Circulating Zinc and Copper Levels are Associated with Sperm Quality in Obese Men after Metabolic Surgery: A Pilot Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Subjects
2.2. Statement of Ethics
2.3. Anthropometric and Analytical Procedures
2.4. Trace Elements and Vitamins Analyses
2.5. Sperm Analysis
2.6. Statistics
3. Results
3.1. Baseline Characteristics
3.2. Zinc, Copper, Other Trace Elements and Sperm Quality at Baseline
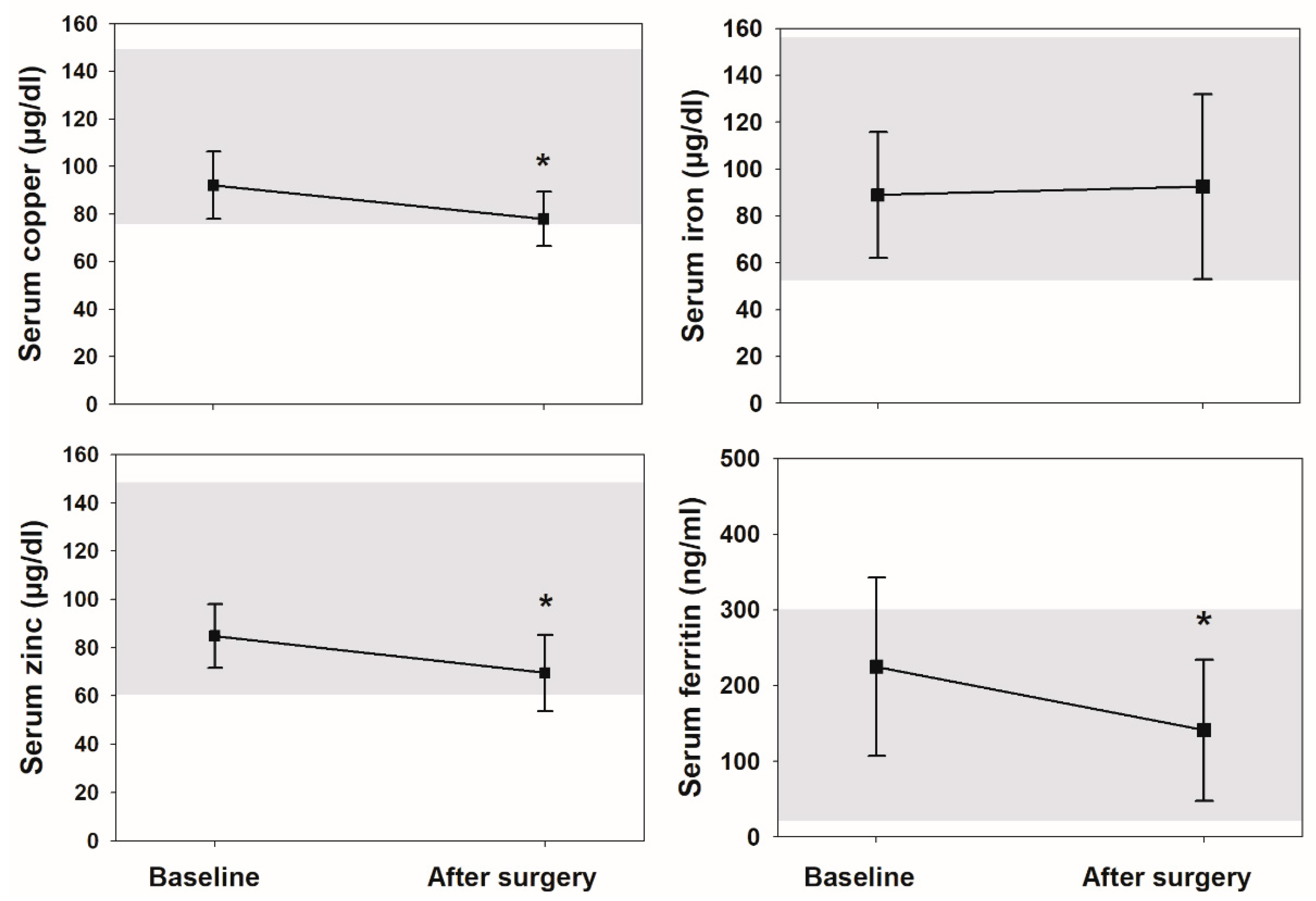
3.3. Changes at Follow-Up
3.4. Zinc, Copper, Other Trace Elements and Sperm Quality after Metabolic Surgery
4. Discussion
Limitations of the Study
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Data Statement
References
- Hogarth, C.A.; Griswold, M.D. The key role of vitamin A in spermatogenesis. J. Clin. Investig. 2010, 120, 956–962. [Google Scholar] [CrossRef]
- Ebisch, I.; Thomas, C.; Peters, W.; Braat, D.; Steegers-Theunissen, R.P. The importance of folate, zinc and antioxidants in the pathogenesis and prevention of subfertility. Hum. Reprod. Update 2006, 13, 163–174. [Google Scholar] [CrossRef] [PubMed]
- Hammiche, F.; Laven, J.S.; Twigt, J.M.; Boellaard, W.P.; Steegers, E.A.; Steegers-Theunissen, R.P. Body mass index and central adiposity are associated with sperm quality in men of subfertile couples. Hum. Reprod. 2012, 27, 2365–2372. [Google Scholar] [CrossRef] [PubMed]
- Samavat, J.; Natali, I.; Degl’Innocenti, S.; Filimberti, E.; Cantini, G.; Di Franco, A.; Danza, G.; Seghieri, G.; Lucchese, M.; Baldi, E.; et al. Acrosome reaction is impaired in spermatozoa of obese men: A preliminary study. Fertil. Steril. 2014, 102, 1274–1281.e2. [Google Scholar] [CrossRef] [PubMed]
- Calderón, B.; Gómez-Martín, J.M.; Vega-Piñero, B.; Martín-Hidalgo, A.; Galindo, J.; Luque-Ramírez, M.; Escobar-Morreale, H.F.; Botella-Carretero, J.I. Prevalence of male secondary hypogonadism in moderate to severe obesity and its relationship with insulin resistance and excess body weight. Andrology 2015, 4, 62–67. [Google Scholar] [CrossRef] [PubMed]
- Gunanti, I.R.; Marks, G.C.; Al Mamun, A.; Long, K.Z. Low Serum Vitamin B-12 and Folate Concentrations and Low Thiamin and Riboflavin Intakes Are Inversely Associated with Greater Adiposity in Mexican American Children. J. Nutr. 2014, 144, 2027–2033. [Google Scholar] [CrossRef]
- Manna, P.; Jain, S.K. Obesity, Oxidative Stress, Adipose Tissue Dysfunction, and the Associated Health Risks: Causes and Therapeutic Strategies. Metab. Syndr. Relat. Disord. 2015, 13, 423–444. [Google Scholar] [CrossRef]
- Sjostrom, L.; Gummesson, A.; Sjostrom, C.D.; Narbro, K.; Peltonen, M.; Wedel, H.; Bengtsson, C.; Bouchard, C.; Carlsson, B.; Dahlgren, S.; et al. Effects of bariatric surgery on cancer incidence in obese patients in sweden (swedish obese subjects study): A prospective, controlled intervention trial. Lancet Oncol. 2009, 10, 653–662. [Google Scholar] [CrossRef]
- Sjostrom, L.; Narbro, K.; Sjostrom, C.D.; Karason, K.; Larsson, B.; Wedel, H.; Lystig, T.; Sullivan, M.; Bouchard, C.; Carlsson, B.; et al. Effects of bariatric surgery on mortality in swedish obese subjects. N. Engl. J. Med. 2007, 357, 741–752. [Google Scholar] [CrossRef]
- Adams, T.D.; Davidson, L.E.; Litwin, S.E.; Kim, J.; Kolotkin, R.L.; Nanjee, M.N.; Gutierrez, J.M.; Frogley, S.J.; Ibele, A.R.; Brinton, E.A.; et al. Weight and metabolic outcomes 12 years after gastric bypass. N. Engl. J. Med. 2017, 377, 1143–1155. [Google Scholar] [CrossRef]
- Escobar-Morreale, H.F.; Santacruz, E.; Luque-Ramírez, M.; Carretero, J.I.B. Prevalence of ’obesity-associated gonadal dysfunction’ in severely obese men and women and its resolution after bariatric surgery: A systematic review and meta-analysis. Hum. Reprod. Update 2017, 23, 390–408. [Google Scholar] [CrossRef] [PubMed]
- Calderón, B.; Galdón, A.; Calañas, A.; Peromingo, R.; Galindo, J.; García-Moreno, F.; Rodríguez-Velasco, G.; Martin, A.; Vázquez, C.; Escobar-Morreale, H.F.; et al. Effects of Bariatric Surgery on Male Obesity-Associated Secondary Hypogonadism: Comparison of Laparoscopic Gastric Bypass with Restrictive Procedures. Obes. Surg. 2014, 24, 1686–1692. [Google Scholar] [CrossRef]
- Di Frega, A.S.; Dale, B.; Di Matteo, L.; Wilding, M. Secondary male factor infertility after Roux-en-Y gastric bypass for morbid obesity: Case report. Hum. Reprod. 2005, 20, 997–998. [Google Scholar] [CrossRef]
- El Bardisi, H.; Majzoub, A.; Arafa, M.; Almalki, A.; Al Said, S.; Khalafalla, K.; Jabbour, G.; Basha, M.; Al Ansari, A.; Sabanegh, E., Jr. Effect of bariatric surgery on semen parameters and sex hormone concentrations: A prospective study. Reprod. Biomed. Online 2016, 33, 606–611. [Google Scholar] [CrossRef]
- Lazaros, L.; Hatzi, E.; Markoula, S.; Takenaka, A.; Sofikitis, N.; Zikopoulos, K.; Georgiou, I. Dramatic reduction in sperm parameters following bariatric surgery: Report of two cases. Andrologia 2012, 44, 428–432. [Google Scholar] [CrossRef]
- Legro, R.S.; Kunselman, A.R.; Meadows, J.W.; Kesner, J.S.; Krieg, E.F.; Rogers, A.M.; Cooney, R.N. Time-related increase in urinary testosterone levels and stable semen analysis parameters after bariatric surgery in men. Reprod. Biomed. Online 2014, 30, 150–156. [Google Scholar] [CrossRef] [PubMed]
- Reis, L.O.; Zani, E.L.; Saad, R.D.; Chaim, E.A.; de Oliveira, L.C.; Fregonesi, A. Bariatric surgery does not interfere with sperm quality—A preliminary long-term study. Reprod. Sci. 2012, 19, 1057–1062. [Google Scholar] [CrossRef] [PubMed]
- Samavat, J.; Cantini, G.; Lotti, F.; Di Franco, A.; Tamburrino, L.; Degl’Innocenti, S.; Maseroli, E.; Filimberti, E.; Facchiano, E.; Lucchese, M.; et al. Massive Weight Loss Obtained by Bariatric Surgery Affects Semen Quality in Morbid Male Obesity: A Preliminary Prospective Double-Armed Study. Obes. Surg. 2017, 28, 69–76. [Google Scholar] [CrossRef] [PubMed]
- Sermondade, N.; Massin, N.; Boitrelle, F.; Pfeffer, J.; Eustache, F.; Sifer, C.; Czernichow, S.; Lévy, R. Sperm parameters and male fertility after bariatric surgery: Three case series. Reprod. Biomed. Online 2012, 24, 206–210. [Google Scholar] [CrossRef]
- Lee, Y.; Dang, J.T.; Switzer, N.; Yu, J.; Tian, C.; Birch, D.W.; Karmali, S. Impact of Bariatric Surgery on Male Sex Hormones and Sperm Quality: A Systematic Review and Meta-Analysis. Obes. Surg. 2018, 29, 334–346. [Google Scholar] [CrossRef]
- Calderón, B.; Huerta, L.; Galindo, J.; Casbas, J.M.G.; Escobar-Morreale, H.F.; Martín-Hidalgo, A.; Botella-Carretero, J.I. Lack of Improvement of Sperm Characteristics in Obese Males After Obesity Surgery Despite the Beneficial Changes Observed in Reproductive Hormones. Obes. Surg. 2019, 29, 2045–2050. [Google Scholar] [CrossRef] [PubMed]
- Vermeulen, A.; Verdonck, L.; Kaufman, J.M. A critical evaluation of simple methods for the estimation of free testosterone in serum. J. Clin. Endocrinol. Metab. 1999, 84, 3666–3672. [Google Scholar] [CrossRef] [PubMed]
- Tocci, A.; Lucchini, C. WHO reference values for human semen. Hum. Reprod. Update 2010, 16, 559. [Google Scholar] [CrossRef] [PubMed]
- Balsa, J.A.; Botella-Carretero, J.I.; Gómez-Martín, J.M.; Peromingo, R.; Arrieta, F.; Santiuste, C.; Zamarron, I.; Vázquez, C. Copper and Zinc Serum Levels after Derivative Bariatric Surgery: Differences Between Roux-en-Y Gastric Bypass and Biliopancreatic Diversion. Obes. Surg. 2011, 21, 744–750. [Google Scholar] [CrossRef]
- Busetto, L.; Dicker, D.; Azran, C.; Batterham, R.L.; Farpour-Lambert, N.; Fried, M.; Hjelmesæth, J.; Kinzl, J.; Leitner, D.R.; Makaronidis, J.M.; et al. Practical Recommendations of the Obesity Management Task Force of the European Association for the Study of Obesity for the Post-Bariatric Surgery Medical Management. Obes. Facts 2017, 10, 597–632. [Google Scholar] [CrossRef]
- Kerns, K.; Zigo, M.; Sutovsky, P. Zinc: A Necessary Ion for Mammalian Sperm Fertilization Competency. Int. J. Mol. Sci. 2018, 19, 4097. [Google Scholar] [CrossRef]
- Lichten, L.A.; Cousins, R.J. Mammalian zinc transporters: Nutritional and physiologic regulation. Annu. Rev. Nutr. 2009, 29, 153–176. [Google Scholar] [CrossRef]
- Chu, D.S. Zinc: A small molecule with a big impact on sperm function. PLoS Biol. 2018, 16, e2006204. [Google Scholar] [CrossRef]
- Zhao, J.; Dong, X.; Hu, X.; Long, Z.; Wang, L.; Liu, Q.; Sun, B.; Wang, Q.; Wu, Q.; Li, L. Zinc levels in seminal plasma and their correlation with male infertility: A systematic review and meta-analysis. Sci. Rep. 2016, 6, 22386. [Google Scholar] [CrossRef]
- Hunt, C.D.; Johnson, P.E.; Herbel, J.; Mullen, L.K. Effects of dietary zinc depletion on seminal volume and zinc loss, serum testosterone concentrations, and sperm morphology in young men. Am. J. Clin. Nutr. 1992, 56, 148–157. [Google Scholar] [CrossRef]
- Dissanayake, D.; Wijesinghe, P.S.; Ratnasooriya, W.D.; Wimalasena, S. Effects of zinc supplementation on sexual behavior of male rats. J. Hum. Reprod. Sci. 2009, 2, 57–61. [Google Scholar] [CrossRef] [PubMed]
- Irani, M.; Amirian, M.; Sadeghi, R.; Lez, J.L.; Latifnejad Roudsari, R. The effect of folate and folate plus zinc supplementation on endocrine parameters and sperm characteristics in sub-fertile men: A systematic review and meta-analysis. Urol. J. 2017, 14, 4069–4078. [Google Scholar] [PubMed]
- Schisterman, E.F.; Sjaarda, L.A.; Clemons, T.; Carrell, D.T.; Perkins, N.J.; Johnstone, E.; Lamb, D.; Chaney, K.; Van Voorhis, B.J.; Ryan, G.; et al. Effect of Folic Acid and Zinc Supplementation in Men on Semen Quality and Live Birth Among Couples Undergoing Infertility Treatment: A randomized clinical trial. JAMA 2020, 323, 35–48. [Google Scholar] [CrossRef] [PubMed]
- Tvrdá, E.; Peer, R.; Sikka, S.C.; Agarwal, A. Iron and copper in male reproduction: A double-edged sword. J. Assist. Reprod. Genet. 2014, 32, 3–16. [Google Scholar] [CrossRef]
- Hellman, N.E.; Gitlin, J.D. Ceruloplasmin metabolism and function. Annu. Rev. Nutr. 2002, 22, 439–458. [Google Scholar] [CrossRef]
- Akinloye, O.; Abbiyesuku, F.M.; Oguntibeju, O.O.; Arowojolu, A.O.; Truter, E.J. The impact of blood and seminal plasma zinc and copper concentrations on spermogram and hormonal changes in infertile nigerian men. Reprod. Biol. 2011, 11, 83–97. [Google Scholar] [CrossRef]
- Wong, W.Y.; Flik, G.; Groenen, P.M.; Swinkels, D.W.; Thomas, C.M.; Copius-Peereboom, J.H.; Merkus, H.M.; Steegers-Theunissen, R.P. The impact of calcium, magnesium, zinc, and copper in blood and seminal plasma on semen parameters in men. Reprod. Toxicol. 2001, 15, 131–136. [Google Scholar] [CrossRef]
- Lyubimov, A.V.; Smith, J.A.; Rousselle, S.D.; Mercieca, M.D.; Tomaszewski, J.E.; Smith, A.C.; Levine, B.S. The effects of tetrathiomolybdate (TTM, NSC-714598) and copper supplementation on fertility and early embryonic development in rats. Reprod. Toxicol. 2004, 19, 223–233. [Google Scholar] [CrossRef]
- Van Niekerk, F.E.; Van Niekerk, C.H. The influence of experimentally induced copper deficiency on the fertility of rams. I. Semen parameters and peripheral plasma androgen concentration. J. S. Afr. Vet. Assoc. 1989, 60, 28–31. [Google Scholar]
- Van Niekerk, F.E.; Van Niekerk, C.H. The influence of experimentally induced copper deficiency on the fertility of rams. II. Macro- and microscopic changes in the testes. J. S. Afr. Vet. Assoc. 1989, 60, 32–35. [Google Scholar]
- Picco, S.; De Luca, J.; Mattioli, G.; Dulout, F. DNA damage induced by copper deficiency in cattle assessed by the Comet assay. Mutat. Res. Toxicol. Environ. Mutagen. 2001, 498, 1–6. [Google Scholar] [CrossRef]
- Lieu, P.T.; Heiskala, M.; Peterson, P.A.; Yang, Y. The roles of iron in health and disease. Mol. Asp. Med. 2001, 22, 1–87. [Google Scholar] [CrossRef]
- Büchter, D.; Behre, H.; Kliesch, S.; Nieschlag, E. Pulsatile GnRH or human chorionic gonadotropin/human menopausal gonadotropin as effective treatment for men with hypogonadotropic hypogonadism: A review of 42 cases. Eur. J. Endocrinol. 1998, 139, 298–303. [Google Scholar] [CrossRef]
- Liu, P.Y.; Baker, H.W.G.; Jayadev, V.; Zacharin, M.; Conway, A.J.; Handelsman, D.J. Induction of Spermatogenesis and Fertility during Gonadotropin Treatment of Gonadotropin-Deficient Infertile Men: Predictors of Fertility Outcome. J. Clin. Endocrinol. Metab. 2009, 94, 801–808. [Google Scholar] [CrossRef]
- Prior, M.; Stewart, J.; McEleny, K.; Dwyer, A.A.; Quinton, R.; Prior, M. Fertility induction in hypogonadotropic hypogonadal men. Clin. Endocrinol. 2018, 89, 712–718. [Google Scholar] [CrossRef]
- Datz, C.; Felder, T.K.; Niederseer, D.; Aigner, E. Iron homeostasis in the Metabolic Syndrome. Eur. J. Clin. Investig. 2013, 43, 215–224. [Google Scholar] [CrossRef]

| At Baseline | After Metabolic Surgery | p | |||||
|---|---|---|---|---|---|---|---|
| Anthropometric characteristics | |||||||
| BMI (kg/m2) | 50 | ± | 10 | 32 | ± | 7 | <0.001 |
| Waist circumference (cm) | 144 | ± | 12 | 106 | ± | 12 | 0.012 |
| Systolic BP (mmHg) | 146 | ± | 15 | 130 | ± | 19 | 0.031 |
| Diastolic BP (mmHg) | 89 | ± | 16 | 77 | ± | 13 | 0.048 |
| Metabolic parameters | |||||||
| Total cholesterol (mg/dL) | 193 | ± | 38 | 170 | ± | 45 | 0.002 |
| HDL (mg/dL) | 39 | ± | 6 | 49 | ± | 11 | 0.002 |
| LDL (mg/dL) | 146 | ± | 32 | 106 | ± | 38 | 0.006 |
| Triglycerides (mg/dL) | 154 | ± | 60 | 92 | ± | 64 | 0.001 |
| Fasting glucose (mg/dL) | 120 | ± | 63 | 86 | ± | 13 | 0.010 |
| Fasting insulin (µU/mL) | 25 | ± | 13 | 6 | ± | 2 | 0.001 |
| HOMA-IR | 7.5 | ± | 5.6 | 1.2 | ± | 0.3 | 0.001 |
| TSH (µU/mL) | 2.3 | ± | 1.3 | 1.9 | ± | 1.3 | 0.018 |
| Reproductive hormones | |||||||
| Total testosterone (ng/dL) | 364 | ± | 163 | 660 | ± | 208 | 0.025 |
| SHBG (µg/dL) | 224 | ± | 83 | 478 | ± | 144 | 0.001 |
| Free testosterone (ng/dL) | 8 | ± | 4 | 11 | ± | 3 | 0.005 |
| Estradiol (pg/mL) | 37 | ± | 12 | 36 | ± | 12 | 0.151 |
| FSH (U/l) | 3.5 | ± | 1.1 | 4.0 | ± | 1.5 | <0.001 |
| LH (U/l) | 3.5 | ± | 1.0 | 3.8 | ± | 1.3 | 0.066 |
| Inhibin B (ng/mL) | 0.56 | ± | 0.25 | 0.70 | ± | 0.24 | 0.041 |
| At Baseline | After Metabolic Surgery | p | |||||
|---|---|---|---|---|---|---|---|
| 25-hydroxyvitamin D (ng/mL) | 24.4 | ± | 10.7 | 23.7 | ± | 6.8 | 0.674 |
| Cobalamin (pg/mL) | 374 | ± | 106 | 335 | ± | 119 | 0.336 |
| Folic acid (ng/mL) | 7.5 | ± | 6.2 | 9.1 | ± | 5.7 | 0.203 |
| Retinol (µg/dL) | 45 | ± | 13 | 37 | ± | 10 | 0.675 |
| Retinol/RBP ratio | 9.8 | ± | 1.7 | 9.9 | ± | 1.5 | 0.232 |
| Alpha tocopherol (µg/mL) | 13 | ± | 3 | 11 | ± | 4 | 0.090 |
| Alpha tocopherol/total cholesterol ratio | 6.7 | ± | 1.3 | 6.6 | ± | 0.9 | 0.667 |
| At Baseline | After Metabolic Surgery | p | |||||
|---|---|---|---|---|---|---|---|
| Sperm volume (ml) * | 2.5 (1.0) | 2.3 (1.8) | 0.041 | ||||
| Sperm concentration (106/mL) | 18 | ± | 69 | 12 | ± | 38 | 0.074 |
| Fluid pH | 8.1 | ± | 0.2 | 8.3 | ± | 0.3 | 0.046 |
| Motility—total (%) | 35 | ± | 35 | 35 | ± | 50 | 0.579 |
| Motility—progressive (%) | 20 | ± | 30 | 25 | ± | 35 | 0.244 |
| Viability (%) | 10 | ± | 5 | 10 | ± | 5 | 0.550 |
| Normal forms (%) | 2 | ± | 4 | 2 | ± | 4 | 0.783 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Calderón, B.; Gómez-Martín, J.M.; Cuadrado-Ayuso, M.; Cobeta, P.; Vega-Piñero, B.; Mateo, R.; Galindo, J.; Botella-Carretero, J.I. Circulating Zinc and Copper Levels are Associated with Sperm Quality in Obese Men after Metabolic Surgery: A Pilot Study. Nutrients 2020, 12, 3354. https://doi.org/10.3390/nu12113354
Calderón B, Gómez-Martín JM, Cuadrado-Ayuso M, Cobeta P, Vega-Piñero B, Mateo R, Galindo J, Botella-Carretero JI. Circulating Zinc and Copper Levels are Associated with Sperm Quality in Obese Men after Metabolic Surgery: A Pilot Study. Nutrients. 2020; 12(11):3354. https://doi.org/10.3390/nu12113354
Chicago/Turabian StyleCalderón, Berniza, Jesús M. Gómez-Martín, Marta Cuadrado-Ayuso, Pilar Cobeta, Belén Vega-Piñero, Raquel Mateo, Julio Galindo, and José I. Botella-Carretero. 2020. "Circulating Zinc and Copper Levels are Associated with Sperm Quality in Obese Men after Metabolic Surgery: A Pilot Study" Nutrients 12, no. 11: 3354. https://doi.org/10.3390/nu12113354
APA StyleCalderón, B., Gómez-Martín, J. M., Cuadrado-Ayuso, M., Cobeta, P., Vega-Piñero, B., Mateo, R., Galindo, J., & Botella-Carretero, J. I. (2020). Circulating Zinc and Copper Levels are Associated with Sperm Quality in Obese Men after Metabolic Surgery: A Pilot Study. Nutrients, 12(11), 3354. https://doi.org/10.3390/nu12113354






