Chitosan Film Functionalized with Grape Seed Oil—Preliminary Evaluation of Antimicrobial Activity
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chitosan Extraction
2.2. Grape Seed Oil Extraction
2.3. Films
2.4. Antimicrobial Activity of Films
3. Results
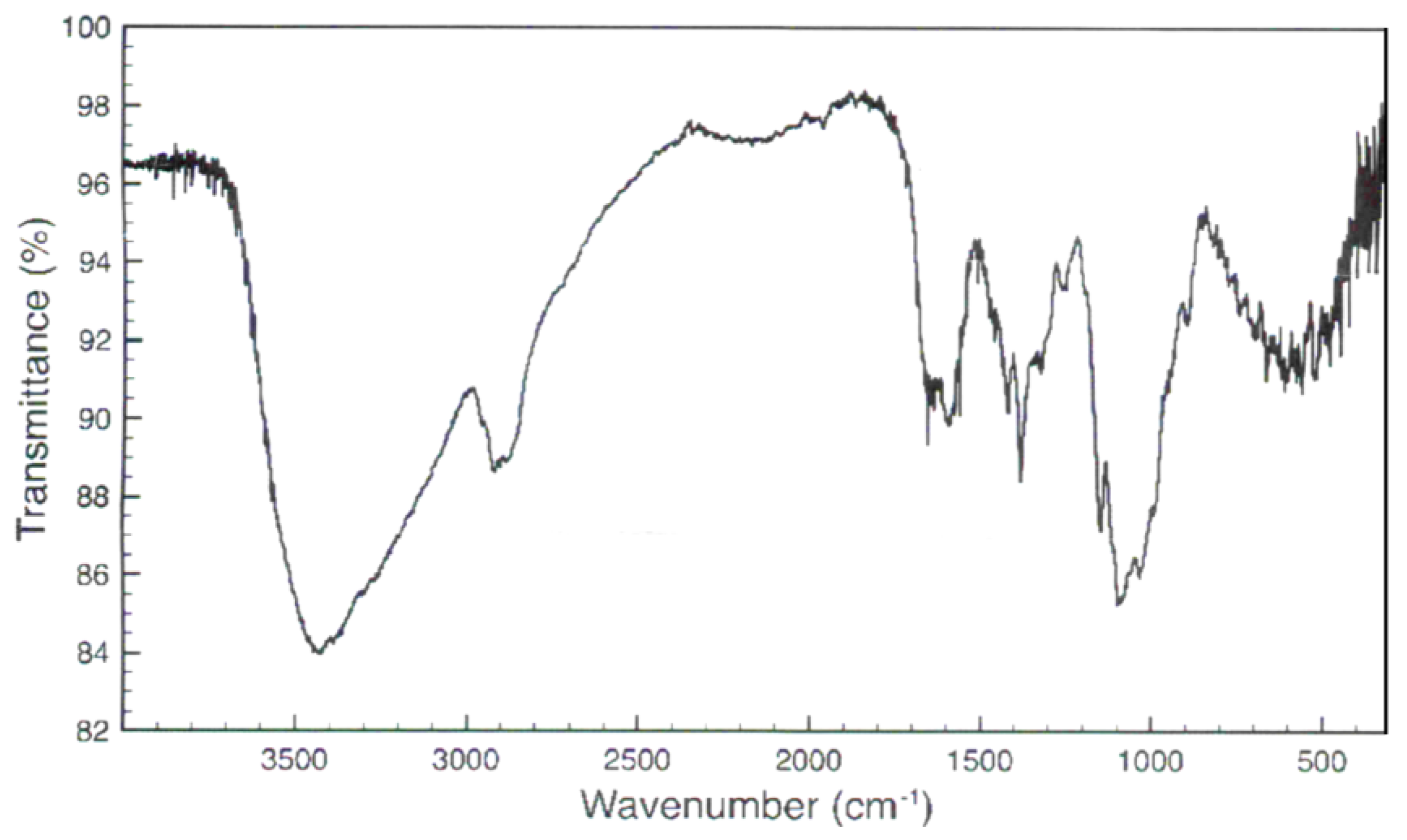
3.1. Chitosan Characterization
3.2. Antimicrobial Activity
4. Discussion
4.1. Chitosan Characterization
4.2. Effects on Gram-Positive Bacteria
4.3. Effects on Gram-Negative Bacteria
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kazemi, K.; Zhang, B.; Lye, L.M.; Zhu, Z. Evaluation of state and evolution of marine fish waste composting by enzyme activities. Can. J. Civ. Eng. 2017, 44, 1–35. [Google Scholar] [CrossRef]
- FAO. The State of World Fisheries and Aquaculture. Sustainability in Action; FAO: Rome, Italy, 2020. [Google Scholar] [CrossRef]
- Gao, L.; Sun, J.A.; Secundo, F.; Gao, X.; Xue, C.H.; Mao, X.Z. Cloning, characterization and substrate degradation mode of a novel chitinase from Streptomyces albolongus ATCC 27414. Food Chem. 2018, 261, 329–336. [Google Scholar] [CrossRef] [PubMed]
- Hamed, I.; Özogul, F.; Regenstein, J.M. Industrial applications of crustacean byproducts (chitin, chitosan, and chitooligosaccharides): A review. Trends Food Sci. Technol. 2016, 48, 40–50. [Google Scholar] [CrossRef]
- Mao, X.Z.; Guo, N.; Sun, J.A.; Xue, C.H. Comprehensive utilization of shrimp waste based on biotechnological methods: A review. J. Clean. Prod. 2017, 143, 814–823. [Google Scholar] [CrossRef]
- Tunkijjanukij, S.; Intarachart, A. Development of green mussel cultivation in Thailand: Sriracha Bay, Chonburi province. Aquac. Asia 2007, 12, 24–25. [Google Scholar]
- Coppola, D.; Lauritano, C.; Palma Esposito, F.; Riccio, G.; Rizzo, C.; de Pascale, D. Fish Waste: From Problem to Valuable Resource. Mar. Drugs 2021, 19, 116. [Google Scholar] [CrossRef]
- United Nations. Special Edition: Progress towards the Sustainable Development Goals. Available online: https://undocs.org/E/2019/68 (accessed on 17 September 2019).
- Wang, W.; Xue, C.; Mao, X. Chitosan: Structural modification, biological activity and application. Int. J. Biol. Macromol. 2020, 164, 4532–4546. [Google Scholar] [CrossRef]
- Shahidi, F.; Kamil, Y.V.A.J. Enzymes from fish and aquatic invertebrates and their application in the food industry. Trends Food Sci. Technol. 2001, 12, 435–464. [Google Scholar] [CrossRef]
- Sivaramakrishna, D.; Bhuvanachandra, B.; Mallakuntla, M.K.; Das, S.N.; Ramakrishna, B.; Podile, A.R. Pretreatment with KOH and KOH-urea enhanced hydrolysis of α-chitin by an endo-chitinase from Enterobacter cloacae subsp. cloacae. Carbohydr. Polym. 2020, 115952, 1–7. [Google Scholar] [CrossRef]
- Xiong, Y.; Luo, B.; Chen, G.; Cai, J.; Jiang, Q.; Gu, B.; Wang, X.Y. CuS@Corn Stalk/Chitin Composite Hydrogel for Photodegradation and Antibacterial. Polymers 2019, 11, 1393. [Google Scholar] [CrossRef] [Green Version]
- Elieh-Ali-Komi, D.; Hamblin, M.R. Chitin and chitosan: Production and application of versatile biomedical nanomaterials. Int. J. Adv. Res. 2016, 4, 411–427. [Google Scholar]
- Shamshina, J.L.; Berton, P.; Rogers, R.D. Advances in functional chitin materials: A review. ACS Sustain. Chem. Eng. 2019, 7, 6444–6457. [Google Scholar] [CrossRef]
- Khor, E.; Lim, L.Y. Implantable applications of chitin and chitosan. Biomaterials 2003, 24, 2339–2349. [Google Scholar] [CrossRef]
- Pillai, C.K.S.; Paul, W.; Sharma, C.P. Chitin and chitosan polymers: Chemistry, solubility and fiber formation. Prog. Polym. Sci. 2009, 34, 641–678. [Google Scholar] [CrossRef]
- Miteluț, A.C.; Tănase, E.E.; Popa, V.I.; Popa, M.E. Sustainable alternative for food packaging: Chitosan biopolymer—A review. AgroLife Sci. J. 2015, 4, 52–61. [Google Scholar]
- Goy, R.C.; de Britto, D.; Assis, O.B.G. A review of the antimicrobial activity of chitosan. Polimeros 2009, 19, 241–247. [Google Scholar] [CrossRef]
- Salah, R.; Michaud, P.; Mati, F.; Harrat, Z.; Lounici, H.; Abdi, N.; Drouiche, N.; Mameri, N. Anticancer activity of chemically prepared shrimp low molecular weight chitin evaluation with the human monocyte leukaemia cell line, THP-1. Int. J. Biol. Macromol. 2013, 52, 333–339. [Google Scholar] [CrossRef]
- Yen, M.T.; Yang, J.H.; Mau, J.L. Antioxidant properties of chitosan from crab shells. Carbohydr. Polym. 2008, 74, 840–844. [Google Scholar] [CrossRef]
- Vongchan, P.; Sajomsang, W.; Kasinrerk, W.; Subyen, D.; Kongtawelert, P. Anticoagulant activities of the chitosan polysulfate synthesized from marine crab shell by semi-heterogeneous conditions. Sci. Asia 2003, 29, 115–120. [Google Scholar] [CrossRef]
- Dash, M.; Chiellini, F.; Ottenbrite, R.M.; Chiellini, E. Chitosan-A versatile semi-synthetic polymer in biomedical applications. Prog. Polym. Sci. 2011, 36, 981–1014. [Google Scholar] [CrossRef]
- Parvez, S.; Rahman, M.M.; Khan, M.A.; Khan, M.A.H.; Islam, J.M.M.; Ahmed, M.; Rahman, M.F.; Ahmed, B. Preparation and characterization of artificial skin using chitosan and gelatin composites for potential biomedical application. Polym. Bull. 2012, 69, 715–731. [Google Scholar] [CrossRef]
- Riva, R.; Ragelle, H.; des Rieux, A.; Duhem, N.; Jérôme, C.; Préat, V. Chitosan and chitosan derivatives in drug delivery and tissue engineering. Adv. Polym. Sci. 2011, 244, 19–44. [Google Scholar]
- Limam, Z.; Selmi, S.; Sadok, S.; El Abed, A. Extraction and characterization of chitin and chitosan from crustacean byproducts: Biological and physicochemical properties. Afr. J. Biotechnol. 2011, 10, 640–647. [Google Scholar]
- Muzzarelli, R.A.A.; Boudrant, J.; Meyer, D.; Manno, N.; Demarchis, M.; Paoletti, M.G. Current views on fungal chitin/chitosan, human chitinases, food preservation, glucans, pectins and inulin: A tribute to Henri Braconnot, precursor of the carbohydrate polymers science, on the chitin bicentennial. Carbohydr. Polym. 2012, 87, 995–1012. [Google Scholar] [CrossRef]
- Bhatnagar, A.; Sillanpää, M. Applications of chitin-and chitosan-derivatives for the detoxification of water and wastewater-a short review. Adv. Colloid Interface 2009, 152, 26–38. [Google Scholar] [CrossRef]
- Jin, R.D.; Suh, J.W.; Park, R.D.; Kim, Y.W.; Krishnan, H.B.; Kim, K.Y. Effect of chitin compost and broth on biological control of Meloidogyne incognita on tomato (Lycopersicon esculentum Mill.). J. Nematol. 2005, 7, 125–132. [Google Scholar] [CrossRef]
- Leceta, I.; Guerrero, P.; Cabezudo, S.; Caba, K.D.L. Environmental assessment of chitosan-based films. J. Clean. Prod. 2013, 41, 312–318. [Google Scholar] [CrossRef]
- Wang, H.; Qian, J.; Ding, F. Emerging Chitosan-Based Films for Food Packaging Applications. J. Agric. Food Chem. 2018, 66, 395–413. [Google Scholar] [CrossRef]
- Kulawik, P.; Jamróz, E.; Özogul, F. Chitosan role for shelf-life extension of seafood. Environ. Chem. Lett. 2020, 18, 61–74. [Google Scholar] [CrossRef]
- Zhang, X.; Li, Y.; Guo, M.; Jin, T.Z.; Ali Arabi, S.; He, Q.; Ismail, B.B.; Hu, Y.; Liu, D. Antimicrobial and UV Blocking Properties of Composite Chitosan Films with Curcumin Grafted Cellulose Nanofiber. Food Hydrocoll. 2021, 112, 1–11. [Google Scholar] [CrossRef]
- Zhang, X.; Ismail, B.B.; Cheng, H.; Jin, T.Z.; Qian, M.; Arabi, S.A.; Liu, D.; Guo, M. Emerging chitosan-essential oil films and coatings for food preservation—A review of advances and applications. Carbohydr. Polym. 2021, 273, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Hromiš, N.M.; Lazić, V.L.; Markov, S.L.; Vaštag, Ž.G.; Popović, S.Z.; Šuput, D.Z.; Džinić, N.R.; Velićanski, A.S.; Popović, L.M. Optimization of chitosan biofilm properties by addition of caraway essential oil and beeswax. J. Food Eng. 2015, 158, 86–93. [Google Scholar] [CrossRef]
- Güneş, S.; Tıhmınlıoğlu, F. Hypericum perforatum incorporated chitosan films as potential bioactive wound dressing material. Int. J. Biol. Macromol. 2017, 102, 933–943. [Google Scholar] [CrossRef] [PubMed]
- Garcia, L.G.S.; da Rocha, M.G.; Lima, L.R.; Cunha, A.P.; de Oliveira, J.S.; de Andrade, A.R.C.; Ricardo, N.M.P.S.; Pereira-Neto, W.A.; Sidrim, J.J.C.; Rocha, M.F.G.; et al. Essential oils encapsulated in chitosan microparticles against Candida albicans biofilms. Int. J. Biol. Macromol. 2021, 166, 621–632. [Google Scholar] [CrossRef]
- Song, X.; Wang, L.; Liu, T.; Liu, Y.; Wu, X.; Liu, L. Mandarin (Citrus reticulata L.) essential oil incorporated into chitosan nanoparticles: Characterization, anti-biofilm properties and application in pork preservation. Int. J. Biol. Macromol. 2021, 185, 620–628. [Google Scholar] [CrossRef]
- Soceanu, A.; Dobrinas, S.; Sirbub, A.; Manea, N.; Popescu, V. Economic aspects of waste recovery in the wine industry. A multidisciplinary approach. Sci. Total Environ. 2021, 759, 143543. [Google Scholar] [CrossRef]
- Kumar, K.; Yadav, A.N.; Kumar, V.; Vyas, P.; Dhaliwal, H.S. Food waste: A potential bioresource for extraction of nutraceuticals and bioactive compounds. Bioresour. Bioprocess. 2017, 4, 1–14. [Google Scholar] [CrossRef] [Green Version]
- Oliveira, D.A.; Salvador, A.A.; Smânia, A., Jr.; Smânia, E.F.A.; Maraschin, M.; Ferreira, S.R.S. Antimicrobial activity and composition profile of grape (Vitis vinifera) pomace extracts obtained by supercritical fluids. J. Biotechnol. 2013, 164, 423–432. [Google Scholar] [CrossRef]
- Adámez, J.D.; Samino, E.G.; Sánchez, E.V.; González-Gómez, D. In vitro estimation of the antibacterial activity and antioxidant capacity of aqueous extracts from grape-seeds (Vitis vinifera L.). Food Control 2012, 24, 136–141. [Google Scholar] [CrossRef]
- Rondeau, P.; Gambier, F.; Jolibert, F.; Brosse, N. Compositions and chemical variability of grape pomaces from French vineyard. Ind. Crops Prod. 2013, 43, 251–254. [Google Scholar] [CrossRef]
- Devesa-Rey, R.; Vecino, X.; Varela-Alende, J.L.; Barral, M.T.; Cruz, J.M.; Moldes, A.B. Valorization of winery waste vs. the costs of not recycling. Waste Manag. 2011, 31, 2327–2335. [Google Scholar] [CrossRef] [PubMed]
- Chouchouli, V.; Kalogeropoulos, N.; Konteles, S.J.; Karvela, E.; Makris, D.P.; Karathanos, V.T. Fortification of yoghurts with grape (Vitis vinifera) seed extracts. LWT Food Sci. Technol. 2013, 53, 522–529. [Google Scholar] [CrossRef]
- Spigno, G.; Marinoni, L.; Garrido, G.D. State of the Art in Grape Processing By-Products. In Handbook of Grape Processing By-Products Sustainable Solutions; Academic Press: Cambridge, MA, USA, 2017; pp. 1–27. [Google Scholar]
- Garavaglia, J.; Markoski, M.M.; Oliveira, A.; Marcadent, A. Grape Seed Oil Compounds: Biological and Chemical Actions for Health. Nutr. Metab. Insights 2016, 16, 9–64. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Di Stefano, V.; Bongiorno, D.; Buzzanca, C.; Indelicato, S.; Santini, A.; Lucarini, M.; Fabbrizio, A.; Mauro, M.; Vazzana, M.; Arizza, V.; et al. Fatty Acids and Triacylglycerols Profiles from Sicilian (Cold Pressed vs. Soxhlet) Grape Seed Oils. Sustainability 2021, 13, 13038. [Google Scholar] [CrossRef]
- Khah, M.D.; Ghanbarzadeh, B.; Nezhad, L.R.; Ostadrahimi, A. Effects of virgin olive oil and grape seed oil on physicochemical and antimicrobial properties of pectin-gelatin blend emulsified films. Int. J. Biol. Macromol. 2021, 171, 262–274. [Google Scholar] [CrossRef]
- Harbeoui, H.; Dakhlaoui, S.; Wannes, W.A.; Bourgou, S.; Hammami, M.; Khan, N.A.; Tounsi, M.S. Does unsaponifiable fraction of grape seed oil attenuate nitric oxide production, oxidant and cytotoxicity activities. J. Food Biochem. 2019, 43, 1–9. [Google Scholar] [CrossRef]
- Bocsan, I.C.; Pop, R.M.; Sabin, O.; Sarkandy, E.; Boarescu, P.M.; Roșian, Ș.H.; Leru, P.M.; Chedea, V.S.; Socaci, S.A.; Buzoianu, A.D. Comparative Protective Effect of Nigella sativa Oil and Vitis vinifera Seed Oil in an Experimental Model of Isoproterenol-Induced Acute Myocardial Ischemia in Rats. Molecules 2021, 26, 3221. [Google Scholar] [CrossRef]
- Sano, A.; Uchida, R.; Saito, M.; Shioya, N.; Komori, Y.; Tho, Y.; Hashizume, N. Beneficial effects of grape seed extract on malondialdehyde-modified LDL. J. Nutr. Sci. Vitaminol. 2007, 53, 174–182. [Google Scholar] [CrossRef] [Green Version]
- Huang, S.; Yang, N.; Liu, Y.; Gao, J.; Huang, T.; Hu, L.; Zhao, J.; Li, Y.; Li, C.; Zhang, X. Grape seed proanthocyanidins inhibit colon cancer-induced angiogenesis through suppressing the expression of VEGF and Ang1. Int. J. Mol. Med. 2012, 30, 1410–1416. [Google Scholar] [CrossRef] [Green Version]
- Duba, K.S.; Fiori, L. Supercritical CO2 extraction of grape seed oil: Effect of process parameters on the extraction kinetics. J. Supercrit. Fluids 2015, 98, 33–43. [Google Scholar] [CrossRef]
- Rombaut, N.; Savoire, R.; Thomasset, B.; Bélliard, T.; Castello, J.; Hecke, É.V.; Lanoisellé, J.L. Grape seed oil extraction: Interest of supercritical fluid extraction and gas-assisted mechanical extraction for enhancing polyphenol co-extraction in oil. C. R. Chim. 2014, 17, 284–292. [Google Scholar] [CrossRef]
- Maier, T.; Schieder, A.; Kammerer, D.R.; Carle, R. Residues of grape (Vitis vinifera L.) seed oil production as a valuable source of phenolic antioxidants. Food Chem. 2009, 112, 551–559. [Google Scholar] [CrossRef]
- Karaman, S.; Karasu, S.; Tornuk, F.; Toker, O.S.; Geçgel, Ü.; Sagdic, O.; Ozcan, N.; Gül, O. Recovery potential of cold press byproducts obtained from the edible oil industry: Physicochemical, bioactive, and antimicrobial properties. J. Agric. Food Chem. 2015, 63, 2305–2313. [Google Scholar] [CrossRef] [PubMed]
- Shinagawa, F.B.; Santana, F.C.; Mancini-Filho, J. Effect of cold pressed grape seed oil on rats biochemical markers and inflammatory profile. Rev. Nutr. 2015, 28, 65–76. [Google Scholar] [CrossRef] [Green Version]
- Li, T.; Cui, F.; Bai, F.; Zhao, G.; Li, J. Involvement of acylated homoserine lactones (AHLs) of aeromonas sobria in spoilage of refrigerated turbot (Scophthalmus maximus L.). Sensors 2016, 16, 1083. [Google Scholar] [CrossRef] [PubMed]
- Schuppler, M.; Loessner, M.J. The opportunistic pathogen Listeria monocytogenes: Pathogenicity and interaction with the mucosal immune system. Int. J. Inflam. 2010, 2010, 1–12. [Google Scholar]
- Orgaz, B.; Lobete, M.M.; Puga, C.H.; San Jose, C. Effectiveness of Chitosan against Mature Biofilms Formed by Food Related Bacteria. Int. J. Mol. Sci. 2011, 12, 817–828. [Google Scholar] [CrossRef] [Green Version]
- Møretrø, T.; Langsrud, S. Residential Bacteria on Surfaces in the Food Industry and Their Implications for Food Safety and Quality. Compr. Rev. Food Sci. Food Saf. 2017, 16, 1022–1041. [Google Scholar] [CrossRef] [Green Version]
- Li, Q.; Li, Y.; Tang, Y.; Meng, C.; Ingmer, H.; Jiao, X. Prevalence and characterization of Staphylococcus aureus and Staphylococcus argenteus in chicken from retail markets in China. Food Control 2019, 96, 158–164. [Google Scholar] [CrossRef]
- Dong, J.; Fang, D.; Zhang, L.; Shan, Q.; Huang, Y. Gallium-doped titania nanotubes elicit anti-bacterial efficacy in vivo against Escherichia coli and Staphylococcus aureus biofilm. Materialia 2019, 5, 100209. [Google Scholar] [CrossRef]
- Amankwaah, C.; Li, J.; Lee, J.; Pascall, M.A. Development of antiviral and bacteriostatic chitosan-based food packaging material with grape seed extract for murine norovirus, Escherichia coli and Listeria innocua control. Food Sci. Nutr. 2020, 8, 6174–6181. [Google Scholar] [CrossRef] [PubMed]
- Cerveny, J.; Meyer, J.D.; Hall, P.A. Microbiological spoilage of meat and poultry products. In Compendium of the Microbiological Spoilage of Foods and Beverages; Springer: Berlin/Heidelberg, Germany, 2009; pp. 69–86. [Google Scholar]
- Rubilar, J.F.; Cruz, R.M.S.; Silva, H.D.; Vicente, A.A.; Khmelinskii, I.; Vieira, M.C. Physico-mechanical properties of chitosan films with carvacrol and grape seed extract. J. Food Eng. 2013, 115, 466–474. [Google Scholar] [CrossRef] [Green Version]
- Bhaskar Reddy, G.V.; Sen, A.R.; Nair, P.N.; Sudhakar Reddy, K.; Kondal Reddy, K.; Kondaiah, N. Effects of grape seed extract on the oxidative and microbial stability of restructured mutton slices. Meat Sci. 2013, 95, 288–294. [Google Scholar] [CrossRef] [PubMed]
- Alves, V.L.C.D.; Rico, B.P.M.; Cruz, R.M.S.; Vicente, A.A.; Khmelinskii, I.; Vieira, M.C. Preparation and characterization of a chitosan film with grape seed extract carvacrol microcapsules and its effect on the shelf-life of refrigerated Salmon (Salmo salar). LWT Food Sci. Technol. 2018, 89, 525–534. [Google Scholar] [CrossRef] [Green Version]
- Guo, M.; Jin, T.Z.; Wang, L.; Scullen, O.J.; Sommers, C.H. Antimicrobial films and coatings for inactivation of Listeria innocua on ready-to-eat deli turkey meat. Food Control 2014, 40, 64–70. [Google Scholar] [CrossRef]
- Leceta, I.; Molinaro, S.; Guerrero, P.; Kerry, J.P.; de la Caba, K. Quality attributes of map packaged ready-to-eat baby carrots by using chitosan-based coatings. Postharvest Biol. Technol. 2015, 100, 142–150. [Google Scholar] [CrossRef]
- Rezaeifar, M.; Mehdizadeh, T.; Langroodi, A.M.; Rezaei, F. Effect of chitosan edible coating enriched with lemon verbena extract and essential oil on the shelf life of vacuum rainbow trout (Oncorhynchus mykiss). J. Food Saf. 2020, 40, 1–9. [Google Scholar] [CrossRef]
- Nawaz, T.; Fatima, M.; Shah, S.Z.H.; Afzal, M. Coating effect of rosemary extract combined with chitosan on storage quality of mori (Cirrhinus mrigala). J. Food Process. Preserv. 2020, 44, 1–10. [Google Scholar] [CrossRef]
- No, H.K.; Meyers, S.P. Crawfish Chitosan as a Coagulant in Recovery of Organic Com-pounds from Seafood Processing Streams. J. Agric. Food Chem. 1989, 37, 580–583. [Google Scholar] [CrossRef]
- Hadi, A.G. Synthesis of Chitosan and Its Use in Metal Removal. Chem. Mater. Res. 2013, 3, 22–26. [Google Scholar]
- Taher, F.A.; Ibrahim, S.A.; El-Aziz, A.A.; Abou El-Nour, M.F.; El-Sheikh, M.A.; El-Husseiny, N.; Mohamed, M.M. Anti-proliferative effect of chitosan nanoparticles (extracted from crayfish Procambarus clarkii, Crustacea: Cambaridae) against MDA-MB-231 and SK-BR-3 human breast cancer cell lines. Int. J. Biol. Macromol. 2019, 126, 478–487. [Google Scholar] [CrossRef] [PubMed]
- Muthu, M.; Gopal, J.; Chun, S.; Devadoss, A.J.P.; Hasan, N.; Sivanesan, I. Crustacean Waste-Derived Chitosan: Antioxidant Properties and Future Perspective. Antioxidants 2021, 10, 228. [Google Scholar] [CrossRef] [PubMed]
- Baxter, A.; Dillon, M.; Taylor, K.D.A.; Roberts, G.A.F. Improved method for i.r. determination of the degree on N-acetylation of chitosan. Int. J. Biol. Macromol. 1992, 14, 166–169. [Google Scholar] [CrossRef]
- Moura, J.M.; Farias, B.S.; Rodrigues, D.A.S.; Moura, C.M.; Dotto, G.L.; Pinto, L.A.A. Preparation of Chitosan with Different Characteristics and Its Application for Biofilms Production. J. Polym. Environ. 2015, 23, 470–477. [Google Scholar] [CrossRef]
- Cé, N.; Noreña, C.P.Z.; Brandelli, A. Antimicrobial activity of chitosan films containing nisin, peptide P34, and natamycin. CyTA J. Food 2012, 10, 21–26. [Google Scholar] [CrossRef]
- Sebti, I.; Ham-Pichavant, F.; Coma, V. Edible Bioactive Fatty Acid-Cellulosic Derivative Composites Used in Food-Packaging Applications. J. Agric. Food Chem. 2002, 50, 4290–4294. [Google Scholar] [CrossRef]
- Moradi, M.; Tajik, H.; Razavi Rohani, S.M.; Oromiehie, A.R. Effectiveness of Zataria multiflora Boiss essential oil and grape seed extract impregnated chitosan film on ready-to-eat mortadella-type sausages during refrigerated storage. J. Sci. Food Agric. 2011, 91, 2850–2857. [Google Scholar] [CrossRef]
- Pranoto, Y.; Rakshit, S.K.; Salokhe, V.M. Enhancing antimicrobial activity of chitosan films by incorporating garlic oil, potassium sorbate and nisin. LWT Food Sci. Technol. 2005, 38, 859–865. [Google Scholar] [CrossRef]
- Hessle, C.C.; Andersson, B.; Wold, A.E. Gram-positive and Gram-negative bacteria elicit different patterns of pro-inflammatory cytokines in human monocytes. Cytokine 2005, 30, 311–318. [Google Scholar] [CrossRef]
- Fernandez-Saiz, P.; Lagaron, J.M.; Ocio, M.J. Optimization of the biocide properties of chitosan for its application in the design of active films of interest in the food area. Food Hydrocoll. 2009, 23, 913–921. [Google Scholar] [CrossRef]
- Kong, M.; Chen, X.G.; Xing, K.; Park, H.J. Antimicrobial properties of chitosan and mode of action: A state of the art review. Int. J. Food Microbiol. 2010, 144, 51–63. [Google Scholar] [CrossRef] [PubMed]
- Mu, H.; Zhang, A.; Zhang, L.; Niu, H.; Duan, J. Inhibitory effects of chitosan in combination with antibiotics on Listeria monocytogenes biofilm. Food Control 2014, 38, 215–220. [Google Scholar] [CrossRef]
- Fernandez-Saiz, P.; Soler, C.; Lagaron, J.M.; Ocio, M.J. Effects of chitosan films on the growth of Listeria monocytogenes, Staphylococcus aureus and Salmonella spp. in laboratory media and in fish soup. Int. J. Food Microbiol. 2010, 137, 287–294. [Google Scholar] [CrossRef] [PubMed]
- Veldhuizen, E.J.A.; Creutzberg, T.O.; Burt, S.A.; Haagsman, H.P. Low Temperature and Binding to Food Components Inhibit the Antibacterial Activity of Carvacrol against Listeria monocytogenes in Steak Tartare. J. Food Prot. 2007, 70, 2127–2132. [Google Scholar] [CrossRef] [PubMed]
- Alzagameem, A.; Klein, S.E.; Bergs, M.; Do, X.T.; Korte, I.; Dohlen, S.; Hüwe, C.; Kreyenschmidt, J.; Kamm, B.; Larkins, M.; et al. Antimicrobial Activity of Lignin and Lignin-Derived Cellulose and Chitosan Composites against Selected Pathogenic and Spoilage Microorganisms. Polymers 2019, 11, 670. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Petrou, S.; Tsiraki, M.; Giatrakou, V.; Savvaidis, I.N. Chitosan dipping or oregano oil treatments, singly or combined on modified atmosphere packaged chicken breast meat. Int. J. Food Microbiol. 2012, 156, 264–271. [Google Scholar] [CrossRef]
- Hosseinnejad, M.; Jafari, S.M. Evaluation of different factors affecting antimicrobial properties of chitosan. Int. J. Biol. Macromol. 2016, 85, 467–475. [Google Scholar] [CrossRef]
- Yuan, G.; Lv, H.; Tang, W.; Zhang, X.; Sun, H. Effect of chitosan coating combined with pomegranate peel extract on the quality of Pacific white shrimp during iced storage. Food Control 2016, 59, 818–823. [Google Scholar] [CrossRef]
- Tsai, G.J.; Tsai, M.T.; Lee, J.M.; Zhong, M.Z. Effects of Chitosan and a Low-Molecular-Weight Chitosan on Bacillus cereus and Application in the Preservation of Cooked Rice. J. Food Prot. 2006, 69, 2168–2175. [Google Scholar] [CrossRef]
- Watnick, P.; Kolter, R. Biofilm, city of microbes. J. Bacteriol. 2000, 182, 2675–2679. [Google Scholar] [CrossRef] [Green Version]
- Muslim, S.N.; Mohammed Ali, A.N.; Muslem, W.H.; Fayyad, R.J. Antimicrobial and Antibiofilm Properties for Chitosan Extracted by Biological Methods. Ann. Rom. Soc. Cell Biol. 2021, 25, 2050–2057. [Google Scholar]
- Ma, Z.; Garrido-Maestu, A.; Jeong, K.C. Application, mode of action, and in vivo activity of chitosan and its micro- and nanoparticles as antimicrobial agents: A review. Carbohydr. Polym. 2017, 176, 257–265. [Google Scholar] [CrossRef] [PubMed]
- Coma, V.; Martial-Gros, A.; Garreau, S.; Copinet, A.; Salin, F.; Deschamps, A. Edible Antimicrobial Films Based on Chitosan Matrix. J. Food Sci. 2002, 67, 1162–1169. [Google Scholar] [CrossRef]
- Duman, F.; Kaya, M. Crayfish chitosan for microencapsulation of coriander (Coriandrum sativum L.) essential oil. Int. J. Biol. Macromol. 2016, 92, 125–133. [Google Scholar] [CrossRef] [PubMed]
- No, H.K.; Young Park, N.; Ho Lee, S.; Meyers, S.P. Antibacterial activity of chitosans and chitosan oligomers with different molecular weights. Int. J. Food Microbiol. 2002, 74, 65–72. [Google Scholar] [CrossRef]
- Garrido-Maestu, A.; Ma, Z.; Paik, S.Y.R.; Chen, N.; Ko, S.; Tong, Z.; Jeong, K.C. Engineering of chitosan-derived nanoparticles to enhance antimicrobial activity against foodborne pathogen Escherichia coli O157:H7. Carbohydr. Polym. 2018, 197, 623–630. [Google Scholar] [CrossRef] [PubMed]
- Kives, J.; Orgaz, B.; Sanjose, C. Polysaccharide differences between planktonic and biofilm-associated EPS from Pseudomonas fluorescens. Colloids Surf. B 2006, 52, 123–127. [Google Scholar] [CrossRef] [PubMed]
- Sutherland, I.W. Comprehensive Glycoscience; Kamerling, J.P., Ed.; Elsevier: Doordrecht, The Netherlands, 2007; Volume 2, pp. 521–558. [Google Scholar]
- Jefferson, K.K. What drives bacteria to produce a biofilm? FEMS Microbiol. Lett. 2004, 236, 163–173. [Google Scholar] [CrossRef]
- Flemming, H.C.; Wingender, J. The biofilm matrix. Nat. Rev. Microbiol. 2010, 8, 623–633. [Google Scholar] [CrossRef]
- Huys, G.; Cnockaert, M.; Abbott, S.L.; Janda, J.M.; Vandamme, P. Hafnia paralvei sp. nov., formerly known as Hafnia alvei hybridization group 2. Int. J. Syst. Evol. Microbiol. 2010, 60, 1725–1728. [Google Scholar] [CrossRef] [Green Version]
- Silalahi, J.; Situmorang, P.; Patilaya, P.; Silalahi, Y.C. Antibacterial activity of chitosan and hydrolyzed coconut oil and their combination against Bacillus cereus and Escherichia coli. Asian J. Pharm. Clin. Res. 2016, 9, 69–73. [Google Scholar] [CrossRef]
- Menconi, A.; Pumford, N.R.; Morgan, M.J.; Bielke, L.R.; Kallapura, G.; Latorre, J.D.; Wolfenden, A.D.; Hernandez-Velasco, X.; Hargis, B.M.; Tellez, G. Effect of Chitosan on Salmonella Typhimurium in Broiler Chickens. Foodborne Pathog. Dis. 2014, 11, 165–169. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chien, R.C.; Yen, M.T.; Mau, J.L. Antimicrobial and antitumor activities of chitosan from shiitake stipes, compared to commercial chitosan from crab shells. Carbohydr. Polym. 2016, 138, 259–264. [Google Scholar] [CrossRef] [PubMed]
- Jeon, S.J.; Oh, M.; Yeo, W.S.; Galvão, K.N.; Jeong, K.C. Underlying Mechanism of Antimicrobial Activity of Chitosan Microparticles and Implications for the Treatment of Infectious Diseases. PLoS ONE 2014, 9, 1–10. [Google Scholar] [CrossRef]
- Dutta, J.; Dutta, P.K. Antimicrobial activity of chitin, chitosan and their oligosaccharides. In Chitosan, Oligosaccharides and Their Derivatives; Kim, S.K., Ed.; CRC Press: New York, NY, USA, 2010; pp. 195–214. [Google Scholar]
- Zheng, L.Y.; Zhu, J.F. Study on antimicrobial activity of chitosan with different molecular weights. Carbohydr. Polym. 2003, 54, 527–530. [Google Scholar] [CrossRef]
- Vilar Junior, J.C.; Ribeaux, D.R.; Alves da Silva, C.A.; De Campos-Takaki, G.M. Physicochemical and Antibacterial Properties of Chitosan Extracted from Waste Shrimp Shells. Int. J. Microbiol. 2016, 2016, 1–8. [Google Scholar] [CrossRef] [Green Version]
- Zarayneh, S.; Sepahi, A.A.; Jonoobi, M.; Rasouli, H. Comparative antibacterial effects of cellulose nanofiber, chitosan nanofiber, chitosan/cellulose combination and chitosan alone against bacterial contamination of Iranian banknotes. Int. J. Biol. Macromol. 2018, 118, 1045–1054. [Google Scholar] [CrossRef]
- Liu, Y.; Jiang, Y.; Zhu, J.; Huang, J.; Zhang, H. Inhibition of bacterial adhesion and biofilm formation of sulfonated chitosan against Pseudomonas aeruginosa. Carbohydr. Polym. 2019, 206, 412–419. [Google Scholar] [CrossRef]
- Jackson, K.D.; Starkey, M.; Kremer, S.; Parsek, M.R.; Wozniak, D.J. Identification of psl, a locus encoding a potential exopolysaccharide that is essential for Pseudomonas aeruginosa PAO1 biofilm formation. J. Bacteriol. 2004, 186, 4466–4475. [Google Scholar] [CrossRef] [Green Version]
- Shokri, D.; Khorasgani, M.R.; Mohkam, M.; Fatemi, S.M.; Ghasemi, Y.; Taheri-Kafrani, A. The inhibition effect of lactobacilli against growth and biofilm formation of Pseudomonas aeruginosa. Probiot. Antimicrob. Proteins 2018, 10, 34–42. [Google Scholar] [CrossRef]
- Martinez, L.R.; Mihu, M.R.; Han, G.; Frases, S.; Cordero, R.J.B.; Casadevall, A.; Friedman, A.J.; Friedman, J.M.; Nosanchuk, J.D. The use of chitosan to damage Cryptococcus neoformans biofilms. Biomaterials 2010, 31, 669–679. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Theivendran, S.; Hettiarachchy, N.S.; Johnson, M.G. Inhibition of Listeria monocytogenes by Nisin Combined with Grape Seed Extract or Green Tea Extract in Soy Protein Film Coated on Turkey Frankfurters. J. Food Sci. 2006, 71, 39–44. [Google Scholar] [CrossRef]
- Yoon, B.K.; Jackman, J.A.; Valle-González, E.R.; Cho, N.J. Antibacterial Free Fatty Acids and Monoglycerides: Biological Activities, Experimental Testing, and Therapeutic Applications. Int. J. Mol. Sci. 2018, 19, 1114. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tajkarimi, M.; Ibrahim, S.; Cliver, D. Antimicrobial herb and spice compounds in food. Food Control 2010, 21, 1199–1218. [Google Scholar] [CrossRef]
- Lv, F.; Liang, H.; Yuan, Q.; Li, C. In vitro antimicrobial effects and mechanism of action of selected plant essential oil combinations against four food-related microorganisms. Int. Food Res. 2011, 44, 3057–3064. [Google Scholar] [CrossRef]
- Shahbazi, Y.; Shavisi, N. Interactions of Ziziphora clinopodioides and Mentha spicata essential oils with chitosan and ciprofloxacin against common food-related pathogens. LWT-Food Sci. Technol. 2016, 71, 364–369. [Google Scholar] [CrossRef]
| Species | Code | Classification | Chitosan Biofilm Inhibition | Chitosan + 0.5 mL GSO Biofilm Inhibition | Chitosan + 1mL GSO Biofilm Inhibition |
|---|---|---|---|---|---|
| Listeria monocytogenes | DHPS 11B0 | Pathogen | +++ | +++ | ++ |
| Listeria monocytogenes | DHPS 13B0 | Pathogen | +++ | +++ | ++ |
| Listeria monocytogenes | ATCC 19114 | Pathogen | − | − | − |
| Bacillus cereus | ICE 170 | Pathogen | ++ | + | ± |
| Staphylococcus Aureus | ATCC 33862 | Pathogen | ++ | ++ | ++ |
| Staphylococcus Epidermidis | ICE 244 | Pathogen | − | − | − |
| Brochothrix Thermosphacta | SP10 | Spoilage | +++ | +++ | +++ |
| Species | Code | Classification | Chitosan Biofilm Inhibition | Chitosan + 0.5mL GSO Biofilm Inhibition | Chitosan + 1mL GSO Biofilm Inhibition |
|---|---|---|---|---|---|
| Escherichia coli | PSL 52 | Pathogen | ++ | ++ | ++ |
| Escherichia coli | ATCC 25922 | Pathogen | + | + | ± |
| Stenotrophomonas maltophilia | ICE 272 | Pathogen | + | ++ | + |
| Acinetobacter guillouiae | ICE 24 | Pathogen | + | ++ | ++ |
| Hafnia alvei | 4G 44 | Pathogen | + | + | + |
| Hafnia paralvei | 4G 53 | Pathogen | ++ | ++ | + |
| Salmonella typhimurium | 50432 | Pathogen | ++ | ++ | + |
| Salmonella enteritidis | ATCC 13076 | Pathogen | + | + | ± |
| Enterobacter amnigenus | 60 A2 | Pathogen | + | + | ++ |
| Pseudomonas aeruginosa | PSA 68 | Pathogen | + | ++ | ++ |
| Pseudomonas lactis | SP 198 | Spoilage | + | + | − |
| Pseudomonas poae | 4G 558 | Spoilage | − | − | − |
| Pseudomonas endophytica | 4G 764 | Spoilage | − | − | − |
| Pseudomonas fluorescens | 4G 628 | Spoilage | +++ | − | − |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mauro, M.; Pinto, P.; Settanni, L.; Puccio, V.; Vazzana, M.; Hornsby, B.L.; Fabbrizio, A.; Di Stefano, V.; Barone, G.; Arizza, V. Chitosan Film Functionalized with Grape Seed Oil—Preliminary Evaluation of Antimicrobial Activity. Sustainability 2022, 14, 5410. https://doi.org/10.3390/su14095410
Mauro M, Pinto P, Settanni L, Puccio V, Vazzana M, Hornsby BL, Fabbrizio A, Di Stefano V, Barone G, Arizza V. Chitosan Film Functionalized with Grape Seed Oil—Preliminary Evaluation of Antimicrobial Activity. Sustainability. 2022; 14(9):5410. https://doi.org/10.3390/su14095410
Chicago/Turabian StyleMauro, Manuela, Paola Pinto, Luca Settanni, Valeria Puccio, Mirella Vazzana, Branwen L. Hornsby, Antonio Fabbrizio, Vita Di Stefano, Giampaolo Barone, and Vincenzo Arizza. 2022. "Chitosan Film Functionalized with Grape Seed Oil—Preliminary Evaluation of Antimicrobial Activity" Sustainability 14, no. 9: 5410. https://doi.org/10.3390/su14095410
APA StyleMauro, M., Pinto, P., Settanni, L., Puccio, V., Vazzana, M., Hornsby, B. L., Fabbrizio, A., Di Stefano, V., Barone, G., & Arizza, V. (2022). Chitosan Film Functionalized with Grape Seed Oil—Preliminary Evaluation of Antimicrobial Activity. Sustainability, 14(9), 5410. https://doi.org/10.3390/su14095410







