Usefulness of Brain Positron Emission Tomography with Different Tracers in the Evaluation of Patients with Idiopathic Normal Pressure Hydrocephalous
Abstract
1. Introduction
2. Results
2.1. Amyloid PET Imaging in iNPH
2.1.1. Correlation between Radiotracer Uptake and Biopsy Samples in iNPH
2.1.2. Uptake in iNPH before Treatment
2.1.3. Role in Prediction of Shunt Response in iNPH
2.2. Perfusion PET Imaging in iNPH
2.2.1. Perfusion Changes before Treatment in iNPH
2.2.2. Perfusion Changes during Infusion Test in iNPH
2.2.3. Perfusion Changes after Shunt Placement in iNPH
2.3. Glucose Metabolism PET Imaging in iNPH
2.3.1. Metabolic Changes at Baseline in iNPH
2.3.2. Metabolic Changes after Treatment in iNPH
2.4. Dopaminergic PET Imaging in iNPH
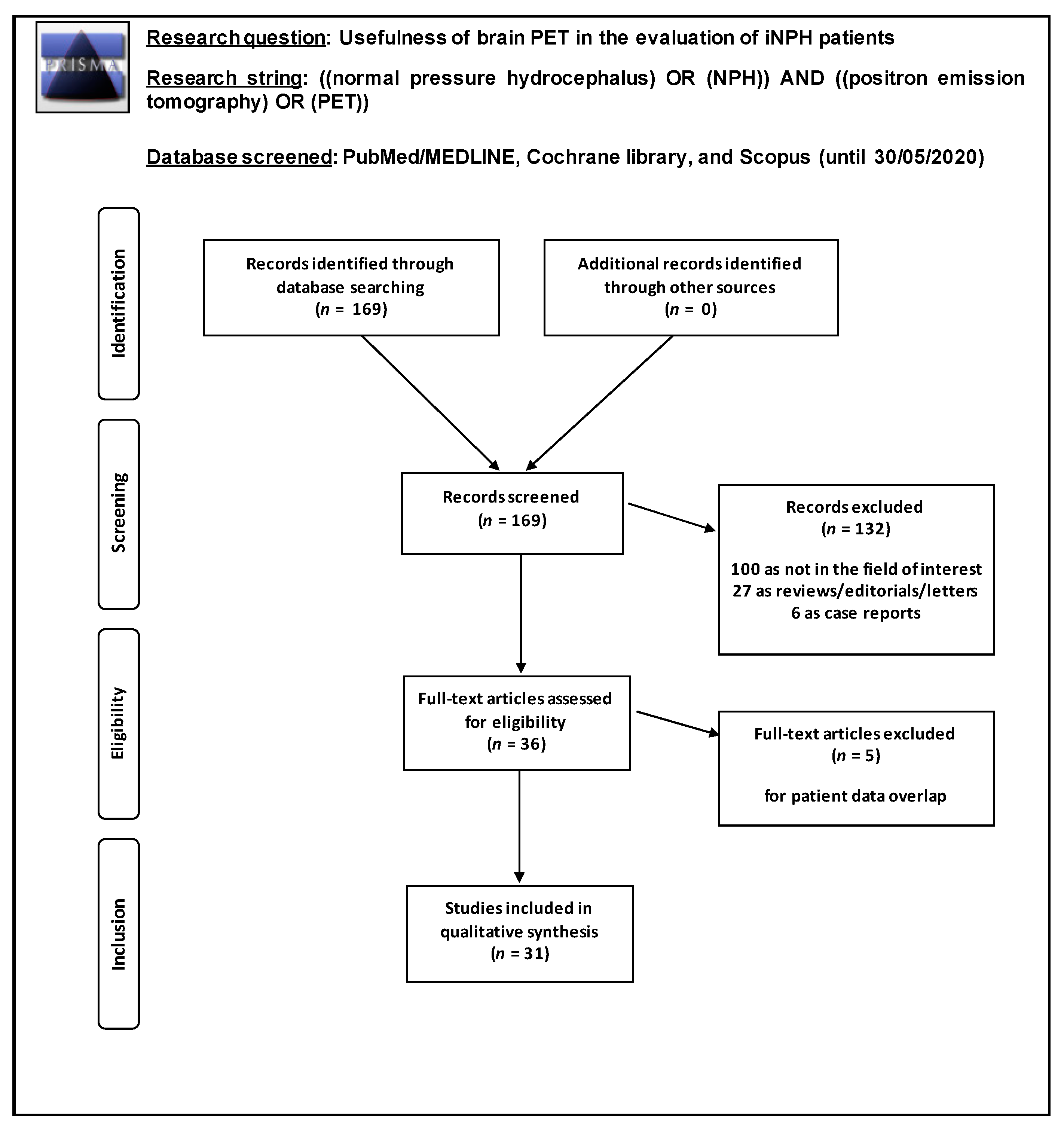
3. Materials and Methods
3.1. Search Strategy
3.2. Study Selection
3.3. Data Abstraction
4. Summary
- PET with amyloid tracers, revealing AD in 20–57% of suspected iNPH patients, could be useful in prediction of surgical outcome;
- PET with perfusion tracers showed a global decreased CBF and regional reduction of CBF in basal ganglia in iNPH patients; preoperative perfusion parameters could predict surgical outcome;
- PET with [18F]FDG showed a global reduction of glucose metabolism without a specific cortical pattern and a hypometabolism in basal ganglia in iNPH patients; [18F]FDG PET may identify a coexisting neurodegenerative disease, helping in the patient selection for surgery; postsurgery increase in glucose metabolism was associated with clinical improvement.
- Dopaminergic PET imaging showed a postsynaptic D2 receptor reduction and a striatal upregulation of D2 receptors after treatment, associated with clinical improvement.
5. Limitations
6. Future Perspectives
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Andersson, J.; Rosell, M.; Kockum, K.; Söderström, L.; Laurell, K. Challenges in diagnosing normal pressure hydrocephalus: Evaluation of the diagnostic guidelines. eNeurologicalSci 2017, 7, 27–31. [Google Scholar] [CrossRef]
- Giordan, E.; Palandri, G.; Lanzino, G.; Murad, M.H.; Elder, B.D. Outcomes and complications of different surgical treatments for idiopathic normal pressure hydrocephalus: A systematic review and meta-analysis. J. Neurosurg. 2019, 131, 1024–1036. [Google Scholar] [CrossRef]
- Anile, C.; De Bonis, P.; Albanese, A.; Di Chirico, A.; Mangiola, A.; Petrella, G.; Santini, P. Selection of patients with idiopathic normal-pressure hydrocephalus for shunt placement: A single-institution experience. J. Neurosurg. 2010, 113, 64–73. [Google Scholar] [CrossRef]
- Eide, P.K. The correlation between pulsatile intracranial pressure and indices of intracranial pressure-volume reserve capacity: Results from ventricular infusion testing. J. Neurosurg. 2016, 125, 1493–1503. [Google Scholar] [CrossRef]
- Katzman, R.; Hussey, F. A simple constant-infusion manometric test for measurement of CSF absorption: I. Rationale and method. Neurology 1970, 20, 534. [Google Scholar] [CrossRef]
- Hussey, F.; Schanzer, B.; Katzman, R. A simple constant-infusion manometric test for measurement of CSF absorption: II. Clinical studies. Neurology 1970, 20, 665. [Google Scholar] [CrossRef]
- Raneri, F.; Zella, M.A.S.; Di Cristofori, A.; Zarino, B.; Pluderi, M.; Spagnoli, D. Supplementary Tests in Idiopathic Normal Pressure Hydrocephalus: A Single-Center Experience with a Combined Lumbar Infusion Test and Tap Test. World Neurosurg. 2017, 100, 567–574. [Google Scholar] [CrossRef]
- Allali, G.; Laidet, M.; Armand, S.; Assal, F. Brain comorbidities in normal pressure hydrocephalus. Eur. J. Neurol. 2018, 25, 542–548. [Google Scholar] [CrossRef]
- Macki, M.; Mahajan, A.; Shatz, R.; Air, E.L.; Novikova, M.; Fakih, M.; Elmenini, J.; Kaur, M.; Bouchard, K.R.; Funk, B.A.; et al. Prevalence of Alternative Diagnoses and Implications for Management in Idiopathic Normal Pressure Hydrocephalus Patients. Neurosurgery 2020, nyaa199. [Google Scholar] [CrossRef]
- Broggi, M.; Redaelli, V.; Tringali, G.; Restelli, F.; Romito, L.; Schiavolin, S.; Tagliavini, F.; Broggi, G. Normal Pressure Hydrocephalus and Parkinsonism: Preliminary Data on Neurosurgical and Neurological Treatment. World Neurosurg. 2016, 90, 348–356. [Google Scholar] [CrossRef]
- Bräutigam, K.; Vakis, A.; Tsitsipanis, C. Pathogenesis of idiopathic Normal Pressure Hydrocephalus: A review of knowledge. J. Clin. Neurosci. 2019, 61, 10–13. [Google Scholar] [CrossRef]
- Lalou, A.D.; Czosnyka, M.; Donnelly, J.; Pickard, J.D.; FMedSci; Nabbanja, E.; Keong, N.C.; Garnett, M.; Czosnyka, Z.H. Cerebral autoregulation, cerebrospinal fluid outflow resistance, and outcome following cerebrospinal fluid diversion in normal pressure hydrocephalus. J. Neurosurg. 2018, 130, 154–162. [Google Scholar] [CrossRef]
- Landau, S.M.; Thomas, B.A.; Thurfjell, L.; Schmidt, M.; Margolin, R.; Mintun, M.; Pontecorvo, M.; Baker, S.L.; Jagust, W.J.; the Alzheimer’s Disease Neuroimaging Initiative. Amyloid PET imaging in Alzheimer’s disease: A comparison of three radiotracers. Eur. J. Nucl. Med. Mol. Imaging 2014, 41, 1398–1407. [Google Scholar] [CrossRef]
- Hamilton, R.; Patel, S.; Lee, E.B.; Jackson, E.M.; Lopinto, J.; Arnold, S.E.; Clark, C.M.; Basil, A.; Shaw, L.M.; Xie, S.X.; et al. Lack of shunt response in suspected idiopathic normal pressure hydrocephalus with Alzheimer disease pathology. Ann. Neurol. 2010, 68, 535–540. [Google Scholar] [CrossRef]
- Leinonen, V.; Alafuzoff, I.; Aalto, S.; Suotunen, T.; Savolainen, S.; Na, K.; Rinne, J.; Jääskeläinen, J.E.; Soininen, H.; Rinne, J.O. Assessment of β-Amyloid in a Frontal Cortical Brain Biopsy Specimen and by Positron Emission Tomography With Carbon 11–Labeled Pittsburgh Compound B. Arch Neurol 2008, 65, 6. [Google Scholar]
- Leinonen, V.; Rauramaa, T.; Johansson, J.; Bottelbergs, A.; Tesseur, I.; van der Ark, P.; Pemberton, D.; Koivisto, A.M.; Jääskeläinen, J.E.; Hiltunen, M.; et al. S-[18F]THK-5117-PET and [11C]PIB-PET Imaging in Idiopathic Normal Pressure Hydrocephalus in Relation to Confirmed Amyloid-β Plaques and Tau in Brain Biopsies. J. Alzheimers Dis. 2018, 64, 171–179. [Google Scholar] [CrossRef]
- Wolk, D.A.; Grachev, I.D.; Buckley, C.; Kazi, H.; Grady, M.S.; Trojanowski, J.Q.; Hamilton, R.H.; Sherwin, P.; McLain, R.; Arnold, S.E. Association between in vivo fluorine 18-labeled flutemetamol amyloid positron emission tomography imaging and in vivo cerebral cortical histopathology. Arch. Neurol. 2011, 68, 1398–1403. [Google Scholar] [CrossRef]
- Wong, D.F.; Moghekar, A.R.; Rigamonti, D.; Brašić, J.R.; Rousset, O.; Willis, W.; Buckley, C.; Smith, A.; Gok, B.; Sherwin, P.; et al. An In Vivo Evaluation of Cerebral Cortical Amyloid with [18F]Flutemetamol Using Positron Emission Tomography Compared with Parietal Biopsy Samples in Living Normal Pressure Hydrocephalus Patients. Mol. Imaging Biol. 2013, 15, 230–237. [Google Scholar] [CrossRef]
- Rinne, J.O.; Wong, D.F.; Wolk, D.A.; Leinonen, V.; Arnold, S.E.; Buckley, C.; Smith, A.; McLain, R.; Sherwin, P.F.; Farrar, G.; et al. [18F]Flutemetamol PET imaging and cortical biopsy histopathology for fibrillar amyloid β detection in living subjects with normal pressure hydrocephalus: Pooled analysis of four studies. Acta Neuropathol. (Berl.) 2012, 124, 833–845. [Google Scholar] [CrossRef]
- Rinne, J.O.; Frantzen, J.; Leinonen, V.; Lonnrot, K.; Laakso, A.; Virtanen, K.A.; Solin, O.; Kotkansalo, A.; Koivisto, A.; Sajanti, J.; et al. Prospective Flutemetamol Positron Emission Tomography and Histopathology in Normal Pressure Hydrocephalus. Neurodegener. Dis. 2013. [Google Scholar] [CrossRef]
- Leinonen, V.; Rinne, J.O.; Virtanen, K.A.; Eskola, O.; Rummukainen, J.; Huttunen, J.; von und zu Fraunberg, M.; Nerg, O.; Koivisto, A.M.; Rinne, J.; et al. Positron emission tomography with [18F]flutemetamol and [11C]PiB for in vivo detection of cerebral cortical amyloid in normal pressure hydrocephalus patients. Eur. J. Neurol. 2013, 20, 1043–1052. [Google Scholar] [CrossRef]
- Kondo, M.; Tokuda, T.; Itsukage, M.; Kuriyama, N.; Matsushima, S.; Yamada, K.; Nakanishi, H.; Ishikawa, M.; Nakagawa, M. Distribution of Amyloid Burden Differs between Idiopathic Normal Pressure Hydrocephalus and Alzheimer’s Disease. Neuroradiol. J. 2013, 26, 41–46. [Google Scholar] [CrossRef]
- Jiménez-Bonilla, J.F.; Quirce, R.; de Arcocha-Torres, M.; Martínez-Rodríguez, I.; Martínez-Amador, N.; Sánchez-Juan, P.; Pozueta, A.; Martín-Láez, R.; Banzo, I.; Rodríguez-Rodríguez, E. 11C-PIB retention patterns in white and grey cerebral matter in idiopathic normal pressure hydrocephalus patients. A visual analysis. Rev. Esp. Med. Nucl. E Imagen Mol. 2018, 37, 87–93. [Google Scholar] [CrossRef]
- Kang, K.; Yoon, U.; Hong, J.; Jeong, S.Y.; Ko, P.-W.; Lee, S.-W.; Lee, H.-W. Amyloid Deposits and Idiopathic Normal-Pressure Hydrocephalus: An 18F-Florbetaben Study. Eur. Neurol. 2018, 79, 192–199. [Google Scholar] [CrossRef]
- Hiraoka, K.; Narita, W.; Kikuchi, H.; Baba, T.; Kanno, S.; Iizuka, O.; Tashiro, M.; Furumoto, S.; Okamura, N.; Furukawa, K.; et al. Amyloid deposits and response to shunt surgery in idiopathic normal-pressure hydrocephalus. J. Neurol. Sci. 2015, 356, 124–128. [Google Scholar] [CrossRef]
- Jang, H.; Park, S.B.; Kim, Y.; Kim, K.W.; Lee, J.I.; Kim, S.T.; Lee, K.H.; Kang, E.-S.; Choe, Y.S.; Seo, S.W.; et al. Prognostic value of amyloid PET scan in normal pressure hydrocephalus. J. Neurol. 2018, 265, 63–73. [Google Scholar] [CrossRef]
- Rinne, J.O.; Suotunen, T.; Rummukainen, J.; Herukka, S.-K.; Nerg, O.; Koivisto, A.M.; Rauramaa, T.; Någren, K.; Hiltunen, M.; Alafuzoff, I.; et al. [11C]PIB PET Is Associated with the Brain Biopsy Amyloid-β Load in Subjects Examined for Normal Pressure Hydrocephalus. J. Alzheimers Dis. JAD 2019, 67, 1343–1351. [Google Scholar] [CrossRef]
- Owler, B.K.; Momjian, S.; Czosnyka, Z.; Czosnyka, M.; Péna, A.; Harris, N.G.; Smielewski, P.; Fryer, T.; Donovan, T.; Coles, J.; et al. Normal Pressure Hydrocephalus and Cerebral Blood Flow: A PET Study of Baseline Values. J. Cereb. Blood Flow Metab. 2004, 24, 17–23. [Google Scholar] [CrossRef]
- Miyamoto, J.; Imahori, Y.; Mineura, K. Cerebral oxygen metabolism in idiopathic-normal pressure hydrocephalus. Neurol. Res. 2007, 29, 830–834. [Google Scholar] [CrossRef]
- Owler, B.K.; Pena, A.; Momjian, S.; Czosnyka, Z.; Czosnyka, M.; Harris, N.G.; Smielewski, P.; Fryer, T.; Donvan, T.; Carpenter, A.; et al. Changes in Cerebral Blood Flow during Cerebrospinal Fluid Pressure Manipulation in Patients with Normal Pressure Hydrocephalus: A Methodological Study. J. Cereb. Blood Flow Metab. 2004, 24, 579–587. [Google Scholar] [CrossRef]
- Momjian, S.; Owler, B.K.; Czosnyka, Z.; Czosnyka, M.; Pena, A.; Pickard, J.D. Pattern of white matter regional cerebral blood flow and autoregulation in normal pressure hydrocephalus. Brain 2004, 127, 965–972. [Google Scholar] [CrossRef]
- Brooks, D.J.; Beaney, R.P.; Powell, M.; Leenders, K.L.; Crockard, H.A.; Thomas, D.G.T.; Marshall, J.; Jones, T. Studies on cerebral oxygen metabolism, blood flow, and blood volume, in patients with hydrocephalus before and after surgical decompression, using positron emission tomography. Brain 1986, 109, 613–628. [Google Scholar] [CrossRef]
- Klinge, P.; Berding, G.; Brinker, T.; Schuhmann, M.; Weckesser, E.; Knapp, W.H.; Samii, M. The Role of Cerebral Blood Flow and Cerebrovascular Reserve Capacity in the Diagnosis of Chronic Hydrocephalus—A PET-Study on 60 Patients. In Intracranial Pressure and Brain Biochemical Monitoring; Czosnyka, M., Pickard, J.D., Kirkpatrick, P.J., Smielewski, P., Hutchinson, P., Eds.; Springer: Vienna, Austria, 2002; pp. 39–41. ISBN 978-3-7091-7397-8. [Google Scholar]
- Klinge, P.M.; Knapp, W.H. A positron emission tomography study of cerebrovascular reserve before and after shunt surgery in patients with idiopathic chronic hydrocephalus. J Neurosurg 1999, 91, 5. [Google Scholar]
- Klinge, P.; Brinker, T.; Brinker, T.; Schuhmann, M.; Knapp, W.H.; Samii, M. PET-Studies in Idiopathic Chronic Hydrocephalus before and after Shunt-Treatment: The Role of Risk Factors for Cerebrovascular Disease (CVD) on Cerebral Hemodynamics. In Intracranial Pressure and Brain Biochemical Monitoring; Czosnyka, M., Pickard, J.D., Kirkpatrick, P.J., Smielewski, P., Hutchinson, P., Eds.; Springer: Vienna, Austria, 2002; pp. 43–45. ISBN 978-3-7091-7397-8. [Google Scholar]
- Klinge, P.; Berding, G.; Brinkerl, T.; Weckesser, E.; Knapp, W.H. Regional Cerebral Blood Flow Profiles of Shunt-Responder in Idiopathic Chronic Hydrocephalus—A 15-0-Water PET-Study. In Intracranial Pressure and Brain Biochemical Monitoring; Springer: Vienna, Austria, 2002; pp. 47–49. [Google Scholar]
- Klinge, P.; Rückert, N.; Schuhmann, M.; Berding, G.; Brinker, T.; Knapp, W.H.; Samii, M. Neuropsychological Sequels to Changes in Global Cerebral Blood Flow and Cerebrovascular Reserve Capacity after Shunt Treatment in Chronic Hydrocephalus—A Quantitative PET-Study. In Intracranial Pressure and Brain Biochemical Monitoring; Czosnyka, M., Pickard, J.D., Kirkpatrick, P.J., Smielewski, P., Hutchinson, P., Eds.; Springer: Vienna, Austria, 2002; pp. 55–57. ISBN 978-3-7091-7397-8. [Google Scholar]
- Klinge, P.M.; Brooks, D.J.; Samii, A.; Weckesser, E.; van den Hoff, J.; Fricke, H.; Brinker, T.; Knapp, W.H.; Berding, G. Correlates of local cerebral blood flow (CBF) in normal pressure hydrocephalus patients before and after shunting—A retrospective analysis of [15O]H2O PET-CBF studies in 65 patients. Clin. Neurol. Neurosurg. 2008, 110, 369–375. [Google Scholar] [CrossRef]
- Miyamoto, J.; Tatsuzawa, K.; Inoue, Y.; Imahori, Y.; Mineura, K. Oxygen metabolism changes in patients with idiopathic normal pressure hydrocephalus before and after shunting operation. Acta Neurol. Scand. 2007, 116, 137–143. [Google Scholar] [CrossRef]
- Klinge, P.; Fischer, J.; Brinker, T.; Heissler, H.E.; Burchert, W.; Berding, G.; Knapp, W.H.; Samii, M. PET and CBF Studies of Chronic Hydrocephalus: A Contribution to Surgical Indication and Prognosis. J. Neuroimaging 1998, 8, 205–209. [Google Scholar] [CrossRef]
- Jagust, W.J.; Friedland, R.P.; Budinger, T.F. Positron emission tomography with [18F]fluorodeoxyglucose differentiates normal pressure hydrocephalus from Alzheimer-type dementia. J. Neurol. Neurosurg. Psychiatry 1985, 48, 1091–1096. [Google Scholar] [CrossRef]
- Tedeschi, E.; Hasselbalch, S.G.; Waldemar, G.; Juhler, M.; Hogh, P.; Holm, S.; Garde, L.; Knudsen, L.L.; Klinken, L.; Gjerris, F. Heterogeneous cerebral glucose metabolism in normal pressure hydrocephalus. J. Neurol. Neurosurg. Psychiatry 1995, 59, 608–615. [Google Scholar] [CrossRef]
- Townley, R.A.; Botha, H.; Graff-Radford, J.; Boeve, B.F.; Petersen, R.C.; Senjem, M.L.; Knopman, D.S.; Lowe, V.; Jack, C.R.; Jones, D.T. 18 F-FDG PET-CT pattern in idiopathic normal pressure hydrocephalus. NeuroImage Clin. 2018, 18, 897–902. [Google Scholar] [CrossRef]
- Miyazaki, K.; Hanaoka, K.; Kaida, H.; Chiba, Y.; Ishii, K. Changes in cerebral glucose metabolism caused by morphologic features of prodromal idiopathic normal pressure hydrocephalus. EJNMMI Res. 2019, 9, 111. [Google Scholar] [CrossRef]
- George, A.E.; de Leon, M.J.; Miller, J.; Klinger, A.; Foo, S.H.; Christman, D.L.; Wolf, A. Positron emission tomography of hydrocephalus. Metabolic effects of shunt procedures. Acta Radiol. Suppl. 1986, 369, 435–439. [Google Scholar]
- Calcagni, M.L.; Lavalle, M.; Mangiola, A.; Indovina, L.; Leccisotti, L.; De Bonis, P.; Marra, C.; Pelliccioni, A.; Anile, C.; Giordano, A. Early evaluation of cerebral metabolic rate of glucose (CMRglu) with 18F-FDG PET/CT and clinical assessment in idiopathic normal pressure hydrocephalus (INPH) patients before and after ventricular shunt placement: Preliminary experience. Eur. J. Nucl. Med. Mol. Imaging 2012, 39, 236–241. [Google Scholar] [CrossRef]
- Calcagni, M.L.; Taralli, S.; Mangiola, A.; Indovina, L.; Lavalle, M.; De Bonis, P.; Anile, C.; Giordano, A. Regional Cerebral Metabolic Rate of Glucose Evaluation and Clinical Assessment in Patients With Idiopathic Normal-Pressure Hydrocephalus Before and After Ventricular Shunt Placement: A Prospective Analysis. Clin. Nucl. Med. 2013, 38, 426–431. [Google Scholar] [CrossRef]
- Fisher, C.M. Hydrocephalus as a cause of disturbances of gait in the elderly. Neurology 1982, 32, 1358–1363. [Google Scholar] [CrossRef]
- Morel, E.; Armand, S.; Assal, F.; Allali, G. Is frontal gait a myth in normal pressure hydrocephalus? J. Neurol. Sci. 2019, 402, 175–179. [Google Scholar] [CrossRef]
- Ouchi, Y.; Nakayama, T.; Kanno, T.; Yoshikawa, E.; Shinke, T.; Torizuka, T. In Vivo Presynaptic and Postsynaptic Striatal Dopamine Functions in Idiopathic Normal Pressure Hydrocephalus. J. Cereb. Blood Flow Metab. 2007, 27, 803–810. [Google Scholar] [CrossRef]
- Nakayama, T.; Ouchi, Y.; Yoshikawa, E.; Sugihara, G.; Torizuka, T.; Tanaka, K. Striatal D2 Receptor Availability After Shunting in Idiopathic Normal Pressure Hydrocephalus. J. Nucl. Med. 2007, 48, 1981–1986. [Google Scholar] [CrossRef]
- Halperin, J.J.; Kurlan, R.; Schwalb, J.M.; Cusimano, M.D.; Gronseth, G.; Gloss, D. Practice guideline: Idiopathic normal pressure hydrocephalus: Response to shunting and predictors of response: Report of the Guideline Development, Dissemination, and Implementation Subcommittee of the American Academy of Neurology. Neurology 2015, 85, 2063–2071. [Google Scholar] [CrossRef]
- Moonis, G.; Subramaniam, R.M.; Trofimova, A.; Burns, J.; Bykowski, J.; Chakraborty, S.; Holloway, K.; Ledbetter, L.N.; Lee, R.K.; Pannell, J.S.; et al. ACR Appropriateness Criteria® Dementia. J. Am. Coll. Radiol. 2020, 17, S100–S112. [Google Scholar] [CrossRef]
- Graff-Radford, N.R.; Jones, D.T. Normal Pressure Hydrocephalus. Contin. Minneap. Minn. 2019, 25, 165–186. [Google Scholar] [CrossRef]
| Authors | N. of Pts | Tracer | Analysis of PET Images | PET Timing | Surgical Procedure | Primary Endpoint | Main Findings |
|---|---|---|---|---|---|---|---|
| Leinonen et al., (2008) [15] | 10 | [11C]PiB | Semiquantitative (SPM) | Postshunt | Shunt + frontal biopsy | To compare [11C]PiB findings in patients with and without AD lesions in frontal cortical biopsy. | 7/10 pts had diagnosis of iNPH; 3/7 iNPH had Aβ aggregates and PET positive (higher uptake in the lateral frontal and lateral temporal cortices, anterior and posterior cingulate gyri, and caudate nucleus). |
| Wolk et al., (2011) [17] | 7 | [18F]Flutemetamol | Qualitative; semiquantitative (SUVratio) | Postshunt | Shunt + frontal biopsy | To compare tracer uptake with amyloid level at biopsy. | 4/7 pts had Aβ aggregates and PET positive. A correlation between Aβ deposition and SUVratio in the VOI contralateral to the biopsy site was evident. |
| Rinne et al., (2012) [19] | 52 | [18F]Flutemetamol | Qualitative and semiquantitative (SUVratio) | Before and postshunt | Shunt + frontal biopsy | To determine the association between tracer uptake and neuritic plaques and fibrillar amyloid β. | 49/52 had sufficient data for analyses. 14/49 pts had Aβ deposition. Significant association between SUVratio and neuritic plaque burden. Se 93% and Sp 100%. [18F]Flutemetamol offers a noninvasive tool that may be useful for identifying the presence of AD-related lesions. |
| Wong et al., (2013) [18] | 12 | [18F]Flutemetamol | Qualitative; semiquantitative (SUVratio) | Baseline | Shunt + parietal biopsy | To determine the association between [18F]Flutemetamol uptake and amyloid levels at biopsy. | 10/12 pts had biopsy sample; 2/10 had Aβ aggregates. Qualitative PET had 100% in Se and Sp. The biopsy site SUVratio was correlated with the biopsy Aβ level and with the composite SUVratio. Amyloid burden in the biopsied region was representative for the entire cortex. |
| Leinonen et al., (2013) [21] | 15 | [18F]Flutemetamol ([11C]PiB in 7 pts) | Qualitative; semiquantitative (SUVratio) | Postshunt | Shunt + frontal biopsy | To determine the correlation between [18F]Flutemetamol and Aβ aggregates levels. | 11/15 pts had diagnosis of iNPH; 2/11 had Aβ aggregates. [18F]Flutemetamol and [11C]PiB SUVratios correlated with Aβ aggregates. NPH pts with PET positive may warrant treatment with AD drugs; |
| Kondo et al., (2013) [22] | 10 (+ 7 AD) | [11C]PiB | Semiquantitative (SUVratio) | Baseline | None | To elucidate the distribution of [11C]PiB in iNPH and clarify the differences with AD pts. | 3/10 iNPH pts had ↑ [11C]PiB uptake (all with MCI). iNPH vs. AD: similar mean cortical SUVratio but different [11C]PiB distribution (high-convexity parasagittal areas in iNPH vs. frontal and parieto-temporal areas in AD). [11C]PiB could be useful to differentiate iNPH from AD. |
| Rinne et al., (2013) [20] | 17 | [18F]Flutemetamol | Qualitative; semiquantitative (SUVratio) | Baseline | Shunt + frontal biopsy | To determine the association between [18F]Flutemetamol uptake and Aβ aggregates levels. | 4/17 pts had Aβ aggregates and [18F]Flutemetamol uptake; Aβ aggregates correlated with SUVratios. Qualitative PET showed 75–100% Se and 100% Sp. In PET positive pts, the response to shunt was variable (good in 1, fair in 1 and transient in 2 pts). |
| Hiraoka et al., (2015) [25] | 10 (+ 10 HC + 10 AD) | [11C]BF-227 | Semiquantitative (SUVratio) | Baseline | Shunt | To evaluate amyloid deposition preshunt and its association with postshunt response. | 5/10 pts had high SUVratios (similar to AD); 5/10 pts had low SUVratios (similar to HC). A significant inverse correlation between neocortical SUVratios and cognitive improvements after shunt surgery was observed. |
| Jiménez-Bonilla et al., (2017) [23] | 13 (+ 7 HC) | [11C]PiB | Qualitative | Baseline | None | To compare the [11C]PiB uptake pattern in iNPH pts and in HC. | 8/13 negative, 3/13 positive, 2/13 equivocal PET scans. 5/13 iNPH pts showed [11C]PiB uptake higher than HC. 4/7 HC had diffuse slight uptake, 3/7 HC had mild uptake in at least one region. WM uptake of iNPH scored lower than in HC. |
| Jang et al., (2018) [26] | 31 | [18F]Florbetaben | Qualitative; semiquantitative (SUVratio) | Baseline | tap test and shunt | To evaluate the prognostic value of [18F]Florbetaben by analyzing the response to the tap test. | 7/31 pts had PET positive. PET negative pts (24/31) had ↑ % of R at tap test and showed ↑ gait scores. PET positivity and CSF p-tau were independently associated with the response to tap test. Amyloid PET can help to determine which iNPH pts will benefit from shunt by discriminating concomitant AD. |
| Kang et al., (2018) [24] | 17 (+ 8 HC) | [18F]Florbetaben | Semiquantitative (SUVratio) | Baseline | None | To investigate the cortical uptake in iNPH and HC; the relationships between [18F]Florbetaben uptake, hippocampal volume and clinical symptoms. | iNPH pts showed ↑ uptake in bilateral frontal, parietal, and occipital regions. In iNPH, right hippocampal volume was negatively correlated with right frontal uptake. ↑ uptake significantly correlated with ↑ CDRS score in the right occipital cortex. iNPH might exhibit a characteristic pattern of cortical uptake. |
| Leinonen et al., (2018) [16] | 14 | S-[18F]-THK-5117 (tau deposit) and [11C]PiB | Semiquantitative (Voxel-based statistical analysis) | Postshunt | Shunt + frontal biopsy | To evaluate the association between CSF, S-[18F]THK-5117, and [11C]PiB PET against tau and amyloid lesions. | 7/14 pts had Aβ lesions, 2/14 pts had both Aβ and tau lesions, 1/14 had tau lesions. Pts with Aβ lesions had higher [11C]PiB uptake. 18F-THK-5117 uptake did not correlate with biopsy tau level or CSF p-tau or t-tau. |
| Rinne et al., (2019) [27] | 21 | [11C]PiB | Semiquantitative (time-activity curves ratio) | Baseline | Shunt + frontal biopsy | To compare the [11C]PiB uptake to Aβ and tau lesions and CSF Aβ. | 11/21 pts had Aβ aggregates; 15 pts underwent shunt (13 clinically improved). AD in 8/21 pts. [11C]PiB uptake was associated with a higher Aβ aggregates and CSF Aβ. |
| Authors | N. of Pts | Tracer | Analysis of PET Images | PET Timing | Surgical Procedure | Primary Endpoint | Main Findings |
|---|---|---|---|---|---|---|---|
| Brooks et al., (1986) [32] | 14 (+ 20 HC) | [15O]CO2 [15O]O2 [11C]CO | Quantitative (regional CMRO2, OER, CBF, CBV) | Baseline and postsurgical decompression | Surgical decompression | To evaluate CMRO2, CBF, and OER in hydrocephalous pts (obstructive and idiopathic) before (vs. HC) and after surgical decompression. | All hydrocephalous pts had ↓ CMRO2and CBF (than HC); iNPH pts had CBF/CMRO2 matched and normal OEF at baseline, and showed no improvement in CBF or CMRO2 or cognitive function after surgical intervention. |
| Klinge et al., (1998) [40] | 21 | [15O]H2O | Quantitative (global and regional CBF) | Baseline and postshunt | Shunt | To evaluate whether CBF was an indicator in selecting pts to undergo shunt. | After shunt, 12/21 pts clinically improved. Pts with lower global CBF showed clinical improvement after 7 months (in contrast with pts with higher CBF); baseline global CBF discriminated in terms of clinical outcome; CBF may be helpful in evaluating the utility of shunt. |
| Klinge et al., (1999) [34] | 10 (+ 10 HC) | [15O]H2O | Quantitative (global CBF and CVR) | Baseline and postshunt | Shunt | To evaluate CBF and CVR in chronic hydrocephalus (vs. HC) after shunt. | Baseline global CBF was ↓ compared to HC; after shunt, 5/10 pts clinically improved; baseline CBF and CVR were not predictive of clinical outcome; early improvement in CVR after shunt indicated a good prognosis. |
| Klinge et al., (2002) [35] | 53 | [15O]H2O | Quantitative (global CBF and CVR) | Baseline and postshunt | Shunt | To investigate the impact of cerebrovascular risk factors in cerebral hemodynamics. | Pts stratified into HR and LR groups; in HR, baseline CBF was ↓ in R, while CVR was marginal in both R and NR; after 1 week, in HR, CVR of R improved; in LR, CVR of NR decreased. |
| Klinge et al., (2002) [33] | 60 | [15O]H2O | Quantitative (global CBF + CVR) | Baseline and postshunt (1 week and 7 months) | Shunt | To evaluate CBF and CVR in chronic hydrocephalus after shunt | After shunt, 31/60 pts clinically improved (R); at baseline, R had ↓ CBF than NR, and CVR was not different between R and NR; after shunting, CVR decreased in NR and increases in R; CBF might substantially contribute to selection of shunt candidates, and neurological sequels may be related to early regeneration of the hemodynamic reserve. |
| Klinge et al., (2002) [36] | 11 | [15O]H2O | Quantitative + SPM (regional CBF + CVR) | Baseline | Shunt | To find out if regional CBF and CVR may indicate shunt response in idiopathic chronic hydrocephalus | CBF ↓ in the frontobasal cortex in R than in NR; CVR ↓ in a variety of cortical regions in R compared with NR, including frontobasal cortex (most ↓ in temporo-dorsal and limbic cortical regions); baseline hemodynamics displayed a regional profile of reduced CBF and CVR in pts with shunt response. |
| Klinge et al., (2002) [37] | 27 | [15O]H2O | Quantitative (global and regional CBF + CVR) | Baseline and postshunt (1 week and 7 months) | Shunt | To evaluate the relationship of neuropsychological deficits before and after shunting with dynamics in cerebral blood flow. | No relationship of test profiles with baseline CBF or CVR; after 1w, improvement of gait related to ↑ CVR; after 7m, improvement in mental tests related to ↑ CVR; neurological sequels after shunting may depend on consecutive improvement of hemodynamics |
| Owler et al., (2004) [28] | 17 (+ 12 HC) | [15O]H2O | Quantitative (global and regional CBF) | Baseline | None | To study the regional CBF in NPH with PET coregistered with MRI (vs. HC). | CBF was significantly ↓ in the cerebrum and cerebellum of iNPH pts than HC; CBF ↓ in basal ganglia and thalamus (not in WM regions); the role of the basal ganglia and thalamus in iNPH may be more prominent than currently appreciated. |
| Momjian et al., (2004) [31] | 12 (+ 10 HC) | [15O]H2O | Quantitative (regional CBF) | Baseline and during infusion test (plateau of raised ICP) | Infusion test | To investigated the distribution of peri- and paraventricular WM CBF in NPH at baseline (vs. HC) and during the infusion test. | At baseline, global CBF in iNPH was ↓ than in HC; in iNPH, the profile of the regional WM CBF at baseline showed an ↑ with distance from the ventricles. In 10 pts, infusion test caused ↓ in CPP and in global CBF; WM CBF was ↓ in iNPH, with an abnormal gradient from the lateral ventricles to the subcortical WM. |
| Owler et al., (2004) [30] | 15 | [15O]H2O | Quantitative (global and regional CBF) | Baseline and during infusion test (plateau of raised ICP) | Infusion test | To investigate the global and regional changes in CBF with changes in CSF pressure (infusion test) | With ↑ in CSF pressure, global CBF was ↓ (including cerebellum). rCBF ↓ in thalamus, basal ganglia, and in WM regions (correlated with changes in the CSF pressure and with proximity to the ventricles). |
| Miyamoto et al., (2007) [39] | 8 | [15O]H2O | Quantitative (regional CMRO2 and OEF) | Baseline and postshunt (3 months) | Shunt | To investigate the changes in cerebral oxygen metabolism before and after shunt. | After shunt, 5/8 good R and 3/8 poor R; the postshunt regional CMRO2 is ↑ in good R; the postshunt regional OEF is ↓ in the poor R. The improvement of r CMRO2 correlated with shunt response; changes in rOEF might predict poor shunt response. |
| Miyamoto et al., (2007) [29] | 9 (+ 10 HC) | [15O]H2O | Quantitative (regional CBF, CBV, OEF and CMRO2) | Baseline | Shunt | To determine the cerebral oxygen metabolism and the cerebral blood flow in patients with iNPH vs. HC. | ↓ CBF in frontal lobe and basal ganglia; ↑ CMRO2 in frontal lobe compare to HC (but CMRO2 in basal ganglia of iNPH was ↓); rCBV and rOEF showed no differences. Reduction of O2 metabolism in basal ganglia might cause symptoms in iNPH; no particular pattern of oxygen metabolism in iNPH. |
| Klinge et al., (2008) [38] | 65 | [15O]H2O | Quantitative + SPM (global and regional CBF, CVR) | Baseline and postshunt (7–10 days) | Shunt | To evaluate whether the functional status before and after shunt treatment might correlate with local blood flow in iNPH pts. | At baseline, ↑ clinical score correlated with ↓ tracer uptake in mesial frontal and anterior temporal areas; at postshunt, in the mesial frontal areas ↑ uptake in R and ↓ uptake in NR. Regional blood flow changes are relevant to NPH and to postshunt functional changes. |
| Authors | N. of Pts | Tracer | Analysis of PET Images | PET Timing | Surgical Procedure | Primary Endpoint | Main Findings |
|---|---|---|---|---|---|---|---|
| Jagust et al., (1985) [41] | 3 (+ 17 AD + 7 HC) | [18F]FDG | Quantitative (rCMRglu) | Baseline | Shunt | To differentiate patients with NPH from AD | Both AD and NPH showed lower CMRglu than HC. The hypometabolic pattern was different: AD had bilateral temporo-parietal hypometabolism, NPH had global hypometabolism. |
| George et al., (1986) [45] | 5 | [18F]FDG | Quantitative (gCMRglu) | Baseline and postshunt | Shunt | To study the glucose metabolism before and after shunt | After shunt, all patients clinically improved. After shunt, ↑ CMRglu in acute hydrocephalus pts; ↓ in chronic hydrocephalus pts despite clinical improvement. |
| Tedeschi et al., (1995) [42] | 18 (+ 11 HC) | [18F]FDG | Quantitative (rCMRglu) and semiquantitative | Baseline | Shunt + frontal biopsy | To evaluate CMRglu at baseline in iNPH underwent shunt (vs. HC) | ↓ rCMRglu in all regions in NPH than HC; no typical metabolic pattern of NPH; AD/CVD in 6/18 pts; after shunt, 6/18 pts clinically improved (2 AD and 2 CVD); no significant different in metabolic pattern between R (6/18) and NR (12/18); high metabolic and histopathological heterogeneity. |
| Calcagni et al., (2012) [46] | 11 | [18F]FDG | Quantitative (gCMRglu) | Baseline and postshunt | Shunt | To evaluate the gCMRglu and clinical assessment before and after shunt | After shunt, ↑ gCMRglu in all pts, and ↓ in all clinical scale scores (significant correlation between CMRglu and clinical assessment). |
| Calcagni et al., (2013) [47] | 20 | [18F]FDG | Quantitative (rCMRglu; gCMRglu) | Baseline and postshunt | Shunt | To evaluate the rCMRglu before and after shunt and to correlated the CMRglu with the clinical scale scores. | No differences in CMRglu between right and left brain regions at baseline and postshunt; at postshunt, R (17/20) had ↓ scale scores and ↑ CMRglu in all regions; in NR (3/20) stable scale scores and ↑ CMRglu in only 3 regions (left frontal, left putamen, and right thalamus). At baseline, no difference in gCMRglu between R and NR. At postshunt, individual % variations of CMRglu correlated with clinical scores. |
| Townley et al., (2018) [43] | 7 (+ 21 HC + 14 AD + 14 DLB/PDD + 7 bvFTD) | [18F]FDG | Semiquantitative + SPM | Baseline | None | To differentiate iNPH from neurodegenerative disorders. | iNPH group had significant hypometabolism in the dorsal striatum (tan other groups), involving the caudate and putamen bilaterally; no specific pattern of significant cortical hypometabolism. This pattern may differentiate iNPH from degenerative diseases. |
| Miyazaki et al., (2019) [44] | 12 (+ 33 PMD + 32 AVIM + 89 HC) | [18F]FDG | Semiquantitative (SUVratio) | Baseline | None | To evaluate the changes in regional cerebral glucose metabolism with respect to the characteristic morphologic features of iNPH. | In the frontal and temporal lobes, SUVr in PMD, AVIM, and iNPH groups were significantly lower than HC; in parietal lobes, the SUVr were significantly higher in PMD and AVIM groups; in the thalami and striata, the SUVr were significantly lower in iNPH. Changes in glucose metabolism in the cortices in preclinical iNPH; basal ganglia hypometabolism only in clinical iNPH. |
| Authors | N. of Pts | Tracer | Analysis of PET Images | PET Timing | Surgical Procedure | Primary Endpoint | Main Finding s |
|---|---|---|---|---|---|---|---|
| Ouchi et al., (2007) [50] | 8 (+ 8 HC) | [11C]CFT (presynaptic marker) | Quantitative (BPs) | Baseline | None | To differentiate impaired gait seen in iNPH from parkinsonian gait; to investigate dopaminergic contribution to iNPH pathophysiology (vs. HC) | ↓ [11C]Raclopride BP in putamen and nucleus accumbens than HC; equal [11C]CFT BP in iNPH and HC; putamen [11C]Raclopride BP negatively correlated with gait severity; nucleus accumbens [11C]Raclopride BP positively correlated with emotional recognition score; postsynaptic receptor reduction and preserved presynaptic status found in NPH. |
| [11C]Raclopride (postsynaptic marker) | |||||||
| Nakayama et al., (2007) [51] | 8 | [11C]Raclopride (postsynaptic marker) | Quantitative (BPs) | Baseline and postshunt (1 month) | Shunt | To evaluate the plasticity of D2 receptor in treating iNPH pts with ventriculoperitoneal shunt | All pts clinically improved after shunt. At postshunt PET, ↑ in [11C]Raclopride BP in nucleus accumbens and dorsal putamen; ↑ [11C]Raclopride BP in striaum correlated with improvement in general cognitive ability. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mattoli, M.V.; Treglia, G.; Calcagni, M.L.; Mangiola, A.; Anile, C.; Trevisi, G. Usefulness of Brain Positron Emission Tomography with Different Tracers in the Evaluation of Patients with Idiopathic Normal Pressure Hydrocephalous. Int. J. Mol. Sci. 2020, 21, 6523. https://doi.org/10.3390/ijms21186523
Mattoli MV, Treglia G, Calcagni ML, Mangiola A, Anile C, Trevisi G. Usefulness of Brain Positron Emission Tomography with Different Tracers in the Evaluation of Patients with Idiopathic Normal Pressure Hydrocephalous. International Journal of Molecular Sciences. 2020; 21(18):6523. https://doi.org/10.3390/ijms21186523
Chicago/Turabian StyleMattoli, Maria Vittoria, Giorgio Treglia, Maria Lucia Calcagni, Annunziato Mangiola, Carmelo Anile, and Gianluca Trevisi. 2020. "Usefulness of Brain Positron Emission Tomography with Different Tracers in the Evaluation of Patients with Idiopathic Normal Pressure Hydrocephalous" International Journal of Molecular Sciences 21, no. 18: 6523. https://doi.org/10.3390/ijms21186523
APA StyleMattoli, M. V., Treglia, G., Calcagni, M. L., Mangiola, A., Anile, C., & Trevisi, G. (2020). Usefulness of Brain Positron Emission Tomography with Different Tracers in the Evaluation of Patients with Idiopathic Normal Pressure Hydrocephalous. International Journal of Molecular Sciences, 21(18), 6523. https://doi.org/10.3390/ijms21186523






