Abstract
Houttuynia cordata Thunb (H. cordata) is a rhizomatous, herbaceous, and perennial plant widely distributed in Asia. It has multiple chemical constituents, such as alkaloids, essential oils, phenolic acids, and flavonoids used against various health problems. The essential oils and flavonoids are the main components of H. cordata that play an essential role in disease treatment and traditional health care. Moreover, the leaves and stems of H. cordata have a long medicinal history in China. In addition, H. cordata is used against several health issues, such as cold, cough, fever, pneumonia, mumps, and tumors, due to its anti-inflammatory, anti-bacterial, anti-viral, anti-oxidant, and anti-tumor effects. It protects organs due to its anti-inflammatory activity. H. cordata regulates immunity by enhancing immune barriers of the oral cavity, vagina, and gastrointestinal tract, and shows broad-spectrum activity against liver, lung, breast, and colon tumors. However, there are some gaps to be filled to understand its pathways and mechanisms. Mechanisms such as its interaction with cells, cell membranes, and various drugs are important. Studies in relation to the blood–brain barrier, lipophilicity, cAMP signaling, and skin permeability, including pharmaceutical effects, will be very useful. This review includes the biological and pharmacological activities of H. cordata based on up-to-date research.
1. Introduction
Eastern countries have a long history of using herbal medicine; in particular, Chinese people have used herbal medicine to treat various diseases for more than 8000 years [1]. The chemical components and biological activities present in the medicinal plant can be used for the prevention and treatment of various diseases [2]. Natural products with a variety of pharmacological targets in numerous diseases have emerged as crucial sources for the development of novel drugs [3]. The most popular anti-malarial drug, artemisinin, which is obtained from plants, is essential for the treatment of malaria [4]. The World Health Organization summarized data in 1985 that showed that, of the world’s total population, eighty percent of people depend on traditional medicine as a treatment, including herbal medicine [5].
Houttuynia Cordata Thunb (H. cordata) is a rhizomatous, herbaceous, and perennial plant found in China, Japan, Korea, and Southwest Asia. It usually grows in shady and moist places. This plant is widely distributed in China, and is present in Tibet, Gansu, Shaanxi, Yunnan, and eastern Taiwan [6]. As its aboveground stems and leaves have a long medicinal history in China and are employed in the treatment of pneumonia and lung abscesses, it plays a significant role in traditional health care and disease treatment [7].
In the past, people used H. cordata as medicine. In India, it is harvested as a medicine and daily food [8]. A significant amount of scientific evidence shows that H. cordata, as a whole plant or in the form of its extracts, has important medicinal effects [9]. Recently, several studies showed and unveiled its anti-allergic [10,11], anti-inflammatory [12,13], anti-viral [14], anti-oxidative [15], anti-leukemic [16], anti-cancer [17], and anti-SARS [18] activities. During the outbreak of severe acute respiratory syndrome (SARS), H. cordata was used for the prevention of SARS by the Health Ministry of China. It also helps to decrease swelling, lower fever, and drain pus, and in urination. Moreover, H. cordata also shows efficacy against methicillin-resistant Staphylococcus aureus (MRSA) [19] and multi-drug resistant Escherichia coli [20].
It is used to treat dysentery, fever, cold, and mumps in clinical medicine. It is also used to protect organs by reducing the release of inflammatory mediators. It boosts immunity by enhancing the immune barriers of the vagina, intestines, and other organs [7]. In this review, we briefly discuss the chemical properties of H. cordata and various effects on different organs of the body.
2. Chemical Components of H. cordata
H. cordata has a variety of chemical constituents with characteristic medicinal properties and belonging to different chemical groups, such as alkaloids, essential oils, and flavonoids as shown in Table 1 [21]. The alkaloids consist of aristolactam A, 3,4-dimethoxy-N-methyl aristolactam, lysicamine, noraritolodione, norcepharadione B, 3,5-didecanoyl-pyridine, 7-chloro-6-demethyl-cepharadione B, cis-N-(4-Hydroxystyryl) benzamide and trans-N-(4-Hydroxystyryl) benzamide, 2-nonyl-5-decanoylpyridine, 3,5-Didecanoyl-4-nonyl-1,4-dihydropyridine, cepharadione B, splendidine, piperolactam A, 3-decanoyl-4-nonyl-5-dodecanoyl-1,4-dihydropyridine, aristolactam B, 3,5-didodecanoyl-4-nonyl-1,4-dihydropyridine, 7-chloro-6-demethylcepharadione B, 3-nonylpyrazole, N-methyl-5-methoxy-pyrrolidin-2-one, phenanthrolactam compounds [22,23]. The flavonoid compounds included quercetin, rutin, hyperin, afzelin, quercitrin, isoquercitrin, kaempferol, quercetin hexoside, avicularin, apigenin, isorhamnetin, phloridzin, quercetin-3-O-β-D-galactoside-7-O-β-D-glucoside, and polyphenols include chlorogenic acid, vanillic acid, protocatechuic acid, catechin, p-hydroxy-benzoic acid methyl ester, chlorogenic acid methyl ester, cryptochlorogenic acid, neochlorogenic acid, procyanidin B, quinic acid, caffeic acid, cis-methyl ferulate, trans-methyl ferulate, methyl vanillate, vanillin, houttuynamide A, and houttuynoside A [24,25,26]. The main components of the essential oil are houttuynin, decanal, trans-caryophyllene, decanoic acid, camphene, β-pinene, lauraldehyde, α-pinene, limonene, nonanol and linalool bornyl acetate, methyl n-nonyl ketones, beta myrcene, monoterpene, 4-terpineol, caryophyllene oxide, phenylpropene derivatives, sesquiterpenes, and oxidized diterpenes [27].
H. cordata has many components, and alkaloids are abundant ingredients [28]. Essential oil and flavonoids are known to be major components that exert pharmacological activities. Moreover, decanoyl acetaldehyde in H. cordata has a fishy smell called Yu-Xing-Cao, and is a herb in traditional Chinese medicine [22]. It has anti-bacterial effects and is easily transformed to 2-undecanone at higher temperatures [29]. Steam distillation extracts of H. cordata contain some important oils, which consist of oxidized diterpenes, monoterpenes, sesquiterpenes, and oxidized diterpenes [30]. Others present in H. cordata include bornyl acetate (0.4–8.61%), ketones (2.10–40.36%), and β-myrcene (2.58–18.47%) [27]. Eleven ingredients have been isolated from leaves of H. cordata, and seven have been isolated from the roots and are not present in the leaves [7]. It is also reported that H. cordata from various areas has various anti-bacterial effects [31]. Flavonoids in H. cordata, such as quercetin, quercitrin, and hyperoside, are mostly combined with rhamnose in glycosides [26]. A new form of hyperoside and houttuynia has been isolated from flavonoid compounds in H. cordata [32]. Other new components are houttuynamide A and houttuynoside A [25]. Caffeic acid derivatives, quinic acid derivatives, chlorogenic acid, neochlorogenic acid, and cryptochlorogenic acid are considered the essential components of H. cordata [24]. Alkaloids such as phenanthrolactam, piperolactam, and aristololactam are key components of H. cordata and play an essential role in pharmacological effects [23].

Table 1.
Therapeutic effect of different extracts of H. cordata.
Table 1.
Therapeutic effect of different extracts of H. cordata.
| Class | Compound | Chemical Structure | Therapeutic Properties | References |
|---|---|---|---|---|
| Essential Oil (Oil) | Terpenoids | 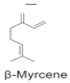 | Anti-bacterial activities, anti-viral activities, and anti-inflammatory activities. | [12,33,34] |
| Hydrocarbons | 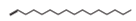 1-Hexadecene | Anti-bacterial activities, anti-viral activities, and anti-inflammatory activities. | [12,30,33] | |
| Esters | 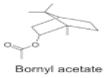 | Anti-bacterial activities and anti--inflammatory activities. | [12,33] | |
| Alcohols | 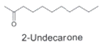 | Anti-viral activities and anti-inflammatory activities. | [12,30,33] | |
| Flavonoids | Quercetin |  | Inflammatory mediator, anti-viral | [9,35] |
| Quercitrin |  | Anti-inflamatory | [36] | |
| Hyperin | 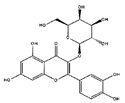 | Anti-viral, anti-inflamatory | [9,37] | |
| Rutin | 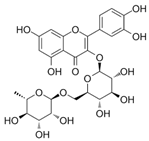 | Inhibition of cholestasis | [9,38] | |
| Afzelin | 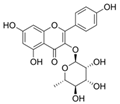 | Lower inflamatory cytokines | [28,39] | |
| Kaempferol | 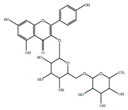 | Anti-inflamatory | [9] | |
| Isoquercitrin | 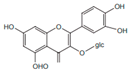 | Anti-inflamatory | [9] | |
| Alkaloids (Alkaloids) | Aristolactam A | 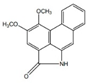 | Anti-tumor activities | [9,17] |
| Aristolactam B | 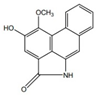 | Anti-inflamatory | [9,23] | |
| Piperolactam A | 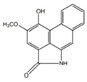 | Anti-bacterial, anti-pyretic, detoxicant, anti-ulcer | [9,17] | |
| Lysicamine | 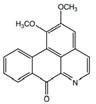 | Anti-parasitic, anti-bacterial | [9,40] | |
| Norcepharadione B | 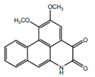 | Anti-inflamatory, anti-pyretic | [9,17] | |
| 3,4-Dimethoxy-N-methyl aristolactam | 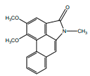 | Anti-inflamatory | [9] | |
| cis-N-(4-Hydroxystyryl) benzamide | 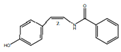 | Inhibitor of platelet aggregation | [9,41]. | |
| 3,5-Didecanoyl-4-nonyl-1, 4-dihydropyridine | 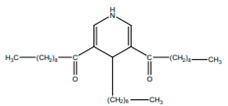 | Anti-inflamatory | [9,21] | |
| 7-chloro-6- demethylcepharadione B | 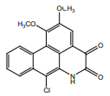 | Anti-oxidant | [9] | |
| trans-N-(4-Hydroxystyryl) benzamide | 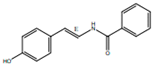 | Inhibitor of platelet aggregation | [41] | |
| Cepharadione B | 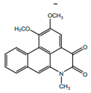 | Anti-oxidant | [9] | |
| Splendidine | 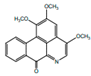 | Inhibitor of platelet aggregation | [9,42] | |
| Organic Acid | 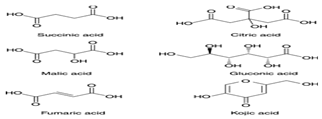 | Anti-bacterial, anti-fungal activities | [43] | |
| Polyphenols | Houttuynamide A | 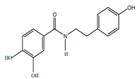 | Anti-inflamatory | [9] |
| Houttuynaside A | 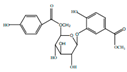 | Anti-inflamatory | [9] | |
| Chlorogenic acid | 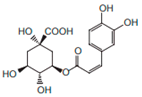 | Anti-hypertensive | [9] | |
| Vanillin | 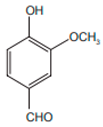 | Anti-oxidant | [9] | |
3. Anti-Inflammatory Effects and Immunomodulatory Activity of H. cordata
Inflammation is a protective response of the body against offending agents such as viruses, bacteria, toxic chemicals, and damaged cells. There are two forms of inflammation. One is acute inflammation and the other is chronic inflammation [44]. Many cells are involved in inflammation, including neutrophils, eosinophils, basophils, mast cells, fibroblasts, and mononuclear cells [45]. Different extracts of H. cordata, such as water extract, n-butanol extract, and ethanol extract, have shown anti-inflammatory results in mice as shown in Table 2. All extracts have good anti-inflammatory activity. It has been found that the anti-inflammatory effects of water extract are better than those of ethnolic extract. Fresh H. cordata extracts showed better pharmacological activity than dry H. cordata extract [9].
Cells involved in body immunity are basophils, eosinophils, mast cells, lymphocytes, and neutrophils. They play vital immune functions. Antibodies, tumor necrosis factors, interferons, and interleukin also play an essential role in body immunity. Abnormal immune functions result in microcirculation, anaphylactic shock, and central nervous system disorders [46]. The polyphenols present in H. cordata show anti-allergic effects. Due to H. cordata extracts, decreased activity of iIgE and FcεRI expression on basophilic cells was observed. Moreover, mRNA activity associated with γ-chains and FcεRI was decreased, and histamine secretions were inhibited [47]. It was observed that H. cordata extract decreased cutaneous anaphylaxis in vivo in mice. The level of cAMP present in mast cells is enhanced by using H. cordata, which shows that H. cordata can speed up the recovery from allergic reactions. HCP-2 polysaccharides extracted from H. cordata regulate the expression of T cells with a dosage of 0.1–25 μg/mL. It increases tumor necrosis factor-α (TNF-α), immune molecule interleukin-1β, and macrophage inhibitory protein-1α and -1β, which increases body immunity. It has been recorded that H. cordata reduces Th2-mediated immune disorders. Ethanol extract of H. cordata decreases the migration of T cells, which ultimately strengthens immune response [10]. H. cordata extract helps in the regulation of immune mediators. After 18 h of treating vaginal epithelial cells with H. cordata, levels of leukocyte protease inhibitor mRNA and human β-defensin 2 were increased. Moreover, an increase in IL-2 and IL-6 and a decrease in CCL5 were observed. These findings show an increase in the overall immune response. H. cordata has the same effects on oral immune mediators by expressing human β-defensin 2, IL-8, CCL20, and secretory leukocyte protease inhibitor. In this manner, H. cordata regulates oral immune response [48].
3.1. In Vitro Anti-Inflamatory Effect of H. cordata
The water extract was reported to reduce nitric oxide and TNF-α production (by up to 30%) in LPS-induced mice macrophages at a dose rate of 0.06 and 0.12 mg/mL [13]. Additionally, it was discovered that the ethnolic extract of H. cordata inhibited the inflammatory biomarkers interleukin-6 and nitric oxide in lung epithelial cells (A549) and alveolar macrophages challenged with LPS (MH-S) [49]. In LPS-treated alveolar macrophages (MH-S) and lung epithelial cells, the ethanolic extract of H. cordata was observed to suppress inflammatory mediators such as interleukin-6 and nitric oxide [35]. In RAW 264.7 cells, essential oils extracted from HC considerably decreased the LPS-induced inflammation. The HC was withdrawn through supercritical CO2 with the concentration of one percent solution, and was found to decrease the PGE2 and nitric oxide levels by 80.4 percent and 98 percent, respectively, in RAW cells treated with LPS [50].
In LPS-primed RAW 264.7 cells, the active biomolecules sodium houttuyfonate and 2-undecanone that were extracted from H. cordata essential oil were demonstrated to have a significant anti-inflammatory impact. At the same concentration, sodium houttuyfonate showed a significant decrease in TNF-α (p < 0.001) and IL-1β level compared to 2-undecanone [29]. One of the crucial molecular pathways thought to be involved in the inflammatory pathophysiology associated with allergy is the mast cell-mediated anaphylactic shock. The primary causes of this condition are alterations in intercellular calcium ions (Ca2+) and histamine produced from degranulated mast cells that cause such severe anaphylactic responses. Research revealed the HCWE’s anti-allergic effects on both systemic rat peritoneal mast cells and acute anaphylactic responses (RPMCs). Histamine release and Ca2+ uptake have been demonstrated to be inhibited by the HCWE in a dose-dependent manner at concentrations of 0, 2.5, 25, and 250 µg [10]. The increase in adenylate cyclase activity and the subsequent rise in the intracellular cAMP level in mast cells was suggested to be the probable mechanism causing the suppression of histamine release and decreased Ca2+ absorption. Additionally, it was shown that IgE-mediated allergic reactions play a significant part in the pathophysiology of inflammation. The Fc epsilon RI receptor, a high-affinity IgR receptor expressed in human basophilic cells, has the ability to trigger allergic responses. Shim et al. demonstrated that the HC water extract decreased both the mRNA expression of both the chains of the Fc epsilon RI receptor in human KU812F cells and the IgE binding activity. Additionally, it was discovered that the HC extract inhibited KU812F cells’ Fc epsilon RI-mediated production of histamine [19].
A study was conducted by isolating polysaccrides from H. cordata and its composition was found to consist of galacturonic acid, glucose, galactose, rhamnose, and arabinose. It was found that H. cordata polysaccharide (HBHP-3) is an active ingredient that inhibits the expression of mRNA, and is involved in inflammatory activity and the secretion of nitric oxide, hence lowering inflammation in a dose-dependent manner [51]. Methanolic and aqueous extract of H. cordata were displayed anti-inflammatory properties. The protein denaturation method was used to assess the plant’s anti-inflammatory potential. Different levels of Houttuynia cordata free radical scavenging and anti-inflammatory activity are present in methanolic and aqueous extracts. The Houttuynia cordata methanolic extract was the most active of the extracts tested in the study compared to the aqueous extract [52].
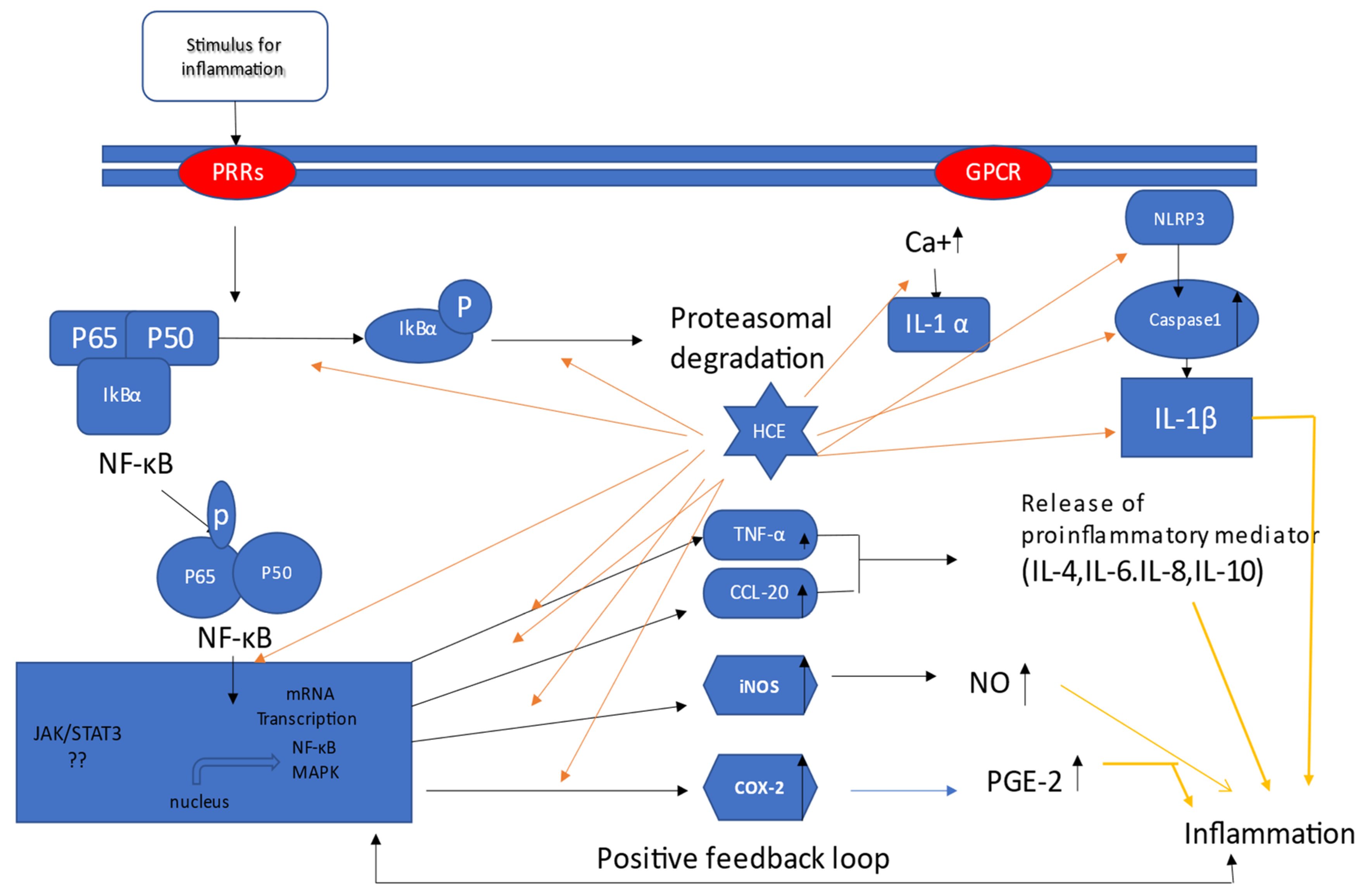
These studies have provided ample evidence that H. cordata has anti-inflammatory capabilities against a variety of inflammagens in various cell lines. Based on the research, Figure 1 depicts a schematic mechanism via which H. cordata functions in several in vitro models.
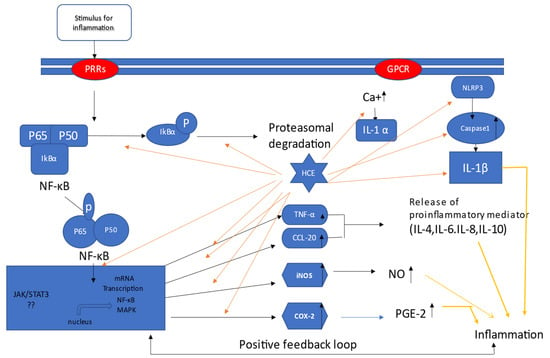
Figure 1.
Mechanism of H. cordata acts as an anti-inflammatory agent in vitro; H. cordata suppresses inflammation through various pathways. It blocks all mentioned signaling pathways, resulting in a decrease in the production of inflammatory mediators such as prostaglandins and interleukins. Moreover, H. cordata suppresses inflammation through down-regulation of NF-κB signals and subsequent COX2, INOS, CCL-20, and TNF- α pathways [7].
3.2. In Vivo Anti-Inflamatory Effect of H. cordata
Ethanolic extract of the aerial parts of H. cordata was found to have anti-inflammatory effects on LPS in male ICR models of mice. Researchers observed that dosages of 100 and 400 milligrams per kilogram (mg/kg) reduced the total cell count by 46.1% and 66.5%, receptively. It was found to be as efficacious as dexamethasone, with a dose rate of 30 mg/kg [35]. Furthermore, when evaluated against a carrageenan-air pouch model, oral use of H. cordata supercritical extract at a 200 mg/kg dose suppressed albumin leakage, exudation, and infiltration of inflammatory cells in the ICR mice model. When the efficacy of this extract was compared with that of dexamethasone (2 mg/kg intraperitoneal) and indomethacin (2 mg/kg intraperitoneal), the extracts were found to be more effective at reducing both the cyclooxygenase 2/PGE2 and TNF-α/NO pathways [50]. In LPS-induced male ICR mice, quercitrin showed the greatest reduction in the cell count in the bronchoalveolar fluid after oral treatment of isolated flavonoids (hyperoside and quercitrin and afzelin at a dose rate of 100 mg/kg body weight) [28].
In a mouse model of D-galactosamine/LPS induction, the afzelin extracted from a methanolic fraction of HC lowered the quantity of inflammatory cytokines (TNF-α and IL-6) and AMPK expression, and enhanced the expression of Sirtuin-1 (Sirt-1) [39].
The effectiveness and potential mechanisms of the Houttuynia cordata (HC) extract in the treatment of (IC/BPS) interstitial cystitis/bladder pain syndrome were studied. A total of 30 adult female rats were divided randomly into three groups: a sham group (n = 10), a cyclophosphamide + saline (CYP + NS) group (n = 10), and a cyclophosphamide and Houttuynia cordata extract (CYP + HC) group (n = 10). The IC/BPS animal model was induced with the CYP + NS group and the CYP + HC group received cyclophosphamide (75 mg/kg, intraperitoneal injection, once every three days for 10 days), and the sham group rats received an equal volume of saline injection. Then, the three groups’ differences in urine frequency, nociceptive behaviours, cystometry, bladder weight, histological alterations, and cytokine (IL-6, IL-8, and TNF- α) concentration were assessed. The CYP + NS group considerably outperformed the sham and CYP + HC groups in terms of inflammatory grade, mast cell number, fraction of activated mast cells, bladder weight, cytokine concentration of bladder homogenates, and frequency of urination. Additionally, compared to the CYP + NS group, the CYP + HC group had a longer intercontraction interval, a larger bladder capacity, a higher nociceptive threshold, fewer mast cells, and a lower proportion of activated mast cells (p < 0.01) [53].
The second-phase anti-edematous effect of 3.08 and 6.16 mL/kg HCFP, which is comparable to that of diclofenac (150 mg/kg), was shown to diminish paw oedema in Wistar rats after 2 h of carrageenan stimulation. By comparison, 6.16 mL/kg HCFP also decreased paw oedema after 1 h of carrageenan stimulation, indicating the first-phase anti-edematous impact. Syringic, vanillic, p-hydroxybenzoic, and ferulic acids were among the active phenolic compounds quantified by HPLC that had anti-inflammatory properties [54]. These studies showed the anti-inflammatory property of H. cordata in vivo.

Table 2.
Different extracts of H. cordata to treat inflammation in vitro and vivo.
Table 2.
Different extracts of H. cordata to treat inflammation in vitro and vivo.
| Extracts | Doses | Standard Drug | Inflammagen Used | Model Used | Time Period | Minimal Active Concentration | Most Potent Biomolecule | Reference |
|---|---|---|---|---|---|---|---|---|
| Aqueous | 0.5–3 g/kg | None | DNA-BSA | ICR mouse | 2 h | 0.5 g/kg | Not mentioned | [55] |
| Aqueous | 1, 10, 20 µg/mL | None | DNA-BSA | RBL-2H3 | 30 min | 1 µg/mL | Not mentioned | [55] |
| Aqueous | 1, 2 g/mL | None | Acetaminophen | BALB/cA mice | 4 weeks | 2 g/L | Not mentioned | [29] |
| Ethanol (100%) | 0.1, 0.2, 1% | None | LTA (1 µg/mL) | RT-7 | 24 h | 0.5% | Not mentioned | [19] |
| Ethanol(80%) | 400, 600, 100 mg/kg | None | Oxaliplatin | Male Sprague Dawley rat | 15 days | 1000 mg/kg/day | Not mentioned | [56] |
| Ethanol | 0.05, 0.1, 0.2 mg/mL | None | PMA + Ca2+ ionophore | HMC-1 | 5 h | 0.2 mg/mL | Not mentioned | [57] |
| Essential oil | 0.01, 0.1, 1, 10, 100 µg/mL | NS-398 | LPS (1 µg/mL) | Mouse peritoneal macrophange | 24 h | 100 µg/mL | Not mentioned | [58] |
| Essential oil (sodium houttuyfonate and two undecanone) | 0.1,1,10,20 µg/mL | None | LPS (1 µg/mL) | RAW 264.7 | 24 h | 1 µg/mL | Sodium houttuyfonate | [29] |
| Essential oil (sodium houttuyfonate and 2 undecanone) | 100, 200, 400 mg/kg | Aspirin | Xylene | Mouse | 30 min | 200 mg/kg | Sodium houttuyfonate | [29] |
| Polysaccrides | 40, 80, 160 mg/kg | Dexamethasone | LPS | BALB/cA mice | 24 h | 40 mg/kg | Not mentioned | [59] |
| Essential oil | 20, 40 mg/kg | Dexamethasone | Xylene, Formaldehyde | Mouse | 7 days | 20 mg/kg | 2-undecane, n-Decanoic acid, Hexadecanoic acid-methyl ester, | [33] |
4. Effect of H. cordata on Different Organs
Lung inflammation is one of the most important signs during lung infection. H. cordata has an anti-inflammatory property that plays a significant role in treating lung inflammation. Quercetin obtained from HC, when administered orally at a dose rate of 100 mg/mL in an LPS-induced model, significantly decreased the production of NO and inflammatory mediators such as cytokines [35]. Researchers compared the effect of different dosage levels of flavonoid glucoside extract of H. cordata at 50, 100, and 200 mg/kg compared with ribavirin 100 mg/kg with the use of acute injury of lung tissues by the H1N1 virus. At 14 days, they found a lower lung index and less weight loss [37]. The oxidative lung damage caused pulmonary fibrosis. In rats, when pulmonary fibrosis was induced by bleomycin, H. cordata aqueous extracts showed a better and stronger anti-oxidant property than vitamin E by decreasing concentration of hydroxyproline, superoxide dismutase, and malondialdehyde [60].
The intestinal barrier is a structure that allows uptake of essential nutrients, while restricting pathogenic molecules and bacteria. The microflora present in the intestine also play a vital role in protecting the intestine [61]. The constituents of H. cordata are polysaccharides, and sodium houttuyfonate is instrumental in reducing or regulating the production of mucus from the goblet cells and wart formation of Secretory IgA (antibodies in the secretions and excretions). Moreover, the protein ZO-1, which forms a gap junction between the intestinal cells, is up-regulated or enhanced to compact intestinal, mechanical, and immunological barriers [62]. Intestinal inflammation induced by Salmonella typhimurium is dampened by sodium houttuyfonate in the form of up-regulating tight junction proteins between the mucosal cells of the intestine and the signaling pathway that leads to interleukin production [63]. These studies showed that H. cordata has a therapeutic effect on the GIT system.
Oxidative damage and inflammation disrupt salt metabolism, and the body’s water is a common cause of kidney diseases [64]. Treatment with 1 to 2% water extracts of H. cordata lowered creatine kinase activity and urea nitrogen in diabetic mice having kidney injury [65]. Inflammation can also cause serious kidney damage. A study found that sodium houttuyfonate dramatically decreased the expression of MCP-1 and NF-kB at a dose rate of 60 to 120 mg/kg in a dose-dependent manner [66]. These findings suggest that H. cordata extract is helpful in various kidney diseases.
Chemical compounds such as sodium houttuyfonate, flavonoids, and polysaccharides present in H. cordata have been used for the treatment of pneumonitis. H. cordata, a pro-inflammatory cytokine (IL 6) and IL 6, were reduced through oral use of quercetin (flavonoid) extracted from H. cordata at a dose rate of 100 mg/mL [35]. It was found that sodium houttuyfonate extracted from H. cordata causes a reduction in mRNA levels of MyD88, TLR4, and NF-κB p65 when used at a dose rate of 24.3 mg/kg. Macromolecular polysaccharides of H. cordata reduce lung injuries, pulmonary oedema, and pneumonia [67]. A combination of cell transplantation with H. cordata is used to treat lung inflammation. The extract at a dose rate of 50 mg/kg reduces the lungs’ inflammatory response. For infection caused by the H1N1 virus, extracts of flavonoid glycoside with a dose rate of 50, 100, and 200 mg/kg cause less weight loss and a lower lung index compared to the anti-viral drug ribavirin with a dose of 100 mg/kg. It causes inhibition of H1N1 neuraminidase activity, which reduces lung inflammation [37]. Water extract of H. cordata remarkably reduces inflammation of the lungs caused by bleomycin-induced pulmonary fibrosis in rats. It also has strong anti-oxidative activity compared to vitamin E. The active ingredients of H. cordata include polysaccharides, flavonoids, and sodium houttuyfonate, which have remarkable anti-inflammatory activity in the lungs [68].
H. cordata protects the intestine various barriers (mucosal barriers, chemical, mechanical, biological, and immune barriers) are present in the intestine. Moreover, intestinal flora also play an essential role in protecting the intestines. Recently, it has been found that sodium houttuyfonate and polysaccharides extracted from H. cordata decrease the expression of sIgA, intestinal goblet cells, and tight junction protein present in the intestines. Sodium houttuyfonate is also involved in reducing inflammation caused by Salmonella typhimurium. Regulation of bacteria, such as Vibrio and Bacillus, also includes polysaccharides made up of galactose, glucose, rhamnose, and arabinose at a 40 mg/kg dosage. These findings show that sodium houttuyfonate and polysaccharides of H. cordata have protective activity by inhibiting NF-κB and regulating intestinal flora in the intestines [69].
Many natural extracts from plants have effective results in preventing and treating various liver ailments. For instance, the chemical components of extracts such as terpenoids, glycosides, coumarins, and alkaloids prevent liver fibrosis. Cholestasis is a common problem inhibited by compounds such as quercetin and rutin. Liver cells are very sensitive to oxidative stress. H. cordata ethyl acetate extract reduces liver damage through its anti-oxidant activity. Ethyl acetate of H. cordata extract at a dose rate of 1000 mg/kg causes an increase in superoxide dismutase, glutathione, and catalase enzymes, and a decrease in malondialdehyde and serum transaminase resulting in liver protection. The mixture of ethanol and water extract of H. cordata at a dosage of 300 mg/kg/day for seven days reduces oxidative factors in the liver [38].
Oxidative damage, inflammation, and infections caused by various pathogenic organisms are the major factors involved in kidney problems. It was observed that 1 to 2% of H. cordata water extract reduced the level of serum creatinine and blood urea nitrogen and oxidative factors in the kidney. Moreover, 2% extract of H. cordata causes inhibition of membrane-anchored receptor made up of end products (RAGE) and glycation, which activate mitogen-activated protein kinase. They induce intracellular reactive oxygen species generation and are involved in renal protection. Sodium houttuyfonate present in H. cordata causes a decrease in expression of MCP-1 and nuclear NF-κB at a dosage of 60–120 mg/kg. It protects against renal glomerulonephritis and kidney oxidative stress [66].
Anti-oxidants such as catechin and procyanidin B present in H. cordata intervene in remodeling of the heart. The use of 2% H. cordata water extract was found to down-regulate cardiac activity related to oxygen, interleukin-6, inflammatory factors, and protein carbonyl. Moreover, 1 and 2% of H. cordata water extract block expression of NF-κB p65, p47phox, and p-p38 in the mouse heart. Sodium houttuyfonate shows activity against myocardial hypertrophy induced by isoprenaline with a dosage of 90 and 180 mg/kg up to 1 week. Cyclic adenosine, left ventricular weight index, heart weight index, and angiotensin 2 were also decreased using sodium houttuyfonate. Moreover, the cross-sectional area of cardiomyocytes and expression of hydroxyproline was also reduced [70]. Sodium houttuyfonate with 50 and 100 mg/kg dosages causes down-regulation of renin-angiotensin-aldosterone, which involves controlling blood pressure. Sodium houttuyfonate is also associated with NF-κB pathway inhibition and adenosine monophosphate-activated protein kinase at the same dosage. It also reduces heart fibrosis and myocardial inflammatory factors. H. cordata reduces the release of inflammatory mediators of the heart and oxidative damage to the heart. Sodium houttuyfonate present in H. cordata also affects the sympathetic nervous system and the renin-angiotensin system by reversing hypertrophy and remodeling of myocardium. Sodium houttuyfonate treatment elevated the activation of adenosine monophosphate-activated protein kinase (AMPK) on post-infarct heart and post-hypoxia H9C2. AMPK did not suppress NF-κB signaling directly; its inhibition of NF-κB was realized indirectly via its downstream mediators, e.g., Sirtuin-1 (SIRT1), Forkhead box O (F oxO) family, and peroxisome proliferator-activated receptor γ co-activator 1α (PGC-1α). Therefore, AMPK activation and suppression of NF-κB and inflammatory cytokines was critically involved in the anti-remodeling effect of SH post-myocardial infarction. [71].
5. Anti-Oxidant Effects of Various Extracts of H. cordata
In this study, anti-oxidant measurement tools for H. cordata were 1,1-Diphenyl-2-picrylhydrazyl (DPPH), 2,2′-azinobis (3-ethylbenzothiazoline-6-sulfonic acid) diammonium salt (ABTS), and β carotene assays, as shown in Table 2. IC50 values are defined as various concentrations of samples needed to scavenge 50% DPPH and ABTS free radicles present in test solutions. The most effective scavenger solution was methanol extract of H. cordata, and the second ranked was ethanol extract. However, there was no significant difference between IC50 values of ethanol and methanol extracts. Standard BHT was present in ABTS assay (p < 0.05). Chloroform and hexane extracts of H. cordata were weak radical scavengers in ABTS and DPPH models (Table 3).

Table 3.
Anti-oxidant effects of different extracts from H. cordata.
In the beta-carotene assay, the lipid peroxidation inhibition (LPI) with a data range of 63.35–86.61% was found in all extracts of H. cordata mentioned in Table 3. Higher LPI values indicate higher anti-oxidant activity. LPI methanol extract was found to have the highest values among all other extracts. Water and hexane extracts of H. cordata had the lowest anti-oxidant values [72]. The significant anti-oxidant effects of methanol extract of H. cordata are associated with many phenolic compounds [71].
6. Anti-Tumor Activity of H. cordata
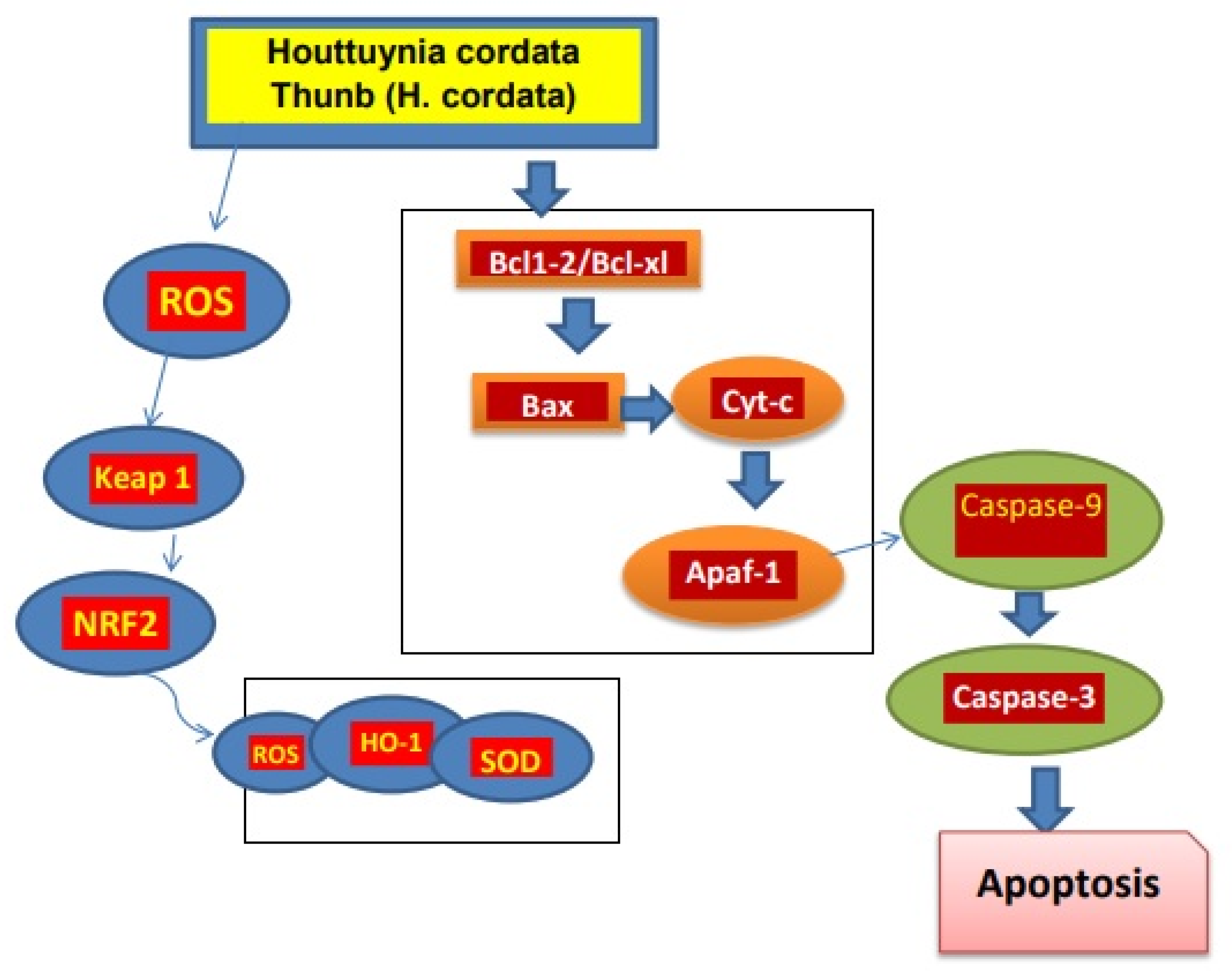
In a study of mice with lung tumors induced by benzo-pyrene, it was found that the active components of H. cordata, such as 2-undecanone, had an anti-tumor effect that may be due to Nrf2-HO-1/NQO-1 pathway activation, which reduces inflammation of lung cells and damage of DNA. In addition, no signs of systemic toxicity were recorded [73]. Moreover, the polysaccharides present in H. cordata exhibited anti-tumor potential. The polysaccharide HCA4S1 inhibited proliferation of tumor cells by cancer cell cycle/A549 lung tumor arrest and apoptosis. Similarly, after HCA4S1 treatment, the activities of cyclin B1 and cleaved caspase3 in cells dramatically reduced [74]. The extracts of H. cordata with the concentration of 0 to 80 µg/mL caused a decrease in lipid accumulation in HepG2 cells when HepG2 cells were merged with a high level of glucose [75]. The ethanolic extract of H. cordata had anti-cancer effects against the colon cancer cell line HT-29. Cancer cell apoptosis was induced when treated with 450 µg/mL extract, which also resulted in lower mitochondrial membrane potential and increased reactive oxygen [76]. H. cordata also has activity against breast cancer. The development and progression of tumors are significantly influenced by the overexpression of the HER2/neu (receptors on breast cells) receptor. With an IC50 of 5.52 µg/mL, Houttuyninum suppressed HER2 phosphorylation in a dose-dependent manner in MDA-MB-453 cells without altering the expression of the HER2/neu protein. Additionally, HER2/neu-mediated signal transduction pathway downstream molecules ERK1/2 and AKT were blocked by houttuyninum from becoming activated [77]. At the concentration of 100 to 500 µg/mL, the ethanolic extract of H. cordata promotes apoptosis in breast cancer cells [78]. These studies showed that H. cordata has anti-tumor activity (Figure 2).
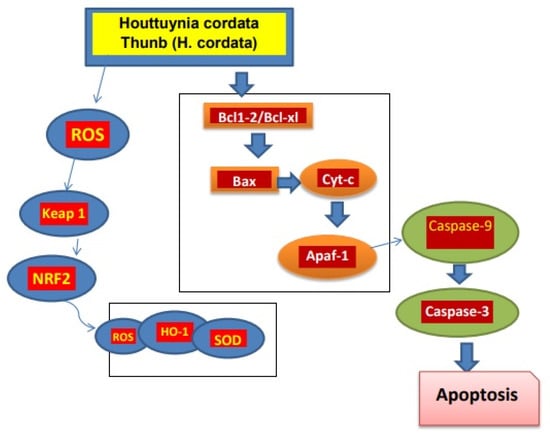
Figure 2.
Mechanism of H. cordata acts as an anti-tumor agent. H. cordata suppresses cancerous cells by blocking NRF2/Bcl1, Bcl-xl signaling pathway and leading to apoptosis of cancerous cells [7].
7. Effect of H. cordata on Viruses
The research on plants for the treatment of AIDS has made significant progress over the last ten years. Many plants and their products, such as H. cordata, have been found to have anti-HIV properties [79]. In vitro, the steam distillate and three main components from H. cordata manifested virucidal effects against HSIV-1 and influenza. The pretreatment with the distillate for 2 and 6 h, respectively, resulted in the inactivation of 20% and 40% of HIV-1 at two-fold dilution [80]. H. cordata aqueous extract has immunomodulatory and anti-SARS properties. H. cordata causes an increase in the spread of mouse splenic lymphocytes. According to flow cytometry, H. cordata enhanced the fraction of CD4+ and CD8+ T cells. Furthermore, it increased the interleukin 2 and interleukin 10 releases by mouse splenic cells. Regarding anti-viral activity, H. cordata inhibited the 3C-like protease of the SARSCOV and RNA-dependent RNA polymerase [81]
The flavonoid quercetin 7-rhamnoside (Q7R) present in H. cordata has anti-viral properties against the porcine epidemic diarrhea virus, the most common cause of severe enteropathogenic diarrhea as shown in Table 4. Using 0.014 μg/mL, it suppressed porcine epidemic diarrhea virus (PEDV) replication by 50% [82]. When tested for neuraminidase activity, H. cordata had anti-viral activity against the influenza virus and it totally suppressed viral neuraminidase with a dosage of 250 mg of herb/mL [83]. The flavonoids of H. cordata had anti-viral activity against the virus H1N1 both in vivo and in vitro. The combined action of hyperoside, rutin, quercitrin, and isoquercitrin in the extract significantly improved the life span and survival rate of mice suffering from H1N1. H. cordata extracts with a dose rate of 50, 100, and 200 mg/mL decreased H1N1 virus effects in lung tissues and decreased neuraminidase action of the virus [37]. Herpes simplex virus (HSV) infection was efficiently suppressed by H. cordata hot water extract, which might be due to the stoppage of the NF-κB pathway [84]. H. cordata inhibits avian infectious bronchitis virus, chicken embryo cells in vero cells and kidney cells, and decreases viral infection by up to 90 percent when detected with plaque reduction and reverse transcription PCR [85]. H. cordata water extract has anti-viral efficacy against the dengue virus serotype 2 strains 16,681. After pre and post-incubation with HepG2 cells, H. cordata at the dose rate of 10 to 100 mg/mL showed a significant reduction in intracellular DEN2 RNA synthesis, corresponding to a drop in dengue protein expression [84].
Coronaviruses are found throughout the world and infect humans and other animals. There are now seven types of coronaviruses that can infect humans. Recently found, severe acute respiratory syndrome coronavirus 2 (SARSCOV2) is one of these, and has caused millions of fatalities globally [86]. Three important proteins of SARS-CoV-2 are papain-like protease, protease (Mpro), and ADP ribose phosphatase. Molecular docking of LigPrep, Epic, and Glide modules of the Schrödinger suite 2020–3 indicated that phytocompound (ligand) 6-hydroxyondansetron had an affinity for PLpro and (Mpro) receptors. Results suggest that H. cordata may have therapeutic potential [87].

Table 4.
Anti-viral effect of H. cordata.
Table 4.
Anti-viral effect of H. cordata.
| Name of Extract | Name of Virus | Reference |
|---|---|---|
| Quercetin 7-rhamnoside (Q7R) | porcine epidemic diarrhea virus | [82] |
| Hyperoside, rutin, quercitrin | H1N1 | [37] |
| Hot water extract | Herpes simplex virus | [84,88] |
| H. cordata water extract | dengue virus serotype 2 strains 16,681 | [84] |
| H. cordata water extract | coronavirus 2 (SARSCOV2) | [84] |
| H. cordata polysaccharides | Murine Norovirus-1 | [89] |
| Houttuynoid A | Herpes Simplex Virus type-1 | [88] |
| Quercetin, quercetrin and cinanserin | Dengue fever virus, Corona virus | [90] |
| H. cordata extract | Enteric Virus | [91] |
| H. cordata essential oil | Avian infectious bronchitis | [92] |
8. Anti-Bacterial Effect of H. cordata
Staphylococcus aureus is a food-borne, gram-positive bacterium that can cause infection of the skin, nasal cavity, GIT, and other human parts. MRSA was synergistically inhibited by sodium houttuyfonate and EDTA-Na2. Mice infected with MRSA were given sodium houttuyfonate combined with EDTA-Na2. After 28 days of MRSA infection, the survival rate of mice with sodium houttuyfonate treatment combined with EDTA-Na2 was 75 percent. It was significantly higher than the 43.75 and 50 percent survival rates of mice treated independently with EDTA-Na2 and sodium houttuyfonate, respectively [93]. At doses of 500 and 50 mg/mL, the aqueous H. cordata extracts demonstrated anti-bacterial effects against isolates of MDR E. coli, with the maximum and minimum zone diameters of inhibition of 29 and 13 mm, respectively. These findings suggest that H. cordata water extract (HCWE) has anti-microbial action against MDR E. coli in vitro [20]
Pseudomonas aeruginosa is a Gram-negative bacterium that infects deep wounds of the body and causes systemic illness. It was reported that sodium houttuyfonate had anti-bacterial activity against pseudomonas aeruginosa. The biosynthesis of alginate, a key ingredient for PA biofilm development, was suppressed, and is linked to sodium houttuyfonate’s down-regulation of algD and algR genes. Simultaneously, electron microscope observations showed that the bacteria’s shape changed after treatment, and the amount of alginate present in bacterial biofilm decreased [94].
Water extracts of H. cordata were found to have anti-bacterial effects against salmonellosis. It was observed that, after 8 h, the anti-bacterial activity of H. cordata increased with concentrations of 25 to 100 mg/mL. Bacterial absorption and morphologic alterations of body cells showed that there was no significant difference in the replication of bacteria. H. cordata showed a decrease in the pathogenicity of salmonella bacterium. The death rate of bacteria at the 7th day in the untreated group was 100%, and with a dose rate of 25, 50, and 100 μg/mL of H. cordata, the extract group lived up to 11, 17, and 23 days, respectively. It was recorded that H. cordata water extract is effective and safe in treating salmonella bacteria infections and various replicating pathogens [95].
9. Toxicity of H. cordata
H. cordata is an edible plant. Therefore, the toxic level of this plant is mostly ignored. However, it has been reported in some studies that aristolactams and aristolochic acid present in H. cordata can cause cancer [60].
Increased levels of aristolochic acid in liver cells also cause toxicity of proximal tubule epithelial cells present in the kidney. Aristolochic acid is also toxic in vivo because of its mutagenicity. A study revealed that 95% ethanol extracts from H. cordata show potential toxicity to zebrafish. A single dose of 2000 mg/kg of H. cordata with oral use had no harmful effects in mice during 14 days of treatment. However, oral administration of H. cordata with a dosage of 500–1000 mg/kg/day for 28 consecutive days led to some rats’ death. The histopathological examination showed inflammatory cell infiltration and vacuum degeneration of liver tissue, and focal necrosis of epithelial cells in kidneys. However, H. cordata has shown a very weak potential for toxicity. There is no evidence that H. cordata causes long-term toxicity. Nonetheless, H. cordata leaves and rhizomes are consumed in South China as an agricultural vegetable [96].
Research was conducted in 2018 to evaluate the toxicological effect of fermented Houttuynia cordata juice (FHJ) in a rodent model. FHJ was prepared by fermentation of Houttuynia cordata for 30 days and its active ingredients were evaluated. Due to lactic acid production, it has a lower pH of 3.63. Rats were fed with FHJ for 60 days and toxicological effects were evaluated using different biochemical, hematological, and histological tests. These revealed that there was no significant biochemical, histological, or hematological change in rats when compared with the control group. Therefore, it was postulated that FHJ did not have any toxicological effect in rats; hence, experiments should be conducted with humans in safety and toxicological studies. [97].
10. Conclusions
H. cordata is an herbal and medicinal plant with various medical and biological effects. It has various functions, such as protecting organs, and has anti-inflammatory effects concerning bacteria, viruses, and tumors, and anti-oxidant activities [98]. Aristolochia components from H. cordata show toxicity to kidneys, which is not considered beneficial for health. However, data about its toxicity is currently insufficient. Similarly, anti-liver activity and liver injury associated with H. cordata are unclear. Overall, H. cordata is considered beneficial in reducing inflammation of various internal organs. Water extract, flavonoids, sodium houttuyfonate, volatile oil, ethanol extract, and polysaccharide components of H. cordata inhibit the release of inflammatory mediators for heart remodeling, lung injury, and other pathological changes in tissues. Apoptosis is the most important function of H. cordata associated with liver, lung, gastric, colon, and breast cancer. H. cordata is also found to be used in eye drops for the treatment of vernal keratoconjunctivitis, and treatment of mild to moderate acne and non-inflammatory skin lesions. It is also used to cure insulin resistance associated with diabetes. However, based on previous studies, it was concluded that the components of H. cordata extracts are still unclear, and their effects are not fully characterized [99].
11. Future Aspects
Due to the different components of H. cordata, its various pharmacological effects seem too optimistic. There are some gaps to be filled in the future to provide and understanding of its pathways and mechanisms, as follows:
- Mechanisms of H. cordata, such as interaction with cells, the cell membrane, and various drugs, will be of great importance.
- Studies on its relation to the blood–brain barrier, lipophilicity, cAMP signaling, and skin permeability with pharmaceutical effects will be of utmost use. However, its possible side-effects and toxic effects should be studied carefully.
- More data are required to study the pharmacological and toxicological activity of H. cordata.
Author Contributions
Writing—original draft preparation, S.R.; supervision and writing review, H.H.; visualization, M.I.; editing and proof reading, A.R. All authors have read and agreed to the published version of the manuscript.
Funding
This work was funded by grants from National natural science foundation of China (32172914), National key research and development program (2021YFD1800600), Fundamental Research Funds for the Central Universities (2662022DKYJC005).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Data sharing not applicable.
Conflicts of Interest
All authors have no conflict of interest.
References
- Drašar, P.; Moravcova, J. Recent Advances in Analysis of Chinese Medical Plants and Traditional Medicines. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2004, 812, 3–21. [Google Scholar] [CrossRef] [PubMed]
- Drasar, P.B.; Khripach, V.A. Growing Importance of Natural Products Research. Molecules 2020, 25, 6. [Google Scholar] [CrossRef] [PubMed]
- Hu, Q.; Zhang, W.; Wu, Z.; Tian, X.; Xiang, J.; Li, L.; Li, Z.; Peng, X.; Wei, S.; Ma, X.; et al. Baicalin and the Liver-Gut System: Pharmacological Bases Explaining Its Therapeutic Effects. Pharmacol. Res. 2021, 165, 105444. [Google Scholar] [CrossRef] [PubMed]
- Talman, A.M.; Clain, J.; Duval, R.; Ménard, R.; Ariey, F. Artemisinin Bioactivity and Resistance in Malaria Parasites. Trends Parasitol. 2019, 35, 953–963. [Google Scholar] [CrossRef]
- Farnsworth, N.R.; Akerele, O.; Bingel, A.S.; Soejarto, D.D.; Guo, Z. Medicinal Plants in Therapy. Bull. World Health Organ. 1985, 63, 965–981. [Google Scholar] [CrossRef]
- Chopra, R.N. Glossary of Indian Medicinal Plants; CSIR: New Delhi, India, 1956. [Google Scholar]
- Wu, Z.; Deng, X.; Hu, Q.; Xiao, X.; Jiang, J.; Ma, X.; Wu, M. Houttuynia Cordata Thunb: An Ethnopharmacological Review. Front. Pharmacol. 2021, 12, 714694. [Google Scholar] [CrossRef]
- Kumar, P.S.; Arivuchelvan, A.; Jagadeeswaran, A.; Punniamurthy, N.; Selvaraj, P.; Jagatheesan, P.N.R.; Mekala, P. Formulation of Enrofloxacin SLNs and Its Pharmacokinetics in Emu (Dromaius novaehollandiae) Birds. Appl. Nanosci. 2015, 5, 661–671. [Google Scholar] [CrossRef]
- Fu, J.; Dai, L.; Lin, Z.; Lu, H. Houttuynia cordata Thunb: A Review of Phytochemistry and Pharmacology and Quality Control. Chin. Med. 2013, 4, 101–123. [Google Scholar] [CrossRef]
- Li, G.Z.; Chai, O.H.; Lee, M.S.; Han, E.H.; Kim, H.T.; Song, C.H. Inhibitory Effects of Houttuynia Cordata Water Extracts on Anaphylactic Reaction and Mast Cell Activation. Biol. Pharm. Bull. 2005, 28, 1864–1868. [Google Scholar] [CrossRef]
- Chen, H.Y.; Lin, Y.H.; Thien, P.F.; Chang, S.C.; Chen, Y.C.; Lo, S.S.; Yang, S.H.; Chen, J.L. Identifying Core Herbal Treatments for Children with Asthma: Implication from a Chinese Herbal Medicine Database in Taiwan. Evid.-Based Complement. Altern. Med. 2013, 2013, 125943. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lu, H.M.; Liang, Y.Z.; Yi, L.Z.; Wu, X.J. Anti-Inflammatory Effect of Houttuynia Cordata Injection. J. Ethnopharmacol. 2006, 104, 245–249. [Google Scholar] [CrossRef]
- Park, E.; Kum, S.; Wang, C.; Park, S.Y.; Kim, B.S.; Schuller-Levis, G. Anti-Inflammatory Activity of Herbal Medicines: Inhibition of Nitric Oxide Production and Tumor Necrosis Factor-α Secretion in an Activated Macrophage-like Cell Line. Am. J. Chin. Med. 2005, 33, 415–424. [Google Scholar] [CrossRef] [PubMed]
- Chiang, L.C.; Chang, J.S.; Chen, C.C.; Ng, L.T.; Lin, C.C. Anti-Herpes Simplex Virus Activity of Bidens Pilosa and Houttuynia Cordata. Am. J. Chin. Med. 2003, 31, 355–362. [Google Scholar] [CrossRef] [PubMed]
- Ng, L.T.; Yen, F.L.; Liao, C.W.; Lin, C.C. Protective Effect of Houttuynia Cordata Extract on Bleomycin-Induced Pulmonary Fibrosis in Rats. Am. J. Chin. Med. 2007, 35, 465–475. [Google Scholar] [CrossRef] [PubMed]
- Chang, J.S.; Chiang, L.C.; Chen, C.C.; Liu, L.T.; Wang, K.C.; Lin, C.C. Atileukemic Activity of Bidenspilosa l. Var. Minor (Blume) Sherff and Houttuynia Cordata Thunb. Am. J. Chin. Med. 2001, 29, 303–312. [Google Scholar] [CrossRef]
- Kim, S.K.; Ryu, S.Y.; No, J.; Choi, S.U.; Kim, Y.S. Cytotoxic Alkaloids from Houttuynia Cordata. Arch. Pharm. Res. 2001, 24, 518–521. [Google Scholar] [CrossRef]
- Zhuang, T.; Li, F.; Huang, L.R.; Liang, J.Y.; Qu, W. Secondary Metabolites from the Plants of the Family Saururaceae and Their Biological Properties. Chem. Biodivers. 2015, 12, 194–220. [Google Scholar] [CrossRef]
- Sekita, Y.; Murakami, K.; Yumoto, H.; Mizuguchi, H.; Amoh, T.; Ogino, S.; Matsuo, T.; Miyake, Y.; Fukui, H.; Kashiwada, Y. Anti-Bacterial and Anti-Inflammatory Effects of Ethanol Extract from Houttuynia Cordata Poultice. Biosci. Biotechnol. Biochem. 2016, 80, 1205–1213. [Google Scholar] [CrossRef]
- Li, J.; Rehman, M.U.; Zhang, H.; Iqbal, M.K.; Mehmood, K.; Huang, S.; Nabi, F.; Sciences, A. Antibacterial Effect of the Water Extract of Houttuynia Cordata Water Extract Against Multi-Drug Resistant. Southeast Asian J. Trop. Med. Public Health 2017, 48, 1260–1266. [Google Scholar]
- Bauer, R.; Pröbstle, A.; Lotter, H.; Wagner-Redecker, W.; Matthiesen, U. Cyclooxygenase Inhibitory Constituents from Houttuynia Cordata. Phytomedicine 1996, 2, 305–308. [Google Scholar] [CrossRef]
- Ma, Q.; Wei, R.; Wang, Z.; Liu, W.; Sang, Z.; Li, Y.; Huang, H. Bioactive Alkaloids from the Aerial Parts of Houttuynia Cordata. J. Ethnopharmacol. 2017, 195, 166–172. [Google Scholar] [CrossRef] [PubMed]
- Probstle, A.; Bauer, R. Aristolactams and a 4,5-Dioxoaporphine Derivative from Houttuynia Cordata. Planta Med. 1992, 58, 568–569. [Google Scholar] [CrossRef]
- Nuengchamnong, N.; Krittasilp, K.; Ingkaninan, K. Rapid Screening and Identification of Antioxidants in Aqueous Extracts of Houttuynia Cordata Using LC-ESI-MS Coupled with DPPH Assay. Food Chem. 2009, 117, 750–756. [Google Scholar] [CrossRef]
- Chou, S.C.; Su, C.R.; Ku, Y.C.; Wu, T.S. The Constituents and Their Bioactivities of Houttuynia Cordata. Chem. Pharm. Bull. 2009, 57, 1227–1230. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Ye, H.; Wang, W.; Yu, L.; Chen, G. Determination of Flavonoids in Houttuynia cordata Thunb. and Saururus chinensis (Lour.) Bail. by Capillary Electrophoresis with Electrochemical Detection. Talanta 2006, 68, 759–764. [Google Scholar] [CrossRef] [PubMed]
- Lu, H.; Wu, X.; Liang, Y.; Zhang, J. Variation in Chemical Composition and Antibacterial Activities of Essential Oils from Two Species of Houttuynia THUNB. Chem. Pharm. Bull. 2006, 54, 936–940. [Google Scholar] [CrossRef]
- Ahn, J.; Chae, H.S.; Chin, Y.W.; Kim, J. Alkaloids from Aerial Parts of Houttuynia Cordata and Their Anti-Inflammatory Activity. Bioorganic Med. Chem. Lett. 2017, 27, 2807–2811. [Google Scholar] [CrossRef]
- Chen, J.; Wang, W.; Shi, C.; Fang, J. A Comparative Study of Sodium Houttuyfonate and 2-Undecanone for Their in Vitro and in Vivo Anti-Inflammatory Activities and Stabilities. Int. J. Mol. Sci. 2014, 15, 22978–22994. [Google Scholar] [CrossRef]
- Řebíčková, K.; Bajer, T.; Šilha, D.; Houdková, M.; Ventura, K.; Bajerová, P. Chemical Composition and Determination of the Antibacterial Activity of Essential Oils in Liquid and Vapor Phases Extracted from Two Different Southeast Asian Herbs-Houttuynia cordata (Saururaceae) and Persicaria odorata (Polygonaceae). Molecules 2020, 25, 2432. [Google Scholar] [CrossRef]
- Verma, R.S.; Joshi, N.; Padalia, R.C.; Singh, V.R.; Goswami, P.; Kumar, A.; Iqbal, H.; Verma, R.K.; Chanda, D.; Chauhan, A.; et al. Chemical Composition and Allelopathic, Antibacterial, Antifungal, and Antiacetylcholinesterase Activity of Fish-Mint (Houttuynia cordata Thunb.) from India. Chem. Biodivers. 2017, 14, 10. [Google Scholar] [CrossRef]
- Chen, S.D.; Gao, H.; Zhu, Q.C.; Wang, Y.Q.; Li, T.; Mu, Z.Q.; Wu, H.L.; Peng, T.; Yao, X.S. Houttuynoids A-E, Anti-Herpes Simplex Virus Active Flavonoids with Novel Skeletons from Houttuynia Cordata. Org. Lett. 2012, 14, 1772–1775. [Google Scholar] [CrossRef]
- Li, W.; Fan, T.; Zhang, Y.; Fan, T.; Zhou, P.; Niu, X.; He, L. Houttuynia Cordata Thunb. Volatile Oil Exhibited Anti-Inflammatory Effects in Vivo and Inhibited Nitric Oxide and Tumor Necrosis Factor-α Production in LPS-Stimulated Mouse Peritoneal Macrophages in Vitro. Phyther. Res. 2013, 27, 1629–1639. [Google Scholar] [CrossRef]
- Yadav, N.; Yadav, R.; Goyal, A. Chemistry of Terpenoids. Int. J. Pharm. Sci. Rev. Res. 2014, 27, 272–278. [Google Scholar]
- Lee, J.H.; Ahn, J.; Kim, J.W.; Lee, S.G.; Kim, H.P. Flavonoids from the Aerial Parts of Houttuynia Cordata Attenuate Lung Inflammation in Mice. Arch. Pharm. Res. 2015, 38, 1304–1311. [Google Scholar] [CrossRef] [PubMed]
- Kumar, M.; Prasad, S.; Hemalatha, S. A Current Update on the Phytopharmacological Aspects of Houttuynia Cordata Thunb. Pharmacogn. Rev. 2014, 8, 22–35. [Google Scholar] [CrossRef] [PubMed]
- Ling, L.-J.; Lu, Y.; Zhang, Y.-Y.; Zhu, H.-Y.; Tu, P.; Li, H.; Chen, D.-F. Flavonoids from Houttuynia Cordata Attenuate H1N1-Induced Acute Lung Injury in Mice via Inhibition of Influenza Virus and Toll-like Receptor Signalling. Phytomedicine 2020, 67, 153150. [Google Scholar] [CrossRef] [PubMed]
- Tian, L.; Shi, X.; Yu, L.; Zhu, J.; Ma, R.; Yang, X. Chemical Composition and Hepatoprotective Effects of Polyphenol-Rich Extract from Houttuynia Cordata Tea. J. Agric. Food Chem. 2012, 60, 4641–4648. [Google Scholar] [CrossRef]
- Lee, S.-B.; Kang, J.W.; Kim, S.J.; Ahn, J.; Kim, J.; Lee, S.M. Afzelin Ameliorates D-Galactosamine and Lipopolysaccharide-Induced Fulminant Hepatic Failure by Modulating Mitochondrial Quality Control and Dynamics. Br. J. Pharmacol. 2017, 174, 195–209. [Google Scholar] [CrossRef]
- Ge, Y.-C.; Wang, K.-W. New Analogues of Aporphine Alkaloids. Mini-Rev. Med. Chem. 2018, 18, 1590–1602. [Google Scholar] [CrossRef]
- Iwahori, A.R. NII-Electronic Library Service. Chem. Pharm. Bull. 1970, 2091. [Google Scholar]
- Qu, W.; Wu, F.H.; Li, J.; Liang, J.Y. Alkaloids from Houttuynia Cordata and Their Antiplatelet Aggregation Activities. Chin. J. Nat. Med. 2011, 9, 425–428. [Google Scholar] [CrossRef]
- Jungmi, K.; Inho, C.; Younggeun, L.; Hongsu. Identification of volatile essential oil components of Eoseongcho, fragrance properties and antibacterial activity of fractions-Ⅱ. Fragrant properties and antibacterial activity of fractions by Prep-HPLC. J. Korean Soc. Food Sci. Nutr. 1997, 26, 214–221. [Google Scholar]
- Kumar, R.; Clermont, G.; Vodovotz, Y.; Chow, C.C. The Dynamics of Acute Inflammation. J. Theor. Biol. 2004, 230, 145–155. [Google Scholar] [CrossRef] [PubMed]
- Roe, K. An Inflammation Classification System Using Cytokine Parameters. Scand. J. Immunol. 2021, 93, 6–10. [Google Scholar] [CrossRef] [PubMed]
- Marshall, G.D. Challenges in Allergy Immunology Practice: Solutions Needed for Persistent Patient Problems. Ann. Allergy Asthma Immunol. 2018, 121, 647–648. [Google Scholar] [CrossRef] [PubMed]
- Shim, S.Y.; Seo, Y.K.; Park, J.R. Down-Regulation of FcεRI Expression by Houttuynia Cordata Thunb Extract in Human Basophilic KU812F Cells. J. Med. Food 2009, 12, 383–388. [Google Scholar] [CrossRef]
- Satthakarn, S.; Chung, W.; Promsong, A.; Nittayananta, W. Houttuynia Cordata Modulates Oral Innate Immune Mediators: Potential Role of Herbal Plant on Oral Health. Oral Dis. 2015, 21, 512–518. [Google Scholar] [CrossRef]
- Saadat, S.; Beigoli, S.; Khazdair, M.R.; Amin, F.; Boskabady, M.H. Experimental and Clinical Studies on the Effects of Natural Products on Noxious Agents-Induced Lung Disorders, a Review. Front. Nutr. 2022, 9, 867914. [Google Scholar] [CrossRef]
- Shin, S.; Joo, S.S.; Jeon, J.H.; Park, D.; Jang, M.J.; Kim, T.O.; Kim, H.K.; Hwang, B.Y.; Kim, K.Y.; Kim, Y.B. Anti-Inflammatory Effects of a Houttuynia Cordata Supercritical Extract. J. Vet. Sci. 2010, 11, 273–275. [Google Scholar] [CrossRef]
- Zou, M.; Hu, X.; Wang, Y.; Wang, J.; Tang, F.; Liu, Y. Structural Characterization and Anti-Inflammatory Activity of a Pectin Polysaccharide HBHP-3 from Houttuynia Cordata. Int. J. Biol. Macromol. 2022, 210, 161–171. [Google Scholar] [CrossRef]
- Pakyntein, C.L.; Syiem, D.; Thabah, D.; Sunn, S.E. Antioxidant, Anti-Inflammatory and Anti-Hyperglycemic Activity of Aqueous and Methanolic Extract of Houttuynia Cordata: An in Vitro and in Vivo Study. GSC Biol. Pharm. Sci. 2021, 16, 145–154. [Google Scholar] [CrossRef]
- Li, W.; Yang, F.; Zhan, H.; Liu, B.; Cai, J.; Luo, Y.; Zhou, X. Houttuynia Cordata Extract Ameliorates Bladder Damage and Improves Bladder Symptoms via Anti-Inflammatory Effect in Rats with Interstitial Cystitis. Evid.-Based Complement. Altern. Med. 2020, 2020, 9026901. [Google Scholar] [CrossRef]
- Woranam, K.; Senawong, G.; Utaiwat, S.; Yunchalard, S.; Sattayasai, J.; Senawong, T. Anti-Inflammatory Activity of the Dietary Supplement Houttuynia Cordata Fermentation Product in RAW264.7 Cells and Wistar Rats. PLoS ONE 2020, 15, e0230645. [Google Scholar] [CrossRef] [PubMed]
- Han, E.H.; Park, J.H.; Kim, J.Y.; Jeong, H.G. Houttuynia Cordata Water Extract Suppresses Anaphylactic Reaction and IgE-Mediated Allergic Response by Inhibiting Multiple Steps of FcεRI Signaling in Mast Cells. Food Chem. Toxicol. 2009, 47, 1659–1666. [Google Scholar] [CrossRef] [PubMed]
- Wan, C.F.; Zheng, L.L.; Liu, Y.; Yu, X. Houttuynia Cordata Thunb Reverses Oxaliplatin-Induced Neuropathic Pain in Rat by Regulating Th17/Treg Balance. Am. J. Transl. Res. 2016, 8, 1609–1614. [Google Scholar]
- Lee, H.J.; Seo, H.S.; Kim, G.J.; Jeon, C.Y.; Park, J.H.; Jang, B.H.; Park, S.J.; Shin, Y.C.; Ko, S.G. Houttuynia Cordata Thunb Inhibits the Production of Pro-Inflammatory Cytokines through Inhibition of the NFκB Signaling Pathway in HMC-1 Human Mast Cells. Mol. Med. Rep. 2013, 8, 731–736. [Google Scholar] [CrossRef]
- Li, W.; Zhou, P.; Zhang, Y.; He, L. Houttuynia Cordata, a Novel and Selective COX-2 Inhibitor with Anti-Inflammatory Activity. J. Ethnopharmacol. 2011, 133, 922–927. [Google Scholar] [CrossRef]
- Xu, Y.Y.; Zhang, Y.Y.; Ou, Y.Y.; Lu, X.X.; Pan, L.Y.; Li, H.; Lu, Y.; Chen, D.F. Houttuynia Cordata Thunb. Polysaccharides Ameliorates Lipopolysaccharide-Induced Acute Lung Injury in Mice. J. Ethnopharmacol. 2015, 173, 81–90. [Google Scholar] [CrossRef]
- Ng, A.W.T.; Poon, S.L.; Huang, M.N.; Lim, J.Q.; Boot, A.; Yu, W.; Suzuki, Y.; Thangaraju, S.; Ng, C.C.Y.; Tan, P.; et al. Aristolochic Acids and Their Derivatives Are Widely Implicated in Liver Cancers in Taiwan and throughout Asia. Sci. Transl. Med. 2017, 9, eaan6446. [Google Scholar] [CrossRef]
- Lu, L.; Li, W.; Chen, L.; Su, Q.; Wang, Y.; Guo, Z.; Lu, Y.; Liu, B.; Qin, S. Radiation-Induced Intestinal Damage: Latest Molecular and Clinical Developments. Futur. Oncol. 2019, 15, 4105–4118. [Google Scholar] [CrossRef]
- Zhu, H.; Lu, X.; Ling, L.; Li, H.; Ou, Y.; Shi, X.; Lu, Y.; Zhang, Y.; Chen, D. Houttuynia Cordata Polysaccharides Ameliorate Pneumonia Severity and Intestinal Injury in Mice with Influenza Virus Infection. J. Ethnopharmacol. 2018, 218, 90–99. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Lv, H.; Li, Y.; Dong, N.; Bi, C.; Shan, A.; Wu, Z.; Shi, B. Sodium Houttuyfonate Enhances the Intestinal Barrier and Attenuates Inflammation Induced by Salmonella Typhimurium through the NF-ΚB Pathway in Mice. Int. Immunopharmacol. 2020, 89, 107058. [Google Scholar] [CrossRef] [PubMed]
- Gong, L.; Pan, Q.; Yang, N. Autophagy and Inflammation Regulation in Acute Kidney Injury. Front. Physiol. 2020, 11, 576463. [Google Scholar] [CrossRef]
- Hsu, C.-C.; Yang, H.-T.; Ho, J.-J.; Yin, M.-C.; Hsu, J.-Y. Houttuynia Cordata Aqueous Extract Attenuated Glycative and Oxidative Stress in Heart and Kidney of Diabetic Mice. Eur. J. Nutr. 2016, 55, 845–854. [Google Scholar] [CrossRef]
- Pan, P.; Wang, Y.J.; Han, L.; Liu, X.; Zhao, M.; Yuan, Y.F. Effects of Sodium Houttuyfonate on Expression of NF-KappaB and MCP-1 in Membranous Glomerulonephritis. J. Ethnopharmacol. 2010, 131, 203–209. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Jiang, Y.; Ling, L.; Zhang, Y.; Li, H.; Chen, D. Beneficial Effects of Houttuynia Cordata Polysaccharides on “Two-Hit” Acute Lung Injury and Endotoxic Fever in Rats Associated with Anti-Complementary Activities. Acta Pharm. Sin. B 2018, 8, 218–227. [Google Scholar] [CrossRef]
- Shilovskiy, I.P.; Yumashev, K.V.; Nikolsky, A.A.; Vishnyakova, L.I.; Khaitov, M.R. Molecular and Cellular Mechanisms of Respiratory Syncytial Viral Infection: Using Murine Models to Understand Human Pathology. Biochemistry 2021, 86, 290–306. [Google Scholar] [CrossRef]
- Chen, M.Y.; Li, H.; Lu, X.X.; Ling, L.J.; Weng, H.B.; Sun, W.; Chen, D.F.; Zhang, Y.Y. Houttuynia Cordata Polysaccharide Alleviated Intestinal Injury and Modulated Intestinal Microbiota in H1N1 Virus Infected Mice. Chin. J. Nat. Med. 2019, 17, 187–197. [Google Scholar] [CrossRef]
- Gao, J.P.; Wang, Y.; Lü, J.; Gu, W.L.; Chen, C.X. Effect of Sodium Houttuyfonate on Myocardial Hypertrophy in Mice and Rats. J. Pharm. Pharmacol. 2009, 61, 677–683. [Google Scholar] [CrossRef]
- Tu, X.; Deng, Y.P.; Chen, J.; Hu, Q.; He, C.S.; Jordan, J.B.; Zhong, S. Screening Study on the Anti-Angiogenic Effects of Traditional Chinese Medicine–Part I: Heat-Clearing and Detoxicating TCM. J. Ethnopharmacol. 2016, 194, 280–287. [Google Scholar] [CrossRef]
- Tuyen, P.T.; Khang, D.T.; Anh, T.T.T.; Trang, P.T.; Xuan, T.D. Antioxidant Properties and Total Phenolic Contents of Various Extracts from Houttuynia Cordata Thunb. Tap Chi Sinh Hoc 2018, 40, 149–154. [Google Scholar] [CrossRef]
- Lou, Y.; Guo, Z.; Zhu, Y.; Kong, M.; Zhang, R.; Lu, L.; Wu, F.; Liu, Z.; Wu, J. Houttuynia Cordata Thunb. and Its Bioactive Compound 2-Undecanone Significantly Suppress Benzo(a)Pyrene-Induced Lung Tumorigenesis by Activating the Nrf2-HO-1/NQO-1 Signaling Pathway. J. Exp. Clin. Cancer Res. 2019, 38, 242. [Google Scholar] [CrossRef] [PubMed]
- Han, K.; Jin, C.; Chen, H.; Wang, P.; Yu, M.; Ding, K. Structural Characterization and Anti-A549 Lung Cancer Cells Bioactivity of a Polysaccharide from Houttuynia Cordata. Int. J. Biol. Macromol. 2018, 120, 288–296. [Google Scholar] [CrossRef] [PubMed]
- Kang, H.; Koppula, S. Houttuynia Cordata Attenuates Lipid Accumulation via Activation of AMP-Activated Protein Kinase Signaling Pathway in HepG2 Cells. Am. J. Chin. Med. 2014, 42, 651–664. [Google Scholar] [CrossRef] [PubMed]
- Routhier, A.; Astuccio, M.; Lahey, D.; Monfredo, N.; Johnson, A.; Callahan, W.; Partington, A.; Fellows, K.; Ouellette, L.; Zhidro, S.; et al. Pharmacological Inhibition of Rho-Kinase Signaling with Y-27632 Blocks Melanoma Tumor Growth. Oncol. Rep. 2010, 23, 861–867. [Google Scholar] [CrossRef]
- Zhou, N.N.; Tang, J.; Chen, W.D.; Feng, G.K.; Xie, B.F.; Liu, Z.C.; Yang, D.; Zhu, X.F. Houttuyninum, an Active Constituent of Chinese Herbal Medicine, Inhibits Phosphorylation of HER2/Neu Receptor Tyrosine Kinase and the Tumor Growth of HER2/Neu-Overexpressing Cancer Cells. Life Sci. 2012, 90, 770–775. [Google Scholar] [CrossRef]
- Subhawa, S.; Chewonarin, T.; Banjerdpongchai, R. The Effects of Houttuynia Cordata Thunb and Piper Ribesioideswall Extracts on Breast Carcinoma Cell Proliferation, Migration, Invasion and Apoptosis. Molecules 2020, 25, 1196. [Google Scholar] [CrossRef]
- Sandip, B.J.J.M.A.P.S. Medicinal Plants with Anti-HIV Potential. Clin. Exp. Pharmacol. Physiol. 2003, 25, 427–440. [Google Scholar]
- Hayashi, K.; Kamiya, M.; Hayashi, T. Virucidal Effects of the Steam Distillate from Houttuynia Cordata and Its Components on HSV-1, Influenza Virus, and HIV. Planta Med. 1995, 61, 237–241. [Google Scholar] [CrossRef]
- Lau, K.M.; Lee, K.M.; Koon, C.M.; Cheung, C.S.F.; Lau, C.P.; Ho, H.M.; Lee, M.Y.H.; Au, S.W.N.; Cheng, C.H.K.; Lau, C.B.S.; et al. Immunomodulatory and Anti-SARS Activities of Houttuynia Cordata. J. Ethnopharmacol. 2008, 118, 79–85. [Google Scholar] [CrossRef]
- Choi, H.J.; Kim, J.H.; Lee, C.H.; Ahn, Y.J.; Song, J.H.; Baek, S.H.; Kwon, D.H. Antiviral Activity of Quercetin 7-Rhamnoside against Porcine Epidemic Diarrhea Virus. Antivir. Res. 2009, 81, 77–81. [Google Scholar] [CrossRef] [PubMed]
- Han, X.; Zhang, D.K.; Guo, Y.M.; Feng, W.W.; Dong, Q.; Zhang, C.E.; Zhou, Y.F.; Liu, Y.; Wang, J.B.; Zhao, Y.L.; et al. Screening and Evaluation of Commonly-Used Anti-Influenza Chinese Herbal Medicines Based on Anti-Neuraminidase Activity. Chin. J. Nat. Med. 2016, 14, 794–800. [Google Scholar] [CrossRef]
- Chen, X.; Wang, Z.; Yang, Z.; Wang, J.; Xu, Y.; Tan, R.-X.; Li, E. Houttuynia Cordata Blocks HSV Infection through Inhibition of NF-ΚB Activation. Antivir. Res. 2011, 92, 341–345. [Google Scholar] [CrossRef] [PubMed]
- Yin, J.; Li, G.; Li, J.; Yang, Q.; Ren, X. In Vitro and in Vivo Effects of Houttuynia Cordata on Infectious Bronchitis Virus. Avian Pathol. 2011, 40, 491–498. [Google Scholar] [CrossRef]
- Adhikari, B.; Marasini, B.P.; Rayamajhee, B.; Bhattarai, B.R.; Lamichhane, G.; Khadayat, K.; Adhikari, A.; Khanal, S.; Parajuli, N. Potential Roles of Medicinal Plants for the Treatment of Viral Diseases Focusing on COVID-19: A Review. Phyther. Res. 2021, 35, 1298–1312. [Google Scholar] [CrossRef]
- Das, S.K.; Mahanta, S.; Tanti, B.; Tag, H.; Hui, P.K. Identification of Phytocompounds from Houttuynia Cordata Thunb. as Potential Inhibitors for SARS-CoV-2 Replication Proteins through GC–MS/LC–MS Characterization, Molecular Docking and Molecular Dynamics Simulation. Mol. Divers. 2022, 26, 365–388. [Google Scholar] [CrossRef]
- Li, T.; Liu, L.; Wu, H.; Chen, S.; Zhu, Q.; Gao, H.; Yu, X.; Wang, Y.; Su, W.; Yao, X.; et al. Anti-Herpes Simplex Virus Type 1 Activity of Houttuynoid A, a Flavonoid from Houttuynia Cordata Thunb. Antivir. Res. 2017, 144, 273–280. [Google Scholar] [CrossRef]
- Cheng, D.; Sun, L.; Zou, S.; Chen, J.; Mao, H.; Zhang, Y.; Liao, N.; Zhang, R. Antiviral Effects of Houttuynia Cordata Polysaccharide Extract on Murine Norovirus-1 (MNV-1)-A Human Norovirus Surrogate. Molecules 2019, 24, 1835. [Google Scholar] [CrossRef]
- Chiow, K.H.; Phoon, M.C.; Putti, T.; Tan, B.K.H.; Chow, V.T. Evaluation of Antiviral Activities of Houttuynia Cordata Thunb. Extract, Quercetin, Quercetrin and Cinanserin on Murine Coronavirus and Dengue Virus Infection. Asian Pac. J. Trop. Med. 2016, 9, 1–7. [Google Scholar] [CrossRef]
- Lin, T.Y.; Liu, Y.C.; Jheng, J.R.; Tsai, H.P.; Jan, J.T.; Wong, W.R.; Horng, J.T. Anti-Enterovirus 71 Activity Screening of Chinese Herbs with Anti-Infection and Inflammation Activities. Am. J. Chin. Med. 2009, 37, 143–158. [Google Scholar] [CrossRef]
- Lelešius, R.; Karpovaite, A.; Mickiene, R.; Drevinskas, T.; Tiso, N.; Ragažinskiene, O.; Kubiliene, L.; Maruška, A.; Šalomskas, A. In Vitro Antiviral Activity of Fifteen Plant Extracts against Avian Infectious Bronchitis Virus. BMC Vet. Res. 2019, 15, 178. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.; Duan, Q.; Li, F.; Shao, J.; Cheng, H.; Wu, D. Sodium Houttuyfonate and EDTA-Na2 in Combination Effectively Inhibits Pseudomonas Aeruginosa, Staphylococcus Aureus and Candida Albicans in Vitro and in Vivo. Bioorganic Med. Chem. Lett. 2015, 25, 142–147. [Google Scholar] [CrossRef] [PubMed]
- Wu, D.Q.; Cheng, H.; Duan, Q.; Huang, W. Sodium Houttuyfonate Inhibits Biofilm Formation and Alginate Biosynthesis-Associated Gene Expression in a Clinical Strain of Pseudomonas Aeruginosa in Vitro. Exp. Ther. Med. 2015, 10, 753–758. [Google Scholar] [CrossRef]
- Kim, G.S.; Kim, D.H.; Ju, L.J.; Lee, J.J.; Han, D.Y.; Lee, W.M.; Jung, W.C.; Min, W.G.; Won, C.G.; Rhee, M.H.; et al. Biological and Antibacterial Activities of the Natural Herb Houttuynia Cordata Water Extract against the Intracellular Bacterial Pathogen Salmonella within the RAW 264.7 Macrophage. Biol. Pharm. Bull. 2008, 31, 2012–2017. [Google Scholar] [CrossRef]
- Chen, H.; Sha, X.; Luo, Y.; Chen, J.; Li, X.; Wang, J.; Cao, G.; Peng, X. Acute and Subacute Toxicity Evaluation of Houttuynia Cordata Ethanol Extract and Plasma Metabolic Profiling Analysis in Both Male and Female Rats. J. Appl. Toxicol. 2021, 41, 2068–2082. [Google Scholar] [CrossRef]
- Chaiyasut, C.; Sivamaruthi, B.S.; Duangjitcharoen, Y.; Kesika, P.; Sirilun, S.; Chaiyasut, K.; Peerajan, S. Assessment of Subchronic Toxicity of Fermented Houttuynia Cordata Thunb. Using Rodent Model System. Asian J. Pharm. Clin. Res. 2018, 11, 307–311. [Google Scholar] [CrossRef]
- Shingnaisui, K.; Dey, T.; Manna, P.; Kalita, J. Therapeutic Potentials of Houttuynia Cordata Thunb. against Inflammation and Oxidative Stress: A Review. J. Ethnopharmacol. 2018, 220, 35–43. [Google Scholar] [CrossRef]
- Heinrich, M.; Appendino, G.; Efferth, T.; Fürst, R.; Izzo, A.A.; Kayser, O.; Pezzuto, J.M.; Viljoen, A. Best Practice in Research–Overcoming Common Challenges in Phytopharmacological Research. J. Ethnopharmacol. 2020, 246, 112230. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).