The Impact of Exercise on Cardiotoxicity in Pediatric and Adolescent Cancer Survivors: A Scoping Review
Abstract
1. Introduction
2. Materials and Methods
2.1. Search Strategy
2.2. Inclusion and Exclusion Criteria
2.3. Information Sources
2.4. Study Selection
2.5. Data Extraction
2.6. Data Synthesis and Analysis
3. Results
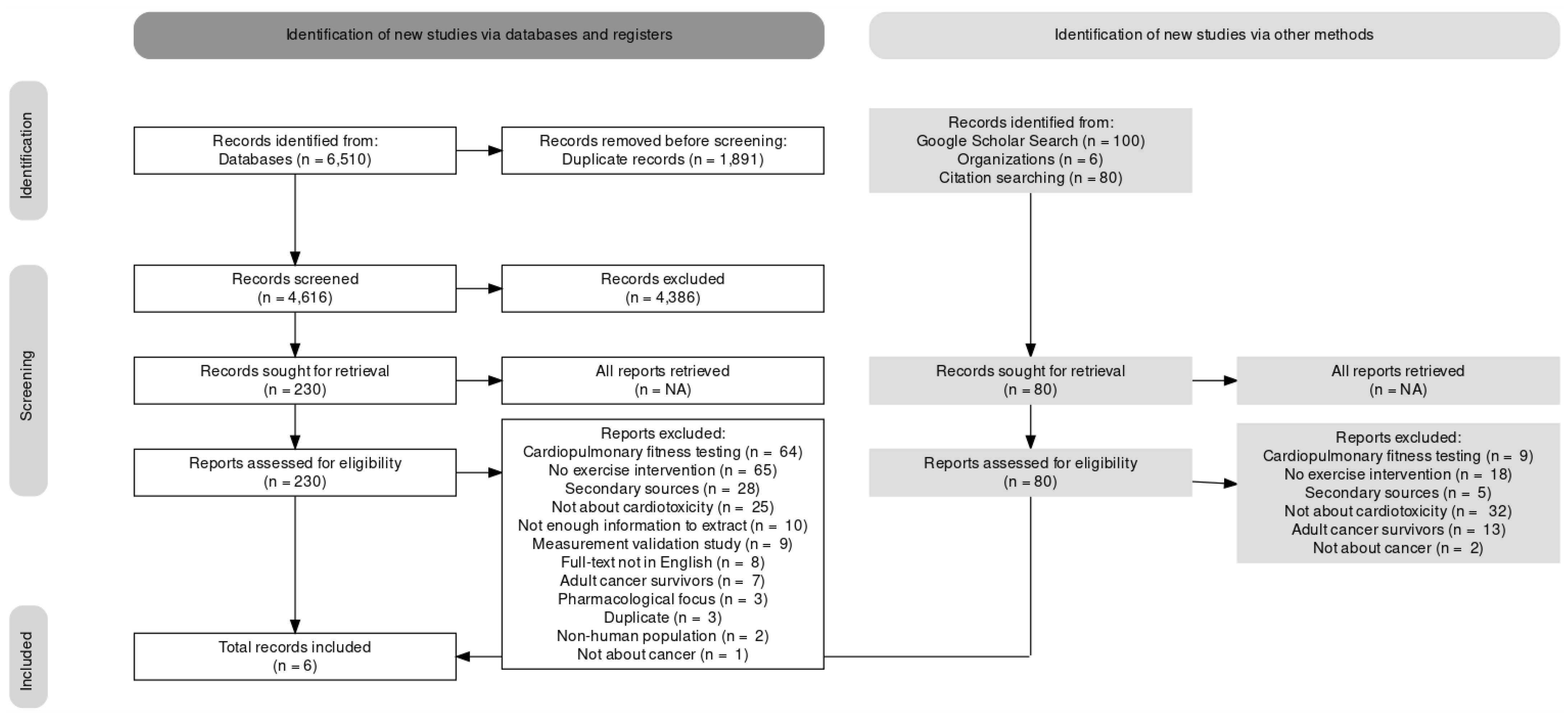
3.1. Study Selection
3.2. Characteristics of Included Studies
3.3. Patient Characteristics of the Included Studies
3.4. Exercise Intervention Characteristics of the Included Studies
3.5. Key Findings of the Included Studies Relating to Peripheral Cardiovascular Health
4. Discussion
4.1. Impact of Reviewed Exercise Interventions
4.2. Exercise Intervention Designs of Reviewed Studies
4.3. Limitations
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| 1 | (Cancer* OR Neoplas* OR Leukemia* OR Leukaemia* OR Tumor* OR Tumour* OR Lymphoma* OR Chemotherap* OR Malignanc* OR anthracycline* OR ‘Antineoplastic Agent*’ OR Immunotherap* OR ‘Monoclonal Antibod*’ OR ‘Tyrosine Kinase Inhibitor*’ OR Radiation OR Radiology) |
| 2 | Child* OR Adolescent* OR Teen* OR ‘Young Adult*’ OR ‘Early Child*’ OR Pediatric* OR Paediatric* OR Infant* OR Toddler* OR Bab* OR Juvenile* OR ‘Pre Pubescent*’ |
| 3 | 1 AND 2 |
| 4 | Exercise* OR ‘Resistance Training*’ OR Aerobic* OR ‘Motor Activity’ OR ‘Exercise Therap*’ OR ‘Physical Activit*’ OR Training OR ‘Physical Fitness’ OR Exertion OR Yoga OR Pilates OR ‘Dance Therap*’ OR ‘Tai Ji’ OR Qigong |
| 5 | Exp Exercise/ |
| 6 | 4 OR 5 |
| 7 | 3 AND 6 |
| 8 | Myocarditis* OR ‘Heart Failure’ OR Cardiotoxic* or Cardiomyopath* OR Heart* OR ‘Radiation Injury*’ |
| 9 | 7 AND 8 |
Appendix B
- Cancer AND Child AND Exercise AND Cardio*
- Cancer AND Pediatric AND Exercise AND Cardio*
- Cancer AND Child AND “Physical Activity” AND Cardio*
- cancer AND child AND exercise AND cardiotoxicity
- cancer AND pediatric AND exercise and cardiotoxicity
- cancer AND child AND ‘Physical Activity’ AND cardiotoxicity
- Canadian cancer society
- American Cancer Society
- Cancer Research UK
- National Health Institute
- American College of Sports Medicine
- Canadian Society for Exercise Physiologies
- Canadian Cardiology Society
Appendix C
- Title
- Contact
- Year
- Country
- Aim
- Design
- Start date
- End date
- Inclusion criteria
- Exclusion criteria
- Method of recruitment
- Cancer type
- Cancer stage
- Time since diagnosis
- Control group details
- Age of participants
- Chemotherapy treatment
- Radiotherapy
- Exercise program
- o
- Setting
- o
- Frequency
- o
- Intensity
- o
- Time
- o
- Type
- o
- Duration
- o
- Location
- o
- Instructor
- o
- Adherence
- Measure of cardiac health
- Other outcome measures
- Results
- Key findings
- Limitations
- Implications
References
- Arruebo, M.; Vilaboa, N.; Sáez-Gutierrez, B.; Lambea, J.; Tres, A.; Valladares, M.; González-Fernández, Á. Assessment of the Evolution of Cancer Treatment Therapies. Cancers 2011, 3, 3279–3330. [Google Scholar] [CrossRef] [PubMed]
- Lipshultz, S.E.; Adams, M.J.; Colan, S.D.; Constine, L.S.; Herman, E.H.; Hsu, D.T.; Hudson, M.M.; Kremer, L.C.; Landy, D.C.; Miller, T.L.; et al. Long-Term Cardiovascular Toxicity in Children, Adolescents, and Young Adults Who Receive Cancer Therapy: Pathophysiology, Course, Monitoring, Management, Prevention, and Research Directions. Circulation 2013, 128, 1927–1995. [Google Scholar] [CrossRef] [PubMed]
- Chow, E.J.; Leger, K.J.; Bhatt, N.S.; Mulrooney, D.A.; Ross, C.J.; Aggarwal, S.; Bansal, N.; Ehrhardt, M.J.; Armenian, S.H.; Scott, J.M.; et al. Paediatric Cardio-Oncology: Epidemiology, Screening, Prevention, and Treatment. Cardiovasc. Res. 2019, 115, 922–934. [Google Scholar] [CrossRef] [PubMed]
- Armenian, S.H.; Armstrong, G.T.; Aune, G.; Chow, E.J.; Ehrhardt, M.J.; Ky, B.; Moslehi, J.; Mulrooney, D.A.; Nathan, P.C.; Ryan, T.D.; et al. Cardiovascular Disease in Survivors of Childhood Cancer: Insights Into Epidemiology, Pathophysiology, and Prevention. J. Clin. Oncol. 2018, 36, 2135–2144. [Google Scholar] [CrossRef]
- Ryan, T.D.; Nagarajan, R.; Godown, J. Pediatric Cardio-Oncology: Development of Cancer Treatment-Related Cardiotoxicity and the Therapeutic Approach to Affected Patients. Curr. Treat. Options Oncol. 2019, 20, 56. [Google Scholar] [CrossRef]
- Mulrooney, D.A.; Yeazel, M.W.; Kawashima, T.; Mertens, A.C.; Mitby, P.; Stovall, M.; Donaldson, S.S.; Green, D.M.; Sklar, C.A.; Robison, L.L.; et al. Cardiac Outcomes in a Cohort of Adult Survivors of Childhood and Adolescent Cancer: Retrospective Analysis of the Childhood Cancer Survivor Study Cohort. BMJ 2009, 339, b4606. [Google Scholar] [CrossRef]
- Lipshultz, S.E.; Colan, S.D.; Gelber, R.D.; Perez-Atayde, A.R.; Sallan, S.E.; Sanders, S.P. Late Cardiac Effects of Doxorubicin Therapy for Acute Lymphoblastic Leukemia in Childhood. N. Engl. J. Med. 1991, 324, 808–815. [Google Scholar] [CrossRef]
- Tan, C.; Tasaka, H.; Yu, K.-P.; Murphy, M.L.; Karnofsky, D.A. Daunomycin, an Antitumor Antibiotic, in the Treatment of Neoplastic Disease. Clinical Evaluation with Special Reference to Childhood Leukemia. Cancer 1967, 20, 333–353. [Google Scholar] [CrossRef]
- Chao, C.; Xu, L.; Bhatia, S.; Cooper, R.; Brar, S.; Wong, F.L.; Armenian, S.H. Cardiovascular Disease Risk Profiles in Survivors of Adolescent and Young Adult (AYA) Cancer: The Kaiser Permanente AYA Cancer Survivors Study. J. Clin. Oncol. 2016, 34, 1626–1633. [Google Scholar] [CrossRef]
- Kang, D.-W.; Wilson, R.L.; Christopher, C.N.; Normann, A.J.; Barnes, O.; Lesansee, J.D.; Choi, G.; Dieli-Conwright, C.M. Exercise Cardio-Oncology: Exercise as a Potential Therapeutic Modality in the Management of Anthracycline-Induced Cardiotoxicity. Front. Cardiovasc. Med. 2022, 8, 2194. [Google Scholar] [CrossRef]
- Chow, E.J.; Antal, Z.; Constine, L.S.; Gardner, R.; Wallace, W.H.; Weil, B.R.; Yeh, J.M.; Fox, E. New Agents, Emerging Late Effects, and the Development of Precision Survivorship. J. Clin. Oncol. 2018, 36, 2231–2240. [Google Scholar] [CrossRef] [PubMed]
- Kopp, L.M.; Womer, R.B.; Schwartz, C.L.; Ebb, D.H.; Franco, V.I.; Hall, D.; Barkauskas, D.A.; Krailo, M.D.; Grier, H.E.; Meyers, P.A.; et al. Effects of Dexrazoxane on Doxorubicin-Related Cardiotoxicity and Second Malignant Neoplasms in Children with Osteosarcoma: A Report from the Children’s Oncology Group. Cardio-Oncology 2019, 5, 15. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Nie, X.; Zhang, P.; Huang, Y.; Ma, L.; Li, F.; Yi, M.; Qin, W.; Yuan, X. Dexrazoxane Ameliorates Radiation-Induced Heart Disease in a Rat Model. Aging 2021, 13, 3699–3711. [Google Scholar] [CrossRef]
- Eneh, C.; Lekkala, M.R. Dexrazoxane. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2022. [Google Scholar]
- Saleh, Y.; Abdelkarim, O.; Herzallah, K.; Abela, G.S. Anthracycline-Induced Cardiotoxicity: Mechanisms of Action, Incidence, Risk Factors, Prevention, and Treatment. Heart Fail. Rev. 2021, 26, 1159–1173. [Google Scholar] [CrossRef] [PubMed]
- Chicco, A.J.; Schneider, C.M.; Hayward, R. Voluntary Exercise Protects against Acute Doxorubicin Cardiotoxicity in the Isolated Perfused Rat Heart. Am. J. Physiol. -Regul. Integr. Comp. Physiol. 2005, 289, R424–R431. [Google Scholar] [CrossRef]
- Guo, S.; Wong, S. Cardiovascular Toxicities from Systemic Breast Cancer Therapy. Front. Oncol. 2014, 4, 346. [Google Scholar] [CrossRef][Green Version]
- Hayward, R.; Lien, C.-Y.; Jensen, B.T.; Hydock, D.S.; Schneider, C.M. Exercise Training Mitigates Anthracycline-Induced Chronic Cardiotoxicity in a Juvenile Rat Model. Pediatr. Blood Cancer 2012, 59, 149–154. [Google Scholar] [CrossRef] [PubMed]
- Hofmann, U.; Frantz, S. How Can We Cure a Heart “in Flame”? A Translational View on Inflammation in Heart Failure. Basic Res. Cardiol. 2013, 108, 356. [Google Scholar] [CrossRef]
- Hydock, D.S.; Lien, C.-Y.; Jensen, B.T.; Parry, T.L.; Schneider, C.M.; Hayward, R. Rehabilitative Exercise in a Rat Model of Doxorubicin Cardiotoxicity. Exp. Biol. Med. 2012, 237, 1483–1492. [Google Scholar] [CrossRef]
- Jones, L.W.; Fels, D.R.; West, M.; Allen, J.D.; Broadwater, G.; Barry, W.T.; Wilke, L.G.; Masko, E.; Douglas, P.S.; Dash, R.C.; et al. Modulation of Circulating Angiogenic Factors and Tumor Biology by Aerobic Training in Breast Cancer Patients Receiving Neoadjuvant Chemotherapy. Cancer Prev. Res. 2013, 6, 925–937. [Google Scholar] [CrossRef]
- Hornsby, W.E.; Douglas, P.S.; West, M.J.; Kenjale, A.A.; Lane, A.R.; Schwitzer, E.R.; Ray, K.A.; Herndon, J.E.; Coan, A.; Gutierrez, A.; et al. Safety and Efficacy of Aerobic Training in Operable Breast Cancer Patients Receiving Neoadjuvant Chemotherapy: A Phase II Randomized Trial. Acta Oncol. 2014, 53, 65–74. [Google Scholar] [CrossRef] [PubMed]
- Kirkham, A.A.; Virani, S.A.; Bland, K.A.; McKenzie, D.C.; Gelmon, K.A.; Warburton, D.E.R.; Campbell, K.L. Exercise Training Affects Hemodynamics Not Cardiac Function during Anthracycline-Based Chemotherapy. Breast Cancer Res. Treat. 2020, 184, 75–85. [Google Scholar] [CrossRef] [PubMed]
- Howden, E.J.; Bigaran, A.; Beaudry, R.; Fraser, S.; Selig, S.; Foulkes, S.; Antill, Y.; Nightingale, S.; Loi, S.; Haykowsky, M.J.; et al. Exercise as a Diagnostic and Therapeutic Tool for the Prevention of Cardiovascular Dysfunction in Breast Cancer Patients. Eur. J. Prev. Cardiol. 2019, 26, 305–315. [Google Scholar] [CrossRef]
- Foulkes, S.; Costello, B.T.; Howden, E.J.; Janssens, K.; Dillon, H.; Toro, C.; Claus, P.; Fraser, S.F.; Daly, R.M.; Elliott, D.A.; et al. Exercise Cardiovascular Magnetic Resonance Reveals Reduced Cardiac Reserve in Pediatric Cancer Survivors with Impaired Cardiopulmonary Fitness. J. Cardiovasc. Magn. Reson. 2020, 22, 64. [Google Scholar] [CrossRef] [PubMed]
- Mijwel, S.; Backman, M.; Bolam, K.A.; Olofsson, E.; Norrbom, J.; Bergh, J.; Sundberg, C.J.; Wengström, Y.; Rundqvist, H. Highly Favorable Physiological Responses to Concurrent Resistance and High-Intensity Interval Training during Chemotherapy: The OptiTrain Breast Cancer Trial. Breast Cancer Res. Treat. 2018, 169, 93–103. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.; Kang, I.; Mack, W.J.; Mortimer, J.; Sattler, F.; Salem, G.; Lu, J.; Dieli-Conwright, C.M. Effects of High Intensity Interval Training on Vascular Endothelial Function and Vascular Wall Thickness in Breast Cancer Patients Receiving Anthracycline-Based Chemotherapy: A Randomized Pilot Study. Breast Cancer Res. Treat. 2019, 177, 477–485. [Google Scholar] [CrossRef]
- Ansund, J.; Mijwel, S.; Bolam, K.A.; Altena, R.; Wengström, Y.; Rullman, E.; Rundqvist, H. High Intensity Exercise during Breast Cancer Chemotherapy—Effects on Long-Term Myocardial Damage and Physical Capacity—Data from the OptiTrain RCT. Cardio-Oncology 2021, 7, 7. [Google Scholar] [CrossRef]
- Kirkham, A.A.; Eves, N.D.; Shave, R.E.; Bland, K.A.; Bovard, J.; Gelmon, K.A.; Virani, S.A.; McKenzie, D.C.; Stöhr, E.J.; Waburton, D.E.R.; et al. The Effect of an Aerobic Exercise Bout 24 h Prior to Each Doxorubicin Treatment for Breast Cancer on Markers of Cardiotoxicity and Treatment Symptoms: A RCT. Breast Cancer Res. Treat. 2018, 167, 719–729. [Google Scholar] [CrossRef]
- Kirkham, A.A.; Lloyd, M.G.; Claydon, V.E.; Gelmon, K.A.; McKenzie, D.C.; Campbell, K.L. A Longitudinal Study of the Association of Clinical Indices of Cardiovascular Autonomic Function with Breast Cancer Treatment and Exercise Training. Oncologist 2019, 24, 273–284. [Google Scholar] [CrossRef]
- Meacham, L.R.; Chow, E.J.; Ness, K.K.; Kamdar, K.Y.; Chen, Y.; Yasui, Y.; Oeffinger, K.C.; Sklar, C.A.; Robison, L.L.; Mertens, A.C. Cardiovascular Risk Factors in Adult Survivors of Pediatric Cancer—A Report from the Childhood Cancer Survivor Study. Cancer Epidemiol. Biomark. Prev. 2010, 19, 170–181. [Google Scholar] [CrossRef]
- Jones, L.W.; Liu, Q.; Armstrong, G.T.; Ness, K.K.; Yasui, Y.; Devine, K.; Tonorezos, E.; Soares-Miranda, L.; Sklar, C.A.; Douglas, P.S.; et al. Exercise and Risk of Major Cardiovascular Events in Adult Survivors of Childhood Hodgkin Lymphoma: A Report from the Childhood Cancer Survivor Study. J. Clin. Oncol. 2014, 32, 3643–3650. [Google Scholar] [CrossRef] [PubMed]
- Peters, M.D.; Godfrey, C.; McInerney, P.; Munn, Z.; Tricco, A.C.; Khalil, H. Chapter 11: Scoping Reviews (2020 Version). In JBI Manual for Evidence Synthesis; Aromataris, E., Munn, Z., Eds.; JBI: Adelaide, Australia, 2020. [Google Scholar]
- Kendall, S.J.; Langley, J.E.; Crooks, B.; Giacomantonio, N.; Heinze-Milne, S.; Johnston, W.J.; Mulvagh, S.L.; Grandy, S.A. The Impact of Exercise on Cardiotoxicity in Pediatric Cancer Survivors: A Scoping Review Protocol. Healthy Popul. J. 2022; in press. [Google Scholar]
- Covidence Systematic Review Software; Veritas Health Innovation: Melbourne, Australia, 2022.
- Järvelä, L.S.; Saraste, M.; Niinikoski, H.; Hannukainen, J.C.; Heinonen, O.J.; Lähteenmäki, P.M.; Arola, M.; Kemppainen, J. Home-Based Exercise Training Improves Left Ventricle Diastolic Function in Survivors of Childhood ALL: A Tissue Doppler and Velocity Vector Imaging Study. Pediatr. Blood Cancer 2016, 63, 1629–1635. [Google Scholar] [CrossRef] [PubMed]
- Järvelä, L.S.; Niinikoski, H.; Heinonen, O.J.; Lähteenmäki, P.M.; Arola, M.; Kemppainen, J. Endothelial Function in Long-Term Survivors of Childhood Acute Lymphoblastic Leukemia: Effects of a Home-Based Exercise Program. Pediatr. Blood Cancer 2013, 60, 1546–1551. [Google Scholar] [CrossRef] [PubMed]
- Järvelä, L.S.; Kemppainen, J.; Niinikoski, H.; Hannukainen, J.C.; Lähteenmäki, P.M.; Kapanen, J.; Arola, M.; Heinonen, O.J. Effects of a Home-Based Exercise Program on Metabolic Risk Factors and Fitness in Long-Term Survivors of Childhood Acute Lymphoblastic Leukemia. Pediatr. Blood Cancer 2012, 59, 155–160. [Google Scholar] [CrossRef] [PubMed]
- Gustafsson, G.; Schmiegelow, K.; Forestier, E.; Clausen, N.; Glomstein, A.; Jonmundsson, G.; Mellander, L.; Mäkipernaa, A.; Nygaard, R.; Saarinen-Pihkala, U.M. Improving Outcome through Two Decades in Childhood ALL in the Nordic Countries: The Impact of High-Dose Methotrexate in the Reduction of CNS Irradiation. Nordic Society of Pediatric Haematology and Oncology (NOPHO). Leukemia 2000, 14, 2267–2275. [Google Scholar] [CrossRef]
- Long, T.M.; Rath, S.R.; Wallman, K.E.; Howie, E.K.; Straker, L.M.; Bullock, A.; Walwyn, T.S.; Gottardo, N.G.; Cole, C.H.; Choong, C.S.; et al. Exercise Training Improves Vascular Function and Secondary Health Measures in Survivors of Pediatric Oncology Related Cerebral Insult. PLoS ONE 2018, 13, e0201449. [Google Scholar] [CrossRef]
- Morales, J.S.; Santana-Sosa, E.; Santos-Lozano, A.; Baño-Rodrigo, A.; Valenzuela, P.L.; Rincón-Castanedo, C.; Fernández-Moreno, D.; González Vicent, M.; Pérez-Somarriba, M.; Madero, L.; et al. Inhospital Exercise Benefits in Childhood Cancer: A Prospective Cohort Study. Scand. J. Med. Sci. Sports 2020, 30, 126–134. [Google Scholar] [CrossRef]
- Sharkey, A.M.; Carey, A.B.; Heise, C.T.; Barber, G. Cardiac Rehabilitation after Cancer Therapy in Children and Young Adults. Am. J. Cardiol. 1993, 71, 1488–1490. [Google Scholar] [CrossRef]
- Smith, W.A.; Ness, K.K.; Joshi, V.; Hudson, M.M.; Robison, L.L.; Green, D.M. Exercise Training in Childhood Cancer Survivors with Subclinical Cardiomyopathy Who Were Treated with Anthracyclines. Pediatr. Blood Cancer 2013, 61, 942–945. [Google Scholar] [CrossRef]
- Patel, N.; Durland, J.; Makaryus, A.N. Physiology, Cardiac Index. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2022. [Google Scholar]
- Heiskanen, J.S.; Ruohonen, S.; Rovio, S.P.; Pahkala, K.; Kytö, V.; Kähönen, M.; Lehtimäki, T.; Viikari, J.S.A.; Juonala, M.; Laitinen, T.; et al. Cardiovascular Risk Factors in Childhood and Left Ventricular Diastolic Function in Adulthood. Pediatrics 2021, 147, e2020016691. [Google Scholar] [CrossRef]
- Unnithan, V.B.; Rowland, T.W.; George, K.; Lord, R.; Oxborough, D. Left Ventricular Function during Exercise in Trained Pre-Adolescent Soccer Players. Scand. J. Med. Sci. Sports 2018, 28, 2330–2338. [Google Scholar] [CrossRef] [PubMed]
- Nystoriak, M.A.; Bhatnagar, A. Cardiovascular Effects and Benefits of Exercise. Front. Cardiovasc. Med. 2018, 5, 135. [Google Scholar] [CrossRef] [PubMed]
- Children’s Oncology Group. Long-Term Follow-Up Guidelines for Survivors of Childhood, Adolescent and Young Adult Cancer; Children’s Oncology Group: Birmingham, UK, 2018. [Google Scholar]
- Barac, A.; Murtagh, G.; Carver, J.R.; Chen, M.H.; Freeman, A.M.; Herrmann, J.; Iliescu, C.; Ky, B.; Mayer, E.L.; Okwuosa, T.M.; et al. Cardiovascular Health of Patients with Cancer and Cancer Survivors. J. Am. Coll. Cardiol. 2015, 65, 2739–2746. [Google Scholar] [CrossRef] [PubMed]
- Cardinale, D.; Colombo, A.; Bacchiani, G.; Tedeschi, I.; Meroni, C.A.; Veglia, F.; Civelli, M.; Lamantia, G.; Colombo, N.; Curigliano, G.; et al. Early Detection of Anthracycline Cardiotoxicity and Improvement with Heart Failure Therapy. Circulation 2015, 131, 1981–1988. [Google Scholar] [CrossRef]
- Zamorano, J.L.; Lancellotti, P.; Rodriguez Muñoz, D.; Aboyans, V.; Asteggiano, R.; Galderisi, M.; Habib, G.; Lenihan, D.J.; Lip, G.Y.H.; Lyon, A.R.; et al. 2016 ESC Position Paper on Cancer Treatments and Cardiovascular Toxicity Developed under the Auspices of the ESC Committee for Practice Guidelines. Eur. J. Heart Fail. 2017, 19, 9–42. [Google Scholar] [CrossRef] [PubMed]
- American College of Sports Medicine. ACSM’s Guidelines for Exercise Testing and Prescription, 10th ed.; Wolters Kluwer: Philadelphia, PA, USA, 2018; ISBN 978-1-4963-3906-5. [Google Scholar]
- Chamorro Viña, C.; Keats, M.R.; Culos-Reed, S.N. POEM for Professionals: Pediatric Oncology Exercise Manual, 1st ed.; Health & Wellness Lab., Faculty of Kinesiology, University of Calgary: Calgary, AB, Canada, 2014; ISBN 978-0-88953-380-6. [Google Scholar]
| Study Identification | Title | Country | Design | Aim | Criteria |
|---|---|---|---|---|---|
| Järvelä 2013 [37] & 2016 [36]. Methodologies as cited in [38]. | Endothelial function in long-term survivors of childhood ALL: Effects of a home-based exercise program [37]; Home-based exercise training improves LV diastolic function in survivors of childhood ALL: A tissue doppler and velocity vector imaging study [36]. | Finland | Case-control study | Assess the effects of a home-based exercise intervention on endothelial structure in survivors of childhood ALL [37]; Determine the effects of an exercise program on anthracycline-induced cardiotoxicity as assessed by tissue doppler imaging and velocity vector imaging in long-term childhood ALL survivors [36]. | Age < 16 years at diagnosis, currently age 16–30 years, first continuous remission without hematopoietic bone marrow transplantation, diagnosed in 1986 or later, treated according to the Nordic regimen [39], and no down syndrome diagnosis. |
| Long 2018 [40] | Exercise training improves vascular function and secondary health measures in survivors of pediatric oncology related cerebral insult. | Australia | Cohort study | Assess the feasibility and effectiveness of a 24-week exercise intervention on cardiovascular health in childhood cancer survivors. | >5-year survivor of pediatric cancer-related cerebral insult, currently aged 15–23, not pregnant and without a current cardiovascular disease diagnosis. |
| Morales 2020 [41] | Inhospital exercise benefits in childhood cancer: A prospective cohort study. | Spain | Cohort study | Assess the effects of aerobic and resistance training in children with leukemia receiving neoadjuvant or intensive chemotherapy. | Currently aged 4–18 years, received a new cancer diagnosis, diagnosed, treated, and followed at the Hospital Infantil Universitario Nino Jesus, and not currently participating in any other interventional trials. |
| Sharkey 1993 [42] | Cardiac rehabilitation after cancer therapy in children and young adults. | United States | Case series | Assess childhood cancer survivors using exercise testing before and after a 12-week aerobic exercise program. | Received >100 mg/m2 of anthracyclines, post-pubertal, ≥1-year post-treatment, and no residual malignancies. |
| Smith 2013 [43] | Exercise training in childhood cancer survivors with subclinical cardiomyopathy who were treated with anthracyclines. | United States | Case series | Assess the effects of a 12-week exercise program on anthracycline-treated childhood cancer survivors with subclinical cardiomyopathy. | 18 years of age, ≥10 years post-diagnosis of childhood cancer, treated with doxorubicin and/or daunorubicin, sedentary (<150 min of moderate-intensity physical activity per week), LVEF ≥ 40 and ≤55%, and not receiving cardiomyopathy treatment or received radiation therapy. |
| Study ID | Group | Participants (Number) | Anthracyclines Dosage (mg/m2) | Radiation Field (Gy) | Age (Years) | Time Since Diagnosis (Years) | Cancer Type |
|---|---|---|---|---|---|---|---|
| Järvelä 2013 & 2016 [36,37] | N/A | M=10 F=11 | n = 21 (Med = 240, range: 120–370) | n = 5 (unspecified dosage) | Med = 21.1 (range: 16.0–28.4) | Med = 15.9 (range: 11.3–21.4) | ALL |
| Long 2018 [40] | N/A | M=6 F=7 | n = 4 (unspecified dosage) | n = 8 (unspecified dosage) | Med = 19 (range: 16–23) | Med = 15 (range: 7–22) | Brain = 9, ALL = 3, Other = 1 |
| Morales 2020 [41] | Controls | M=63 F=38 | n = 41 (unspecified dosage) | n = 30, (range: 1–≥50) | mean = 11 (range: 4–18) | On treatment | 15 various types |
| Exercise | M=61 F=27 | n = 27 (unspecified dosage | n = 27 (range: 1–≥50) | mean = 11 (range: 4–17) | |||
| Sharkey 1993 [42] | N/A | M=5 F=5 | n = 10 (mean = 349 ± 69 | n = 9 (range: 18–55) | mean = 19+/−3 | mean = 11 (range: 4–18) | 5 various types |
| Smith 2013 [43] | N/A | M=3 F=2 | n = 4 (range: 5, 298) | n = 0 | Range: 33–41 | Range: 25–30 | Osteosarcoma = 4, Ewing sarcoma = 1 |
| Study ID | Mode | Frequency (Sessions/Week) | Intensity | Time (min) | Type | Location | Duration (Weeks) | Instructor |
|---|---|---|---|---|---|---|---|---|
| Järvelä 2013 & 2016 [36,37]. Exercise protocol as cited in [38] | Resistance | 3–4 | 3 sets, as many repetitions as possible, no rest stated. | Not stated | Eight exercises to strengthen the gluteal, lower limb, shoulders, upper limb, abdominal, and back muscles. | Home | 12 | Experts in sports science |
| Aerobic | At least 3 | Not specified | 30 | Participant choice (i.e., walking or jogging). | ||||
| Long 2018 [40] | Resistance | 2 to 3 | 3 sets, 10 repetitions, 60–70% 3-RM, with 3 to 5 min of rest between exercises. | 75–80 | Circuit including 6 to 10 exercises targeting the chest, back, shoulders, arms, and legs. | Not stated | 24 | Exercise physiologist |
| Aerobic | 2 to 3 | 40–60% HRmaxwith individualized progressive increase. | 10–15 | Three sets of 4 consecutive sprint-rest bouts, with 3 to 5 min of rest between each set. Rowing ergometer, stationary bike, or arm ergometer. | ||||
| Morales 2020 [41] | Resistance | 2 to 3 | 1 to 3 sets of 6–15 repetitions, 5% to 10% load increases as needed with 1 min rest between sets. | 30 | Shoulder, chest and leg press, side-arm rowing extension and flexion, knee extension and flexion and abdominal, lumbar and shoulder adduction. | Hospital | Med duration 22 weeks (IQR: 14, 28) | Exercise physiologist |
| Aerobic | 2 to 3 | 65–80% HRreserve with individualized progressive increase. | 30–40 | Ten minutes each of cycle ergometer leg pedalling, treadmill running, or arm cranking in those missing a lower limb. Ten minutes of aerobic games. | ||||
| Sharkey 1993 [42] | Aerobic | Two sessions for weeks 1–6 and 3 sessions for weeks 7–12. | 60% to 80% HRmax progressive increase. | 45–60 | Not stated | Hospital and home | 12 | Not stated |
| Smith 2013 [43] | Resistance | 3–5 | 1 set of 12–15 repetitions on 8 to 10 exercises. | Not stated | Not stated | Home | 12 | Exercise physiologist |
| Aerobic | 2–3 | 40–70%HRreserve. | 20–45 | Not stated |
| Study ID | LVEF | Valve Velocity | Strain | FS | CI | SV |
|---|---|---|---|---|---|---|
| Järvelä 2013 & 2016 [36,37] |  |  ↑ ↑ |  ↑ ↑ |  | N/A | N/A |
| Long 2020 [40] | N/A | N/A | N/A | N/A | N/A | N/A |
| Morales 2020 [41] |  ↑ ↑ | N/A | N/A |  ↑ ↑ | N/A | N/A |
| Sharkey 1993 [42] | N/A | N/A | N/A | N/A |  |  |
| Smith 2013 [43] |  ↑ ↑ | N/A | N/A | N/A | N/A | N/A |
 , insignificant change; ↑, improvement
, insignificant change; ↑, improvement  , significant change; LVEF, left ventricle ejection fraction; FS, fractional shortening; CI, cardiac index; SV, stroke volume; N/A, not applicable.
, significant change; LVEF, left ventricle ejection fraction; FS, fractional shortening; CI, cardiac index; SV, stroke volume; N/A, not applicable.| Study ID | IMT | FMD | Oxygen Pulse |
|---|---|---|---|
| Järvelä 2013 & 2016 [36,37] |  ↓ ↓ |  ↑ ↑ | N/A |
| Long 2020 [40] | N/A |  ↑ ↑ | N/A |
| Morales 2020 [41] | N/A | N/A | N/A |
| Sharkey 1993 [42] | N/A | N/A | N/A |
| Smith 2013 [43] | N/A | N/A |  ↑ ↑ |
 , significant change IMT, intima-media thickness; FMD, flow-mediated dilation; NA, not applicable; ↑, increased; ↓, decreased.
, significant change IMT, intima-media thickness; FMD, flow-mediated dilation; NA, not applicable; ↑, increased; ↓, decreased.Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kendall, S.J.; Langley, J.E.; Aghdam, M.; Crooks, B.N.; Giacomantonio, N.; Heinze-Milne, S.; Johnston, W.J.; Keats, M.R.; Mulvagh, S.L.; Grandy, S.A. The Impact of Exercise on Cardiotoxicity in Pediatric and Adolescent Cancer Survivors: A Scoping Review. Curr. Oncol. 2022, 29, 6350-6363. https://doi.org/10.3390/curroncol29090500
Kendall SJ, Langley JE, Aghdam M, Crooks BN, Giacomantonio N, Heinze-Milne S, Johnston WJ, Keats MR, Mulvagh SL, Grandy SA. The Impact of Exercise on Cardiotoxicity in Pediatric and Adolescent Cancer Survivors: A Scoping Review. Current Oncology. 2022; 29(9):6350-6363. https://doi.org/10.3390/curroncol29090500
Chicago/Turabian StyleKendall, Stephanie J., Jodi E. Langley, Mohsen Aghdam, Bruce N. Crooks, Nicholas Giacomantonio, Stefan Heinze-Milne, Will J. Johnston, Melanie R. Keats, Sharon L. Mulvagh, and Scott A. Grandy. 2022. "The Impact of Exercise on Cardiotoxicity in Pediatric and Adolescent Cancer Survivors: A Scoping Review" Current Oncology 29, no. 9: 6350-6363. https://doi.org/10.3390/curroncol29090500
APA StyleKendall, S. J., Langley, J. E., Aghdam, M., Crooks, B. N., Giacomantonio, N., Heinze-Milne, S., Johnston, W. J., Keats, M. R., Mulvagh, S. L., & Grandy, S. A. (2022). The Impact of Exercise on Cardiotoxicity in Pediatric and Adolescent Cancer Survivors: A Scoping Review. Current Oncology, 29(9), 6350-6363. https://doi.org/10.3390/curroncol29090500






