Towards Mobile Gait Analysis: Concurrent Validity and Test-Retest Reliability of an Inertial Measurement System for the Assessment of Spatio-Temporal Gait Parameters
Abstract
:1. Introduction
2. Materials and Methods
2.1. Subjects
2.2. Study Protocol
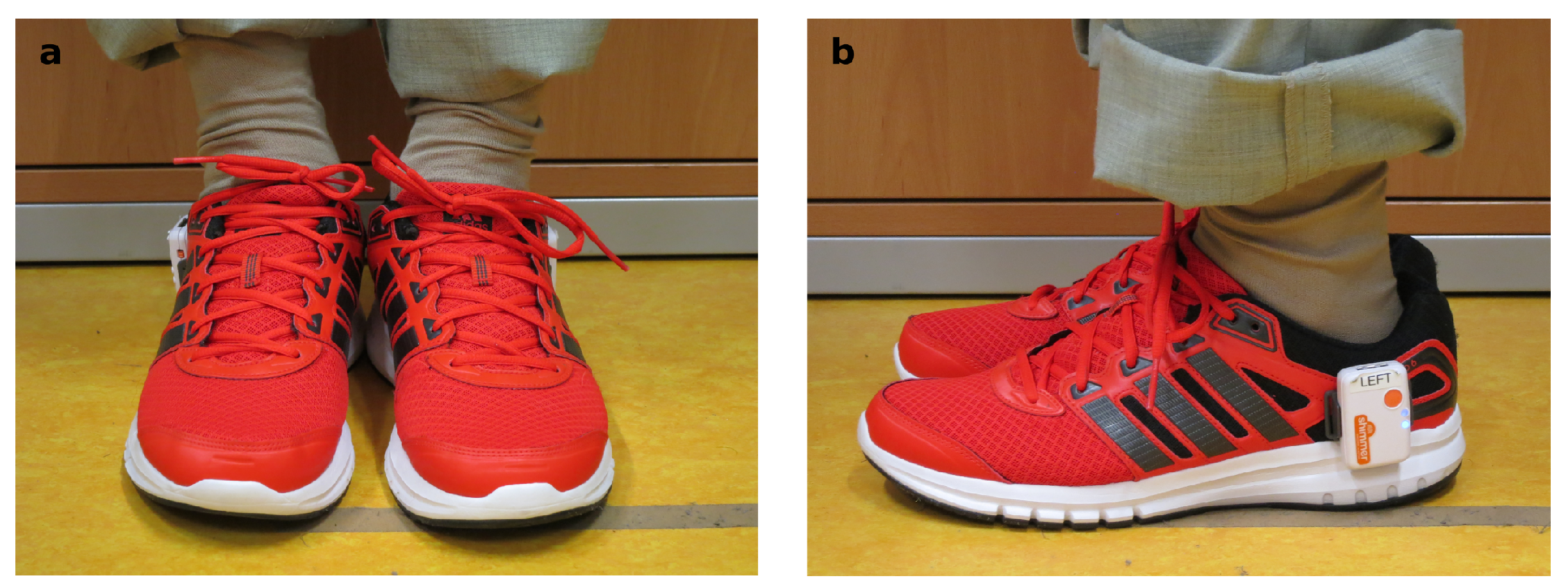
2.3. Measurement Setup
2.4. Sensor-Based Gait Analysis
2.5. Camera-Based Gait Analysis
2.6. Statistical Analysis
3. Results
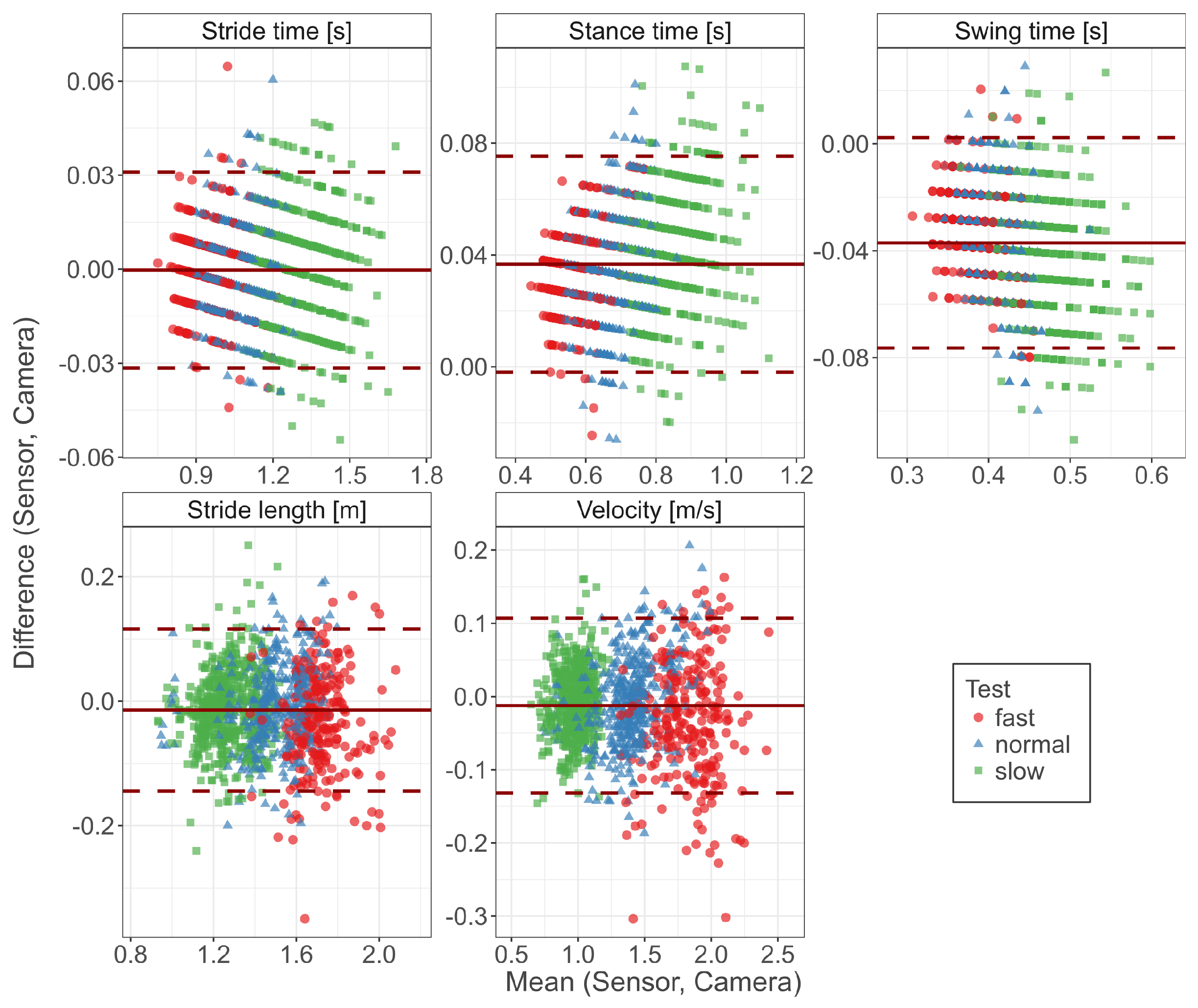
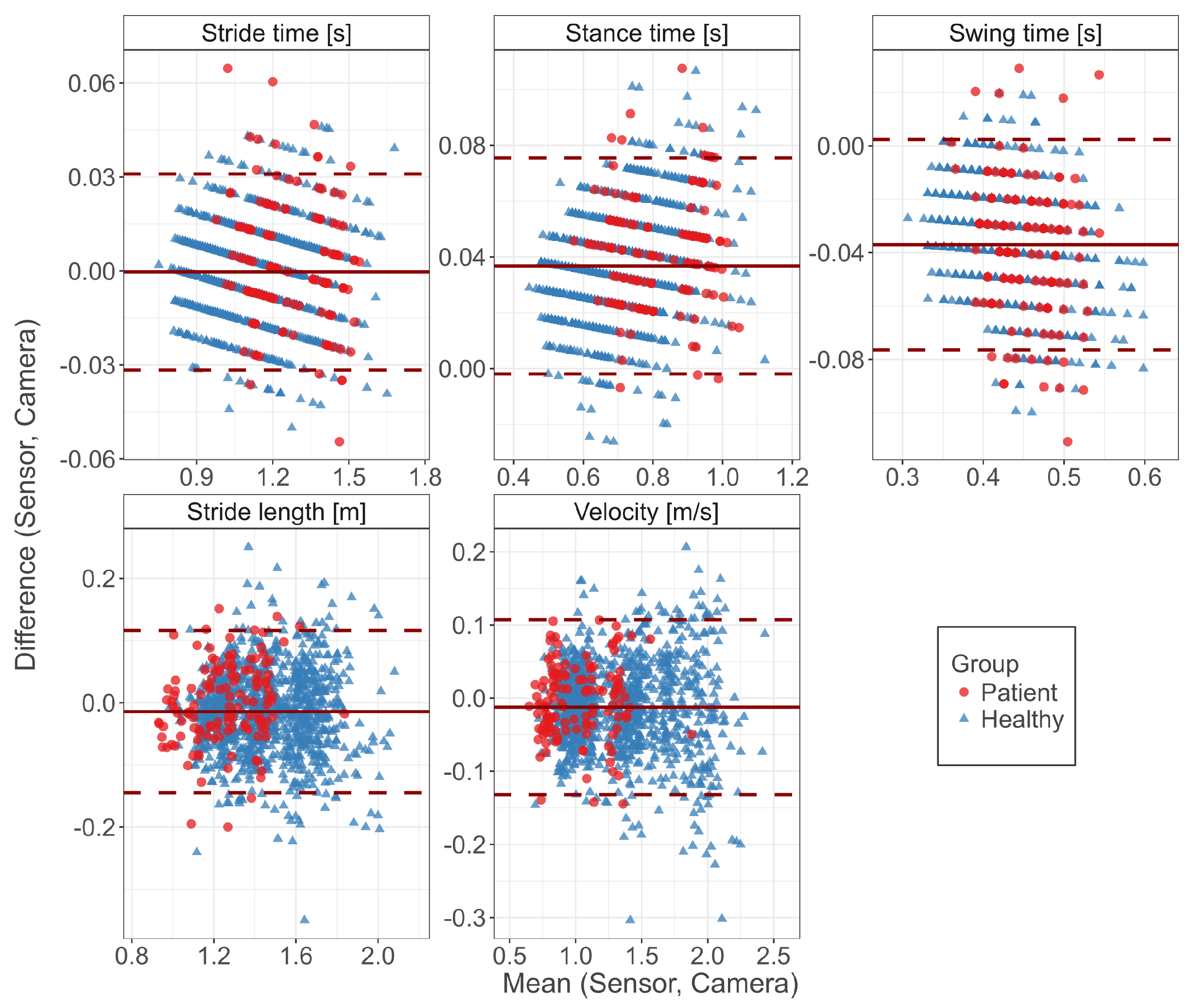
3.1. Concurrent Validity
3.2. Retest Reliability
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Pringsheim, T.; Jette, N.; Frolkis, A.; Steeves, T.D.L. The prevalence of Parkinson’s disease: A systematic review and meta-analysis. Mov. Disord. 2014, 29, 1583–1590. [Google Scholar] [CrossRef] [PubMed]
- Jankovic, J. Parkinson’s disease: Clinical features and diagnosis. J. Neurol. Neurosurg. Psychiatry 2008, 79, 368–376. [Google Scholar] [CrossRef] [PubMed]
- Movement Disorder Society Task Force on Rating Scales for Parkinson’s Disease. The Unified Parkinson’s Disease Rating Scale (UPDRS): Status and recommendations. Mov. Disord. 2003, 18, 738–750. [Google Scholar]
- Krebs, D.E.; Edelstein, J.E.; Fishman, S. Reliability of observational kinematic gait analysis. Phys. Ther. 1985, 65, 1027–1033. [Google Scholar] [CrossRef] [PubMed]
- Coutts, F. Gait analysis in the therapeutic environment. Man. Ther. 1999, 4, 2–10. [Google Scholar] [CrossRef]
- Chen, S.; Lach, J.; Lo, B.; Yang, G.Z. Toward Pervasive Gait Analysis with Wearable Sensors: A Systematic Review. IEEE J. Biomed. Health Inform. 2016, 20, 1521–1537. [Google Scholar] [CrossRef] [PubMed]
- Robles-García, V.; Corral-Bergantiños, Y.; Espinosa, N.; Jácome, M.A.; García-Sancho, C.; Cudeiro, J.; Arias, P. Spatiotemporal Gait Patterns during Overt and Covert Evaluation in Patients with Parkinson’s Disease and Healthy Subjects: Is There a Hawthorne Effect? J. Appl. Biomech. 2015, 31, 189–194. [Google Scholar] [CrossRef] [PubMed]
- Dobkin, B.H.; Dorsch, A. The Promise of mHealth: Daily Activity Monitoring and Outcome Assessments by Wearable Sensors. Neurorehabil. Neural Repair 2011, 25, 788–798. [Google Scholar] [CrossRef] [PubMed]
- Pasluosta, C.F.; Gassner, H.; Winkler, J.; Klucken, J.; Eskofier, B.M. An emerging era in the management of Parkinson’s disease: Wearable technologies and the internet of things. IEEE J. Biomed. Health Inform. 2015, 19, 1873–1881. [Google Scholar] [CrossRef] [PubMed]
- Weiss, A.; Sharifi, S.; Plotnik, M.; van Vugt, J.P.P.; Giladi, N.; Hausdorff, J.M. Toward Automated, At-Home Assessment of Mobility among Patients with Parkinson Disease, Using a Body-Worn Accelerometer. Neurorehabil. Neural Repair 2011, 25, 810–818. [Google Scholar] [CrossRef] [PubMed]
- Hamacher, D.; Hamacher, D.; Taylor, W.R.; Singh, N.B.; Schega, L. Towards clinical application: Repetitive sensor position re-calibration for improved reliability of gait parameters. Gait Posture 2014, 39, 1146–1148. [Google Scholar] [CrossRef] [PubMed]
- Donath, L.; Faude, O.; Lichtenstein, E.; Pagenstert, G.; Nüesch, C.; Mündermann, A. Mobile inertial sensor based gait analysis: Validity and reliability of spatiotemporal gait characteristics in healthy seniors. Gait Posture 2016, 49, 371–374. [Google Scholar] [CrossRef] [PubMed]
- Orlowski, K.; Eckardt, F.; Herold, F.; Aye, N.; Edelmann-Nusser, J.; Witte, K. Examination of the reliability of an inertial sensor-based gait analysis system. Biomed. Eng./Biomed. Tech. 2017. [Google Scholar] [CrossRef] [PubMed]
- Bruening, D.A.; Ridge, S.T. Automated event detection algorithms in pathological gait. Gait Posture 2014, 39, 472–477. [Google Scholar] [CrossRef] [PubMed]
- Calliess, T.; Bocklage, R.; Karkosch, R.; Marschollek, M.; Windhagen, H.; Schulze, M. Clinical Evaluation of a Mobile Sensor-Based Gait Analysis Method for Outcome Measurement after Knee Arthroplasty. Sensors 2014, 14, 15953–15964. [Google Scholar] [CrossRef] [PubMed]
- Tadano, S.; Takeda, R.; Sasaki, K.; Fujisawa, T.; Tohyama, H. Gait characterization for osteoarthritis patients using wearable gait sensors (H-Gait systems). J. Biomech. 2016, 49, 684–690. [Google Scholar] [CrossRef] [PubMed]
- Muro-de-la Herran, A.; García-Zapirain, B.; Méndez-Zorrilla, A. Gait Analysis Methods: An Overview of Wearable and Non-Wearable Systems, Highlighting Clinical Applications. Sensors 2014, 14, 3362–3394. [Google Scholar] [CrossRef] [PubMed]
- Papi, E.; Osei-Kuffour, D.; Chen, Y.M.A.; McGregor, A.H. Use of wearable technology for performance assessment: A validation study. Med. Eng. Phys. 2015, 37, 698–704. [Google Scholar] [CrossRef] [PubMed]
- Kobsar, D.; Osis, S.T.; Phinyomark, A.; Boyd, J.E.; Ferber, R. Reliability of gait analysis using wearable sensors in patients with knee osteoarthritis. J. Biomech. 2016, 49, 3977–3982. [Google Scholar] [CrossRef] [PubMed]
- Kitagawa, N.; Ogihara, N. Estimation of foot trajectory during human walking by a wearable inertial measurement unit mounted to the foot. Gait Posture 2016, 45, 110–114. [Google Scholar] [CrossRef] [PubMed]
- Hughes, A.J.; Daniel, S.E.; Kilford, L.; Lees, A.J. Accuracy of clinical diagnosis of idiopathic Parkinson’s disease: A clinico-pathological study of 100 cases. J. Neurol. Neurosurg. Psychiatry 1992, 55, 181–184. [Google Scholar] [CrossRef] [PubMed]
- Hoehn, M.M.; Yahr, M.D. Parkinsonism: Onset, progression, and mortality. Neurology 1967, 17, 427–442. [Google Scholar] [CrossRef] [PubMed]
- Combs, S.A.; Diehl, M.D.; Filip, J.; Long, E. Short-distance walking speed tests in people with Parkinson disease: Reliability, responsiveness, and validity. Gait Posture 2014, 39, 784–788. [Google Scholar] [CrossRef] [PubMed]
- Kluge, F.; Pasluosta, C.; Gassner, H.; Klucken, J.; Eskofier, B.M. MotionLab@Home: Complementary Measurement of Gait Characteristics Using Wearable Technology and Markerless Video Tracking—A Study Protocol. Adv. Eng. Forum 2016, 19, 149–155. [Google Scholar] [CrossRef]
- ActivityNet Database. Available online: www.activitynet.org (accessed on 30 May 2017).
- Burns, A.; Greene, B.R.; McGrath, M.J.; O’Shea, T.J.; Kuris, B.; Ayer, S.M.; Stroiescu, F.; Cionca, V. SHIMMER—A Wireless Sensor Platform for Noninvasive Biomedical Research. IEEE Sens. J. 2010, 10, 1527–1534. [Google Scholar] [CrossRef]
- Menant, J.C.; Steele, J.R.; Menz, H.B.; Munro, B.J.; Lord, S.R. Effects of walking surfaces and footwear on temporo-spatial gait parameters in young and older people. Gait Posture 2009, 29, 392–397. [Google Scholar] [CrossRef] [PubMed]
- Klucken, J.; Barth, J.; Kugler, P.; Schlachetzki, J.; Henze, T.; Marxreiter, F.; Kohl, Z.; Steidl, R.; Hornegger, J.; Eskofier, B.; et al. Unbiased and Mobile Gait Analysis Detects Motor Impairment in Parkinson’s Disease. PLoS ONE 2013, 8, e56956. [Google Scholar] [CrossRef] [PubMed]
- Ferraris, F.; Grimaldi, U.; Parvis, M. Procedure for effortless in-field calibration of three-axial rate gyro and accelerometers. Sens. Mater. 1995, 7, 311–330. [Google Scholar]
- Barth, J.; Oberndorfer, C.; Pasluosta, C.; Schülein, S.; Gassner, H.; Reinfelder, S.; Kugler, P.; Schuldhaus, D.; Winkler, J.; Klucken, J.; et al. Stride Segmentation during Free Walk Movements Using Multi-Dimensional Subsequence Dynamic Time Warping on Inertial Sensor Data. Sensors 2015, 15, 6419–6440. [Google Scholar] [CrossRef] [PubMed]
- Rampp, A.; Barth, J.; Schülein, S.; Gaßmann, K.G.; Klucken, J.; Eskofier, B.M. Inertial Sensor Based Stride Parameter Calculation from Gait Sequences in Geriatric Patients. IEEE Trans. Biomed. Eng. 2014, 62, 1089–1097. [Google Scholar] [CrossRef] [PubMed]
- Euston, M.; Coote, P.; Mahony, R.; Kim, J.; Hamel, T. A complementary filter for attitude estimation of a fixed-wing UAV. In Proceedings of the 2008 IEEE/RSJ International Conference on Intelligent Robots and Systems, IROS, Nice, France, 22–26 September 2008; pp. 340–345. [Google Scholar]
- Zok, M.; Mazzà, C.; Della Croce, U. Total body centre of mass displacement estimated using ground reactions during transitory motor tasks: Application to step ascent. Med. Eng. Phys. 2004, 26, 791–798. [Google Scholar] [CrossRef] [PubMed]
- Perry, J. Gait Analysis: Normal and Pathological Function; SLACK: Thorofare, NJ, USA, 1992. [Google Scholar]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2016. [Google Scholar]
- Bland, J.M.; Altman, D.G. Statistical methods for assessing agreement between two methods of clinical measurement. Lancet 1986, 327, 307–310. [Google Scholar] [CrossRef]
- Shrout, P.E.; Fleiss, J.L. Intraclass correlations: Uses in assessing rater reliability. Psychol. Bull. 1979, 86, 420–428. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Zeng, L.; Lin, Z.J.; Cazzell, M.; Liu, H. Tutorial on use of intraclass correlation coefficients for assessing intertest reliability and its application in functional near-infrared spectroscopy-based brain imaging. J. Biomed. Opt. 2015, 20, 050801. [Google Scholar] [CrossRef] [PubMed]
- Cicchetti, D.V. Guidelines, criteria, and rules of thumb for evaluating normed and standardized assessment instruments in psychology. Psychol. Assess. 1994, 6, 284–290. [Google Scholar] [CrossRef]
- Bohannon, R.W.; Williams Andrews, A. Normal walking speed: A descriptive meta-analysis. Physiotherapy 2011, 97, 182–189. [Google Scholar] [CrossRef] [PubMed]
- Sofuwa, O.; Nieuwboer, A.; Desloovere, K.; Willems, A.M.; Chavret, F.; Jonkers, I. Quantitative Gait Analysis in Parkinson’s Disease: Comparison with a Healthy Control Group. Arch. Phys. Med. Rehabil. 2005, 86, 1007–1013. [Google Scholar] [CrossRef] [PubMed]
- Lanovaz, J.L.; Oates, A.R.; Treen, T.T.; Unger, J.; Musselman, K.E. Validation of a commercial inertial sensor system for spatiotemporal gait measurements in children. Gait Posture 2017, 51, 14–19. [Google Scholar] [CrossRef] [PubMed]
- Zeni, J.A.; Higginson, J.S. Gait parameters and stride-to-stride variability during familiarization to walking on a split-belt treadmill. Clin. Biomech. 2010, 25, 383–386. [Google Scholar] [CrossRef] [PubMed]
- Hausdorff, J.M.; Cudkowicz, M.E.; Firtion, R.; Wei, J.Y.; Goldberger, A.L. Gait variability and basal ganglia disorders: Stride-to-stride variations of gait cycle timing in parkinson’s disease and Huntington’s disease. Mov. Disord. 1998, 13, 428–437. [Google Scholar] [CrossRef] [PubMed]
- Kiss, R.M. Effect of severity of knee osteoarthritis on the variability of gait parameters. J. Electromyogr. Kinesiol. 2011, 21, 695–703. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.; Patten, C.; Kothari, D.H.; Zajac, F.E. Gait differences between individuals with post-stroke hemiparesis and non-disabled controls at matched speeds. Gait Posture 2005, 22, 51–56. [Google Scholar] [CrossRef] [PubMed]
- Becker, L.; Russ, P. Accuracy of joint angles using markerless silhouette-based tracking and hybrid tracking vs. traditional marker based tracking. In Proceedings of the 21st Annual Congress of the European College of Sport Science, Vienna, Austria, 6–9 July 2016; p. 404. [Google Scholar]
- Becker, L.; Russ, P. Genauigkeit markerloser und hybrider Bewegungsanalyse im Vergleich zu marker basierten Verfahren bei der Erfassung von Gelenkwinkeln. In Proceedings of the 11 Symposium der dvs Sportinformatik, Magdeburg, Germany, 14–16 September 2016; p. 28. [Google Scholar]
- Ceseracciu, E.; Sawacha, Z.; Cobelli, C. Comparison of markerless and marker-based motion capture technologies through simultaneous data collection during gait: Proof of concept. PLoS ONE 2014, 9, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Corazza, S.; Mündermann, L.; Gambaretto, E.; Ferrigno, G.; Andriacchi, T.P. Markerless motion capture through visual hull, articulated ICP and subject specific model generation. Int. J. Comput. Vis. 2010, 87, 156–169. [Google Scholar] [CrossRef]
- Gorton, G.E.; Hebert, D.A.; Gannotti, M.E. Assessment of the kinematic variability among 12 motion analysis laboratories. Gait Posture 2009, 29, 398–402. [Google Scholar] [CrossRef] [PubMed]
- Osis, S.T.; Hettinga, B.A.; Macdonald, S.; Ferber, R. Effects of simulated marker placement deviations on running kinematics and evaluation of a morphometric-based placement feedback method. PLoS ONE 2016, 11, 1–13. [Google Scholar] [CrossRef] [PubMed]

| Healthy Subjects | PD Patients | |
|---|---|---|
| Gender (m:f) | 6:5 | 2:2 |
| Age (years) | 33.6 ± 5.7 | 70.5 ± 6.6 |
| Mass (kg) | 77.1 ± 20.7 | 72.6 ± 5.3 |
| Height (cm) | 180.3 ± 9.9 | 172.8 ± 6.7 |
| UPDRS-III | - | 20.0 ± 6.4 |
| Hoehn & Yahr | - | 2.4 ± 0.8 |
| Parameter | Sensor | Camera | r | Bias | Abs. Error | Abs. Error (%) |
|---|---|---|---|---|---|---|
| Stride time (s) | 1.15 (0.18) | 1.15 (0.18) | −0.000 (0.016) | 0.013 (0.010) | ||
| Stance time (s) | 0.74 (0.14) | 0.70 (0.13) | 0.037 (0.020) | 0.037 (0.019) | ||
| Swing time (s) | 0.41 (0.05) | 0.45 (0.05) | −0.037 (0.020) | 0.037 (0.019) | ||
| Stride length (m) | 1.43 (0.22) | 1.45 (0.22) | −0.014 (0.067) | 0.053 (0.043) | ||
| Velocity (m/s) | 1.30 (0.37) | 1.31 (0.37) | −0.012 (0.061) | 0.048 (0.040) |
| Parameter | Sensor | Camera | r | Bias | Abs. Error | Abs. Error (%) |
|---|---|---|---|---|---|---|
| Stride time (s) | ||||||
| Healthy | 1.13 (0.18) | 1.13 (0.18) | −0.001 (0.015) | 0.012 (0.009) | ||
| Patient | 1.27 (0.15) | 1.27 (0.15) | 0.003 (0.020) | 0.016 (0.013) | ||
| Stance time (s) | ||||||
| Healthy | 0.72 (0.13) | 0.69 (0.13) | 0.036 (0.020) | 0.037 (0.019) | ||
| Patient | 0.84 (0.12) | 0.80 (0.12) | 0.042 (0.020) | 0.042 (0.020) | ||
| Swing time (s) | ||||||
| Healthy | 0.41 (0.05) | 0.44 (0.05) | −0.037 (0.019) | 0.037 (0.019) | ||
| Patient | 0.43 (0.04) | 0.47 (0.04) | −0.039 (0.026) | 0.041 (0.023) | ||
| Stride length (m) | ||||||
| Healthy | 1.45 (0.21) | 1.47 (0.21) | −0.016 (0.066) | 0.053 (0.044) | ||
| Patient | 1.25 (0.18) | 1.26 (0.17) | −0.001 (0.065) | 0.052 (0.039) | ||
| Velocity (m/s) | ||||||
| Healthy | 1.34 (0.37) | 1.35 (0.37) | −0.013 (0.062) | 0.049 (0.041) | ||
| Patient | 1.01 (0.24) | 1.02 (0.24) | −0.004 (0.052) | 0.041 (0.031) |
| Sensor System | Camera System | |||
|---|---|---|---|---|
| ICC(2,1) | ICC(2,k) | ICC(2,1) | ICC(2,k) | |
| Stride time (s) | ||||
| fast | 0.89 | 0.96 | 0.91 | 0.97 |
| normal | 0.92 | 0.97 | 0.91 | 0.97 |
| slow | 0.94 | 0.98 | 0.93 | 0.98 |
| Stance time (s) | ||||
| fast | 0.87 | 0.95 | 0.89 | 0.96 |
| normal | 0.90 | 0.97 | 0.92 | 0.97 |
| slow | 0.94 | 0.98 | 0.91 | 0.97 |
| Swing time (s) | ||||
| fast | 0.92 | 0.97 | 0.83 | 0.94 |
| normal | 0.92 | 0.97 | 0.81 | 0.93 |
| slow | 0.86 | 0.95 | 0.88 | 0.96 |
| Stride length (m) | ||||
| fast | 0.87 | 0.95 | 0.87 | 0.95 |
| normal | 0.81 | 0.93 | 0.83 | 0.94 |
| slow | 0.87 | 0.95 | 0.92 | 0.97 |
| Velocity (m/s) | ||||
| fast | 0.75 | 0.90 | 0.72 | 0.88 |
| normal | 0.78 | 0.92 | 0.74 | 0.89 |
| slow | 0.55 | 0.79 | 0.55 | 0.79 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kluge, F.; Gaßner, H.; Hannink, J.; Pasluosta, C.; Klucken, J.; Eskofier, B.M. Towards Mobile Gait Analysis: Concurrent Validity and Test-Retest Reliability of an Inertial Measurement System for the Assessment of Spatio-Temporal Gait Parameters. Sensors 2017, 17, 1522. https://doi.org/10.3390/s17071522
Kluge F, Gaßner H, Hannink J, Pasluosta C, Klucken J, Eskofier BM. Towards Mobile Gait Analysis: Concurrent Validity and Test-Retest Reliability of an Inertial Measurement System for the Assessment of Spatio-Temporal Gait Parameters. Sensors. 2017; 17(7):1522. https://doi.org/10.3390/s17071522
Chicago/Turabian StyleKluge, Felix, Heiko Gaßner, Julius Hannink, Cristian Pasluosta, Jochen Klucken, and Björn M. Eskofier. 2017. "Towards Mobile Gait Analysis: Concurrent Validity and Test-Retest Reliability of an Inertial Measurement System for the Assessment of Spatio-Temporal Gait Parameters" Sensors 17, no. 7: 1522. https://doi.org/10.3390/s17071522
APA StyleKluge, F., Gaßner, H., Hannink, J., Pasluosta, C., Klucken, J., & Eskofier, B. M. (2017). Towards Mobile Gait Analysis: Concurrent Validity and Test-Retest Reliability of an Inertial Measurement System for the Assessment of Spatio-Temporal Gait Parameters. Sensors, 17(7), 1522. https://doi.org/10.3390/s17071522








