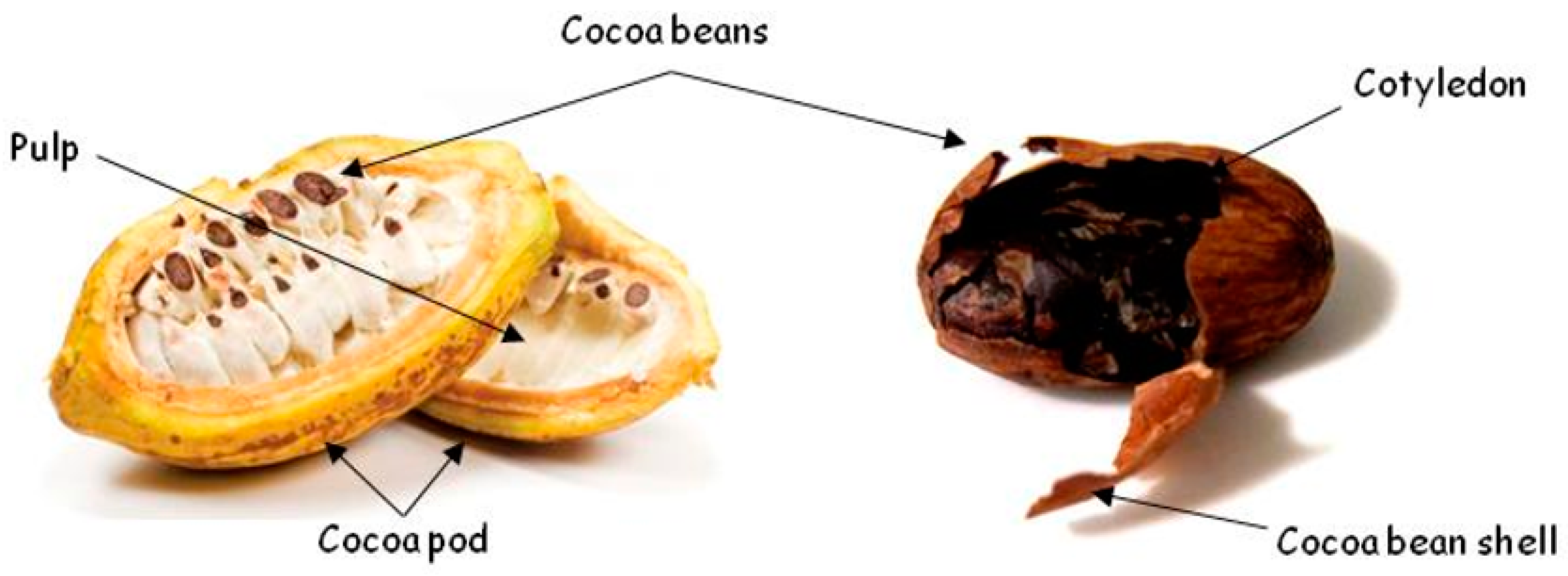
Cocoa Bean Shell—A By-Product with Nutritional Properties and Biofunctional Potential
Abstract
1. Introduction
2. Methods and Literature Search
3. Nutritional and Chemical Composition
3.1. Proximate composition—Moisture, Ashes, Proteins, Fats, and Carbohydrates
3.2. Dietary Fiber
3.3. Phenolic Compounds
3.4. Methylxanthines
3.5. Minerals and Vitamins
4. Applications
4.1. Food Applications
4.2. Utilization as Feedstuffs
4.3. Uses in Industry and Other Applications
5. Biofunctionality and Potential Health Benefits
5.1. Antibacterial Activity and Anticariogenic Effects
5.2. Antiviral Properties
5.3. Action on Cardiovascular Health
5.4. Anticarcinogenic Action
5.5. Antidiabetic Activity
5.6. Other Biofunctional Properties
6. Safety Aspects
6.1. Heavy Metals
6.2. Mycotoxins
7. Conclusions and Future Trends
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
References
- González, J.; Pérez, D.; Gutiérrez, Y.I.; Scull, R. Pharmacognostic and Physicochemical Studies of Theobroma cacao bean husk in Cuba. Int. Invent.Sci. J. 2018, 2, 262–267. [Google Scholar]
- ICCO. Quarterly Bulletin of Cocoa Statistics (Cocoa year 2018/19); International Cocoa Organization: Abidjan, Ivory Coast, 30 August 2019. [Google Scholar]
- Alimentarius, C. Standard for cocoa (cacao) mass (cocoa/chocolate liquour) and cocoa cake. Codex Stan 2016, 141. [Google Scholar]
- Quelal-Vásconez, M.A.; Lerma-García, M.J.; Pérez-Esteve, É.; Arnau-Bonachera, A.; Barat, J.M.; Talens, P. Fast detection of cocoa shell in cocoa powders by near infrared spectroscopy and multivariate analysis. Food Control 2019, 99, 68–72. [Google Scholar] [CrossRef]
- Beckett, S.T. The Science of Chocolate; Royal Society of Chemistry: London, UK, 2018. [Google Scholar]
- Battegazzore, D.; Bocchini, S.; Alongi, J.; Frache, A. Plasticizers, antioxidants and reinforcement fillers from hazelnut skin and cocoa by-products: Extraction and use in PLA and PP. Polym. Degrad. Stab. 2014, 108, 297–306. [Google Scholar] [CrossRef]
- Chandrasekaran, M. Valorization of Food Processing by-Products; CRC Press: Boca Raton, FL, USA, 2012. [Google Scholar]
- Hashimoto, J.C.; Lima, J.C.; Celeghini, R.M.; Nogueira, A.B.; Efraim, P.; Poppi, R.J.; Pallone, J.A. Quality control of commercial cocoa beans (Theobroma cacao L.) by near-infrared spectroscopy. Food Anal. Methods 2018, 11, 1510–1517. [Google Scholar] [CrossRef]
- Zhao, J.; Fleet, G. Yeasts are essential for cocoa bean fermentation. Int. J. Food Microbiol. 2014, 174, 72–87. [Google Scholar]
- Gardner, R. Nutritional Outlook. Available online: https://www.nutritionaloutlook.com/herbs-botanicals/cacao-pulp-its-not-just-waste-product-cocoa-anymore (accessed on 20 January 2020).
- Baker, T.L. Cocoa Nibs. Available online: http://www.thelonebaker.com/journal/2011/9/12/cocoa-nibs.html (accessed on 20 January 2020).
- Afrane, G.; Ntiamoah, A. Use of pesticides in the cocoa industry and their impact on the environment and the food chain. In Pesticides in the Modern World-Risks and Benefits; IntechOpen: London, UK, 2011. [Google Scholar]
- Rojo-Poveda, O.; Barbosa-Pereira, L.; Mateus-Reguengo, L.; Bertolino, M.; Stévigny, C.; Zeppa, G. Effects of particle size and extraction methods on cocoa bean shell functional beverage. Nutrients 2019, 11, 867. [Google Scholar] [CrossRef]
- Kofink, M.; Papagiannopoulos, M.; Galensa, R. (-)-Catechin in cocoa and chocolate: occurence and analysis of an atypical flavan-3-ol enantiomer. Molecules 2007, 12, 1274–1288. [Google Scholar] [CrossRef]
- Adamafio, N. Theobromine toxicity and remediation of cocoa by-products: An overview. J. Biol. Sci. 2013, 13, 570–576. [Google Scholar] [CrossRef]
- Olaifa, F.; Hamzat, R.; Oyetoyan, O. Acute toxicity of ethanol extracts of cocoa bean shell on Sarotherodon galilaeus juveniles. J. Fish. Int. 2008, 3, 56–60. [Google Scholar]
- Martín-Cabrejas, M.A.; Valiente, C.; Esteban, R.M.; Mollá, E.; Waldron, K. Cocoa hull: a potential source of dietary fibre. J. Sci. Food Agric. 1994, 66, 307–311. [Google Scholar] [CrossRef]
- Badrie, N.; Bekele, F.; Sikora, E.; Sikora, M. Cocoa agronomy, quality, nutritional, and health aspects. Crit. Rev. Food Sci. Nutr. 2015, 55, 620–659. [Google Scholar] [CrossRef] [PubMed]
- Kowalska, H.; Czajkowska, K.; Cichowska, J.; Lenart, A. What’s new in biopotential of fruit and vegetable by-products applied in the food processing industry. Trends Food Sci. Technol. 2017, 67, 150–159. [Google Scholar] [CrossRef]
- Ravindran, R.; Jaiswal, A.K. Exploitation of food industry waste for high-value products. Trends Biotechnol. 2016, 34, 58–69. [Google Scholar] [CrossRef] [PubMed]
- Okiyama, D.C.G.; Navarro, S.L.B.; Rodrigues, C.E.C. Cocoa shell and its compounds: Applications in the food industry. Trends Food Sci. Technol. 2017, 63, 103–112. [Google Scholar] [CrossRef]
- Panak Balentić, J.; Ačkar, Đ.; Jokić, S.; Jozinović, A.; Babić, J.; Miličević, B.; Šubarić, D.; Pavlović, N. Cocoa shell: A by-product with great potential for wide application. Molecules 2018, 23, 1404. [Google Scholar] [CrossRef] [PubMed]
- Thangaraj, P. Proximate Composition Analysis. In Pharmacological Assays of Plant-Based Natural Products; Springer: Berlin, Germany, 2016; pp. 21–31. [Google Scholar]
- Pérez, E.; Méndez, A.; León, M.; Hernández, G.; Sívoli, L. Proximal composition and the nutritional and functional properties of cocoa by-products (pods and husks) for their use in the food industry. Cocoa By-Prod. Technol. Rheol. Styl. Nutr. 2015, 10, 219–234. [Google Scholar]
- Diomande, D.; Antheaume, I.; Leroux, M.; Lalande, J.; Balayssac, S.; Remaud, G.S.; Tea, I. Multi-element, multi-compound isotope profiling as a means to distinguish the geographical and varietal origin of fermented cocoa (Theobroma cacao L.) beans. Food Chem. 2015, 188, 576–582. [Google Scholar] [CrossRef]
- Agus, B.A.P.; Mohamad, N.N.; Hussain, N. Composition of unfermented, unroasted, roasted cocoa beans and cocoa shells from Peninsular Malaysia. J. Food Meas. Charact. 2018, 12, 2581–2589. [Google Scholar] [CrossRef]
- Bonvehı, J.S.; Coll, F.V. Protein quality assessment in cocoa husk. Food Res. Int. 1999, 32, 201–208. [Google Scholar] [CrossRef]
- Osundahunsi, O.; Bolade, M.; Akinbinu, A. Effect of cocoa shell ash as an alkalizing agent on cocoa products. J. Appl. Sci. 2007, 7, 1674–1678. [Google Scholar]
- WHO. Quality Control Methods for Herbal Materials; WHO: Geneva, Switzerland, 2011. [Google Scholar]
- Arlorio, M.; Coisson, J.; Restani, P.; Martelli, A. Characterization of pectins and some secondary compounds from Theobroma cacao hulls. J. Food Sci. 2001, 66, 653–656. [Google Scholar] [CrossRef]
- Barbosa-Pereira, L.; Rojo-Poveda, O.; Ferrocino, I.; Giordano, M.; Zeppa, G. Analytical dataset on volatile compounds of cocoa bean shells from different cultivars and geographical origins. Data Brief 2019, 25, 104268. [Google Scholar] [CrossRef] [PubMed]
- Vojvodić, A.; Komes, D.; Vovk, I.; Belščak-Cvitanović, A.; Bušić, A. Compositional evaluation of selected agro-industrial wastes as valuable sources for the recovery of complex carbohydrates. Food Res. Int. 2016, 89, 565–573. [Google Scholar] [CrossRef]
- Bonvehí, J.S.; Benería, M.A. Composition of dietary fibre in cocoa husk. Z. Für Lebensm. Und-Forsch. A 1998, 207, 105–109. [Google Scholar]
- Mancini, G.; Papirio, S.; Lens, P.N.; Esposito, G. Anaerobic Digestion of Lignocellulosic Materials Using Ethanol-Organosolv Pretreatment. Environ. Eng. Sci. 2018, 35, 953–960. [Google Scholar] [CrossRef]
- Martínez, R.; Torres, P.; Meneses, M.; Figueroa, J.; Pérez-Álvarez, J.; Viuda-Martos, M. Chemical, technological and in vitro antioxidant properties of cocoa (Theobroma cacao L.) co-products. Food Res. Int. 2012, 49, 39–45. [Google Scholar] [CrossRef]
- Okiyama, D.C.; Soares, I.D.; Toda, T.A.; Oliveira, A.L.; Rodrigues, C.E. Effect of the temperature on the kinetics of cocoa bean shell fat extraction using pressurized ethanol and evaluation of the lipid fraction and defatted meal. Ind. Crops Prod. 2019, 130, 96–103. [Google Scholar] [CrossRef]
- Belitz, H.-D.; Grosch, W.; Schieberle, P. Food Chemistry. In Cocoa and Chocolate, 4th ed.; Springer: Berlin, Germany, 2009; pp. 959–969. [Google Scholar]
- Abarca, D.; Martínez, R.; Muñoz, J.J.; Torres, M.P.; Vargas, G. Residuos de café, cacao y cladodio de tuna: Fuentes promisorias de fibra dietaria. Rev. Tecnológica-Espol 2010, 23, 63–69. [Google Scholar]
- Bonvehí, J.S. Constituents of cocoa husks. Z. Für Nat. C 1998, 53, 785–792. [Google Scholar] [CrossRef]
- Lecumberri, E.; Mateos, R.; Izquierdo-Pulido, M.; Rupérez, P.; Goya, L.; Bravo, L. Dietary fibre composition, antioxidant capacity and physico-chemical properties of a fibre-rich product from cocoa (Theobroma cacao L.). Food Chem. 2007, 104, 948–954. [Google Scholar] [CrossRef]
- Nsor-Atindana, J.; Zhong, F.; Mothibe, K.J.; Bangoura, M.L.; Lagnika, C. Quantification of total polyphenolic content and antimicrobial activity of cocoa (Theobroma cacao L.) Bean Shells. Pak. J. Nutr. 2012, 11, 574. [Google Scholar]
- Belščak-Cvitanović, A.; Vojvodić, A.; Bušić, A.; Keppler, J.; Steffen-Heins, A.; Komes, D. Encapsulation templated approach to valorization of cocoa husk, poppy and hemp macrostructural and bioactive constituents. Ind. Crops Prod. 2018, 112, 402–411. [Google Scholar] [CrossRef]
- Ogunsipe, M.; Balogun, K.; Oladepo, A.; Ayoola, M.; Arikewuyo, M. Nutritive value of cocoa bean shell meal and its effect on growth and haematology of weaning rabbits. Niger. J Agric Food Environ. 2017, 13, 23–28. [Google Scholar]
- Sandoval, A.J.; Barreiro, J.A.; De Sousa, A.; Valera, D.; López, J.V.; Alejandro, J. Composition and Thermogravimetric Characterization of Components of Venezuelan Fermented and dry Trinitario Cocoa Beans (Theobroma cacao L.): Whole Beans, Peeled Beans and Shells. Rev. Técnica De La Fac. De Ing. Univ. Del Zulia 2019, 1, 39–47. [Google Scholar] [CrossRef]
- El-Saied, H.M.; Morsi, M.; Amer, M. Composition of cocoa shell fat as related to cocoa butter. Für Ernährungswiss. 1981, 20, 145–151. [Google Scholar] [CrossRef] [PubMed]
- Jentzsch, P.V.; Ciobotă, V.; Salinas, W.; Kampe, B.; Aponte, P.M.; Rösch, P.; Popp, J.; Ramos, L.A. Distinction of Ecuadorian varieties of fermented cocoa beans using Raman spectroscopy. Food Chem. 2016, 211, 274–280. [Google Scholar] [CrossRef]
- Nair, K.P. Cocoa (Theobroma cacao L.)—The Agronomy and Economy of Important Tree Crops of the Developing World; Elsevier: Amsterdam, The Netherlands, 2010; pp. 131–180. [Google Scholar]
- Santana, D.P.; Sanchez, J.L.R.; Calle, J.; de Villavicencio, M.N.; Ortega, L.D.; Llanes, L.H. Utilización de la cascarilla de cacao como fuente de fibra dietética y antioxidantes en la elaboración de galletas dulces/Use of cocoa bean shell as a source of dietetic fiber and antioxidants in the production of sweet cookies. Cienc. Y Tecnol. De Aliment. 2018, 28, 62–67. [Google Scholar]
- Chan, S.-Y.; Choo, W.-S. Effect of extraction conditions on the yield and chemical properties of pectin from cocoa husks. Food Chem. 2013, 141, 3752–3758. [Google Scholar] [CrossRef]
- Mollea, C.; Chiampo, F.; Conti, R. Extraction and characterization of pectins from cocoa husks: A preliminary study. Food Chem. 2008, 107, 1353–1356. [Google Scholar] [CrossRef]
- Nazaruddin, R. Effect of ammonium oxalate and acetic acid at several extraction time and pH on some physicochemical properties of pectin from cocoa husks (Theobroma cacao). Afr. J. Food Sci. 2011, 5, 790–798. [Google Scholar] [CrossRef]
- Chung, B.Y.; Iiyama, K.; Han, K.W. Food Science; Compositional Characterization Of Cacao (Theobroma cacao L.) Hull. J. Appl. Biol. Chem. 2003, 46, 12–16. [Google Scholar]
- Vītola, V.; Ciproviča, I. The effect of cocoa beans heavy and trace elements on safety and stability of confectionery products. Rural Sustain. Res. 2016, 35, 19–23. [Google Scholar] [CrossRef]
- Knapp, A.W.; Coward, K.H. The vitamin D activity of cacao shell: the effect of the fermenting and drying of cacao on the vitamin D potency of cacao shell. II. The origin of vitamin D in cacao shell. Biochem. J. 1935, 29, 2728. [Google Scholar]
- Manzano, P.; Hernández, J.; Quijano-Avilés, M.; Barragán, A.; Chóez-Guaranda, I.; Viteri, R.; Valle, O. Polyphenols extracted from Theobroma cacao waste and its utility as antioxidant. Emir. J. Food Agric. 2017, 29, 45. [Google Scholar] [CrossRef]
- Baharum, Z.; Akim, A.; Taufiq-Yap, Y.; Hamid, R.; Kasran, R. In vitro antioxidant and antiproliferative activities of methanolic plant part extracts of Theobroma cacao. Molecules 2014, 19, 18317–18331. [Google Scholar] [CrossRef]
- Grillo, G.; Boffa, L.; Binello, A.; Mantegna, S.; Cravotto, G.; Chemat, F.; Dizhbite, T.; Lauberte, L.; Telysheva, G. Cocoa bean shell waste valorisation; extraction from lab to pilot-scale cavitational reactors. Food Res. Int. 2019, 115, 200–208. [Google Scholar] [CrossRef]
- Jokić, S.; Pavlović, N.; Jozinović, A.; Ačkar, Đ.; Babić, J.; Šubarić, D. High-Voltage Electric Discharge Extraction of Bioactive Compounds from the Cocoa Bean Shell. Chem. Biochem. Eng. Q. 2019, 33, 271–280. [Google Scholar] [CrossRef]
- Barbosa-Pereira, L.; Guglielmetti, A.; Zeppa, G. Pulsed electric field assisted extraction of bioactive compounds from cocoa bean shell and coffee silverskin. Food Bioprocess Technol. 2018, 11, 818–835. [Google Scholar] [CrossRef]
- Mazzutti, S.; Rodrigues, L.G.G.; Mezzomo, N.; Venturi, V.; Ferreira, S.R.S. Integrated green-based processes using supercritical CO2 and pressurized ethanol applied to recover antioxidant compouds from cocoa (Theobroma cacao) bean hulls. J. Supercrit. Fluids 2018, 135, 52–59. [Google Scholar] [CrossRef]
- Pavlović, N.; Jakovljević, M.; Miškulin, M.; Molnar, M.; Ačkar, Đ.; Jokić, S. Green extraction techniques of bioactive components from cocoa shell. Croat. J. Food Sci. Technol. 2019, 11, 11–20. [Google Scholar] [CrossRef]
- Hernández-Hernández, C.; Morales-Sillero, A.; Fernández-Bolaños, J.; Bermúdez-Oria, A.; Morales, A.A.; Rodríguez-Gutiérrez, G. Cocoa bean husk: Industrial source of antioxidant phenolic extract. J. Sci. Food Agric. 2019, 99, 325–333. [Google Scholar] [CrossRef] [PubMed]
- Jokić, S.; Gagić, T.; Knez, Ž.; Šubarić, D.; Škerget, M. Separation of Active Compounds from Food by-Product (Cocoa Shell) Using Subcritical Water Extraction. Molecules 2018, 23, 1408. [Google Scholar] [CrossRef] [PubMed]
- Okiyama, D.C.; Soares, I.D.; Cuevas, M.S.; Crevelin, E.J.; Moraes, L.A.; Melo, M.P.; Oliveira, A.L.; Rodrigues, C.E. Pressurized liquid extraction of flavanols and alkaloids from cocoa bean shell using ethanol as solvent. Food Res. Int. 2018, 114, 20–29. [Google Scholar] [CrossRef] [PubMed]
- Papillo, V.A.; Locatelli, M.; Travaglia, F.; Bordiga, M.; Garino, C.; Coïsson, J.D.; Arlorio, M. Cocoa hulls polyphenols stabilized by microencapsulation as functional ingredient for bakery applications. Food Res. Int. 2019, 115, 511–518. [Google Scholar] [CrossRef] [PubMed]
- Barišić, V.; Flanjak, I.; Križić, I.; Jozinović, A.; Šubarić, D.; Babić, J.; Miličević, B.; Ačkar, Đ. Impact of high-voltage electric discharge treatment on cocoa shell phenolic components and methylxanthines. J. Food Process Eng. 2019, 43, e13057. [Google Scholar] [CrossRef]
- Hartati, I. Hydrotopic extraction of theobromine from cocoa bean shell. Momentum 2010, 6, 17–20. [Google Scholar]
- Barbosa-Pereira, L.; Rojo-Poveda, O.; Ferrocino, I.; Giordano, M.; Zeppa, G. Assessment of volatile fingerprint by HS-SPME/GC-qMS and E-nose for the classification of cocoa bean shells using chemometrics. Food Res. Int. 2019, 123, 684–696. [Google Scholar] [CrossRef]
- Bentil, J.A. Enhancement of the nutritive value of cocoa (Theobroma cacao) bean shells for use as feed for animals through a two-stage solid state fermentation with Pleurotus ostreatus and Aspergillus niger. Int. J. Appl. Microbiol. Biotechnol. Res. 2012, 3, 20–30. [Google Scholar]
- Pätzold, R.; Brückner, H. Gas chromatographic determination and mechanism of formation of D-amino acids occurring in fermented and roasted cocoa beans, cocoa powder, chocolate and cocoa shell. Amino Acids 2006, 31, 63. [Google Scholar] [CrossRef]
- González-Alejo, F.A.; Barajas-Fernández, J.; Olán-Acosta, M.d.l.Á.; Lagunes-Gálvez, L.M.; García-Alamilla, P. Supercritical Fluid Extraction of Fat and Caffeine with Theobromine Retention in the Cocoa Shell. Processes 2019, 7, 385. [Google Scholar] [CrossRef]
- Münch, M.; Schieberle, P. A sensitive and selective method for the quantitative determination of fatty acid tryptamides as shell indicators in cocoa products. Z. Für Lebensm. Und-Forsch. A 1999, 208, 39–46. [Google Scholar] [CrossRef]
- Janßen, K.; Matissek, R. Fatty acid tryptamides as shell indicators for cocoa products and as quality parameters for cocoa butter. Eur. Food Res. Technol. 2002, 214, 259–264. [Google Scholar] [CrossRef]
- Grillo, G.; Boffa, L.; Binello, A.; Mantegna, S.; Cravotto, G.; Chemat, F.; Dizhbite, T.; Lauberte, L.; Telysheva, G. Analytical dataset of Ecuadorian cocoa shells and beans. Data Brief 2019, 22, 56–64. [Google Scholar] [CrossRef]
- Lessa, O.A.; dos Santos Reis, N.; Leite, S.G.F.; Gutarra, M.L.E.; Souza, A.O.; Gualberto, S.A.; de Oliveira, J.R.; Aguiar-Oliveira, E.; Franco, M. Effect of the solid state fermentation of cocoa shell on the secondary metabolites, antioxidant activity, and fatty acids. Food Sci. Biotechnol. 2018, 27, 107–113. [Google Scholar] [CrossRef]
- Ruesgas-Ramón, M.; Figueroa-Espinoza, M.C.; Durand, E.; Suárez-Quiroz, M.L.; González-Ríos, O.; Rocher, A.; Reversat, G.; Vercauteren, J.; Oger, C.; Galano, J.-M. Identification and quantification of phytoprostanes and phytofurans of coffee and cocoa by-and co-products. Food Funct. 2019, 10, 6882–6891. [Google Scholar] [CrossRef]
- Redgwell, R.; Trovato, V.; Merinat, S.; Curti, D.; Hediger, S.; Manez, A. Dietary fibre in cocoa shell: characterisation of component polysaccharides. Food Chem. 2003, 81, 103–112. [Google Scholar] [CrossRef]
- Lecumberri, E.; Goya, L.; Mateos, R.; Alía, M.; Ramos, S.; Izquierdo-Pulido, M.; Bravo, L. A diet rich in dietary fiber from cocoa improves lipid profile and reduces malondialdehyde in hypercholesterolemic rats. Nutrition 2007, 23, 332–341. [Google Scholar] [CrossRef]
- Nsor-Atindana, J.; Zhong, F.; Mothibe, K.J. In vitro hypoglycemic and cholesterol lowering effects of dietary fiber prepared from cocoa (Theobroma cacao L.) shells. Food Funct. 2012, 3, 1044–1050. [Google Scholar] [CrossRef]
- Rossin, D.; Barbosa-Pereira, L.; Iaia, N.; Testa, G.; Sottero, B.; Poli, G.; Zeppa, G.; Biasi, F. A Dietary Mixture of Oxysterols Induces In Vitro Intestinal Inflammation through TLR2/4 Activation: The Protective Effect of Cocoa Bean Shells. Antioxidants 2019, 8, 151. [Google Scholar] [CrossRef]
- Sarah, M.; Hanum, F.; Rizky, M.; Hisham, M. Microwave-assisted extraction of pectin from cocoa peel. In Proceedings of the IOP Conference Series: Earth and Environmental Science, Medan, Indonesia, 7–8 November 2017; p. 012079. [Google Scholar]
- Mollea, C.; Chiampo, F. Valorization of Cocoa Husks: Pectin Recovery. Int. J. Food Sci. 2019, 2019. [Google Scholar] [CrossRef] [PubMed]
- Wollgast, J.; Anklam, E. Review on polyphenols in Theobroma cacao: changes in composition during the manufacture of chocolate and methodology for identification and quantification. Food Res. Int. 2000, 33, 423–447. [Google Scholar] [CrossRef]
- Vauzour, D.; Rodriguez-Mateos, A.; Corona, G.; Oruna-Concha, M.J.; Spencer, J.P. Polyphenols and human health: prevention of disease and mechanisms of action. Nutrients 2010, 2, 1106–1131. [Google Scholar] [CrossRef] [PubMed]
- Hussain, T.; Tan, B.; Yin, Y.; Blachier, F.; Tossou, M.C.; Rahu, N. Oxidative stress and inflammation: what polyphenols can do for us? Oxidative Med. Cell. Longev. 2016, 2016. [Google Scholar] [CrossRef]
- Martin, M.Á.; Goya, L.; Ramos, S. Antidiabetic actions of cocoa flavanols. Mol. Nutr. Food Res. 2016, 60, 1756–1769. [Google Scholar] [CrossRef]
- Rodriguez-Mateos, A.; Vauzour, D.; Krueger, C.G.; Shanmuganayagam, D.; Reed, J.; Calani, L.; Mena, P.; Del Rio, D.; Crozier, A. Bioavailability, bioactivity and impact on health of dietary flavonoids and related compounds: an update. Arch. Toxicol. 2014, 88, 1803–1853. [Google Scholar] [CrossRef]
- Vauzour, D. Effect of flavonoids on learning, memory and neurocognitive performance: relevance and potential implications for Alzheimer’s disease pathophysiology. J. Sci. Food Agric. 2014, 94, 1042–1056. [Google Scholar] [CrossRef]
- Neshatdoust, S.; Saunders, C.; Castle, S.M.; Vauzour, D.; Williams, C.; Butler, L.; Lovegrove, J.A.; Spencer, J.P. High-flavonoid intake induces cognitive improvements linked to changes in serum brain-derived neurotrophic factor: two randomised, controlled trials. Nutr. Healthy Aging 2016, 4, 81–93. [Google Scholar] [CrossRef]
- Arlorio, M.; Coïsson, J.; Travaglia, F.; Varsaldi, F.; Miglio, G.; Lombardi, G.; Martelli, A. Antioxidant and biological activity of phenolic pigments from Theobroma cacao hulls extracted with supercritical CO2. Food Res. Int. 2005, 38, 1009–1014. [Google Scholar] [CrossRef]
- Hernández, S.M.P.; Estévez, J.J.; Giraldo, L.J.L.; Méndez, C.J.M. Supercritical extraction of bioactive compounds from cocoa husk: study of the main parameters. Rev. Fac. De Ing. Univ. De Antioq. 2019, 95–105. [Google Scholar] [CrossRef]
- Yusof, M.; Huzaimi, A.; Abd Gani, S.S.; Zaidan, U.H.; Halmi, M.I.E.; Zainudin, B.H. Optimization of an Ultrasound-Assisted Extraction Condition for Flavonoid Compounds from Cocoa Shells (Theobroma cacao) Using Response Surface Methodology. Molecules 2019, 24, 711. [Google Scholar] [CrossRef] [PubMed]
- Zhong, J.-L.; Muhammad, N.; Gu, Y.-C.; Yan, W.-D. A simple and efficient method for enrichment of cocoa polyphenols from cocoa bean husks with macroporous resins following a scale-up separation. J. Food Eng. 2019, 243, 82–88. [Google Scholar] [CrossRef]
- Bruna, C.; Eichholz, I.; Rohn, S.; Kroh, L.; Huyskens-Keil, S. Bioactive compounds and antioxidant activity of cocoa hulls (Theobroma cacao L.) from different origins. J. Appl. Bot. Food Qual. 2009, 83, 9–13. [Google Scholar]
- Utami, R.; Armunanto, R.; Supriyanto, S. Effects of cocoa bean (Theobroma cacao L.) fermentation on phenolic content, antioxidant activity and functional group of cocoa bean shell. Pak. J. Nutr 2016, 15, 948–953. [Google Scholar]
- Hernández-Hernández, C.; Viera-Alcaide, I.; Morales-Sillero, A.M.; Fernández-Bolaños, J.; Rodríguez-Gutiérrez, G. Bioactive compounds in Mexican genotypes of cocoa cotyledon and husk. Food Chem. 2018, 240, 831–839. [Google Scholar] [CrossRef] [PubMed]
- Karim, A.A.; Azlan, A.; Ismail, A.; Hashim, P.; Abdullah, N.A. Antioxidant properties of cocoa pods and shells. Malays. Cocoa J. 2014, 8, 49–56. [Google Scholar]
- Azizah, A.; Ruslawati, N.N.; Tee, T.S. Extraction and characterization of antioxidant from cocoa by-products. Food Chem. 1999, 64, 199–202. [Google Scholar] [CrossRef]
- Awarikabey, E.; Amponsah, I.K.; Woode, M.Y. The value of the cocoa bean shell (hull) and the effect of various processing methods on the phyto-constituents and antioxidant activity of the nib and shell. J. Nat. Prod. Plant Resour. 2014, 4, 58–64. [Google Scholar]
- Granato, D.; Shahidi, F.; Wrolstad, R.; Kilmartin, P.; Melton, L.D.; Hidalgo, F.J.; Miyashita, K.; van Camp, J.; Alasalvar, C.; Ismail, A.B. Antioxidant activity, total phenolics and flavonoids contents: Should we ban in vitro screening methods? Food Chem. 2018, 264, 471–475. [Google Scholar] [CrossRef]
- González, J.; Coronela, L.; Lisa, L. UHPLC-DAD-ESI-MS/MS Analysis of Flavonoids from Ethanolic Extracts of Theobroma cacao Husk in Cuba. Open Access Libr. J. 2019, 6, 1. [Google Scholar] [CrossRef]
- Rebollo-Hernanz, M.; Zhang, Q.; Aguilera, Y.; Martín-Cabrejas, M.A.; de Mejia, E.G. Cocoa Shell Aqueous Phenolic Extract Preserves Mitochondrial Function and Insulin Sensitivity by Attenuating Inflammation between Macrophages and Adipocytes In Vitro. Mol. Nutr. Food Res. 2019, 63, 1801413. [Google Scholar] [CrossRef] [PubMed]
- Heck, C.I.; De Mejia, E.G. Yerba Mate Tea (Ilex paraguariensis): a comprehensive review on chemistry, health implications, and technological considerations. J. Food Sci. 2007, 72, R138–R151. [Google Scholar] [CrossRef] [PubMed]
- Bispo, M.S.; Veloso, M.C.C.; Pinheiro, H.L.C.; De Oliveira, R.F.; Reis, J.O.N.; De Andrade, J.B. Simultaneous determination of caffeine, theobromine, and theophylline by high-performance liquid chromatography. J. Chromatogr. Sci. 2002, 40, 45–48. [Google Scholar] [CrossRef] [PubMed]
- Authority, E.F.S. Theobromine as undesirable substances in animal feed-Scientific opinion of the Panel on Contaminants in the Food Chain. EFSA J. 2008, 6, 725. [Google Scholar]
- Baggott, M.J.; Childs, E.; Hart, A.B.; de Bruin, E.; Palmer, A.A.; Wilkinson, J.E.; de Wit, H. Psychopharmacology of theobromine in healthy volunteers. Psychopharmacology 2013, 228, 109–118. [Google Scholar] [CrossRef]
- Ashihara, H.; Sano, H.; Crozier, A. Caffeine and related purine alkaloids: biosynthesis, catabolism, function and genetic engineering. Phytochemistry 2008, 69, 841–856. [Google Scholar] [CrossRef]
- Zheng, X.-Q.; Koyama, Y.; Nagai, C.; Ashihara, H. Biosynthesis, accumulation and degradation of theobromine in developing Theobroma cacao fruits. J. Plant Physiol. 2004, 161, 363–369. [Google Scholar] [CrossRef]
- Hue, C.; Gunata, Z.; Breysse, A.; Davrieux, F.; Boulanger, R.; Sauvage, F.-X. Impact of fermentation on nitrogenous compounds of cocoa beans (Theobroma cacao L.) from various origins. Food Chem. 2016, 192, 958–964. [Google Scholar] [CrossRef]
- Júnior, P.C.G.; dos Santos, V.B.; Lopes, A.S.; de Souza, J.P.I.; Pina, J.R.S.; Júnior, G.C.A.C.; Marinho, P.S.B. Determination of theobromine and caffeine in fermented and unfermented Amazonian cocoa (Theobroma cacao L.) beans using square wave voltammetry after chromatographic separation. Food Control 2020, 108, 106887. [Google Scholar] [CrossRef]
- Sansone, R.; Ottaviani, J.I.; Rodriguez-Mateos, A.; Heinen, Y.; Noske, D.; Spencer, J.P.; Crozier, A.; Merx, M.W.; Kelm, M.; Schroeter, H. Methylxanthines enhance the effects of cocoa flavanols on cardiovascular function: randomized, double-masked controlled studies. Am. J. Clin. Nutr. 2017, 105, 352–360. [Google Scholar] [CrossRef]
- Coimbra, M.C.; Jorge, N. Proximate composition of guariroba (Syagrus oleracea), jerivá (Syagrus romanzoffiana) and macaúba (Acrocomia aculeata) palm fruits. Food Res. Int. 2011, 44, 2139–2142. [Google Scholar] [CrossRef]
- Thyssen, G.M.; Keil, C.; Wolff, M.; Sperling, M.; Kadow, D.; Haase, H.; Karst, U. Bioimaging of the elemental distribution in cocoa beans by means of LA-ICP-TQMS. J. Anal. At. Spectrom. 2018, 33, 187–194. [Google Scholar] [CrossRef]
- Mandrile, L.; Barbosa-Pereira, L.; Sorensen, K.M.; Giovannozzi, A.M.; Zeppa, G.; Engelsen, S.B.; Rossi, A.M. Authentication of cocoa bean shells by near-and mid-infrared spectroscopy and inductively coupled plasma-optical emission spectroscopy. Food Chem. 2019, 292, 47–57. [Google Scholar] [CrossRef] [PubMed]
- Wickramasuriya, A.M.; Dunwell, J.M. Cacao biotechnology: current status and future prospects. Plant Biotechnol. J. 2018, 16, 4–17. [Google Scholar] [CrossRef] [PubMed]
- Kon, S.K.; Henry, K.M. The effect of feeding cacao shell to cows on the vitamin D content of butter (milk). Biochem. J. 1935, 29, 2051. [Google Scholar] [CrossRef] [PubMed]
- Collar, C.; Rosell, C.M.; Muguerza, B.; Moulay, L. Breadmaking performance and keeping behavior of cocoa-soluble fiber-enriched wheat breads. Food Sci. Technol. Int. 2009, 15, 79–87. [Google Scholar] [CrossRef]
- Kārkliņa, D.; Gedrovica, I.; Reca, M.; Kronberga, M. Production of biscuits with higher nutritional value. Proc. Latvian Acad. Sci. Sec. B. Nat. Exact Appl.Sci. 2012, 66, 113–116. [Google Scholar] [CrossRef]
- Öztürk, E.; Ova, G. Evaluation of Cocoa Bean Hulls as a Fat Replacer On Functional Cake Production. Turk. J. Agric. -Food Sci. Technol. 2018, 6, 1043–1050. [Google Scholar] [CrossRef]
- Martínez-Cervera, S.; Salvador, A.; Muguerza, B.; Moulay, L.; Fiszman, S. Cocoa fibre and its application as a fat replacer in chocolate muffins. LWT-Food Sci. Technol. 2011, 44, 729–736. [Google Scholar] [CrossRef]
- Eggen, I.B. Cocoa Shell Extract. US Patent No. US4156030A, 22 May 1979. [Google Scholar]
- Quijano-Aviles, M.F.; Franco-Agurto, G.L.; Suárez-Quirumbay, K.B.; Barragán-Lucas, A.D.; Manzano-Santana, P.I. Linear programming formulation of a dairy drink made of cocoa, coffee and orange by-products. Emir. J. Food Agric. 2016, 554–559. [Google Scholar] [CrossRef]
- Jozinović, A.; Panak Balentić, J.; Ačkar, Đ.; Babić, J.; Pajin, B.; Miličević, B.; Guberac, S.; Vrdoljak, A.; Šubarić, D. Cocoa husk application in the enrichment of extruded snack products. J. Food Process. Preserv. 2019, 43, e13866. [Google Scholar] [CrossRef]
- Altin, G.; Gültekin-Özgüven, M.; Ozcelik, B. Chitosan coated liposome dispersions loaded with cacao hull waste extract: Effect of spray drying on physico-chemical stability and in vitro bioaccessibility. J. Food Eng. 2018, 223, 91–98. [Google Scholar] [CrossRef]
- Altin, G.; Gültekin-Özgüven, M.; Ozcelik, B. Liposomal dispersion and powder systems for delivery of cocoa hull waste phenolics via Ayran (drinking yoghurt): Comparative studies on in-vitro bioaccessibility and antioxidant capacity. Food Hydrocoll. 2018, 81, 364–370. [Google Scholar] [CrossRef]
- Ismail, A.; Yee, C.L. Antioxidative effects of extracts of cocoa shell, roselle seeds and a combination of both extracts on the susceptibility of cooked beef to lipid oxidation. J. Food Technol. 2006, 4, 10–15. [Google Scholar]
- Hernández-Hernández, C.; Morales-Sillero, A.; Fernández-Prior, M.Á.; Fernández-Bolaños, J.; de la Paz Aguilera-Herrera, M.; Rodríguez-Gutiérrez, G. Extra virgin olive oil jam enriched with cocoa bean husk extract rich in theobromine and phenols. LWT 2019, 111, 278–283. [Google Scholar] [CrossRef]
- Handojo, L.; Cherilisa; Indarto, A. Cocoa bean skin waste as potential raw material for liquid smoke production. Environ. Technol. 2018, 41, 1044–1153. [Google Scholar] [CrossRef]
- Tran, T.N.; Heredia-Guerrero, J.A.; Mai, B.T.; Ceseracciu, L.; Marini, L.; Athanassiou, A.; Bayer, I.S. Bioelastomers based on cocoa shell waste with antioxidant ability. Adv. Sustain. Syst. 2017, 1, 1700002. [Google Scholar] [CrossRef]
- Adeyemo, G.; Ajayi, A.; Longe, O.; Olubamiwa, O. Gut morphology and internal organs of broiler birds fed graded levels of bio-detheobrominized cocoa bean shell (CBS) based diets. J. Exp. Agric. Int. 2015, 172–177. [Google Scholar] [CrossRef]
- Drolet, R.; Arendt, T.; Stowe, C. Cacao bean shell poisoning in a dog. J. Am. Vet. Med Assoc. 1984, 185, 902. [Google Scholar]
- Adeyemo, G.; Ajayi, A.; Olubamiwa, O. Performance of broilers fed graded levels of bio-detheobrominized cocoa bean shell (CBS) based diets. Am. J. Exp. Agric. 2015, 5, 540–545. [Google Scholar] [CrossRef]
- Emiola, I.; Ojebiyi, O.; Akande, T. Performance and organ weights of laying hens fed diets containing graded levels of sun-dried cocoa bean shell (CBS). Int.J. Poult. Sci. 2011, 10, 986–989. [Google Scholar] [CrossRef]
- Olumide, M.D.; Akinsoyinu, A.; Hamzat, R.A. Egg quality characteristics of layers fed raw, fermented and enzyme-treated cocoa bean shell based diets. Pac. J. Sci. Technol. 2014, 15, 289–296. [Google Scholar]
- Oduniyi, O.S. Egg weight and shell quality characteristics of laying hens fed with graded levels of cocoa bean shell. In Proceedings of the Sixth International Scientific Agricultural Symposium" Agrosym 2015", Jahorina, Bosnia and Herzegovina, 15–18 October 2015; pp. 1684–1692. [Google Scholar]
- Day, E.J.; Dilworth, B.C. Toxicity of jimson weed seed and cocoa shell meal to broilers. Poult. Sci. 1984, 63, 466–468. [Google Scholar] [CrossRef]
- Adeyina, A.; Apata, D.; Annongu, A.; Olatunde, O.; Alli, O.; Okupke, K. Performance and physiological response of weaner rabbits fed hot water treated cocoa bean shell-based diet. Res. J. Anim. Vet. Sci. 2010, 5, 53–57. [Google Scholar]
- Ayinde, O.; Ojo, V.; Adeyina, A.; Adesoye, O. Economics of using cocoa bean shell as feed supplement for rabbits. Pak. J. Nutr. 2010, 9, 195–197. [Google Scholar] [CrossRef]
- Amin, M.; Cahyono, A. The Use of Cocoa Bean Waste as a Supplement in Male Bali Cattle Feeding. Proc. Int. sem. LPVT 2016, 45–49. [Google Scholar]
- Yajima, A.; Owada, H.; Kobayashi, S.; Komatsu, N.; Takehara, K.; Ito, M.; Matsuda, K.; Sato, K.; Itabashi, H.; Sugimura, S. Cacao bean husk: an applicable bedding material in dairy free-stall barns. Asian-Australas. J. Anim. Sci. 2017, 30, 1048. [Google Scholar] [CrossRef][Green Version]
- Magistrelli, D.; Zanchi, R.; Malagutti, L.; Galassi, G.; Canzi, E.; Rosi, F. Effects of cocoa husk feeding on the composition of swine intestinal microbiota. J. Agric. Food Chem. 2016, 64, 2046–2052. [Google Scholar] [CrossRef]
- Bamba, Y.; Ouattara, N.; Soro, Y.; Ouattara, A.; Yao, K.; Gourène, G. Evaluation of production efficiency of Nile tilapia (Oreochromis niloticus L.) fed diets containing crop residues in combination with cocoa bean shell and coconut oil cake in Côte d’Ivoire. Livest. Res. Rural Dev. 2014, 26, 10. [Google Scholar]
- Olubamiwa, O.; Ikyo, S.; Adebowale, B.; Omojola, A.; Hamzat, R. Effect of boiling time on the utilization of cocoa bean shell in laying hen feeds. Int. J. Poult. Sci. 2006, 5, 1137–1139. [Google Scholar]
- Makinde, O.J.; Okunade, S.A.; Opoola, E.; Sikiru, A.B.; Ajide, S.O.; Elaigwu, S. Exploration of Cocoa (Theobroma cacao) By-Products as Valuable Potential Resources in Livestock Feeds and Feeding Systems. In Theobroma cacao-Deploying Science for Sustainability of Global Cocoa Economy; IntechOpen: London, UK, 2019. [Google Scholar]
- Aromolaran, O.; Ogunsakin, F.M. Degradation of Theobromine in Cocoa (Theobroma cacao) by-products by Fermentation with Aspergillus niger. South Asian J. Res. Microbiol. 2018, 2, 1–6. [Google Scholar] [CrossRef]
- Oduro-Mensah, D.; Ocloo, A.; Lowor, S.T.; Mingle, C.; Okine, L.K.-A.; Adamafio, N.A. Bio-detheobromination of cocoa pod husks: reduction of ochratoxin A content without change in nutrient profile. Microb. Cell Factories 2018, 17, 79. [Google Scholar] [CrossRef]
- Mancini, G.; Papirio, S.; Lens, P.N.; Esposito, G. Effect of N-methylmorpholine-N-oxide pretreatment on biogas production from rice straw, cocoa shell, and hazelnut skin. Environ. Eng. Sci. 2016, 33, 843–850. [Google Scholar] [CrossRef]
- Ilham, M.; Fazil, A. Performance and kinetic study of the anaerobic co-digestion of cocoa husk and digested cow manure with high organic loading rate. INMATEH-Agric. Eng. 2018, 55, 131–140. [Google Scholar]
- Awolu, O.; Oyeyemi, S.O. Optimization of bioethanol production from cocoa (Theobroma cacao) bean shell. Int. J. Curr. Microbiol. App. Sci 2015, 4, 506–514. [Google Scholar]
- Papadopoulou, E.L.; Paul, U.C.; Tran, T.N.; Suarato, G.; Ceseracciu, L.; Marras, S.; d’Arcy, R.; Athanassiou, A. Sustainable Active Food Packaging from Poly (lactic acid) and Cocoa Bean Shells. Acs Appl. Mater. Interfaces 2019, 11, 31317–31327. [Google Scholar] [CrossRef]
- Puglia, D.; Dominici, F.; Badalotti, M.; Santulli, C.; Kenny, J. Tensile, thermal and morphological characterization of cocoa bean shells (CBS)/polycaprolactone-based composites. J. Renew. Mater. 2016, 4, 199–205. [Google Scholar] [CrossRef]
- Lik, H. Development of particleboard from cocoa shells. Malays. Cocoa J. 2006, 2, 22–27. [Google Scholar]
- Olabisi, A.I.; Adam, A.N.; Okechukwu, O.M. Development and assessment of composite brake pad using pulverized cocoa beans shells filler. Int. J. Mater. Sci. Appl. 2016, 5, 66–78. [Google Scholar]
- Olabisi, A.I. Development of asbestos-free automotive brake pad using ternary agro-waste fillers. J. Multidiscip. Eng. Sci. Technol. (JMEST) 2016, 3, 5307–5323. [Google Scholar]
- Olabisi, A.I.; Boye, T.E.; Eyere, E. Evaluation of Pure Aluminium Inoculated with Varying Grain Sizes of an Agro-waste based Inoculant. Adv. Sci. Technol. Eng. Syst. J. 2017, 2, 14–25. [Google Scholar] [CrossRef]
- Plaza-Recobert, M.; Trautwein, G.; Pérez-Cadenas, M.; Alcañiz-Monge, J. Preparation of binderless activated carbon monoliths from cocoa bean husk. Microporous Mesoporous Mater. 2017, 243, 28–38. [Google Scholar] [CrossRef]
- Pérez-Cadenas, M.; Plaza-Recobert, M.; Trautwein, G.; Alcañiz-Monge, J. Development of tailored mesoporosity in carbonised cocoa bean husk. Microporous Mesoporous Mater. 2018, 256, 128–139. [Google Scholar] [CrossRef]
- Ahmad, F.; Daud, W.M.A.W.; Ahmad, M.A.; Radzi, R. Cocoa (Theobroma cacao) shell-based activated carbon by CO2 activation in removing of Cationic dye from aqueous solution: Kinetics and equilibrium studies. Chem. Eng. Res. Des. 2012, 90, 1480–1490. [Google Scholar] [CrossRef]
- Takam, B.; Acayanka, E.; Kamgang, G.Y.; Pedekwang, M.T.; Laminsi, S. Enhancement of sorption capacity of cocoa shell biomass modified with non-thermal plasma for removal of both cationic and anionic dyes from aqueous solution. Environ. Sci. Pollut. Res. 2017, 24, 16958–16970. [Google Scholar] [CrossRef]
- Fioresi, F.; Vieillard, J.; Bargougui, R.; Bouazizi, N.; Fotsing, P.N.; Woumfo, E.D.; Brun, N.; Mofaddel, N.; Le Derf, F. Chemical modification of the cocoa shell surface using diazonium salts. J. Colloid Interface Sci. 2017, 494, 92–97. [Google Scholar] [CrossRef]
- Diaza, V.J.M.; Nakayoa, J.L.J.; Benites, E. Use of coffee grind with cocoa shell as the basis for a filter to reduce lead from contaminated water from a river, Peru. Environ. Sci. 2018, 2, 24–29. [Google Scholar]
- International Cocoa Organization. Products That can be Made from Cocoa. Available online: https://www.icco.org/faq/52-by-products/115-products-that-can-be-made-from-cocoa.html (accessed on 6 September 2019).
- Watson, R.R.; Preedy, V.R.; Zibadi, S. Chocolate in Health and Nutrition; Springer: Berlin, Germany, 2013. [Google Scholar]
- Rosmiza, M.; Davies, W.; CR, R.A.; Jabil, M.; Mazdi, M. Prospects for increasing commercial mushroom production in Malaysia: challenges and opportunities. Mediterr. J. Soc. Sci. 2016, 7, 406. [Google Scholar] [CrossRef]
- Prathibha, V.; Sharadraj, K.; Nidhina, K.; Hegde, V. Evaluation of locally available substrates for mass production of Trichoderma. J. plant. crops 2015, 43, 168–170. [Google Scholar]
- Silva, T.; Souza, L.; Reis, N.; Assis, S.; Ferreira, M.; Oliveira, J.; Aguiar-Oliveira, E.; Franco, M. Cultivation of Penicillium roqueforti in cocoa shell to produce and characterize its lipase extract. Rev. Mex. De Ing. Química 2017, 16, 745–756. [Google Scholar]
- Oliveira, P.; de Brito, A.; Pimentel, A.; Soares, G.; Pacheco, C.; Santana, N.; da Silva, E.; Fernandes, A.d.A.; Ferreira, M.; Oliveira, J. Cocoa shell for the production of endoglucanase by Penicillium roqueforti ATCC 10110 in solid state fermentation and biochemical properties. Rev. Mex. De Ing. Química 2019, 18, 777–787. [Google Scholar] [CrossRef]
- Tu, C. Study about Stability of Cacao Husk Pigment and Its Dyeing Properties on Cotton. In Key Engineering Materials; Trans Tech Publications Ltd.: Bäch, Switzarland, 2016; pp. 133–138. [Google Scholar]
- Fontes, C.; Silva, R.; Lima, P. Characterization and Effect of Using Bottom and Fly Ashes from Co-combustion of Cocoa Waste as Mineral Addition in Concrete. Waste Biomass Valorization 2019, 10, 223–233. [Google Scholar] [CrossRef]
- Zumbé, A. Polyphenols in cocoa: are there health benefits? Nutr. Bull. 1998, 23, 94–102. [Google Scholar] [CrossRef]
- Martín, M.A.; Ramos, S. Cocoa polyphenols in oxidative stress: Potential health implications. J. Funct. Foods 2016, 27, 570–588. [Google Scholar] [CrossRef]
- Martin, M.A.; Goya, L.; Ramos, S. Potential for preventive effects of cocoa and cocoa polyphenols in cancer. Food Chem. Toxicol. 2013, 56, 336–351. [Google Scholar] [CrossRef]
- Daglia, M. Polyphenols as antimicrobial agents. Curr. Opin. Biotechnol. 2012, 23, 174–181. [Google Scholar] [CrossRef]
- Sies, H.; Schewe, T.; Heiss, C.; Kelm, M. Cocoa polyphenols and inflammatory mediators. Am. J. Clin. Nutr. 2005, 81, 304S–312S. [Google Scholar] [CrossRef]
- Jalil, A.M.M.; Ismail, A. Polyphenols in cocoa and cocoa products: is there a link between antioxidant properties and health? Molecules 2008, 13, 2190–2219. [Google Scholar] [CrossRef]
- Weisburger, J.H. Chemopreventive effects of cocoa polyphenols on chronic diseases. Exp. Biol. Med. 2001, 226, 891–897. [Google Scholar] [CrossRef]
- Kim, K.H.; Lee, K.W.; Kim, D.Y.; Park, H.H.; Kwon, I.B.; Lee, H.J. Extraction and fractionation of glucosyltransferase inhibitors from cacao bean husk. Process Biochem. 2004, 39, 2043–2046. [Google Scholar] [CrossRef]
- Matsumoto, M.; Tsuji, M.; Okuda, J.; Sasaki, H.; Nakano, K.; Osawa, K.; Shimura, S.; Ooshima, T. Inhibitory effects of cacao bean husk extract on plaque formation in vitro and in vivo. Eur. J. Oral Sci. 2004, 112, 249–252. [Google Scholar] [CrossRef] [PubMed]
- Osawa, K.; Miyazaki, K.; Shimura, S.; Okuda, J.; Matsumoto, M.; Ooshima, T. Identification of cariostatic substances in the cacao bean husk: their anti-glucosyltransferase and antibacterial activities. J. Dent. Res. 2001, 80, 2000–2004. [Google Scholar] [CrossRef]
- Babu, N.V.; Vivek, D.; Ambika, G. Comparative evaluation of chlorhexidine mouthrinse versus cacao bean husk extract mouthrinse as antimicrobial agents in children. Eur. Arch. Paediatr. Dent. 2011, 12, 245–249. [Google Scholar] [CrossRef] [PubMed]
- Ooshima, T.; Osaka, Y.; Sasaki, H.; Osawa, K.; Yasuda, H.; Matsumura, M.; Sobue, S.; Matsumoto, M. Caries inhibitory activity of cacao bean husk extract in in-vitro and animal experiments. Arch. Oral Biol. 2000, 45, 639–645. [Google Scholar] [CrossRef]
- Badiyani, B.K.; Kumar, A.; Bhat, P.K.; Sarkar, S. Chocolate disinfectant: effectiveness of cocoa bean husk extract on Streptococcus mutans in used toothbrushes. Int. J. Oral Care Res. 2013, 1, 7–10. [Google Scholar]
- Sakagami, H.; Satoh, K.; Fukamachi, H.; Ikarashi, T.; Shimizu, A.; Yano, K.; Kanamoto, T.; Terakubo, S.; Nakashima, H.; Hasegawa, H. Anti-HIV and vitamin C-synergized radical scavenging activity of cacao husk lignin fractions. In Vivo 2008, 22, 327–332. [Google Scholar] [PubMed]
- Sakagami, H.; Matsuta, T. Biological Activity of Cacao Husk and Mass Lignin-Carbohydrate Complexes. In Chocolate in Health and Nutrition; Springer: Berlin, Germany, 2013; pp. 247–262. [Google Scholar]
- Unten, S.; Ushijima, H.; Shimizu, H.; Tsuchie, H.; Kitamura, T.; Moritome, N.; Sakagami, H. Effect of cacao husk extract on human immunodeficiency virus infection. Lett. Appl. Microbiol. 1991, 13, 251–254. [Google Scholar] [CrossRef]
- Lee, K.W.; Hwang, E.-S.; Kang, N.J.; Kim, K.H.; Lee, H.J. Extraction and chromatographic separation of anticarcinogenic fractions from cacao bean husk. Biofactors 2005, 23, 141–150. [Google Scholar] [CrossRef]
- Zainal, B.; Abdah, M.; Taufiq-Yap, Y.; Roslida, A.; Rosmin, K. Anticancer agents from non-edible parts of Theobroma cacao. Nat. Prod. Chem. Res. 2014, 2, 134–142. [Google Scholar]
- Zainal, B.; Abdah, M.; Taufiq Yap, Y.; Roslida, A.; Mohd Redzuan, S.; Kasran, R. Bioactivity-guided fractionation of potent anti-cancer properties from non-edible tissues of Theobroma cacao. Malasyan Cocoa J. 2016, 9, 170–181. [Google Scholar]
- Guil-Guerrero, J.; Ramos, L.; Moreno, C.; Zúñiga-Paredes, J.; Carlosama-Yepez, M.; Ruales, P. Antimicrobial activity of plant-food by-products: A review focusing on the tropics. Livest. Sci. 2016, 189, 32–49. [Google Scholar] [CrossRef]
- Kim, D.Y.; Park, H.J.; Park, H.H.; Kim, H.S.; Kwon, I.B. Manufacturing Process of Glucosyltransferase Inhibitors from Cacao Bean Husk. U.S. Patent No. 6,159,451, 12 December 2000. [Google Scholar]
- Kwon, I.B.; Park, H.H.; An, B.J. Chewing Gum Designed to Prevent Tooth Decay by Blending a Soluble Extract of Cacao Bean Husk. U.S. Patent No. 4,908,212, 13 March 1990. [Google Scholar]
- Kris-Etherton, P.M.; Keen, C.L. Evidence that the antioxidant flavonoids in tea and cocoa are beneficial for cardiovascular health. Curr. Opin. Lipidol. 2002, 13, 41–49. [Google Scholar] [CrossRef] [PubMed]
- Steinberg, F.M.; Bearden, M.M.; Keen, C.L. Cocoa and chocolate flavonoids: implications for cardiovascular health. J. Am. Diet. Assoc. 2003, 103, 215–223. [Google Scholar] [CrossRef] [PubMed]
- Yamagishi, M.; Natsume, M.; Osakabe, N.; Nakamura, H.; Furukawa, F.; Imazawa, T.; Nishikawa, A.; Hirose, M. Effects of cacao liquor proanthocyanidins on PhIP-induced mutagenesis in vitro, and in vivo mammary and pancreatic tumorigenesis in female Sprague–Dawley rats. Cancer Lett. 2002, 185, 123–130. [Google Scholar] [CrossRef]
- Carnésecchi, S.; Schneider, Y.; Lazarus, S.A.; Coehlo, D.; Gossé, F.; Raul, F. Flavanols and procyanidins of cocoa and chocolate inhibit growth and polyamine biosynthesis of human colonic cancer cells. Cancer Lett. 2002, 175, 147–155. [Google Scholar] [CrossRef]
- Lee, H.J.; Lee, K.W.; Kang, K.S.; Kim, D.; Park, H.H.; Lee, M.J.; Kim, H.S.; Kwon, I.B. Extracts of Cacao and Cacao Bean Husk with Inhibitory Effects on Carcinogenesis. U.S. Patent Application No. 10/202,586, 19 February 2004. [Google Scholar]
- ICCO, International Cocoa Organization. Integrated Management of Cocoa Pests and Pathogens in Africa: Controlling Indigenous Pests and Diseases and Preventing the Introduction of Exogenous Ones. In Proceedings of the Report of Project Inception Workshop, Accra, Ghana, 15–18 April 2013. [Google Scholar]
- Dankyi, E.; Carboo, D.; Gordon, C.; Fomsgaard, I.S. Application of the QuEChERS procedure and LC–MS/MS for the assessment of neonicotinoid insecticide residues in cocoa beans and shells. J. Food Compos. Anal. 2015, 44, 149–157. [Google Scholar] [CrossRef]
- Owusu-Boateng, G.; Owusu, S. Methods of cocoa harvesting to drying of bean in Ghana and polycyclic aromatic hydrocarbon concentration in the nib and shell of the cocoa bean. Acad. J. Agric. Res. 2015, 3, 176–183. [Google Scholar]
- Bertoldi, D.; Barbero, A.; Camin, F.; Caligiani, A.; Larcher, R. Multielemental fingerprinting and geographic traceability of Theobroma cacao beans and cocoa products. Food Control 2016, 65, 46–53. [Google Scholar] [CrossRef]
- Kruszewski, B.; Obiedziński, M.W.; Kowalska, J. Nickel, cadmium and lead levels in raw cocoa and processed chocolate mass materials from three different manufacturers. J. Food Compos. Anal. 2018, 66, 127–135. [Google Scholar] [CrossRef]
- Meunier, N.; Laroulandie, J.; Blais, J.; Tyagi, R. Cocoa shells for heavy metal removal from acidic solutions. Bioresour. Technol. 2003, 90, 255–263. [Google Scholar] [CrossRef]
- Meunier, N.; Blais, J.-F.; Tyagi, R.D. Removal of heavy metals from acid soil leachate using cocoa shells in a batch counter-current sorption process. Hydrometallurgy 2004, 73, 225–235. [Google Scholar] [CrossRef]
- Taylor, D.A. Lead in Cocoa Products: Where Does Contamination Come From; National Institute of Environmental Health Sciences: Triangle Park, NC, USA, 2005. [Google Scholar]
- Rankin, C.W.; Nriagu, J.O.; Aggarwal, J.K.; Arowolo, T.A.; Adebayo, K.; Flegal, A.R. Lead contamination in cocoa and cocoa products: isotopic evidence of global contamination. Environ. Health Perspect. 2005, 113, 1344–1348. [Google Scholar] [CrossRef] [PubMed]
- Assa, A.; Noor, A.; Yunus, M.; Djide, M. Heavy metal concentrations in cocoa beans (Theobroma cacao L.) originating from East Luwu, South Sulawesi, Indonesia. In Proceedings of Journal of Physics: Conference Series; IOP Publishing: Bristol, UK, 2018; p. 012011. [Google Scholar]
- Lewis, C.; Lennon, A.M.; Eudoxie, G.; Umaharan, P. Genetic variation in bioaccumulation and partitioning of cadmium in Theobroma cacao L. Sci. Total Environ. 2018, 640, 696–703. [Google Scholar] [CrossRef] [PubMed]
- FAO/WHO. Codex commitee on contaminants in foods. In Proposed draft maximum levels for cadmium in chocolate and cocoa-derived products (at step 4); Codex Alimentarius Commission: Utrecht, The Netherlands, 2018; Volume CF12/CRD08. [Google Scholar]
- Amézqueta, S.; Gonzalez-Penas, E.; Lizarraga, T.; Murillo-Arbizu, M.; De Cerain, A.L. A simple chemical method reduces ochratoxin A in contaminated cocoa shells. J. Food Prot. 2008, 71, 1422–1426. [Google Scholar] [CrossRef] [PubMed]
- Copetti, M.V.; Iamanaka, B.T.; Nester, M.A.; Efraim, P.; Taniwaki, M.H. Occurrence of ochratoxin A in cocoa by-products and determination of its reduction during chocolate manufacture. Food Chem. 2013, 136, 100–104. [Google Scholar] [CrossRef] [PubMed]
- Coulibaly, A.; Biego, G.H.M.; Dembele, A.; Bohoussou, K.M.; Toure, A. Cocoa beans and cocoa derivatives from Cote-D’Ivoire: investigating ochratoxin a level and assessing dietary intake adults. Sustain. Agric. Res. 2013, 2, 173–180. [Google Scholar] [CrossRef]
- Amezqueta, S.; Gonzalez-Penas, E.; Murillo, M.; Lopez de Cerain, A. Occurrence of ochratoxin A in cocoa beans: effect of shelling. Food Addit. Contam. 2005, 22, 590–596. [Google Scholar] [CrossRef]
- Serra Bonvehí, J. Occurrence of ochratoxin A in cocoa products and chocolate. J. Agric. Food Chem. 2004, 52, 6347–6352. [Google Scholar] [CrossRef]
- Copetti, M.V.; Iamanaka, B.T.; Pereira, J.L.; Lemes, D.P.; Nakano, F.; Taniwaki, M.H. Determination of aflatoxins in by-products of industrial processing of cocoa beans. Food Addit. Contam. Part A 2012, 29, 972–978. [Google Scholar] [CrossRef]
- Raters, M.; Matissek, R. Analysis and occurrence of deoxynivalenol (DON) in cocoa. Eur. Food Res. Technol. 2008, 226, 1107–1112. [Google Scholar] [CrossRef]
- Aroyeun, S.; Adegoke, G. Reduction of ochratoxin A (OTA) in spiked cocoa powder and beverage using aqueous extracts and essential oils of Aframomum danielli. Afr. J. Biotechnol. 2007, 6, 612. [Google Scholar]
- Manda, P.; Dano, D.S.; Kouadio, J.H.; Diakite, A.; Sangare-Tigori, B.; Ezoulin, M.J.M.; Soumahoro, A.; Dembele, A.; Fourny, G. Impact of industrial treatments on ochratoxin A content in artificially contaminated cocoa beans. Food Addit. Contam. 2009, 26, 1081–1088. [Google Scholar] [CrossRef] [PubMed]
- Kreibich, H.; Moecke, E.O.E.; Scussel, V. Stereo and scanning electron microscopy of cocoa beans (Theobroma cacao L.): fungi spoilage susceptibility. In Microscopy and Imaging Science: Practical Approaches to Applied Research and Education; Méndez-Vilas, A., Ed.; Formatex Research Center S.L.: Badajoz, Spain, 2017; pp. 329–336. [Google Scholar]
| Parameter | Amount a | References |
|---|---|---|
| Energy (kcal/100 g) | 122.00 | [15] |
| Moisture (%) | 3.60–13.13 | [13,26,27,30,31,32,33,34,35,36,37,38] |
| Ash (g/100 g) | 5.96–11.42 | [13,30,33,35,36,37,38,39,40,41] |
| Proteins (g/100 g) | 10.30–27.40 | [13,26,27,30,32,34,35,36,37,38,40,41,42,43,44] |
| Fats (g/100 g) | 1.50–8.49 | [13,30,32,33,35,37,38,40,41,42,43,44,45] |
| Carbohydrates (g/100 g) | 7.85–70.25 | [13,15,32,35,38,42] |
| - Starch (g/100 g) | 0–2.80 | [24,27,39,46,47] |
| - Soluble sugars (g/100 g) | 0.16–1.66 | [27,32,40] |
| Dietary fiber (g/100 g) | 39.25–66.33 | [13,17,30,33,35,38,40,43,48] |
| - Soluble fiber (g/100 g) | 7.03–16.91 | [13,17,33,35,36,38,40,41] |
| - Insoluble fiber (g/100 g) | 28.34–50.42 | [13,17,33,35,36,38,40,41] |
| Pectin (g/100 g) | 7.62–15.59 | [30,49,50,51] |
| Minerals | ||
| - Calcium (g/100 g) | 0.23–0.44 | [39,52] |
| - Phosphorus (g/100 g) | 0.58–1.00 | [39] |
| - Magnesium (g/100 g) | 0.48–1.29 | [39,52] |
| - Potassium (g/100 g) | 1.25–1.82 | [39,52] |
| - Sodium (mg/100 g) | 16.00–192.20 | [39,52] |
| - Iron (mg/100 g) | 27.60–80.50 | [39,52] |
| - Manganese (mg/100 g) | 4.53 | [52] |
| - Copper (mg/100 g) | 2.35–6.62 | [39,52] |
| - Selenium (mg/100 g) | 0.21 | [52] |
| - Cobalt (mg/100 g) | 0.10 | [52] |
| - Zinc (mg/100 g) | 2.75–19.00 | [39,52,53] |
| - Chromium (mg/100 g) | 0.67‒4.86 | [39,52] |
| Vitamins | ||
| - B1 (μg/g) | 0.70–3.10 | [39] |
| - B2 (μg/g) | 0.90–3.10 | [39] |
| - B6 (μg/g) | tr | [39] |
| - D (μg/g) | tr–0.53 | [39,54] |
| - E (μg total tocopherols/g CBS fat) | 1.02 | [36] |
| Polyphenol content | ||
| - Total phenolic content b | 3.12–94.95 | [13,40,41,55,56,57,58,59,60,61] |
| - Total flavonoid content c | 1.65–40.72 | [13,41,59] |
| - Total tannin content c | 1.70–25.30 | [13,39,41,59] |
| Flavanols | ||
| - Epicatechin (mg/g) | 0.21–34.97 | [59,62,63,64,65] |
| - Catechin (mg/g) | 0.18–4.50 | [62,63,64,65] |
| - Procyanidin B1 (mg/g) | 0.55–0.83 | [65] |
| - Procyanidin B2 (mg/g) | 0.23–1.38 | [64,65] |
| Methylxanthines | ||
| - Theobromine (g/100 g) | 0.39–1.83 | [30,37,39,47,57,59,62,64,66,67] |
| - Caffeine (g/100 g) | 0.04–0.42 | [39,47,57,59,63,64,66] |
| Volatile organic compounds (aromatics; μg/g) | 4.92–16.10 | [31,68] |
| Extract/Fraction (Dose) | Application | Mechanism of Action | References |
|---|---|---|---|
| Acetone, ethanol, methanol (80%), and water extracts of defatted CBS (100 mg dry CBS/mL) | Antibacterial | Weak antibacterial activity against E. coli, S. aureus, Salmonella, and B. cereus (acetone > ethanol = methanol > water) when compared to a positive control (cephadex) with the inhibition zone diameter technique. Minimum inhibitory concentrations (MIC) of 0.468–3.750 mg/mL when using microdilution technique. | [41] |
| CBS ethanol, methanol, and acetone (50%, v/v) dried extracts (0.1 g/mL) | Antibacterial (anti-cariogenic) | Inhibitory activity against glucosyltransferase (GTF) from S. mutans. | [177] |
| Cellulase-treated CBS extracted with 30% ethanol, re-dissolved with1% ethanol (1.0 mg dry extract/mL)—in vitro | Antibacterial (anti-cariogenic) | In vitro: Avoid plaque deposition by inhibiting the adherence of S. mutans to saliva-coated hydroxiapatite (inhibition of glucosyltransferase due to epicatechin polymers) and reducing S. mutans in plaque (antibacterial activity of unsaturated fatty acids on CBS). | [178] |
| CBS dissolved in 20% ethanol (2 0mg/mL), diluted with water to 1 mg/mL in 1% ethanol (mouth rinses)—in vivo | In vivo: CBS mouth rinse more efficient than just 1% ethanol mouth rinse. Inhibition of plaque deposition and S. mutans in saliva. | ||
| Cellulase-treated CBS extracted with 50% ethanol and fractionation of the dried extract by chromatography | Antibacterial (anti-cariogenic) | Antibacterial activity due to oleic and linoleic acids on CBS. | [179] |
| Inhibition of bacterial adhesion due to the glucosyltransferase inhibition by polymeric epicatechins with C-43 and C-8 intermolecular bonds estimated to be 4636 in molecular weight in an acetylated form. | |||
| 0.1% CBS extract mouth rinse prepared as in [178] | Antibacterial (anti-cariogenic) | In vivo reduction of S. mutans in saliva significantly similar to that obtained with 0.2% chlorexidine. | [180] |
| Cellulase-treated CBS extracted with 30% ethanol and resuspended on water (1 mg/mL) | Antibacterial (anti-cariogenic) | In vitro and in vivo reduction of oral Streptococci (S. mutans and S. sobrinus) growth rate and reduction of plaque deposition by decreasing sucrose-dependent adherence (inhibition of GTF). Minimum cariostatic concentration of 1.0 mg dry extract/mL. | [181] |
| Cellulase-treated CBS extracted with 50% ethanol and resuspended in water (1 mg/mL) | Antibacterial | CBS extract used as toothbrush disinfectant, reducing up to 32.25% of bacterial contamination by S. mutans. | [182] |
| Lignin fractions of CBS extracted with 1% NaOH and precipitated with acetic acid and ethanol | Antiviral(HIV and influenza) | Carbohydrate-rich fractions showed a high selectivity index against HIV (SI = 30–10000). Inhibition of cytopathic effects produced by the influenza virus against MDCK cells (Madin-Darby Canine Kidney cells). | [183,184] |
| Enhanced radical scavenging activity synergistically with vitamin C. | |||
| Anti-carcinogenic | 100–1000 μg/mL of the CBS lignin fraction stimulates the proliferation of human normal gingival fibroblasts (HGFs), but not that of the human oral squamous cell carcinoma cell line (HSC-2). | ||
| CBS extracted with 0.1 N NaOH, then lyophilized and fractionated according to the molecular weight | Antiviral (HIV) | Anti-HIV activity via inhibition of virus adsorption, and, therefore, inhibition of the cytopathic effect on MT-2 and MT-4 cells (highly sensible to HIV-1) when treated with 31.2–250 μg DW CBS/mL. Inhibition of syncytium formation between uninfected and HIV-infected MOLT-4 cells (lymphoblastoid T-cell line). | [185] |
| Ground CBS (60% of total dietary fiber) | Effects on cardiovascular health | Hypolipidemic and cholesterol reducing action in vivo: Reduction of total and low-density lipoprotein cholesterol and reduction of the lipid peroxidation in serum and liver. Mostly soluble fraction of the dietary fiber. | [78] |
| Fractionated (20%, 40%, 60%, and 80% ethanol) CBS freeze-dried extracts obtained with 50% methanol, ethanol, and acetone (0.1 g/mL) | Anti-carcinogenic | CBS polyphenolic fractions reduce the DNA synthesis of cancer cells and the inhibition of the gap-junction intracellular communication (GJIC). | [186] |
| Dried methanolic extract of CBS (5 g/200 mL) | Anti-carcinogenic | Anti-proliferative action against breast, liver, colon, lung, and cervical cancer cell lines. | [56,187,188] |
| Soluble dietary fiber (SDF), insoluble dietary fiber (IDF), and total dietary fiber (TDF) powders from CBS | Anti-carcinogenic | Binding capacity for bile acids (potential carcinogens) resulting in their detoxification. | [79] |
| Effects on cardiovascular health | Binding capacity for oil and cholesterol, decreasing their bioavailabilities. | ||
| Antidiabetic | Absorption of glucose retarding its diffusion and α-amylase inhibition. | ||
| Aqueous extracts of CBS | Antidiabetic | Inhibition of α-glucosidase enzyme close to that of acarbose 0.5 mM. | [13] |
| Desugared ground CBS | Antidiabetic | Sugar retention on the SDF fraction. | [40] |
| Supercritical CO2 extracts of CBS dissolved in acetone | Neuroprotective action | Protective action against ischemic oxidative damage in neuronal phenotype differentiated cells. | [90] |
| Dried ground CBS | Neuroprotective action and anti-inflammatory | Great content on phytoprostanes (474.3 ng/g DW CBS) phytofurans (278.0 ng/g DW CBS) with cytoprotective activity in immature brain cells and involved in anti-inflammatory processes. | [76] |
| Methanol and acetone CBS fractions of ethanolic extract (rich in epicatechin and tannins; 5 mg dried extract/mL) | Anti-inflammatory | Prevention of oxysterol mixture-induced IL-8 release (pro-inflammatory cytokine) on Caco-2 intestinal cell models and prevention of exaggerated toll-like receptor 2 and 4 (TRL2 and TRL4) responses, which may contribute to induce oxysterol-dependent intestinal inflammation. | [80] |
| Freeze-dried CBS aqueous extract (0.02 g/mL) | Action against obesity and inflammation-related disorders | Adipogenesis modulation and inhibition of adipokine production (responsible for inflammation processes and insulin resistance). | [102] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rojo-Poveda, O.; Barbosa-Pereira, L.; Zeppa, G.; Stévigny, C. Cocoa Bean Shell—A By-Product with Nutritional Properties and Biofunctional Potential. Nutrients 2020, 12, 1123. https://doi.org/10.3390/nu12041123
Rojo-Poveda O, Barbosa-Pereira L, Zeppa G, Stévigny C. Cocoa Bean Shell—A By-Product with Nutritional Properties and Biofunctional Potential. Nutrients. 2020; 12(4):1123. https://doi.org/10.3390/nu12041123
Chicago/Turabian StyleRojo-Poveda, Olga, Letricia Barbosa-Pereira, Giuseppe Zeppa, and Caroline Stévigny. 2020. "Cocoa Bean Shell—A By-Product with Nutritional Properties and Biofunctional Potential" Nutrients 12, no. 4: 1123. https://doi.org/10.3390/nu12041123
APA StyleRojo-Poveda, O., Barbosa-Pereira, L., Zeppa, G., & Stévigny, C. (2020). Cocoa Bean Shell—A By-Product with Nutritional Properties and Biofunctional Potential. Nutrients, 12(4), 1123. https://doi.org/10.3390/nu12041123