Industrial Waste Treatment by ETS-10 Ion Exchanger Material
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Characterization
2.3. Preparation of the Samples and Aqueous Systems
2.4. Ion Exchanger Procedures
3. Results
3.1. Characterization of Zinc Ferrite
3.1.1. ICP-MS: Elementary Analysis
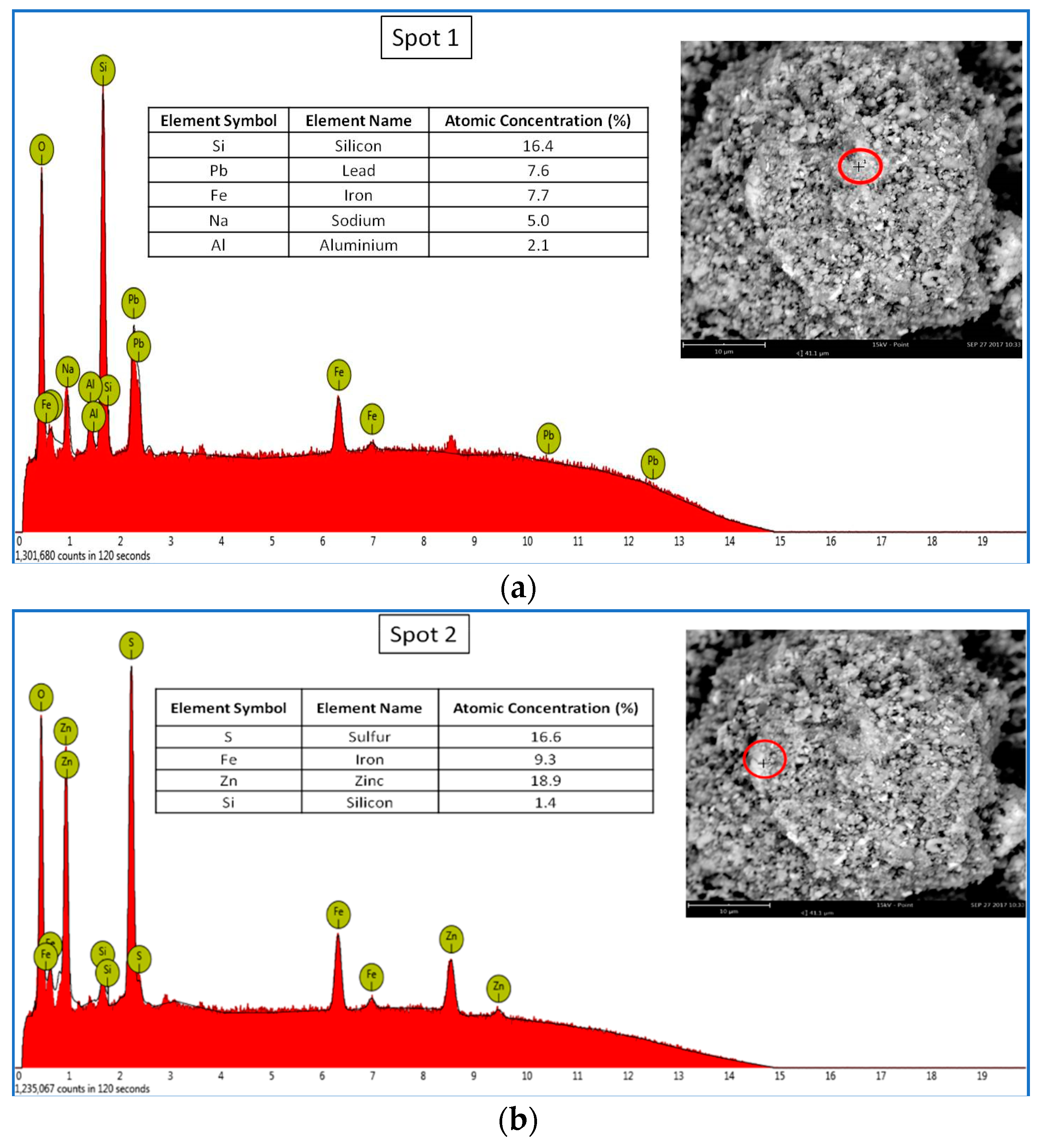
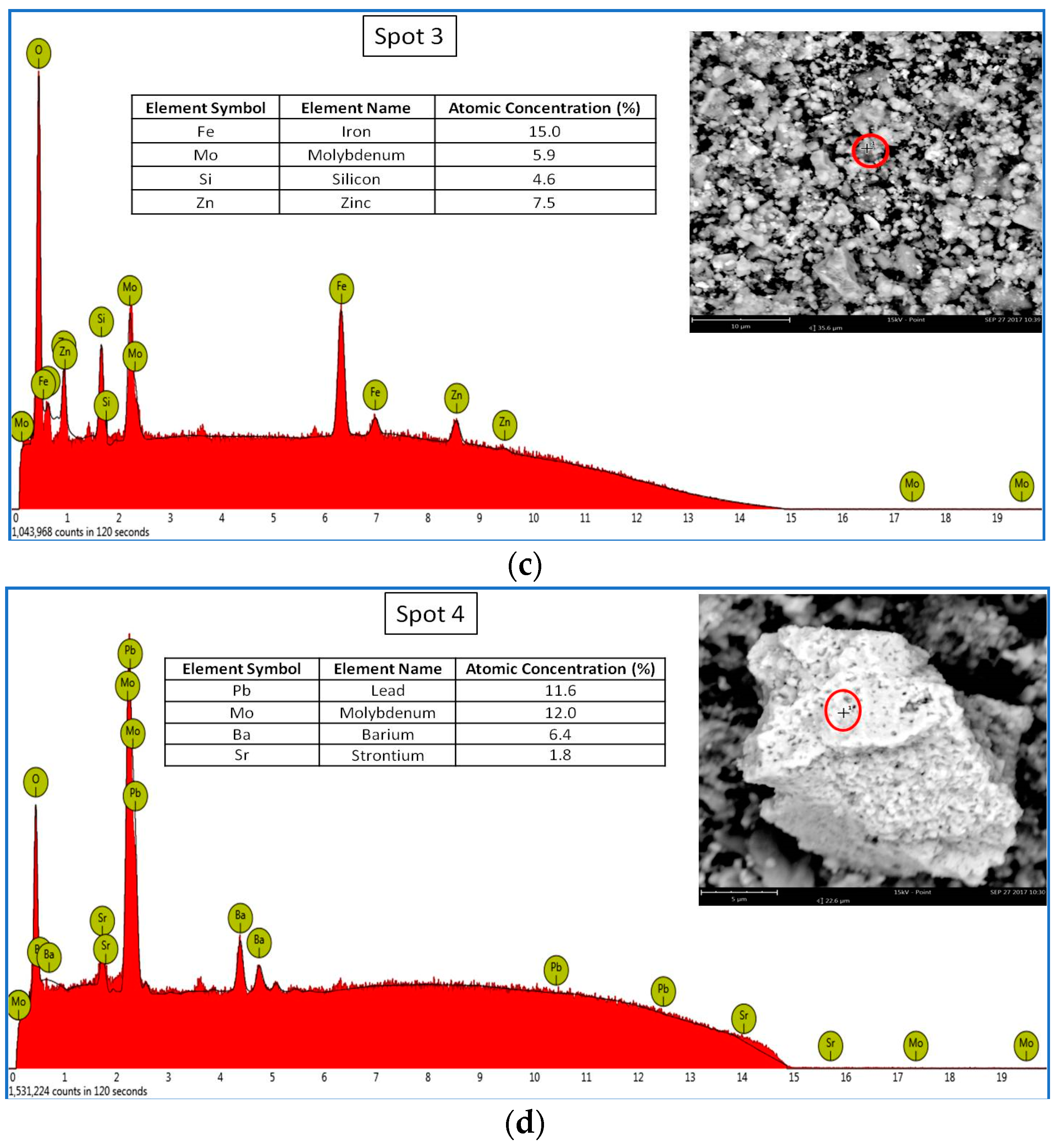
3.1.2. Scanning Electron Microscopy and Microanalysis
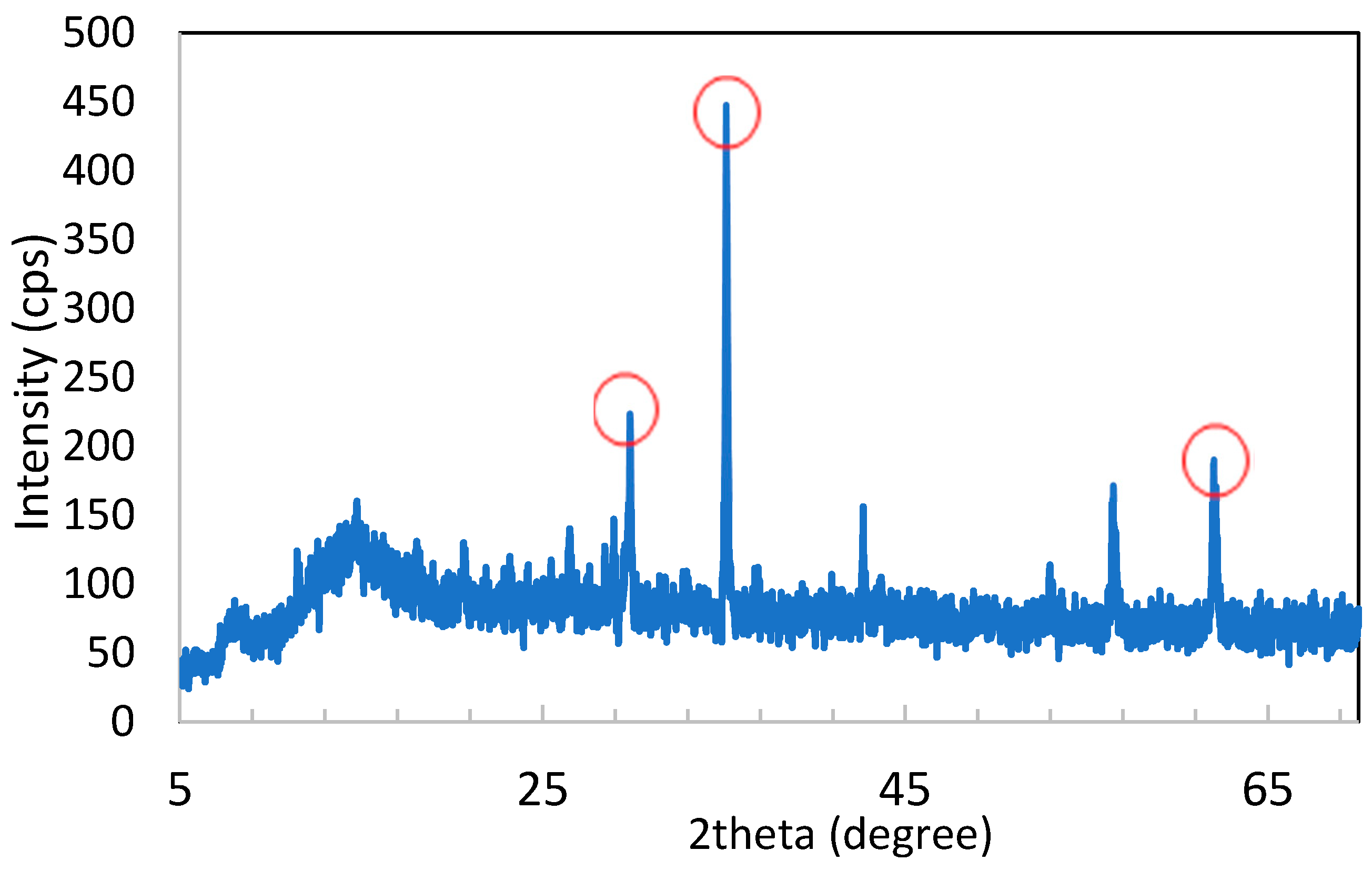
3.1.3. X-ray Diffraction Analysis
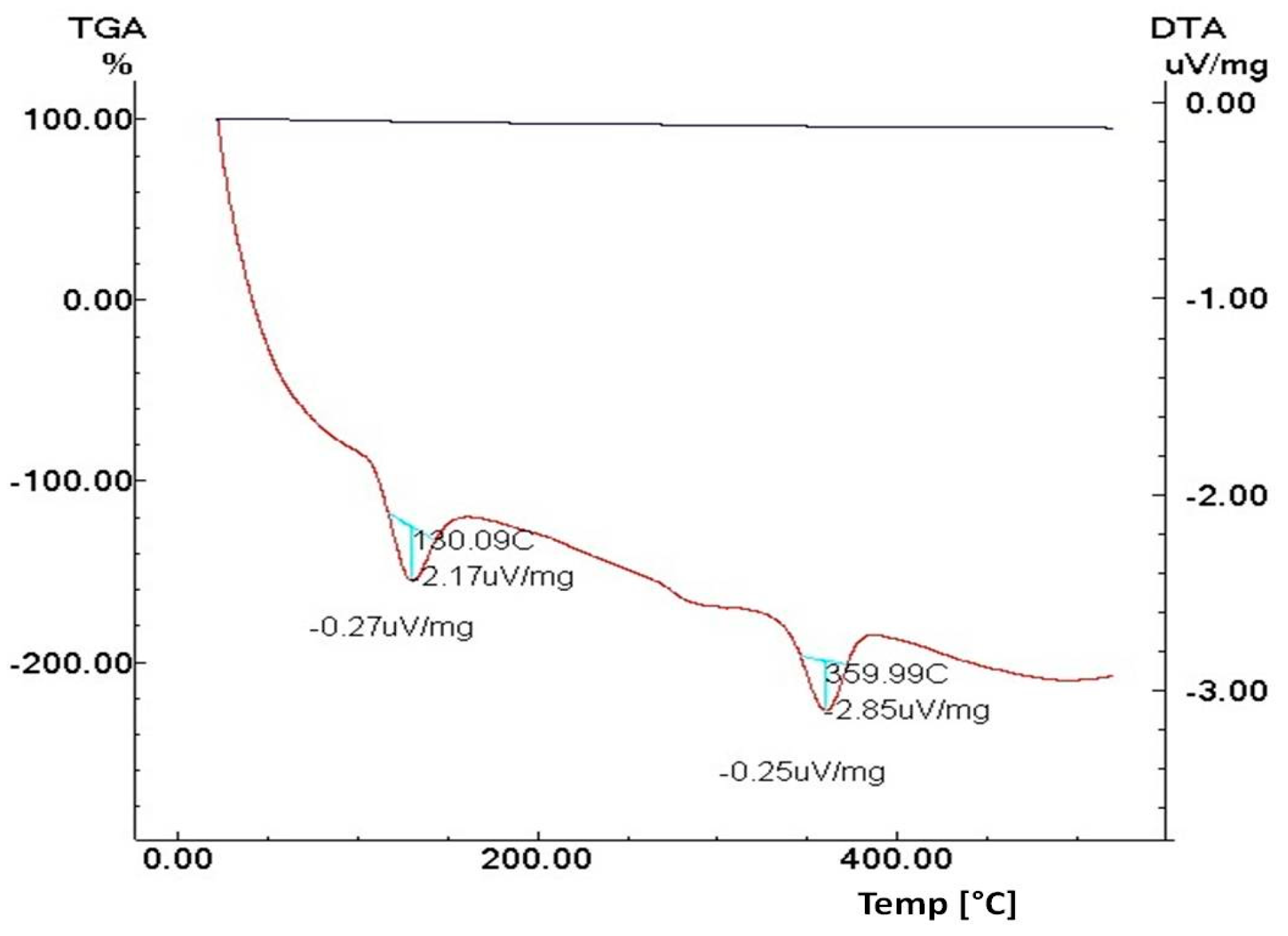
3.1.4. Thermal Analysis
3.1.5. pH and Conductivity
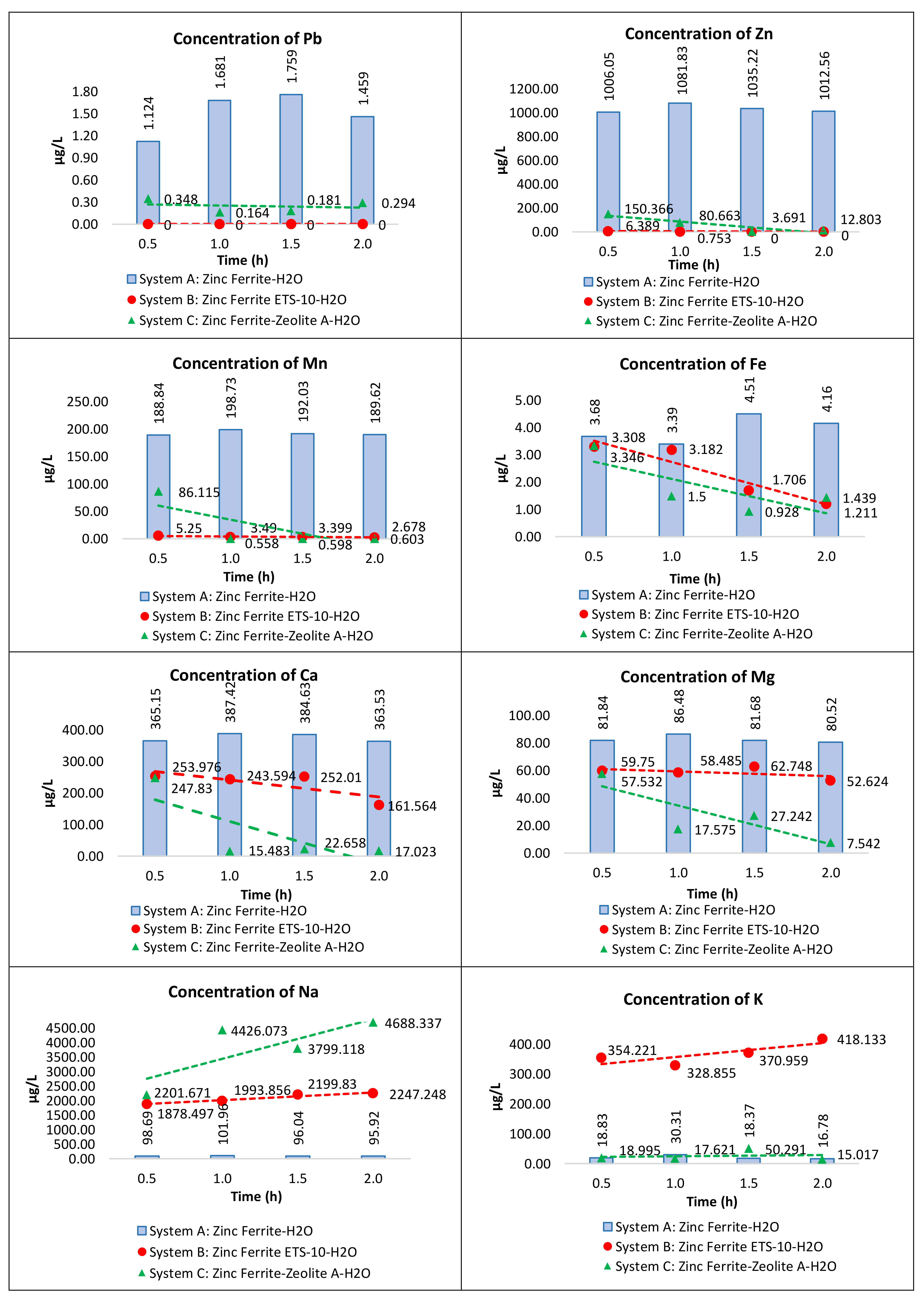
3.2. Removal of Elements Released by Zinc Ferrite in Aqueous Systems by ETS-10 Ion Exchanger
3.3. Comparison between the ETS-10 Phase and Commercial Zeolite A (LTA) in the Removal of Metals from Water Contaminated by Zinc Ferrite
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Giusti, L. A review of waste management practices and their impact on human health. Waste Manag. 2009, 29, 2227–2239. [Google Scholar] [CrossRef] [PubMed]
- Anand Kumar Varma, S. Principles of Industrial Waste Management; Covali, A., Ed.; LAP Lambert Academic Publishing: Saarbrücken, Germany, 2017; ISBN 978-620-2-01127-3. [Google Scholar]
- Donat, R.; Akdogan, A.; Erdem, E.; Cetisli, H. Thermodynamics of Pb2+ and Ni2+ adsorption onto natural bentonite from aqueous solutions. J. Colloid Interface Sci. 2005, 286, 43–52. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, K.M.; Akashah, T. Lead removal from wastewater using faujasite tuff. Environ. Geol. 2004, 46, 865–870. [Google Scholar] [CrossRef]
- Sels, B.; Kustov, L. Zeolites and Zeolite-like Materials, 1st ed.; Elsevier: Amsterdam, The Netherlands, 2016; ISBN 9780444635143. [Google Scholar]
- Wang, S.; Peng, Y. Natural zeolites as effective adsorbents in water and wastewater treatment. Chem. Eng. J. 2010, 156, 11–24. [Google Scholar]
- Breck, D.W. Zeolite Molecular Sieves; Wiley: New York, NY, USA, 1974. [Google Scholar]
- Al-Attar, L.; Dyer, A.; Harjula, R. Uptake of radionuclides on microporous and layered ion exchange materials. J. Mater. Chem. 2003, 13, 2963–2968. [Google Scholar] [CrossRef]
- Al-Harahsheh, M.S.; Al Zboon, K.; Al-Makhadmeh, L.; Hararah, M.; Mahasneh, M. Fly ash based geopolymer for heavy metal removal: A case study on copper removal. J. Environ. Chem. Eng. 2015, 3, 1669–1677. [Google Scholar] [CrossRef]
- Sebastiano, C.; De Luca, P.; Frontera, P.; Crea, F. Production of Geopolymeric Mortars Containing Forest Biomass Ash as Partial Replacement of Metakaolin. Environments 2017, 4, 7. [Google Scholar] [CrossRef]
- Davidovits, J. Geopolymer Chemistry & Application; Institute Géopolymère: Saint-Quentin, France, 2015; ISBN 9782951482098. [Google Scholar]
- Huang, C.P.; Blankenship, D.W. The removal of mercury(II) from dilute aqueous solution by activated carbon. Water Res. 1984, 18, 37–46. [Google Scholar] [CrossRef]
- Logar, N.Z.; Kaucic, V. Nanoporous materials: From catalysis and hydrogen storage to wastewater treatment. Acta Chim. Slov. 2006, 53, 117–135. [Google Scholar]
- Policicchio, A.; Vuono, D.; Rugiero, T.; De Luca, P.; Nagy, J.B. Study of MWCNTs adsorption perfomances in gas processes. J. CO2 Util. 2015, 10, 30–39. [Google Scholar] [CrossRef]
- Ghori, Z.; Iftikhar, H.; Bhatti, M.F.; um-Minullah, N.; Sharma, I.; Kazi, A.G.; Ahmad, P. Phytoextraction: The use of plants to remove heavy metals from soil. In Plant Metal Interaction: Emerging Remediation Techniques; Elsevier: Amsterdam, The Netherlands, 2016; pp. 385–409. [Google Scholar]
- Kang, S.Y.; Lee, J.U.; Moon, S.H.; Kim, K.W. Competitive adsorption characteristics of Co2+, Ni2+ and Cr3+ by IRN-77 cation exchange resin in synthesized wastewater. Chemosphere 2004, 56, 141–147. [Google Scholar] [CrossRef] [PubMed]
- Motsi, T.; Rowson, N.A.; Simmons, M.J.H. Adsorption of heavy metals from acid mine drainage by natural zeolite. Int. J. Miner. Process. 2009, 92, 42–48. [Google Scholar] [CrossRef]
- Ostroski, I.C.; Barros, M.A.S.D.; Silvab, E.A.; Dantas, J.H.; Arroyo, P.A.; Lima, O.C.M. A comparative study for the ion exchange of Fe(III) and Zn(II) on zeolite NaY. J. Hazard. Mater. 2009, 161, 1404–1421. [Google Scholar] [CrossRef] [PubMed]
- Taffarel, S.R.; Rubio, J. On the removal of Mn2+ ions by adsorption onto natural and activated Chilean zeolites. Miner. Eng. 2009, 22, 336–343. [Google Scholar] [CrossRef]
- Barakat, M.A. New trends in removing heavy metals from industrial wastewater. Arab. J. Chem. 2011, 4, 361–377. [Google Scholar] [CrossRef]
- Fenglian, F.; Qui, W. Removal of heavy metal ions from wastewaters: A review. J. Environ. Manag. 2011, 92, 407–418. [Google Scholar]
- De Raffele, G.; Aloise, A.; De Luca, P.; Vuono, D.; Tagarelli, A.; Nagy, J.B. Kinetic and thermodynamic effects during the adsorption of heavy metals on ETS-4 and ETS-10 microporous materials. J. Porous Mater. 2016, 23, 389–400. [Google Scholar] [CrossRef]
- Anderson, M.W.; Terasaki, O.; Ohsuna, T.; Philippou, A.; MacKay, S.P.; Ferreira, A.; Rocha, J.; Lidin, S. Structure of the microporous titanosilicate ETS-10. Nature 1994, 367, 347–351. [Google Scholar] [CrossRef]
- Kuznicki, S.M. Large-Pored Crystalline Titanium Molecular Sieve Zeolites. US Patent 4853202A, 8 September 1987. [Google Scholar]
- Kuznicki, S.M. Preparation of Small-Pored Crystalline Titanium Molecular Sieve Zeolites 1990. US Patent 4938939A, 3 July 1990. [Google Scholar]
- Anderson, M.W.; Terasaki, O.; Ohsuna, T.; Malley, P.J.O.; Philippou, A.; MacKay, S.P.; Ferreira, A.; Rocha, J.; Lidin, S. Microporous Titanosilicate ETS-10: A Structural Survey. Philos. Mag. B 1995, 71, 813–841. [Google Scholar] [CrossRef]
- Rocha, J.; Anderson, M.W. Microporous Titanosilicates and Other Novel Mixed Octahedral-Tetrahedral Framework Oxides. Eur. J. Inorg. Chem. 2000, 2000, 801–818. [Google Scholar] [CrossRef]
- De Luca, P.; Nastro, A. Synthesis of ETS-10 molecular sieve from systems containing TAABr salts. Stud. Surf. Sci. Catal. 1997, 105, 221–228. [Google Scholar]
- Wang, X.; Jacobson, A.J. Crystal structure of the microporous titanosilicate ETS-10 refined from single crystal X-ray diffraction data. Chem. Commun. 1999, 973–974. [Google Scholar] [CrossRef]
- Turta, N.A.; De Luca, P.; Bilba, N.; Nagy, J.B. Synthesis of titanosilicate ETS-10 in presence of cetyltrimethylammonium bromide. Microporous Mesoporous Mater. 2008, 112, 425–431. [Google Scholar] [CrossRef]
- Turta, N.A.; Veltri, M.; Vuono, D.; De Luca, P.; Bilba, N.; Nastro, A. Effect of crystallization temperature on the synthesis of ETS-4 and ETS-10 titanosilicates. J. Porous Mater. 2009, 16, 527–536. [Google Scholar] [CrossRef]
- Pavel, C.C.; De Luca, P.; Bilba, N.; Nagy, J.B.; Nastro, A. On the crystallization mechanism of the ETS-10 titanosilicate synthesized in gels containing TAABr. Thermochim. Acta 2005, 435, 213–221. [Google Scholar] [CrossRef]
- Yilmaz, B.; Shattuck, K.G.; Miraglia, P.Q.; Warzywoda, J.; Sacco, A. Synthesis of titanosilicate ETS-4 utilizing organic precursors. Stud. Surf. Sci. Catal. 2004, 154, 763–769. [Google Scholar]
- Yilmaz, B.; Miraglia, P.Q.; Warzywoda, J.; Sacco, A. Synthesis of titanosilicate ETS-4 with controlled morphology and investigation of its crystallization kinetics. Microporous Mesoporous Mater. 2004, 71, 167–175. [Google Scholar] [CrossRef]
- Yang, X.; Paillaud, J.L.; van Breukelen, H.F.W.J.; Kessler, H.; Duprey, E. Synthesis of microporous titanosilicate ETS-10 with TiF4 or TiO2. Microporous Mesoporous Mater. 2001, 46, 1–11. [Google Scholar] [CrossRef]
- Ismail, I.H.; El-Maksod, I.H.A.; Ezzat, H. Synthesis and characterization of titanosilicates from white sand silica and its hydrogen uptake. J. Hydrogen Energy Int. 2010, 35, 10359–10365. [Google Scholar] [CrossRef]
- Pavel, C.C.; Vuono, D.; De Luca, P.; Bilba, N.; Nagy, J.B.; Nastro, A. Synthesis and characterization of ET(P)S-4 and ET(P)S-10. Microporous Mesoporous Mater. 2005, 80, 263–268. [Google Scholar] [CrossRef]
- Catanzaro, L.; De Luca, P.; Nagy, J.B.; Nastro, A. Microporous titanosilicate synthesized with vanadium. Stud. Surf. Sci. Catal. 2004, 154, 746–752. [Google Scholar]
- Craveiro, R.; Lin, Z. The influence of Fe on the formation of titanosilicate ETS-4. J. Solid State Chem. 2012, 190, 162–168. [Google Scholar] [CrossRef]
- Ferdov, S.; Lin, Z.; Ferreira, R.A.S.; Correia, M.R. Hydrothermal synthesis, structural, and spectroscopic studies of vanadium substituted ETS-4. Microporous Mesoporous Mater. 2008, 110, 436–441. [Google Scholar] [CrossRef]
- Sudheesh, N.; Shukla, R.S. Rhodium exchanged ETS-10 and ETS-4: Efficient heterogeneous catalyst for hydroaminomethylation. Appl. Catal. A Gen. 2014, 473, 116–124. [Google Scholar] [CrossRef]
- Choi, J.H.; Kim, S.D.; Kwon, Y.J.; Kim, W.J. Adsorption behaviors of ETS-10 and its variant, ETAS-10 on the removal of heavy metals, Cu2+, Co2+, Mn2+ and Zn2+from a waste water. Microporous Mesoporous Mater. 2006, 96, 157–167. [Google Scholar] [CrossRef]
- Zhao, G.X.S.; Lee, J.L.; Chia, P.A. Unusual adsorption properties of microporous titanosilicate ETS-10 toward heavy metal lead. Langmuir 2003, 19, 1977–1979. [Google Scholar] [CrossRef]
- Lopes, C.B.; Coimbra, J.; Otero, M.; Pereira, E.; Duarte, A.C.; Lin, Z.; Rocha, J. Uptake of Hg2+ from aqueous solutions by microporous titano- and zircono-silicates. Quimica Gradiškan. 2008, 31, 321–325. [Google Scholar] [CrossRef]
- Lv, L.; Su, F.; Zhao, X.S. Microporous titanosilicate ETS-10 for the removal of divalentheavy metals. Stud. Surf. Sci. Catal. 2005, 156, 933–940. [Google Scholar]
- Al-Attar, L.; Dyer, A.; Blackburn, R. Uptake of uranium on ETS-10 microporous titanosilicate. J. Radioanal. Nucl. Chem. 2000, 246, 451–455. [Google Scholar] [CrossRef]
- Pavel, C.C.; Popa, K.; Bilba, N.; Cecal, A.; Cozman, D.; Pui, A. The sorption of some radiocations on microporous titaniosilicate ETS-10. J. Radioanal. Nucl. Chem. 2003, 258, 243–248. [Google Scholar] [CrossRef]
- De Luca, P.; Poulsen, T.G.; Salituro, A.; Tedeschi, A.; Vuono, D.; Kònya, Z.; Madaràsz, D.; Nagy, J.B. Evaluation and comparison of the ammonia adsorption capacity of titanosilicates ETS-4 and ETS-10 and aluminotitanosilicates ETAS-4 and ETAS-10. J. Therm. Anal. Calorim. 2015, 122, 1257–1267. [Google Scholar] [CrossRef]
- Bagnasco, G.; Turco, M.; Busca, G.; Armaroli, T.; Nastro, A.; De Luca, P. Characterization of the structural and gas adsorption properties of ETS-10 molecular sieve. Adsorpt. Sci. Technol. 2003, 21, 683–696. [Google Scholar] [CrossRef]
- Areàn, C.O.; Palomino, G.T.; Zecchina, A.; Bordiga, S.; Xamena, F.X.L.L.; Pazé, C. Vibrational spectroscopy of carbon monoxide and dinitrogen adsorbed on magnesium-exchanged ETS-10 molecular sieve. Catal. Lett. 2000, 66, 231–235. [Google Scholar] [CrossRef]
- De Luca, P.; Chiodo, A.; Nagy, J.B. Activated ceramic materials with deposition of photocatalytic titano-silicate micro-crystals. WIT Trans. Ecol. Environ. 2011, 154, 155–165. [Google Scholar]
- Guan, G.; Kida, T.; Kusakabe, K.; Kimura, K.; Abe, E.; Yoshida, A. Photocatalytic activity of CdS nanoparticles incorporated in titanium silicate molecular sieves of ETS-4 and ETS-10. Appl. Catal. A Gen. 2005, 295, 71–78. [Google Scholar] [CrossRef]
- Crea, F.; Aiello, R.; Nastro, A.; Nagy, J.B. Synthesis of ZSM-5 zeolite from very dense systems: Formation of pelleted ZSM-5 zeolite from (Na, Li, TPA, Si, Al) hydrogels. Zeolites 1991, 11, 521–527. [Google Scholar] [CrossRef]
- Frontera, P.; Macario, A.; Katovic, A.; Crea, F.; Giordano, G. Direct synthesis of zeolites self-bonded pellets for biocatalyst immobilization. Stud. Surf. Sci. Catal. 2005, 158, 383–390. [Google Scholar]
- Macario, A.; Giordano, G.; Frontera, P.; Crea, F.; Setti, L. Hydrolysis of alkyl ester on lipase/silicalite-1 catalyst. Catal. Lett. 2008, 122, 43–52. [Google Scholar] [CrossRef]
- Aiello, R.; Nastro, A.; Crea, F.; Colella, C. Use of natural products for zeolite synthesis. Self-bonded zeolite pellets from rhyolitic pumice. Zeolites 1985, 2, 290–294. [Google Scholar] [CrossRef]
- Gilson, J.P. Method of Preparing Silicate Composition. US Patent 4537866, 29 March 1984. [Google Scholar]
- De Luca, P.; Crea, F.; Fonseca, A.; Nagy, J.B. Direct formation of self-bonded pellets during the synthesis of mordenite and ZSM-11 zeolites from low water content systems. Microporous Mesoporous Mater. 2001, 42, 37–48. [Google Scholar] [CrossRef]
- De Luca, P.; Mastroianni, C.; Nagy, J.B. Synthesis of self-bonded pellets of ETS-4 phase by new methodology of preparation. IOP Conf. Ser. Mater. Sci. Eng. 2018, 347, 012003. [Google Scholar] [CrossRef]
- Vuono, D.; De Luca, P.; Nagy, J.B.; Nastro, A. Synthesis and characterization of self-bonded ETS-4 and ETS-10 pellets. Microporous Mesoporous Mater. 2008, 109, 118–137. [Google Scholar] [CrossRef]
- Vuono, D.; Guzzo, M.; De Luca, P.; Nagy, J.B. Physico-chemical characterization of zirconium-based self-bonded ETS-4 pellets. J. Therm. Anal. Calorim. 2014, 116, 169–182. [Google Scholar] [CrossRef]
- De Luca, P.; Vuono, D.; Filice, M. Self-bonded ETS-10 pellets containing iron. Environ. Eng. Manag. J. 2009, 8, 1009–1015. [Google Scholar] [CrossRef]
- De Luca, P.; Nappo, G.; Siciliano, C.; Nagy, J.B. The role of carbon nanotubes and cobalt in the synthesis of pellets of titanium silicates. J. Porous Mater. 2018, 25, 283–296. [Google Scholar] [CrossRef]
- Youcai, Z.; Stanforth, R. Extraction of zinc from zinc ferrites by fusion with caustic soda. Miner. Eng. 2000, 13, 1417–1421. [Google Scholar] [CrossRef]
- Candamano, S.; Frontera, P.; Macario, A.; Crea, F. Effect of commercial LTA type zeolite inclusion in properties of structural epoxy adhesive. Adv. Sci. Lett. 2017, 6, 5927–5930. [Google Scholar] [CrossRef]
- Jo, D.; Park, G.T.; Ryu, T.; Hong, S.B. Economical synthesis of high-silica LTA zeolites: A step forward in developing a new commercial NH3-SCR catalyst. Appl. Catal. B Environ. 2019, 243, 212–219. [Google Scholar] [CrossRef]
- Frontera, P.; Crea, F.; Testa, F.; Aiello, R. Zeolite LTA deposition on silicon wafer. J. Porous Mater. 2007, 14, 325–329. [Google Scholar] [CrossRef]
- Xue, Z.; Ma, J.; Hao, W.; Bai, X.; Kang, Y.; Liu, J.; Li, R. Synthesis and characterization of ordered mesoporous zeolite LTA with high ion exchange ability. J. Mater. Chem. 2012, 22, 2532–2538. [Google Scholar] [CrossRef]
- Hashemiam, S.; Hossein, S.H.; Salehifar, H.; Salari, K. Adosorption of Fe(III) from acqueous solution by Linde Type-A zeolite. Am. J. Anal. Chem. 2013, 4, 123–126. [Google Scholar] [CrossRef]
- Hui, K.S.; Chao, C.Y.H.; Kot, S.C. Removal of mixed heavy ions in wastewater by zeolite 4A and residual products from recycled coal fly ash. J. Hazard. Mater. 2005, 127, 89–101. [Google Scholar] [CrossRef] [PubMed]
- Istituto di Ricerca Sulle Acque. Metodi Analitici per I Fanghi—Parametri Chimic-Fisici; Quaderni/Istituto di Ricerca Sulle Acque; CNR: Roma, Italy, 1985; ISSN 0390-6329. [Google Scholar]
- Ribeiro, F.R. (Ed.) Zeolites: Science and Technology; Martinus Nijhoff Publisher: The Hague, The Netherlands, 1984. [Google Scholar]
- Nightingale, E.R. Phenomenological theory of ion solvation. Effective radii of hydrated ions. J. Phys. Chem. 1959, 63, 1381–1387. [Google Scholar] [CrossRef]
- Persson, I. Hydrated metal ions in aqueous solution: How regular are their structures? Pure Appl. Chem. 2010, 82, 1901–1917. [Google Scholar] [CrossRef]

| Systems | Time (h) | System Composition |
|---|---|---|
| 1A | 0.5 | 5 g Zinc ferrite + 100 mL H2O |
| 2A | 1.0 | |
| 3A | 1.5 | |
| 4A | 2.0 | |
| 1B | 0.5 | 5 g Zinc ferrite + 10 g ETS-10 + 100 mL H2O |
| 2B | 1.0 | |
| 3B | 1.5 | |
| 4B | 2.0 | |
| 1C | 0.5 | 5 g Zinc ferrite + 10 g zeolite A + 100 mL H2O |
| 2C | 1.0 | |
| 3C | 1.5 | |
| 4C | 2.0 |
| Element | Concentration (mg/L) | % Weight |
|---|---|---|
| Fe | 1168.000 | 23.360 |
| Zn | 768.000 | 15.360 |
| Pb | 265.000 | 5.300 |
| Ca | 103.000 | 2.060 |
| Mn | 59.800 | 1.196 |
| Al | 31.300 | 0.626 |
| Mg | 19.000 | 0.380 |
| Cu | 15.300 | 0.306 |
| Na | 14.800 | 0.296 |
| Sr | 11.100 | 0.222 |
| K | 8.000 | 0.160 |
| Cd | 3.070 | 0.061 |
| Ba | 1.950 | 0.039 |
| In | 1.130 | 0.026 |
| Ni | 0.559 | 0.011 |
| Cr | 0.549 | 0.011 |
| Ga | 0.450 | 0.009 |
| Co | 0.302 | 0.006 |
| Li | 0.020 | 4 × 10−4 |
| Rb | 0.018 | 3.6 × 10−4 |
| Cs | 0.009 | 1.8 × 10−4 |
| Peak | 2theta (degree) | Intensity (cps) | d-Spacing |
|---|---|---|---|
| 1 | 29.8 | 227.1429 | 2.5546 |
| 2 | 35.1 | 440.1429 | 2.9957 |
| 3 | 61.98 | 190.3463 | 1.4961 |
| Concentration (µg/L) | ||||
|---|---|---|---|---|
| Time | 0.5 h | 1.0 h | 1.5 h | 2.0 h |
| Zn | 1006.045 | 1081.831 | 1035.219 | 1012.561 |
| Ca | 365.153 | 387.416 | 384.632 | 363.531 |
| Mn | 188.840 | 198.733 | 192.026 | 189.617 |
| Na | 98.691 | 101.962 | 96.040 | 95.917 |
| Mg | 81.840 | 86.480 | 81.676 | 80.522 |
| K | 18.829 | 30.309 | 18.367 | 16.780 |
| Fe | 3.683 | 3.387 | 4.505 | 4.158 |
| Pb | 1.124 | 1.681 | 1.759 | 1.459 |
| Element | Wt % Released |
|---|---|
| Na | 6.48 × 10–2 |
| Mg | 4.24 × 10–2 |
| Ca | 3.26 × 10–2 |
| Mn | 3.17 × 10–2 |
| K | 2.09 × 10–2 |
| Zn | 1.32 × 10–2 |
| Pb | 5.47 × 10–5 |
| Fe | 3.55 × 10–5 |
| Concentration (µg/L) | ||||
|---|---|---|---|---|
| Time | 0.5 h | 1.0 h | 1.5 h | 2.0 h |
| Na | 1878.497 | 1993.856 | 2199.830 | 2247.248 |
| K | 354.221 | 328.855 | 370.959 | 418.133 |
| Ca | 253.976 | 243.594 | 252.010 | 161.564 |
| Mg | 59.750 | 58.485 | 62.748 | 52.624 |
| Zn | 6.389 | 0.753 | 0.000 | 0.000 |
| Mn | 5.250 | 3.490 | 3.399 | 2.678 |
| Fe | 3.308 | 3.182 | 1.706 | 1.211 |
| Pb | 0.000 | 0.000 | 0.000 | 0.000 |
| Element | Efficiency | |
|---|---|---|
| ETS-10 | Zeolite A | |
| Pb | 100.00% | 69.10% |
| Zn | 97.71% | 84.95% |
| Mn | 97.65% | 54.38% |
| Fe | 10.45% | 10.70% |
| Ca | 30.22% | 32.41% |
| Mg | 26.98% | 29.61% |
| Element | Efficiency | |
|---|---|---|
| ETS-10 | Hydrated Radii (Å) [73] | |
| Pb2+ | 100.00% | 4.01 |
| Zn2+ | 97.71% | 4.30 |
| Mn2+ | 97.65% | 4.38 |
| Fe2+/Fe3+ | 10.45% | 4.28/4.57 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
De Luca, P.; Bernaudo, I.; Elliani, R.; Tagarelli, A.; Nagy, J.B.; Macario, A. Industrial Waste Treatment by ETS-10 Ion Exchanger Material. Materials 2018, 11, 2316. https://doi.org/10.3390/ma11112316
De Luca P, Bernaudo I, Elliani R, Tagarelli A, Nagy JB, Macario A. Industrial Waste Treatment by ETS-10 Ion Exchanger Material. Materials. 2018; 11(11):2316. https://doi.org/10.3390/ma11112316
Chicago/Turabian StyleDe Luca, Pierantonio, Ivano Bernaudo, Rosangela Elliani, Antonio Tagarelli, Jànos B. Nagy, and Anastasia Macario. 2018. "Industrial Waste Treatment by ETS-10 Ion Exchanger Material" Materials 11, no. 11: 2316. https://doi.org/10.3390/ma11112316





