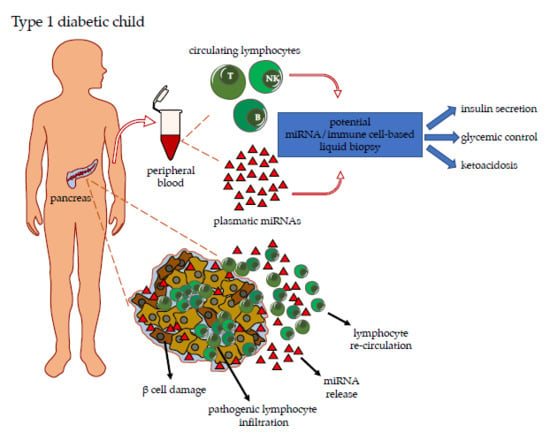
Blood Co-Circulating Extracellular microRNAs and Immune Cell Subsets Associate with Type 1 Diabetes Severity
Abstract
1. Introduction
2. Results
2.1. Plasmatic miRNAs Are Better Than Peripheral Blood Circulating Immune Cells in Discriminating Type 1 Diabetics from Healthy Children
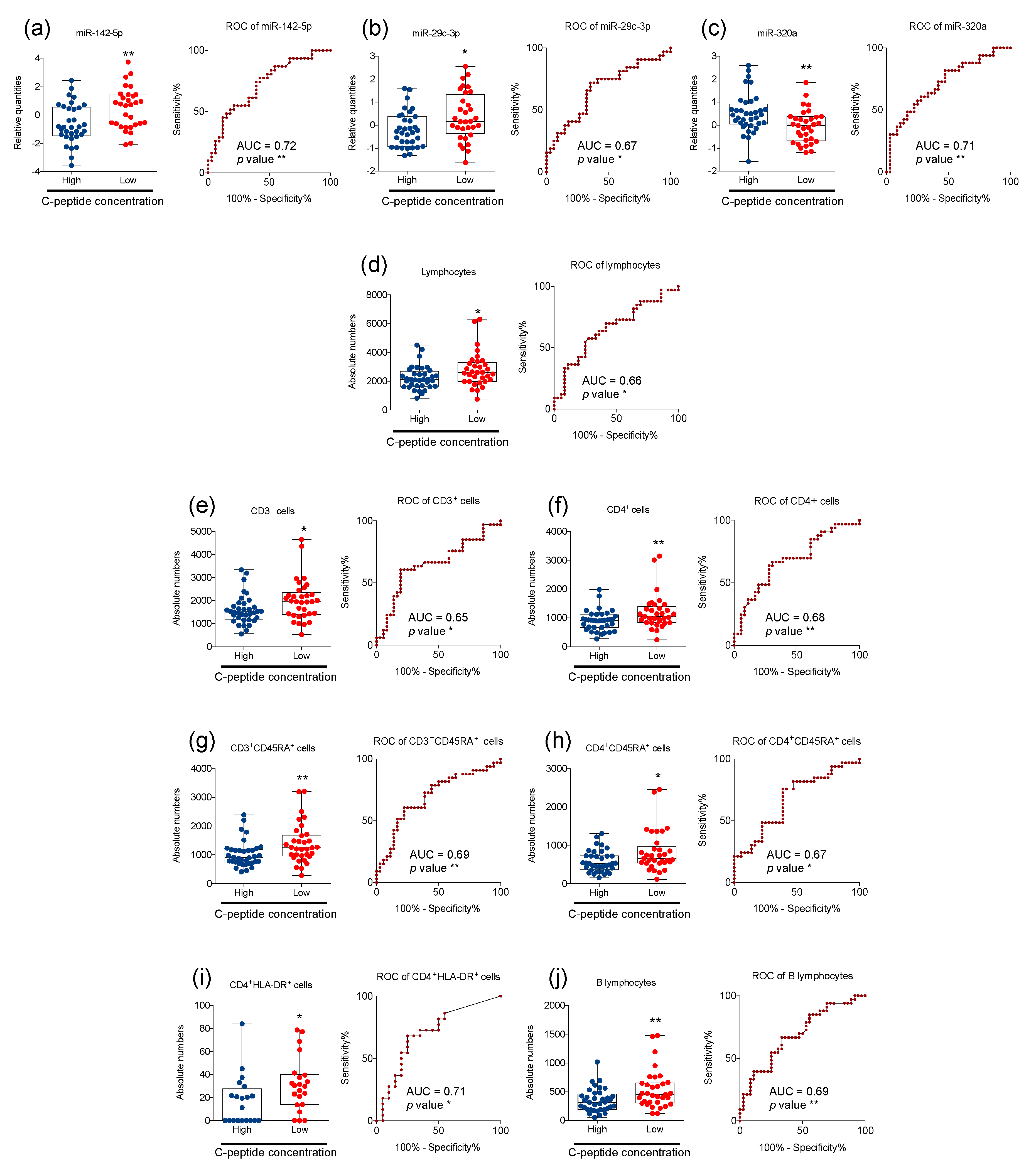
2.2. Significant Change in Plasmatic miRNAs and Peripheral Blood Circulating Immune Cells is Associated with Different Capability of Insulin Secretion in T1D Children at Diagnosis
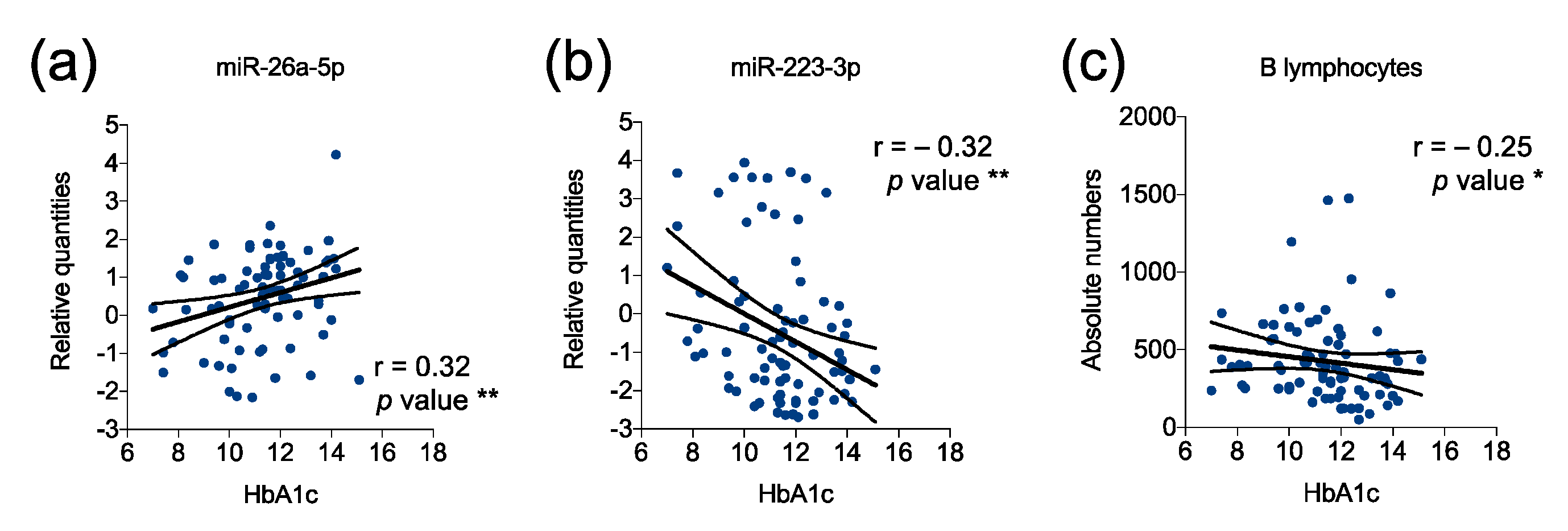
2.3. Correlation of Plasmatic miRNAs with Glycemic Control in Children with T1D at Diagnosis
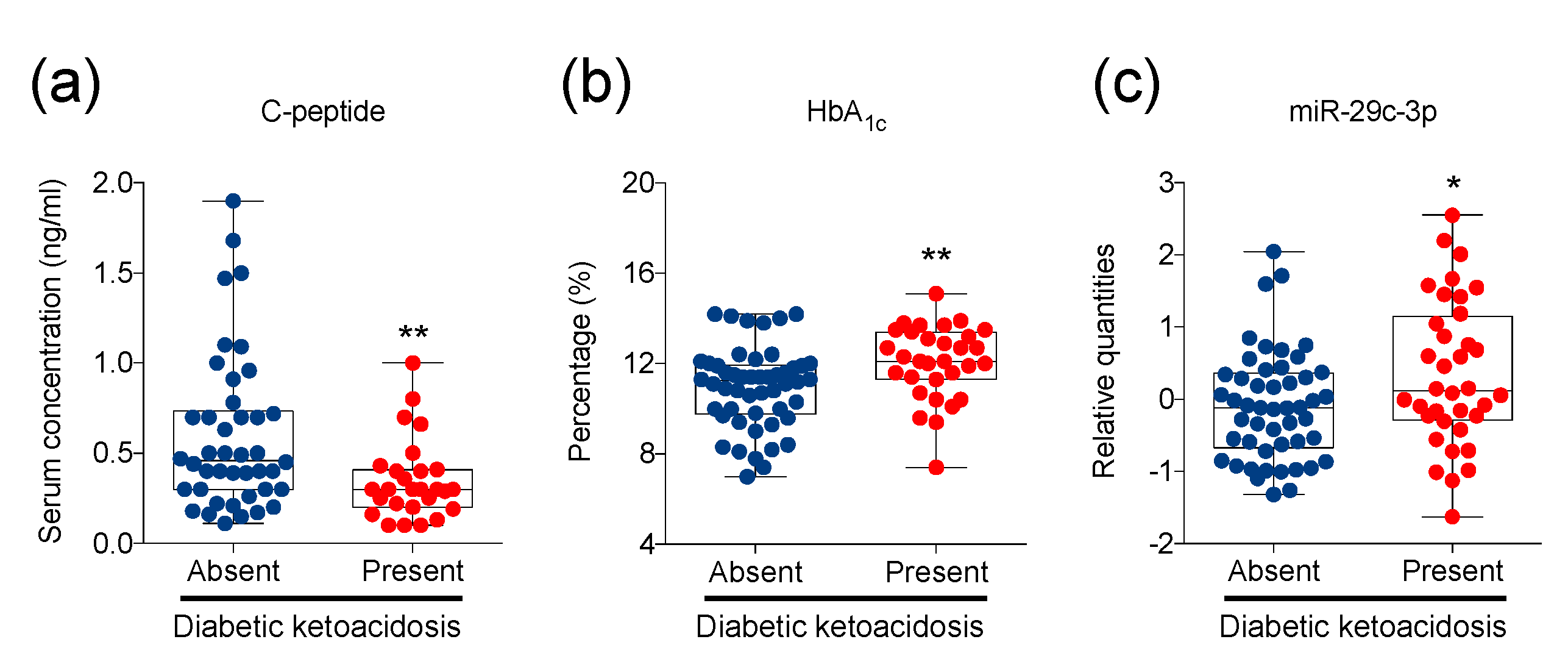
2.4. Plasmatic miR-29c-3p Is Significantly Associated with Diabetic Ketoacidosis in T1D Children at Diagnosis
2.5. Correlation Analysis Reveals a T1D-Specific Interconnection between Plasmatic miRNAs and Peripheral Blood Circulating Immune Cells
3. Discussion
4. Materials and Methods
4.1. Subjects
4.2. Laboratory Testing
4.3. Immunophenotypic Analysis
4.4. Plasmatic miRNA Quantification
4.5. MiRNA Target Gene Analysis
4.6. Statistics
4.7. Study Approval
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| miRNA | microRNAs |
| LNA | locked nucleic acid |
| cel-miR-39-3p | Caenorhabditis elegans miR-39 |
| ROC | receiver operating characteristics |
References
- Eisenbarth, G.S. Type I diabetes mellitus. A chronic autoimmune disease. N. Engl. J. Med. 1986, 314, 1360–1368. [Google Scholar] [CrossRef] [PubMed]
- Gale, E.A. Type 1 diabetes in the young: The harvest of sorrow goes on. Diabetologia 2005, 48, 1435–1438. [Google Scholar] [CrossRef] [PubMed]
- Moltchanova, E.V.; Schreier, N.; Lammi, N.; Karvonen, M. Seasonal variation of diagnosis of Type 1 diabetes mellitus in children worldwide. Diabet. Med. 2009, 26, 673–678. [Google Scholar] [CrossRef] [PubMed]
- Kolb, H.; Kolb-Bachofen, V.; Roep, B.O. Autoimmune versus inflammatory Type I diabetes: A controversy? Immunol. Today 1995, 16, 170–172. [Google Scholar] [CrossRef]
- DiMeglio, L.A.; Evans-Molina, C.; Oram, R.A. Type 1 diabetes. Lancet 2018, 391, 2449–2462. [Google Scholar] [CrossRef]
- Greenbaum, C.J.; Beam, C.A.; Boulware, D.; Gitelman, S.E.; Gottlieb, P.A.; Herold, K.C.; Lachin, J.M.; McGee, P.; Palmer, J.P.; Pescovitz, M.D.; et al. Fall in C-peptide during first 2 years from diagnosis: Evidence of at least two distinct phases from composite Type 1 diabetes trialnet data. Diabetes 2012, 61, 2066–2073. [Google Scholar] [CrossRef]
- Steffes, M.W.; Sibley, S.; Jackson, M.; Thomas, W. Beta-cell function and the development of diabetes-related complications in the diabetes control and complications trial. Diabetes Care 2003, 26, 832–836. [Google Scholar] [CrossRef]
- Fitas, A.L.; Martins, C.; Borrego, L.M.; Lopes, L.; Jorns, A.; Lenzen, S.; Limbert, C. Immune cell and cytokine patterns in children with Type 1 diabetes mellitus undergoing a remission phase: A longitudinal study. Pediatr. Diabetes 2018, 19, 963–971. [Google Scholar] [CrossRef]
- Klingensmith, G.J.; Tamborlane, W.V.; Wood, J.; Haller, M.J.; Silverstein, J.; Cengiz, E.; Shanmugham, S.; Kollman, C.; Wong-Jacobson, S.; Beck, R.W.; et al. Diabetic ketoacidosis at diabetes onset: Still an all too common threat in youth. J. Pediatr. 2013, 162, 330–334. [Google Scholar] [CrossRef]
- Rewers, A.; Klingensmith, G.; Davis, C.; Petitti, D.B.; Pihoker, C.; Rodriguez, B.; Schwartz, I.D.; Imperatore, G.; Williams, D.; Dolan, L.M.; et al. Presence of diabetic ketoacidosis at diagnosis of diabetes mellitus in youth: The search for diabetes in youth study. Pediatrics 2008, 121, e1258–e1266. [Google Scholar] [CrossRef]
- Komulainen, J.; Lounamaa, R.; Knip, M.; Kaprio, E.A.; Akerblom, H.K. Ketoacidosis at the diagnosis of Type 1 (insulin dependent) diabetes mellitus is related to poor residual beta cell function. Childhood diabetes in finland study group. Arch. Dis. Child. 1996, 75, 410–415. [Google Scholar] [CrossRef]
- Mortensen, H.B.; Swift, P.G.; Holl, R.W.; Hougaard, P.; Hansen, L.; Bjoerndalen, H.; de Beaufort, C.E.; Knip, M.; Hvidoere Study Group on Childhood, D. Multinational study in children and adolescents with newly diagnosed Type 1 diabetes: Association of age, ketoacidosis, HLA status, and autoantibodies on residual beta-cell function and glycemic control 12 months after diagnosis. Pediatr. Diabetes 2010, 11, 218–226. [Google Scholar] [CrossRef]
- Chen, X.; Makala, L.H.; Jin, Y.; Hopkins, D.; Muir, A.; Garge, N.; Podolsky, R.H.; She, J.X. Type 1 diabetes patients have significantly lower frequency of plasmacytoid dendritic cells in the peripheral blood. Clin. Immunol. 2008, 129, 413–418. [Google Scholar] [CrossRef] [PubMed]
- Deng, C.; Xiang, Y.; Tan, T.; Ren, Z.; Cao, C.; Huang, G.; Wen, L.; Zhou, Z. Altered peripheral B-lymphocyte subsets in Type 1 diabetes and latent autoimmune diabetes in adults. Diabetes Care 2016, 39, 434–440. [Google Scholar] [CrossRef] [PubMed]
- Nieminen, J.K.; Vakkila, J.; Salo, H.M.; Ekstrom, N.; Harkonen, T.; Ilonen, J.; Knip, M.; Vaarala, O. Altered phenotype of peripheral blood dendritic cells in pediatric Type 1 diabetes. Diabetes Care 2012, 35, 2303–2310. [Google Scholar] [CrossRef] [PubMed]
- Qin, H.; Lee, I.F.; Panagiotopoulos, C.; Wang, X.; Chu, A.D.; Utz, P.J.; Priatel, J.J.; Tan, R. Natural killer cells from children with Type 1 diabetes have defects in NKG2D-dependent function and signaling. Diabetes 2011, 60, 857–866. [Google Scholar] [CrossRef]
- Oras, A.; Peet, A.; Giese, T.; Tillmann, V.; Uibo, R. A study of 51 subtypes of peripheral blood immune cells in newly diagnosed young Type 1 diabetes patients. Clin. Exp. Immunol. 2019, 198, 57–70. [Google Scholar] [CrossRef]
- Galgani, M.; Nugnes, R.; Bruzzese, D.; Perna, F.; De Rosa, V.; Procaccini, C.; Mozzillo, E.; Cilio, C.M.; Elding Larsson, H.; Lernmark, A.; et al. Meta-immunological profiling of children with Type 1 diabetes identifies new biomarkers to monitor disease progression. Diabetes 2013, 62, 2481–2491. [Google Scholar] [CrossRef]
- Osipova, J.; Fischer, D.C.; Dangwal, S.; Volkmann, I.; Widera, C.; Schwarz, K.; Lorenzen, J.M.; Schreiver, C.; Jacoby, U.; Heimhalt, M.; et al. Diabetes-associated microRNAs in pediatric patients with Type 1 diabetes mellitus: A cross-sectional cohort study. J. Clin. Endocrinol. Metab. 2014, 99, E1661–E1665. [Google Scholar] [CrossRef]
- Bijkerk, R.; Duijs, J.M.; Khairoun, M.; Ter Horst, C.J.; van der Pol, P.; Mallat, M.J.; Rotmans, J.I.; de Vries, A.P.; de Koning, E.J.; de Fijter, J.W.; et al. Circulating microRNAs associate with diabetic nephropathy and systemic microvascular damage and normalize after simultaneous pancreas-kidney transplantation. Am. J. Transplant. 2015, 15, 1081–1090. [Google Scholar] [CrossRef]
- Zampetaki, A.; Willeit, P.; Burr, S.; Yin, X.; Langley, S.R.; Kiechl, S.; Klein, R.; Rossing, P.; Chaturvedi, N.; Mayr, M. Angiogenic microRNAs linked to incidence and progression of diabetic retinopathy in Type 1 diabetes. Diabetes 2016, 65, 216–227. [Google Scholar] [CrossRef] [PubMed]
- Joglekar, M.V.; Januszewski, A.S.; Jenkins, A.J.; Hardikar, A.A. Circulating microRNA biomarkers of diabetic retinopathy. Diabetes 2016, 65, 22–24. [Google Scholar] [CrossRef] [PubMed]
- Marchand, L.; Jalabert, A.; Meugnier, E.; Van den Hende, K.; Fabien, N.; Nicolino, M.; Madec, A.M.; Thivolet, C.; Rome, S. MiRNA-375 a sensor of glucotoxicity is altered in the serum of children with newly diagnosed Type 1 diabetes. J. Diabetes Res. 2016, 2016, 1869082. [Google Scholar] [CrossRef] [PubMed]
- Barutta, F.; Bruno, G.; Matullo, G.; Chaturvedi, N.; Grimaldi, S.; Schalkwijk, C.; Stehouwer, C.D.; Fuller, J.H.; Gruden, G. MicroRNA-126 and micro-/macrovascular complications of Type 1 diabetes in the EURODIAB prospective complications study. Acta Diabetol. 2017, 54, 133–139. [Google Scholar] [CrossRef] [PubMed]
- Samandari, N.; Mirza, A.H.; Nielsen, L.B.; Kaur, S.; Hougaard, P.; Fredheim, S.; Mortensen, H.B.; Pociot, F. Circulating microRNA levels predict residual beta cell function and glycaemic control in children with Type 1 diabetes mellitus. Diabetologia 2017, 60, 354–363. [Google Scholar] [CrossRef] [PubMed]
- Erener, S.; Marwaha, A.; Tan, R.; Panagiotopoulos, C.; Kieffer, T.J. Profiling of circulating microRNAs in children with recent onset of Type 1 diabetes. JCI Insight 2017, 2, e89656. [Google Scholar] [CrossRef]
- Lakhter, A.J.; Pratt, R.E.; Moore, R.E.; Doucette, K.K.; Maier, B.F.; DiMeglio, L.A.; Sims, E.K. Beta cell extracellular vesicle miR-21-5p cargo is increased in response to inflammatory cytokines and serves as a biomarker of Type 1 diabetes. Diabetologia 2018, 61, 1124–1134. [Google Scholar] [CrossRef]
- Satake, E.; Pezzolesi, M.G.; Md Dom, Z.I.; Smiles, A.M.; Niewczas, M.A.; Krolewski, A.S. Circulating miRNA profiles associated with hyperglycemia in patients with Type 1 diabetes. Diabetes 2018, 67, 1013–1023. [Google Scholar] [CrossRef]
- Assmann, T.S.; Recamonde-Mendoza, M.; Costa, A.R.; Punales, M.; Tschiedel, B.; Canani, L.H.; Bauer, A.C.; Crispim, D. Circulating miRNAs in diabetic kidney disease: Case-control study and in silico analyses. Acta Diabetol. 2019, 56, 55–65. [Google Scholar] [CrossRef]
- Malachowska, B.; Wyka, K.; Nowicka, Z.; Bartlomiejczyk, M.A.; Mlynarski, W.; Fendler, W. Temporal dynamics of serum let-7g expression mirror the decline of residual beta-cell function in longitudinal observation of children with Type 1 diabetes. Pediatr. Diabetes 2018, 19, 1407–1415. [Google Scholar] [CrossRef]
- Jaeger, A.; Zollinger, L.; Saely, C.H.; Muendlein, A.; Evangelakos, I.; Nasias, D.; Charizopoulou, N.; Schofield, J.D.; Othman, A.; Soran, H.; et al. Circulating microRNAs-192 and -194 are associated with the presence and incidence of diabetes mellitus. Sci. Rep. 2018, 8, 14274. [Google Scholar] [CrossRef] [PubMed]
- Bartel, D.P. Metazoan microRNAs. Cell 2018, 173, 20–51. [Google Scholar] [CrossRef] [PubMed]
- Rottiers, V.; Naar, A.M. MicroRNAs in metabolism and metabolic disorders. Nat. Rev. Mol. Cell Biol. 2012, 13, 239–250. [Google Scholar] [CrossRef] [PubMed]
- Friedman, R.C.; Farh, K.K.; Burge, C.B.; Bartel, D.P. Most mammalian mRNAs are conserved targets of microRNAs. Genome Res. 2009, 19, 92–105. [Google Scholar] [CrossRef]
- Guay, C.; Regazzi, R. Exosomes as new players in metabolic organ cross-talk. Diabetes Obes. Metab. 2017, 19, 137–146. [Google Scholar] [CrossRef]
- Turchinovich, A.; Weiz, L.; Langheinz, A.; Burwinkel, B. Characterization of extracellular circulating microRNA. Nucleic Acids Res. 2011, 39, 7223–7233. [Google Scholar] [CrossRef]
- Chen, X.; Ba, Y.; Ma, L.; Cai, X.; Yin, Y.; Wang, K.; Guo, J.; Zhang, Y.; Chen, J.; Guo, X.; et al. Characterization of microRNAs in serum: A novel class of biomarkers for diagnosis of cancer and other diseases. Cell Res. 2008, 18, 997–1006. [Google Scholar] [CrossRef]
- Arroyo, J.D.; Chevillet, J.R.; Kroh, E.M.; Ruf, I.K.; Pritchard, C.C.; Gibson, D.F.; Mitchell, P.S.; Bennett, C.F.; Pogosova-Agadjanyan, E.L.; Stirewalt, D.L.; et al. Argonaute2 complexes carry a population of circulating microRNAs independent of vesicles in human plasma. Proc. Natl. Acad. Sci. USA 2011, 108, 5003–5008. [Google Scholar] [CrossRef]
- Mitchell, P.S.; Parkin, R.K.; Kroh, E.M.; Fritz, B.R.; Wyman, S.K.; Pogosova-Agadjanyan, E.L.; Peterson, A.; Noteboom, J.; O’Briant, K.C.; Allen, A.; et al. Circulating microRNAs as stable blood-based markers for cancer detection. Proc. Natl. Acad. Sci. USA 2008, 105, 10513–10518. [Google Scholar] [CrossRef]
- Thomou, T.; Mori, M.A.; Dreyfuss, J.M.; Konishi, M.; Sakaguchi, M.; Wolfrum, C.; Rao, T.N.; Winnay, J.N.; Garcia-Martin, R.; Grinspoon, S.K.; et al. Adipose-derived circulating miRNAs regulate gene expression in other tissues. Nature 2017, 542, 450–455. [Google Scholar] [CrossRef]
- Sebastiani, G.; Nigi, L.; Grieco, G.E.; Mancarella, F.; Ventriglia, G.; Dotta, F. Circulating microRNAs and diabetes mellitus: A novel tool for disease prediction, diagnosis, and staging? J. Endocrinol. Invest. 2017, 40, 591–610. [Google Scholar] [CrossRef] [PubMed]
- International Expert, C. International expert committee report on the role of the A1C assay in the diagnosis of diabetes. Diabetes Care 2009, 32, 1327–1334. [Google Scholar] [CrossRef] [PubMed]
- de Candia, P.; Torri, A.; Gorletta, T.; Fedeli, M.; Bulgheroni, E.; Cheroni, C.; Marabita, F.; Crosti, M.; Moro, M.; Pariani, E.; et al. Intracellular modulation, extracellular disposal and serum increase of miR-150 mark lymphocyte activation. PLoS ONE 2013, 8, e75348. [Google Scholar] [CrossRef] [PubMed]
- Vecchio, F.; Messina, G.; Giovenzana, A.; Petrelli, A. New evidence of exocrine pancreatopathy in pre-symptomatic and symptomatic Type 1 diabetes. Curr. Diabetes Rep. 2019, 19, 92. [Google Scholar] [CrossRef]
- Alexandre-Heymann, L.; Mallone, R.; Boitard, C.; Scharfmann, R.; Larger, E. Structure and function of the exocrine pancreas in patients with Type 1 diabetes. Rev. Endocr. Metab. Disord. 2019, 20, 129–149. [Google Scholar] [CrossRef]
- Tschen, S.I.; Zeng, C.; Field, L.; Dhawan, S.; Bhushan, A.; Georgia, S. Cyclin D2 is sufficient to drive beta cell self-renewal and regeneration. Cell cycle 2017, 16, 2183–2191. [Google Scholar] [CrossRef]
- Thompson, P.J.; Shah, A.; Ntranos, V.; Van Gool, F.; Atkinson, M.; Bhushan, A. Targeted elimination of senescent beta cells prevents Type 1 diabetes. Cell Metab. 2019, 29, 1045–1060.e10. [Google Scholar] [CrossRef]
- de Candia, P.; Torri, A.; Pagani, M.; Abrignani, S. Serum microRNAs as biomarkers of human lymphocyte activation in health and disease. Front. Immunol. 2014, 5, 43. [Google Scholar] [CrossRef]
- de Candia, P.; Torri, A.; Fedeli, M.; Vigano, V.; Carpi, D.; Gorletta, T.; Casorati, G.; Pagani, M.; Dellabona, P.; Abrignani, S. The circulating microRNome demonstrates distinct lymphocyte subset-dependent signatures. Eur. J. Immunol. 2016, 46, 725–731. [Google Scholar] [CrossRef]
- Torri, A.; Carpi, D.; Bulgheroni, E.; Crosti, M.C.; Moro, M.; Gruarin, P.; Rossi, R.L.; Rossetti, G.; Di Vizio, D.; Hoxha, M.; et al. Extracellular microRNA signature of human helper T cell subsets in health and autoimmunity. J. Biol. Chem. 2017, 292, 2903–2915. [Google Scholar] [CrossRef]
- Frost, R.J.; Olson, E.N. Control of glucose homeostasis and insulin sensitivity by the let-7 family of microRNAs. Proc. Natl. Acad. Sci. USA 2011, 108, 21075–21080. [Google Scholar] [CrossRef] [PubMed]
- Zhu, H.; Shyh-Chang, N.; Segre, A.V.; Shinoda, G.; Shah, S.P.; Einhorn, W.S.; Takeuchi, A.; Engreitz, J.M.; Hagan, J.P.; Kharas, M.G.; et al. The Lin28/let-7 axis regulates glucose metabolism. Cell 2011, 147, 81–94. [Google Scholar] [CrossRef] [PubMed]
- Pobezinsky, L.A.; Etzensperger, R.; Jeurling, S.; Alag, A.; Kadakia, T.; McCaughtry, T.M.; Kimura, M.Y.; Sharrow, S.O.; Guinter, T.I.; Feigenbaum, L.; et al. Let-7 microRNAs target the lineage-specific transcription factor PLZF to regulate terminal NKT cell differentiation and effector function. Nat. Immunol. 2015, 16, 517–524. [Google Scholar] [CrossRef]
- Almanza, G.; Fernandez, A.; Volinia, S.; Cortez-Gonzalez, X.; Croce, C.M.; Zanetti, M. Selected microRNAs define cell fate determination of murine central memory CD8 T cells. PLoS ONE 2010, 5, e11243. [Google Scholar] [CrossRef]
- Swaminathan, S.; Suzuki, K.; Seddiki, N.; Kaplan, W.; Cowley, M.J.; Hood, C.L.; Clancy, J.L.; Murray, D.D.; Mendez, C.; Gelgor, L.; et al. Differential regulation of the let-7 family of microRNAs in CD4+ T cells alters IL-10 expression. J. Immunol. 2012, 188, 6238–6246. [Google Scholar] [CrossRef]
- Marcais, A.; Blevins, R.; Graumann, J.; Feytout, A.; Dharmalingam, G.; Carroll, T.; Amado, I.F.; Bruno, L.; Lee, K.; Walzer, T.; et al. MicroRNA-mediated regulation of mTOR complex components facilitates discrimination between activation and anergy in CD4 T cells. J. Exp. Med. 2014, 211, 2281–2295. [Google Scholar] [CrossRef] [PubMed]
- Okoye, I.S.; Coomes, S.M.; Pelly, V.S.; Czieso, S.; Papayannopoulos, V.; Tolmachova, T.; Seabra, M.C.; Wilson, M.S. MicroRNA-containing T-regulatory-cell-derived exosomes suppress pathogenic T helper 1 cells. Immunity 2014, 41, 89–103. [Google Scholar] [CrossRef]
- Kimura, K.; Hohjoh, H.; Fukuoka, M.; Sato, W.; Oki, S.; Tomi, C.; Yamaguchi, H.; Kondo, T.; Takahashi, R.; Yamamura, T. Circulating exosomes suppress the induction of regulatory T cells via let-7i in multiple sclerosis. Nat. Commun. 2018, 9, 17. [Google Scholar] [CrossRef]
- Esteves, J.V.; Yonamine, C.Y.; Pinto-Junior, D.C.; Gerlinger-Romero, F.; Enguita, F.J.; Machado, U.F. Diabetes modulates microRNAs 29b-3p, 29c-3p, 199a-5p and 532-3p expression in muscle: possible role in GLUT4 and HK2 repression. Front. Endocrinol. 2018, 9, 536. [Google Scholar] [CrossRef]
- Esteves, J.V.; Enguita, F.J.; Machado, U.F. MicroRNAs-mediated regulation of skeletal muscle GLUT4 expression and translocation in insulin resistance. J. Diabetes Res. 2017, 2017, 7267910. [Google Scholar] [CrossRef]
- Torella, D.; Iaconetti, C.; Tarallo, R.; Marino, F.; Giurato, G.; Veneziano, C.; Aquila, I.; Scalise, M.; Mancuso, T.; Cianflone, E.; et al. MiRNA regulation of the hyperproliferative phenotype of vascular smooth muscle cells in diabetes. Diabetes 2018, 67, 2554–2568. [Google Scholar] [CrossRef] [PubMed]
- Dooley, J.; Garcia-Perez, J.E.; Sreenivasan, J.; Schlenner, S.M.; Vangoitsenhoven, R.; Papadopoulou, A.S.; Tian, L.; Schonefeldt, S.; Serneels, L.; Deroose, C.; et al. The microRNA-29 family dictates the balance between homeostatic and pathological glucose handling in diabetes and obesity. Diabetes 2016, 65, 53–61. [Google Scholar] [CrossRef] [PubMed]
- Medina, C.O.; Nagy, N.; Bollyky, P.L. Extracellular matrix and the maintenance and loss of peripheral immune tolerance in autoimmune insulitis. Curr. Opin. Immunol. 2018, 55, 22–30. [Google Scholar] [CrossRef] [PubMed]
- Bogdani, M.; Korpos, E.; Simeonovic, C.J.; Parish, C.R.; Sorokin, L.; Wight, T.N. Extracellular matrix components in the pathogenesis of Type 1 diabetes. Curr. Diab. Rep. 2014, 14, 552. [Google Scholar] [CrossRef] [PubMed]
- Craig, M.E.; Hattersley, A.; Donaghue, K.C. Definition, epidemiology and classification of diabetes in children and adolescents. Pediatr. Diabetes 2009, 10, 3–12. [Google Scholar] [CrossRef] [PubMed]
- Chou, C.H.; Shrestha, S.; Yang, C.D.; Chang, N.W.; Lin, Y.L.; Liao, K.W.; Huang, W.C.; Sun, T.H.; Tu, S.J.; Lee, W.H.; et al. MiRTarBase update 2018: A resource for experimentally validated microRNA-target interactions. Nucleic Acids Res. 2018, 46, D296–D302. [Google Scholar] [CrossRef] [PubMed]
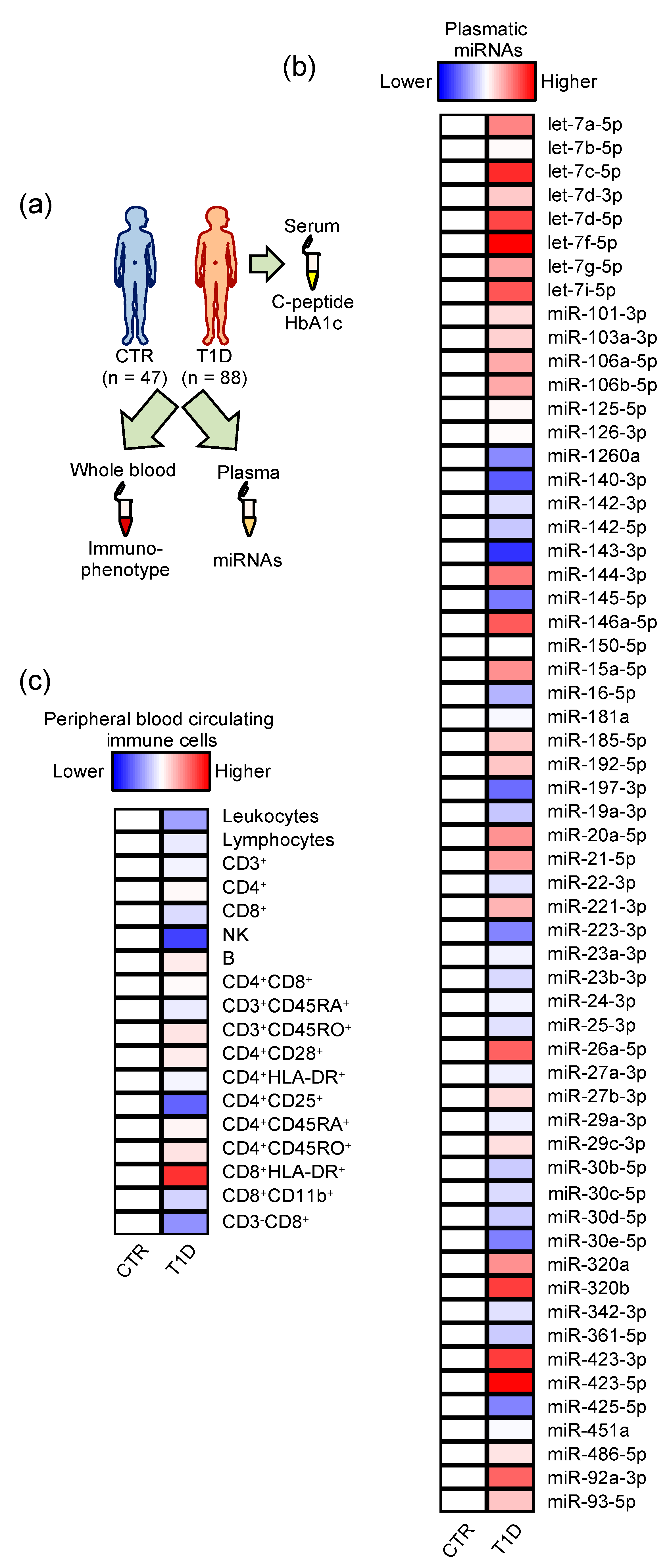

| Baseline Characteristics | CTR | T1D | p value |
|---|---|---|---|
| Numbers of subjects | 47 | 88 | - |
| Age (years) | 8.44 ± 3.25 | 8.97 ± 3.68 | n.s. |
| Gender (%M) | 45.65 | 54.55 | n.s.(§) |
| Body mass index (kg/m2) | 19.72 ± 4.82 | 17.43 ± 3.44 | * |
| C-peptide (ng/mL) | n.a. | 0.50 ± 0.39 | - |
| Ketoacidosis at diagnosis (yes/no/n.a.) | n.a. | 35/52/1 | - |
| Glycated hemoglobin (%) | n.a. | 11.34 ± 1.79 | - |
| Insulin dose (IU/kg/day) | n.a. | 0.65 ± 0.30 | - |
| Other autoimmune disorders (yes/no) | 0/47 | 15/73 | *(§) |
| Variable | CTR (Mean ± SD) | T1D (Mean ± SD) | Adj. OR (95% CI) for One Unit of Parameter Increase | p Value | Adjusted for | AUC |
|---|---|---|---|---|---|---|
| let-7c-5p* | 0 ± 0.73 | 0.5 ± 0.91 | 2.81 (1.37–5.74) | 0.0046 | BMI | 0.769 |
| let-7d-5p* | 0 ± 0.83 | 0.44 ± 0.9 | 2.49 (1.19–5.19) | 0.0150 | BMI | 0.732 |
| let-7f-5p* | 0 ± 0.85 | 0.81 ± 0.88 | 4.15 (1.82–9.48) | 0.0007 | BMI | 0.845 |
| let-7i-5p* | 0 ± 0.94 | 0.41 ± 0.61 | 3.05 (1.56–5.98) | 0.0012 | BMI* | 0.770 |
| miR-140-3p* | 0 ± 1 | −0.57 ± 0.93 | 0.36 (0.22–0.62) | 0.0001 | BMI* | 0.793 |
| miR-143-3p* | 0 ± 0.8 | −0.74 ± 0.86 | 0.41 (0.20–0.85) | 0.0170 | BMI | 0.700 |
| miR-146a-5p* | 0 ± 0.94 | 0.41 ± 0.84 | 2.51 (1.47–4.28) | 0.0008 | BMI* | 0.742 |
| miR-423-3p* | 0 ± 0.72 | 0.46 ± 0.83 | 2.27 (1.15–4.47) | 0.0181 | BMI | 0.729 |
| miR-423-5p* | 0 ± 1.1 | 0.56 ± 1.01 | 2.24 (1.38–3.65) | 0.0011 | BMI* | 0.763 |
| Leukocytes | 6472.79 ± 2624.42 | 5233.22 ± 1818.89 | 0.75 (0.61–0.93) ° | 0.0080 | BMI | 0.698 |
| NK | 286.39 ± 210.22 | 179.23 ± 108.48 | 0.68 (0.49–0.93) § | 0.0155 | BMI* | 0.693 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Garavelli, S.; Bruzzaniti, S.; Tagliabue, E.; Prattichizzo, F.; Di Silvestre, D.; Perna, F.; La Sala, L.; Ceriello, A.; Mozzillo, E.; Fattorusso, V.; et al. Blood Co-Circulating Extracellular microRNAs and Immune Cell Subsets Associate with Type 1 Diabetes Severity. Int. J. Mol. Sci. 2020, 21, 477. https://doi.org/10.3390/ijms21020477
Garavelli S, Bruzzaniti S, Tagliabue E, Prattichizzo F, Di Silvestre D, Perna F, La Sala L, Ceriello A, Mozzillo E, Fattorusso V, et al. Blood Co-Circulating Extracellular microRNAs and Immune Cell Subsets Associate with Type 1 Diabetes Severity. International Journal of Molecular Sciences. 2020; 21(2):477. https://doi.org/10.3390/ijms21020477
Chicago/Turabian StyleGaravelli, Silvia, Sara Bruzzaniti, Elena Tagliabue, Francesco Prattichizzo, Dario Di Silvestre, Francesco Perna, Lucia La Sala, Antonio Ceriello, Enza Mozzillo, Valentina Fattorusso, and et al. 2020. "Blood Co-Circulating Extracellular microRNAs and Immune Cell Subsets Associate with Type 1 Diabetes Severity" International Journal of Molecular Sciences 21, no. 2: 477. https://doi.org/10.3390/ijms21020477
APA StyleGaravelli, S., Bruzzaniti, S., Tagliabue, E., Prattichizzo, F., Di Silvestre, D., Perna, F., La Sala, L., Ceriello, A., Mozzillo, E., Fattorusso, V., Mauri, P., Puca, A. A., Franzese, A., Matarese, G., Galgani, M., & de Candia, P. (2020). Blood Co-Circulating Extracellular microRNAs and Immune Cell Subsets Associate with Type 1 Diabetes Severity. International Journal of Molecular Sciences, 21(2), 477. https://doi.org/10.3390/ijms21020477