Consolidation Chemotherapy Rather than Induction Chemotherapy Can Prolong the Survival Rate of Inoperable Esophageal Cancer Patients Who Received Concurrent Chemoradiotherapy
Abstract
1. Introduction
2. Materials and Methods
2.1. Pretreatment Staging
2.2. Chemotherapy
2.3. Radiation Therapy
2.4. Endpoints and Follow-Up
2.5. Statistics
3. Results
3.1. Patients and Tumor Characteristics
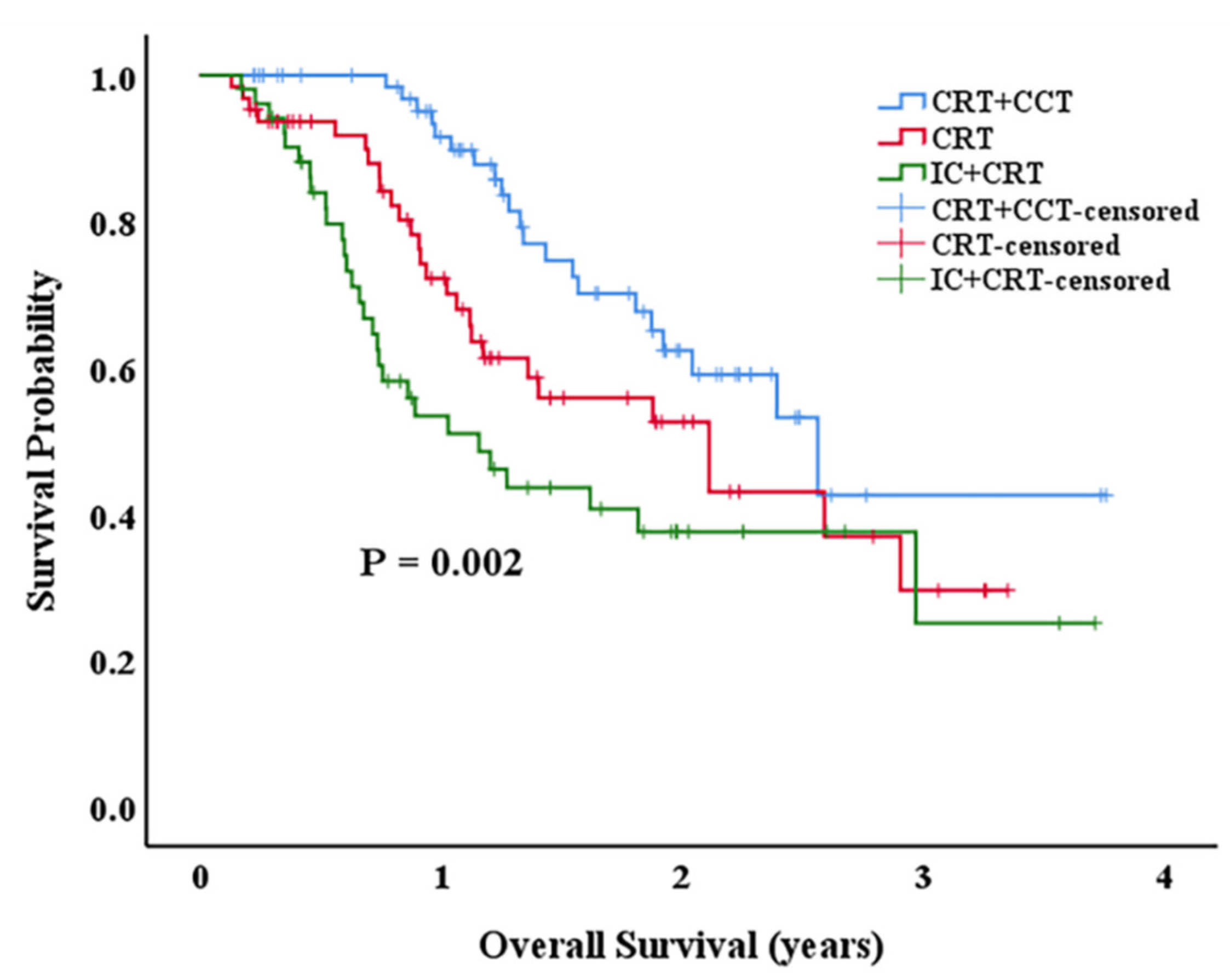
3.2. Survival Outcomes
3.3. Toxicity of Treatment
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Yennurajalingam, S.; Kang, J.H.; Cheng, H.Y.; Chisholm, G.B.; Kwon, J.H.; Palla, S.L.; Bruera, E. Characteristics of advanced cancer patients with cancer-related fatigue enrolled in clinical trials and patients referred to outpatient palliative care clinics. J. Pain Symptom Manag. 2013, 45, 534–541. [Google Scholar] [CrossRef]
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef] [PubMed]
- Short, M.W.; Burgers, K.G.; Fry, V.T. Esophageal Cancer. Am. Fam. Physician 2017, 95, 22–28. [Google Scholar] [PubMed]
- van Hagen, P.; Hulshof, M.C.; van Lanschot, J.J.; Steyerberg, E.W.; van Berge Henegouwen, M.I.; Wijnhoven, B.P.; Richel, D.J.; Nieuwenhuijzen, G.A.; Hospers, G.A.; Bonenkamp, J.J.; et al. Preoperative chemoradiotherapy for esophageal or junctional cancer. N. Engl. J. Med. 2012, 366, 2074–2084. [Google Scholar] [CrossRef] [PubMed]
- Shapiro, J.; van Lanschot, J.J.B.; Hulshof, M.; van Hagen, P.; van Berge Henegouwen, M.I.; Wijnhoven, B.P.L.; van Laarhoven, H.W.M.; Nieuwenhuijzen, G.A.P.; Hospers, G.A.P.; Bonenkamp, J.J.; et al. Neoadjuvant chemoradiotherapy plus surgery versus surgery alone for oesophageal or junctional cancer (CROSS): Long-term results of a randomised controlled trial. Lancet. Oncol. 2015, 16, 1090–1098. [Google Scholar] [CrossRef]
- Eyck, B.M.; van Lanschot, J.J.B.; Hulshof, M.; van der Wilk, B.J.; Shapiro, J.; van Hagen, P.; van Berge Henegouwen, M.I.; Wijnhoven, B.P.L.; van Laarhoven, H.W.M.; Nieuwenhuijzen, G.A.P.; et al. Ten-Year Outcome of Neoadjuvant Chemoradiotherapy Plus Surgery for Esophageal Cancer: The Randomized Controlled CROSS Trial. J. Clin. Oncol. 2021, 39, 1995–2004. [Google Scholar] [CrossRef]
- Sasaki, Y.; Kato, K. Chemoradiotherapy for esophageal squamous cell cancer. Jpn. J. Clin. Oncol. 2016, 46, 805–810. [Google Scholar] [CrossRef]
- Pennathur, A.; Gibson, M.K.; Jobe, B.A.; Luketich, J.D. Oesophageal carcinoma. Lancet 2013, 381, 400–412. [Google Scholar] [CrossRef]
- Gwynne, S.; Hurt, C.; Evans, M.; Holden, C.; Vout, L.; Crosby, T. Definitive chemoradiation for oesophageal cancer--a standard of care in patients with non-metastatic oesophageal cancer. Clin. Oncol. (R. Coll. Radiol.) 2011, 23, 182–188. [Google Scholar] [CrossRef]
- Putora, P.M.; Bedenne, L.; Budach, W.; Eisterer, W.; Van Der Gaast, A.; Jäger, R.; Van Lanschot, J.J.; Mariette, C.; Schnider, A.; Stahl, M.; et al. Oesophageal cancer: Exploring controversies overview of experts’ opinions of Austria, Germany, France, Netherlands and Switzerland. Radiat Oncol. 2015, 10, 116. [Google Scholar] [CrossRef]
- Hui, E.P.; Ma, B.B.; Leung, S.F.; King, A.D.; Mo, F.; Kam, M.K.; Yu, B.K.; Chiu, S.K.; Kwan, W.H.; Ho, R.; et al. Randomized phase II trial of concurrent cisplatin-radiotherapy with or without neoadjuvant docetaxel and cisplatin in advanced nasopharyngeal carcinoma. J. Clin. Oncol. 2009, 27, 242–249. [Google Scholar] [CrossRef]
- Luo, L.L.; Xi, M.; Yang, Y.D.; Li, Q.Q.; Zhao, L.; Zhang, P.; Liu, S.L.; Liu, M.Z. Comparative Outcomes of Induction Chemotherapy Followed By Definitive Chemoradiotherapy versus Chemoradiotherapy Alone In Esophageal Squamous Cell Carcinoma. J. Cancer 2017, 8, 3441–3447. [Google Scholar] [CrossRef] [PubMed]
- Satake, H.; Tahara, M.; Mochizuki, S.; Kato, K.; Hara, H.; Yokota, T.; Kiyota, N.; Kii, T.; Chin, K.; Zenda, S.; et al. A prospective, multicenter phase I/II study of induction chemotherapy with docetaxel, cisplatin and fluorouracil (DCF) followed by chemoradiotherapy in patients with unresectable locally advanced esophageal carcinoma. Cancer Chemother. Pharm. 2016, 78, 91–99. [Google Scholar] [CrossRef]
- Chen, M.Q.; Lin, Q.L.; Chen, Y.G.; Guo, J.H.; Xu, B.H.; Tian, Y. Neoadjuvant chemotherapy may not benefit esophageal squamous cell carcinoma patients treated with definitive chemoradiotherapy. J. Chin. Med. Assoc. 2017, 80, 636–643. [Google Scholar] [CrossRef] [PubMed]
- Yoon, D.H.; Jang, G.; Kim, J.H.; Kim, Y.H.; Kim, J.Y.; Kim, H.R.; Jung, H.Y.; Lee, G.H.; Song, H.Y.; Cho, K.J.; et al. Randomized phase 2 trial of S1 and oxaliplatin-based chemoradiotherapy with or without induction chemotherapy for esophageal cancer. Int. J. Radiat Oncol. Biol. Phys. 2015, 91, 489–496. [Google Scholar] [CrossRef] [PubMed]
- Grossi, F.; Aita, M.; Follador, A.; Defferrari, C.; Brianti, A.; Sinaccio, G.; Belvedere, O. Sequential, alternating, and maintenance/consolidation chemotherapy in advanced non-small cell lung cancer: A review of the literature. Oncologist 2007, 12, 451–464. [Google Scholar] [CrossRef]
- Mabuchi, S.; Isohashi, F.; Okazawa, M.; Kitada, F.; Maruoka, S.; Ogawa, K.; Kimura, T. Chemoradiotherapy followed by consolidation chemotherapy involving paclitaxel and carboplatin and in FIGO stage IIIB/IVA cervical cancer patients. J. Gynecol. Oncol. 2017, 28, e15. [Google Scholar] [CrossRef]
- Liu, L.; Bi, N.; Ji, Z.; Li, J.; Wang, J.; Wang, X.; Hui, Z.; Lv, J.; Liang, J.; Zhou, Z.; et al. Consolidation chemotherapy may improve survival for patients with locally advanced non-small-cell lung cancer receiving concurrent chemoradiotherapy--retrospective analysis of 203 cases. BMC Cancer 2015, 15, 715. [Google Scholar] [CrossRef]
- Wang, X.; Ding, X.; Kong, D.; Zhang, L.; Guo, Y.; Ren, J.; Hu, X.; Yang, J.; Gao, S. The effect of consolidation chemotherapy after concurrent chemoradiotherapy on the survival of patients with locally advanced non-small cell lung cancer: A meta-analysis. Int. J. Clin. Oncol. 2017, 22, 229–236. [Google Scholar] [CrossRef]
- Xia, X.; Liu, Z.; Qin, Q.; Di, X.; Zhang, Z.; Sun, X.; Ge, X. Long-Term Survival in Nonsurgical Esophageal Cancer Patients Who Received Consolidation Chemotherapy Compared With Patients Who Received Concurrent Chemoradiotherapy Alone: A Systematic Review and Meta-Analysis. Front. Oncol. 2020, 10, 604657. [Google Scholar] [CrossRef]
- Wang, J.; Xiao, L.; Wang, S.; Pang, Q.; Wang, J. Addition of Induction or Consolidation Chemotherapy in Definitive Concurrent Chemoradiotherapy Versus Concurrent Chemoradiotherapy Alone for Patients With Unresectable Esophageal Cancer: A Systematic Review and Meta-Analysis. Front. Oncol. 2021, 11, 665231. [Google Scholar] [CrossRef]
- Liu, S.; Luo, L.; Zhao, L.; Zhu, Y.; Liu, H.; Li, Q.; Cai, L.; Hu, Y.; Qiu, B.; Zhang, L.; et al. Induction chemotherapy followed by definitive chemoradiotherapy versus chemoradiotherapy alone in esophageal squamous cell carcinoma: A randomized phase II trial. Nat. Commun. 2021, 12, 4014. [Google Scholar] [CrossRef] [PubMed]
- Harada, G.; Bonadio, R.; de Araújo, F.C.C.; Victor, C.R.; Sallum, R.A.A.; Junior, U.R.; Cecconello, I.; Takeda, F.R.; de Castria, T.B. Induction Chemotherapy for Locally Advanced Esophageal Cancer. J. Gastrointest. Cancer 2020, 51, 498–505. [Google Scholar] [CrossRef] [PubMed]
- Minsky, B.D.; Neuberg, D.; Kelsen, D.P.; Pisansky, T.M.; Ginsberg, R.; Benson, A., 3rd. Neoadjuvant chemotherapy plus concurrent chemotherapy and high-dose radiation for squamous cell carcinoma of the esophagus: A preliminary analysis of the phase II intergroup trial 0122. J. Clin. Oncol. 1996, 14, 149–155. [Google Scholar] [CrossRef]
- Yoon, H.H.; Ou, F.S.; Soori, G.S.; Shi, Q.; Wigle, D.A.; Sticca, R.P.; Miller, R.C.; Leenstra, J.L.; Peller, P.J.; Ginos, B.; et al. Induction versus no induction chemotherapy before neoadjuvant chemoradiotherapy and surgery in oesophageal adenocarcinoma: A multicentre randomised phase II trial (NCCTG N0849 [Alliance]). Eur. J. Cancer 2021, 150, 214–223. [Google Scholar] [CrossRef]
- Steffen, T.; Dietrich, D.; Schnider, A.; Kettelhack, C.; Huber, O.; Marti, W.R.; Furrer, M.; Gloor, B.; Schiesser, M.; Thierstein, S.; et al. Recurrence Patterns and Long-term Results After Induction Chemotherapy, Chemoradiotherapy, and Curative Surgery in Patients With Locally Advanced Esophageal Cancer. Ann. Surg. 2019, 269, 83–87. [Google Scholar] [CrossRef] [PubMed]
- Terada, M.; Hara, H.; Daiko, H.; Mizusawa, J.; Kadota, T.; Hori, K.; Ogawa, H.; Ogata, T.; Sakanaka, K.; Sakamoto, T.; et al. Phase III study of tri-modality combination therapy with induction docetaxel plus cisplatin and 5-fluorouracil versus definitive chemoradiotherapy for locally advanced unresectable squamous-cell carcinoma of the thoracic esophagus (JCOG1510: TRIANgLE). Jpn. J. Clin. Oncol. 2019, 49, 1055–1060. [Google Scholar] [CrossRef]
- Wu, S.X.; Li, X.Y.; Xu, H.Y.; Xu, Q.N.; Luo, H.S.; Du, Z.S.; Huang, H.C.; Wu, Z.Y. Effect of consolidation chemotherapy following definitive chemoradiotherapy in patients with esophageal squamous cell cancer. Sci. Rep. 2017, 7, 16870. [Google Scholar] [CrossRef]
- Koh, H.K.; Park, Y.; Koo, T.; Park, H.J.; Lee, M.Y.; Chang, A.R.; Hong, S.; Bae, H. Adjuvant Chemotherapy and Dose Escalation in Definitive Concurrent Chemoradiotherapy for Esophageal Squamous Cell Carcinoma. Anticancer. Res. 2020, 40, 1771–1778. [Google Scholar] [CrossRef]
- Zhang, A.D.; Su, X.H.; Shi, G.F.; Han, C.; Wang, L.; Liu, H.; Zhang, J.; Zhang, R.H. Survival Comparision of Three-dimensional Radiotherapy Alone vs. Chemoradiotherapy for Esophageal Squamous Cell Carcinoma. Arch. Med. Res. 2020, 51, 419–428. [Google Scholar] [CrossRef]
- Chen, M.; Shen, M.; Lin, Y.; Liu, P.; Liu, X.; Li, X.; Li, A.; Yang, R.; Ni, W.; Zhou, X.; et al. Adjuvant chemotherapy does not benefit patients with esophageal squamous cell carcinoma treated with definitive chemoradiotherapy. Radiat Oncol. 2018, 13, 150. [Google Scholar] [CrossRef] [PubMed]
- ChiCtr. A Prospective, Open-Label, Multicenter, Randomized and Controlled PHASE III Trial to Compare Radical Concurrent Chemoradiotherapy alone with Radical Concurrent Chemoradiotherapy followed by Consolidation Chemotherapy for the Locally Advanced Esophageal Squamous Cell Carcinoma. 2018. Available online: https://trialsearch.who.int/Trial2.aspx?TrialID=ChiCTR1800017646 (accessed on 31 March 2019).
- Xiang, M.; Liu, B.; Zhang, G.; Gong, H.; Han, D.; Ma, C. Induction Chemotherapy Followed by Chemoradiotherapy With or Without Consolidation Chemotherapy Versus Chemoradiotherapy Followed by Consolidation Chemotherapy for Esophageal Squamous Cell Carcinoma. Front. Oncol. 2022, 12, 813021. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Zhou, L.; Yang, Y.; Yang, L.; Chen, L. Clinical Effect of Radiotherapy Combined with Chemotherapy for Non-Surgical Treatment of the Esophageal Squamous Cell Carcinoma. Med. Sci. Monit. 2018, 24, 4183–4191. [Google Scholar] [CrossRef] [PubMed]
- Xia, Y.; Li, Y.H.; Chen, Y.; Zhang, J.H.; Liu, Q.; Deng, J.Y.; Ai, T.S.; Zhu, H.T.; Fan, J.H.; Badakhshi, H.; et al. A phase II study of concurrent chemoradiotherapy combined with a weekly paclitaxel and 5-fluorouracil regimen to treat patients with advanced oesophageal carcinoma. Radiat Oncol. 2017, 12, 47. [Google Scholar] [CrossRef][Green Version]
| Project | Patient (Range, %) | P | |||||
|---|---|---|---|---|---|---|---|
| IC + CRT (n = 52) | CRT (n = 64) | CRT + CCT (n = 70) | |||||
| Age | 0.41 | ||||||
| Median | 66 (49–80) | 76 (53–88) | 65 (55–85) | ||||
| <60 | 9 | 17.31% | 6 | 9.38% | 11 | 15.71% | |
| ≥60 | 43 | 82.69% | 58 | 90.63% | 59 | 84.29% | |
| Gender | 0.472 | ||||||
| Female | 16 | 30.77% | 18 | 28.13% | 15 | 21.43% | |
| Male | 36 | 69.23% | 46 | 71.88% | 55 | 78.57% | |
| Smoking | 0.97 | ||||||
| Yes | 28 | 53.85% | 33 | 51.56% | 37 | 52.86% | |
| No | 24 | 46.15% | 31 | 48.44% | 33 | 47.14% | |
| Alcohol status | 0.942 | ||||||
| Yes | 26 | 50.00% | 31 | 48.44% | 36 | 51.43% | |
| No | 26 | 50.00% | 33 | 51.56% | 34 | 48.57% | |
| ECOG | 0.386 | ||||||
| 0 | 18 | 34.62% | 19 | 29.69% | 24 | 34.29% | |
| 1 | 32 | 61.54% | 38 | 59.38% | 36 | 51.43% | |
| 2 | 2 | 3.85% | 7 | 10.94% | 10 | 14.29% | |
| Tumor diameter | 0.661 | ||||||
| <5cm | 12 | 23.08% | 16 | 25.00% | 21 | 30.00% | |
| ≥5cm | 40 | 76.92% | 48 | 75.00% | 49 | 70.00% | |
| Location | 0.098 | ||||||
| Cervical | 1 | 1.92% | 3 | 4.69% | 3 | 4.29% | |
| Upper | 14 | 26.92% | 15 | 23.44% | 32 | 45.71% | |
| Middle | 15 | 28.85% | 15 | 23.44% | 14 | 20.00% | |
| Lower | 21 | 40.38% | 28 | 43.75% | 21 | 30.00% | |
| Two pieces | 1 | 1.92% | 3 | 4.69% | 0 | 0.00% | |
| T-staging | 0.156 | ||||||
| T1 | 0 | 0.00% | 1 | 1.56% | 1 | 1.43% | |
| T2 | 5 | 9.62% | 3 | 4.69% | 13 | 18.57% | |
| T3 | 21 | 40.38% | 32 | 50.00% | 30 | 42.86% | |
| T4 | 26 | 50.00% | 28 | 43.75% | 26 | 37.14% | |
| N-staging | 0.609 | ||||||
| N0 | 19 | 36.54% | 33 | 51.56% | 32 | 45.71% | |
| N1 | 24 | 46.15% | 23 | 35.94% | 26 | 37.14% | |
| N2 | 8 | 15.38% | 5 | 7.81% | 9 | 12.86% | |
| N3 | 1 | 1.92% | 3 | 4.69% | 3 | 4.29% | |
| Clinical staging | 0.554 | ||||||
| Ⅰ | 0 | 0.00% | 1 | 1.56% | 1 | 1.43% | |
| II | 13 | 25.00% | 22 | 34.38% | 26 | 37.14% | |
| III | 13 | 25.00% | 11 | 17.19% | 17 | 24.29% | |
| IVA | 26 | 50.00% | 30 | 46.88% | 26 | 37.14% | |
| Radiation dose | 0.074 | ||||||
| ≤60 Gy | 49 | 94.23% | 64 | 100.00% | 65 | 92.86% | |
| >60 Gy | 3 | 5.77% | 0 | 0.00% | 5 | 7.14% | |
| Toxicity | IC + CRT (n = 52) | CRT (n = 64) | CRT + CCT (n = 70) | p Value | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Grade 0–1 | Grade 2 | Grade 3 | Grade 4 | Grade 0–1 | Grade 2 | Grade 3 | Grade 4 | Grade 0–1 | Grade 2 | Grade 3 | Grade 4 | ||
| leukopenia | 26 | 12 | 12 | 0 | 23 | 9 | 9 | 0 | 34 | 13 | 14 | 1 | 0.997 |
| thrombocytopenia | 28 | 8 | 14 | 0 | 24 | 9 | 8 | 0 | 36 | 14 | 12 | 0 | 0.769 |
| anemia | 28 | 22 | 0 | 0 | 24 | 16 | 1 | 0 | 36 | 24 | 1 | 0 | 0.913 |
| ALT/AST | 6 | 2 | 1 | 1 | 9 | 2 | 1 | 0 | 10 | 3 | 2 | 0 | 0.957 |
| nausea | 37 | 9 | 4 | 0 | 28 | 6 | 6 | 1 | 17 | 10 | 9 | 1 | 0.091 |
| vomiting | 23 | 20 | 2 | 1 | 22 | 18 | 1 | 0 | 25 | 27 | 2 | 0 | 0.936 |
| radiation esophagitis | 27 | 23 | 1 | 0 | 23 | 16 | 1 | 0 | 28 | 25 | 1 | 1 | 0.98 |
| radiation pneumonia | 34 | 5 | 3 | 0 | 32 | 3 | 3 | 0 | 49 | 6 | 5 | 0 | 0.995 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xia, X.; Wu, M.; Gao, Q.; Sun, X.; Ge, X. Consolidation Chemotherapy Rather than Induction Chemotherapy Can Prolong the Survival Rate of Inoperable Esophageal Cancer Patients Who Received Concurrent Chemoradiotherapy. Curr. Oncol. 2022, 29, 6342-6349. https://doi.org/10.3390/curroncol29090499
Xia X, Wu M, Gao Q, Sun X, Ge X. Consolidation Chemotherapy Rather than Induction Chemotherapy Can Prolong the Survival Rate of Inoperable Esophageal Cancer Patients Who Received Concurrent Chemoradiotherapy. Current Oncology. 2022; 29(9):6342-6349. https://doi.org/10.3390/curroncol29090499
Chicago/Turabian StyleXia, Xiaojie, Mengxing Wu, Qing Gao, Xinchen Sun, and Xiaolin Ge. 2022. "Consolidation Chemotherapy Rather than Induction Chemotherapy Can Prolong the Survival Rate of Inoperable Esophageal Cancer Patients Who Received Concurrent Chemoradiotherapy" Current Oncology 29, no. 9: 6342-6349. https://doi.org/10.3390/curroncol29090499
APA StyleXia, X., Wu, M., Gao, Q., Sun, X., & Ge, X. (2022). Consolidation Chemotherapy Rather than Induction Chemotherapy Can Prolong the Survival Rate of Inoperable Esophageal Cancer Patients Who Received Concurrent Chemoradiotherapy. Current Oncology, 29(9), 6342-6349. https://doi.org/10.3390/curroncol29090499




