Antibacterial Efficacy and Physiochemical Effects of Ozone Microbubble Water on Tomato
Abstract
:1. Introduction
2. Material and Methods
2.1. Preparation of Bacterial Suspension
2.2. Preparation of Ozone Microbubble (OMB)
2.3. Inoculation and Inactivation of Bacteria on Tomatoes
2.4. Measurement of pH, Oxidation Reduction Potential (ORP), Electrical Conductivity (EC), and Ozone (O3) of OMB Water
2.5. Analyses of Tomato Texture, Color, and Sensory Characteristics
2.5.1. Analyses of Tomato Texture
2.5.2. Analyses of Tomato Color
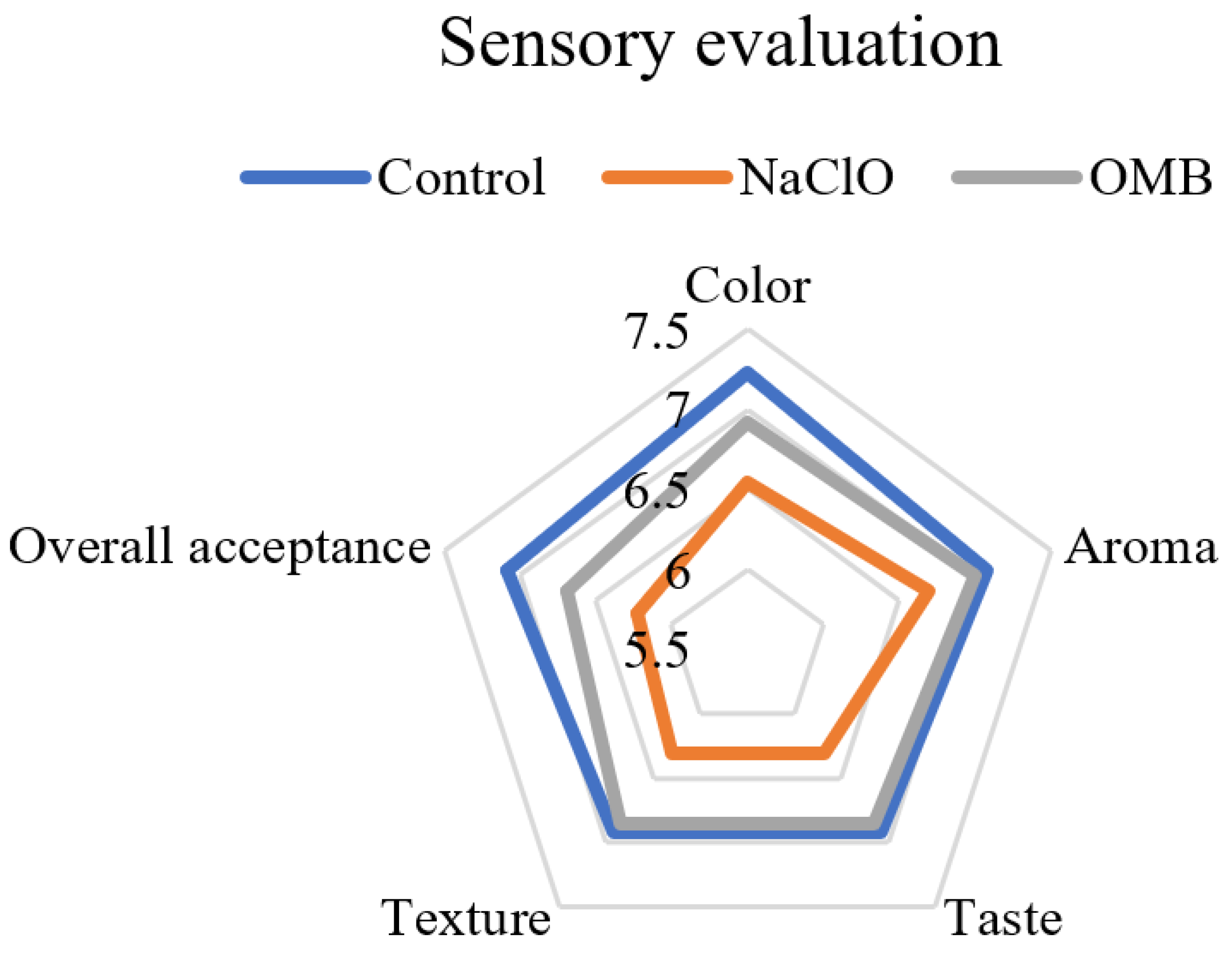
2.5.3. Sensory Evaluation of Tomatoes
2.6. Statistical Analyses
3. Results and Discussion
3.1. Antibacterial Effects of Various Treatments
3.2. Physiochemical Characteristics of Treatment Water at Various Parameters
3.3. The Texture, Color, and Sensory Characteristics of Treated Tomatoes
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wright, A.; Uprety, B.; Shaw, A.; Shama, G.; Iza, F.; Bandulasena, H. Effect of Humic Acid on E. coli Disinfection in a Microbubble-Gas Plasma Reactor. J. Water Process Eng. 2019, 31, 100881. [Google Scholar] [CrossRef]
- Dewey-Mattia, D.; Manikonda, K.; Hall, A.J.; Wise, M.E.; Crowe, S.J. Surveillance for Foodborne Disease Outbreaks—United States, 2009–2015. MMWR Surveill. Summ. 2018, 67, 1. [Google Scholar] [CrossRef] [PubMed]
- de Oliveira Elias, S.; Tombini Decol, L.; Tondo, E.C. Foodborne Outbreaks in Brazil Associated with Fruits and Vegetables: 2008 through 2014. Food Qual. Saf. 2018, 2, 173–181. [Google Scholar] [CrossRef] [Green Version]
- Centers for Disease Control and Prevention (CDC). Surveillance for Foodborne Disease Outbreaks; Annual Report; US Department of Health and Human Services, CDC: Atlanta, GA, USA, 2017. [Google Scholar]
- Bennett, S.D.; Littrell, K.W.; Hill, T.A.; Mahovic, M.; Behravesh, C.B. Multistate Foodborne Disease Outbreaks Associated with Raw Tomatoes, United States, 1990–2010: A Recurring Public Health Problem. Epidemiol. Infect. 2015, 143, 1352–1359. [Google Scholar] [CrossRef]
- Bartz, J.A.; Yuk, H.-G.; Mahovic, M.J.; Warren, B.R.; Sreedharan, A.; Schneider, K.R. Internalization of Salmonella Enterica by Tomato Fruit. Food Control 2015, 55, 141–150. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention (CDC). Outbreaks of Salmonella Infections Associated with Eating Roma Tomatoes--United States and Canada, 2004. MMWR. Morb. Mortal. Wkly. Rep. 2005, 54, 325–328. [Google Scholar]
- Centers for Disease Control and Prevention (CDC). Multistate Outbreaks of Salmonella Infections Associated with Raw Tomatoes Eaten in Restaurants—United States, 2005–2006. MMWR. Morb. Mortal. Wkly. Rep. 2007, 56, 909–911. [Google Scholar]
- Le, H.H.T.; Dalsgaard, A.; Andersen, P.S.; Nguyen, H.M.; Ta, Y.T.; Nguyen, T.T. Large-Scale Staphylococcus aureus Foodborne Disease Poisoning Outbreak among Primary School Children. Microbiol. Res. 2021, 12, 43–52. [Google Scholar] [CrossRef]
- Takahashi, M. Potential of Microbubbles in Aqueous Solutions: Electrical Properties of the Gas−Water Interface. J. Phys. Chem. B 2005, 109, 21858–21864. [Google Scholar] [CrossRef]
- Tsuge, H. Micro-and Nanobubbles-Fundamentals and Applications; Jenny Stanford Publishing: New York, NY, USA, 2014; ISBN 9789814463102. [Google Scholar] [CrossRef]
- Parmar, R.; Majumder, S.K. Microbubble Generation and Microbubble-Aided Transport Process Intensification—A State-of-the-Art Report. Chem. Eng. Process. Process Intensif. 2013, 64, 79–97. [Google Scholar] [CrossRef]
- Sharma, P.K.; Gibcus, M.J.; van derMei, H.C.; Busscher, H.J. Influence of Fluid Shear and Microbubbles on Bacterial Detachment from a Surface. Appl. Environ. Microbiol. 2005, 71, 3668–3673. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Klintham, P.; Tongchitpakdee, S.; Chinsirikul, W.; Mahakarnchanakul, W. Two-Step Washing with Commercial Vegetable Washing Solutions, and Electrolyzed Oxidizing Microbubbles Water to Decontaminate Sweet Basil and Thai Mint: A Case Study. Food Control 2018, 94, 324–330. [Google Scholar] [CrossRef]
- Chuajedton, A.; Aoyagi, H.; Uthaibutra, J.; Pengphol, S.; Whangchai, K. Inactivation of Escherichia coli O157: H7 by Treatment with Different Temperatures of Micro-Bubbles Ozone Containing Water. Int. Food Res. J. 2017, 24, 1006–1010. [Google Scholar]
- Ikeura, H.; Hamasaki, S.; Tamaki, M. Effects of Ozone Microbubble Treatment on Removal of Residual Pesticides and Quality of Persimmon Leaves. Food Chem. 2013, 138, 366–371. [Google Scholar] [CrossRef] [PubMed]
- Kwack, Y.; Kim, K.; Hwang, H.; Chun, C. An Ozone Micro-Bubble Technique for Seed Sterilization in Alfalfa Sprouts. Korean J. Hortic. Sci. Technol. 2014, 32, 901–905. [Google Scholar] [CrossRef] [Green Version]
- Pandiselvam, R.; Kaavya, R.; Jayanath, Y.; Veenuttranon, K.; Lueprasitsakul, P.; Divya, V.; Kothakota, A.; Ramesh, S.V. Ozone as a Novel Emerging Technology for the Dissipation of Pesticide Residues in Foods–a Review. Trends Food Sci. Technol. 2020, 97, 38–54. [Google Scholar] [CrossRef]
- Phaephiphat, A.; Mahakarnchanakul, W. Surface Decontamination of Salmonella Typhimurium and Escherichia coli on Sweet Basil by Ozone Microbubbles. Cogent Food Agric. 2018, 4, 1558496. [Google Scholar] [CrossRef]
- Pongprasert, N.; Jitareerat, P.; Srilaong, V. Effect of Carbon Dioxide Micro Bubbles in Combination with Chlorine Dioxide Solution to Reduce Microbial Contamination and Browning of Fresh-Cut Cos Lettuce (Lactuca Sativa L.). Acta Hortic. 2020, 1275, 39–44. [Google Scholar] [CrossRef]
- Ikeura, H.; Kobayashi, F.; Tamaki, M. Removal of Residual Pesticide, Fenitrothion, in Vegetables by Using Ozone Microbubbles Generated by Different Methods. J. Food Eng. 2011, 103, 345–349. [Google Scholar] [CrossRef]
- Li, P.; Wu, C.; Yang, Y.; Wang, Y.; Yu, S.; Xia, S.; Chu, W. Effects of Microbubble Ozonation on the Formation of Disinfection By-Products in Bromide-Containing Water from Tai Lake. Sep. Purif. Technol. 2018, 193, 408–414. [Google Scholar] [CrossRef]
- Zhang, H.; Tikekar, R.V. Air Microbubble Assisted Washing of Fresh Produce: Effect on Microbial Detachment and Inactivation. Postharvest Biol. Technol. 2021, 181, 111687. [Google Scholar] [CrossRef]
- Ushida, A.; Koyama, T.; Nakamoto, Y.; Narumi, T.; Sato, T.; Hasegawa, T. Antimicrobial Effectiveness of Ultra-Fine Ozone-Rich Bubble Mixtures for Fresh Vegetables Using an Alternating Flow. J. Food Eng. 2017, 206, 48–56. [Google Scholar] [CrossRef]
- Staehelin, J.; Hoigne, J. Decomposition of Ozone in Water: Rate of Initiation by Hydroxide Ions and Hydrogen Peroxide. Environ. Sci. Technol. 1982, 16, 676–681. [Google Scholar] [CrossRef]
- Seki, M.; Ishikawa, T.; Terada, H.; Nashimoto, M. Microbicidal Effects of Stored Aqueous Ozone Solution Generated by Nano-Bubble Technology. In Vivo 2017, 31, 579–583. [Google Scholar] [CrossRef] [Green Version]
- Takahashi, M.; Chiba, K.; Li, P. Formation of Hydroxyl Radicals by Collapsing Ozone Microbubbles under Strongly Acidic Conditions. J. Phys. Chem. B 2007, 111, 11443–11446. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Fan, W.; Huo, M.; Zhao, H.; Lu, Y. Hydroxyl Radical Generation and Contaminant Removal from Water by the Collapse of Microbubbles Under Different Hydrochemical Conditions. Water Air Soil Pollut. 2018, 229, 86. [Google Scholar] [CrossRef]
- Xiong, X.; Wang, B.; Zhu, W.; Tian, K.; Zhang, H.A. Review on Ultrasonic Catalytic Microbubbles Ozonation Processes: Properties, Hydroxyl Radicals Generation Pathway and Potential in Application. Catalysts 2019, 9, 10. [Google Scholar] [CrossRef] [Green Version]
- Kobayashi, F.; Ikeura, H.; Ohsato, S.; Goto, T.; Tamaki, M. Disinfection Using Ozone Microbubbles to Inactivate Fusarium oxysporum f. Sp. melonis and Pectobacterium carotovorum Subsp. carotovorum. Crop Prot. 2011, 30, 1514–1518. [Google Scholar] [CrossRef]



| Treatments/Bacteria | S. Enteritidis | S. Typhimurium | E. coli | S. aureus |
|---|---|---|---|---|
| Untreated | 8.50 ± 0.26 Ca | 9.29 ± 0.15 Ba | 9.54 ± 0.11 Aa | 9.90 ± 0.11 ABa |
| Water washed | 8.05 ± 0.42 Ba | 8.70 ± 0.42 Ab | 8.51 ± 0.32 ABb | 8.09 ± 0.24 Bb |
| MB 20 min-30 s | 6.32 ± 0.35 Bde | 6.86 ± 0.16 Ac | 6.20 ± 0.03 Bc | 6.80 ± 0.24 Ad |
| MB 20 min-60 s | 5.17 ± 0.51 Af | 5.85 ± 0.09 Ad | 5.98 ± 1.30 Ac | 6.29 ± 0.25 Ae |
| MB 10 min-30 s | 6.48 ± 0.79 Ade | 6.65 ± 0.29 Ac | 6.29 ± 0.56 Ac | 6.79 ± 0.11 Ad |
| MB 10 min-60 s | 6.00 ± 0.48 Ae | 5.89 ± 0.27 Ad | 5.70 ± 0.22 Ac | 6.28 ± 0.32 Ae |
| Ozone 20 min-30 s | 7.25 ± 0.32 Bb | 8.52 ± 0.41 Ab | 8.26 ± 0.53 Ab | 7.90 ± 0.30 ABb |
| Ozone 20 min-60 s | 7.12 ± 0.24 Cbc | 8.49 ± 0.76 Ab | 8.15 ± 0.39 ABb | 7.40 ± 0.11 BCc |
| Ozone 10 min-30 s | 7.31 ± 0.58 Bb | 8.37 ± 0.57 Ab | 8.28 ± 0.30 Ab | 7.90 ± 0.09 ABb |
| Ozone 10 min-60 s | 6.90 ± 0.26 Bbcd | 8.25 ± 0.88 Ab | 8.15 ± 0.57 Ab | 7.29 ± 0.36 ABc |
| OMB 20 min-30 s | 4.00 ± 0.05 Ag | 3.84 ± 0.25 Ae | 4.45 ± 0.58 Ad | 3.94 ± 0.31 Af |
| OMB 20 min-60 s | 3.66 ± 0.16 Bg | 3.79 ± 0.36 ABe | 4.30 ± 0.36 Ad | 3.71 ± 0.32 Bf |
| OMB 10 min-30 s | 3.94 ± 0.40 Ag | 3.87 ± 0.11 Ae | 4.20 ± 0.29 Ad | 3.97 ± 0.47 Af |
| OMB 10 min-60 s | 3.60 ± 0.07 Ag | 3.79 ± 0.21 Ae | 4.16 ± 0.46 Ad | 3.83 ± 0.41 Af |
| Treatments | pH Values | Conductivity | ORP | Ozone |
|---|---|---|---|---|
| Water | 7.29 ± 0.203 A | 477 ± 18.6 A | 302.8 ± 15.61 C | 0.05 ± 0.02 C |
| MB 20 min-30 s | 7.13 ± 0.021 A | 660 ± 10.2 B | 310.4 ± 36.60 C | 0.10 ± 0.06 C |
| MB 20 min-60 s | 7.13 ± 0.044 A | 606 ± 20.5 B | 309.6 ± 29.90 C | 0.04 ± 0.02 C |
| MB 10 min-30 s | 7.27 ± 0.007 A | 666 ± 15.6 B | 317.2 ± 10.06 C | 0.05 ± 0.02 C |
| MB 10 min-60 s | 7.26 ± 0.021 A | 657 ± 17.0 B | 323.0 ± 57.61 C | 0.10 ± 0.05 C |
| Ozone 20 min-30 s | 7.15 ± 0.021 A | 634 ± 13.54 B | 426.7 ± 57.60 B | 2.24 ± 0.80 B |
| Ozone 20 min-60 s | 7.14 ± 0.042 A | 619 ± 21.5 B | 410.7 ± 14.84 B | 2.25 ± 0.90 B |
| Ozone 10 min-30 s | 7.23 ± 0.021 A | 662 ± 12.8 B | 415.3 ± 8.08 B | 1.83 ± 0.32 B |
| Ozone 10 min-60 s | 7.22 ± 0.021 A | 666 ± 12.4 B | 426.3 ± 8.62 B | 1.94 ± 0.46 B |
| OMB 20 min-30 s | 7.46 ± 0.044 A | 708 ± 13.8 B | 764.4 ± 47.84 A | >10.0 ± 0.00 A |
| OMB 20 min-60 s | 7.41 ± 0.093 A | 651 ± 18.3 B | 746.7 ± 28.45 A | >10.0 ± 0.00 A |
| OMB 10 min-30 s | 7.25 ± 0.012 A | 678 ± 17.6 B | 747.0 ± 36.12 A | >10.0 ± 0.00 A |
| OMB 10 min-60 s | 7.46 ± 0.14 A | 683 ± 20.0 B | 788.8 ± 24.14 A | >10.0 ± 0.00 A |
| Treatments | L* | a* | b* | ΔE |
|---|---|---|---|---|
| Control | 41.5 ± 4.30 A | 24.3 ± 3.91 A | 38.2 ± 3.87 A | 6.63 ± 3.42 A |
| NaClO | 37.4 ± 3.65 B | 24.2 ± 2.65 A | 38.1 ± 4.71 A | 6.89 ± 2.31 A |
| OMB | 37.0 ± 3.42 B | 25.8 ± 4.01 A | 40.4 ± 2.88 A | 6.95 ± 3.52 A |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hou, C.-Y.; Chen, Y.-R.; Wu, J.-S.; Chen, H.-L.; Hsiao, C.-P.; Liu, C.-T.; Lin, C.-M. Antibacterial Efficacy and Physiochemical Effects of Ozone Microbubble Water on Tomato. Sustainability 2022, 14, 6549. https://doi.org/10.3390/su14116549
Hou C-Y, Chen Y-R, Wu J-S, Chen H-L, Hsiao C-P, Liu C-T, Lin C-M. Antibacterial Efficacy and Physiochemical Effects of Ozone Microbubble Water on Tomato. Sustainability. 2022; 14(11):6549. https://doi.org/10.3390/su14116549
Chicago/Turabian StyleHou, Chih-Yao, Yun-Ru Chen, Jong-Shinn Wu, Hsiu-Ling Chen, Chun-Ping Hsiao, Chih-Tung Liu, and Chia-Min Lin. 2022. "Antibacterial Efficacy and Physiochemical Effects of Ozone Microbubble Water on Tomato" Sustainability 14, no. 11: 6549. https://doi.org/10.3390/su14116549
APA StyleHou, C.-Y., Chen, Y.-R., Wu, J.-S., Chen, H.-L., Hsiao, C.-P., Liu, C.-T., & Lin, C.-M. (2022). Antibacterial Efficacy and Physiochemical Effects of Ozone Microbubble Water on Tomato. Sustainability, 14(11), 6549. https://doi.org/10.3390/su14116549










