Strain-Specific Probiotic Properties of Bifidobacteria and Lactobacilli for the Prevention of Diarrhea Caused by Rotavirus in a Preclinical Model
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Experimental Design
2.3. Sample Collection and Clinical Indices
2.4. Fecal SA11 Shedding
2.5. Fecal pH Measurement
2.6. Specific Humoral Response
2.7. Statistical Analysis
3. Results
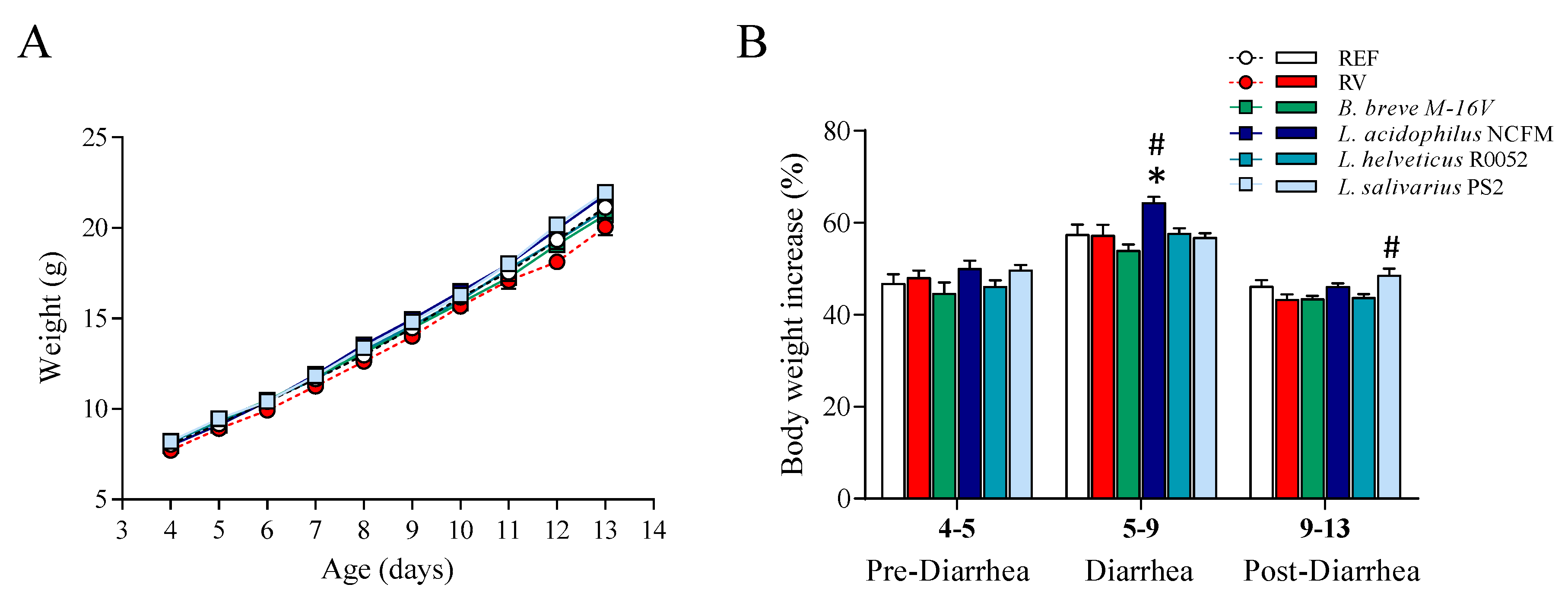
3.1. Growth and Morphometry
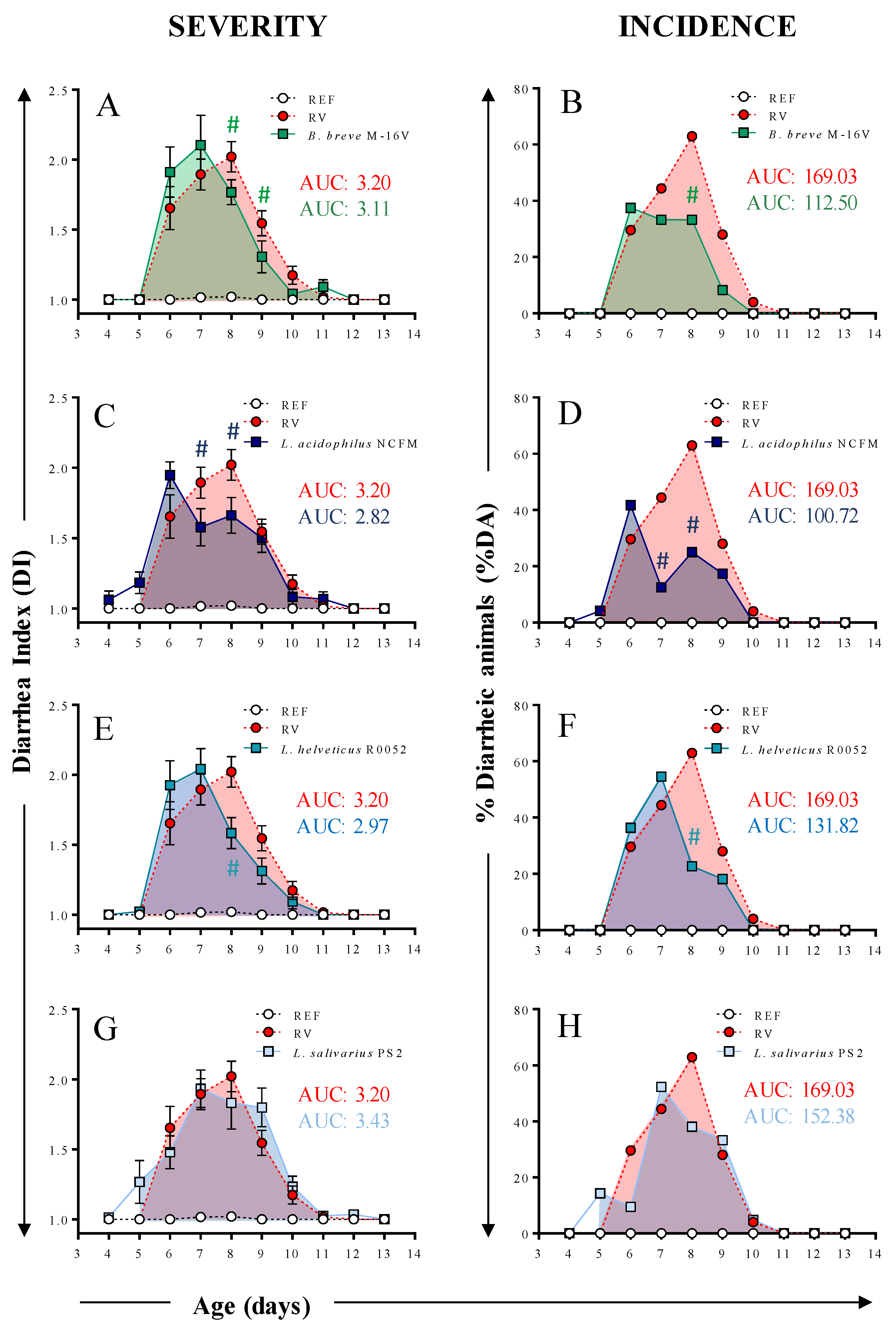
3.2. Clinical Evaluation of Diarrhea
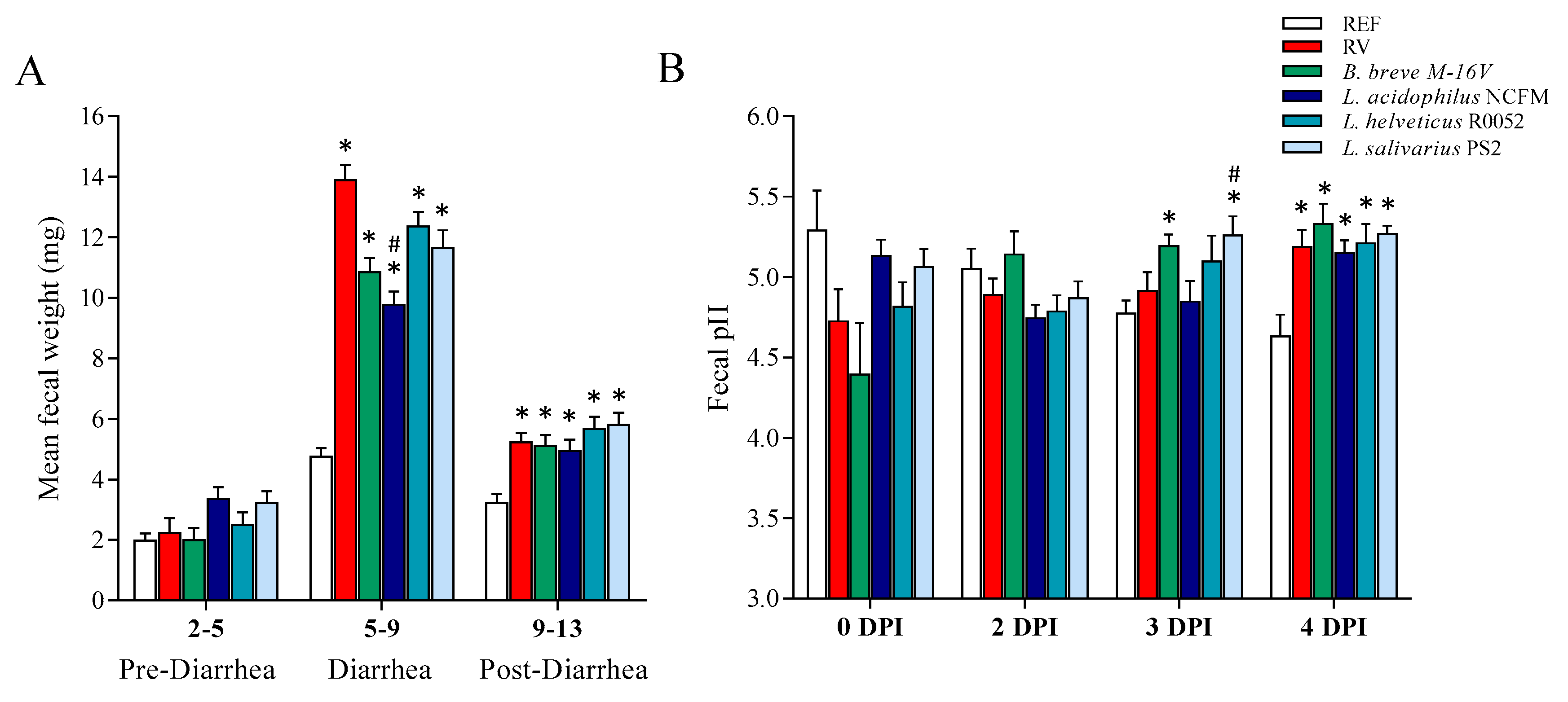
3.3. Fecal Weight and pH
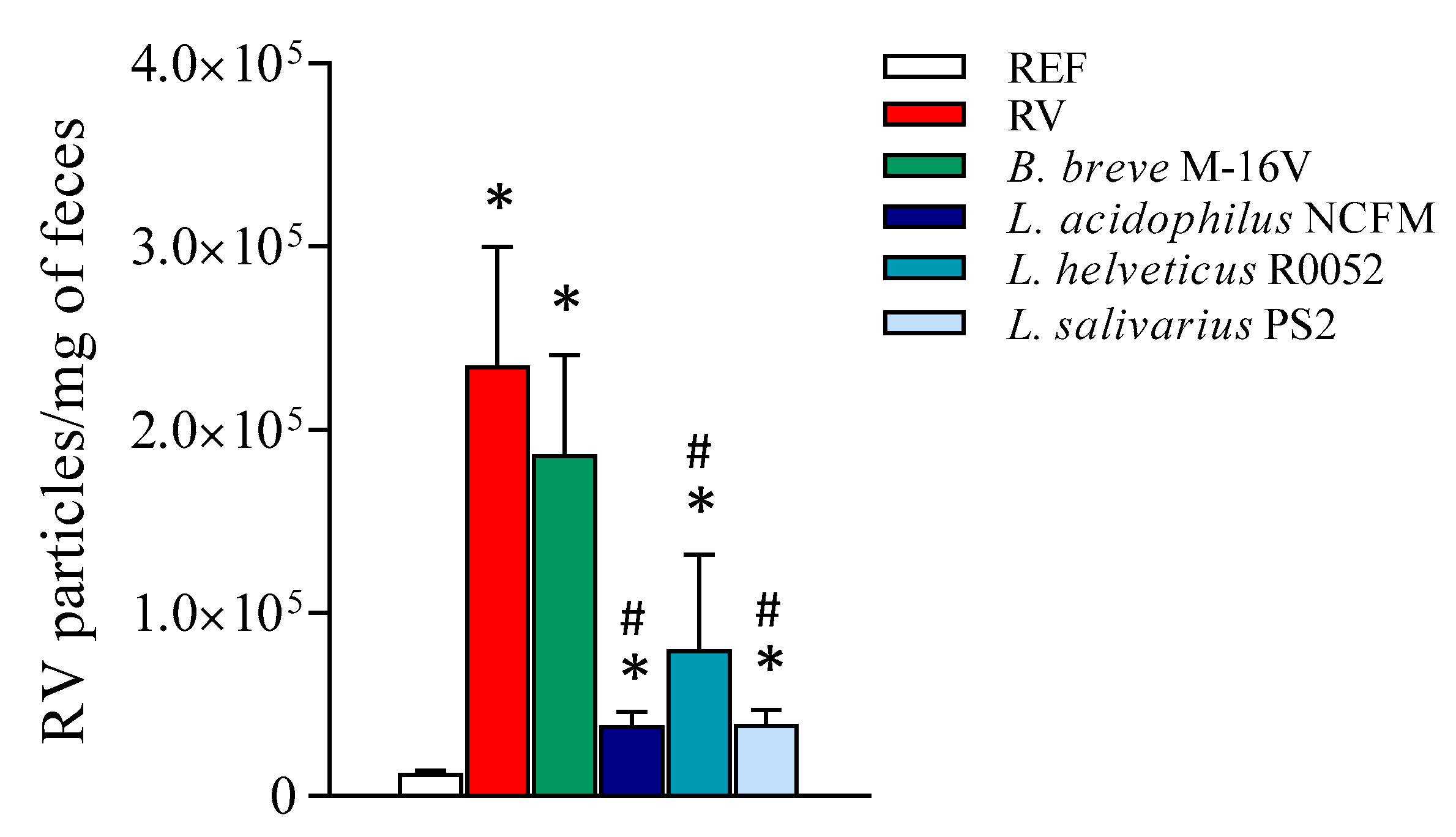
3.4. Viral Shedding
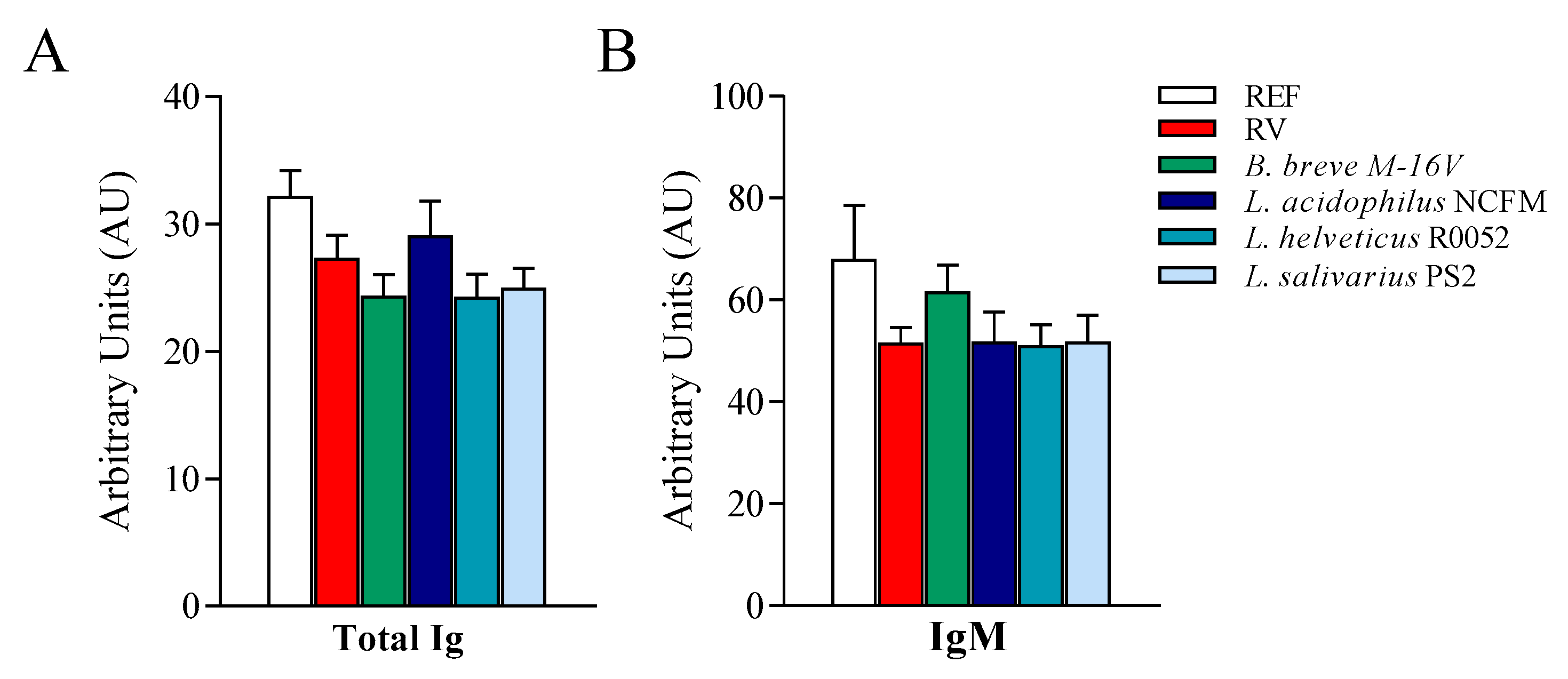
3.5. Anti-RV Humoral Response
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hill, C.; Guarner, F.; Reid, G.; Gibson, G.R.; Merenstein, D.J.; Pot, B.; Morelli, L.; Canani, R.B.; Flint, H.J.; Salminen, S.; et al. The International Scientific Association for Probiotcs and Prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat. Rev. Gastroenterol. Hepatol. 2014, 11, 506–514. [Google Scholar] [CrossRef]
- Salas-Cárdenas, S.P.; Olaya-Galán, N.N.; Fernández, K.; Velez, F.; Guerrero, C.A.; Gutiérrez, M.F. Decreased rotavirus infection of MA104 cells via probiotic extract binding to Hsc70 and ß3 integrin receptors. Univ. Sci. 2018, 23, 219–239. [Google Scholar] [CrossRef]
- Verna, E.C.; Lucak, S. Use of probiotics in gastrointestinal disorders: What to recommend? Therap. Adv. Gastroenterol. 2010, 3, 307–319. [Google Scholar] [CrossRef]
- Britton, R.A.; Versalovic, J. Probiotics and gastrointestinal infections. Interdiscip. Perspect. Infect. Dis. 2008, 2008, 290769. [Google Scholar] [CrossRef] [PubMed]
- Pant, N.; Marcotte, H.; Brüssow, H.; Svensson, L.; Hammarström, L. Effective prophylaxis against rotavirus diarrhea using a combination of Lactobacillus rhamnosus GG and antibodies. BMC Microbiol. 2007, 7, 86. [Google Scholar] [CrossRef] [PubMed]
- Isolauri, E.; Kaila, M.; Mykkänen, H.; Ling, W.H.; Salminen, S. Oral bacteriotherapy for viral gastroenteritis. Dig. Dis. Sci. 1994, 39, 2595–2600. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Ochoa, G.; Flores-Mendoza, L.K.; Icedo-Garcia, R.; Gomez-Flores, R.; Tamez-Guerra, P. Modulation of rotavirus severe gastroenteritis by the combination of probiotics and prebiotics. Arch. Microbiol. 2017, 199, 953–961. [Google Scholar] [CrossRef]
- Tran, A.; Talmud, D.; Lejeune, B.; Jovenin, N.; Renois, F.; Payan, C.; Leveque, N.; Andreoletti, L. Prevalence of rotavirus, adenovirus, norovirus, and astrovirus infections and coinfections among hospitalized children. J. Clin. Microbiol. 2010, 48, 1943–1946. [Google Scholar] [CrossRef]
- GBD. 2015 Mortality and Causes of Death Collaborators Global, regional, and national life expectancy, all-cause mortality, and cause-specific mortality for 249 causes of death, 1980−2015: A systematic analysis for the Global Burden of Disease Study 2015. Lancet 2016, 388, 1459–1544. [Google Scholar] [CrossRef]
- Bishop, R. Discovery of rotavirus: Implications for child health. J. Gastroenterol. Hepatol. 2009, 24, S81–S85. [Google Scholar] [CrossRef]
- Crawford, S.E.; Ramani, S.; Tate, J.E.; Parashar, U.D.; Svensson, L.; Hagbom, M.; Franco, M.A.; Greenberg, H.B.; O’Ryan, M.; Kang, G.; et al. Rotavirus infection. Nat. Rev. Dis. Prim. 2018, 3, 17083. [Google Scholar] [CrossRef] [PubMed]
- Latipov, R.; Utegenova, E.; Kuatbayeva, A.; Kasymbekova, K.; Abdykarimov, S.; Juraev, R.; Ismailov, U.; Flem, E. Epidemiology and burden of rotavirus disease in Central Asia. Int. J. Infect. Dis. 2011, 15, e464–e469. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Rigo-Adrover, M.; Saldaña-Ruíz, S.; van Limpt, K.; Knipping, K.; Garssen, J.; Knol, J.; Franch, A.; Castell, M.; Pérez-Cano, F.J. A combination of scGOS/lcFOS with Bifidobacterium breve M-16V protects suckling rats from rotavirus gastroenteritis. Eur. J. Nutr. 2017, 56, 1657–1670. [Google Scholar] [CrossRef] [PubMed]
- Kandasamy, S.; Vlasova, A.N.; Fischer, D.; Kumar, A.; Chattha, K.S.; Rauf, A.; Shao, L.; Langel, S.N.; Rajashekara, G.; Saif, L.J. Differential effects of Escherichia coli Nissle and Lactobacillus rhamnosus strain GG on human rotavirus binding, infection, and B cell immunity. J. Immunol. 2016, 196, 1780–1789. [Google Scholar] [CrossRef]
- Telmesani, A.M.A. Oral rehydration salts, zinc supplement and rotavirus vaccine in the management of childhood acute diarrhea. J. Fam. Community Med. 2010, 17, 79–82. [Google Scholar] [CrossRef]
- Grandy, G.; Medina, M.; Soria, R.; Terán, C.G.; Araya, M. Probiotics in the treatment of acute rotavirus diarrhoea. A randomized, double-blind, controlled trial using two different probiotic preparations in Bolivian children. BMC Infect. Dis. 2010, 10, 253. [Google Scholar] [CrossRef]
- Sindhu, K.N.C.; Sowmyanarayanan, T.V.; Paul, A.; Babji, S.; Ajjampur, S.S.R.; Priyadarshini, S.; Sarkar, R.; Balasubramanian, K.A.; Wanke, C.A.; Ward, H.D.; et al. Immune response and intestinal permeability in children with acute gastroenteritis treated with Lactobacillus rhamnosus GG: A randomized, double-blind, placebo-controlled trial. Clin. Infect. Dis. 2014, 58, 1107–1115. [Google Scholar] [CrossRef]
- Gänzle, M.G.; Höltzel, A.; Walter, J.; Jung, G.; Hammes, W.P. Characterization of reutericyclin produced by Lactobacillus reuteri LTH2584. Appl. Environ. Microbiol. 2000, 66, 4325–4333. [Google Scholar] [CrossRef]
- Reid, G. Probiotics in the treatment of diarrheal diseases. Curr. Infect. Dis. Rep. 2000, 2, 78–83. [Google Scholar] [CrossRef]
- Teran, C.G.; Teran-escalera, C.N.; Villarroel, P. Nitazoxanide vs. probiotics for the treatment of acute rotavirus diarrhea in children: A randomized, single-blind, controlled trial in Bolivian children. Int. J. Infect. Dis. 2009, 13, 518–523. [Google Scholar] [CrossRef]
- Isolauri, E. The role of probiotics in paediatrics. Curr. Paediatr. 2004, 14, 104–109. [Google Scholar] [CrossRef]
- Kaila, M.; Isolauri, E.; Saxelin, M.; Arvilommi, H.; Vesikari, T. Viable versus inactivated Lactobacillus strain GG in acute rotavirus diarrhoea. Arch. Dis. Child. 1995, 72, 51–53. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Li, G.; Wen, K.; Bui, T.; Cao, D.; Zhang, Y.; Yuan, L. Porcine small intestinal epithelial cell line (IPEC-J2) of rotavirus infection as a new model for the study of innate immune responses to rotaviruses and probiotics. Viral Immunol. 2010, 23, 135–149. [Google Scholar] [CrossRef] [PubMed]
- Macpherson, C.; Audy, J.; Mathieu, O.; Tompkins, T.A. Multistrain probiotic modulation of intestinal epithelial cells’ immune response to a double-stranded RNA ligand, poly (IC). Appl. Environ. Microbiol. 2014, 80, 1692–1700. [Google Scholar] [CrossRef]
- Varyukhina, S.; Freitas, M.; Bardin, S.; Robillard, E.; Tavan, E.; Sapin, C.; Grill, J.-P.; Trugnan, G. Glycan-modifying bacteria-derived soluble factors from Bacteroides thetaiotaomicron and Lactobacillus casei inhibit rotavirus infection in human intestinal cells. Microbes Infect. 2012, 14, 273–278. [Google Scholar] [CrossRef]
- Maragkoudakis, P.A.; Chingwaru, W.; Gradisnik, L.; Tsakalidou, E.; Cencic, A. Lactic acid bacteria efficiently protect human and animal intestinal epithelial and immune cells from enteric virus infection. Int. J. Food Microbiol. 2010, 141, S91–S97. [Google Scholar] [CrossRef]
- Moreno Muñoz, J.A.; Chenoll, E.; Casinos, B.; Bataller, E.; Ramón, D.; Genovés, S.; Montava, R.; Ribes, J.M.; Buesa, J.; Fàbrega, J.; et al. Novel probiotic Bifidobacterium longum subsp. infantis CECT 7210 strain active against rotavirus infections. Appl. Environ. Microbiol. 2011, 77, 8775–8783. [Google Scholar]
- Buccigrossi, V.; Laudiero, G.; Russo, C.; Miele, E.; Sofia, M.; Monini, M.; Ruggeri, F.M.; Guarino, A. Chloride secretion induced by rotavirus is oxidative stress-dependent and inhibited by Saccharomyces boulardii in human enterocytes. PLoS ONE 2014, 9, e99830. [Google Scholar] [CrossRef]
- Qiao, H.; Duffy, L.C.; Griffiths, E.; Dryja, D.; Leavens, A.; Rossman, J.; Rich, G.; Riepenhoff-Talty, M.; Locniskar, M. Immune responses in rhesus rotavirus-challenged Balb/c mice treated with bifidobacteria and prebiotic supplements. Pediatr. Res. 2002, 51, 750–755. [Google Scholar] [CrossRef]
- Del Rigo-Adrover, M.M.; Knipping, K.; Garssen, J.; van Limpt, K.; Knol, J.; Franch, À.; Castell, M.; Rodríguez-Lagunas, M.J.; Pérez-Cano, F.J. Prevention of rotavirus diarrhea in suckling rats by a specific fermented milk concentrate with prebiotic mixture. Nutrients 2019, 11, 189. [Google Scholar] [CrossRef]
- Thompson, A.; Van Moorlehem, E.; Aich, P. Probiotic-induced priming of innate immunity to protect against rotaviral infection. Probiotics Antimicro Prot. 2010, 2, 90–97. [Google Scholar] [CrossRef] [PubMed]
- Gaon, D.; Garcia, H.; Winter, L.; Rodriguez, N.; Quintas, R.; Gonzalez, S.N.; Oliver, G. Effect of Lactobacillus strains and Saccharomyces boulardii on persistent diarrhea in children. Medicina 2003, 63, 293–298. [Google Scholar] [PubMed]
- Guérin-Danan, C.; Meslin, J.; Chambard, A.; Charpilienne, A.; Relano, P.; Bouley, C.; Cohen, J.; Andrieux, C. Nutrient interactions and toxicity food supplementation with milk fermented by Lactobacillus casei DN-114 001 protects suckling rats from rotavirus-associated diarrhea. Nutr. Interact. Toxic. 2001, 131, 111–117. [Google Scholar]
- Olaya Galán, N.N.; Ulloa Rubiano, J.C.; Velez Reyes, F.A.; Fernandez Duarte, K.P.; Salas Cárdenas, S.P.; Gutierrez Fernandez, M.F. In vitro antiviral activity of Lactobacillus casei and Bifidobacterium adolescentis against rotavirus infection monitored by NSP4 protein production. J. Appl. Microbiol. 2016, 120, 1041–1051. [Google Scholar] [CrossRef]
- Chattha, K.S.; Vlasova, A.N.; Kandasamy, S.; Rajashekara, G.; Saif, L.J. Divergent immunomodulating effects of probiotics on T cell responses to oral attenuated human rotavirus vaccine and virulent human rotavirus infection in a neonatal gnotobiotic piglet disease model. J. Immunol. 2013, 191, 2446–2456. [Google Scholar] [CrossRef]
- Del Rigo-Adrover, M.M.; van Limpt, K.; Knipping, K.; Garssen, J.; Knol, J.; Costabile, A.; Franch, À.; Castell, M.; Pérez-cano, F.J. Preventive effect of a synbiotic combination of galacto- and fructooligosaccharides Mixture With Bifidobacterium breve M-16V in a model of multiple rotavirus infections. Front. Immunol. 2018, 9, 1318. [Google Scholar] [CrossRef]
- Lee, D.K.; Park, J.E.; Kim, M.J.; Seo, J.G.; Lee, J.H.; Ha, N.J. Probiotic bacteria, B. longum and L. acidophilus inhibit infection by rotavirus in vitro and decrease the duration of diarrhea in pediatric patients. Clin. Res. Hepatol. Gastroenterol. 2015, 39, 237–244. [Google Scholar] [CrossRef]
- Shornikova, A.-V.; Casas, I.A.; Mykkänen, H.; Salo, E.; Vesikari, T. Bacterotherapy with Lactobacillus reuteri in rotavirus gastroenteritis. Pediatr. Infect. Dis. J. 1997, 16, 1103–1107. [Google Scholar] [CrossRef]
- Wanke, M.; Szajewska, H. Probiotics for preventing healthcare-associated diarrhea in children: A meta-analysis of randomized controlled trials. Pediatr. Pol. 2014, 89, 8–16. [Google Scholar] [CrossRef]
- Aggarwal, S.; Upadhyay, A.; Shah, D.; Teotia, N.; Agarwal, A.; Jaiswal, V. Lactobacillus GG for treatment of acute childhood diarrhoea: An open labelled, randomized controlled trial. Indian J. Med. Res. 2014, 379–385. [Google Scholar]
- Guandalini, S.; Pensabene, L.; Zikri, M.A.; Dias, J.A.; Casali, L.G.; Hoekstra, H.; Kolacek, S.; Massar, K.; Micetic–Turk, D.; Papadopoulou, A.; et al. Lactobacillus GG administered in oral rehydration solution to children with acute diarrhea: A multicenter european trial. J. Pediatr. Gastroenterol. Nutr. 2000, 30, 54–60. [Google Scholar] [CrossRef] [PubMed]
- Guarino, A.; CAnani, R.B.; Spagnuolo, M.I.; Albano, F.; Di Benedetto, L. Oral bacterial therapy reduces the duration of symptoms and of viral excretion in children with mild diarrhea. J. Pediatr. Gastroenterol. Nutr. 1997, 25, 516–519. [Google Scholar] [CrossRef] [PubMed]
- Isolauri, E.; Juntunen, M.; Rautanen, T.; Sillanaukee, P.; Koivula, T. A human Lactobacillus strain (Lactobacillus casei sp strain GG) promotes recovery from acute diarrhea in children. Pediatrics 1991, 88, 90–97. [Google Scholar] [PubMed]
- Chandra, R.K. Effect of Lactobacillus on the incidence and severity of acute rotavirus diarrhoea in infants. A prospective placebo-controlled double-blind study. Nutr. Res. 2002, 22, 65–69. [Google Scholar] [CrossRef]
- Dutta, P.; Mitra, U.; Dutta, S.; Rajendran, K.; Saha, T.K.; Chatterjee, M.K. Randomised controlled clinical trial of Lactobacillus sporogenes (Bacillus coagulans), used as probiotic in clinical practice, on acute watery diarrhoea in children. Trop. Med. Int. Health 2011, 16, 555–561. [Google Scholar] [CrossRef] [PubMed]
- Mao, M.; Yu, T.; Xiong, Y.; Wang, Z.; Liu, H.; Gotteland, M.; Brunser, O. Effect of a lactose-free milk formula supplemented with bifidobacteria and streptococci on the recovery from acute diarrhoea. Asia Pac. J. Clin. Nutr. 2008, 17, 30–34. [Google Scholar]
- Phuapradit, P.; Vathanophas, K.; Podhipak, A.; Nopchinda, S.; Haschke, F.; Varavithya, W.; Sangchai, R.; Suthutvoravut, U.; Chantraruksa, V. Reduction of rotavirus infection in children receiving bifidobacteria-supplemented formula. J. Med. Assoc. Thai. 1999, 82, S43–S48. [Google Scholar]
- Erdogan, Ö.; Tanyeri, B.; Torun, E.; Gönüllü, E.; Arslan, H.; Erenberk, U.; Öktem, F. The comparition of the efficacy of two different probiotics in rotavirus gastroenteritis in children. J. Trop. Med. 2012, 2012, 787240. [Google Scholar] [CrossRef]
- Reeves, P.G.; Nielsen, F.H.; Fahey, G.C. AIN-93 purified diets for laboratory rodents: Final report of the American Institute of Nutrition ad hoc Writing Committee on the reformulation of the AIN-76A rodent diet. J. Nutr. 1993, 123, 1939–1951. [Google Scholar] [CrossRef]
- Wong, C.B.; Iwabuchi, N.; Xiao, J. Exploring the science behind Bifidobacterium breve M-16V in infant health. Nutrients 2019, 11, 1724. [Google Scholar] [CrossRef]
- Gilliland, S.E.; Speck, M.L.; Morgan, C.G. Detection of Lactobacillus acidophilus in feces of humans, pigs, and chickens. Appl. Microbiol. 1975, 30, 541–545. [Google Scholar] [CrossRef] [PubMed]
- Tompkins, T.A.; Barreau, G.; Broadbent, J.R. Complete genome sequence of Lactobacillus helveticus R0052, a commercial probiotic strain. J. Bacteriol. 2012, 194, 6349. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Espinosa-Martos, I.; Jiménez, E.; de Andrés, J.; Rodríguez-Alcalá, L.M.; Tavárez, S.; Manzano, S.; Fernández, L.; Alonso, E.; Fontecha, J.; Rodríguez, J.M. Milk and blood biomarkers associated to the clinical efficacy of a probiotic for the treatment of infectious mastitis. Benef. Microbes 2016, 7, 305–318. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Cano, F.J.; Castell, M.; Castellote, C.; Franch, À. Characterization of clinical and immune response in a rotavirus diarrhea model in suckling Lewis rats. Pediatr. Res. 2007, 62, 658–663. [Google Scholar] [CrossRef] [PubMed]
- Rigo-Adrover, M.; Pérez-Berezo, T.; Ramos-Romero, S.; van Limpt, K.; Knipping, K.; Garssen, J.; Knol, J.; Franch, À.; Castell, M.; Pérez-Cano, F.J. A fermented milk concentrate and a combination of short-chain galacto-oligosaccharides/long-chain fructo-oligosaccharides/pectin-derived acidic oligosaccharides protect suckling rats from rotavirus gastroenteritis. Br. J. Nutr. 2017, 117, 209–217. [Google Scholar] [CrossRef] [PubMed]
- Azagra-Boronat, I.; Massot-Cladera, M.; Knipping, K.; van’t Land, B.; Stahl, B.; Garssen, J.; Rodríguez-Lagunas, M.J.; Franch, À.; Castell, M.; Pérez-Cano, F.J. Supplementation with 2’-FL and scGOS/lcFOS ameliorates rotavirus-induced diarrhea in suckling rats. Front. Cell Infect. Microbiol. 2018, 8, 372. [Google Scholar] [CrossRef] [PubMed]
- Abrahamse-Berkeveld, M.; Alles, M.; Franke-Beckmann, E.; Helm, K.; Knecht, R.; Köllges, R.; Sandner, B.; Knol, J.; Ben Amor, K.; Bufe, A. Infant formula containing galacto-and fructo-oligosaccharides and Bifidobacterium breve M-16V supports adequate growth and tolerance in healthy infants in a randomised, controlled, double-blind, prospective, multicentre study. J. Nutr. Sci. 2016, 5, e42. [Google Scholar] [CrossRef]
- Sanders, M.E.; Klaenhammer, T.R. Invited review: The scientific basis of Lactobacillus acidophilus NCFM functionality as a probiotic. J. Dairy Sci. 2001, 84, 319–331. [Google Scholar] [CrossRef]
- Lee, H.; Hu, J.; Hou, S.; Liu, H.; Fang, H.; Free, R.; Hospital, M.M. Dose-dependent effect of Lactobacillus rhamnosus on quantitative reduction of faecal rotavirus shedding in children. J. Trop. Pediatr. 2009, 55, 297–301. [Google Scholar]
- Basu, S.; Chatterjee, M.; Ganguly, S.; Chandra, P.K. Efficacy of Lactobacillus rhamnosus GG in acute watery diarrhoea of Indian children: A randomised controlled trial. J. Paediatr. Child Health 2007, 43, 837–842. [Google Scholar] [CrossRef]
- Wolf, J.L.; Cukor, G.; Blacklow, N.R.; Dambrauskas, R.; Trier, J.S. Susceptibility of mice to rotavirus infection: Effects of age and administration of corticosteroids. Infect. Immun. 1981, 33, 565–574. [Google Scholar] [CrossRef] [PubMed]
- Ciarlet, M.; Conner, M.E.; Finegold, M.J.; Estes, M.K. Group A rotavirus infection and age-dependent diarrheal disease in rats: A new animal model to study the pathophysiology of rotavirus infection. J. Virol. 2002, 76, 41–57. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Wen, K.; Li, G.; Yang, X.; Kocher, J.; Bui, T.; Jones, D.; Pelzer, K.; Clark-Deener, S.; Yuan, L. Dual functions of Lactobacillus acidophilus NCFM at the intermidiate dose in protection against rotavirus diarrhea in gnotobiotic pigs vaccinated with a human rotavirus vaccine. J. Pediatr. Gastroenterol. Nutr. 2014, 58, 171–178. [Google Scholar] [CrossRef] [PubMed]
- Fernández, L.; Cárdenas, N.; Arroyo, R.; Manzano, S.; Jiménez, E.; Martín, V.; Rodríguez, J.M. Prevention of infectious mastitis by oral administration of Lactobacillus salivarius PS2 during late pregnancy. Clin. Infect. Dis. 2016, 62, 568–573. [Google Scholar] [CrossRef] [PubMed]
- Sack, D.A.; Rhoads, M.; Molla, A.; Molla, A.M.; Wahed, M.A. Carbohydrate malabsorption in infants with rotavirus diarrhea. AM J Clin Nutr 1982, 36, 1112–1118. [Google Scholar] [CrossRef] [PubMed]
- Gómez-gómez, M.; Danglot-banck, C.; Vega-franco, L. Intolerancia transitoria a lactosa: Criterios y procedimientos de diagnóstico. Rev. Mex. Pediatría 2007, 74, 24–31. [Google Scholar]
- Rigo-Adrover, M.D.M.; Knipping, K.; Garssen, J.; Saldaña-Ruíz, S.; Franch, À.; Castell, M.; Pérez-Cano, F.J. Rotavirus double infection model to study preventive dietary interventions. Nutrients 2019, 11, 131. [Google Scholar] [CrossRef]
- Saavedra, J.M.; Bauman, N.A.; Perman, J.A.; Yolken, R.H.; Saavedra, J.M.; Bauman, N.A.; Oung, I. Feeding of Bifidobacterium bifidum and Streptococcus thermophilus to infants in hospital for prevention of diarrhoea and shedding of rotavirus. Lancet 1994, 344, 1046–1049. [Google Scholar] [CrossRef]
- Duffy, L.C.; Zielezny, M.A.; Riepenhoff-Talty, M.; Dryja, D.; Sayahtaheri-Altaie, S.; Griffiths, E.; Ruffin, D.; Barrett, H.; Rossman, J.; Ogra, P.L. Effectiveness of Bifidobacterium bifidum in mediating the clinical course of murine rotavirus diarrhea. Pediatr. Res. 1994, 35, 690–695. [Google Scholar] [CrossRef]
- Morozov, V.; Hansman, G.; Hanisch, F.G.; Schroten, H.; Kunz, C. Human milk oligosaccharides as promising antivirals. Mol. Nutr. Food Res. 2018, 62, 1700679. [Google Scholar] [CrossRef]
- Bode, L. Human milk oligosaccharides: Every baby needs a sugar mama. Glycobiology 2012, 22, 1147–1162. [Google Scholar] [CrossRef] [PubMed]
- Guarner, F.; Khan, A.G.; Garisch, J.; Eliakim, R.; Gangl, A.; Thomson, A.; Krabshuis, J.; Le Mair, T.; Kaufmann, P.; de Paula, J.A.; et al. World Gastroenterology Organisation practice guideline: Probiotics and prebiotics. Arab J. Gastroenterol. 2009, 10, 33–42. [Google Scholar]
- Fijan, S. Microorganisms with claimed probiotic properties: An overview of recent literature. Int. J. Environ. Res. Public Health 2014, 11, 4745–4767. [Google Scholar] [CrossRef] [PubMed]
| REF | RV | B. breve M-16V | L. acidophilus NCFM | L. helveticus R0052 | L. salivarius PS2 | |
|---|---|---|---|---|---|---|
| Morphometrics | ||||||
| BMI (g/cm2) | 0.31 ± 0.02 | 0.29 ± 0.01 * | 0.30 ± 0.01 | 0.30 ± 0.00 | 0.30 ± 0.00 | 0.32 ± 0.01 # |
| Body/tail length ratio | 2.12 ± 0.02 | 2.11 ± 0.04 | 2.05 ± 0.03 | 2.09 ± 0.02 | 2.03 ± 0.02 | 2.07 ± 0.03 |
| Lee Index (g0.33/cm × 103) | 330.86 ± 2.90 | 323.97 ± 2.92 | 325.50 ± 2.51 | 326.77 ± 2.03 | 329.64 ± 2.44 | 335.44 ± 3.79 # |
| RV | B. breve M-16V | L. acidophilus NCFM | L. helveticus R0052 | L. salivarius PS2 | |
|---|---|---|---|---|---|
| Severity | |||||
| MS | 2.20 ± 0.07 | 2.14 ± 0.13 | 2.06 ± 0.10 | 2.13 ± 0.12 | 2.23 ± 0.14 |
| MSd | 7.40 ± 0.18 | 6.87 ± 0.21 # | 6.82 ± 0.21 # | 6.72 ± 0.23 # | 7.14 ± 0.26 |
| Incidence | |||||
| MI | 62.96 | 37.50 # | 41.66 # | 54.54 | 52.38 |
| MId | 8 | 6 | 6 | 7 | 7 |
| Duration | |||||
| DDB | 7.00 ± 0.18 | 6.84 ± 0.21 | 6.58 ± 0.23 | 6.62 ± 0.18 | 6.87 ± 0.31 |
| DDE | 8.04 ± 0.19 | 7.29 ± 0.23 | 7.27 ± 0.30 | 7.63 ± 0.24 | 8.06 ± 0.32 |
| DP | 0.93 ± 0.22 | 0.36 ± 0.12 # | 0.36 ± 0.17 # | 0.68 ± 0.23 # | 0.91 ± 0.28 |
| DwD | 1.67 ± 0.21 | 1.14 ± 0.17 # | 0.86 ± 0.15 # | 1.35 ± 0.26 | 1.52 ± 0.29 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Azagra-Boronat, I.; Massot-Cladera, M.; Knipping, K.; Garssen, J.; Ben Amor, K.; Knol, J.; Franch, À.; Castell, M.; Rodríguez-Lagunas, M.J.; Pérez-Cano, F.J. Strain-Specific Probiotic Properties of Bifidobacteria and Lactobacilli for the Prevention of Diarrhea Caused by Rotavirus in a Preclinical Model. Nutrients 2020, 12, 498. https://doi.org/10.3390/nu12020498
Azagra-Boronat I, Massot-Cladera M, Knipping K, Garssen J, Ben Amor K, Knol J, Franch À, Castell M, Rodríguez-Lagunas MJ, Pérez-Cano FJ. Strain-Specific Probiotic Properties of Bifidobacteria and Lactobacilli for the Prevention of Diarrhea Caused by Rotavirus in a Preclinical Model. Nutrients. 2020; 12(2):498. https://doi.org/10.3390/nu12020498
Chicago/Turabian StyleAzagra-Boronat, Ignasi, Malén Massot-Cladera, Karen Knipping, Johan Garssen, Kaouther Ben Amor, Jan Knol, Àngels Franch, Margarida Castell, María J. Rodríguez-Lagunas, and Francisco J. Pérez-Cano. 2020. "Strain-Specific Probiotic Properties of Bifidobacteria and Lactobacilli for the Prevention of Diarrhea Caused by Rotavirus in a Preclinical Model" Nutrients 12, no. 2: 498. https://doi.org/10.3390/nu12020498
APA StyleAzagra-Boronat, I., Massot-Cladera, M., Knipping, K., Garssen, J., Ben Amor, K., Knol, J., Franch, À., Castell, M., Rodríguez-Lagunas, M. J., & Pérez-Cano, F. J. (2020). Strain-Specific Probiotic Properties of Bifidobacteria and Lactobacilli for the Prevention of Diarrhea Caused by Rotavirus in a Preclinical Model. Nutrients, 12(2), 498. https://doi.org/10.3390/nu12020498









