Salmon Fillet Intake Led to Higher Serum Triacylglycerol in Obese Zucker Fa/Fa Rats But Not in Normolipidemic Long-Evans Rats
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethical Statement
2.2. Design
2.3. Diets
2.4. Euthanasia and Sampling
2.5. Analyses of Diets
2.6. Analyses in Serum
2.7. Analyses of Fatty Acids in Serum Lipids
2.8. Statistical Analysis
3. Results
3.1. Bodyweight, Growth, White Adipose Tissue Weight, and Energy and Fat Intake
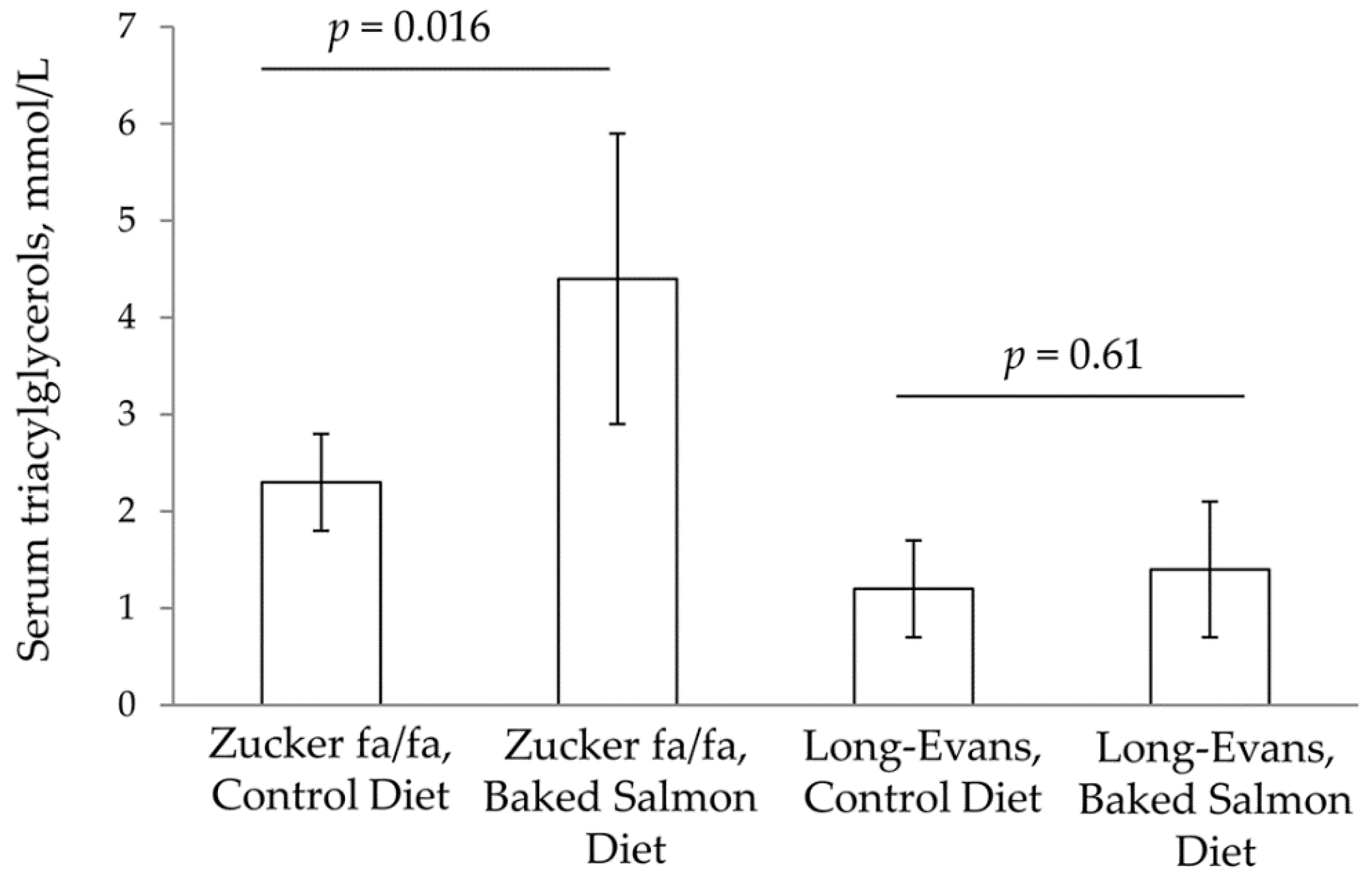
3.2. Serum Analyses
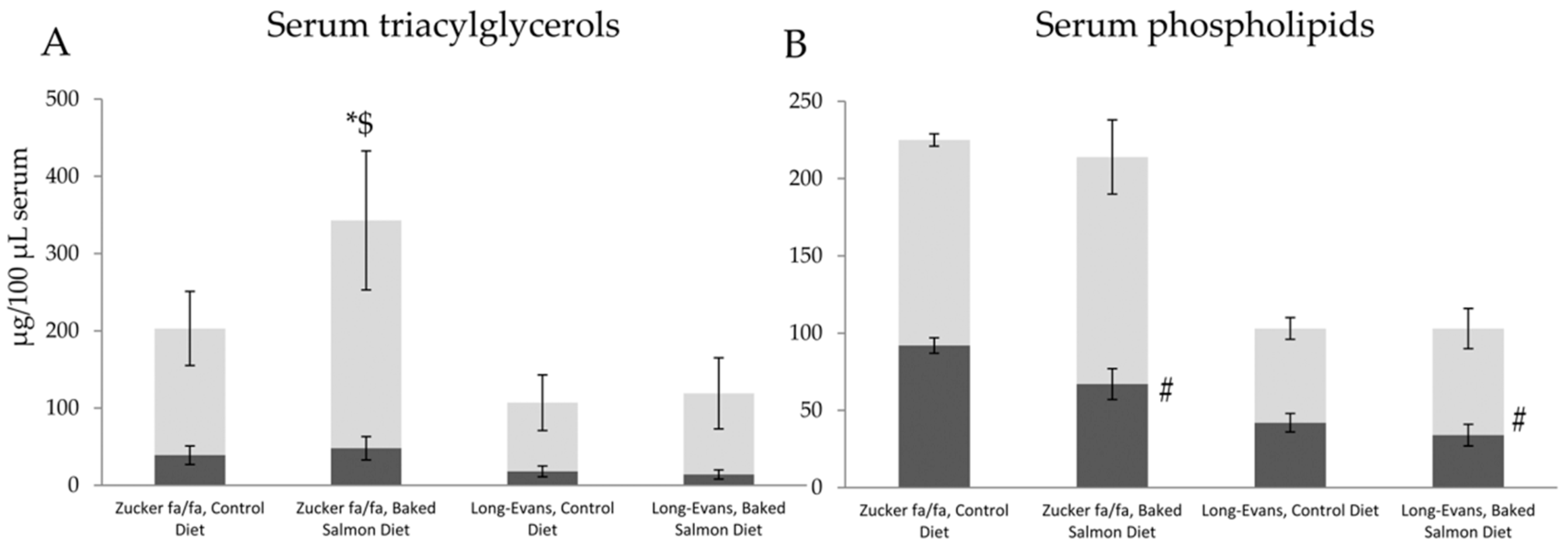
3.3. Fatty Acids in Triacylglycerols and Phospholipids in Serum
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Bansal, S.; Buring, J.E.; Rifai, N.; Mora, S.; Sacks, F.M.; Ridker, P.M. Fasting compared with nonfasting triglycerides and risk of cardiovascular events in women. JAMA 2007, 298, 309–316. [Google Scholar] [PubMed]
- Perez-Martinez, P.; Perez-Jimenez, F.; Lopez-Miranda, J. n-3 PUFA and lipotoxicity. Biochim. Biophys. Acta 2010, 1801, 362–366. [Google Scholar] [CrossRef] [PubMed]
- Telle-Hansen, V.H.; Larsen, L.N.; Hostmark, A.T.; Molin, M.; Dahl, L.; Almendingen, K.; Ulven, S.M. Daily intake of cod or salmon for 2 weeks decreases the 18:1n-9/18:0 ratio and serum triacylglycerols in healthy subjects. Lipids 2012, 47, 151–160. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, E.B.; Skou, H.A.; Christensen, J.H.; Dyerberg, J. N-3 fatty acids from fish and coronary artery disease: implications for public health. Public Health Nutr. 2000, 3, 91–98. [Google Scholar] [PubMed]
- Mori, T.A.; Bao, D.Q.; Burke, V.; Puddey, I.B.; Watts, G.F.; Beilin, L.J. Dietary fish as a major component of a weight-loss diet: effect on serum lipids, glucose, and insulin metabolism in overweight hypertensive subjects. Am. J. Clin. Nutr. 1999, 70, 817–825. [Google Scholar] [CrossRef] [PubMed]
- Gunnarsdottir, I.; Tomasson, H.; Kiely, M.; Martinez, J.A.; Bandarra, N.M.; Morais, M.G.; Thorsdottir, I. Inclusion of fish or fish oil in weight-loss diets for young adults: effects on blood lipids. Int. J. Obes. 2008, 32, 1105–1112. [Google Scholar] [CrossRef] [PubMed]
- Lindqvist, H.M.; Langkilde, A.M.; Undeland, I.; Sandberg, A.S. Herring (Clupea harengus) intake influences lipoproteins but not inflammatory and oxidation markers in overweight men. Br. J. Nutr. 2009, 101, 383–390. [Google Scholar] [CrossRef] [PubMed]
- Tinker, L.F.; Parks, E.J.; Behr, S.R.; Schneeman, B.O.; Davis, P.A. (n−3) fatty acid supplementation in moderately hypertriglyceridemic adults changes postprandial lipid and apolipoprotein B responses to a standardized test meal. J. Nutr. 1999, 129, 1126–1134. [Google Scholar] [CrossRef] [PubMed]
- Nettleton, J.A.; Katz, R. n-3 long-chain polyunsaturated fatty acids in type 2 diabetes: a review. J. Am. Diet. Assoc. 2005, 105, 428–440. [Google Scholar] [PubMed]
- Lara, J.J.; Economou, M.; Wallace, A.M.; Rumley, A.; Lowe, G.; Slater, C.; Caslake, M.; Sattar, N.; Lean, M.E. Benefits of salmon eating on traditional and novel vascular risk factors in young, non-obese healthy subjects. Atherosclerosis 2007, 193, 213–221. [Google Scholar] [CrossRef] [PubMed]
- Eslick, G.D.; Howe, P.R.; Smith, C.; Priest, R.; Bensoussan, A. Benefits of fish oil supplementation in hyperlipidemia: a systematic review and meta-analysis. Int. J. Cardiol. 2009, 136, 4–16. [Google Scholar] [CrossRef] [PubMed]
- Whelton, S.P.; He, J.; Whelton, P.K.; Muntner, P. Meta-analysis of observational studies on fish intake and coronary heart disease. Am. J. Cardiol. 2004, 93, 1119–1123. [Google Scholar] [CrossRef] [PubMed]
- Harris, W.S. Fish oils and plasma lipid and lipoprotein metabolism in humans: a critical review. J. Lipid Res. 1989, 30, 785–807. [Google Scholar] [PubMed]
- Harris, W.S. n-3 fatty acids and serum lipoproteins: human studies. Am. J. Clin. Nutr. 1997, 65, 1645S–1654S. [Google Scholar] [CrossRef] [PubMed]
- Smith, K.M.; Barraj, L.M.; Kantor, M.; Sahyoun, N.R. Relationship between fish intake, n-3 fatty acids, mercury and risk markers of CHD (National Health and Nutrition Examination Survey 1999–2002). Public Health Nutr. 2009, 12, 1261–1269. [Google Scholar] [CrossRef] [PubMed]
- Balk, E.M.; Lichtenstein, A.H.; Chung, M.; Kupelnick, B.; Chew, P.; Lau, J. Effects of omega-3 fatty acids on serum markers of cardiovascular disease risk: a systematic review. Atherosclerosis 2006, 189, 19–30. [Google Scholar] [CrossRef] [PubMed]
- Siscovick, D.S.; Barringer, T.A.; Fretts, A.M.; Wu, J.H.; Lichtenstein, A.H.; Costello, R.B.; Kris-Etherton, P.M.; Jacobson, T.A.; Engler, M.B.; Alger, H.M.; et al. Omega-3 Polyunsaturated Fatty Acid (Fish Oil) Supplementation and the Prevention of Clinical Cardiovascular Disease: A Science Advisory From the American Heart Association. Circulation 2017, 135, e867–e884. [Google Scholar] [CrossRef] [PubMed]
- Mohan, P.F.; Phillips, F.C.; Cleary, M.P. Metabolic effects of coconut, safflower, or menhaden oil feeding in lean and obese Zucker rats. Br. J. Nutr. 1991, 66, 285–299. [Google Scholar] [CrossRef] [PubMed]
- Kromhout, D.; de Goede, J. Update on cardiometabolic health effects of omega-3 fatty acids. Curr. Opin. Lipidol. 2014, 25, 85–90. [Google Scholar] [CrossRef] [PubMed]
- Glauber, H.; Wallace, P.; Griver, K.; Brechtel, G. Adverse metabolic effect of omega-3 fatty acids in non-insulin-dependent diabetes mellitus. Ann. Intern. Med. 1988, 108, 663–668. [Google Scholar] [CrossRef] [PubMed]
- Popp-Snijders, C.; Schouten, J.A.; Heine, R.J.; van der Meer, J.; van der Veen, E.A. Dietary supplementation of omega-3 polyunsaturated fatty acids improves insulin sensitivity in non-insulin-dependent diabetes. Diabetes Res. 1987, 4, 141–147. [Google Scholar] [PubMed]
- Fredrickson, D.S.; Lees, R.S. A System for Phenotyping Hyperlipoproteinemia. Circulation 1965, 31, 321–327. [Google Scholar] [CrossRef] [PubMed]
- De Artinano, A.A.; Castro, M.M. Experimental rat models to study the metabolic syndrome. Br. J. Nutr. 2009, 102, 1246–1253. [Google Scholar] [CrossRef] [PubMed]
- Boulange, A.; Planche, E.; de Gasquet, P. Onset and development of hypertriglyceridemia in the Zucker rat (fa/fa). Metabolism 1981, 30, 1045–1052. [Google Scholar] [CrossRef]
- Redgrave, T.G. Catabolism of chylomicron triacylglycerol and cholesteryl ester in genetically obese rats. J. Lipid Res. 1977, 18, 604–612. [Google Scholar] [PubMed]
- Vikoren, L.A.; Drotningsvik, A.; Bergseth, M.T.; Mjos, S.A.; Mola, N.; Leh, S.; Mellgren, G.; Gudbrandsen, O.A. Effects of baked and raw salmon fillet on lipids and n−3 PUFAs in serum and tissues in Zucker fa/fa rats. Food Nutr. Res. 2017, 61, 1333395. [Google Scholar] [CrossRef] [PubMed]
- Vikoren, L.A.; Drotningsvik, A.; Mwakimonga, A.; Leh, S.; Mellgren, G.; Gudbrandsen, O.A. Diets containing salmon fillet delay development of high blood pressure and hyperfusion damage in kidneys in obese Zucker fa/fa rats. J. Am. Soc. Hypertens. 2018, 12, 294–302. [Google Scholar] [CrossRef] [PubMed]
- Reeves, P.G.; Nielsen, F.H.; Fahey, G.C., Jr. AIN-93 purified diets for laboratory rodents: final report of the American Institute of Nutrition ad hoc writing committee on the reformulation of the AIN-76A rodent diet. J. Nutr. 1993, 123, 1939–1951. [Google Scholar] [CrossRef] [PubMed]
- Reeves, P.G. AIN-93 Purified Diets for the Study of Trace Element Metabolism in Rodents, in Trace Elements in Laboratory Rodents; Watson, R.R., Ed.; CRC Press Inc.: Boca Raton, FL, USA, 1996; pp. 3–37. [Google Scholar]
- Sissener, N.H.; Waagbø, R.; Rosenlund, G.; Tvenning, L.; Susort, S.; Lea, T.B.; Oaland, Ø.; Chen, L.; Breck, O. Reduced n−3 long chain fatty acid levels in feed for Atlantic salmon (Salmo salar L.) do not reduce growth, robustness or product quality through an entire full scale commercial production cycle in seawater. Aquaculture 2016, 464, 236–245. [Google Scholar]
- Bligh, E.G.; Dyer, W.J. A rapid method of total lipid extraction and purification. Can. J. Biochem. Physiol. 1959, 37, 911–917. [Google Scholar] [CrossRef] [PubMed]
- Meier, S.; Mjos, S.A.; Joensen, H.; Grahl-Nielsen, O. Validation of a one-step extraction/methylation method for determination of fatty acids and cholesterol in marine tissues. J. Chromatogr. A 2006, 1104, 291–298. [Google Scholar] [CrossRef] [PubMed]
- Sciotto, C.; Mjos, S.A. Trans isomers of EPA and DHA in omega-3 products on the European market. Lipids 2012, 47, 659–667. [Google Scholar] [CrossRef] [PubMed]
- Wasta, Z.; Mjos, S.A. A database of chromatographic properties and mass spectra of fatty acid methyl esters from omega-3 products. J. Chromatogr. A 2013, 1299, 94–102. [Google Scholar] [CrossRef] [PubMed]
- Drotningsvik, A.; Mjos, S.A.; Hogoy, I.; Remman, T.; Gudbrandsen, O.A. A low dietary intake of cod protein is sufficient to increase growth, improve serum and tissue fatty acid compositions, and lower serum postprandial glucose and fasting non-esterified fatty acid concentrations in obese Zucker fa/fa rats. Eur. J. Nutr. 2015, 54, 1151–1160. [Google Scholar] [CrossRef] [PubMed]
- Mangold, H.K. Aliphatic Lipids in Thin-layer Chromatography, in A Laboratory Handbook, 2nd ed.; Stahl, E., Ed.; Springer: Berlin, Germany, 1969; pp. 363–421. [Google Scholar]
- Batetta, B.; Griinari, M.; Carta, G.; Murru, E.; Ligresti, A.; Cordeddu, L.; Giordano, E.; Sanna, F.; Bisogno, T.; Uda, S.; et al. Endocannabinoids may mediate the ability of (n-3) fatty acids to reduce ectopic fat and inflammatory mediators in obese Zucker rats. J. Nutr. 2009, 139, 1495–1501. [Google Scholar] [CrossRef] [PubMed]
- Shearer, G.C.; Savinova, O.V.; Harris, W.S. Fish oil—How does it reduce plasma triglycerides? Biochim. Biophys. Acta 2012, 1821, 843–851. [Google Scholar] [CrossRef] [PubMed]
- Park, Y.; Harris, W.S. Omega-3 fatty acid supplementation accelerates chylomicron triglyceride clearance. J. Lipid Res. 2003, 44, 455–463. [Google Scholar] [CrossRef] [PubMed]
- Carlsson, M.; Wessman, Y.; Almgren, P.; Groop, L. High levels of nonesterified fatty acids are associated with increased familial risk of cardiovascular disease. Arterioscler. Thromb. Vasc. Biol. 2000, 20, 1588–1594. [Google Scholar] [CrossRef] [PubMed]
- Sparks, J.D.; Sparks, C.E. Insulin modulation of hepatic synthesis and secretion of apolipoprotein B by rat hepatocytes. J. Biol. Chem. 1990, 265, 8854–8862. [Google Scholar] [PubMed]

| g/kg Diet | Control Diet | Baked Salmon Diet |
|---|---|---|
| Casein protein 1 | 216.2 | 162.2 |
| Freeze dried baked salmon 2 | - | 100.0 |
| Cornstarch | 512.0 | 466.0 |
| Sucrose | 90.0 | 90.0 |
| Cellulose | 50.0 | 50.0 |
| Soybean Oil | 70.0 | 70.0 |
| t-Butylhydroquinone | 0.014 | 0.014 |
| Mineral Mix (AIN-93-MX) | 35.0 | 35.0 |
| Vitamin Mix (AIN-93-VX) | 10.0 | 10.0 |
| L-Methionine | 1.60 | 1.60 |
| L-Cystine | 3.0 | 3.0 |
| Choline Bitartrate 3 | 2.5 | 2.5 |
| Growth and Maintenance Supplement 4 | 10.0 | 10.0 |
| g/kg Diet | Control Diet | Baked Salmon Diet |
|---|---|---|
| 16:0 | 7.5 | 9.6 |
| 18:0 | 2.5 | 3.0 |
| 18:1n−7 | 0.87 | 1.6 |
| 18:1n−9 | 13.9 | 24.8 |
| 18:2n−6 | 32.7 | 34.5 |
| 18:3n−3 | 3.9 | 5.0 |
| 20:5n−3 | ND | 0.53 |
| 22:5n−3 | ND | 0.22 |
| 22:6n−3 | ND | 1.0 |
| Zucker fa/fa Rats | Long-Evans Rats | |||||
|---|---|---|---|---|---|---|
| Control Group | Baked Salmon Group | p Diets | Control Group | Baked Salmon Group | p Diets | |
| Relative amount of WAT 1, g/100 g bodyweight | 7.43 ± 0.75 | 7.58 ± 0.80 | 0.77 | 4.97 ± 0.90 | 5.80 ± 1.32 | 0.23 |
| Energy intake, kcal/24 h | 144 ± 17 | 126 ± 20 | 0.15 | 123 ± 18 | 122 ± 21 | 0.91 |
| Fat intake, g/24 h | 2.7 ± 0.3 | 3.3 ± 0.5 | 0.045 | 2.3 ± 0.3 | 3.2 ± 0.5 | 0.0062 |
| Zucker Fa/Fa Rats | Long-Evans Rats | |||||
|---|---|---|---|---|---|---|
| Control Group | Baked Salmon Group | p Diets | Control Group | Baked Salmon Group | p Diets | |
| Total cholesterol, mmol/L | 6.3 ± 0.6 | 4.9 ± 0.8 | 0.011 | 2.9 ± 0.3 | 2.8 ± 0.5 | 0.58 |
| HDL cholesterol, mmol/L | 5.3 ± 0.3 | 2.7 ± 0.2 | 4.4 × 10−8 | 2.5 ± 0.2 | 2.3 ± 0.2 | 0.18 |
| LDL cholesterol, mmol/L | 1.5 ± 0.3 | 0.9 ± 0.3 | 0.0025 | 0.7 ± 0.1 | 0.6 ± 0.1 | 0.21 |
| Cholesteryl ester, mmol/L | 4.9 ± 0.5 | 3.7 ± 0.5 | 0.0045 | 2.2 ± 0.2 | 2.2 ± 0.4 | 0.74 |
| Total bile acids, umol/L | 46 ± 21 | 16 ± 7 | 0.0084 | 7 ± 5 | 5 ± 4 | 0.44 |
| NEFA, mmol/L | 0.91 ± 0.13 | 0.84 ± 0.12 | 0.38 | 0.43 ± 0.18 | 0.19 ± 0.14 | 0.030 |
| ApoB-100, ng/mL | 163 ± 53 | 96 ± 32 | 0.036 | 61 ± 6 | 68 ± 11 | 0.22 |
| ApoB-48, ng/mL | 7 ± 3 | 10 ± 3 | 0.12 | 20 ± 3 | 21 ± 4 | 0.69 |
| CRP, mg/mL | 1.18 ± 0.08 | 0.96 ± 0.14 | 0.014 | 0.96 ± 0.05 | 0.98 ± 0.07 | 0.62 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vikøren, L.A.; Drotningsvik, A.; Mjøs, S.A.; Mellgren, G.; Gudbrandsen, O.A. Salmon Fillet Intake Led to Higher Serum Triacylglycerol in Obese Zucker Fa/Fa Rats But Not in Normolipidemic Long-Evans Rats. Nutrients 2018, 10, 1459. https://doi.org/10.3390/nu10101459
Vikøren LA, Drotningsvik A, Mjøs SA, Mellgren G, Gudbrandsen OA. Salmon Fillet Intake Led to Higher Serum Triacylglycerol in Obese Zucker Fa/Fa Rats But Not in Normolipidemic Long-Evans Rats. Nutrients. 2018; 10(10):1459. https://doi.org/10.3390/nu10101459
Chicago/Turabian StyleVikøren, Linn Anja, Aslaug Drotningsvik, Svein Are Mjøs, Gunnar Mellgren, and Oddrun Anita Gudbrandsen. 2018. "Salmon Fillet Intake Led to Higher Serum Triacylglycerol in Obese Zucker Fa/Fa Rats But Not in Normolipidemic Long-Evans Rats" Nutrients 10, no. 10: 1459. https://doi.org/10.3390/nu10101459
APA StyleVikøren, L. A., Drotningsvik, A., Mjøs, S. A., Mellgren, G., & Gudbrandsen, O. A. (2018). Salmon Fillet Intake Led to Higher Serum Triacylglycerol in Obese Zucker Fa/Fa Rats But Not in Normolipidemic Long-Evans Rats. Nutrients, 10(10), 1459. https://doi.org/10.3390/nu10101459





