Bioactive Glass-Based Endodontic Sealer as a Promising Root Canal Filling Material without Semisolid Core Materials
Abstract
1. Introduction
1.1. Bioceramics
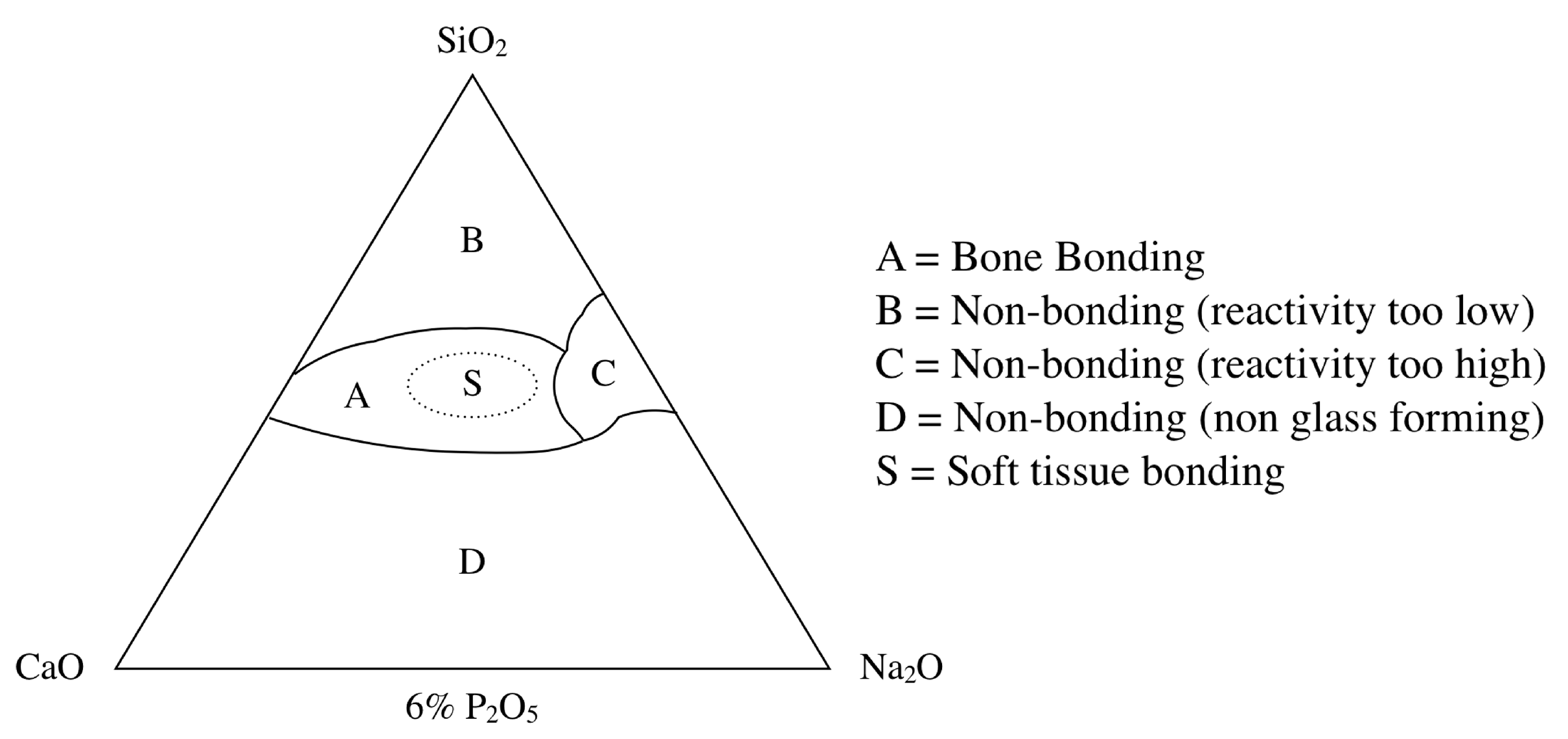
1.2. Bioactive Glass
1.3. Bioceramic-Based Root Canal Sealer
1.4. Bioactive Glass-Based Root Canal Sealer
2. Physicochemical Properties
2.1. Physical Properties
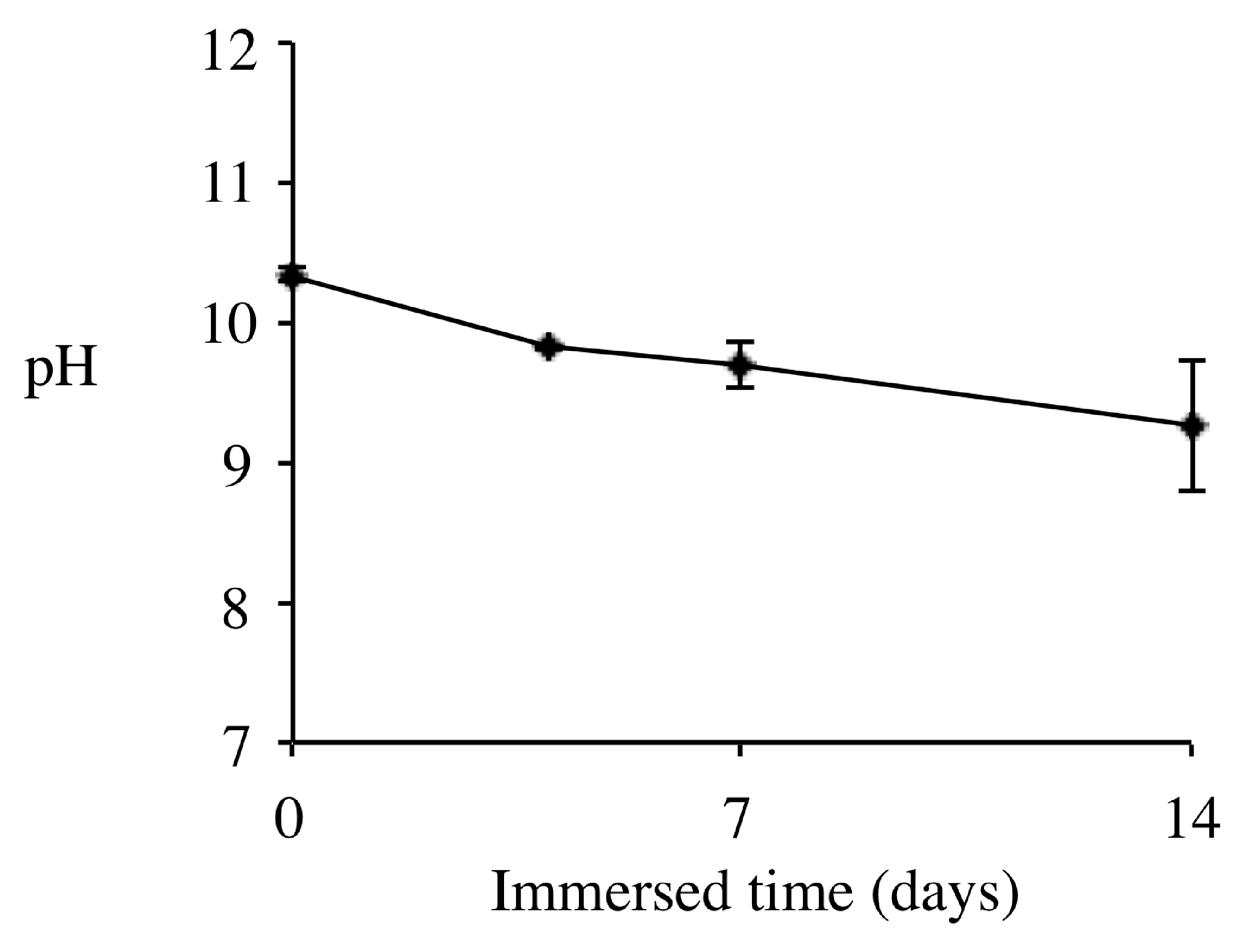
2.2. pH Change
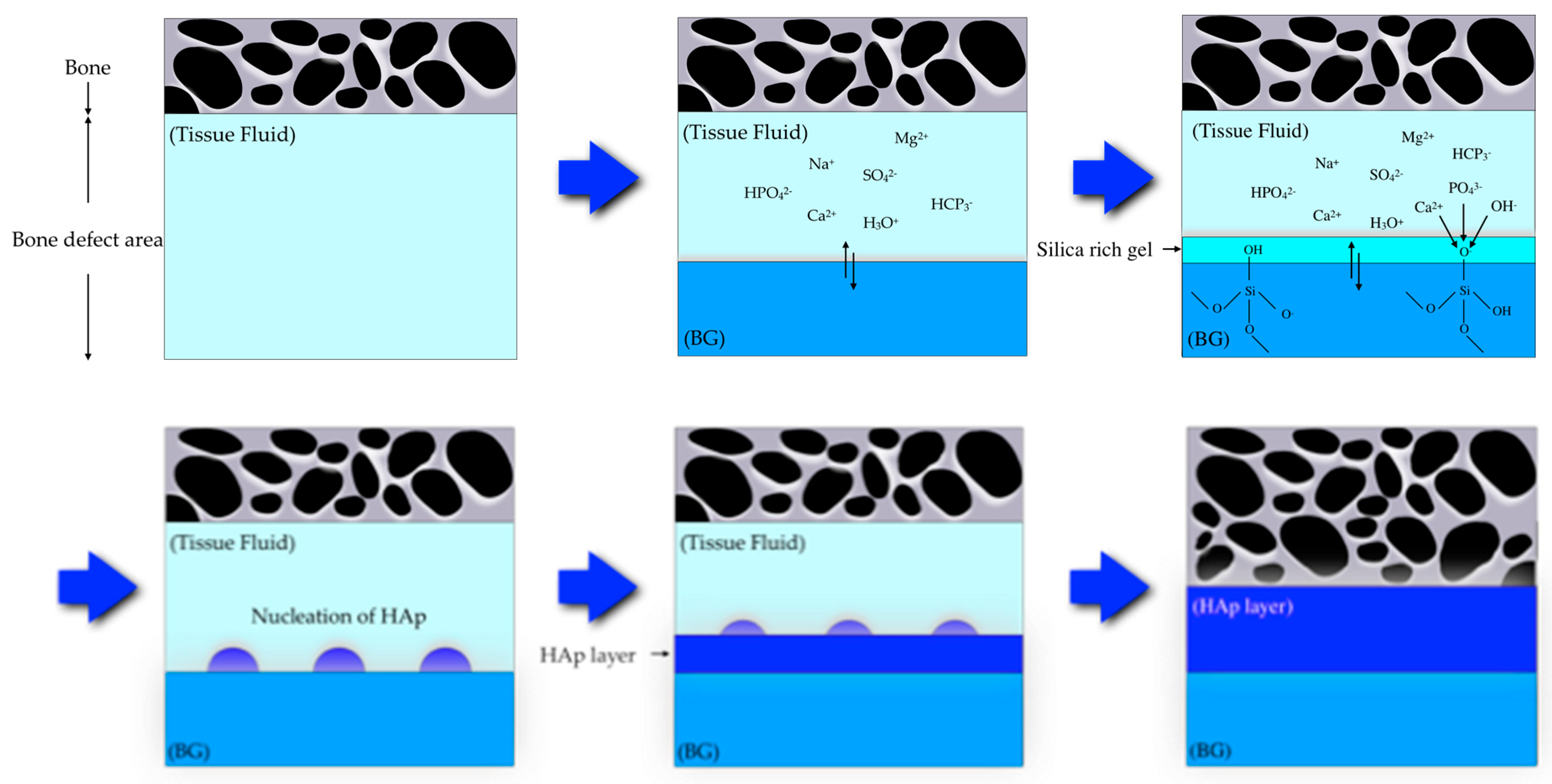
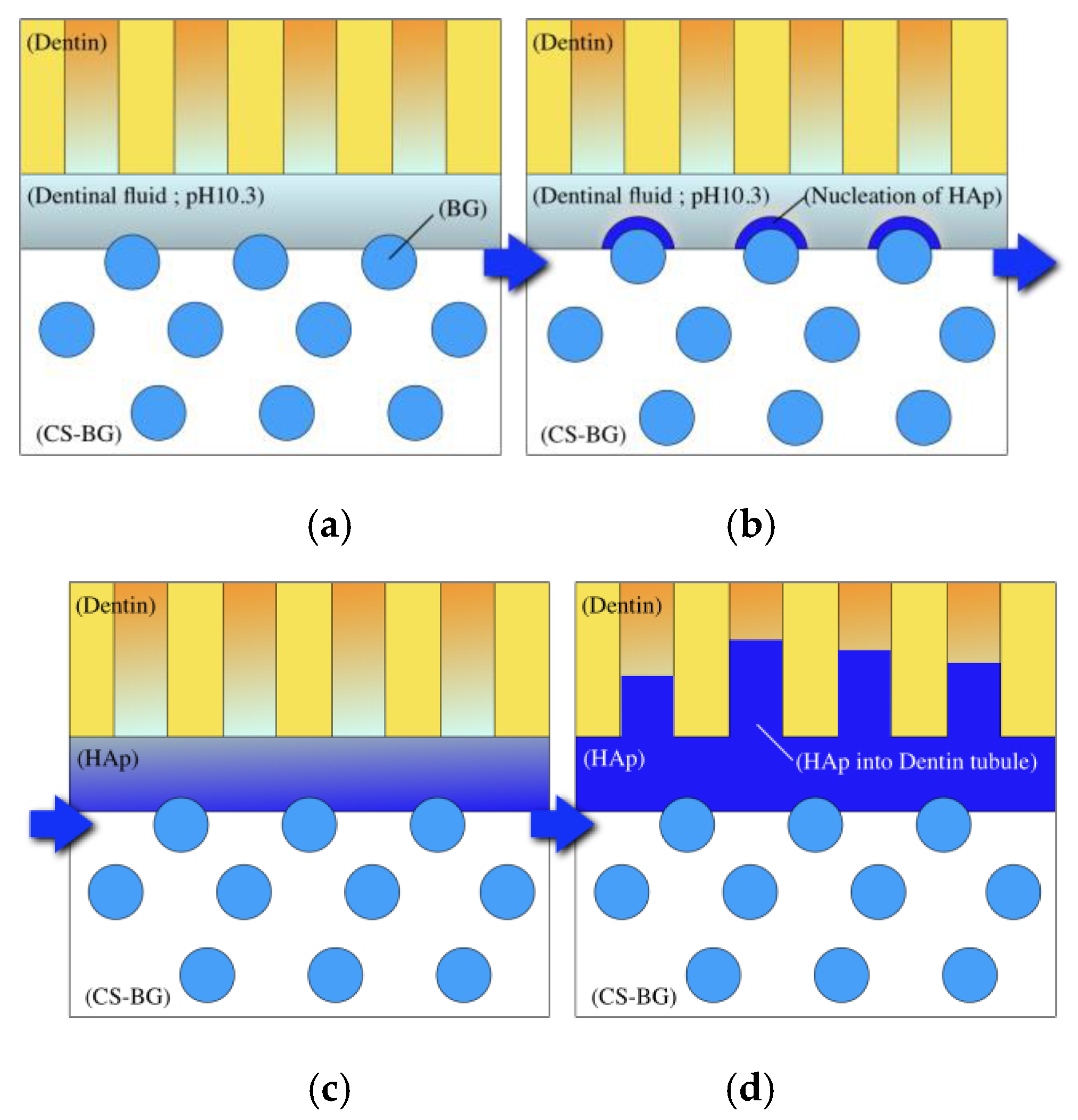
2.3. HAp Formation on the Surface of CS-BG in SBF
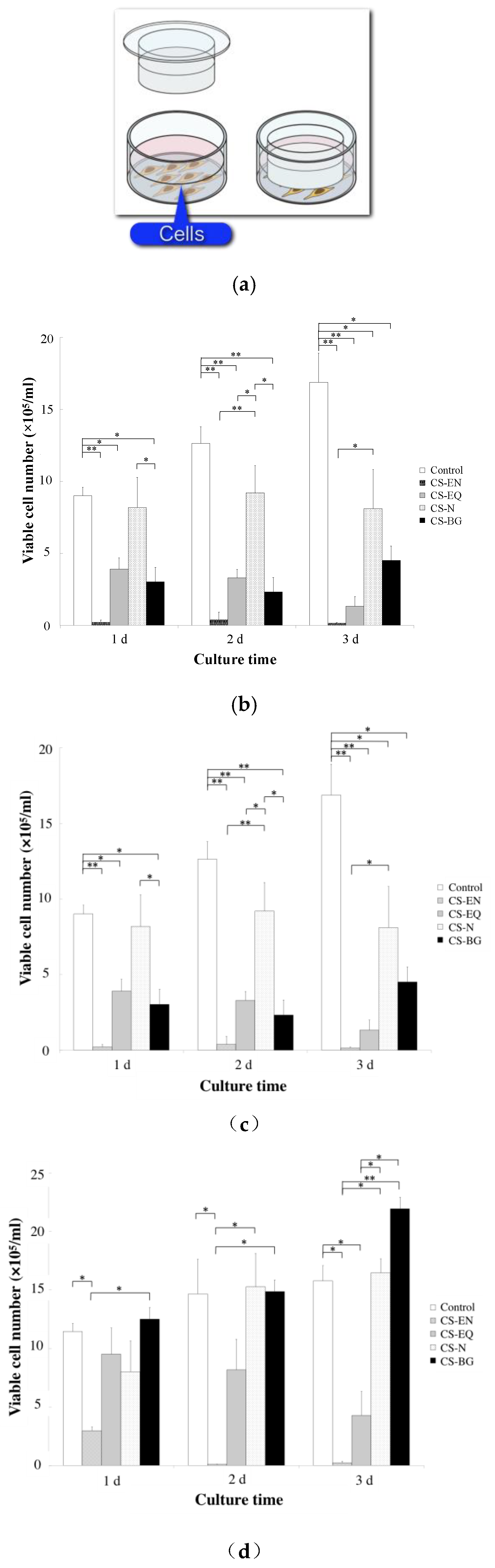
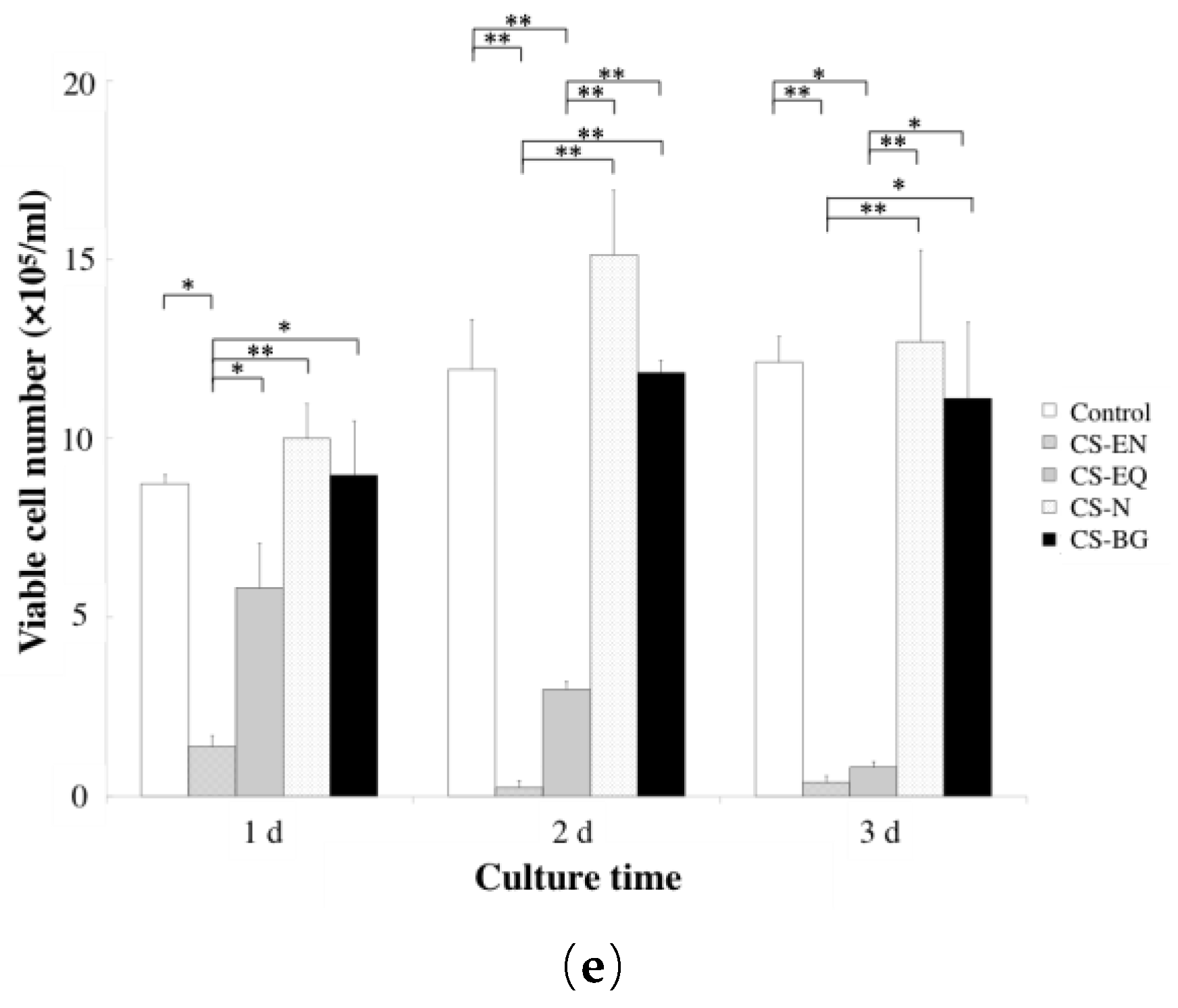
3. Biocompatibility
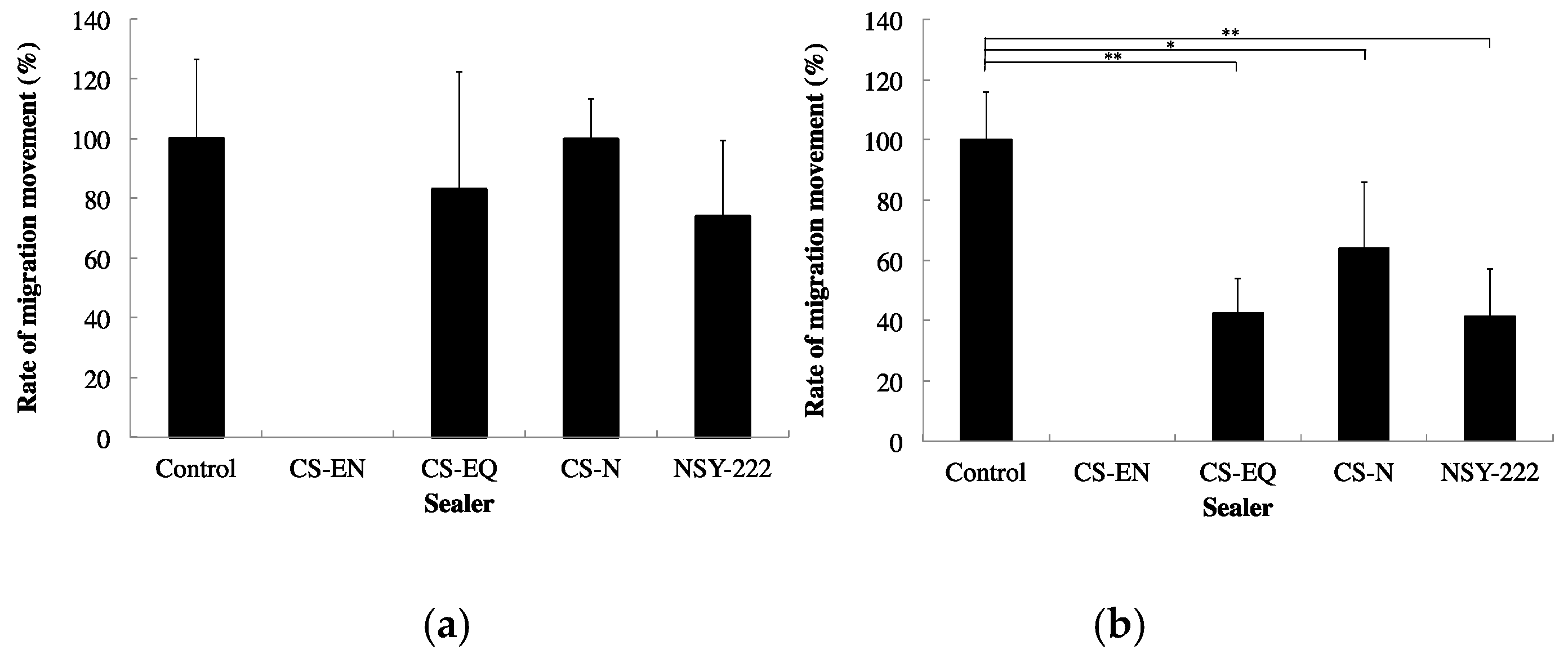
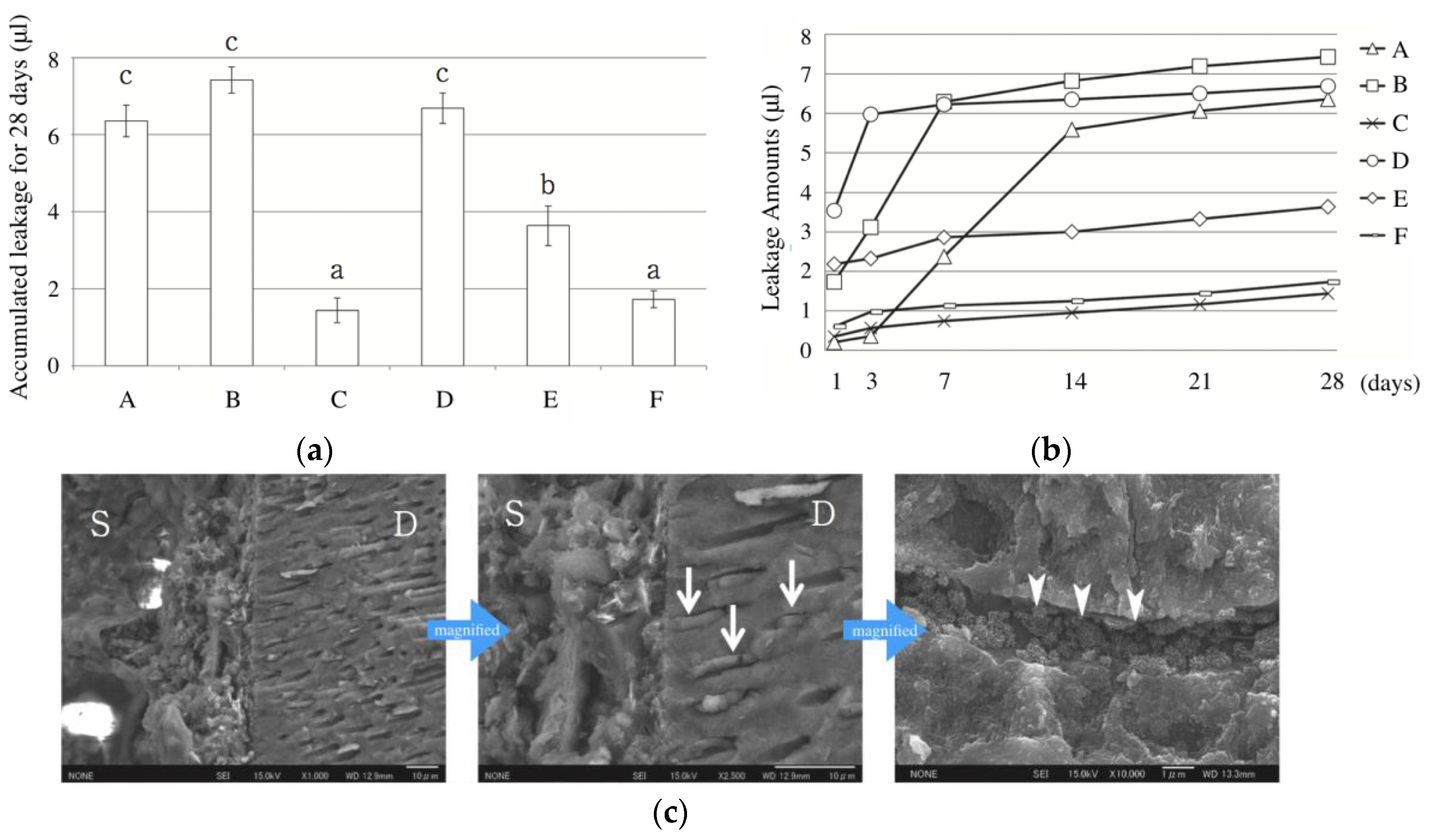
4. Sealing Ability
5. Removability
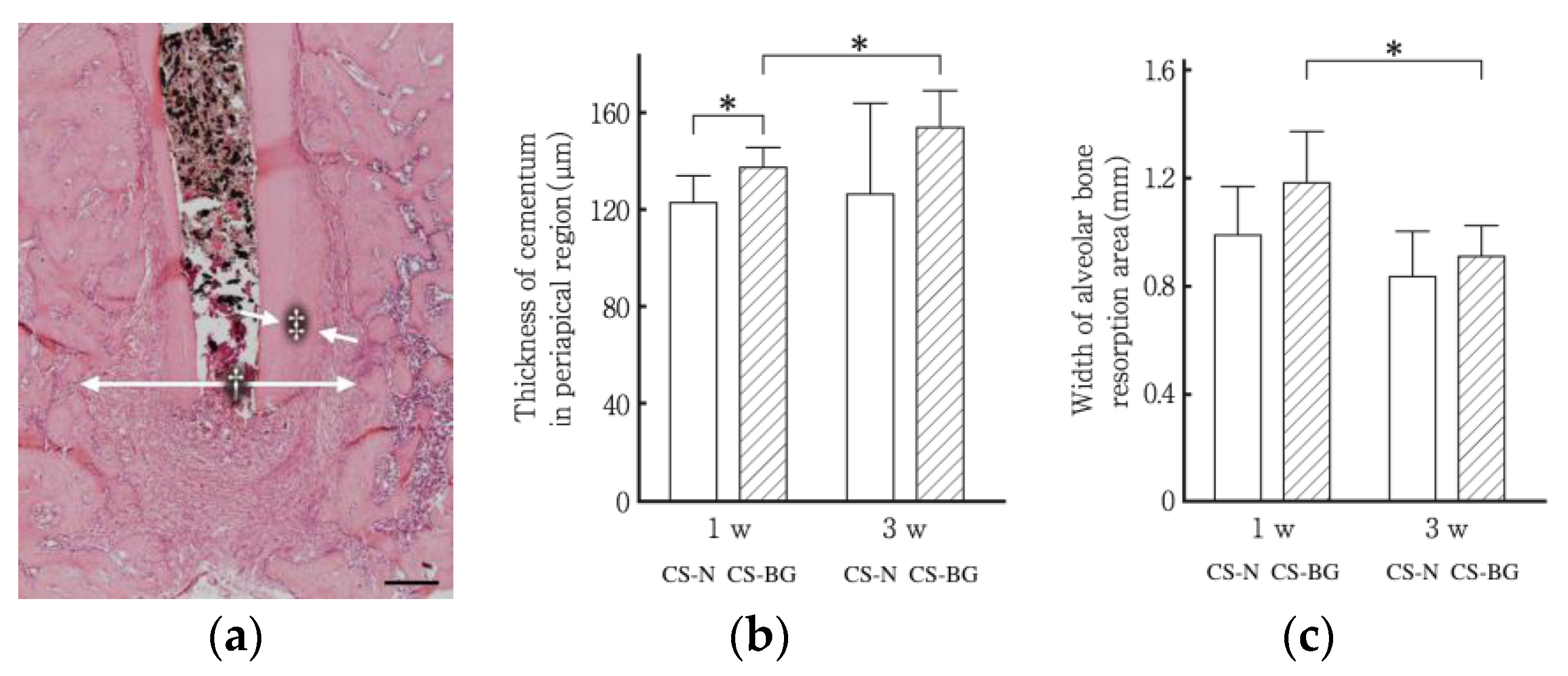
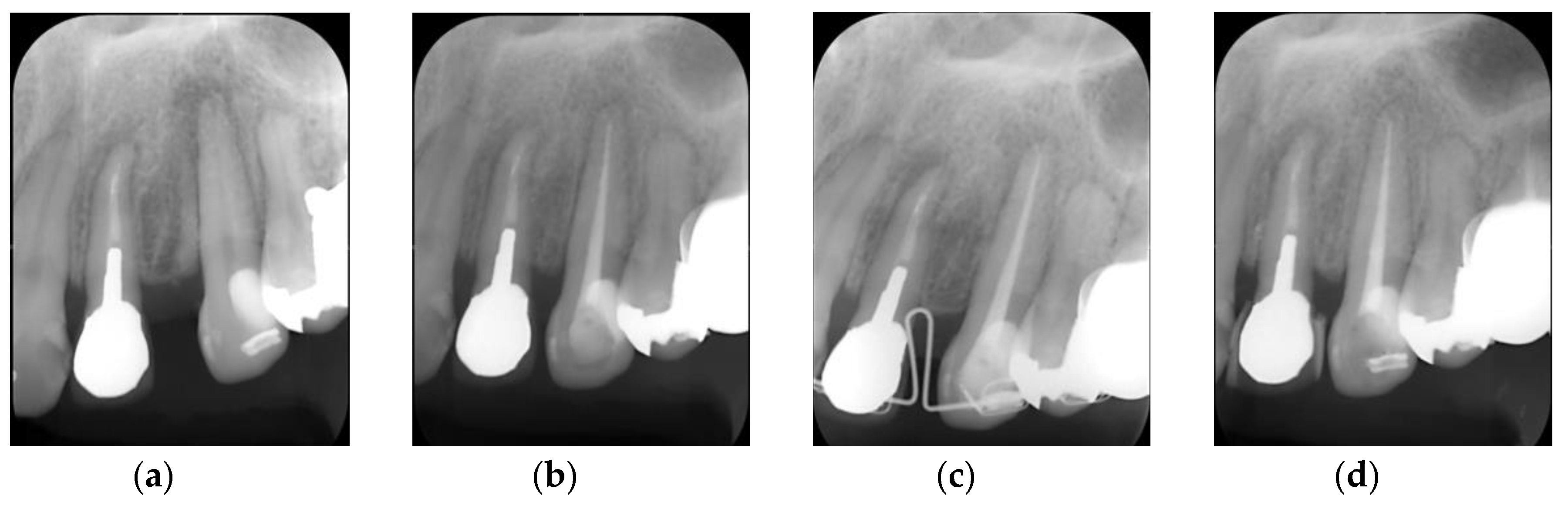
6. Clinical Performance of Bioactive Glass-Based Root Canal Sealer
7. Potential of Bioactive Glass-Based Sealer as Root Canal Filling Material Without Semisolid Core Materials
8. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Sjogren, U.; Figdor, D.; Persson, S.; Sundqvist, G. Influence of infection at the time of root filling on the outcome of endodontic treatment of teeth with apical periodontitis. Int. Endod. J. 2012, 30, 297–306. [Google Scholar] [CrossRef]
- Van Nieuwenhuysen, J.P.; Aouar, M.; D’Hoore, W. Retreatment or radiographic monitoring in endodontics. Int. Endod. J. 1994, 27, 75–81. [Google Scholar] [CrossRef]
- Rotstein, I.; Salehrabi, R.; Forrest, J.L. Endodontic treatment outcome: Survey of oral health care professionals. J. Endod. 2006, 32, 399–403. [Google Scholar] [CrossRef] [PubMed]
- Imura, N.; Pinheiro, E.T.; Gomes, B.P.; Zaia, A.A.; Ferraz, C.C.; Souza-Filho, F.J. The outcome of endodontic treatment: A retrospective study of 2000 cases performed by a specialist. J. Endod. 2007, 33, 1278–1282. [Google Scholar] [CrossRef] [PubMed]
- Ng, Y.L.; Mann, V.; Gulabivala, K. Outcome of secondary root canal treatment: A systematic review of the literature. Int. Endod. J. 2008, 41, 1026–1046. [Google Scholar] [CrossRef] [PubMed]
- Ng, Y.L.; Mann, V.; Rahbaran, S.; Lewsey, J.; Gulabivala, K. Outcome of primary root canal treatment: Systematic review of the literature. Part 2. Influence of clinical factors. Int. Endod. J. 2008, 41, 6–31. [Google Scholar] [CrossRef] [PubMed]
- Santos, J.M.; Palma, P.J.; Ramos, J.C.; Cabrita, A.S.; Friedman, S. Periapical inflammation subsequent to coronal inoculation of dog teeth root filled with resilon/epiphany in 1 or 2 treatment sessions with chlorhexidine medication. J. Endod. 2014, 40, 837–841. [Google Scholar] [CrossRef] [PubMed]
- Emmanuel, S.; Shantaram, K.; Sushil, K.C.; Manoj, L. An in vitro evaluation and comparision of apical sealing ability of three different obturation technique-lateral condensation, Obtura II, and ThermaFil. J. Int. Oral Health 2013, 5, 35–43. [Google Scholar]
- Sarkar, N.K.; Caicedo, R.; Ritwik, P.; Moiseyeva, R.; Kawashima, I. Physicochemical basis of the biologic properties of mineral trioxide aggregate. J. Endod. 2005, 31, 97–100. [Google Scholar] [CrossRef]
- Dammaschke, T.; Gerth, H.U.; Züchner, H.; Schäfer, E. Chemical and physical surface and bulk material characterization of white ProRoot MTA and two Portland cements. Dent. Mater. 2005, 21, 731–738. [Google Scholar] [CrossRef]
- Zhang, H.; Shen, Y.; Ruse, N.D.; Haapasalo, M. Antibacterial activity of endodontic sealers by modified direct contact test against Enterococcus faecalis. J. Endod. 2009, 35, 1051–1055. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.M.; Shen, Y.; Zheng, W.; Li, L.; Zheng, Y.F.; Haapasalo, M. Physical properties of 5 root canal sealers. J. Endod. 2013, 39, 1281–1286. [Google Scholar] [CrossRef] [PubMed]
- Candeiro, G.T.; Correia, F.C.; Duarte, M.A.; Ribeiro-Siqueira, D.C.; Gavini, G. Evaluation of radiopacity, pH, release of calcium ions, and flow of a bioceramic root canal sealer. J. Endod. 2012, 38, 842–845. [Google Scholar] [CrossRef] [PubMed]
- Bozeman, T.B.; Lemon, R.R.; Eleazer, P.D. Elemental analysis of crystal precipitate from gray and white MTA. J. Endod. 2006, 32, 425–428. [Google Scholar] [CrossRef]
- Reyes-Carmona, J.F.; Felippe, M.S.; Felippe, W.T. Biomineralization ability and interaction of mineral trioxide aggregate and white Portland cement with dentin in a phosphate-containing fluid. J. Endod. 2009, 35, 731–736. [Google Scholar] [CrossRef]
- Tay, F.R.; Pashley, D.H.; Rueggeberg, F.A.; Loushine, R.J.; Weller, R.N. Calcium phosphate phase transformation produced by the interaction of the Portland cement component of white mineral trioxide aggregate with a phosphate-containing fluid. J. Endod. 2007, 33, 1347–1351. [Google Scholar] [CrossRef]
- Bramante, C.M.; Demarchi, A.C.; de MOraes, I.G.; Bernadineli, N.; Garcia, R.B.; Spangberg, L.S.; Duarte, M.A. Presence of arsenic in different types of MTA and white and gray Portland cement. Oral. Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 2008, 106, 909–913. [Google Scholar] [CrossRef]
- Torabinejad, M.; Chivian, N. Clinical applications of mineral trioxide aggregate. J. Endod. 1999, 25, 197–205. [Google Scholar] [CrossRef]
- Williams, D.F. The Williams Dictionary of Biomaterials; Liverpool University Press: Liverpool, UK, 1999. [Google Scholar]
- Dubok, V.A. Bioceramics—Yesterday, Today, Tomorrow. Powder Metall. Met. Ceram. 2000, 39, 381–394. [Google Scholar] [CrossRef]
- Hulbert, S.F.; Hench, L.L.; Wilson, J. An Introduction to Bioceramics; World Scientific: Singapore, 1993; pp. 25–40. [Google Scholar]
- Hench, L.L. Bioceramics: From Concept to Clinic. J. Am. Ceram. Soc. 1991, 74, 1487–1510. [Google Scholar] [CrossRef]
- Hench, L.L. Genetic design of bioactive glass. J. Eur. Ceram. Soc. 2009, 29, 1257–1265. [Google Scholar] [CrossRef]
- Jones, J.R.; Brauer, D.S.; Hupa, L.; Greenspan, D.C. Bioglass and bioactive glasses and their impact on healthcare. Int. J. Appl. Glass Sci. 2016, 7, 423–434. [Google Scholar] [CrossRef]
- Hench, L.L. The story of Bioglass®. J. Mater. Sci. 2006, 17, 967–978. [Google Scholar] [CrossRef] [PubMed]
- Jell, G.; Stevens, M.M. Gene activation by bioactive glasses. J. Mater. Sci. Mater. Med. 2006, 17, 997–1002. [Google Scholar] [CrossRef]
- Xynos, I.D.; Edgar, A.J.; Buttery, L.D.K.; Hench, L.L.; Polak, J.M. Gene expression profiling of human osteoblasts following treatment with the ionic products of Bioglass 45S5 dissolution. J. Biomed. Mater. Res. 2001, 55, 151–157. [Google Scholar] [CrossRef]
- Hoeland, W.; Vogel, W.; Waumann, K.; Gummel, J. Interface reactions between machinable bioactive glass-ceramics and bone. J. Biomed. Mater. Res. 1985, 19, 303–312. [Google Scholar] [CrossRef]
- Hench, L.L.; Polack, J.M. Third-generation biomedical materials. Science 2002, 295, 1014–1017. [Google Scholar] [CrossRef]
- Baino, F.; Novajra, G.; Miguez-Pacheco, V.; Boccaccini, A.R.; Vitale-Brovarone, C. Bioactive glasses: Special applications outside the skeletal system. J. Non-Cryst. Solids 2016, 432, 15–30. [Google Scholar] [CrossRef]
- Kokubo, T.; Takadama, H. How useful is SBF in predicting in vivo bone bioactivity? Biomaterials 2006, 27, 2907–2915. [Google Scholar] [CrossRef]
- Gilchrist, T.; Glasby, M.; Healy, D.; Kelly, G.; Lenihan, D.; McDowall, K.; Miller, I.; Myles, L. In vitro nerve repair-In vivo. The reconstruction of peripheral nerves by entubulation with biodegradable glass tubes—A preliminary report. Br. J. Plast. Surg. 1998, 51, 231–237. [Google Scholar] [CrossRef]
- Oguntebi, B.; Clark, A.; Wilsin, J. Pulp capping with bioglass and autologous demineralized dentin in miniature swine. J. Dent. Res. 1993, 72, 484–489. [Google Scholar] [CrossRef] [PubMed]
- Damas, B.A.; Wheater, M.A.; Bringas, J.S.; Hoen, M.M. Cytotoxicity comparison of mineral trioxide aggregates and EndoSequence bioceramic root repair materials. J. Endod. 2011, 37, 372–375. [Google Scholar] [CrossRef] [PubMed]
- Leal, F.; De-Deus, G.; Brandão, C.; Luna, A.S.; Fidel, S.R.; Souza, E.M. Comparison of the root-end seal provided by bioceramic repair cements and White MTA. Int. Endod. J. 2011, 44, 662–668. [Google Scholar] [CrossRef] [PubMed]
- Koch, K.; Brave, D. Bioceramic technology: The game changer in endodontics. Endod. Pract. 2009, 2, 17–21. [Google Scholar]
- Hess, D.; Solomon, E.; Spears, R.; He, J. Retreatability of a bioceramic root canal sealing material. J. Endod. 2011, 37, 1547–1549. [Google Scholar] [CrossRef] [PubMed]
- Loushine, B.A.; Bryan, T.E.; Looney, S.W.; Gillen, B.M.; Loushine, R.J.; Weller, R.N.; Pashley, D.H.; Tay, F.R. Setting properties and cytotoxicity evaluation of a premixed bioceramic root canal sealer. J. Endod. 2011, 37, 673–677. [Google Scholar] [CrossRef]
- Massi, S.; Tanomaru-Filho, M.; Silva, G.F.; Duarte, M.A.; Grizzo, L.T.; Buzalaf, M.A.; Guerreiro-Tanomaru, J.M. pH, calcium ion release, and setting time of an experimental mineral trioxide aggregate-based root canal sealer. J. Endod. 2011, 37, 844–846. [Google Scholar] [CrossRef]
- Walker, M.P.; Diliberto, A.; Lee, C. Effect of setting conditions on mineral trioxide aggregate flexural strength. J. Endod. 2006, 32, 334–336. [Google Scholar] [CrossRef]
- Chen, L.; Suh, B.I. Cytotoxicity and biocompatibility of resin-free and resin modified direct pulp capping materials: A state-of-the-art review. Dent. Mater. J. 2017, 36, 1–7. [Google Scholar] [CrossRef]
- Jafari, F.; Aghazadeh, M.; Jafari, S.; Khaki, F.; Kabiri, F. In vitro Cytotoxicity Comparison of MTA Fillapex, AH-26 and Apatite Root Canal Sealer at Different Setting Times. Iran. Endod. J. 2017, 12, 162–167. [Google Scholar]
- Silva, E.J.; Rosa, T.P.; Herrera, D.R.; Jacinto, R.C.; Gomes, B.P.; Zaia, A.A. Evaluation of cytotoxicity and physicochemical properties of calcium silicate-based endodontic sealer MTA Fillapex. J. Endod. 2013, 39, 274–277. [Google Scholar] [CrossRef] [PubMed]
- Oltra, E.; Cox, T.C.; LaCourse, M.R.; Johnson, J.D.; Paranjpe, A. Retreatability of two endodontic sealers, EndoSequence BC Sealer and AH Plus: A micro-computed tomographic comparison. Restor. Dent. Endod. 2017, 42, 19–26. [Google Scholar] [CrossRef]
- Gandolfi, M.G.; Siboni, F.; Prati, C. Properties of a novel polysiloxane-guttapercha calcium silicate-bioglass-containing root canal sealer. Dent. Mater. 2016, 32, 113–126. [Google Scholar] [CrossRef] [PubMed]
- Akcay, M.; Arslan, H.; Durmus, N.; Mese, M.; Capar, I.D. Dentinal tubule penetration of AH Plus, iRoot SP, MTA Fillapex, and GuttaFlow Bioseal root canal sealers after different final irrigation procedures: A confocal microscopic study. Lasers. Surg. Med. 2016, 48, 70–76. [Google Scholar] [CrossRef]
- Collado-Gonzalez, M.; Tomas-Catala, C.J.; Onate-Sanchez, R.E.; Moraleda, J.M.; Rodríguez-Lozano, F.J. Cytotoxicity of GuttaFlow Bioseal, GuttaFlow2, MTA Fillapex, and AH Plus on human periodontal ligament stem cells. J. Endod. 2017, 43, 816–822. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Lozano, F.J.; Collado-González, M.; Tomás-Catalá, C.J.; García-Bernal, D.; López, S.; Oñate-Sánchez, R.E.; Moraleda, J.M.; Murcia, L. GuttaFlow Bioseal promotes spontaneous differentiation of human periodontal ligament stem cells into cementoblast-like cells. Dent. Mater. 2019, 35, 114–124. [Google Scholar] [CrossRef]
- Kokubo, T.; Kushitani, H.; Sakka, S.; Kitsugi, T.; Yamamuro, T. Solutions able to reproduce in vivo surface-structure changes in bioactive glass-ceramic A-W. J. Biomed. Mater. Res. 1990, 24, 721–734. [Google Scholar] [CrossRef]
- Kokubo, T.; Ito, S.; Huang, Z.T.; Hayashi, T.; Sakka, S.; Kitsugi, T.; Yamamuro, T. Ca, P-rich layer formed on high-strength bioactive glass-ceramic A-W. J. Biomed. Mater. Res. 1990, 24, 331–343. [Google Scholar] [CrossRef]
- Washio, A.; Nakagawa, A.; Nishihara, T.; Maeda, H.; Kitamura, C. Physicochemical properties of newly developed bioactive glass cement and its effects on various cells. J. Biomed. Mater. Res. B Appl. Biomater. 2015, 103, 373–380. [Google Scholar] [CrossRef]
- Orstavik, D. Materials used for root canal obturation: Technical, biological and clinical testing. Endod. Top. 2005, 12, 25–38. [Google Scholar] [CrossRef]
- Al-Haddad, A.; Ab Aziz, C.; Zeti, A. Bioceramic-Based Root Canal Sealers: A Review. Int. J. Biomater. 2016. [Google Scholar] [CrossRef] [PubMed]
- Williams, D.F. Definitions in Biomaterials: Proceedings of a Consensus Conference of the European Society for Biomaterials, Chester, UK, 3–5 March 1986; Elsevier: Amsterdam, The Netherlands; New York, NY, USA, 1987; Volume 4. [Google Scholar]
- Schmalz, G. Use of cell cultures for toxicity testing of dental materials—Advantages and limitations. J. Dent. 1994, 22, S6–S11. [Google Scholar] [CrossRef]
- Salles, L.P.; Gomes-Cornélio, A.L.; Guimarães, F.C.; Herrera, B.S.; Bao, S.N.; Rossa-Junior, C. Guerreiro-Tanomaru, J.M.; Tanomaru-Filho, M. Mineral trioxide aggregate-based endodontic sealer stimulates hydroxyapatite nucleation in human osteoblast-like cell culture. J. Endod. 2012, 38, 971–976. [Google Scholar] [CrossRef]
- Jung, S.; Sielker, S.; Hanisch, M.R.; Libricht, V.; Schäfer, E.; Dammaschke, T. Cytotoxic effects of four different root canal sealers on human osteoblasts. PLoS ONE 2018, 13, e0194467. [Google Scholar] [CrossRef] [PubMed]
- Bae, W.J.; Chang, S.W.; Lee, S.I.; Kum, K.Y.; Bae, K.S.; Kim, E.C. Human periodontal ligament cell response to a newly developed calcium phosphate-based root canal sealer. J. Endod. 2010, 36, 1658–1663. [Google Scholar] [CrossRef]
- Collado-González, M.; García-Bernal, D.; Oñate-Sánchez, R.E.; Ortolani-Seltenerich, P.S.; Lozano, A.; Forner, L.; Llena, C.; Rodríguez-Lozano, F.J. Biocompatibility of three new calcium silicate-based endodontic sealers on human periodontal ligament stem cells. Int. Endod. J. 2017, 50, 875–884. [Google Scholar] [CrossRef]
- Lee, J.K.; Kim, S.; Lee, S.; Kim, H.C.; Kim, E. In Vitro Comparison of Biocompatibility of Calcium Silicate-Based Root Canal Sealers. Materials 2019, 12, 2411. [Google Scholar] [CrossRef]
- Washio, A.; Yoshii, S.; Morotomi, T.; Maeda, H.; Kitamura, C. Effects of bioactive glass based sealer on cell migration ability and viability of periodontal ligament cells and osteoblast-like cells. Jpn. J. Conserv. Dent. 2017, 60, 96–104. [Google Scholar]
- Washio, A.; Kitamura, C. The next-generation bioacvite glass-based root canal sealer inducing the ideal wound healing environment of periapical tissue 〜Nishika Canal Sealer BG〜. Dent. Diam. 2017, 42, 178–183. [Google Scholar]
- Morotomi, T.; Hanada, K.; Washio, A.; Yoshii, S.; Matsuo, K.; Kitamura, C. Effect of newly-developed bioactive glass root canal sealer on periapical tissue of rat’s molar. Jpn. J. Conserv. Dent. 2017, 60, 120–127. [Google Scholar]
- Wilcox, L.R. Endodontic retreatment: Ultrasonics and chloroformas the final step in reinstrumentation. J. Endod. 1989, 15, 125–128. [Google Scholar] [CrossRef]
- Schirrmeister, J.F.; Wrbas, K.T.; Meyer, K.M.; Altenburger, M.J.; Hellwig, E. Efficacy of different rotary instruments for gutta-percha removal in root canal retreatment. J. Endod. 2006, 32, 469–472. [Google Scholar] [CrossRef] [PubMed]
- Gomes-Filho, J.E.; Watanabe, S.; Cintra, L.T.; Nery, M.J.; Dezan-Júnior, E.; Queiroz, I.O.; Lodi, C.S.; Basso, M.D. Effect of MTA-based sealer on the healing of periapical lesions. J. Appl. Oral Sci. 2013, 21, 235–242. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, R.S. Adhesive dentistry and endodontics. Part 2: Bonding in the root canal system—The promise and the problems: A review. J. Endod. 2016, 32, 1125–1134. [Google Scholar] [CrossRef]
- Nagas, E.; Altundasar, E.; Serper, A. The effect of master point taper on bond strength and apical sealing ability of different root canal sealers. Oral. Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 2009, 107, 61–64. [Google Scholar] [CrossRef]
- Lin, Z.; Ling, J.; Fang, J.; Liu, F.; He, J. Physicochemical properties, sealing ability, bond strength and cytotoxicity of a new dimethacrylate-based root canal sealer. J. Formos. Med. Assoc. 2010, 109, 819–827. [Google Scholar] [CrossRef]
- Fang, J.; Mai, S.; Ling, J.; Lin, Z.; Huang, X. In vitro evaluation of bond strength and sealing ability of a new low-shrinkage, methacrylate resin-based root canal sealer. J. Formos. Med. Assoc. 2012, 111, 340–346. [Google Scholar] [CrossRef][Green Version]
- Yoshii, S.; Washio, A.; Morotomi, T.; Kitamura, C. Root canal sealing ability of bioactive glass-based sealer and its effects on dentin. Jpn. J. Conserv. Dent. 2016, 59, 463–471. [Google Scholar]
- Wilcox, L.R.; Krell, K.V.; Madison, S.; Rittman, B. Endodontic retreatment: Evaluation of gutta-percha and sealer removal and canal reinstrumentation. J. Endod. 1987, 9, 453–457. [Google Scholar] [CrossRef]
- Reddy, S.; Neelakantan, P.; Saghiri, M.A.; Lotfi, M.; Subbarao, C.V.; Garcia-Godoy, F.; Gutmann, J.L. Removal of gutta-percha/zinc-oxide-eugenol sealer or gutta-percha/epoxy resin sealer from severely curved canals: An in vitro study. Int. J. Dent. 2011, 2011, 541831. [Google Scholar] [CrossRef]
- Uzunoglu, E.; Yilmaz, Z.; Sungur, D.D.; Altundasar, E. Retreatability of Root Canals Obturated Using Gutta-Percha with Bioceramic, MTA and Resin-Based Sealers. Iran. Endod. J. 2015, 10, 93–98. [Google Scholar]
- Washio, A.; Yoshii, S.; Morotomi, T.; Kitamura, C. Evaluation of removability of root canals filled using bioactive glass based sealer. Jpn. J. Conserv. Dent. 2017, 60, 14–21. [Google Scholar]
- Endodontics: Colleagues for Excellence. Canal preparation and obturation: An updated view of the two pillars of nonsurgical endodontics. In American Association of Endodontists; Dental Professional Community: Chicago, IL, USA, 2016; pp. 1–8. [Google Scholar]
- Treatment standards. In American Association of Endodontists; Dental Professional Community: Chicago, IL, USA, 2018; pp. 1–20.
- Michelle, L.; Johnathon, W.; Gary, H.; Jeffrey, S.; Rufus, C. Current Trends in Endodontic Practice: Emergency Treatments and Technological Armamentarium. J. Endod. 2009, 35, 35–39. [Google Scholar]
- Gina, M.S.; Wael, S.; Christine, M.S.; Brian, W. Current Trends in Endodontic Treatment by General Dental Practitioners: Report of a United States National Survey. J. Endod. 2014, 40, 618–624. [Google Scholar]
- Raoof, M.; Zeini, N.; Haghani, J.; Sadr, S.; Mohammadalizadeh, S. Preferred materials and methods employed for endodontic treatment by Iranian general practitioners. Iran. Endod. J. 2015, 10, 112–116. [Google Scholar]
- Al-Omari, W.M. Survey of attitudes, materials and methods employed in endodontic treatment by general dental practitioners in North Jordan. BMC Oral Health 2004, 4, 1. [Google Scholar] [CrossRef]
- Gupta, R.; Rai, R. The adoption of new endodontic technology by Indian dental practitioners: A questionnaire survey. J. Clin. Diagn. Res. 2013, 7, 2610–2614. [Google Scholar] [CrossRef]
- Germain, S.; Meetu, K.; Issam, K.; Alfred, N.; Carla, Z. Impact of the Root Canal Taper on the Apical Adaptability of Sealers used in a Single-cone Technique: A Micro-Computed Tomography Study. J. Contemp. Dent. Pract. 2018, 19, 808–815. [Google Scholar]
- Chybowski, E.A.; Glickman, G.N.; Patel, Y.; Fleury, A.; Solomon, E.; He, J. Clinical Outcome of Non-Surgical Root Canal Treatment Using a Single-cone Technique with EndoSequence Bioceramic Sealer: A Retrospective Analysis. J. Endod. 2018, 44, 941–945. [Google Scholar] [CrossRef]

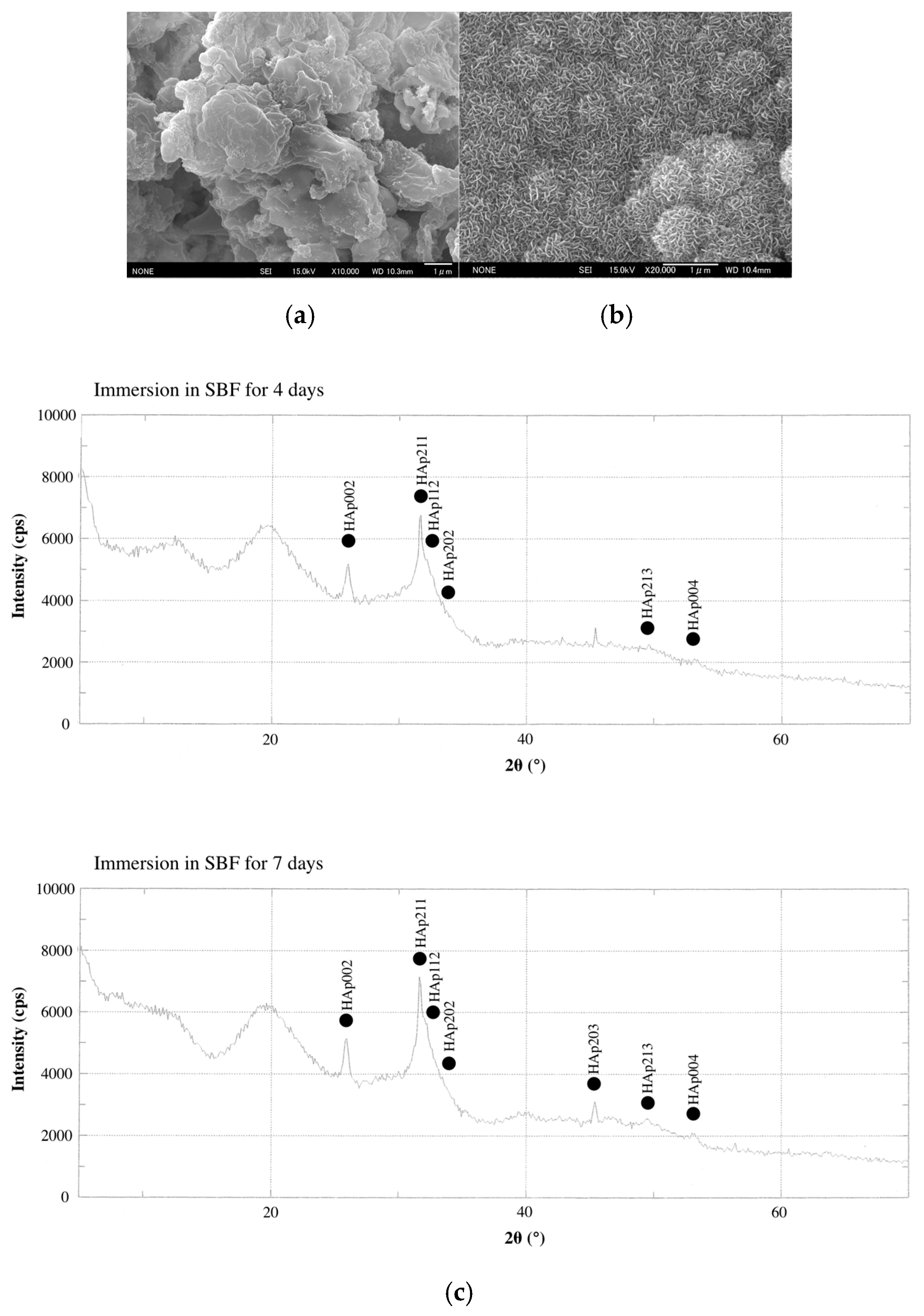
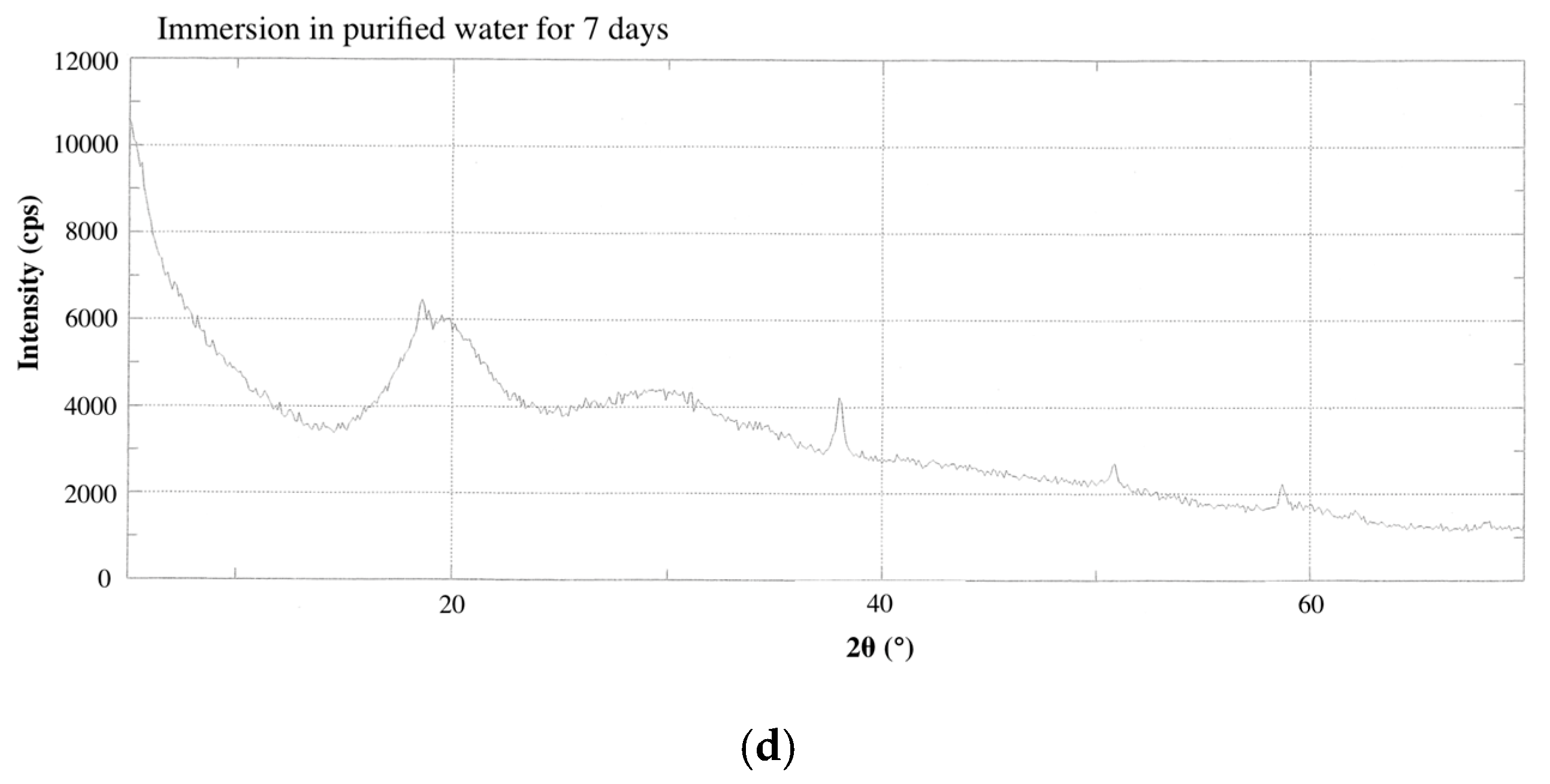


| Flow | 28.7 mm | Solubility | 0.5% |
|---|---|---|---|
| Working time | 15 mm | Disintegration | None |
| Setting time | 180 min | Radiopacity | 5 mmAl. |
| Film thickenss | 27.9 μm |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Washio, A.; Morotomi, T.; Yoshii, S.; Kitamura, C. Bioactive Glass-Based Endodontic Sealer as a Promising Root Canal Filling Material without Semisolid Core Materials. Materials 2019, 12, 3967. https://doi.org/10.3390/ma12233967
Washio A, Morotomi T, Yoshii S, Kitamura C. Bioactive Glass-Based Endodontic Sealer as a Promising Root Canal Filling Material without Semisolid Core Materials. Materials. 2019; 12(23):3967. https://doi.org/10.3390/ma12233967
Chicago/Turabian StyleWashio, Ayako, Takahiko Morotomi, Shinji Yoshii, and Chiaki Kitamura. 2019. "Bioactive Glass-Based Endodontic Sealer as a Promising Root Canal Filling Material without Semisolid Core Materials" Materials 12, no. 23: 3967. https://doi.org/10.3390/ma12233967
APA StyleWashio, A., Morotomi, T., Yoshii, S., & Kitamura, C. (2019). Bioactive Glass-Based Endodontic Sealer as a Promising Root Canal Filling Material without Semisolid Core Materials. Materials, 12(23), 3967. https://doi.org/10.3390/ma12233967