Improving the Health Benefits of Snap Bean: Genome-Wide Association Studies of Total Phenolic Content
Abstract
1. Introduction
2. Materials and Methods
2.1. Snap Bean Diversity Panel
2.2. Total Phenolics Analysis
2.3. Phenotyping
2.4. Genotyping
2.5. Genome-Wide Association Mapping
2.6. Candidate Gene Search
3. Results
3.1. Phenotypic Data
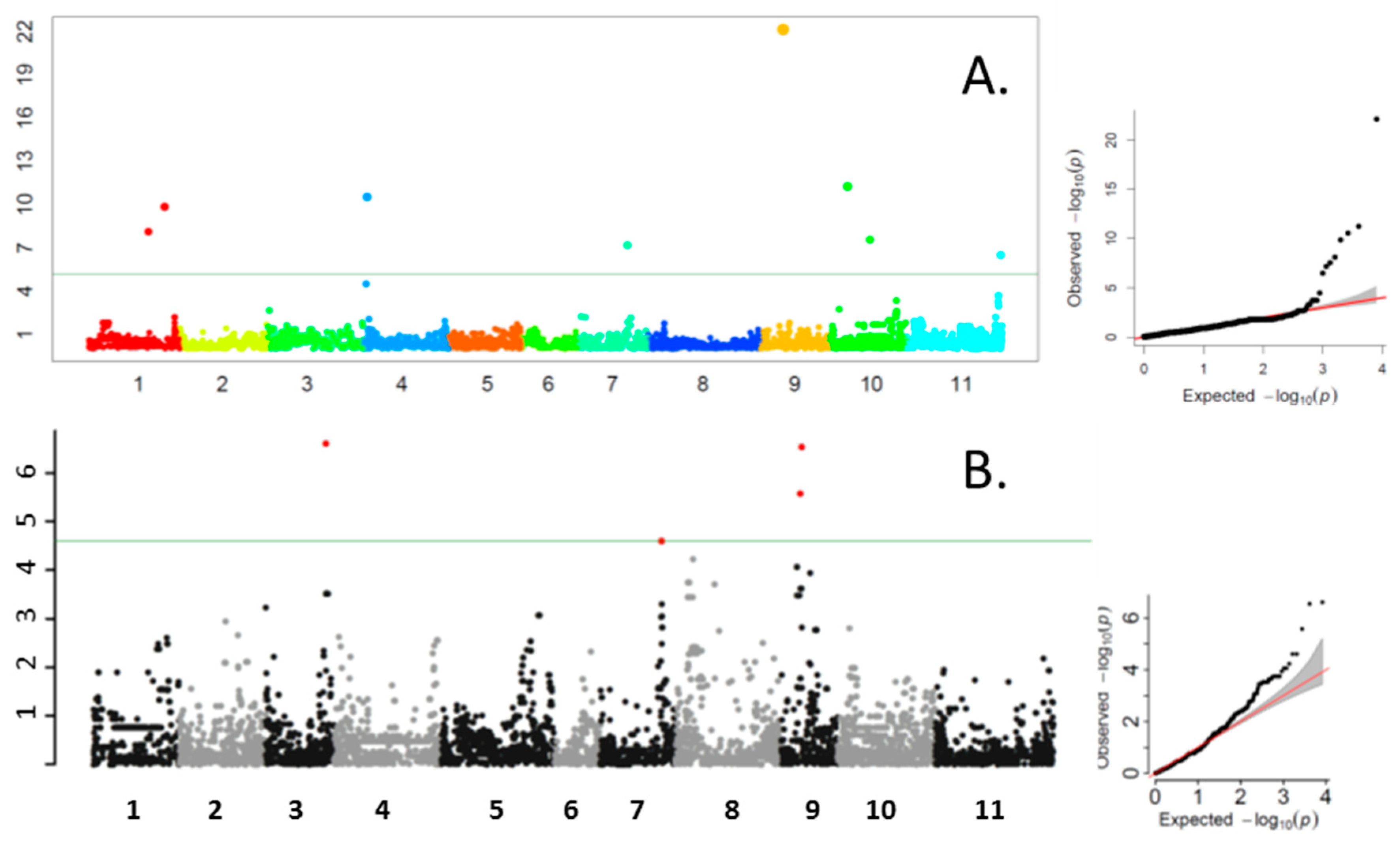
3.2. Genome-Wide Association Study
3.3. Total Phenolic Content QTN
3.4. Pod Color QTN
3.5. Flower Color SNPs
3.6. Candidate Genes in Proximity to Traits
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Thompson, M.D.; Thompson, H.J. Biomedical agriculture: A systematic approach to food crop improvement for chronic disease prevention. Adv. Agron. 2009, 102, 1–54. [Google Scholar]
- Hefferon, K. Nutritionally enhanced food crops; progress and perspectives. Int. J. Mol. Sci. 2015, 16, 3895–3914. [Google Scholar] [CrossRef] [PubMed]
- FDA. Guidance for Industry: Guide for Developing and Using Data Bases for Nutrition Labeling. Available online: http://www.fda.gov/regulatory-information/search-fda-guidance-documents/guidance-industry-guide-developing-and-using-data-bases-nutrition-labeling (accessed on 16 September 2019).
- Bond, C.A.; Thilmany, D.D.; Bond, J.K. What to choose? The value of label claims to fresh produce consumers. J. Agric. Resour. Econ. 2008, 33, 402–427. [Google Scholar]
- Laborde, L.; McGee, W. Food for Profit: Food Labels. Penn State Ext. 2019. Code UK153 04/19pod. [Google Scholar]
- USDA. USDA National Nutrient Database for Standard Reference. Available online: https://www.ars.usda.gov/northeast-area/beltsville-md-bhnrc/beltsville-human-nutrition-research-center/nutrient-data-laboratory/docs/usda-national-nutrient-database-for-standard-reference/ (accessed on 12 July 2019).
- Myers, J.R.; Kmiecik, K. Common Bean: Economic importance and relevance to biological science research. In The Common Bean Genome; Springer: Berlin, Germany, 2017; pp. 1–20. [Google Scholar]
- USDA. Food Availability and Consumption. Available online: https://www.ers.usda.gov/data-products/ag-and-food-statistics-charting-the-essentials/food-availability-and-consumption/ (accessed on 12 July 2019).
- Crozier, A.B.; Jaganath, I.N.; Clifford, M. Dietary phenolics: Chemistry, bioavailability and effects on health. Nat. Prod. Rep. 2009, 26, 1001–1043. [Google Scholar] [CrossRef] [PubMed]
- Hounsome, N.; Hounsome, B.; Tomos, D.; Edwards-Jones, G. Plant metabolites and nutritional quality of vegetables. J. Food Sci. 2008, 73, R48–R65. [Google Scholar] [CrossRef]
- Parr, A.J.; Bolwell, G.P. Phenols in the plant and in man. The potential for possible nutritional enhancement of the diet by modifying the phenols content or profile. J. Sci. Food Agric. 2000, 80, 985–1012. [Google Scholar] [CrossRef]
- Shahidi, F.; Ambigaipalan, P. Phenolics and polyphenolics in foods, beverages and spices: Antioxidant activity and health effects—A review. J. Funct. Foods 2015, 18, 820–897. [Google Scholar] [CrossRef]
- Amarowicz, R.; Pegg, R.B. Legumes as a source of natural antioxidants. Eur. J. Lipid Sci. Technol. 2008, 110, 865–878. [Google Scholar] [CrossRef]
- Xu, B.J.; Chang, S.K.C. A comparative study on phenolic profiles and antioxidant activities of legumes as affected by extraction solvents. J. Food Sci. 2007, 72, S159–S166. [Google Scholar] [CrossRef]
- Yang, Q.-Q.; Gan, R.-Y.; Ge, Y.-Y.; Zhang, D.; Corke, H. Polyphenols in common beans (Phaseolus vulgaris L.): Chemistry, analysis, and factors affecting composition. Compr. Rev. Food Sci. Food Saf. 2018, 17, 1518–1539. [Google Scholar] [CrossRef]
- Bazzano, L.A.; He, J.; Ogden, L.G.; Loria, C.; Vupputuri, S.; Myers, L.; Whelton, P.K. Legume consumption and risk of coronary heart disease in US men and women: NHANES I Epidemiologic Follow-up Study. Arch. Intern. Med. 2001, 161, 2573–2578. [Google Scholar] [CrossRef] [PubMed]
- Câmara, C.; Urrea, C.; Schlegel, V. Pinto beans (Phaseolus vulgaris L.) as a functional food: Implications on human health. Agriculture 2013, 3, 90–111. [Google Scholar]
- Thompson, M.D.; Brick, M.A.; McGinley, J.N.; Thompson, H.J. Chemical composition and mammary cancer inhibitory activity of dry bean. Crop. Sci. 2009, 49, 179–186. [Google Scholar] [CrossRef]
- Ganesan, K.; Xu, B. Polyphenol-rich dry common beans (Phaseolus vulgaris L.) and their health benefits. Int. J. Mol. Sci. 2017, 18, 2331. [Google Scholar] [CrossRef]
- Abu-Reidah, I.M.; Arráez-Román, D.; Lozano-Sánchez, J.; Segura-Carretero, A.; Fernández-Gutiérrez, A. Phytochemical characterisation of green beans (Phaseolus vulgaris L.) by using high-performance liquid chromatography coupled with time-of-flight mass spectrometry. Phytochem. Anal. Pca 2013, 24, 105–116. [Google Scholar] [CrossRef]
- Escarpa, A.; González, M.C. Identification and quantitation of phenolics from green beans by high-performance liquid chromatography. Chromatographia 2000, 52, 33–38. [Google Scholar] [CrossRef]
- Mattila, P.; Hellström, J. Phenolic acids in potatoes, vegetables, and some of their products. J. Food Compos. Anal. 2007, 20, 152–160. [Google Scholar] [CrossRef]
- Hertog, M.G.L.; Hollman, P.C.H.; Katan, M.B. Content of potentially anticarcinogenic flavonoids of 28 vegetables and fruits commonly consumed in The Netherlands. J. Agric. Food Chem. 1992, 40, 2379–2383. [Google Scholar] [CrossRef]
- Hempel, J.; Böhm, H. Quality and quantity of prevailing flavonoid glycosides of yellow and green French beans (Phaseolus vulgaris L.). J. Agric. Food Chem. 1996, 44, 2114–2116. [Google Scholar] [CrossRef]
- Kleintop, A.E.; Myers, J.R.; Echeverria, D.; Thompson, H.J.; Brick, M.A. Total phenolic content and associated phenotypic traits in a diverse collection of snap bean cultivars. J. Am. Soc. Hortic. Sci. 2016, 141, 3–11. [Google Scholar] [CrossRef]
- Myers, J.R.; Baggett, J.R. Improvement of Snap Bean. In Common Bean Improvement in the Twenty-First Century; Singh, S.P., Ed.; Kluwer: Dordrecht, The Netherlands, 1999; pp. 289–329. [Google Scholar]
- McClean, P.E.; Bett, K.E.; Stonehouse, R.; Lee, R.; Pflieger, S.; Moghaddam, S.M.; Geffroy, V.; Miklas, P.; Mamidi, S. White seed color in common bean (Phaseolus vulgaris) results from convergent evolution in the P (pigment) gene. New Phytol. 2018, 219, 1112–1123. [Google Scholar] [CrossRef] [PubMed]
- Dickson, M.H. Breeding beans, Phaseolus vulgaris L., for improved germination under unfavorable low temperature conditions. Crop. Sci. 1971, 11, 848–850. [Google Scholar]
- Powell, A.A.; Oliveira, M.d.A.; Matthews, S. Seed vigour in cultivars of dwarf French bean (Phaseolus vulgaris) in relation to the colour of the testa. J. Agric. Sci. 1986, 106, 419–425. [Google Scholar] [CrossRef]
- Bassett, M.J. Genetics of seed coat color and pattern in common bean. In Plant Breeding Reviews; John Wiley & Sons: New York, NY, USA, 2007; Volume 28, pp. 239–315. ISBN 978-0-471-99798-6. [Google Scholar]
- Bassett, M.J.; McClean, P.E. A brief review of the genetics of partly colored seed coats in common bean. Annu. Rep. Bean Improv. Coop. 2000, 43, 99–101. [Google Scholar]
- Hagerty, C.H.; Cuesta-Marcos, A.; Cregan, P.; Song, Q.; McClean, P.; Myers, J.R. Mapping snap bean pod and color traits, in a dry bean x snap bean rcombinant inbred population. J. Am. Soc. Hortic. Sci. 2016, 141, 131–138. [Google Scholar] [CrossRef]
- McClean, P.E.; Lee, R.K.; Otto, C.; Gepts, P.; Bassett, M.J. Molecular and phenotypic mapping of genes controlling seed coat pattern and color in common bean (Phaseolus vulgaris L.). J. Hered. 2002, 93, 148–152. [Google Scholar] [CrossRef]
- Bassett, M.J.; Miklas, P.N.; Caldas, G.V.; Blair, M.W. A dominant gene for garnet brown seed coats at the Rk locus in ‘Dorado’common bean and mapping Rk to linkage group 1. Euphytica 2010, 176, 281–290. [Google Scholar] [CrossRef]
- Reinprecht, Y.; Yadegari, Z.; Perry, G.E.; Siddiqua, M.; Wright, L.C.; McClean, P.E.; Pauls, P.K. In silico comparison of genomic regions containing genes coding for enzymes and transcription factors for the phenylpropanoid pathway in Phaseolus vulgaris L. and Glycine max L. Merr. Front. Plant Sci. 2013, 4, 317. [Google Scholar] [CrossRef]
- Ates, D.; Asciogul, T.K.; Nemli, S.; Erdogmus, S.; Esiyok, D.; Tanyolac, M.B. Association mapping of days to flowering in common bean (Phaseolus vulgaris L.) revealed by DArT markers. Mol. Breed. 2018, 38, 113. [Google Scholar] [CrossRef]
- Cichy, K.A.; Porch, T.G.; Beaver, J.S.; Cregan, P.; Fourie, D.; Glahn, R.P.; Grusak, M.A.; Kamfwa, K.; Katuuramu, D.N.; McClean, P.; et al. A Phaseolus vulgaris diversity panel for Andean bean improvement. Crop. Sci. 2015, 55, 2149–2160. [Google Scholar] [CrossRef]
- Nascimento, M.; Nascimento, A.C.C.; Silva, F.F.E.; Barili, L.D.; Vale, N.M.D.; Carneiro, J.E.; Cruz, C.D.; Carneiro, P.C.S.; Serão, N.V.L. Quantile regression for genome-wide association study of flowering time-related traits in common bean. PLoS ONE 2018, 13, e0190303. [Google Scholar] [CrossRef] [PubMed]
- Kamfwa, K.; Cichy, K.A.; Kelly, J.D. Genome-wide association study of agronomic traits in common bean. Plant. Genome 2015, 8. [Google Scholar] [CrossRef]
- Moghaddam, S.M.; Mamidi, S.; Osorno, J.M.; Lee, R.; Brick, M.; Kelly, J.; Miklas, P.; Urrea, C.; Song, Q.; Cregan, P.; et al. Genome-wide association study identifies candidate loci underlying agronomic traits in a Middle American diversity panel of common bean. Plant. Genome 2016, 9. [Google Scholar] [CrossRef] [PubMed]
- Kamfwa, K.; Cichy, K.A.; Kelly, J.D. Genome-wide association analysis of symbiotic nitrogen fixation in common bean. Appl. Genet. 2015, 128, 1999–2017. [Google Scholar] [CrossRef]
- Resende, R.T.; de Resende, M.D.V.; Azevedo, C.F.; E Silva, F.F.; Melo, L.C.; Pereira, H.S.; Souza, T.L.P.; Valdisser, P.A.M.; Brondani, C.; Vianello, R.P. Genome-wide association and regional heritability mapping of plant architecture, lodging and productivity in Phaseolus vulgaris. G3 Genes Genomes Genet. 2018, 8, 2841–2854. [Google Scholar] [CrossRef]
- Cichy, K.A.; Wiesinger, J.A.; Mendoza, F.A. Genetic diversity and genome-wide association analysis of cooking time in dry bean (Phaseolus vulgaris L.). Appl. Genet. 2015, 128, 1555–1567. [Google Scholar] [CrossRef]
- Katuuramu, D.N.; Hart, J.P.; Porch, T.G.; Grusak, M.A.; Glahn, R.P.; Cichy, K.A. Genome-wide association analysis of nutritional composition-related traits and iron bioavailability in cooked dry beans (Phaseolus vulgaris L.). Mol. Breed. 2018, 38, 44. [Google Scholar] [CrossRef]
- Soltani, A.; MafiMoghaddam, S.; Walter, K.; Restrepo-Montoya, D.; Mamidi, S.; Schroder, S.; Lee, R.; McClean, P.E.; Osorno, J.M. Genetic architecture of flooding tolerance in the dry bean Middle-American diversity panel. Front. Plant. Sci. 2017, 8. [Google Scholar] [CrossRef]
- Soltani, A.; MafiMoghaddam, S.; Oladzad-Abbasabadi, A.; Walter, K.; Kearns, P.J.; Vasquez-Guzman, J.; Mamidi, S.; Lee, R.; Shade, A.L.; Jacobs, J.L.; et al. Genetic analysis of flooding tolerance in an Andean diversity panel of dry bean (Phaseolus vulgaris L.). Front. Plant. Sci. 2018, 9. [Google Scholar] [CrossRef]
- Perseguini, J.M.K.C.; Oblessuc, P.R.; Rosa, J.R.B.F.; Gomes, K.A.; Chiorato, A.F.; Carbonell, S.A.M.; Garcia, A.A.F.; Vianello, R.P.; Benchimol-Reis, L.L. Genome-wide association studies of anthracnose and angular leaf spot resistance in common bean (Phaseolus vulgaris L.). PLoS ONE 2016, 11, e0150506. [Google Scholar] [CrossRef] [PubMed]
- Tock, A.J.; Fourie, D.; Walley, P.G.; Holub, E.B.; Soler, A.; Cichy, K.A.; Pastor-Corrales, M.A.; Song, Q.; Porch, T.G.; Hart, J.P. Genome-wide linkage and association mapping of halo blight resistance in common bean to race 6 of the globally important bacterial pathogen. Front. Plant. Sci. 2017, 8, 1170. [Google Scholar] [CrossRef] [PubMed]
- Zuiderveen, G.H.; Padder, B.A.; Kamfwa, K.; Song, Q.; Kelly, J.D. Genome-wide association study of anthracnose resistance in Andean beans (Phaseolus vulgaris). PLoS ONE 2016, 11, e0156391. [Google Scholar] [CrossRef] [PubMed]
- Cai, S.; Han, Z.; Huang, Y.; Chen, Z.-H.; Zhang, G.; Dai, F. Genetic diversity of individual phenolic acids in barley and their correlation with barley malt quality. J. Agric. Food Chem. 2015, 63, 7051–7057. [Google Scholar] [CrossRef]
- Mohammadi, M.; Endelman, J.B.; Nair, S.; Chao, S.; Jones, S.S.; Muehlbauer, G.J.; Ullrich, S.E.; Baik, B.-K.; Wise, M.L.; Smith, K.P. Association mapping of grain hardness, polyphenol oxidase, total phenolics, amylose content, and β-glucan in US barley breeding germplasm. Mol. Breed. 2014, 34, 1229–1243. [Google Scholar] [CrossRef]
- Jin, L.; Lu, Y.; Xiao, P.; Sun, M.; Corke, H.; Bao, J. Genetic diversity and population structure of a diverse set of rice germplasm for association mapping. Appl. Genet. 2010, 121, 475–487. [Google Scholar] [CrossRef]
- Shao, Y.; Tang, F.; Huang, Y.; Xu, F.; Chen, Y.; Tong, C.; Chen, H.; Bao, J. Analysis of genotype x environment interactions for polyphenols and antioxidant capacity of rice by association mapping. J. Agric. Food Chem. 2014, 62, 5361–5368. [Google Scholar] [CrossRef]
- Xu, F.; Bao, J.; Kim, T.-S.; Park, Y.-J. Genome-wide association mapping of polyphenol contents and antioxidant capacity in whole-grain rice. J. Agric. Food Chem. 2016, 64, 4695–4703. [Google Scholar] [CrossRef]
- Shao, Y.; Jin, L.; Zhang, G.; Lu, Y.; Shen, Y.; Bao, J. Association mapping of grain color, phenolic content, flavonoid content and antioxidant capacity in dehulled rice. Appl. Genet. 2011, 122, 1005–1016. [Google Scholar] [CrossRef]
- Nigro, D.; Laddomada, B.; Mita, G.; Blanco, E.; Colasuonno, P.; Simeone, R.; Gadaleta, A.; Pasqualone, A.; Blanco, A. Genome-wide association mapping of phenolic acids in tetraploid wheats. J. Cereal Sci. 2017, 75, 25–34. [Google Scholar] [CrossRef]
- Rhodes, D.H.; Hoffmann, L., Jr.; Rooney, W.L.; Ramu, P.; Morris, G.P.; Kresovich, S. Genome-wide association study of grain polyphenol concentrations in global sorghum (Sorghum bicolor (L.) Moench) germplasm. J. Agric. Food Chem. 2014, 62, 10916–10927. [Google Scholar] [CrossRef] [PubMed]
- Wen, W.; Li, D.; Li, X.; Gao, Y.; Li, W.; Li, H.; Liu, J.; Liu, H.; Chen, W.; Luo, J. Metabolome-based genome-wide association study of maize kernel leads to novel biochemical insights. Nat. Commun. 2014, 5, 3438. [Google Scholar] [CrossRef] [PubMed]
- Ruggieri, V.; Francese, G.; Sacco, A.; D’Alessandro, A.; Rigano, M.M.; Parisi, M.; Milone, M.; Cardi, T.; Mennella, G.; Barone, A. An association mapping approach to identify favourable alleles for tomato fruit quality breeding. BMC Plant. Biol. 2014, 14, 337. [Google Scholar] [CrossRef] [PubMed]
- Rezaeizad, A.; Wittkop, B.; Snowdon, R.; Hasan, M.; Mohammadi, V.; Zali, A.; Friedt, W. Identification of QTLs for phenolic compounds in oilseed rape (Brassica napus L.) by association mapping using SSR markers. Euphytica 2011, 177, 335–342. [Google Scholar] [CrossRef]
- Kaya, H.B.; Cetin, O.; Kaya, H.S.; Sahin, M.; Sefer, F.; Tanyolac, B. Association mapping in Turkish olive cultivars revealed significant markers related to some important agronomic traits. Biochem. Genet. 2016, 54, 506–533. [Google Scholar] [CrossRef]
- Singleton, V.L.; Rossi, J.A. Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am. J. Enol. Vitic. 1965, 16, 144–158. [Google Scholar]
- CIE. CIE Colorimetry—Part 4: 1976 L*a*b* Colour Space. Available online: http://cie.co.at/index.php?i_ca_id=485 (accessed on 1 December 2018).
- Wallace, L.; Arkwazee, H.; Vining, K.; Myers, J. Genetic diversity within snap beans and their relation to dry beans. Genes 2018, 9, 587. [Google Scholar] [CrossRef]
- Liu, X.; Huang, M.; Fan, B.; Buckler, E.S.; Zhang, Z. Iterative usage of fixed and random effect models for powerful and efficient genome-wide association studies. PLoS Genet. 2016, 12, e1005767. [Google Scholar] [CrossRef]
- Li, S. Transcriptional control of flavonoid biosynthesis. Plant. Signal. Behav. 2014, 9, e27522. [Google Scholar] [CrossRef]
- Xu, W.; Dubos, C.; Lepiniec, L. Transcriptional control of flavonoid biosynthesis by MYB–bHLH–WDR complexes. Trends Plant. Sci. 2015, 20, 176–185. [Google Scholar] [CrossRef]
- Chen, L.; Hu, B.; Qin, Y.; Hu, G.; Zhao, J. Advance of the negative regulation of anthocyanin biosynthesis by MYB transcription factors. Plant. Physiol. Biochem. 2019, 136, 178–187. [Google Scholar] [CrossRef] [PubMed]
- Allan, A.C.; Espley, R.V. MYBs drive novel consumer traits in fruits and vegetables. Trends Plant. Sci. 2018, 23, 693–705. [Google Scholar] [CrossRef] [PubMed]
- Ma, D.; Constabel, C.P. MYB repressors as regulators of phenylpropanoid metabolism in plants. Trends Plant. Sci. 2019. [Google Scholar] [CrossRef] [PubMed]
- Cheynier, V.; Comte, G.; Davies, K.M.; Lattanzio, V.; Martens, S. Plant phenolics: Recent advances on their biosynthesis, genetics, and ecophysiology. Plant. Physiol. Biochem. 2013, 72, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Schmutz, J.; McClean, P.E.; Mamidi, S.; Wu, G.A.; Cannon, S.B.; Grimwood, J.; Jenkins, J.; Shu, S.; Song, Q.; Chavarro, C. A reference genome for common bean and genome-wide analysis of dual domestications. Nat. Genet. 2014, 46, 707. [Google Scholar] [CrossRef]
- Flint-Garcia, S.A.; Thornsberry, J.M.; Buckler, E.S. Structure of linkage disequilibrium in plants. Annu. Rev. Plant. Biol. 2003, 54, 357–374. [Google Scholar] [CrossRef]
- Diniz, A.L.; Giordani, W.; Costa, Z.P.; Margarido, G.R.; Perseguini, J.M.K.; Benchimol-Reis, L.L.; Chiorato, A.F.; Garcia, A.A.F.; Vieira, M.L.C. Evidence for strong kinship influence on the extent of linkage disequilibrium in cultivated common beans. Genes 2019, 10, 5. [Google Scholar] [CrossRef]
- Blair, M.W.; Cortés, A.J.; Farmer, A.D.; Huang, W.; Ambachew, D.; Penmetsa, R.V.; Carrasquilla-Garcia, N.; Assefa, T.; Cannon, S.B. Uneven recombination rate and linkage disequilibrium across a reference SNP map for common bean (Phaseolus vulgaris L.). PLoS ONE 2018, 13, e0189597. [Google Scholar] [CrossRef]
- Valdisser, P.A.; Pereira, W.J.; Almeida Filho, J.E.; Müller, B.S.; Coelho, G.R.; de Menezes, I.P.; Vianna, J.P.; Zucchi, M.I.; Lanna, A.C.; Coelho, A.S. In-depth genome characterization of a Brazilian common bean core collection using DArTseq high-density SNP genotyping. BMC Genom. 2017, 18, 423. [Google Scholar] [CrossRef]
- Myers, J.R.; Davis, J.; Arkwazee, H.; Wallace, L.; Lee, R.; Moghaddam, S.M.; McClean, P. Why wax beans lack carotenoids. Annu. Rep. Bean Improv. Coop. 2018, 61, 29–30. [Google Scholar]
- Barkan, A.; Small, I. Pentatricopeptide repeat proteins in plants. Annu. Rev. Plant. Biol. 2014, 65, 415–442. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, A.; Niwa, Y.; Goto, S.; Ogawa, T.; Shimizu, M.; Suzuki, A.; Kobayashi, K.; Kobayashi, H. bHLH106 integrates functions of multiple genes through their G-box to confer salt tolerance on Arabidopsis. PLoS ONE 2015, 10, e0126872. [Google Scholar] [CrossRef] [PubMed]
- Ma, Z.-H.; Li, W.-F.; Mao, J.; Li, W.; Zuo, C.-W.; Zhao, X.; Dawuda, M.M.; Shi, X.-Y.; Chen, B.-H. Synthesis of light-inducible and light-independent anthocyanins regulated by specific genes in grape ‘Marselan’ (V. vinifera L.). PeerJ. 2019, 7, e6521. [Google Scholar] [CrossRef] [PubMed]
- Berim, A.; Gang, D.R. The roles of a flavone-6-hydroxylase and 7-O-demethylation in the flavone biosynthetic network of sweet basil. J. Biol. Chem. 2013, 288, 1795–1805. [Google Scholar] [CrossRef]
- Yuan, T.; Fujioka, S.; Takatsuto, S.; Matsumoto, S.; Gou, X.; He, K.; Russell, S.D.; Li, J. BEN1, a gene encoding a dihydroflavonol 4-reductase (DFR)-like protein, regulates the levels of brassinosteroids in Arabidopsis thaliana. Plant. J. 2007, 51, 220–233. [Google Scholar] [CrossRef] [PubMed]
- Zheng, S.; Lin, Y.; Lai, Z.; Lin, J. Isolation and characterisation of a MYB transcription factor associated with epigallocatechin-3-gallate biosynthesis in Camellia sinensis L. J. Hortic. Sci. Biotechnol. 2019, 94, 41–48. [Google Scholar] [CrossRef]
- String. String Database. Available online: https://string-db.org/cgi/network.pl?taskId=RHIDbolr9GEA (accessed on 12 July 2019).
| Gene 2 | Chrom. 3 | Marker/Gene Model | Linkage 4 | Start | End | Length 5 |
|---|---|---|---|---|---|---|
| cM | bp | |||||
| B | 2 | ss715645998 | 0.0 | 48,634,623 | 48,634,743 | 121 |
| Z | 3 | OAM10560 | 1.4 | 31,467,887 | 31,467,909 | 22 |
| G | 4 | OAP7850 | 0.0 | 30,171,480 | 30,172,322 | 842 |
| G | 4 | OAP31400 | 0.0 | 30,172,301 | 30,172,322 | 21 |
| G | 4 | OU14900 | 0.0 | 31,093,347 | 31,093,369 | 22 |
| V | 6 | OD12800 | 0.0 | 9,288,398 | 9,288,419 | 21 |
| P | 7 | Phvul.007G171333 + Phvul.007G171466 | − | 28,752,132 | 28,774,743 | 22,611 |
| Gy | 8 | OW17600 | 1.6 | 3,230,983 | 3,231,005 | 22 |
| C | 8 | OAP2700 | 4.9 | 9,694,328 | 9,695,007 | 679 |
| T | 9 | OM19400 | 1.4 | 11,731,567 | 11,731,944 | 377 |
| Ana | 10 | OM9200 | 5.4 | 11,563,621 | 11,563,800 | 179 |
| L | 10 | OL4525 | 1.2 | 41,443,673 | 41,443,694 | 21 |
| QTN Designation 1 | Method | ss ID No. 2 | Chrom. 3 | Position (bp) | Prob. Value | -Log10 P | MAF 4 | SNP 5 |
|---|---|---|---|---|---|---|---|---|
| TPC1.1 | FarmCPU | 715648077 | 1 | 33,470,635 | 7.952E-09 | 8.10 | 0.05 | C-T |
| TPC1.2 | FarmCPU | 715646871 | 1 | 42,698,218 | 1.357E-10 | 9.87 | 0.07 | T-C |
| TPC3.1 | GAPIT | 715646623 | 3 | 40,370,083 | 2.448E-07 | 6.61 | 0.05 | T-C |
| TPC4.1 | FarmCPU | 715647815 | 4 | 2,190,413 | 2.912E-11 | 10.54 | 0.07 | G-A |
| TPC7.1 | FarmCPU | 715647649 | 7 | 28,427,257 | 6.783E-08 | 7.17 | 0.29 | T-C |
| TPC9.1 | GAPIT | 715647263 | 9 | 12,159,056 | 2.614E-06 | 5.58 | 0.09 | C-T |
| TPC9.2 | GAPIT | 715646560 | 9 | 12,776,010 | 2.872E-07 | 6.54 | 0.08 | A-G |
| TPC9.3 | FarmCPU | 715646559 | 9 | 13,053,019 | 7.182E-23 | 22.14 | 0.08 | G-A |
| TPC10.1 | FarmCPU | 715639693 | 10 | 11,130,696 | 5.660E-12 | 11.25 | 0.06 | T-C |
| TPC10.2 | FarmCPU | 715643755 | 10 | 23,719,802 | 2.746E-08 | 7.56 | 0.06 | C-T |
| TPC11.1 | FarmCPU | 715650328 | 11 | 52,968,750 | 3.222E-07 | 6.49 | 0.05 | T-C |
| QTN Designation 1 | Method | ss ID No. 2 | Chrom. 3 | Position (bp) | Prob. Value | -Log10 P | MAF 4 | SNP 5 |
|---|---|---|---|---|---|---|---|---|
| A*2.1 | GAPIT | 715646673 | 2 | 542,087 | 2.684E-06 | 5.57 | 0.11 | G-T |
| L*2.1 | GAPIT | 715639371 | 2 | 729,615 | 1.324E-06 | 5.88 | 0.18 | C-A |
| L*2.1 | GAPIT | 715639372 | 2 | 766,293 | 1.324E-06 | 5.88 | 0.18 | C-T |
| B*3.1 | FarmCPU | 715649460 | 3 | 3,463,964 | 2.601E-09 | 8.58 | 0.07 | T-C |
| B*3.2 | GAPIT | 715646623 | 3 | 40,370,083 | 1.790E-07 | 6.75 | 0.05 | T-C |
| B*5.1 | FarmCPU | 715645117 | 5 | 887,360 | 3.322E-06 | 5.48 | 0.14 | A-G |
| QTN Designation 1 | Method | ss ID No.2 | Chrom. 3 | Position (bp) | Prob. Value | -Log10 P | MAF 4 | SNP 5 |
|---|---|---|---|---|---|---|---|---|
| FC1.1 | FarmCPU | 715648402 | 1 | 8,955,838 | 3.088E-21 | 20.51 | 0.06 | A-C |
| FC1.2 | FarmCPU | 715645902 | 1 | 48,430,158 | 3.215E-10 | 9.49 | 0.09 | T-C |
| FC3.1 | FarmCPU | 715646620 | 3 | 40,621,263 | 1.087E-08 | 7.96 | 0.07 | T-C |
| FC3.1 | FarmCPU | 715646619 | 3 | 40,634,185 | 1.087E-08 | 7.96 | 0.07 | A-G |
| FC3.2 | FarmCPU | 715650182 | 3 | 40,901,620 | 1.087E-08 | 7.96 | 0.07 | T-C |
| FC3.2 | FarmCPU | 715650183 | 3 | 40,922,819 | 1.087E-08 | 7.96 | 0.07 | A-C |
| FC3.3 | FarmCPU | 715639426 | 3 | 41,081,362 | 1.087E-08 | 7.96 | 0.07 | C-T |
| FC3.3 | FarmCPU | 715639423 | 3 | 41,169,500 | 1.087E-08 | 7.96 | 0.07 | T-G |
| FC3.4 | FarmCPU | 715640920 | 3 | 43,174,196 | 1.087E-08 | 7.96 | 0.07 | A-C |
| FC9.1 | GAPIT | 715647263 | 9 | 12,159,056 | 2.326E-07 | 6.63 | 0.09 | C-T |
| FC9.2 | GAPIT | 715646560 | 9 | 12,776,010 | 7.518E-08 | 7.12 | 0.08 | A-G |
| FC9.3 | FarmCPU | 715646559 | 9 | 13,053,019 | 8.124E-21 | 20.09 | 0.08 | G-A |
| FC9.3 | GAPIT | 715648638 | 9 | 13,339,943 | 1.028E-06 | 5.99 | 0.19 | G-A |
| QTN Designation | Gene Models ± 350 kb of QTN | Annotated Gene Models | Putative Phenolics Related Gene Models | Unknown Gene Models |
|---|---|---|---|---|
| no. | ||||
| TPC1.1 | 19 | 18 | 1 | 1 |
| TPC1.2 | 61 | 55 | 0 | 6 |
| TPC3.1 | 67 | 61 | 0 | 6 |
| TPC4.1 | 67 | 61 | 10 | 6 |
| TPC7.1 | 56 | 48 | 2 | 8 |
| TPC9.1 | 65 | 55 | 2 | 10 |
| TPC9.2 | 61 | 45 | 1 | 16 |
| TPC9.3 | 65 | 50 | 1 | 15 |
| TPC10.1 | 13 | 13 | 0 | 0 |
| TPC10.2 | 19 | 18 | 0 | 1 |
| TPC11.1 | 52 | 40 | 2 | 12 |
| QTN 1 | SNP Position | Distance 2 | P. vulgaris Gene Model | Start | End | A. thaliana Gene Model | Gene Abbreviation | Gene Function |
|---|---|---|---|---|---|---|---|---|
| bp | bp | |||||||
| TPC1.1 | 33,470,635 | 83,056 | Phvul.001G121200 | 33,553,691 | 33,555,719 | AT2G41130.1 | BHLH106, STC8 | Basic helix-loop-helix (bHLH) DNA-binding superfamily protein |
| TPC1.2 | 42,698,218 | -- | No candidates | -- | -- | -- | -- | -- |
| TPC3.1 | 40,370,083 | -- | No candidates | -- | -- | -- | -- | -- |
| TPC4.1 | 2,190,413 | 269,553 | Phvul.004G021100 | 2,459,966 | 2,462,674 | AT4G31940.1 | CYP82C4 | Cytochrome P450, family 82, subfamily C, polypeptide 4 |
| 275,490 | Phvul.004G021200 | 2,465,903 | 2,467,865 | AT4G31940.1 | CYP82C4 | Cytochrome P450, family 82, subfamily C, polypeptide 4 | ||
| 283,486 | Phvul.004G021300 | 2,473,899 | 2,476,387 | AT4G31940.1 | CYP82C4 | Cytochrome P450, family 82, subfamily C, polypeptide 4 | ||
| 289,437 | Phvul.004G021400 | 2,479,850 | 2,482,645 | AT4G31940.1 | CYP82C4 | Cytochrome P450, family 82, subfamily C, polypeptide 4 | ||
| 298,067 | Phvul.004G021500 | 2,488,480 | 2,491,886 | AT4G31940.1 | CYP82C4 | Cytochrome P450, family 82, subfamily C, polypeptide 4 | ||
| 304,827 | Phvul.004G021600 | 2,495,240 | 2,497,544 | AT4G31940.1 | CYP82C4 | Cytochrome P450, family 82, subfamily C, polypeptide 4 | ||
| 312,938 | Phvul.004G021700 | 2,503,351 | 2,505,988 | AT4G31970.1 | CYP82C2 | Cytochrome P450, family 82, subfamily C, polypeptide 2 | ||
| 321,346 | Phvul.004G021800 | 2,511,759 | 2,513,935 | AT4G31970.1 | CYP82C2 | Cytochrome P450, family 82, subfamily C, polypeptide 2 | ||
| 325,444 | Phvul.004G021900 | 2,515,857 | 2,518,683 | AT4G31940.1 | CYP82C4 | Cytochrome P450, family 82, subfamily C, polypeptide 4 | ||
| 338,085 | Phvul.004G022000 | 2,528,498 | 2,531,156 | AT4G31940.1 | CYP82C4 | Cytochrome P450, family 82, subfamily C, polypeptide 4 | ||
| TPC7.1 | 28,427,257 | 324,875 | Phvul.007G171333 | 28,752,132 | 28,766,155 | AT4G09820.1 | BHLH42, TT8 | Basic helix-loop-helix (bHLH) DNA-binding superfamily protein |
| 345,156 | Phvul.007G171466 | 28,772,413 | 28,774,743 | -- | -- | Basic helix-loop-helix (bHLH) DNA-binding superfamily protein | ||
| TPC7.1 | 28,433,796 | 318,336 | Phvul.007G171333 | 28,752,132 | 28,766,155 | AT4G09820.1 | BHLH42, TT8 | Basic helix-loop-helix (bHLH) DNA-binding superfamily protein |
| 338,617 | Phvul.007G171466 | 28,772,413 | 28,774,743 | -- | -- | Basic helix-loop-helix (bHLH) DNA-binding superfamily protein | ||
| TPC9.1 | 12,159,056 | 30,719 | Phvul.009G069401 | 12,122,701 | 12,128,337 | AT5G19440.1 | -- | NAD(P)-binding Rossmann-fold superfamily protein |
| 28,423 | Phvul.009G069500 | 12,129,317 | 12,130,633 | AT1G51410.1 | -- | NAD(P)-binding Rossmann-fold superfamily protein | ||
| TPC9.2 | 12,776,010 | 242,880 | Phvul.009G077300 | 13,018,890 | 13,020,479 | AT5G56840.1 | -- | myb-like transcription factor family protein |
| TPC9.3 | 13,053,019 | 32,540 | Phvul.009G077300 | 13,018,890 | 13,020,479 | AT5G56840.1 | -- | myb-like transcription factor family protein |
| TPC10.1 | 11,130,696 | -- | No candidates | -- | -- | -- | -- | -- |
| TPC10.2 | 23,719,802 | -- | No candidates | -- | -- | -- | -- | -- |
| TPC11.1 | 52,968,750 | 287,108 | Phvul.011G212600 | 53,255,858 | 53,257,674 | AT2G45400.1 | BEN1 | NAD(P)-binding Rossmann-fold superfamily protein |
| 291,448 | Phvul.011G212800 | 53,260,198 | 53,262,370 | AT2G45400.1 | BEN1 | NAD(P)-binding Rossmann-fold superfamily protein | ||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Myers, J.R.; Wallace, L.T.; Mafi Moghaddam, S.; Kleintop, A.E.; Echeverria, D.; Thompson, H.J.; Brick, M.A.; Lee, R.; McClean, P.E. Improving the Health Benefits of Snap Bean: Genome-Wide Association Studies of Total Phenolic Content. Nutrients 2019, 11, 2509. https://doi.org/10.3390/nu11102509
Myers JR, Wallace LT, Mafi Moghaddam S, Kleintop AE, Echeverria D, Thompson HJ, Brick MA, Lee R, McClean PE. Improving the Health Benefits of Snap Bean: Genome-Wide Association Studies of Total Phenolic Content. Nutrients. 2019; 11(10):2509. https://doi.org/10.3390/nu11102509
Chicago/Turabian StyleMyers, James R., Lyle T. Wallace, Samira Mafi Moghaddam, Adrienne E. Kleintop, Dimas Echeverria, Henry J. Thompson, Mark A. Brick, Rian Lee, and Phillip E. McClean. 2019. "Improving the Health Benefits of Snap Bean: Genome-Wide Association Studies of Total Phenolic Content" Nutrients 11, no. 10: 2509. https://doi.org/10.3390/nu11102509
APA StyleMyers, J. R., Wallace, L. T., Mafi Moghaddam, S., Kleintop, A. E., Echeverria, D., Thompson, H. J., Brick, M. A., Lee, R., & McClean, P. E. (2019). Improving the Health Benefits of Snap Bean: Genome-Wide Association Studies of Total Phenolic Content. Nutrients, 11(10), 2509. https://doi.org/10.3390/nu11102509






