived diet on gut and immune homeostasis: An overview. Front. Immunol. 2017, 8, 548. [Google Scholar] [CrossRef]
- Cardona, F.; Andrés-Lacueva, C.; Tulipania, S.; Tinahones, F.J.; Queipo-Ortuño, M.I. Benefits of polyphenols on gut microbiota and implications in human health. J. Nutr. Biochem. 2013, 24, 1415–1422. [Google Scholar] [CrossRef]
- Blumberg, J.B.; Ding, E.L.; Dixon, R.; Pasinetti, G.M.; Villarreal, F. The science of cocoa flavanols: Bioavailability, emerging evidence, and proposed mechanisms. Adv. Nutr. 2014, 5, 547–549. [Google Scholar] [CrossRef]
- Del Rio, D.; Costa, L.G.; Lean, M.E.J.; Crozier, A. Polyphenols and health: What compounds are involved? Nutr. Metab. Cardiovasc. Dis. 2010, 20, 1–6. [Google Scholar] [CrossRef]
- Gibson, G.R.; Hutkins, R.; Sanders, M.E.; Prescott, S.L.; Reimer, R.A.; Salminen, S.J.; Scott, K.; Stanton, C.; Swanson, K.S.; Cani, P.D.; et al. Expert consensus document: The international scientific association for probiotics and prebiotics (ISAPP) consensus statement on the definition and scope of prebiotics. Nat. Rev. Gastroenterol. Hepatol. 2017, 14, 491–502. [Google Scholar] [CrossRef] [PubMed]
- Sonnenburg, E.D.; Sonnenburg, J.L. Starving our microbial self: The deleterious consequences of a diet deficient in microbiota-accessible carbohydrates. Cell Metab. 2014, 20, 779–786. [Google Scholar] [CrossRef] [PubMed]
- Colombo, M.L.; Pinorini-Godly, M.T.; Conti, A. Botany and Pharmacognosy of the Cacao Tree. In Chocolate and Health; Springer Milan: Milano, Italy, 2012; pp. 41–62. [Google Scholar]
- Redovnikovic, I.R.; Delonga, K.; Mazor, S.; Verica, D.U.; Caric, M.; Vorkapic-Furac, J. Polyphenolic Content and Composition and Antioxidative Activity of Different Cocoa Liquors. Czech J. Food Sci. 2009, 27, 330–337. [Google Scholar] [CrossRef]
- Schwan, R.F.; Wheals, A.E. The microbiology of cocoa fermentation and its role in chocolate quality. Crit. Rev. food Sci. Nutr. 2004, 44, 205–221. [Google Scholar] [CrossRef]
- Close, D.C.; McArthur, C. Rethinking the role of many plant phenolics – protection from photodamage not herbivores? Oikos 2002, 99, 166–172. [Google Scholar] [CrossRef]
- Flores, M.E.J. Cocoa Flavanols: Natural Agents with Attenuating Effects on Metabolic Syndrome Risk Factors. Nutrients 2019, 11, 751. [Google Scholar]
- Borchers, A.T.; Keen, C.L.; Hannum, S.M.; Gershwin, M.E. Cocoa and chocolate: Composition, bioavailability, and health implications. J. Med. Food 2000, 3, 77–105. [Google Scholar] [CrossRef]
- Martín, M.A.; Ramos, S. Cocoa polyphenols in oxidative stress: Potential health implications. J. Funct. Foods 2016, 27, 570–588. [Google Scholar] [CrossRef]
- Miller, K.B.; Hurst, W.J.; Payne, M.J.; Stuart, D.A.; Apgar, J.; Sweigart, D.S.; Ou, B. Impact of alkalization on the antioxidant and flavanol content of commercial cocoa powders. J. Agric. Food Chem. 2008, 56, 8527–8533. [Google Scholar] [CrossRef]
- Rocha, I.S.; de Santana, L.R.R.; Soares, S.E.; Bispo, E.S. Effect of the roasting temperature and time of cocoa beans on the sensory characteristics and acceptability of chocolate. Food Sci. Technol. 2017, 37, 522–530. [Google Scholar] [CrossRef]
- Giacometti, J.; Jolić, S.M.; Josić, D. Chapter 73—Cocoa Processing and Impact on Composition. In Processing and Impact on Active Components in Food; Preedy, V., Ed.; Academic Press: San Diego, CA, USA, 2015; pp. 605–612. [Google Scholar]
- Jolic, S.; Redovniković, I.R.; Marković, K.; Šipušić, D.I.; Delonga, K. Changes of phenolic compounds and antioxidant capacity in cocoa beans processing. Int. J. Food Sci. Technol. 2011, 46, 1793–1800. [Google Scholar] [CrossRef]
- Wollgast, J.; Anklam, E. Review on polyphenols in theobroma cacao: Changes in composition during the manufacture of chocolate and methodology for identification and quantification. Food Res. Int. 2000, 33, 423–447. [Google Scholar] [CrossRef]
- Hurst, W.J.; Krake, S.H.; Bergmeier, S.C.; Payne, M.J.; Miller, K.B.; Stuart, D.A. Impact of fermentation, drying, roasting and Dutch processing on flavan-3-ol stereochemistry in cacao beans and cocoa ingredients. Chem. Cent. J. 2011, 5, 53. [Google Scholar] [CrossRef] [PubMed]
- Miller, K.B.; Stuart, D.A.; Smith, N.L.; Lee, C.Y.; McHale, N.L.; Flanagan, J.A.; Ou, B.; Hurst, W.J. Antioxidant activity and polyphenol and procyanidin contents of selected commercially available cocoa-containing and chocolate products in the United States. J. Agric. Food Chem. 2006, 54, 4062–4068. [Google Scholar] [CrossRef] [PubMed]
- Natsume, M.; Osakabe, N.; Yamagishi, M.; Takizawa, T.; Nakamura, T.; Miyatake, H.; Hatano, T.; Yoshida, T. Analyses of polyphenols in cacao liquor, cocoa, and chocolate by normal-phase and reversed-phase HPLC. Biosci. Biotechnol. Biochem. 2000, 64, 2581–2587. [Google Scholar] [CrossRef] [PubMed]
- Tomas-Barberan, F.A.; Cienfuegos-jovellanos, E.; Marìn, A.; Muguerza, B.; Gil-Izquierdo, A.; Cerda, B.; Zafrilla, P.; Morillas, J.; Mulero, J.; Ibarra, A.; et al. A new process to develop a cocoa powder with higher flavonoid monomer content and enhanced bioavailability in healthy humans. J. Agric. Food Chem. 2007, 55, 3926–3935. [Google Scholar] [CrossRef]
- Andres-Lacueva, C.; Monagas, M.; Khan, N.; Izquierdo-Pulido, M.; Urpi-Sarda, M.; Permanyer, J.; Lamuela-Raventós, R.M. Flavanol and flavonol contents of cocoa powder products: Influence of the manufacturing process. J. Agric. Food Chem. 2008, 56, 3111–3117. [Google Scholar] [CrossRef]
- Larson, A.; Symons, J.; Jalili, T. Therapeutic Potential of Quercetin to Decrease Blood Pressure: Review of Efficacy and Mechanisms. Adv. Nutr. (Bethesda, Md.) 2012, 3, 39–46. [Google Scholar] [CrossRef]
- Krings, U.; Zelena, K.; Wu, S.; Berger, R.G. Thin-layer high-vacuum distillation to isolate volatile flavour compounds of cocoa powder. Eur. Food Res. Technol. 2006, 223, 675–681. [Google Scholar] [CrossRef]
- Li, Y.; Feng, Y.; Zhu, S.; Luo, C.; Ma, J.; Zhong, F. The effect of alkalization on the bioactive and flavor related components in commercial cocoa powder. J. Food Compos. Anal. 2012, 25, 17–23. [Google Scholar] [CrossRef]
- Ramli, N.; Yatim, A.; Hok, H. HPLC Determination of Methylxanthines and Polyphenols Levels In Cocoa and Chocolate Products. Mal. J. Anal. Sci. 2001, 7, 377–386. [Google Scholar]
- Lang, R.; Mueller, C.; Hofmann, T. Development of a stable isotope dilution analysis with liquid chromatography-tandem mass spectrometry detection for the quantitative analysis of di- and trihydroxybenzenes in foods and model systems. J. Agric. Food Chem. 2006, 54, 5755–5762. [Google Scholar] [CrossRef] [PubMed]
- Arts, I.C.; van de Putte, B.; Hollman, P.C. Catechin contents of foods commonly consumed in The Netherlands. 1. Fruits, vegetables, staple foods, and processed foods. J. Agric. Food Chem. 2000, 48, 1746–1751. [Google Scholar] [CrossRef] [PubMed]
- D’Archivio, M.; Filesi, C.; Varì, R.; Scazzocchio, B.; Masella, R. Bioavailability of the polyphenols: Status and controversies. Int. J. Mol. Sci. 2010, 11, 1321–1342. [Google Scholar] [CrossRef]
- Rechner, A.R.; Smith, M.A.; Kuhnle, G.; Gibson, G.R.; Debnam, E.S.; Srai, S.K.S.; Moore, K.P.; Rice-Evans, C.A. Colonic metabolism of dietary polyphenols: Influence of structure on microbial fermentation products. Free Radic. Biol. Med. 2004, 36, 212–225. [Google Scholar] [CrossRef]
- D’Archivio, M.; Filesi, C.; Di benedetto, R.; Gargiulo, R.; Giovannini, C.; Masella, R. Polyphenols, dietary sources and bioavailability. Ann. Ist. Super Sanita 2007, 43, 348–361. [Google Scholar]
- Oracz, J.; Nebesny, E.; Zyzelewicz, D.; Budryn, G.; Luzak, B. Bioavailability and metabolism of selected cocoa bioactive compounds: A comprehensive review. Crit. Rev. food Sci. Nutr. 2020, 60, 1947–1985. [Google Scholar] [CrossRef]
- Sorrenti, V.; Fortinguerra, S.; Caudullo, G.; Buriani, A. Deciphering the Role of Polyphenols in Sports Performance: From Nutritional Genomics to the Gut Microbiota toward Phytonutritional Epigenomics. Nutrients 2020, 12, 1265. [Google Scholar] [CrossRef]
- Metere, A.; Giacomelli, L. Absorption, metabolism and protective role of fruits and vegetables polyphenols against gastric cancer. Eur. Rev. Med. Pharmacol. Sci. 2017, 21, 5850–5858. [Google Scholar]
- Williamson, G.; Manach, C. Bioavailability and bioefficacy of polyphenols in humans. I. Review of 93 bioavailability studies. Am. J. Clin. Nutr. 2005, 81 (Suppl. 1), 230S–242S. [Google Scholar] [CrossRef]
- Luca, S.V.; Macovei, I.; Bujor, A.; Miron, A.; Skalicka-Woźniak, K.; Aprotosoaie, A.C.; Trifan, A. Bioactivity of dietary polyphenols: The role of metabolites. Crit. Rev. Food Sci. Nutr. 2020, 60, 626–659. [Google Scholar] [CrossRef] [PubMed]
- Scalbert, A.; Williamson, G. Dietary intake and bioavailability of polyphenols. J Nutr. 2000, 130 (Suppl. 8S), 2073S–2085S. [Google Scholar] [CrossRef]
- Montagnana, M.; Danese, E.; Angelino, D.; Mena, P.; Rosi, A.; Benati, M.; Gelati, M.; Salvagno, G.L.; Favaloro, E.J.; Del Rio, D.; et al. Dark chocolate modulates platelet function with a mechanism mediated by flavan-3-ol metabolites. Medicine (Baltimore) 2018, 97, e13432. [Google Scholar] [CrossRef] [PubMed]
- Kay, C.D.; Mazza, G.; Holub, B.J.; Wang, J. Anthocyanin metabolites in human urine and serum. Br. J. Nutr. 2004, 91, 933–942. [Google Scholar] [CrossRef] [PubMed]
- Mena, P.; Bresciani, L.; Brindani, N.; Ludwig, I.A.; Pereira-Caro, G.; Angelino, D.; Llorach, R.; Calani, L.; Brighenti, F.; Clifford, M.N.; et al. Phenyl-γ-valerolactones and phenylvaleric acids, the main colonic metabolites of flavan-3-ols: Synthesis, analysis, bioavailability, and bioactivity. Nat. Prod. Rep. 2019, 36, 714–752. [Google Scholar] [CrossRef]
- Stoupi, S.; Williamson, G.; Drynan, J.W.; Barron, D.; Clifford, M.N. A comparison of the in vitro biotransformation of (-)-epicatechin and procyanidin B2 by human faecal microbiota. Mol. Nutr. Food Res. 2010, 54, 747–759. [Google Scholar] [CrossRef]
- Gómez-Juaristi, M.; Sarria, B.; Martínez-López, S.; Bravo Clemente, L.; Mateos, R. Flavanol Bioavailability in Two Cocoa Products with Different Phenolic Content. A Comparative Study in Humans. Nutrients 2019, 11, 1441. [Google Scholar] [CrossRef]
- Petyaev, I.M.; Dovgalevsky, P.Y.; Chalyk, N.E.; Klochkov, V.; Kyle, N.H. Reduction in blood pressure and serum lipids by lycosome formulation of dark chocolate and lycopene in prehypertension. Food Sci. Nutr. 2014, 2, 744–750. [Google Scholar] [CrossRef]
- Vitaglione, P.; Lumaga, R.B.; Ferracane, R.; Sellitto, S.; Morelló, J.R.; Miranda, J.R.; Shimoni, E.; Fogliano, V. Human bioavailability of flavanols and phenolic acids from cocoa-nut creams enriched with free or microencapsulated cocoa polyphenols. Br. J. Nutr. 2013, 109, 1832–1843. [Google Scholar] [CrossRef]
- Kawabata, K.; Yoshioka, Y.; Terao, J. Role of Intestinal Microbiota in the Bioavailability and Physiological Functions of Dietary Polyphenols. Molecules 2019, 24, 370. [Google Scholar] [CrossRef]
- Yang, Q.; Liang, Q.; Balakrishnan, B.; Belobrajdic, D.P.; Feng, Q.-J.; Zhang, W. Role of Dietary Nutrients in the Modulation of Gut Microbiota: A Narrative Review. Nutrients 2020, 12, 381. [Google Scholar] [CrossRef] [PubMed]
- Kumar Singh, A.; Cabral, C.; Kumar, R.; Ganguly, R.; Kumar Rana, H.; Gupta, A.; Rosaria Lauro, M.; Carbone, C.; Reis, F.; Pandey, A.K. Beneficial Effects of Dietary Polyphenols on Gut Microbiota and Strategies to Improve Delivery Efficiency. Nutrients 2019, 11, 2216. [Google Scholar] [CrossRef] [PubMed]
- Tzounis, X.; Rodriguez-Mateos, A.; Vulevic, J.; Gibson, G.R.; Kwik-Uribe, C.; Spencer, J.P.E. Prebiotic evaluation of cocoa-derived flavanols in healthy humans by using a randomized, controlled, double-blind, crossover intervention study. Am. J. Clin. Nutr. 2011, 93, 62–72. [Google Scholar] [CrossRef]
- Ma, G.; Chen, Y. Polyphenol supplementation benefits human health via gut microbiota: A systematic review via meta-analysis. J. Funct. Foods 2020, 66, 103829. [Google Scholar] [CrossRef]
- Tzounis, X.; Vulevic, J.; Kuhnle, G.G.C.; George, T.; Leonczak, J.; Gibson, G.R.; Kwik-Uribe, C.; Spencer, J.P.E. Flavanol monomer-induced changes to the human faecal microflora. Br. J. Nutr. 2008, 99, 782–792. [Google Scholar] [CrossRef] [PubMed]
- Fogliano, V.; Corollaro, M.L.; Vitaglione, P.; Napolitano, A.; Ferracane, R.; Travaglia, F.; Arlorio, M.; Costabile, A.; Klinder, A.; Gibson, G. In vitro bioaccessibility and gut biotransformation of polyphenols present in the water-insoluble cocoa fraction. Mol. Nutr. Food Res. 2011, 55 (Suppl. 1), S44–S55. [Google Scholar] [CrossRef]
- Peng, M.; Aryal, U.; Cooper, B.; Biswas, D. Metabolites produced during the growth of probiotics in cocoa supplementation and the limited role of cocoa in host-enteric bacterial pathogen interactions. Food Control. 2015, 53, 124–133. [Google Scholar] [CrossRef]
- Massot-Cladera, M.; Pérez-Berezo, T.; Franch, A.; Castell, M.; Pérez-Cano, F.J. Cocoa modulatory effect on rat faecal microbiota and colonic crosstalk. Arch. Biochem. Biophys. 2012, 527, 105–112. [Google Scholar] [CrossRef]
- Magistrelli, D.; Zanchi, R.; Malagutti, L.; Galassi, G.; Canzi, E.; Rosi, F. Effects of Cocoa Husk Feeding on the Composition of Swine Intestinal Microbiota. J. Agric. Food Chem. 2016, 64, 2046–2052. [Google Scholar] [CrossRef]
- Jang, S.; Sun, J.; Chen, P.; Lakshman, S.; Molokin, A.; Harnly, J.M.; Vinyard, B.T.; Urban, J.F.; Davis, C.D.; Solano-Aguilar, G. Flavanol-Enriched Cocoa Powder Alters the Intestinal Microbiota, Tissue and Fluid Metabolite Profiles, and Intestinal Gene Expression in Pigs. J. Nutr. 2016, 146, 673–680. [Google Scholar] [CrossRef]
- Álvarez-Cilleros, D.; Ramos, S.; López-Oliva, M.E.; Escrivá, F.; Álvarez, C.; Fernández-Millán, E.; Martín, M.A. Cocoa diet modulates gut microbiota composition and improves intestinal health in Zucker diabetic rats. Food Res. Int. 2020, 132, 109058. [Google Scholar] [CrossRef] [PubMed]
- Camps-Bossacoma, M.; Pérez-Cano, F.J.; Franch, A.; Castell, M. Gut Microbiota in a Rat Oral Sensitization Model: Effect of a Cocoa-Enriched Diet. Oxidative Med. Cell. Longev. 2017, 2017, 7417505. [Google Scholar] [CrossRef] [PubMed]
- Abril-Gil, M.; Pérez-Cano, F.J.; Franch, A.; Castell, M. Effect of a cocoa-enriched diet on immune response and anaphylaxis in a food allergy model in Brown Norway rats. J. Nutr. Biochem. 2016, 27, 317–326. [Google Scholar] [CrossRef] [PubMed]
- Massot-Cladera, M.; Abril-Gil, M.; Torres, S.; Franch, A.; Castell, M.; Pérez-Cano, F.J. Impact of cocoa polyphenol extracts on the immune system and microbiota in two strains of young rats. Br. J. Nutr. 2014, 112, 1944–1954. [Google Scholar] [CrossRef] [PubMed]
- Martín-Peláez, S.; Camps-Bossacoma, M.; Massot-Cladera, M.; Rigo-Adrover, M.; Franch, A.; Pérez-Cano, F.J.; Castell, M. Effect of cocoa’s theobromine on intestinal microbiota of rats. Mol. Nutr. Food Res. 2017, 61. [Google Scholar] [CrossRef]
- Castell, M.; Saldaña-Ruíz, S.; Rodríguez-Lagunas, M.J.; Franch, À.; Pérez-Cano, F.J. Second International Congress on Chocolate and Cocoa in Medicine Held in Barcelona, Spain, 25–26th September 2015. Nutrients 2015, 7, 9785–9803. [Google Scholar] [CrossRef]
- Urpi-Sarda, M.; Llorach, R.; Khan, N.; Monagas, M.; Rotches-Ribalta, M.; Lamuela-Raventos, R.; Estruch, R.; Tinahones, F.J.; Andres-Lacueva, C. Effect of milk on the urinary excretion of microbial phenolic acids after cocoa powder consumption in humans. J. Agric. Food Chem. 2010, 58, 4706–4711. [Google Scholar] [CrossRef]
- Orrhage, K.; Nord, C.E. Bifidobacteria and lactobacilli in human health. Drugs Exp. Clin. Res. 2000, 26, 95–111. [Google Scholar]
- Lavefve, L.; Howard, L.; Carbonero, F. Berry polyphenols metabolism and impact on human gut microbiota and health. Food Funct. 2019, 11, 45–65. [Google Scholar] [CrossRef]
- Wiese, M.; Bashmakov, Y.; Chalyk, N.; Nielsen, D.S.; Krych, Ł.; Kot, W.; Klochkov, V.; Pristensky, D.; Bandaletova, T.; Chernyshova, M.; et al. Prebiotic Effect of Lycopene and Dark Chocolate on Gut Microbiome with Systemic Changes in Liver Metabolism, Skeletal Muscles and Skin in Moderately Obese Persons. Biomed. Res. Int. 2019, 2019, 4625279. [Google Scholar] [CrossRef]
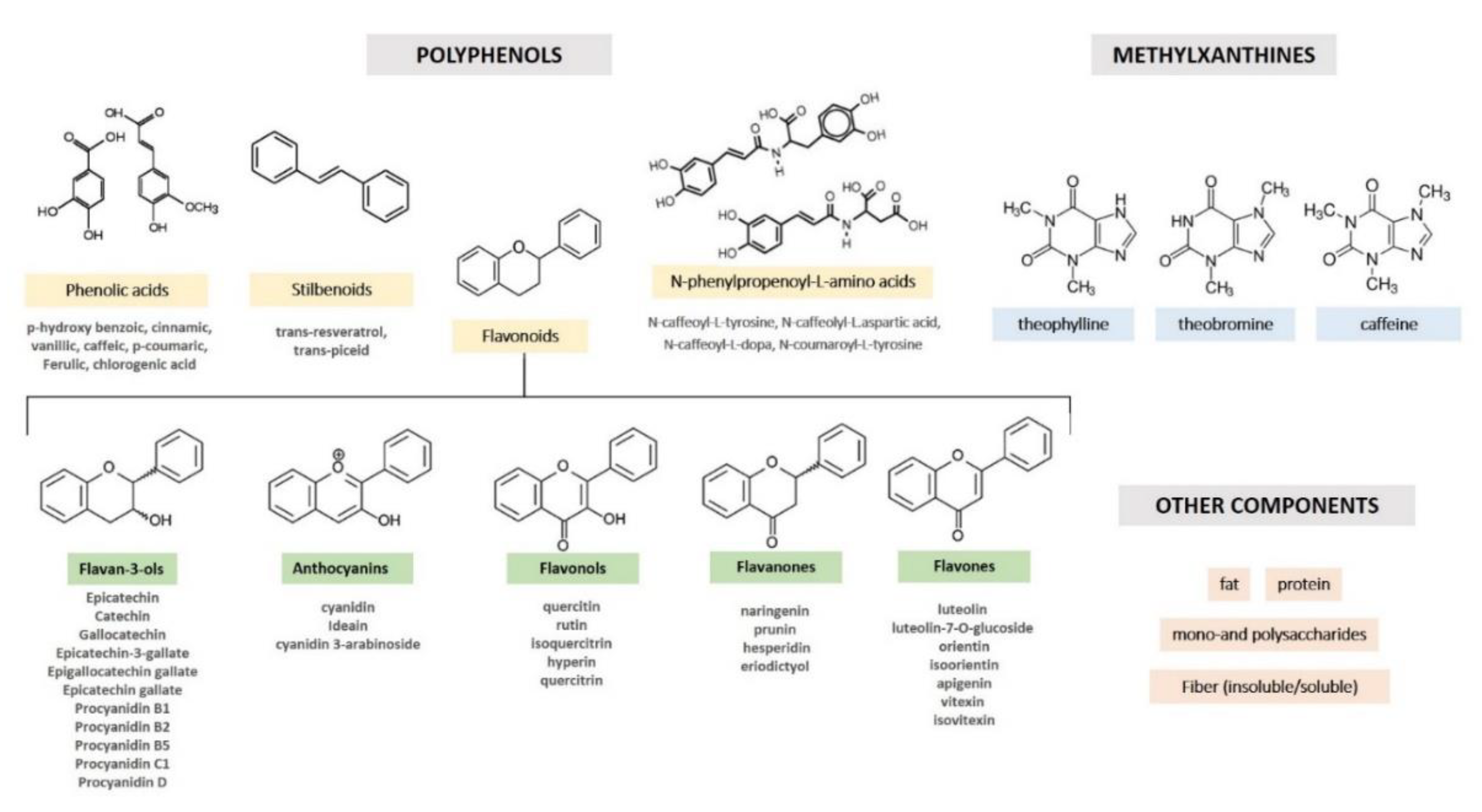
| Phytochemical | Mg/g of Fresh Weight | Reference | |
|---|---|---|---|
| Flavanols | (−)-Epicatechin | 0.63- 3.3 | [37,38] |
| (+)-Catechin | 0.61–2.02 | [36,38] | |
| Procyanidin B1 (dimer) | 1.12 | [38] | |
| Procyanidin B2 (dimer) | 0.13- 2.62 | [37,38] | |
| Procyanidin C1 (trimer) | 0.05- 0.36 | [37] | |
| Cinnamtannin A2 (tetramer) | 0.31–0.56 | [37] | |
| Galactopyranosyl-ent-(−)-epicatechin (2alpha-->7, 4alpha->8)-(−)-epicatechin (Gal-EC-EC) | 0.02–0.07 | [37] | |
| Flavonols | Quercetin | 0.0032–0.2 | [39,40] |
| Isoquercitrin | 0.03334–0.04664 | [39] | |
| Quercetin-3-Arabinoside | 0.05258–0.09212 | [39] | |
| Quercetin 3-O-glucuronide | 0.00938–0.01512 | [39] | |
| Phenolic acids | |||
| Hydroxybenzoic acids | Benzoic acid | 0.00017–0.00097 | [41] |
| Hydroxycinnamic acids | Caffeoyl aspartate | 0.37 | [38] |
| Hydroxybenzaldehydes | Vanillin | 0.00014–0.00232 | [41] |
| Methylxanthines | Theobromine | 4.11–27.69 | [42,43] |
| Caffeine | 0.946–6.58 | [42,43] | |
| Others | Catechol | 0.0012 | [44] |
| Pyrogallol | 0.0018 | [44] |
| Source | Quantity (mg/g) | Reference | |
|---|---|---|---|
| (+)-Catechin | (−)-Epicatechin | ||
| Cocoa bean | 0.28 | 2.23 | [33] |
| Roasted cocoa bean | 0.33 | 1.63 | [33] |
| Alkalised cocoa nibs | 0.26 | 0.58 | [33] |
| Alkalised cocoa powder | 0.60 | 0.88 | [33] |
| Dried alkalised cocoa nibs | 0.33 | 0.49 | [33] |
| Cocoa liquor | 0.34 | 1.22 | [33] |
| Cocoa liquor (Venezuela) | 0.14 | 0.74 | [37] |
| Cocoa liquor (Brazil) | 0.63 | 5.77 | [37] |
| Baking chocolate | 0.24 | 0.52 | [33] |
| Dark chocolate (Albert Heijn) | 0.1324 | 0.3274 | [45] |
| Dark chocolate (Verkade) | 0.1075 | 0.5025 | [45] |
| Milk chocolate | 0.05–0.12 | 0.18–0.24 | [36] |
| Milk chocolate (Albert Heijn) | 0.0383 | 0.1249 | [45] |
| Milk chocolate (Verkade) | 0.0269 | 0.1261 | [45] |
| Baking chips | 0.26–0.50 | 0.66–1.07 | [36] |
| Chocolate candy bar | 0.0217 | 0.0625 | [45] |
| Type of Cocoa | Gut Bacteria | Growth Rate | Health Outcome | Reference | |||
|---|---|---|---|---|---|---|---|
| Phylum | Genus | Species | |||||
| In vitro studies | Water-insoluble cocoa fraction (WICF) of alkali-treated commercial cocoa powder. | Actinobacteria | Bifidobacterium | Increased | Exertion of antioxidant action by insoluble polyphenols | [68] | |
| Firmicutes | Lactobacillus | ||||||
| Commercial, non-alkali treated, defatted cocoa powder | Firmicutes | Lactobacillus | casei | Increased | - Diminished adherence and invasion ability of pathogenic gut bacteria | [69] | |
| Lactobacillus | rhamnosus | ||||||
| Lactobacillus | plantarum | ||||||
| Lactobacillus | acidophilus | ||||||
| Bacillus | subtilis | ||||||
| Enterococcus | faecalis | ||||||
| Streptococcus | thermophilus | ||||||
| Proteobacteria | Escherichia | coli O157:H7 | Decreased | ||||
| Salmonella | Typhimurium | ||||||
| Firmicutes | Listeria | monocytogenes | |||||
| In vivo studies | Natural Forastero cocoa powder | Bacteroidetes | Bacteroides | Decreased | - Lower body weight gain - Downregulated colon TLR2 and TLR7 and TLR9 - Reduced intestinal IgA concentration - Prevented age-related percentage increase of IgA-coating bacteria | [70] | |
| Firmicutes | Staphylococcus | ||||||
| Clostridium | |||||||
| Cocoa husks | Bacteroidetes | Bacteroides−Prevotella | Increased | - Increased fibrous fractions of the diet, mainly ADL | [71] | ||
| Faecalibacterium | prausnitzii | ||||||
| Firmicutes | Lactobacillus−Enterococcus | Decreased | |||||
| Clostridium | histolyticum | ||||||
| Flavanol-enriched cocoa powder (Acticoa) | Firmicutes | Lactobacillus | Increased | - Decreased TNF-a, TLR2, TLR4 and TLR9 expression of intestinal tissues | [72] | ||
| Actinobacteria | Bifidobacterium | ||||||
| Natural Forastero cocoa powder | Firmicutes | Blautia | Hansenii wexleare others | Increased | - Reduced body weight - Decreased glycaemia, insulinaemia and HbA1c - Lower insulin resistance state and enhanced beta-cell function - Improved glucose tolerance - Greater crypt depth - Promoted colonocyte proliferation and apoptosis - Elevated mucin glycoprotein Increased Zonula occludens-1 (ZO-1) proteins - Partially dropped level of TNFα, IL-6, MCP-1 and CD45 lin the colonic mucosa | [73] | |
| Bacteroidetes | Flavobacterium | ||||||
| Deferribacteres | |||||||
| Cyanobacteries | |||||||
| Firmicutes | Enterococcus | Decreased | |||||
| Lactobacillus | |||||||
| Bacteroidetes | Parabacteroides | Goldsteinii distasonis others | |||||
| Proteobacteria | Sutterella | ||||||
| Cocoa powder | Firmicutes | Lactobacillus | reuteri | Increased | - Lower intestinal IgA concentration | [74] | |
| Anaerostipes | |||||||
| Bacteroidetes | Prevotella | ||||||
| Bacteroides | uniformis | ||||||
| Tenericutes | |||||||
| Cyanobacteria | |||||||
| Firmicutes | Ruminococcus | flavefaciens | Decreased | ||||
| Holdemania | |||||||
| Proteobacteria | |||||||
| Natural Forastero cocoa powder | Firmicutes | Butyrivibrio | Increased | - Lower body weight gain - Higher SCFA (acetic, propionic, butyric and formic acids) - Smaller percentages of fecal IgA-coated bacteria | [77] | ||
| Bacteroidetes | Prevotella | ||||||
| Cyanobacteria | |||||||
| Firmicutes | Anaerotruncus | Decreased | |||||
| Bacteroidetes | Bacteroides | acidifaciens | |||||
| Human studies | Dairy-based cocoa-beverage mixes (standardized flavanol content) | Actinobacteria | Bifidobacterium | Increased | - Decreased total cholesterol concentrations - Reduced plasma triacylglycerol concentrations - Lowered Plasma CRP concentrations | [65] | |
| Firmicutes | Lactobacillus | ||||||
| Enterococcus | |||||||
| Eubacterium | rectale | ||||||
| Clostridium | coccoides | ||||||
| Firmicutes | Clostridium | histolyticum | Decreased | ||||
| Dark chocolate made from Trinitario cocoa beans | Firmicutes | Lactobacillus | Increased | - Diminished Inflammatory Oxidative Damage (IOD) - Reduced LDL-Px - Increased lipoprotein O2 - Reduced corneocyte exfoliation | [82] | ||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sorrenti, V.; Ali, S.; Mancin, L.; Davinelli, S.; Paoli, A.; Scapagnini, G. Cocoa Polyphenols and Gut Microbiota Interplay: Bioavailability, Prebiotic Effect, and Impact on Human Health. Nutrients 2020, 12, 1908. https://doi.org/10.3390/nu12071908
Sorrenti V, Ali S, Mancin L, Davinelli S, Paoli A, Scapagnini G. Cocoa Polyphenols and Gut Microbiota Interplay: Bioavailability, Prebiotic Effect, and Impact on Human Health. Nutrients. 2020; 12(7):1908. https://doi.org/10.3390/nu12071908
Chicago/Turabian StyleSorrenti, Vincenzo, Sawan Ali, Laura Mancin, Sergio Davinelli, Antonio Paoli, and Giovanni Scapagnini. 2020. "Cocoa Polyphenols and Gut Microbiota Interplay: Bioavailability, Prebiotic Effect, and Impact on Human Health" Nutrients 12, no. 7: 1908. https://doi.org/10.3390/nu12071908
APA StyleSorrenti, V., Ali, S., Mancin, L., Davinelli, S., Paoli, A., & Scapagnini, G. (2020). Cocoa Polyphenols and Gut Microbiota Interplay: Bioavailability, Prebiotic Effect, and Impact on Human Health. Nutrients, 12(7), 1908. https://doi.org/10.3390/nu12071908







