In Vitro Antithrombotic and Hypocholesterolemic Activities of Milk Fermented with Specific Strains of Lactococcus lactis
Abstract
1. Introduction
2. Materials and Methods
2.1. Substrates and Chemicals
2.2. Bacterial Strains and Growth Conditions
2.3. Preparation of Fermented Milk
2.4. Simulated Gastrointestinal Digestion Model
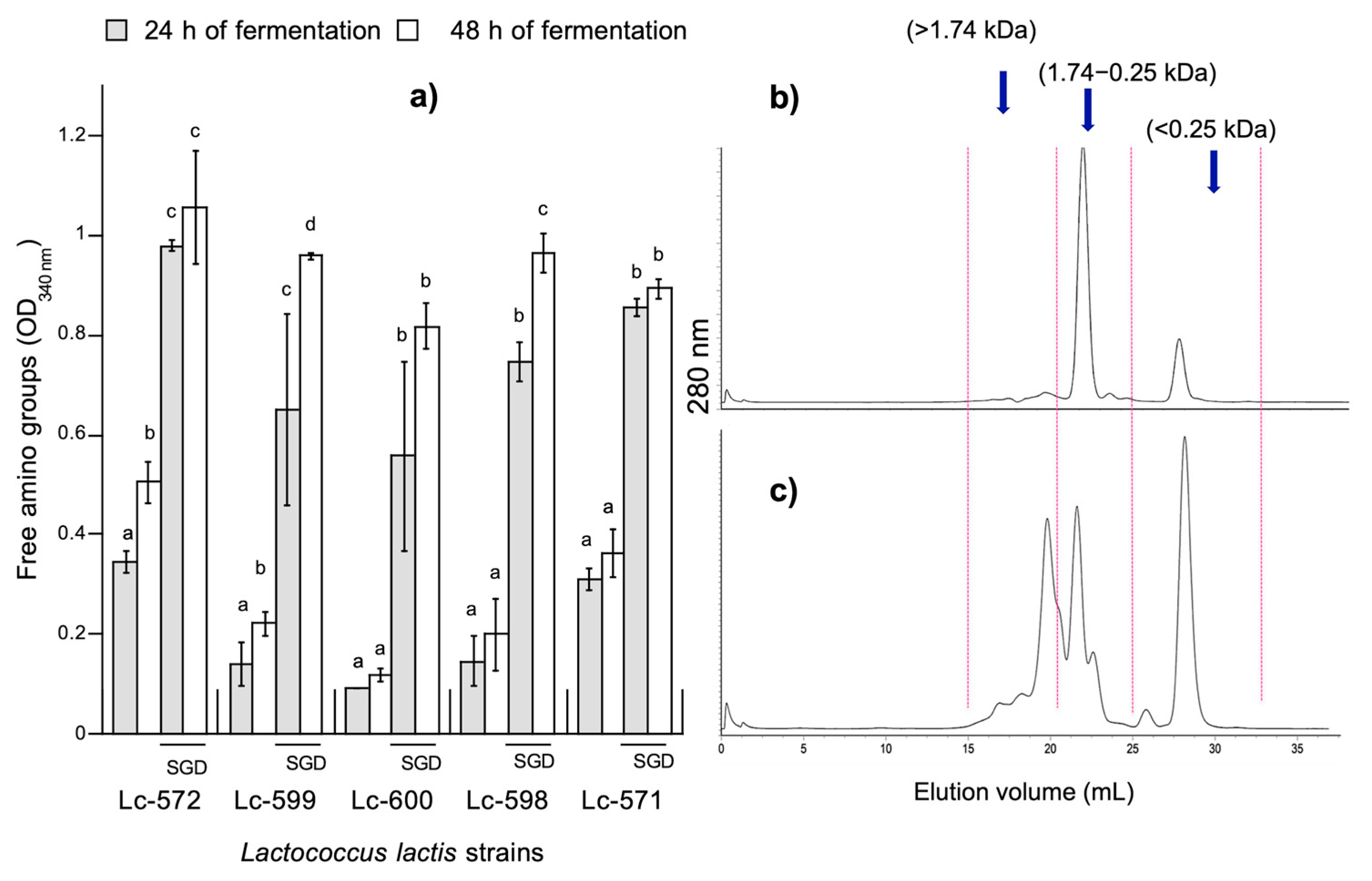
2.5. Determination of Free Amino Groups
2.6. Preparation of Water-Soluble Peptidic Fraction
2.7. Size Exclusion Chromatography (SEC)
2.8. Determination of Antithrombotic Activity
2.8.1. Inhibition of Thrombin Induced Fibrin Polymerization
2.8.2. In Vitro Anticoagulant Activity in Plasma
2.9. Determination of Hypocholesterolemic Activity
2.9.1. Inhibition of Micellar Solubility of Cholesterol
2.9.2. In Vitro Bile Acid-Binding Capacity
2.10. Statistical Analysis
3. Results and Discussion
3.1. Determination of Free Amino Groups in Fermented Milk
3.2. Antithrombotic Activity
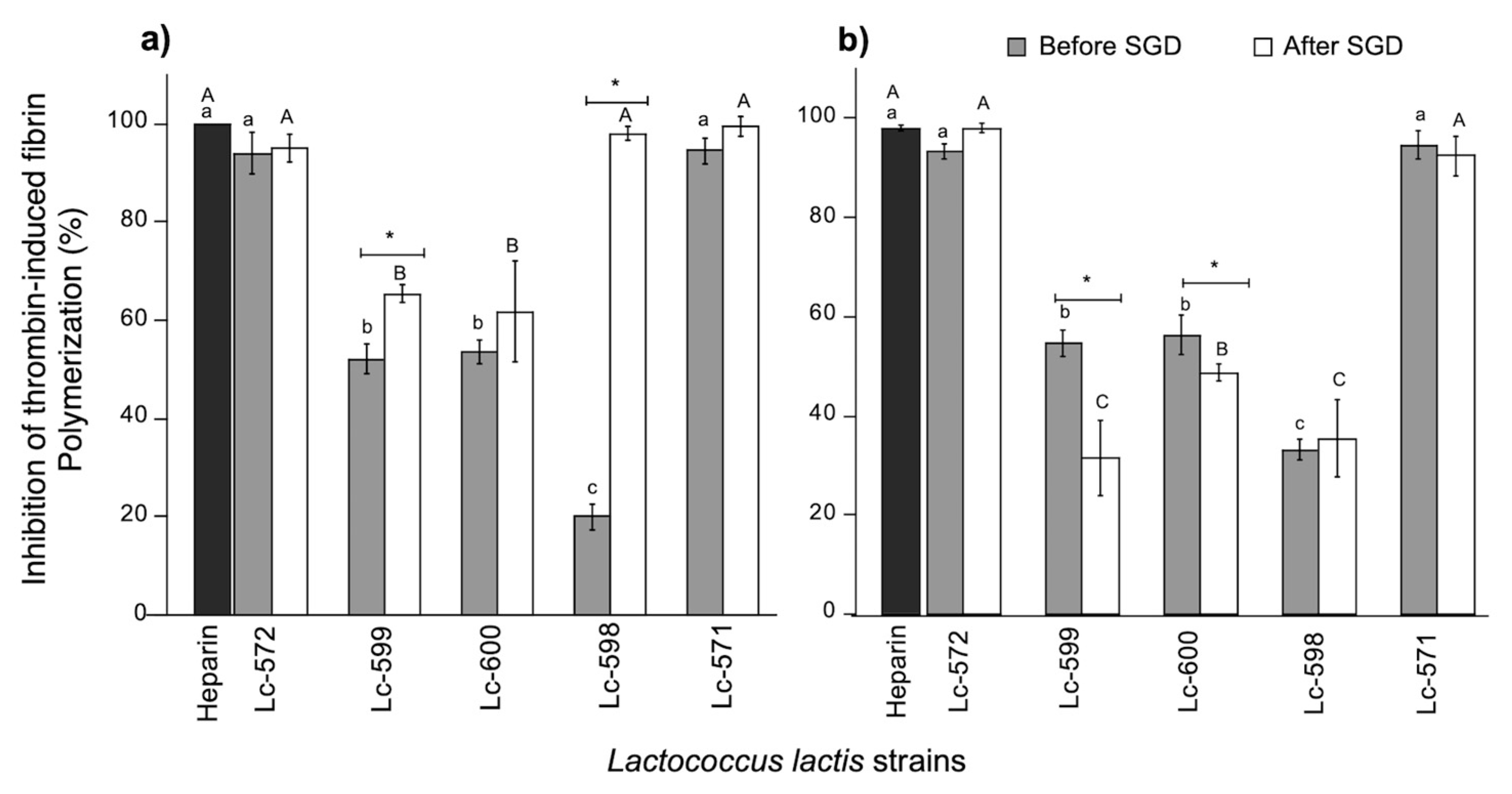
3.2.1. Inhibition of Thrombin-Induced Fibrin Polymerization
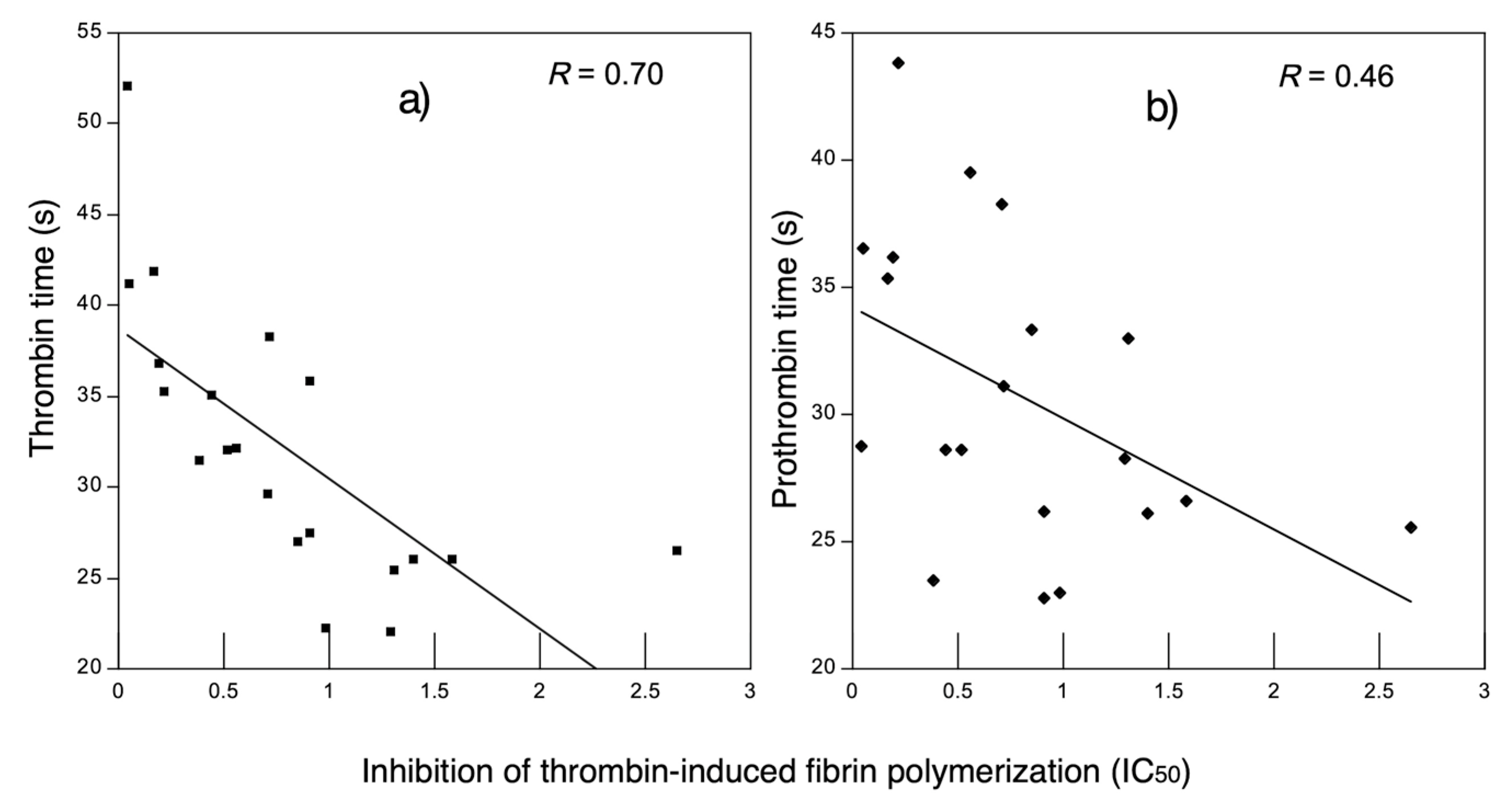
3.2.2. Anticoagulant Activity in Decalcified Plasma
3.3. Hypocholesterolemic Activity
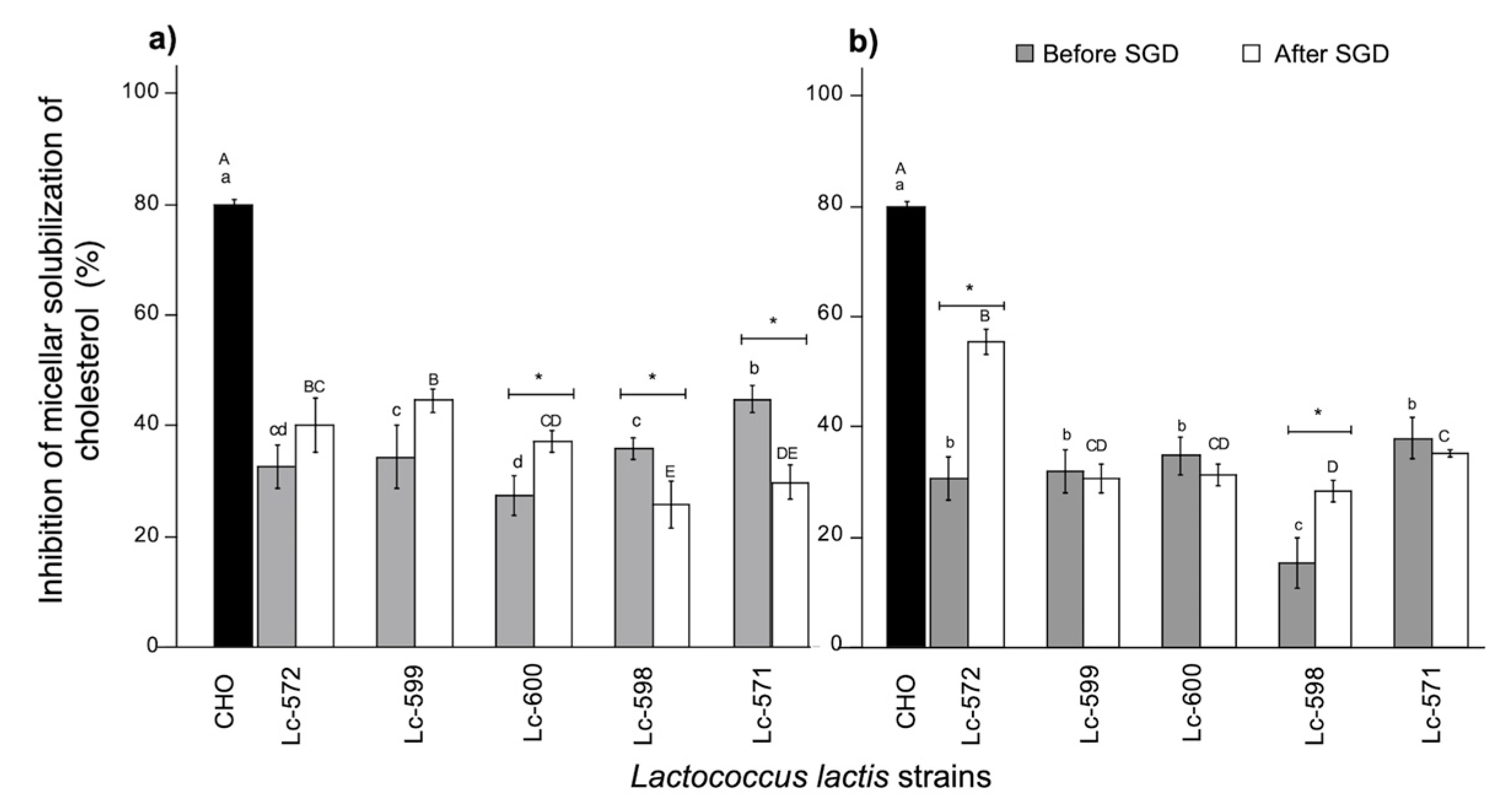
3.3.1. Micellar Solubility of Cholesterol
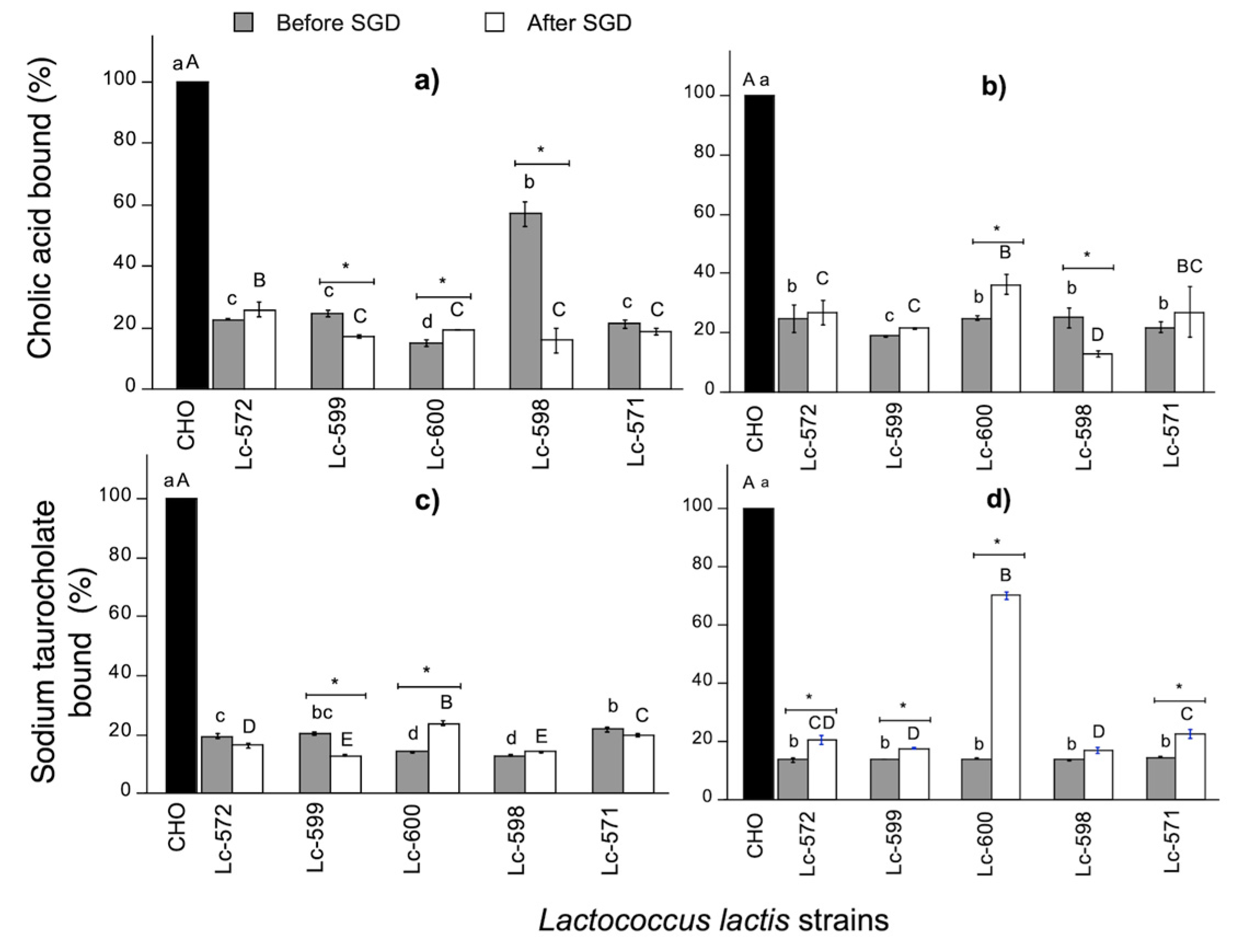
3.3.2. Bile Acid Binding Capacity
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wang, T.; Butany, J. Pathogenesis of atherosclerosis. Diagn. Histopathol. 2018, 23, 447–478. [Google Scholar] [CrossRef]
- Perez-Calahorra, S.; Lacaustra, M.; Marco-Benedí, V.; Lamiquiz-Moneo, J.P.; Plana, N.; Sanchez-Hernandez, R.M.; Amor, J.A.; Almagro, F.; Fuentes, F.; Suarez-Tembra, M.; et al. Effect of lipid-lowering treatment in cardiovascular disease prevalence in familial hypercholesterolemia. Atherosclerosis 2019, 284, 245–252. [Google Scholar] [CrossRef]
- Balakumar, P.; Maung-U, K.; Jadadeesh, G. Prevalence and prevention of cardiovascular disease and diabetes mellitus. Pharmacol. Res. 2016, 113, 600–609. [Google Scholar] [CrossRef]
- Salvo, F.; Bezin, J.; Bosco-Levy, P.; Letinier, L.; Blin, P.; Pariente, A.; Moore, N. Pharmacological treatments of cardiovascular diseases: Evidence from real-life studies. Pharmacol. Res. 2017, 118, 43–52. [Google Scholar] [CrossRef]
- Valdés, A.; Cifuentes, A.; León, C. Foodomics evaluation of bioactive compounds in foods. Trends Anal. Chem. 2017, 96, 2–13. [Google Scholar] [CrossRef]
- Erdmann, K.; Cheung, W.Y.B.; Shroder, H. The possible roles of food-derived bioactive peptides in reducing the risk of cardiovascular disease. J. Nutr. Biochem. 2008, 19, 643–654. [Google Scholar] [CrossRef]
- Lammi, C.; Aiello, G.; Boschin, G.; Arnoldi, A. Multifunctional peptides for the prevention of cardiovascular disease: A new concept in the are of bioactive food-derived peptides. J. Funct. Foods. 2019, 55, 135–145. [Google Scholar] [CrossRef]
- Toldrá, F.; Reing, M.; Aristoy, M.C.; Mora, L. Generation of bioactive peptides during food processing. Food Chem. 2018, 267, 395–404. [Google Scholar] [CrossRef]
- Singh, B.; Ram, C.; Singh, D.; Singh, N.P.; Singh, A.; Singh, R.; Verma, R.R. Potential of novel bioactive peptides as functional ingredients in preventing cardiovascular disease. In Alternative and Replacement Foods, 1st ed.; Holban, A., Mihai, A., Eds.; Academic Press: San Diego, CA, USA, 2018; pp. 411–431. [Google Scholar]
- Nagaoka, S.; Futamura, Y.; Miwa, K.; Awano, T.; Yamauchi, K.; Kanamaru, Y.; Tadashi, K.; Kuwato, T. Identification of novel hypocholesterolemic peptides derived from bovine milk β-Lactoglobulin. Biochem. Biophys. Res. Commun. 2001, 281, 11–17. [Google Scholar] [CrossRef]
- Tu, M.; Feng, L.; Wang, Z.; Qiao, M.; Shahidi, F.; Lu, W.; Du, M. Sequence analysis and molecular docking of antithrombotic peptides from casein hydrolysates by trypsin digestion. J. Funct. Foods. 2017, 32, 313–323. [Google Scholar] [CrossRef]
- Howard, A.; Udenigwe, C. Mechanism and prospect of food protein hydrolysates and peptide-induced hypolipidaemia. Food Funct. 2013, 83, 40–51. [Google Scholar] [CrossRef]
- Fan, M.; Guo, T.; Li, W.; Chen, J.; Li, F.; Wang, C.; Shi, Y.; Xian, L.D.; Zhang, S. Isolation and identification of novel casein-derived bioactive peptides and potential functions in fermented casein with Lactobacillus helveticus. Food Sci. Hum. Wellness 2019. [Google Scholar] [CrossRef]
- Liu, M.; Bayjanov, J.B.; Renckens, B.; Nauta, A.; Siezen, R.J. The proteolytic system of lactic acid bacteria revisited: A genomic comparison. BMC Genom. 2010, 11, 36. [Google Scholar] [CrossRef]
- Mohanty, D.P.; Mohapatra, S.; Misra, S.; Sahu, P.S. Milk derived bioactive peptides and their impact on human health: A review. Saudi J. Biol. Sci. 2015, 23, 577–583. [Google Scholar] [CrossRef]
- Meisel, H. Multifunctional peptides encrypted in milk proteins. Biofactors 2014, 21, 55–61. [Google Scholar] [CrossRef]
- Aguilar-Toalá, J.E.; Santiago-López, L.; Peres, C.M.; Peres, C.; García, H.S.; Vallejo-Córdoba, B.; González-Córdova, A.F.; Hernández-Mendoza, A. Assessment of multifunctional activity of bioactive peptides derived from fermented milk by specific Lactobacillus plantarum strains. J. Dairy Sci. 2017, 100, 65–75. [Google Scholar] [CrossRef]
- Banan-Mwine, D.; Oh, D.H.; Lee, B.H. Bioactive peptides. Foods 2017, 6, 35. [Google Scholar]
- Hafeez, Z.; Cakir-Kiefer, C.; Roux, E.; Perrin, C.; Miclo, L.; Dary-Morout, A. Strategies of producing bioactive peptides from milk proteins to functionalize fermented milk products. Food Res. Int. 2014, 63, 71–80. [Google Scholar] [CrossRef]
- Hosono, A.; Otani, H.; Yasui, H.; Watanuki, M. Impact of fermented milk on human health: Cholesterol-lowering and immunomodulatory properties of fermented milk. Anim. Sci. J. 2002, 73, 241–256. [Google Scholar] [CrossRef]
- Prasanna, P.H.P.; Rastall, R.A. Potential applications of prebiotics to yogurt and impact on health. In Yogurt in Health and Disease Prevention, 1st ed.; Shah, N., Ed.; Academic Press: London, UK, 2017; pp. 171–183. [Google Scholar]
- Guan-Wen, C.; Jenn-Shou, T.; Bonnie, S.P. Cardiovascular effects of whey prozyme 6-facilitaed lactic acid bacteria fermentation of milk. J. Food Biochem. 2006, 31, 639–655. [Google Scholar]
- Rodríguez-Figueroa, J.C.; González-Córdova, A.F.; Astiazarán-García, H.; Hernández-Mendoza, A.; Vallejo-Cordoba, B. Antihypertensive and hypolipidemic effect of milk fermented by Lactococcus lactis strains. J. Dairy Sci. 2013, 96, 4094–4099. [Google Scholar] [CrossRef] [PubMed]
- Jolles, P.; Levy-Toledano, S.; Fiat, A.M.; Soria, C.; Gillessen, D.; Thomanidis, A.; Caen, J.P. Analogy between fibrinogen and casein. Effect of a undecapeptide isolated from kappa-casein on platelet function. Eur. J. Biochem. 1986, 158, 379–382. [Google Scholar] [CrossRef] [PubMed]
- Levy-Toledano, S.; Caen, J.; Grelac, F.; Maclouf, J. KRDS, a peptide derived from human lactotransferrin, inhibits thrombin-induced thromboxane synthesis by cyclooxygenase independent mechanism. Thromb. Haemost. 1995, 73, 857–861. [Google Scholar] [CrossRef] [PubMed]
- Rojas-Ronquillo, R.; Cruz-Guerrero, A.; Flores-Najera, A.; Rodríguez-Serrano, G.; Gómez-Ruiz, L.; Reyes-Grajeda, J.P.; Jimenez-Guzmán, J.; García-Garibay, M. Antithrombotic and angiotensin-converting enzyme inhibitory properties of peptides release from bovine casein by Lactobacillus casei shirota. Int. Dairy J. 2012, 26, 147–154. [Google Scholar] [CrossRef]
- Corrochano, A.R.; Saricay, Y.; Arranz, E.; Kelly, P.M.; Buckin, V.; Giblin, L. Comparison of antioxidant activities of bovine whey proteins before and after simulated gastrointestinal digestion. J. Dairy Sci. 2019, 102, 57–62. [Google Scholar] [CrossRef]
- Wang, B.; Li, B. Charge and hydrophobicity of casein peptides influence transpitelial transport and bioavailability. Food Chem. 2017, 245, 646–652. [Google Scholar] [CrossRef]
- Singh, B.P.; Vij, S. In vitro stability of bioactive peptides derived from fermented soy milk against heat treatment, pH and gastrointestinal enzymes. LWT Food Sci. Technol. 2018, 91, 303–307. [Google Scholar] [CrossRef]
- Cardoso, C.; Alfonso, C.; Lourenco, H. Bioaccesibility assessment methodologies and their consequences for the risk-benefit evaluation of food. Trends Food Sci. Technol. 2015, 41, 5–23. [Google Scholar] [CrossRef]
- Beltrán-Barrientos, L.M.; Hernández-Mendoza, A.; González-Córdova, A.F.; Astiazarán-García, H.; Esparza-Romero, J.; Vallejo-Cordoba, B. Mechanistic pathways underlying the antihypertensive effect of fermented milk with Lactococcus lactis NRRL B-50571 in spontaneously hypertensive rats. Nutrients 2018, 10, 262. [Google Scholar] [CrossRef]
- Kopf-Bolanz, K.A.; Shwander, F.; Gijs, M.; Vergeres, G.; Portmann, R.; Egger, L. Validation of in vitro digestive system for studying macronutrient decomposition in humans. J. Nutr. 2012, 142, 245–250. [Google Scholar] [CrossRef]
- Church, C.F.; Swaisgood, H.E.; Porter, D.H.; Ctignani, L. Spectrophotometric assay using o-Phthaldialdehyde for determination of proteolysis in milk and isolated milk proteins. J. Dairy Sci. 1983, 66, 1219–1227. [Google Scholar] [CrossRef]
- Mora-Cortes, W.G. Potencial Antihipertensivo y Antidiabético de Hidrolizados de Lactosuero Producidos con Enzimas Proteolíticas de Plantas. Master’s Thesis, Centro de Investigación en Alimentación y Desarrollo, A.C., Sonora, Mexico, 2018. [Google Scholar]
- Sabbione, A.C.; Scilingo, A.; Añon, M.C. Potential antithrombotic activity in amaranth proteins and peptides and its hydrolysates. LWT Food Sci. Technol. 2015, 60, 171–177. [Google Scholar] [CrossRef]
- Kirana, C.; Rogers, P.F.; Bennett, L.E.; Aveywardena, M.Y.; Patten, S.G. Naturally derived micelles for rapid in vitro screening of potential cholesterol-Lowering bioactives. J. Agric. Food Chem. 2005, 53, 4623–4627. [Google Scholar] [CrossRef]
- Yoshie-Stark, Y.; Wasche, A. In vitro binding of bile acids by lupin protein isolates and their hydrolysates. Food Chem. 2004, 88, 179–184. [Google Scholar] [CrossRef]
- Hernández-Ledesma, B.; Contreras, M.M.; Recio, I. Antihypertensive peptides: Production, bioavailability and incorporation into foods. Adv. Colloid Interface Sci. 2011, 9, 23–35. [Google Scholar] [CrossRef]
- Sánchez-Rivera, L.; Matínez-Maqueda, D.; Cruz-Huerta, E.; Miralles, B.; Recio, I. Peptidomics for discovery, bioavailability and monitoring of dairy bioactive peptides. Food Res. Int. 2014, 63, 170–181. [Google Scholar] [CrossRef]
- Gaertner, F.; Massberg, S. Blood coagulation in immunothrombosis—At the frontline of intravascular immunity. Semin. Immunol. 2016, 28, 561–569. [Google Scholar] [CrossRef]
- Marcone, S.; Fitzgerald, D. Milk-derived bioactive peptides and their health promoting effects: A potential role in atherosclerosis. Br. J. Clin. Pharmacol. 2016, 83, 152–163. [Google Scholar] [CrossRef]
- Rodriguez-Figueroa, J.C.; González-Córdova, A.F.; Torres-Llanez, M.J.; Garcia, H.S.; Vallejo-Cordoba, B. Novel angiotensin I-converting enzyme inhibitory peptides produced in fermented milk by specific wild Lactococcus lactis strains. J. Dairy Sci. 2012, 95, 5536–5543. [Google Scholar] [CrossRef]
- Bhagavan, N.V.; Chung-Eun, H. Biochemistry of hemostasis. In Essentials of Medical Biochemistry, 2nd ed.; Bhagavan, N.V., Chung-Eun, H., Eds.; Academic Press: San Diego, CA, USA, 2015; pp. 637–699. [Google Scholar]
- Beltrán-Barrientos, L.M.; González-Cordova, A.F.; Hernández-Mendoza, A.; Torres-Inguanzo, E.H.; Astiazarán-Garcia, H.; Esparza-Romero, J.; Vallejo-Cordoba, B. Randomized double-blind controlled clinical trial of the blood pressure-lowering effect of fermented milk with Lactococcus lactis: A pilot study. J. Dairy Sci. 2018, 4, 2819–2825. [Google Scholar] [CrossRef]
- Norata, G.D.; Catapano, A.L. Lipid lowering activity of drugs affecting cholesterol absortion. Nutr. Metab. Cardiovasc. Dis. 2003, 13, 42–51. [Google Scholar]
- Mohan, A.; Udenigwe, C. Towards the desing of hypolipidemic peptides: Deoxycholate binding affinity of hydrophobic peptide aggregates of casein plastein. J. Funct. Foods. 2015, 18, 129–136. [Google Scholar] [CrossRef]
- Nagaoka, S.; Nakamura, A.; Shibata, H.; Kanamaru, Y. Soystatin (VAWWMY), a novel bile acid-binding peptide, decreased micellar solubility and inhibited cholesterol abortion in rats. Biosci. Biotechnol. Biochem. 2010, 74, 1738–1741. [Google Scholar] [CrossRef]
- Kahlon, T.S.; Woodruff, C.L. In vitro binding of bile acids by soy protein pinto beans, black beans and wheat gluten. Food Chem. 2002, 79, 425–429. [Google Scholar] [CrossRef]
- Yao, S.; Agyei, D.; Udenigwe, C. Structural basis of bioactivity of food peptides in promoting metabolic health. Adv. Food Nutr. Res. 2018, 84, 145–181. [Google Scholar]
- Kongo-Dia, M.J.; Nsor-Atindana, J.; Zhang, H. Hypocholesterolemic activity and characterization of protein hydrolysates from defatted corn protein. Asian J. Biochem. 2011, 6, 439–449. [Google Scholar]
| WSF | 24 h | 48 h | ||
|---|---|---|---|---|
| Before | After | Before | After | |
| Lc-572 | 0.19 ± 0.02 d | 0.17 ± 0.07 d | 0.71 ± 0.03 b | 0.049 ± 0.00 c,* |
| Lc-599 | 1.29 ± 0.15 a | 0.38 ± 0.01 c,* | 0.91 ± 0.00 b | >1.0 |
| Lc-600 | 0.52 ± 0.02 c | 0.85 ± 0.01 b,* | 0.44 ± 0.032 c | 0.98 ± 0.0 a,* |
| Lc-598 | 2.65 ± 0.26 a | 0.91 ± 0.04 a,* | 1.31 ± 0.17 a | >1.0 |
| Lc-571 | 0.56 ± 0.00 c | 0.045 ± 0.02 e,* | 0.45 ± 0.03 b | 0.22 ± 0.00 b,* |
| Thrombin Time (s) | Pro-Thrombin Time (s) | |||||||
|---|---|---|---|---|---|---|---|---|
| WSF | 24 h | 48 h | 24 h | 48 h | ||||
| Before | After | Before | After | Before | After | Before | After | |
| Lc-572 | 36.83 ± 0.17 | 41.87 ± 0.26 * | 29.65 ± 0.54 | 41.17 ± 1.23 | 36.19 ± 0.97 | 35.33 ± 2.28 | 38.3 ± 3.11 | 36.5 ± 2.82 |
| Lc-599 | 29.50 ± 0.48 | 31.50 ± 0.92 | 35.85 ± 0.32 | 26.00 ± 0.10 * | 28.25 ± 0.35 | 23.5 ± 0.90 * | 22.78 ± 1.71 | 26.61 ± 1.92 * |
| Lc-600 | 32.05 ± 0.23 | 27.00 ± 0.20 | 35.03 ± 1.02 | 22.2 ± 0.20 * | 28.62 ± 0.02 | 33.25 ± 4.71 | 28.63 ± 2.68 | 22.98 ± 3.51 * |
| Lc-598 | 26.50 ± 0.23 | 27.50 ± 0.23 | 25.46 ± 0.43 | 26.00 ± 0.76 | 25.57 ± 0.79 | 26.17 ± 0.24 | 33.00 ± 0.70 | 26.13 ± 1.26 * |
| Lc-571 | 32.14 ± 0.56 | 52.07 ± 1.23 * | 38.27 ± 1.20 | 35.27 ± 0.34 | 39.52 ± 1.44 | 28.74 ± 3.81 * | 31.45 ± 0.70 | 43.85 ± 4.41 * |
| NC | 23 ± 2 | 18.9 ± 0.56 | ||||||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rendon-Rosales, M.Á.; Torres-Llanez, M.J.; González-Córdova, A.F.; Hernández-Mendoza, A.; Mazorra-Manzano, M.A.; Vallejo-Cordoba, B. In Vitro Antithrombotic and Hypocholesterolemic Activities of Milk Fermented with Specific Strains of Lactococcus lactis. Nutrients 2019, 11, 2150. https://doi.org/10.3390/nu11092150
Rendon-Rosales MÁ, Torres-Llanez MJ, González-Córdova AF, Hernández-Mendoza A, Mazorra-Manzano MA, Vallejo-Cordoba B. In Vitro Antithrombotic and Hypocholesterolemic Activities of Milk Fermented with Specific Strains of Lactococcus lactis. Nutrients. 2019; 11(9):2150. https://doi.org/10.3390/nu11092150
Chicago/Turabian StyleRendon-Rosales, Miguel Ángel, María J. Torres-Llanez, Aarón F. González-Córdova, Adrián Hernández-Mendoza, Miguel A. Mazorra-Manzano, and Belinda Vallejo-Cordoba. 2019. "In Vitro Antithrombotic and Hypocholesterolemic Activities of Milk Fermented with Specific Strains of Lactococcus lactis" Nutrients 11, no. 9: 2150. https://doi.org/10.3390/nu11092150
APA StyleRendon-Rosales, M. Á., Torres-Llanez, M. J., González-Córdova, A. F., Hernández-Mendoza, A., Mazorra-Manzano, M. A., & Vallejo-Cordoba, B. (2019). In Vitro Antithrombotic and Hypocholesterolemic Activities of Milk Fermented with Specific Strains of Lactococcus lactis. Nutrients, 11(9), 2150. https://doi.org/10.3390/nu11092150





