1. Introduction
Stroke survivors often suffer from severe long-term disability and cognitive decline [
1,
2,
3]. Thrombolysis and thrombectomy are the most effective therapies in acute ischemic stroke [
2,
4]. Both therapies restore cerebral blood flow, with the goal of rescuing the perilesional tissue that has not sustained irreversible damage. Such a mismatch can be observed in the early hours following cerebral ischemia, making only a minority of stroke patients eligible for such therapy [
2]. Despite clinical improvement due to recanalization, the 90-day mortality of treated and untreated stroke survivors has not improved substantially [
5], and 30–50% of patients suffer from cognitive decline, impeding a return to work [
6].
It is known that infiltration of immune cells contributes to secondary brain damage; however, so far, studies aiming to improve stroke outcome by modulating immunological or neuroregenerative pathways have not translated into clinical application. As a consequence, a more detailed understanding of the processes in the post-acute stroke phase is required to identify new therapeutic targets.
The immunological consequences of ischemic stroke include both impaired bacterial defense mechanisms facilitating post-stroke infections and excessive activation of cells of the adaptive immune system, including T cells [
7]. It is known that T cells are of major importance for functional physiologic cognitive processes [
8], and T cell interaction with brain cells and immune cells has been suggested to modulate cognitive recovery after stroke [
9,
10]. Following ischemic stroke, T cells upregulate activation markers, which are not controlled due to disturbed upregulation of negative feedback loops; one such example is cytotoxic T-lymphocyte-associated protein (CTLA)-4 [
7]. Therefore, the activation of T cells persists for several weeks [
7,
9,
11,
12,
13,
14]. Nevertheless, our knowledge of the course of T cell activation in patients post stroke is still very limited: Data are available mainly on CD4
+ T cells, few activation markers (HLA-DR, CD25, CTLA-4), and for a limited time span of 14 days [
7]. While it is undisputed that activated lymphocytes migrate to the ischemic lesion and contribute to secondary lesion growth, detailed knowledge about the role of the immune system during long-term recovery is still scarce. To extend current knowledge beyond the acute and subacute phases, we included a three-month time point representing the early chronic recovery period after stroke. Previous studies have reported persistent lymphocyte activation for several weeks but lack data beyond 14 days [
7,
11,
12]. We therefore aimed to capture the later stage when inhibitory feedback mechanisms, such as CTLA-4 and programmed cell death 1 ligand (PD-L1) expression, may become re-established. The three-month time point represents the post-acute stage of stroke recovery, where sustained systemic immune alterations have been associated with long-term neurological and cognitive outcomes.
The current study was designed to investigate CD25 expression (the alpha chain of the IL-2 receptor) as well as the activation inhibitory feedback loop providers CTLA-4 and PD-L1 on CD4+ and the CD8+ T cell subgroup. In addition, we focused on the small but functionally relevant double-negative (CD4- CD8-) T cells (DNTs).
Microglia are brain-resident immune cells either shaping a pro- (M1 microglia) or anti-inflammatory (M2 microglia) micro milieu [
15,
16,
17,
18]. While the recent literature has rightly criticized the oversimplified M1/M2 dichotomy as unable to capture the full spectrum of microglial phenotypes revealed by modern transcriptomic and proteomic profiling [
19,
20], we retain this nomenclature here for pragmatic reasons. Despite its limitations, the M1/M2 framework remains a broadly understood and functional shorthand for distinguishing pro- and anti-inflammatory microglial responses in the post-stroke setting, particularly in translational and therapeutic contexts [
19,
20]. We emphasize that this nomenclature is retained solely as a conventional and practical framework, acknowledging that microglial activation represents a dynamic continuum rather than a strict binary state.
Starting 5 days post stroke, M1 outnumber M2 microglia in the infarction area [
16]. M1 microglia downregulate the chemokine receptor (CX3CR)1, which arrests microglia cells in an inactive state upon binding of its ligand CX3CL1 [
21]. M1 microglia substantially contribute to persisting neuroinflammation post stroke and produce major proportions of TNF-α in the infarct [
22]. By means of a reduced rate of phagocytosis and the inflammatory micromilieu, M1 microglia exacerbate the ramifications of oxygen and glucose deprivation in the infarct. [
16,
23]. Subsequently, lymphocytes infiltrate the affected brain parenchyma; both CD4
+ and CD8
+ T cells migrate to the infarct. By secreting inflammatory Th1 cytokines such as IL-1β and interferon (IFN)-γ, T cells facilitate adhesion of leukocytes and thrombus formation and substantially contribute to neuroinflammation [
24]. The resulting elevated IFN-γ concentrations and the correlating temporal course foster the hypothesis that infiltrating T cells shape M1 activation of microglia and maintain their proinflammatory phenotype. So far, this detrimental interaction has only been shown in murine organisms [
25].
Within the population of T lymphocytes, CD4- and CD8-double-negative T cells (DNTs) represent a small subset (1.5–7.5%) which exhibit both inflammatory and anti-inflammatory properties [
26,
27]. Meng et al. observed a significant increase in DNTs in the acute phase post stroke. However, the role of DNTs during stroke-induced immune alteration has not been fully elucidated [
28].
To investigate T cell function within the post-acute stroke recovery phase as well as the interaction of T cells, especially that of DNTs with microglia cells, we investigated T cell activation until 3 months post stroke and measured the ex vivo effects of T cells obtained from stroke patients (herein, also DNTs) on human microglia cells and vice versa. In addition, we quantified the release of the proinflammatory cytokine IL-1β by coculturing T cells and microglia cells.
This study addresses the current knowledge gap regarding delayed dysregulation of immune checkpoints after ischemic stroke. We hypothesized that reduced negative feedback via CTLA-4 and PD-L1 contributes to sustained T cell activation three months after the event and may be further modulated by interactions with microglia, including double-negative T cells.
3. Discussion
Stroke alters immune function in several ways. Among them, CD4+ T cell activation has been described by several groups within the first weeks after stroke, but it has remained unknown for how long such activation lasts in patients, which pathways could be of importance for chronic activation, and if such activation also applies for CD8+ T cells. Extending observation to 3 months post stroke thus enabled assessment of the transition from persistent activation to re-emerging inhibitory signaling through CTLA-4 and PD-L1. Other inhibitory checkpoint pathways such as PD-1, TIM-3, LAG-3, and TIGIT likely contribute to T cell regulation after stroke but were beyond the scope of this focused analysis and should be addressed in future studies.
These observations are consistent with recent reports demonstrating prolonged immune checkpoint alteration and chronic neuroinflammation after stroke in both human and pre-clinical studies [
29,
30]. While decreased checkpoint expression has been reported in ischemic conditions, the definitive mechanistic pathways through which stroke alters CTLA-4 and PD-L1 signaling remain insufficiently understood. Therefore, our findings should be considered an indication of possible checkpoint dysregulation rather than proof of impaired signaling function.
It is also plausible that sustained T cell activation itself leads to reduced checkpoint expression, indicating a bidirectional relationship rather than a unidirectional failure of inhibition.
This suggests potential implications not only for immune dysregulation but also for cognitive recovery trajectories.
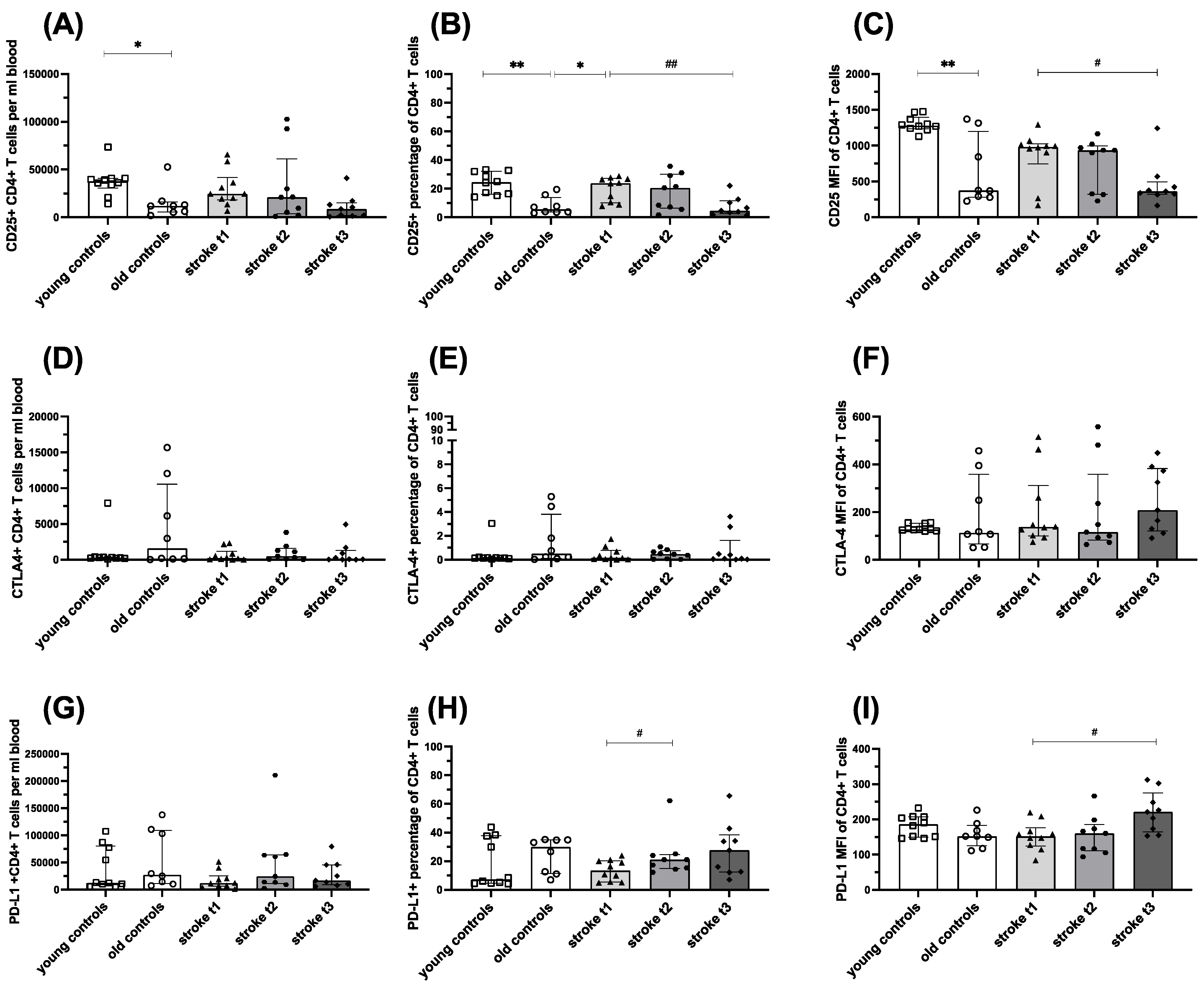
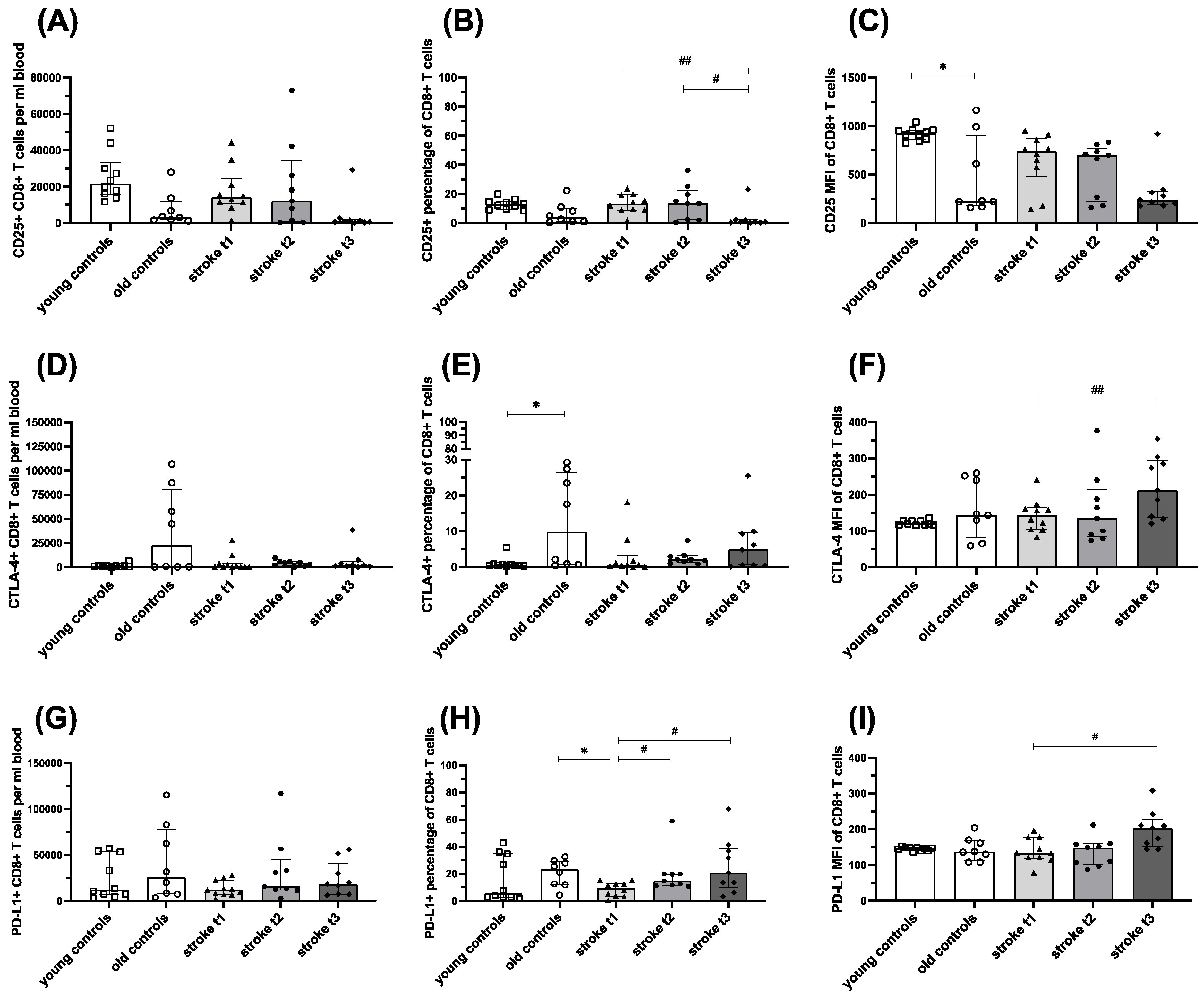
3.1. Persistent Activation of CD4+ and CD8+ T Cells Post Stroke Might Be Terminated via the Late Upregulation of Inhibitory CTLA-4 and PD-L1
In the acute post-stroke phase (t
1), CD25 expression was significantly increased on both CD4
+ and CD8
+ T cells and only declined at t
3. This expands findings from earlier studies [
7] to a period of 3 months and to CD8
+ T cells. Activation of CD8
+ T cells has so far only been described in a murine stroke model and only considering the early activation marker CD69 [
31]. This chronic T cell activation within the first month is accompanied by a lack of CTLA-4 upregulation, which normally functions as a negative feedback regulator of activation. While at t
3, the activation of T cells is diminished, CTLA-4 tended to be upregulated again on CD4
+ T cells and was significantly upregulated on CD8
+ T cells. The inhibitory activation marker PD-L1 is decreased in the acute post-stroke phase followed by subsequent upregulation. These findings indicate a potential role for PD-L1 and CTLA-4 in the late termination of T cell activation post stroke.
Our findings complement the concept of post-stroke immunosuppression: persistent T cell activation in the peripheral blood can coexist with systemic immune dampening, indicating compartmentalized immune regulation, as suggested by Haeusler et al. (2012) [
12]. While altered expression suggests reduced inhibitory feedback, functional assays (e.g., checkpoint blockade or cytokine profiling) would provide stronger causal evidence and are warranted in future studies. Whether persistent T cell activation contributes to secondary complications or poorer functional outcomes after stroke remains to be evaluated in larger, clinically correlated cohorts.
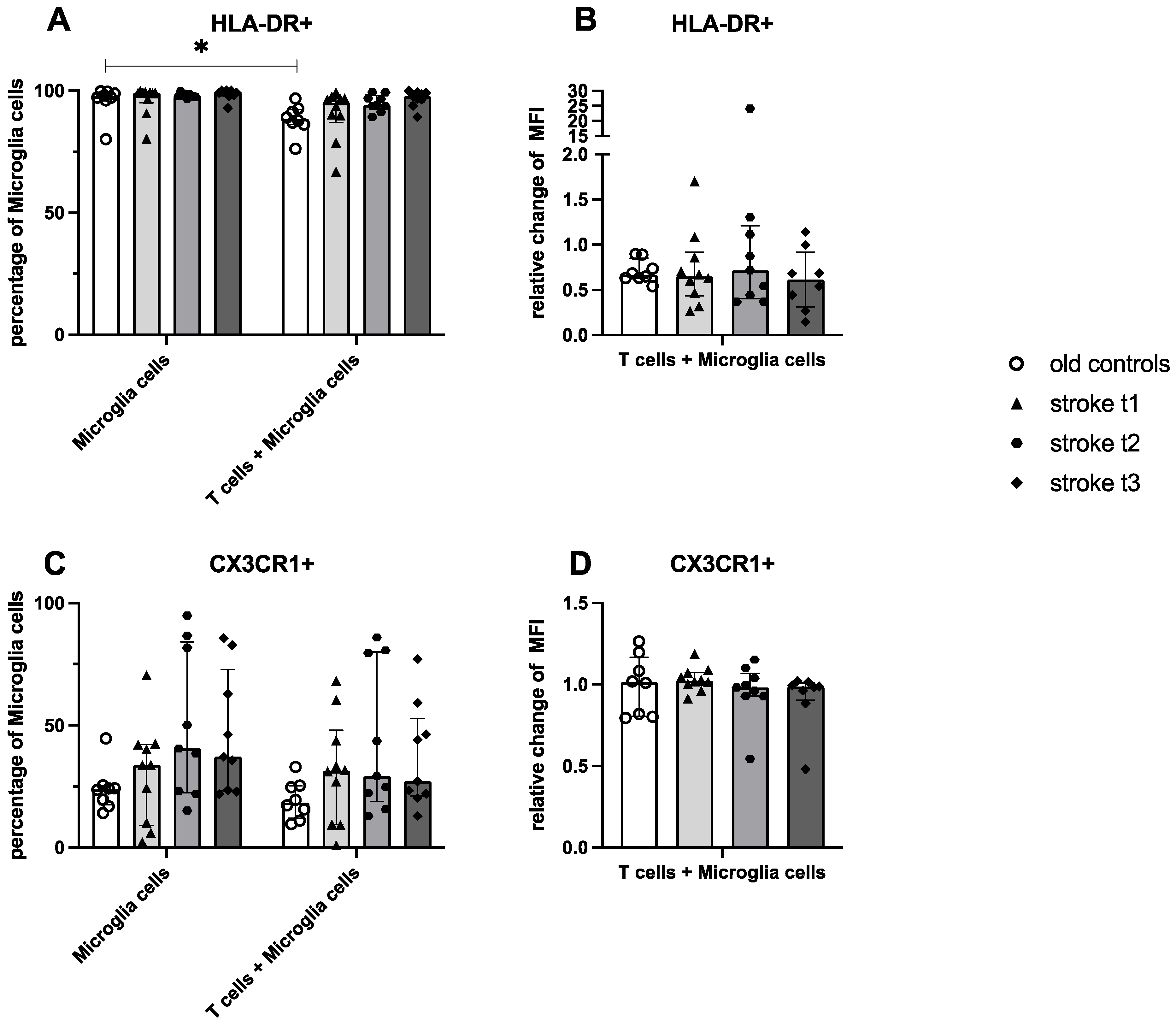
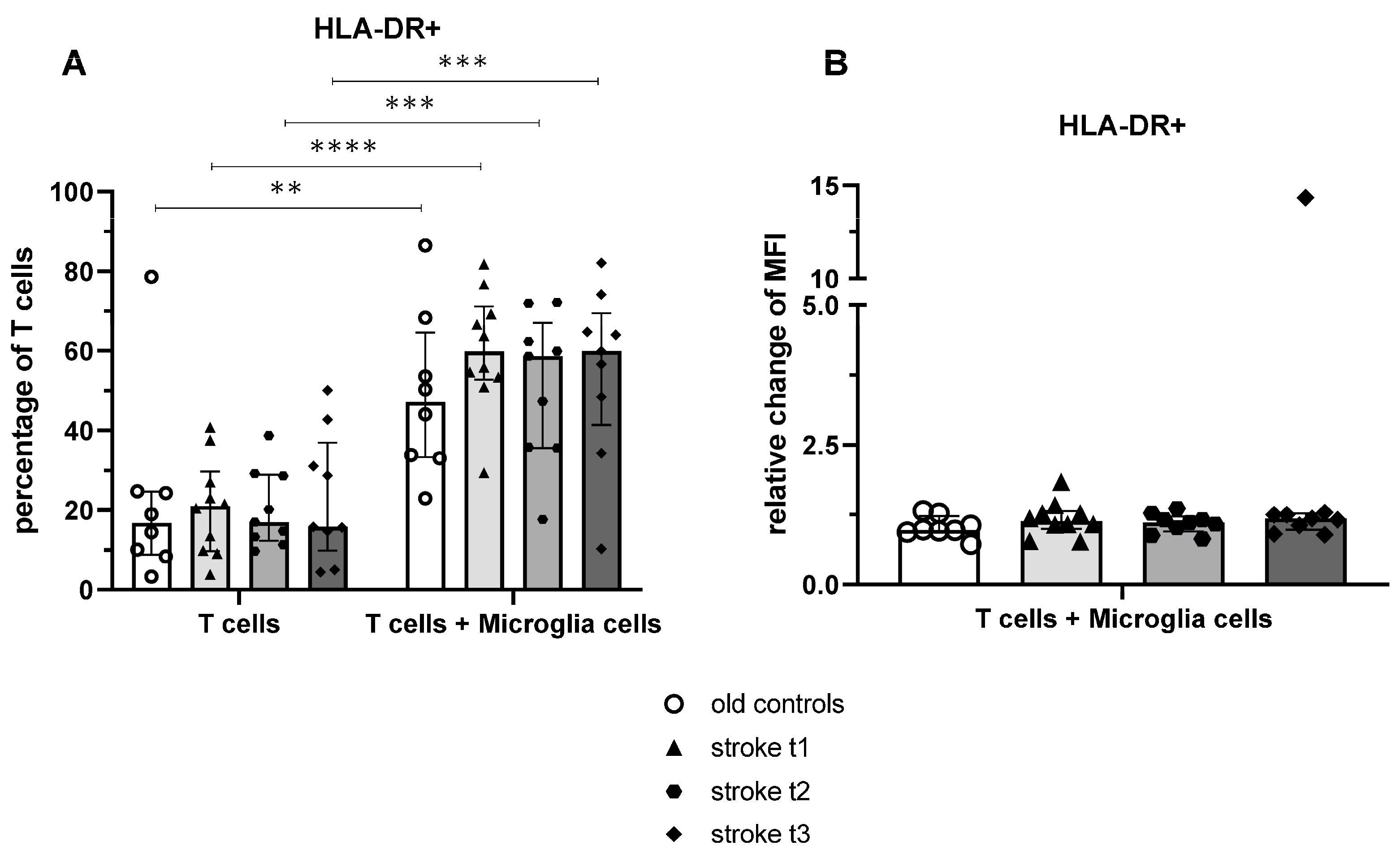
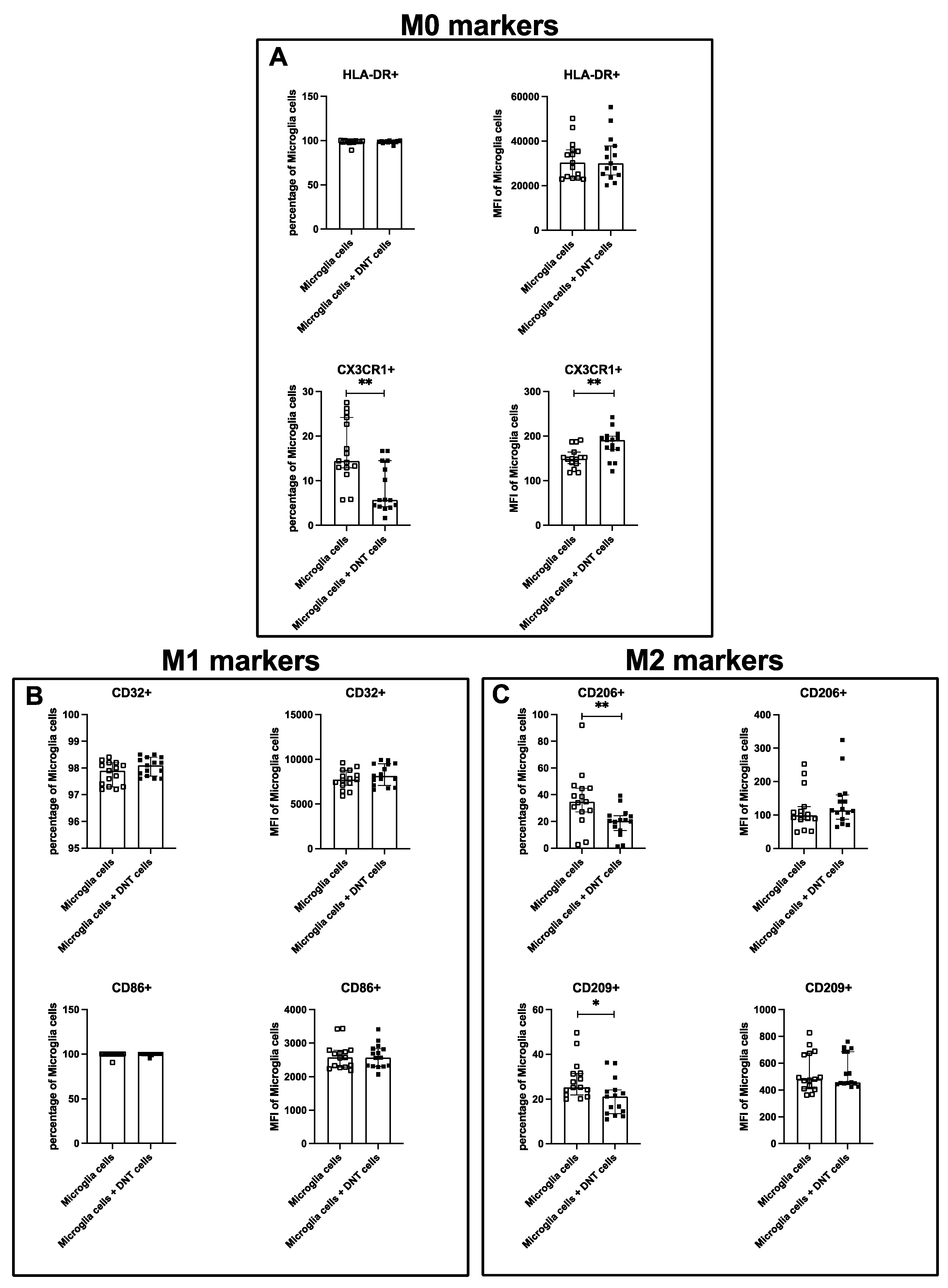
3.2. Microglia Cells Activate Both Pan-T Cells and DNTs, but Only DNTs Modulate Microglia Activation Status Ex Vivo
To elucidate potential interactions between microglia and T cells, activation of both cell types after culture alone and coculture was analysed using flow cytometry.
None of the six analysed markers on microglia (HLA-DR, CX3CR1, CD32, CD86, CD206, CD209) showed significant changes in percentage or MFI after coculture with pan-T cells. Thus, pan-T cells from stroke patients were not able to modulate the activation status of healthy microglia ex vivo, in contrast to findings in the murine organism [
32,
33]. The provided microglia cells showed high percentages of activation markers when incubated alone, which was not described in other studies [
34,
35]. As a consequence, potential modulatory T cell effects might have been masked by a high degree of pre-activation induced by the isolation procedure performed by the supplier as seen in other studies [
36]. In addition, a control experiment for allo-activation was performed (
Figure S7). Commercially derived human microglia are known to display partial pre-activation due to isolation-related stress [
36]. Resting periods prior to coculture did not further reduce activation levels. While these cells differ from primary microglia in some transcriptomic features [
15], they provide a standardized and reproducible human model suitable for ex vivo interaction studies. We have included this limitation explicitly. Since hypoxia is a known activating stimulus for microglia cells [
37], we also compared normoxic to hypoxic cell culture conditions in a control experiment but did not find any differences. That said, endotoxicity after stroke causes additional damage-associated molecular patterns which were not included within our ex vivo experiment.
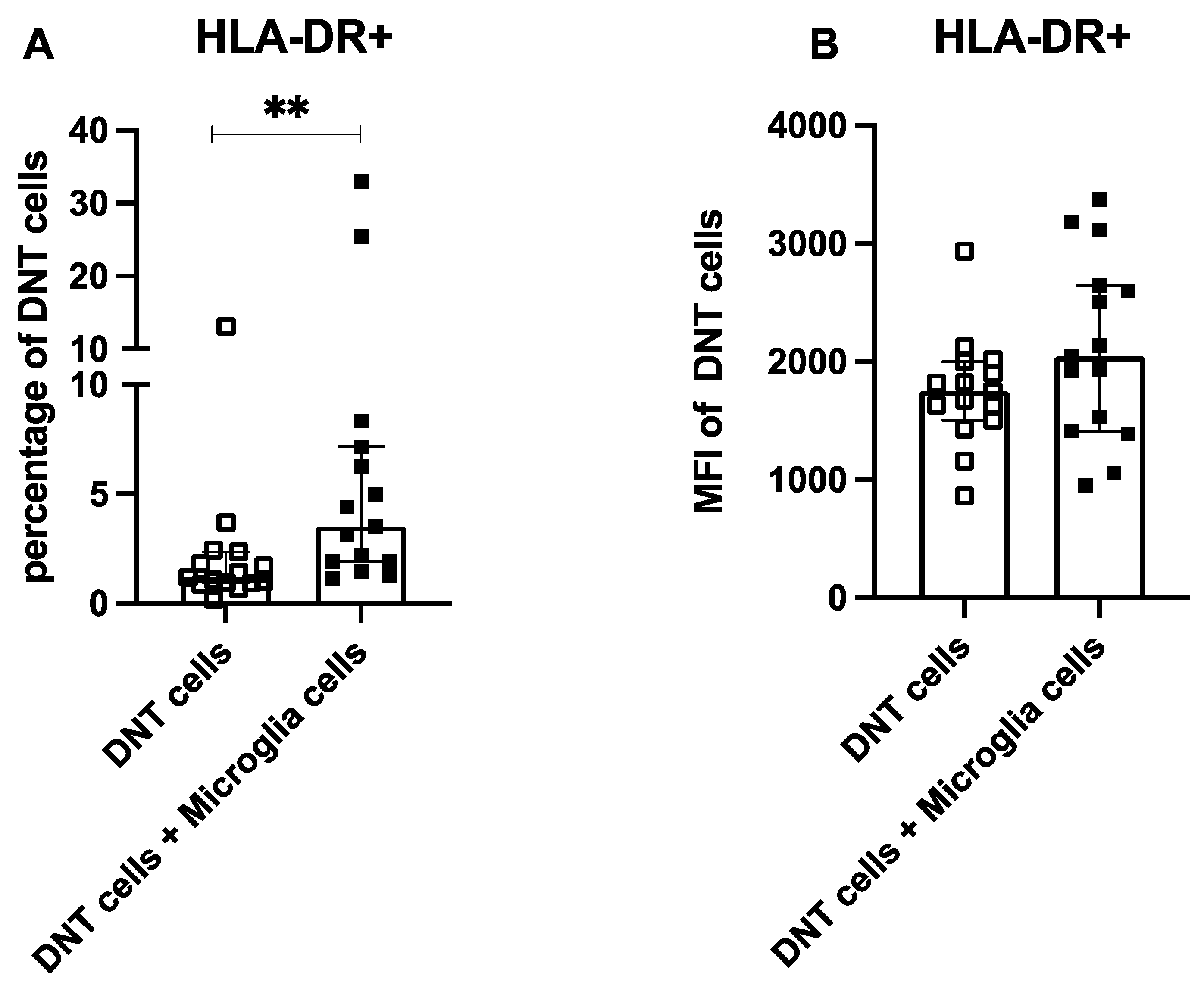
On the other hand, microglia cells effectively activated HLA-DR upregulation on pan-T cells after coculture. This significant effect was not specific for T cells from stroke patients but also observable on T cells from healthy controls; however, the effect size of activation was larger in the stroke cohort.
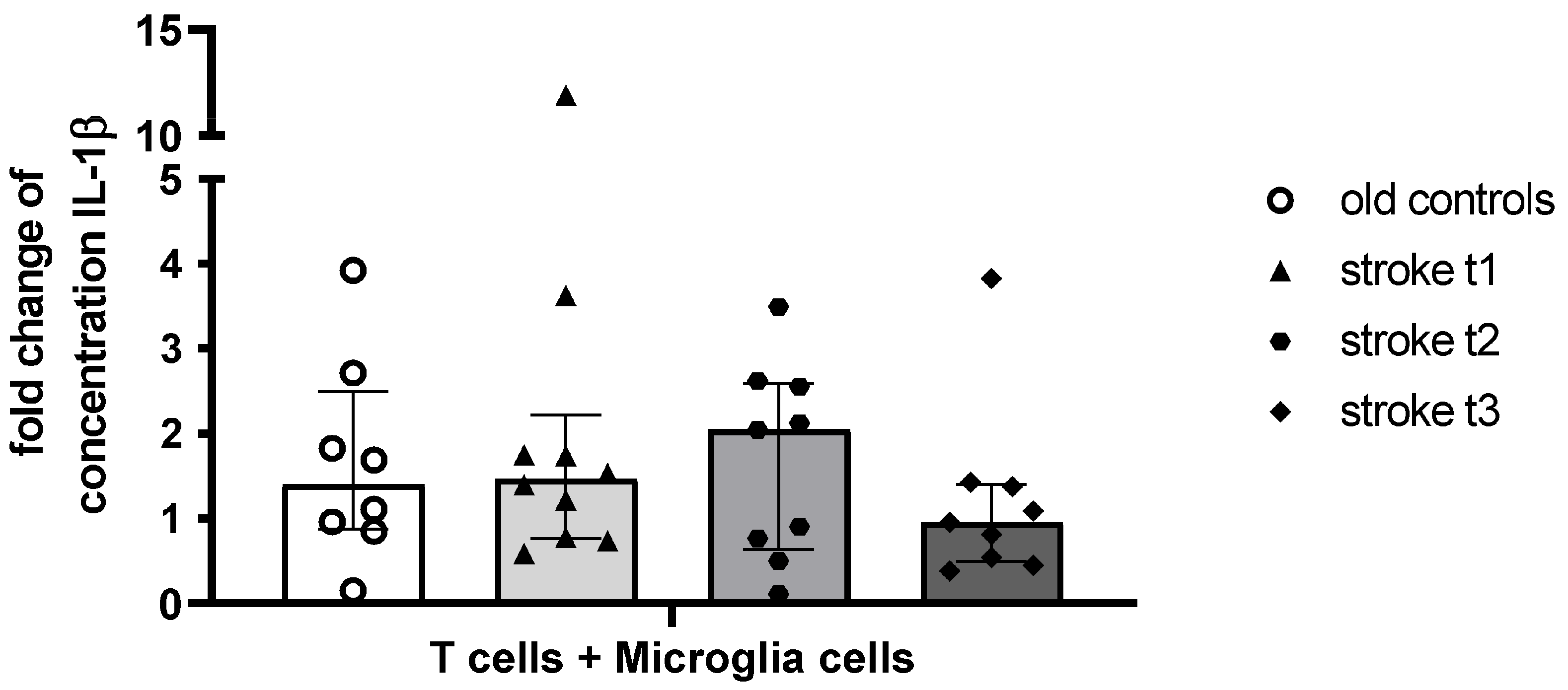
The absence of statistically significant IL-1β differences likely reflects inter-individual variability and assay sensitivity limits rather than lack of biological effect, as observed effects were concordant with cellular activation markers. Given that other cytokines were below detection, IL-1β served as a feasible indicator of inflammatory activation but warrants confirmation with higher-sensitivity assays in larger cohorts.
T cells and microglia can interact via different pathways either direct cell-to=cell contact or soluble mediators (reviewed in [
25]). While Schetters et al. and Chabot et al. showed that Th1 cells were able to induce the inflammatory M1 phenotype, Meng et al. described that not all CD3
+ T cells (but only the CD4- CD8-DNTs) were colocalized with microglia cells post stroke and were the only T cell subset that polarized microglia activation towards the M1 phenotype [
25,
28,
33]. As a consequence, the coculture setup with pan-T cells might have harbored too few DNTs per well to modulate microglia activation.
We could show that the findings regarding the role of DNTs from murine stroke models [
28] coincided with findings using human cells: While HLA-DR did not change after coculture with DNTs, the percentage of CX3CR1 decreased significantly. Since CX3CR1 arrests microglia cells in an idle state and is only downregulated after activation of microglia cells, it is sensible that DNTs activated the microglia cells from the coculture [
21]. In our study, CX3CR1 modulation is interpreted as an indirect indicator of microglial activation rather than evidence of direct ligand–receptor signaling, since CX3CL1 concentrations were not measured.
Mechanistically, DNTs may secrete pro-inflammatory mediators such as IFN-γ or IL-17, which promote M1-like microglial activation [
28]. This hypothesis aligns with the observed decrease in CX3CR1 and reduction of M2 markers following coculture. Microglia cells increased percentage of HLA-DR on DNTs from coculture significantly, but MFI of HLA-DR did not change significantly.
3.3. Limitations
We acknowledge several methodological constraints. Pharmacological treatments such as statins or antihypertensives may have modulated immune activation, although no patient received immunosuppressants. The limited sample size reflects the exploratory design and logistical complexity of longitudinal sampling combined with ex vivo coculture experiments. Effect-size calculations, however, indicated medium-to-large differences between groups, suggesting adequate power for detecting biologically relevant trends. Larger multicenter studies are needed to confirm these observations and assess correlations with clinical parameters.
This study was not capable of sex-specific analyses (43.8% female). As sex hormones influence T cell responses and stroke-induced effects, future studies should address potential sex differences in post-stroke immune activation.
Differences in cardiovascular risk profiles between stroke and control groups may influence baseline immune activation; thus, our results should be interpreted within this clinical context. Further studies will be needed to find whether the results can be found in larger and sex-matched patient cohorts as well.
The use of commercially derived, non-autologous human microglia provides standardized conditions but cannot fully reproduce patient-specific microglial phenotypes or the influence of ischemic priming. This compromise was necessary for ethical and technical reasons yet should be considered when extrapolating to in vivo interactions. Ex vivo coculture systems cannot fully reproduce the hypoxic, vascular, and cytokine gradients of the post-ischemic brain.
Additionally, the cytokine panel was restricted to IL-1β for follow-up analyses, meaning they did not capture the full breadth of inflammatory mediators. Clinical confounders such as comorbidities and ongoing medications might further affect immune marker expression.
The results herein should therefore be interpreted as mechanistic indications rather than direct representations of in vivo dynamics.
4. Methods
4.1. Patients and Controls
Samples were obtained from a total of 16 patients and 28 controls. Patient and control characteristics are listed in
Table 1. Participants included in the individual analysis are stated in the respective figure caption.
Patients were recruited at the Department of Neurology of the University Medicine Greifswald (UMG) as well as Department of Neurology of the AMEOS Clinic Ueckermuende. Inclusion criteria were as follows: National Institute of Health Stroke Scale (NIHSS) score of ≥4; no signs of systemic infection or hyperthyreosis on admission (C-reactive protein (CRP) < 50 mg/L; thyroid-stimulating hormone (TSH) > 0.27 µIU/mL); and written informed consent obtained from the patient or through a surrogate, when applicable. Exclusion criteria were defined as follows: patients receiving immunosuppressive drugs; those diagnosed with a malignancy or autoimmune disease; previous cerebral infarction within the past 6 months; clinically significant anemia; and no written informed consent by the patient themselves or through a surrogate.
To minimize systemic inflammatory confounders, patients with elevated CRP > 50 mg/L, clinical infection, or autoimmune disease were excluded. Most received standard secondary prevention (antihypertensives, statins, platelet inhibitors) but no immunosuppressive medication.
To investigate age-related effects, two control groups were included in addition to stroke patients aged 50 and older. Eight sex- and age-matched control individuals (mean age ± SD: 70.3 ± 9.8 years) were recruited from the ophthalmology clinic, either undergoing treatment for age-related macular degeneration or scheduled for cataract surgery. In addition, 20 young neurologically and immunologically healthy control individuals (mean age ± SD: 23.9 ± 3.2 years) were recruited to specifically examine the influence of age on neurobiological and immunological markers.
4.2. Blood Sampling
All venous blood samples were obtained from patients and controls between 7:00 and 10:30 am and processed within 3 h under identical conditions to reduce circadian and pre-analytical variation in immune marker expression.
CRP (C-reactive protein) and thyroid-stimulating hormone (TSH) values (quantified with Dimension Vista, Siemens Healthcare Diagnostics; Eschborn, Germany) were quantified in the central laboratory facility of the UMG. Cell isolation of peripheral blood mononuclear cells (PBMCs) started within 3 h of blood collection.
4.3. Pan-T Cell and DNT Isolation
PBMCs were obtained from EDTA-anticoagulated whole blood by separation on a Biocoll density gradient (Biocoll GmbH, Planegg/Martinsried, Germany) using standard centrifugation procedures. Pan-T cells, i.e., all CD3+ cells, were magnetically isolated from PBMCs using a Pan-T Cell Isolation Kit (Miltenyi Biotec; Bergisch-Gladbach, Germany) according to the manufacturer’s instructions and resuspended in 1 mL of Microglia Cell Medium (Pelo Biotech; Planegg, Germany).
Where applicable, DNTs were sorted using BD FACSAriaTM III (Becton Dickinson and Company; Franklin Lakes, NJ, USA). In total, 360,000 DNTs and 1,000,000 non-DNTs were sorted into two separate tubes. For separation, cells were stained with PE/Cy7 anti-human CD3—Clone: UCHT1, FITC anti-human CD4—Clone: RPA-T4 and PerCP/Cy5.5 anti-human CD8—Clone: SK1 (all BioLegend; San Diego, CA, USA).
4.4. Cell Culture
For all cell culture experiments, Costar® 24-well Clear Flat Bottom Ultra-Low-Attachment Multiple-Well Plates (Corning Incorporated; Corning, NY, USA) were used. The used human microglia cells (Pelo Biotech; Planegg, Germany) were stored in liquid nitrogen and thawed in a water bath for 105 s and afterwards counted using a Neubauer chamber. Cell viability (>95%) was verified by trypan blue exclusion and confirmed by flow cytometric gating on live CD11b+CD3− cells. The commercial microglia culture (Pelo Biotech) was free of peripheral macrophages or lymphocytes. Thawed cells were adjusted to a concentration of 15,000 cells per ml using Microglia Cell Medium, and 2 mL of cell suspension was added to the respective wells. After incubation at 37 °C and 5% CO2 for 8 h, plates were centrifuged (300× g, 5 min, Acc. 9, Dec. 7, 37 °C), and 1 mL of old medium was replaced with fresh medium. Plates were then cultured for a further 16 h and centrifuged afterwards (300× g, 5 min, Acc. 9, Dec. 7, 37 °C).
For coculture of pan-T cells and DNTs with microglia cells, 1 mL of medium was removed from each well, and 1 mL of pan-T cell or DNT suspension (concentration: 30,000 cells per mL) was added to each well. For only pan-T cell or DNT wells, 2 mL of pan-T cell or DNT suspension (concentration: 30,000 cells per mL) was seeded in empty wells. Microglia cells only served as the control.
4.5. Preparation of Whole Blood for Staining
EDTA whole blood was lysed using ACK lysis buffer (1000 mL Aqua destillata—Milli-Q® by Merck; Darmstadt, Germany; plus 8.02 g NH4Cl—Carl Roth GmbH; Karlsruhe, Germany; plus 1.0 g KHCO3—Merck KGaA; Darmstadt, Germany; plus 0.02 g EDTA—Sigma Aldrich; St. Louis, MO, USA; pH = 7.2) in a 1:10 ratio for 12 min. Lysed blood was centrifuged (300× g, 5 min, Acc. 9, Dec. 9, 21 °C) and the supernatant discarded. Cells were washed twice using DPBS (Biochrom GmbH; Berlin, Germany) (300× g, 5 min, Acc. 9, Dec. 9, 21 °C).
4.6. Analysis of Cell Activation
Cell activation was analysed after 24 h of incubation for cell culture experiments or immediately after peripheral blood withdrawal. Cells from cell culture were harvested using cell scrapers (Greiner Bio One; Kremsmünster, Austria) to regain all cells; cell-free supernatant was collected and stored at −80 °C. Cells were washed twice using DPBS (Biochrom GmbH; Berlin, Germany) (300× g, 5 min, Acc. 9, Dec. 9, 21 °C). A Zombie NIR™ Fixable Viability Kit (BioLegend; San Diego, CA, USA) was used for live/dead staining. To prevent nonspecific interactions with Fc receptors, cells were first incubated with a human FcR blocking reagent (Miltenyi Biotec) following the supplier’s recommendations. After this blocking step, surface staining was performed using the corresponding monoclonal antibodies. The panel for surface markers after cell culture included the following: PE/Cy7 anti-human CD3—Clone: UCHT1, BV510 anti-human CD11b—Clone: M1/70, AF488 anti-human HLA-DR—Clone: L243, BV650 anti-human CX3CR1—Clone: 2A9-1, AF647 anti-human CD32—Clone: FUN-2, BV421 anti-human CD86—Clone: IT2.2, PE anti-human CD206—Clone: 15-2, and PerCP/Cy5.5 anti-human CD209—Clone: 9E9A8 (all BioLegend; San Diego, CA, USA). Percentage and median fluorescence intensity (MFI) values were used to quantify marker expression on CD3−CD11b+ microglia. In parallel, HLA-DR expression was evaluated on CD3+ T cells. Fluorescence minus one (FMO) controls were included to verify gating accuracy and exclude spillover-related false positives. Relative changes in MFI were calculated as a quotient of MFIMicroglia/MFIcoculture or MFIT cells/MFIcoculture.
The panel for surface markers for whole blood included the following: FITC anti-human CD4—Clone: RPA-T4, PerCP/Cy5.5 anti-human CD8—Clone: SK1, PE/Cy7 anti-human CD25—Clone: M-A251, PE anti-human CD57—Clone: HCD57, APC anti-human CTLA-4 (CD152)—Clone: L3D10, and PE/Dazzle594 anti-human PD-L1 (CD274)—Clone: 29E.2A3 (all BioLegend; San Diego, CA, USA). Percentage; expression density of activation markers CD25, CTLA-4, and PD-L1 determined by the MFI; and absolute cell counts per ml blood were analysed for CD4+ and CD8+ T cells. Staining was controlled by using FMO samples for CD25, CTLA-4, and PD-L1.
Data acquisition was performed on an LSR II flow cytometer (BD Biosciences, NY, USA), and subsequent analysis was carried out using FlowJo™ software version 10.5.0 (BD, Ashland, OR, USA). Representative graphs for gating strategy from cell culture and whole blood are shown in the
Supplemental Figure (Figures S1 and S2). Further staining controls for expression of T cell activation markers are shown in
Supplemental Figures S3 and S4.
All samples were processed using identical instrument settings and compensation controls; nonetheless, inter-day variability cannot be fully excluded.
4.7. Analysis of Cytokine Production
We performed an initial screening by cytokine plex (LEGENDplex™ HU Th Cytokine Panel (12-plex), BioLegend, San Diego, CA, USA) within 4 different samples based on where we expected the most effects due to differences observed in cell activation markers. IL-2 (<0.82 pg/mL)), IL-4 (<1.25 pg/mL), IL-5 (<1.44 pg/mL), IL-13 (<0.91 pg/mL), IL-17A (<0.48 pg/mL), IL-17F (<1.17 pg/mL), IL-22 (V1.14 pg/mL), and IFN-g (<1.31 pg/mL) were always below detection limits (see value in brackets). The other cytokines IL-6, IL-9, IL-10, and TNF-a were detectable in some but never in all of the screened samples, revealing rather individual differences between patients than differences based on cell culture conditions. We therefore did not proceed to investigate these prescreened cytokines but continued with IL-1β. IL-1β was selected because of its central role in microglial activation and its reliable detection in preliminary multiplex screening in our cohort.
A high-sensitivity IL-1β ELISA (Thermo Fisher; Darmstadt, Germany) was performed according to the manufacturer’s instructions. Only for the standard curve was a top standard of 20 pg/mL included instead of 10 pg/mL. For analyses, the fold change in IL-1β concentration was calculated as concentration (T cells + Microglia cells)/concentration (Microglia cells).
4.8. Statistics
For visualization and statistical analyses, Graphpad Prism 8.2 (GraphPad Software Inc., San Diego, CA, USA) was used. The normality of the data distributions was assessed using the Shapiro–Wilk test. In case of normally distributed data subsets within one analysis, Levene’s test was performed to test for homoscedasticity. To compare more than two groups, a one-way ANOVA or repeated-measures ANOVA was performed in case of homoscedastic data subsets, while the Kruskal–Wallis test or Friedman test was performed in case of heteroscedastic data. Dunn’s post hoc test and Dunnett’s post hoc test was used.
For pairwise comparisons, normality and homoscedasticity were tested as described above. Afterwards, Student’s t-test or the Wilcoxon–Mann–Whitney test were performed. For effect sizes, Cohen’s d was calculated. For all analyses, a p-value < 0.05 was regarded as significant.
Given the exploratory nature of this study and the limited cohort size, p-values were interpreted descriptively, and effect sizes were reported where appropriate. Formal multiple-comparison corrections were not applied.
5. Conclusions
Collectively, our findings suggest a model in which the physiological negative-feedback regulation of post-ischemic T cell activation is weakened. Under normal conditions, engagement of CTLA-4 and PD-L1 limits excessive T cell responses by inhibiting costimulatory signaling and cytokine release. In the post-acute stroke phase, reduced expression of these checkpoints may permit sustained activation and interaction with microglia, contributing to prolonged neuroinflammation. This concept is supported by prior studies linking impaired CTLA-4 or PD-1/PD-L1 signaling to post-stroke immune dysregulation and poor neurological recovery [
7,
38].
The chronic activation of T cells fades at 3 months post stroke. The lack of negative feedback loops of CTLA-4 and PD-L1 could emerge as key mechanisms and should be studied in future studies including cognitive readouts. DNTs might influence microglia’s activation state through CX3CR1; thus, they inhibit the M2 state ex vivo, which might contribute to cerebral inflammation post-stroke. These mechanisms remain hypothetical, and confirmation in larger longitudinal and functional studies will be required to establish causal links.
The delayed normalization of CTLA-4 and PD-L1 expression may represent a potential therapeutic window within which to restore immune homeostasis after stroke. Pharmacological activation of these checkpoint pathways could theoretically temper chronic neuroinflammation and deserves further preclinical evaluation.
Taken together, these observations suggest that disrupted CTLA-4 and PD-L1 signaling and altered DNT–microglia crosstalk may contribute to persistent T cell activation after stroke. Targeting such pathways could offer novel opportunities for post-stroke immunomodulation, pending validation in future mechanistic studies.