Abstract
Increases in adipocyte volume and tissue mass due to obesity can result in inflammation, further dysregulation in adipose tissue function, and eventually adipose tissue fibrosis. Like other fibrotic diseases, adipose tissue fibrosis is the accumulation and increased production of extracellular matrix (ECM) proteins. Adipose tissue fibrosis has been linked to decreased insulin sensitivity, poor bariatric surgery outcomes, and difficulty in weight loss. With the rising rates of obesity, it is important to create accurate models for adipose tissue fibrosis to gain mechanistic insights and develop targeted treatments. This article discusses recent research in modeling adipose tissue fibrosis using in vivo and in vitro (2D and 3D) methods with considerations for biomaterial selections. Additionally, this article outlines the importance of adipose tissue in treating other fibrotic diseases and methods used to detect and characterize adipose tissue fibrosis.
1. Introduction
Fibrosis is characteristically defined as the thickening and scarring of tissues from a pathological repair process. While often fibrosis is thought to occur following an injury, in adipose tissue changes in metabolism can trigger an inflammatory cascade that initiates the maladaptive fibrotic repair process. Once initiated, fibrotic changes in adipose tissue have devastating effects for patients. In most cases, adipose tissue fibrosis is undetected, difficult to reverse, and interferes with treatment options for obesity and other comorbidities.
White adipose tissue is responsible for storing and releasing energy in the body by regulating lipogenesis and lipolysis, respectively [1]. Lipogenesis is the process where free fatty acids and glycerol are taken up from the blood stream and stored in adipocytes as triglycerides. Conversely, lipolysis is the process in which triglycerides are catabolized into free fatty acids and glycerol that are released into the blood stream where they are used as an energy source by other organs [2]. An imbalance between these two processes leads to obesity and metabolic diseases, such as type 2 diabetes [3].
Obesity is associated with adipocyte enlargement (hypertrophy) and formation of new adipocytes (hyperplasia) [4]. This increase in adipocyte volume and tissue mass triggers inflammation, further dysregulation in adipose tissue, and eventually adipose tissue fibrosis [5]. Like other fibrotic diseases, adipose tissue fibrosis is the accumulation and increased production of extracellular matrix (ECM) proteins [6,7]. In healthy adipose tissue, the ECM can be remodeled to accommodate normal fluctuations in adipocyte size. However, when adipose tissue becomes fibrotic the stiff ECM cannot be dynamically remodeled.
Historically, obesity has been linked to an overall increase in lipolysis that results in ectopic accumulation of lipids in other tissues and insulin resistance [8,9]. An increase is observed when lipolysis is normalized to overall lean body mass; however, when normalized to lipid content there is a decrease in basal lipolysis rate per cell [10,11,12]. Furthermore, starved hypertrophic adipocytes only lose a portion of their stored triglycerides (<50%) without supplemental factors [13]. There is limited literature investigating the effect adipose tissue fibrosis has on lipolysis, so most studies assume the same trends as obesity. There are several theories as to why this may not be valid. One being that as adipocytes shrink, stress between the ECM and cells can increase, eventually inhibiting lipolysis, leading to enlarged adipocytes without a mechanism for depleting their lipid stores [7].
With decreased lipolytic function, it is not surprising that fibrotic adipose tissue has been linked to difficulties losing fat mass [14] and insulin resistance [15]. For example, gastric bypass surgery, which generally causes rapid fat loss and increases in insulin sensitivity, is less effective for patients with high degrees of adipose tissue fibrosis [14,16]. Specifically, insulin resistance is linked to higher levels of adipose tissue inflammation and increased ECM deposition [15]. Initially, as insulin sensitivity decreases, adipocytes begin to undergo enhanced lipolysis and release free fatty acids into the environment. These free fatty acids cause further insulin resistance and can lead to inflammation by signaling macrophages and other immune cells to the area [17]. Over time this inflammation increases ECM protein production leading to interstitial fibrosis in adipose tissue. The increased ECM stiffness prevents the adipocytes from expanding in a healthy manner, causing the tissue to be metabolically dysfunctional, including adipocyte death, decreased lipolysis, and disrupted cell-cell interactions [6,14].
Adipose tissue fibrosis has a complicated and critical role in metabolic dysfunction and obesity. With the growing obesity epidemic, it is important to understand the mechanisms behind the development and progression of adipose tissue fibrosis. Furthermore, targeted therapeutic treatments are required for patients with fibrotic adipose tissue. This article discusses recent research in modeling adipose tissue fibrosis using in vitro (2D and 3D) and in vivo methods with considerations for biomaterial selections. Additionally, this article will outline the importance of adipose tissue in treating other fibrotic diseases and methods used to detect and characterize adipose tissue fibrosis.
2. Mechanisms
While there are a number of cell types, environmental factors, and cellular pathways that contribute to the development of adipose tissue fibrosis, the overarching cause is obesity (Figure 1) [18,19,20,21,22,23]. However, the link between obesity and fibrosis is complex and incompletely understood. While there is an increased prevalence of adipose tissue fibrosis in cases of severe obesity [19,24], not all obese patients will develop fibrotic tissue. Primarily based on the correlative nature of human studies, there remains many open questions surrounding the link between obesity and fibrotic tissue. This synopsis aims to introduce several mechanisms that have been thoroughly researched.
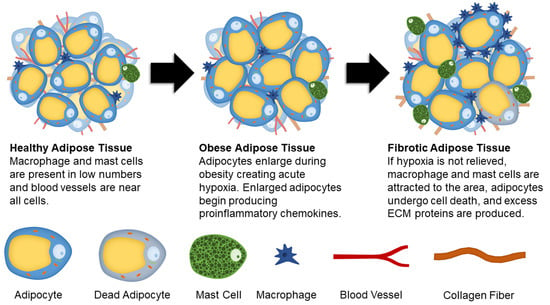
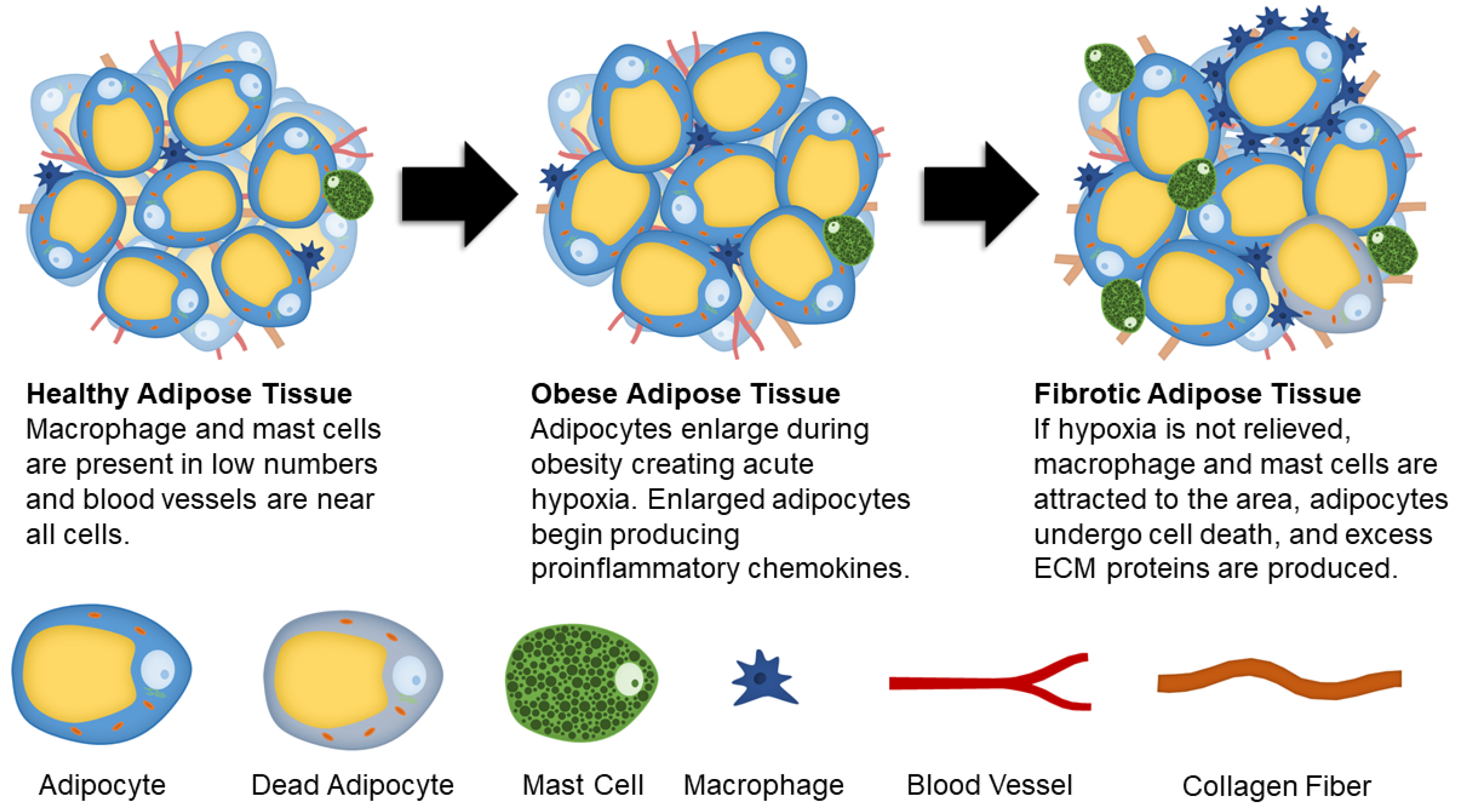
Figure 1.
Schematic showing changes to adipose tissue during obesity and fibrosis development.
Proper adipose tissue ECM composition and remodeling is crucial for cellular function. During the development of fibrosis, excess ECM proteins are produced while ECM degradation is limited. Several cell types in adipose tissue including adipocyte progenitors, adipocytes, fibroblasts, and myofibroblasts are responsible for the production of ECM proteins [25].
Fibronectin and collagens are the most prevalent ECM proteins found in healthy and fibrotic adipose tissue. Comparing obese and diabetic mice, it was determined that type I, III, V, and VI collagens are present in higher concentrations compared to healthy mice [26]. Collagen I (COL1) is responsible for providing structure within the tissue [14], while collagen V (COL5) in high concentrations inhibits angiogenesis in adipose tissues [27]. Specific to adipose tissue fibrosis, collagen VI (COL6) has been investigated for its fibrotic role, with the absence of COL6 (in a knockout model) resulting in lower inflammation and uninhibited enlargement of individual adipocytes [28]. Furthermore, a component of COL6, COL6a3 named endotrophin, stimulates fibrotic collagen deposition, inflammation, and insulin resistance [22]. In times of energy surplus, adipocytes undergo hypertrophy (enlargements of cells) and hyperplasia (an increase in the number of cells) [29]. As adipocytes become larger, they not only exhibit morphological changes but also become metabolically dysfunctional [30,31], producing several proinflammatory chemokines. These chemokines include tumor necrosis factor-alpha (TNFα), inducible nitric oxide synthase (iNOS), interleukin 6 (IL-6), interleukin 8 (IL-8), C-reactive protein, Transforming growth factor beta 1 (TGFβ1), soluble intercellular adhesion molecule (ICAM), and monocyte chemoattractant protein 1 (MCP-1) [31,32,33,34,35,36,37,38,39,40]. Additionally, adipocytes exhibit increased gene expression for ECM production under a high fat diet [41]. Similar to mature adipocytes, adipose derived stem cells (ASCs) also undergo significant changes during obesity. After being fed a high fat diet, mice had higher concentrations of ASCs that expressed platelet-derived growth factor receptor α (PDGFRα) [42]. PDGFRα activation opposes adipogenesis and causes cells to differentiate into a profibrotic phenotype [43]. The PDGFRα+ ASCs were found to promote fibrosis and are associated with insulin resistance [42].
Hypoxia is another major contributor in the development of adipose tissue fibrosis. As adipocytes expand due to lipid accumulation, they eventually reach the diffusion limit of oxygen. This results in hypoxia that causes stress signals to increase angiogenesis and remodel ECM proteins in an attempt to mitigate the oxygen shortage. In hypoxic adipose tissue, hypoxia-inducible factor 1-alpha (HIF1α) is expressed. Instead of initiating proangiogenic conditions in adipocytes, it enhances synthesis of ECM components. HIF1α is also suggested to play a role in collagen crosslinking and stabilization [7]. Both adipocytes and adipocyte progenitor cells produce angiogenic factors, such as vascular endothelial growth factor (VEGF), leptin, fibroblast growth factor 2 (FGF-2), and hepatocyte growth factor (HGF) [44,45]. Interestingly, endothelial cells derived from obese adipose tissue have increased gene expression related to inflammation and senescence, and impaired angiogenesis [46], suggesting that initial signals to promote angiogenesis are reversed if hypoxia is unresolved. Therefore, prolonged hypoxic conditions cause injury and long-term damage, fibrosis, cellular senescence, and necrotic adipocyte death [47].
Unresolved hypoxia can also attract proinflammatory cell types to the area. A population of cells that have a high concentration in fibrotic adipose tissue are M1 macrophages. Resident macrophages comprise between 10–15% of stromal cells in the adipose tissue of healthy, lean individuals [48]. This concentration jumps to between 45–60% of stromal cells in obese individuals [35,49,50]. Macrophages are typically observed in “crown-like structures” surrounding dead or dying adipocytes in obese adipose tissue [51,52]. The increase in macrophage concentration causes an increased recruitment of monocytes to the area; where the monocytes then differentiate into macrophages [35,48]. In the presence of M1 macrophages, ASCs are predisposed to differentiate into proinflammatory cells. This change occurs due to pro-inflammatory cytokines secreted by the macrophages [49,53], where it has been shown that they are the primary producers of TNFα, iNOS, and IL-6 [44].
ECM degradation is regulated by metalloproteinases (MMPs) and tissue inhibitors of metalloproteinases (TIMPs). MMPs have the ability to solubilize ECM components while specific TIMPs are able to inhibit MMPs activity. Obesity and type 2 diabetes have been linked to increased circulating TIMPs concentration causing MMP activity to be lower than in healthy individuals [54,55]. The increase in proinflammatory cytokines that occurs during hypoxia and inflammation has also been linked to regulation of MMPs expression [56].
Another cell population that has been linked to adipose tissue fibrosis is mast cells [57,58]. Mast cells can promote fibroblast growth and collagen production by releasing cytokines, chemokines, proteases, etc. [59,60]. By comparing adipose tissue from obese patients with the metabolic syndrome to healthy patients, it was determined that there is an accumulation of mast cells in the subcutaneous adipose tissue of obese patients. Related to fibrotic changes, there were positive correlations to collagen, leptin, and glucose concentrations, as well as waist circumference [57,61]. Overall, more research is needed to determine the role of mast cells and macrophages in the fibrotic transition of adipose tissues.
One method of better understanding the role of different cell types in ECM production, immune reactions, and fibrosis is single-cell RNA (scRNA) sequencing. When looking at adipocytes specifically single nuclei adipocyte RNA sequencing (SNAP-seq) is used [62]. These techniques have been used to better understand variability in cells from different fat deposits or pathological settings [63,64,65,66,67]. It offers a method of characterizing different subpopulations of adipose tissue cells and detecting heterogeneity in the populations [68].
3. In Vitro Adipose Tissue Fibrosis Modeling
Whenever possible, in vitro models are utilized to refine, reduce, and replace animal models. In vitro models can be quickly developed (compared to animal models), are easily manipulated with highly defined conditions, and have a high likelihood of clinical translation (when developed with human cells). Having accurate biomimetic in vitro adipose tissue fibrosis models can serve as useful tools for generating a better understanding of the mechanisms behind fibrosis development and progression, as well as drug screening for effective anti-fibrotic drugs.
3.1. 2D Models
White adipocytes have large unilocular lipid droplets in their cytoplasm. This causes the mature adipocytes to float in media when cultured using 2-dimensional (2D) approaches. Unable to meet their nutritional requirements, the floating adipocytes will lyse in a matter of days [69]. Techniques have been developed to combat this unique problem, including a method termed “ceiling culture” where flasks are filled with media and adipocytes attach to the ceiling of the cell culture flask [69,70,71]. However, cells cultured in this way do not function the same as in vivo adipocytes and lose their lipid stores [72]. As an alternative to culturing mature adipocytes ex vivo, ASCs are differentiated on 2D substrates to an immature multilocular phenotype (full differentiation cannot be achieved in these systems as lipid laden cells will detach from the surface).
Two-dimensional culture systems have been used to model cellular changes during obesity and adipose tissue fibrosis. Many systems use macrophages to mimic the inflammatory effects associated with obesity that trigger adipose tissue fibrosis [53,73,74,75,76,77]. One model system used media that was conditioned by macrophages from an obese human patient with or without supplementation of lipopolysaccharide (LPS), to study the secretory effects on ASCs from a healthy patient. The results indicated that ASCs have a lower capacity to differentiate in the presence of macrophage byproducts. Additionally, in the presence of LPS the ASCs became proinflammatory and secreted higher concentrations of inflammatory factors [77]. Following a similar experimental setup, another study found that ASCs exposed to factors secreted by M1 macrophages increased ECM remodeling. The ASCs had a proinflammatory phenotype, increased proliferation and migration, but a decreased ability to differentiate [53]. Together, these results indicate that the presence of secretory factors from M1 polarized macrophages primes ASCs to develop a proinflammatory phenotype that is pro-fibrotic, rather than pro-adipogenic.
Another study corroborated these results with a co-culture model. Proinflammatory macrophages (CD14+) and human ASCs (hASCs) were investigated to determine how stem cell differentiation was affected by their interaction. Consistently, there was a significant decrease in differentiation after 14 days in the presence of proinflammatory macrophages, with a decrease in adiponectin, CEBPβ, and GLUT4 and an increase in IL6 gene expression [74]. These results have an increased physiological relevance compared to similar studies [5,75,78] because both cell types were sourced from the same patient. It was proposed that the cytokine levels would be similar to native human adipose tissue because the same cell source was used. This suggests that 2D models could be used to study other fibrotic mechanisms related to ASC commitment and proinflammatory factors.
3.2. Three-Dimensional Models
Biomaterials Used for 3D Adipose Tissue Culture
Due to the decreased physiological relevance of culturing adipocytes using 2D techniques, 3D scaffolds are often used to grow and model adipose tissue. Several synthetic and natural biomaterials have been used to successfully culture ASCs and whole adipose tissue ranging from synthetic to natural polymers (Table 1). While some progress has been made modeling fibrotic adipose tissue using 3D in vitro platforms, this area of research is still at early stages of development with large potential for growth.

Table 1.
Biomaterials that have been used in vitro to culture adipose tissue.
Diabetic patients have higher degrees of adipose tissue fibrosis [79]. To better understand the link between the ECM and adipocyte metabolic regulation, researchers used human decellularized adipose tissue from patients with and without type II diabetes mellitus as a model system. Combining ECM from a nondiabetic patient and adipocytes from a diabetic patient resulted in adipocytes that had fully restored basal lipolysis and insulin stimulated glucose uptake. Conversely, combining ECM from a diabetic patient and adipocytes from a nondiabetic patient decreased insulin sensitivity, but had no effect on lipolysis [80]. This suggests that dysfunctional fibrotic cues are found in the matrix and are not an intrinsic property of the cells.
The interaction of matrix stiffness and architecture with adipocytes was also tested in an in vitro model. Researchers incorporated ethylene glycol-bis-succinic acid N-hydroxysuccinimide ester (PEGDS) into a collagen hydrogel system. The addition of PEGDS created a more organized and interconnected ECM by establishing intrafibrillar and interfibrillar crosslinks. This was used to investigate the effect mechanical changes, like those that occur during adipose tissue fibrosis, have on adipocyte morphology and function. In stiffer hydrogels, adipocytes experienced increased profibrotic gene expression and ECM deposition. By inhibiting actin contractility, the researchers determined that the adipocyte dysfunction was regulated by actin cytoskeletal stress fibers that registered the mechanical properties of the surrounding environment [81]. This research indicates that if fibrosis could be reversed cells could be rescued from the fibrotic phenotype. Additionally, this research further supports that fibrotic cues are found in the extracellular matrix.
To mimic in vivo fibrosis in another system, researchers created hypoxic clusters of ASCs that grew into 3D cell masses comprised of ASCs, alpha smooth muscle actin positive (αSMA-positive) cells, necrotic cells, and collagen [82]. Researchers compared these 3D cell masses to 2D cultures and found that there was a significant increase in TGFβ1 secretion, hydroxyproline concentration, and lactate dehydrogenase secretion. These changes indicate an increase in expression of the fibrotic phenotype, ECM production, and cell death which align with physiological changes that occur during adipose tissue fibrosis development [82].
3.3. Biomaterial Considerations for In Vitro Modeling
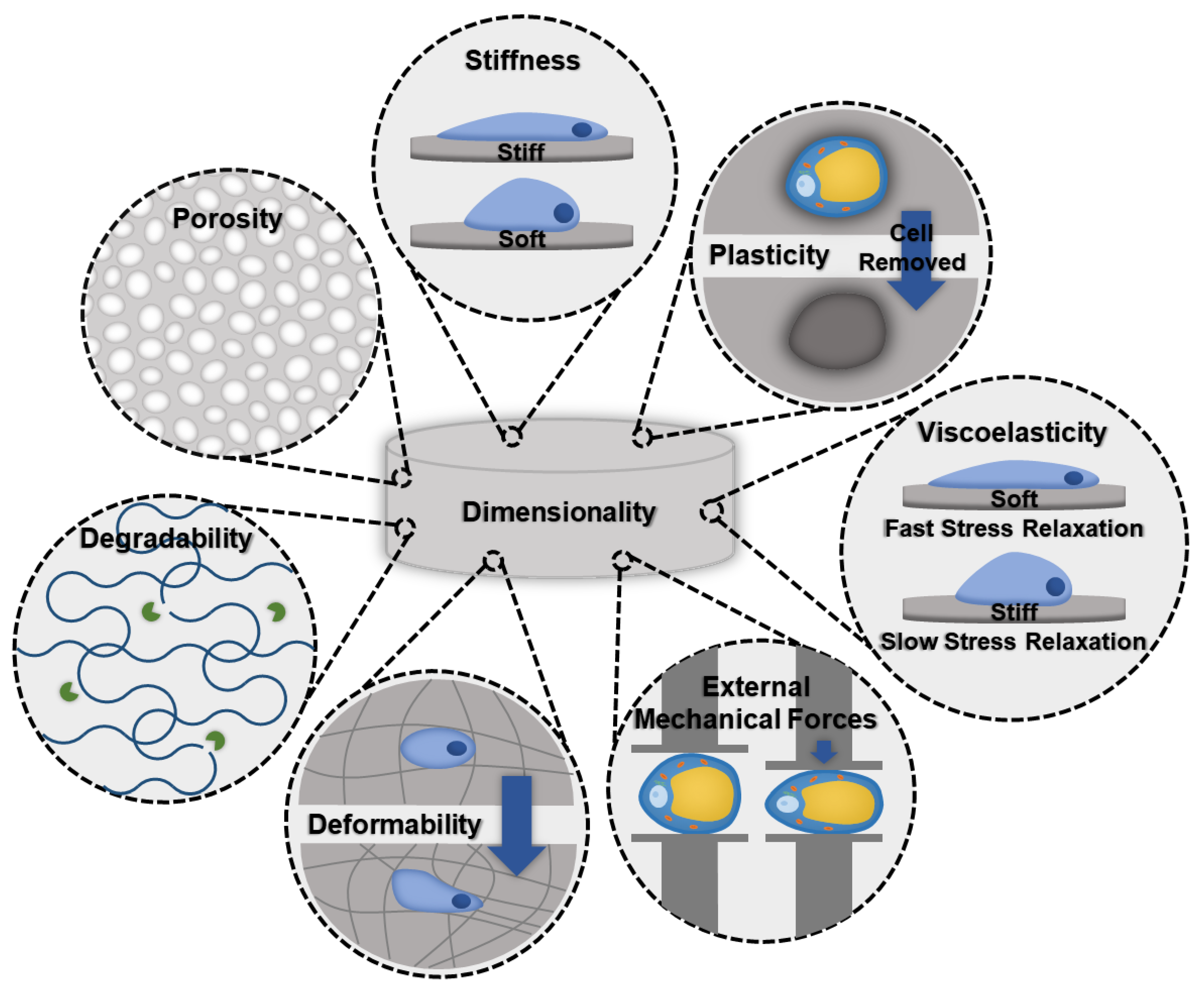
When designing a biomaterial for modeling adipose tissue fibrosis, evaluating biomaterials that successfully grow healthy adipose tissue, as well as models that are used for other fibrotic tissues, can be a good starting point. Important biomaterial characteristics that should be considered follow (Figure 2).
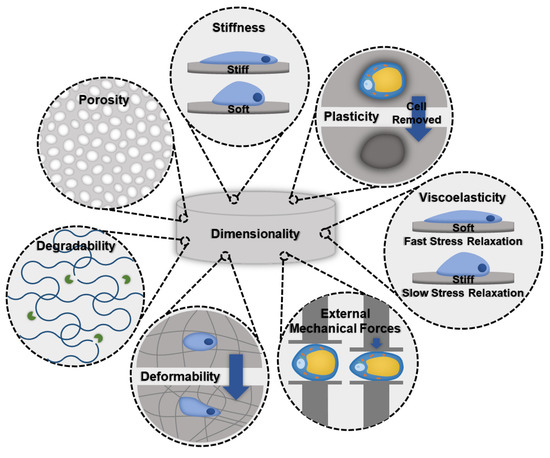
Figure 2.
Schematic illustrating important biomaterial properties that should be considered when designing an in vitro fibrotic adipose tissue model.
- Stiffness: Stiffness is an intrinsic property of a material and is defined as the resistance of an elastic material to deformation by an applied force. Adipocytes and ASCs are mechanosensitive and mechanoresponsive [100,101]. In the body, mechanical forces are balanced internally by the cytoskeleton and externally through the ECM. Scaffolding materials act as the ECM in in vitro models, therefore, the stiffness should be carefully considered as it will influence the cellular cytoskeleton and phenotype [100]. Scaffold stiffness can be controlled by adding other polymers or additives [81]. A recent study found that stiffer biomaterials triggered fibrotic traits in adipocytes, such as increased profibrotic gene expression and ECM deposition [81]. Additionally, stiffer matrices were found to promote osteogenic differentiation of ASCs, while softer matrices promoted adipogenic commitment [101].
- Viscoelasticity: Under deformation, a viscoelastic biomaterial will exhibit both elastic and viscous behavior making it a time-dependent response. Similar to stiffness, viscoelasticity is an intrinsic material property. The stress response of the material will vary based on strain and history of deformation and allows viscoelastic materials to exhibit stress relaxation, hysteresis, and creep. Soft tissues, like adipose tissue, are composed of solids and liquids and naturally behave like viscoelastic materials. An increase in collagen content results in a greater elastic component to the adipose tissue, and therefore fibrotic tissues would exhibit a decrease in the time-dependent viscous behavior.
- Degradability: A biomaterials degradability is also an intrinsic trait. It is dependent on the molecular properties of the biomaterial. Factors, such as cross-linking, can decrease the degradability. As fibrosis involves significant deposition of ECM proteins, degradability should be considered to ensure that cells have enough time and the ability to remodel their environment before the scaffolding degrades.
- Dimensionality: Dimensionality is an extrinsic material property that defines the number of dimensions an object occupies. To make the most accurate model for adipose tissue fibrosis a 3-dimensional model is necessary. Typically, growing cells in 3D constructs is more difficult than 2D, but due to adipocytes’ unique buoyancy issues, and the fact that 2D cultures results in different morphological (multilocular lipid droplets as opposed to the single unilocular lipid droplet observed in vivo) and functional adipocytes, 3D culture is often pursued as a more physiologically relevant option. In particular, the largest difference is in cell volume; 2D culture results in adipocytes with less than 20% the total cell volume of lean subcutaneous adipocytes and less than 3% the total cell volume of obese subcutaneous adipocytes [102,103,104,105,106].
- Deformability: Deformability is an intrinsic material property that is defined as the ability for the material to change shape. The goal of designing a biomaterial to model adipose tissue fibrosis is to encapsulate adipocytes and the stromal vascular fraction (SVF) in a scaffold with properties similar to the ECM found in adipose tissues. One method is to begin with a highly deformable biomaterial that has similar properties to healthy adipose tissue and allow cells to remodel their environment by triggering fibrosis. Alternatively, by beginning with a biomaterial scaffold that has properties of fibrotic adipose tissue, and a low degree of deformability, fibrotic adipose tissue can be modeled. Depending on the experiment and the end goal, each method offers unique advantages.
- Plasticity: Plasticity is an intrinsic material property that is related to stiffness and viscoelasticity. Several studies have found that cells are able to plastically remodel certain biomaterial environments, such as collagen and fibrin gels [107,108,109]. Cells align and compact the fibers around them to the point where, when the cells are removed, voids remain [110]. Healthy adipose tissue is a dynamic organ that is remodeled constantly to allow fluctuations in adipocyte size to meet energy storage and demand needs. However, adipose tissue fibrosis limits the capacity of adipose tissue to remodel [111]. The addition of crosslinks can lower the accessibility of cells to remodel the biomaterial [112].
- Porosity: Porosity is an extrinsic property that is defined as the percentage of void space in a material. Generally, biomaterial scaffolds used to culture adipose tissue have high porosities (>90%) [83,84] to accommodate the large size of lipid-laden adipocytes. However, pore sizes vary considerably based on the method of formation (135–633 µm) [83,93]. For example, gas foaming results in a larger range of pore sizes and is more difficult to control compared to salt leaching [113]. Researchers that have used salt leaching to generate their scaffolds have chosen pore sizes ranging from 500–600 µm [93]. Electrospun scaffolds had slightly lower porosity (~88%) and considerably smaller pore sizes (6–70 µm); however, these scaffolds were used with murine ASC cell lines that are smaller and do not contain the large unilocular lipid droplet [89]. Three-dimensional printing can be used to create specific pore sizes but depends heavily on the print resolution.
- Processing: There are many methods of processing biomaterials. Some researchers have used electrospinning as a method of making scaffolds for in vitro adipose tissue modeling [89,90]. Electrospinning was chosen because the resulting scaffold has similar structure to collagen fibers found in the ECM matrix of adipose tissue. However, electrospinning resulted in lower porosity and smaller pore sizes. Three-dimensional bioprinting has been used to model other fibrotic diseases [114]. Printing with cells allows for more complex tissues to be created. Currently, printing vasculature is difficult, which limits the size of the print, as necrotic cores develop if cells do not have access to nutrients in large tissue constructs without vasculature. Three-dimensional printing also is not an option for all biomaterials. Decellularized tissue matrices have been used to study adipose tissue fibrosis [80], with conduits for vasculature to help sustain long term culture. However, decellularized tissue matrices can limit the cell-cell interactions and only represent the final stages of the disease [115]. Hydrogels are useful for tissue culture due to ease of fabrication and can be made using ECM proteins [81]. However, the mechanical properties of hydrogels have lower tunability.
- External Mechanical Forces: Including mechanical forces, such as tensile or compressive strains, is independent of material properties. Applying tensile strains of about 12% to adipocyte cultures resulted in faster accumulation of lipids and larger lipid droplets compared to adipocytes cultured under no external stresses [116]. This could be a useful technique to expedite the formation of a lipid-rich model.
4. Current Methods of Modeling Fibrotic Adipose Tissue In Vivo
Rodent
Animal models have been extensively used in obesity research. While it is beyond the scope of this review to delve into all animal models of obesity, many excellent reviews exist [117,118,119]. To investigate adipose tissues dynamics, mice are the most common animal model used. Mice are advantageous as they are readily available with well-established protocols for the development of monogenic (single genetic cause) and polygenic (surgical, chemical, dietary, or environmental causes) models [117], comparative historical data, a short lifespan and breeding cycle, and the ability to procure genetically identical strains [120]. Murine models can provide important whole-body information and have served as a useful tool in learning about adipose tissue fibrosis. Particularly relevant for exploring the dynamics of fibrotic adipose tissue, systemic effects (i.e., dynamic signaling with muscle, liver, etc.) and immune interactions can be explored that are not feasible in in vitro models.
Despite the extensive use of murine models, there are some significant drawbacks. For example, modeling obesity in mice requires dietary, genetic, and chemical modifications that limit their applicability to human translation [117]. Additionally, there are significant differences in location and purpose of fat deposits. Most murine research uses perigonadal fat pads as the adipose tissue source; however, humans do not have an analogous fat deposit [121]. Finally, differences between male and female mice and lipolysis mechanisms are not translatable to humans [121]. For example, adult female fat deposits are strongly influenced by reproductive hormones, while this is not seen in female rodents. Additionally, mice and human adipocytes respond differently to some lipolytic agents [122,123,124].
Among some of the most common animal models of obesity are: the leptin-deficient ob/ob mouse [125,126,127]; the leptin receptor deficient db/db mouse [125,128] and (the more specific) ss/ss mouse [129,130]; rat analogs with mutated leptin receptor domains: the Zucker and Koletsky rats [131,132]; deficits downstream from the leptin receptor: the proopiomelanocortin knockout [133,134], the melanocortin 4 receptor deficient mouse, and the Agouti related protein knockout [135]; and models of diet induced obesity [117]. Depending on the study design each model has advantages. Diet induced obesity in animals likely mimics human obesity more than genetically modified models (as mutations are very rare in humans) and are therefore a good fit for prospective therapeutics [117]. High fat diet models (>30% of energy from fat) are highly prevalent as dietary fat intake increases adiposity [136]. In particular, many diet-induced obesity models either use fat- and sugar-rich supermarket foods (cafeteria diet) or focus on saturated fatty acids and simple sugars (western diet). On the other hand, transgenic or spontaneous mutations are beneficial for exploring the role of specific molecular targets in the progression of obesity.
Obesity is closely linked to fibrosis, and thus many animal models of obesity naturally develop fibrosis. For example, leptin deficiency, as occurs in the ob/ob mouse results in adipose tissue fibrosis [28,137]. Likewise, collagens are highly upregulated in adipose tissue during metabolic challenges in the db/db mouse [28]. Currently, obese models of rats are being used to study fibrosis in other tissues, such as the pancreas (Zucker rats + high fat diet) [138] and liver (wistar rats + high fat diet) [139]; however, more research needs to focus on adipose tissue fibrosis in obese animal models.
Knockout models and overexpression models of genes are well-suited to study specific molecular targets involved in adipose tissue fibrosis. One study targeted periostin, an ECM protein that is secreted following a high fat diet in mice that amplifies inflammation, collagen cross-linking, and degradation of the ECM. In periostin knockout mice, there were fewer crown-like structures and reduced fibrosis compared to wildtype mice [140]. In another study, the same profibrotic genes (COL1A1, COL6A3, Lumican (LUM), Tensascin C (TNC)) overexpressed in humans with adipose tissue fibrosis [141] were overexpressed in C3H mice. In conjunction with a high fat diet, the C3H mice exhibited insulin resistance, collagen overproduction, increased macrophage activity, and adipocyte metabolic dysfunction. It was also determined that an important factor to consider in adipose tissue fibrosis models is the strain of mouse used, as different strains vary in levels of gene expression. Compared to the C57BL/6J mouse strain, which is often used in obesity research, the C3H strain showed earlier profibrotic gene expression [141], indicating it was a more accurate model of fibrosis. In another study, researchers studied gender differences with a knockout model. In mice, female adipose tissue has higher insulin sensitivities, less susceptibility to inflammation, and a higher expression of estrogen receptors, compared to male adipose tissue [142,143,144]. Additionally, in mice, adipose tissue estrogen receptor α (ERα) regulates body fat distribution, inflammation, and fibrosis [145]. They found that ERα knockout (αERKO) mice had enlarged adipocytes and higher degrees of inflammation and fibrosis in both males and females compared to wildtype [145]. Notably, female αERKO mice had a significantly higher expression of the COL6 gene, a gene related to ECM production and fibrosis. Taken together, these models highlight the important role of key proteins in the pro-fibrotic profile.
Another approach to model fibrosis is to target microRNAs (miRNAs), which are small noncoding RNAs that regulate gene expression [146,147]. Inflammatory and dysfunctional metabolic processes related to obesity have been linked to miRNAs. Specifically, it has been found that miRNA155 (miR155) plays an important role in adipocyte differentiation into the white phenotype and activation of proinflammatory pathways [148,149]. By deleting miR155 and feeding male mice a high fat diet to induce obesity, it was found that this deletion caused mice to store less visceral adipose tissue but exacerbated adipose tissue fibrosis compared to wildtype mice fed a high fat diet [150]. This was surprising because other groups have shown that miR155 plays a role in promoting macrophage polarization to the M1 phenotype [151,152,153]. By preventing the polarization of macrophages to the proinflammatory phenotype, it was thought that the prevalence of adipose tissue fibrosis would be decreased. It is important to note that the combination of miRNA155 deletion and a low-fat diet did not result in adipose tissue fibrosis. Conflicting results were gathered when a similar study was performed using female mice. Feeding miR155 female knockout mice a high fat diet resulted in lower degrees of obesity. Additionally, these mice had reduced inflammation and cell hypertrophy [154]. These results further illustrate that adipose tissue is sexually dimorphic.
The long-term goal of modeling fibrosis in animal models is not only to explore mechanisms of fibrosis but also to target treatments. Knockout models can be combined with traditional models of obesity to explore therapeutic targets. For example, knockout of collagen VI was explored in the ob/ob environment and with exposure to a high fat diet [28]. Interestingly, collagen VI deficiency resulted in an improvement of the metabolic phenotype in both the high-fat diet and ob/ob background. Similarly, in another knockout model, iNOS ablation in leptin-deficient mice (ob/ob) decreased fibrosis and metabolic dysfunction [137]. Towards the goal of clinical translation, a murine adipose tissue fibrosis model was used to investigate the effect of drugs on modulating adipose tissue fibrosis in a diet induced obesity model. In another study, Isoliquiritigenin (ILG) was used to treat adipose tissue fibrosis in a C57BL/6 diet induced obesity model. Mice fed a high fat diet and given ILG showed reduced fibrotic area, TNFα, COL1, and TGFβ1 expression compared to control mice. Importantly, there were no significant differences between high fat diet plus ILG and normal diet [155]. Overall, these studies emphasize that while rodent models may not directly translate to human clinical outcomes, they provide an important pre-clinical tool for controlling confounding variables and determining systemic interactions, immune responses, and genetic influences.
5. Human Epidemiological Studies
Human epidemiological studies are essential for correlating mechanistic data from in vitro and in vivo studies to fibrotic outcomes clinically. However, human studies on adipose tissue fibrosis are limited and inconsistent.
In human adipose tissue fibrosis studies, one of the most well studied comorbidities is the effect of diabetes. One study examined the effect of bariatric surgery on diabetic versus insulin-sensitive patients. Using patients with similar age, body mass index, and fat mass, the study showed that before surgery diabetic patients had a significantly higher degree of adipose tissue fibrosis. Six months after bariatric surgery, fibrosis levels had not decreased, even with significant weight loss [79]. However, contradictory conclusions were gathered from another research group looking at adipose tissue samples taken during bariatric surgery. It was concluded that adipose tissue fibrosis was less prevalent in obese patients with diabetes, but adipose hypertrophy was more common in these patients [21]. Additionally, fewer stem cells were found in the adipose tissue samples taken from the diabetic patients [21]. These conflicting results indicate that comorbidities have varying effects on adipose tissue fibrosis and patient demographics should be more closely considered when drawing conclusions. A study comparing healthy, non-obese patients with a predisposition for type 2 diabetes to control patients found that patients with a predisposition had higher degrees of adipocyte hypertrophy, inflammation, and Wnt-signal activation [156]. All these factors contribute to adipose tissue dysfunction. This study further supports that patient demographics should be heavily considered when generalizing adipose tissue fibrosis mechanisms and effects.
In recent years, more diverse patient demographics (race, gender, disease status, etc.) have been used to investigate their correlation to adipose tissue fibrosis. A recent report investigated the link between obesity, insulin resistance, and adipose tissue fibrosis in Chinese Americans [157]. Asian Americans tend to develop type 2 diabetes at lower BMI values compared to White Americans [157,158]. The researchers found that insulin resistance was more strongly correlated to subcutaneous adipose tissue fibrosis than the patient’s body mass index in the Asian American population. Another study examined if there was a difference between patients that had been infected with human immunodeficiency virus (HIV) versus uninfected patients [159]. Patients with HIV related lipoatrophy had a higher degree of fibrosis and adipocyte apoptosis compared to uninfected patients but were unable to conclude if the fibrosis caused insulin resistance [111,159]. Future studies are required to categorize different demographics and their predisposition for adipose tissue fibrosis.
Clinically, adipose tissue fibrosis can be used as a predictor of health-related outcomes, demonstrating the importance of considering it in treatment options. A subcutaneous adipose tissue scoring model was developed to predict weight-loss outcomes after gastric bypass based on the degree of fibrosis in a patient’s adipose tissue [160]. These scores were correlated to the patients’ weight loss after gastric bypass to train a machine learning algorithm, providing semiquantitative, reproducible fibrosis scores. The researchers also determined that the higher the fibrosis score, or the more fibrotic the tissue, the less successful weight-loss was after gastric bypass [160]. Together, these studies reinforce the importance of diagnosing fibrosis in patients to inform better treatment strategies.
6. Adipose Tissue’s Role in Treating Other Fibrotic Diseases
Cells sourced from adipose tissue, particularly the SVF, have been used to treat other fibrotic diseases and tissues (Table 2). The SVF consists of all non-adipocyte cell types, including ASCs, endothelial cells, immune cells, and fibroblasts. ASCs are an attractive therapeutic option as they secrete growth factors, cytokines, proteins, and exosomes that promote regeneration [161,162] and have been used to treat systemic sclerosis, as well as dermal, liver, cardiac, renal, muscular, and lung fibrosis [163,164,165,166,167,168,169,170,171,172]. For example, injecting hASCs systemically reduced kidney fibrosis, improved renal functions, and reduced profibrotic gene expression [168,169]. Many of these treatments are effective by inhibiting the inflammatory response through the TGFβ1 signaling pathway [168,171] and reducing collagen deposition [171].

Table 2.
Synopsis of research that used cells and vesicles sourced from adipose tissue to treat other fibrotic diseases.
Adipose derived exosomes are another potential therapeutic option. The effect of media conditioned in the presence of ASCs and ASC injections were investigated for treatment of pulmonary fibrosis. Not only did ASCs improve pulmonary fibrosis [173], but secretions in conditioned media also successfully had therapeutic effects [172]. The secretions in the conditioned media included exosomes, which several researchers have used to treat liver fibrosis [174,175]. Exosomes offer a cell free therapy for treating fibrotic diseases. Furthermore, the mechanism is similar, as microRNA181 in the exosomes, reduced TGFβ1 expression, and had in vitro and in vivo therapeutic effects [175].
The significance of these treatments underscores the importance of investigating and modeling adipose tissue fibrosis. These treatments all rely on adipose derived cells; however, adipose tissue fibrosis significantly alters ASCs function and morphology. For these treatments to effectively work a stronger understanding of how adipose tissue fibrosis affects adipose derived stem cells and their secretome at different stages of fibrosis is required.
7. Methods for Evaluating Degree of Fibrosis
Detecting and evaluating adipose tissue fibrosis using reproducible, standardized techniques is essential for model development. Information gathered from these techniques in situ can be applied to engineered models to increase their clinical relevance, demonstrating consistency with clinical outcomes. Researchers have used multiple methods to evaluate the presence or degree of fibrosis through staining, imaging, mechanical testing, etc. (Table 3). Each of these techniques offers unique information and has advantages and disadvantages.

Table 3.
Techniques used by researchers to evaluate the presence and degree of adipose tissue fibrosis.
8. Conclusions
While adipose tissue fibrosis has been investigated for several decades, there is still an incomplete understanding of causes, effects, and mechanisms. Under periods of high caloric consumption, the increase in adipocyte volume and tissue mass results in inflammation, metabolic dysregulation, and adipose tissue fibrosis in some individuals, but not all.
To address many of the open-ended questions surrounding adipose tissue fibrosis, in vitro and in vivo model systems should be carefully chosen to meet the needs of the research question. Three-dimensional in vitro models are an area of adipose tissue fibrosis research that has a high potential for growth. Improvements to this field would drastically enhance our understanding of the disease by creating physiologically relevant models where factors can be carefully manipulated, and treatments can be investigated in human samples. While using a biomaterial scaffold to model adipose tissue fibrosis would offer a high throughput, inexpensive disease model, the tradeoff is a lack of the complexities found in in vivo animal models, including systemic effects and immune responses [81]. Human epidemiological studies vary considerably depending on experimental setup and subject demographics. The effects of race, gender, and comorbidities have just begun to be investigated and appear to have a significant effect on adipose tissue fibrosis [157,158,159]. More in-depth studies and integration of existing datasets [160] are needed to further investigate how patient demographics affect adipose tissue fibrosis.
Currently, there is also a large amount of research in treating other fibrotic diseases using cells and secreted vesicles from adipose tissue. As these treatments rely on cells functioning similarly in all patients the decreased ability to differentiate and exhibit a proinflammatory phenotype in fibrotic tissues is concerning.
Overall, adipose tissue fibrosis is not routinely screened for; however, it can have profound effects on patient outcomes and therapeutic options. With the rate of obesity increasing globally, it is essential that we develop a better understanding of fibrotic causes and mechanisms, as well as create better models to study potential treatments.
Author Contributions
Conceptualization, M.K.D. and R.D.A.; Literature Review, M.K.D. and R.D.A.; Figures and Tables, M.K.D.; Writing—Original Draft Preparation, M.K.D.; Writing—Review & Editing, R.D.A. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Saely:, C.H.; Geiger, K.; Drexel, H. Brown versus white adipose tissue: A mini-review. Gerontology 2012, 58, 15–23. [Google Scholar] [CrossRef] [PubMed]
- Lass, A.; Zimmermann, R.; Oberer, M.; Zechner, R. Lipolysis—A highly regulated multi-enzyme complex mediates the catabolism of cellular fat stores. Prog. Lipid Res. 2011, 50, 14–27. [Google Scholar] [CrossRef]
- Lelliott, C.; Vidal-Puig, A. Lipotoxicity, an imbalance between lipogenesis de novo and fatty acid oxidation. Int. J. Obes. 2004, 28, S22–S28. [Google Scholar] [CrossRef] [PubMed]
- Verboven, K.; Wouters, K.; Gaens, K.; Hansen, D.; Bijnen, M.; Wetzels, S.; Stehouwer, C.D.; Goossens, G.H.; Schalkwijk, C.G.; Blaak, E.E.; et al. Abdominal subcutaneous and visceral adipocyte size, lipolysis and inflammation relate to insulin resistance in male obese humans. Sci. Rep. 2018, 8, 4677. [Google Scholar] [CrossRef] [PubMed]
- Gustafson, B.; Gogg, S.; Hedjazifar, S.; Jenndahl, L.; Hammarstedt, A.; Smith, U. Inflammation and impaired adipogenesis in hypertrophic obesity in man. Am. J. Physiol. Metab. 2009, 297, E999–E1003. [Google Scholar] [CrossRef] [PubMed]
- Buechler, C.; Krautbauer, S.; Eisinger, K. Adipose tissue fibrosis. World J. Diabetes 2015, 6, 548–553. [Google Scholar] [CrossRef]
- Mariman, E.C.M.; Wang, P. Adipocyte extracellular matrix composition, dynamics and role in obesity. Cell. Mol. Life Sci. 2010, 67, 1277–1292. [Google Scholar] [CrossRef]
- Berger, J.J.; Barnard, R.J. Effect of diet on fat cell size and hormone-sensitive lipase activity. J. Appl. Physiol. 1999, 87, 227–232. [Google Scholar] [CrossRef]
- Wueest, S.; Rapold, R.A.; Rytka, J.M.; Schoenle, E.J.; Konrad, D. Basal lipolysis, not the degree of insulin resistance, differentiates large from small isolated adipocytes in high-fat fed mice. Diabetologia 2008, 52, 541–546. [Google Scholar] [CrossRef]
- Jocken, J.W.E.; Goossens, G.H.; Van Hees, A.M.J.; Frayn, K.N.; Van Baak, M.; Stegen, J.; Pakbiers, M.T.W.; Saris, W.H.M.; Blaak, E.E. Effect of beta-adrenergic stimulation on whole-body and abdominal subcutaneous adipose tissue lipolysis in lean and obese men. Diabetologia 2007, 51, 320–327. [Google Scholar] [CrossRef]
- Reynisdottir, S.; Ellerfeldt, K.; Wahrenberg, H.; Lithell, H.; Arner, P. Multiple lipolysis defects in the insulin resistance (metabolic) syndrome. J. Clin. Investig. 1994, 93, 2590–2599. [Google Scholar] [CrossRef]
- Campbell, P.J.; Carlson, M.G.; Nurjhan, N. Fat metabolism in human obesity. Am. J. Physiol. Metab. 1994, 266, E600–E605. [Google Scholar] [CrossRef] [PubMed]
- Renes, J.; Bouwman, F.; Noben, J.-P.; Evelo, C.; Robben, J.; Mariman, E. Protein profiling of 3T3-L1 adipocyte differentiation and (tumor necrosis factor alpha-mediated) starvation. Cell. Mol. Life Sci. 2005, 62, 492–503. [Google Scholar] [CrossRef] [PubMed]
- Sun, K.; Tordjman, J.; Clément, K.; Scherer, P.E. Fibrosis and adipose tissue dysfunction. Cell Metab. 2013, 18, 470–477. [Google Scholar] [CrossRef] [PubMed]
- Lawler, H.M.; Underkofler, C.M.; Kern, P.A.; Erickson, C.; Bredbeck, B.; Rasouli, N. Adipose tissue hypoxia, inflammation, and fibrosis in obese insulin-sensitive and obese insulin-resistant subjects. J. Clin. Endocrinol. Metab. 2016, 101, 1422–1428. [Google Scholar] [CrossRef] [PubMed]
- Buchwald, H.; Avidor, Y.; Braunwald, E. Bariatric surgery. A systematic review and meta-analysis. ACC Curr. J. Rev. 2005, 14, 1724–1737. [Google Scholar] [CrossRef]
- Dandona, P.; Aljada, A.; Chaudhuri, A.; Mohanty, P.; Garg, R. Metabolic syndrome: A comprehensive perspective based on interactions between obesity, diabetes, and inflammation. Circulation 2005, 111, 1448–1454. [Google Scholar] [CrossRef]
- Dankel, S.N.; Svard, J.; Mattha, S.; Claussnitzer, M.; Kloting, N.; Glunk, V.; Fandalyuk, Z.; Grytten, E.; Solsvik, M.H.; Nielsen, H.J.; et al. COL6A3 expression in adipocytes associates with insulin resistance and depends on PPARgamma and adipocyte size. Obesity 2014, 22, 1807–1813. [Google Scholar] [CrossRef]
- Divoux, A.; Tordjman, J.; Lacasa, D.; Veyrie, N.; Hugol, D.; Aissat, A.; Basdevant, A.; Guerre-Millo, M.; Poitou, C.; Zucker, J.D.; et al. Fibrosis in human adipose tissue: Composition, distribution, and link with lipid metabolism and fat mass loss. Diabetes 2010, 59, 2817–2825. [Google Scholar] [CrossRef]
- Lackey, D.E.; Burk, D.H.; Ali, M.R.; Mostaedi, R.; Smith, W.H.; Park, J.; Scherer, P.E.; Seay, S.A.; McCoin, C.S.; Bonaldo, P.; et al. Contributions of adipose tissue architectural and tensile properties toward defining healthy and unhealthy obesity. Am. J. Physiol. Metab. 2014, 306, E233–E246. [Google Scholar] [CrossRef]
- Muir, L.A.; Neeley, C.K.; Meyer, K.A.; Baker, N.A.; Brosius, A.M.; Washabaugh, A.R.; Varban, O.A.; Finks, J.F.; Zamarron, B.F.; Flesher, C.G.; et al. Adipose tissue fibrosis, hypertrophy, and hyperplasia: Correlations with diabetes in human obesity. Obesity 2016, 24, 597–605. [Google Scholar] [CrossRef] [PubMed]
- Sun, K.; Park, J.; Gupta, O.T.; Holland, W.L.; Auerbach, P.; Zhang, N.; Marangoni, R.G.; Nicoloro, S.M.; Czech, M.P.; Varga, J.; et al. Endotrophin triggers adipose tissue fibrosis and metabolic dysfunction. Nat. Commun. 2014, 5, 3485. [Google Scholar] [CrossRef] [PubMed]
- Spencer, M.; Yao-Borengasser, A.; Unal, R.; Rasouli, N.; Gurley, C.M.; Zhu, B.; Peterson, C.A.; Kern, P.A. Adipose tissue macrophages in insulin-resistant subjects are associated with collagen VI and fibrosis and demonstrate alternative activation. Am. J. Physiol. Metab. 2010, 299, E1016–E1027. [Google Scholar] [CrossRef]
- Divoux, A.; Clement, K. Architecture and the extracellular matrix: The still unappreciated components of the adipose tissue. Obes. Rev. 2011, 12, 494–503. [Google Scholar] [CrossRef] [PubMed]
- Datta, R.; Podolsky, M.J.; Atabai, K. Fat fibrosis: Friend or foe? JCI Insight 2018, 3, 19. [Google Scholar] [CrossRef] [PubMed]
- Huber, J.; Löffler, M.; Bilban, M.; Reimers, M.; Kadl, A.; Todoric, J.; Zeyda, M.; Geyeregger, R.; Schreiner, M.; Weichhart, T.; et al. Prevention of high-fat diet-induced adipose tissue remodeling in obese diabetic mice by n-3 polyunsaturated fatty acids. Int. J. Obes. 2006, 31, 1004–1013. [Google Scholar] [CrossRef]
- Spencer, M.; Unal, R.; Zhu, B.; Rasouli, N.; McGehee, R.E.; Peterson, C.A.; Kern, P.A. Adipose tissue extracellular matrix and vascular abnormalities in obesity and insulin resistance. J. Clin. Endocrinol. Metab. 2011, 96, E1990–E1998. [Google Scholar] [CrossRef]
- Khan, T.; Muise, E.S.; Iyengar, P.; Wang, Z.V.; Chandalia, M.; Abate, N.; Zhang, B.B.; Bonaldo, P.; Chua, S.; Scherer, P.E. Metabolic dysregulation and adipose tissue fibrosis: Role of collagen VI. Mol. Cell. Biol. 2008, 29, 1575–1591. [Google Scholar] [CrossRef]
- Marcelin, G.; Silveira, A.L.M.; Martins, L.B.; Ferreira, A.V.; Clément, K. Deciphering the cellular interplays underlying obesity-induced adipose tissue fibrosis. J. Clin. Investig. 2019, 129, 4032–4040. [Google Scholar] [CrossRef]
- Giordano, A.; Murano, I.; Mondini, E.; Perugini, J.; Smorlesi, A.; Severi, I.; Barazzoni, R.; Scherer, P.E.; Cinti, S. Obese adipocytes show ultrastructural features of stressed cells and die of pyroptosis. J. Lipid Res. 2013, 54, 2423–2436. [Google Scholar] [CrossRef]
- Jernås, M.; Palming, J.; Sjöholm, K.; Jennische, E.; Svensson, P.A.; Gabrielsson, B.G.; Levin, M.; Sjögren, A.; Rudemo, M.; Lystig, T.; et al. Separation of human adipocytes by size: Hypertrophic fat cells display distinct gene expression. FASEB J. 2006, 20, 1540–1542. [Google Scholar] [CrossRef] [PubMed]
- Sartipy, P.; Loskutoff, D.J. Monocyte chemoattractant protein 1 in obesity and insulin resistance. Proc. Natl. Acad. Sci. USA 2003, 100, 7265–7270. [Google Scholar] [CrossRef] [PubMed]
- Jang, J.E.; Ko, M.S.; Yun, J.Y.; Kim, M.O.; Kim, J.H.; Park, H.S.; Kim, A.R.; Kim, H.J.; Kim, B.J.; Ahn, Y.E.; et al. Nitric oxide produced by macrophages inhibits adipocyte differentiation and promotes profibrogenic responses in preadipocytes to induce adipose tissue fibrosis. Diabetes 2016, 65, 2516–2528. [Google Scholar] [CrossRef] [PubMed]
- Trayhurn, P.; Wang, B.; Wood, I.S. Hypoxia in adipose tissue: A basis for the dysregulation of tissue function in obesity? Br. J. Nutr. 2008, 100, 227–235. [Google Scholar] [CrossRef]
- Weisberg, S.P.; McCann, D.; Desai, M.; Rosenbaum, M.; Leibel, R.L.; Ferrante, A.W., Jr. Obesity is associated with macrophage accumulation in adipose tissue. J. Clin. Investig. 2003, 112, 1796–1808. [Google Scholar] [CrossRef] [PubMed]
- Hotamisligil, G.S.; Arner, P.; Caro, J.F.; Atkinson, R.L.; Spiegelman, B.M. Increased adipose tissue expression of tumor necrosis factor-alpha in human obesity and insulin resistance. J. Clin. Investig. 1995, 95, 2409–2415. [Google Scholar] [CrossRef]
- Hotamisligil, G.; Shargill, N.; Spiegelman, B. Adipose expression of tumor necrosis factor-alpha: Direct role in obesity-linked insulin resistance. Science 1993, 259, 87–91. [Google Scholar] [CrossRef]
- Fried, S.K.; Bunkin, D.A.; Greenberg, A.S. Omental and subcutaneous adipose tissues of obese subjects release interleukin-6: Depot difference and regulation by glucocorticoid. J. Clin. Endocrinol. Metab. 1998, 83, 847–850. [Google Scholar] [CrossRef]
- Perreault, M.; Marette, A. Targeted disruption of inducible nitric oxide synthase protects against obesity-linked insulin resistance in muscle. Nat. Med. 2001, 7, 1138–1143. [Google Scholar] [CrossRef]
- Visser, M.; Bouter, L.M.; McQuillan, G.M.; Wener, M.H.; Harris, T.B. Elevated C-reactive protein levels in overweight and obese adults. JAMA 1999, 282, 2131–2135. [Google Scholar] [CrossRef]
- Jones, J.E.C.; Rabhi, N.; Orofino, J.; Gamini, R.; Perissi, V.; Vernochet, C.; Farmer, S.R. The adipocyte acquires a fibroblast-like transcriptional signature in response to a high fat diet. Sci. Rep. 2020, 10, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Marcelin, G.; Ferreira, A.; Liu, Y.; Atlan, M.; Aron-Wisnewsky, J.; Pelloux, V.; Botbol, Y.; Ambrosini, M.; Fradet, M.; Rouault, C.; et al. A PDGFRalpha-mediated switch toward CD9(high) adipocyte progenitors controls obesity-induced adipose tissue fibrosis. Cell Metab. 2017, 25, 673–685. [Google Scholar] [CrossRef] [PubMed]
- Iwayama, T.; Steele, C.; Yao, L.; Dozmorov, M.G.; Karamichos, D.; Wren, J.D.; Olson, L.E. PDGFRalpha signaling drives adipose tissue fibrosis by targeting progenitor cell plasticity. Genes Dev. 2015, 29, 1106–1119. [Google Scholar] [CrossRef]
- Engin, A. Adipose tissue hypoxia in obesity and its impact on preadipocytes and macrophages: Hypoxia hypothesis. Adv. Exp. Med. Biol. 2017, 960, 305–326. [Google Scholar] [CrossRef]
- Cao, Y. Angiogenesis modulates adipogenesis and obesity. J. Clin. Investig. 2007, 117, 2362–2368. [Google Scholar] [CrossRef]
- Pellegrinelli, V.; Rouault, C.; Veyrie, N.; Clement, K.; Lacasa, D. Endothelial cells from visceral adipose tissue disrupt adipocyte functions in a three-dimensional setting: Partial rescue by angiopoietin-1. Diabetes 2013, 63, 535–549. [Google Scholar] [CrossRef]
- Crewe, C.; An, Y.A.; Scherer, P.E. The ominous triad of adipose tissue dysfunction: Inflammation, fibrosis, and impaired angiogenesis. J. Clin. Investig. 2017, 127, 74–82. [Google Scholar] [CrossRef]
- Lumeng, C.N.; Bodzin, J.L.; Saltiel, A. Obesity induces a phenotypic switch in adipose tissue macrophage polarization. J. Clin. Investig. 2007, 117, 175–184. [Google Scholar] [CrossRef]
- Wynn, T.A.; Chawla, A.; Pollard, J.W. Macrophage biology in development, homeostasis and disease. Nature 2013, 496, 445–455. [Google Scholar] [CrossRef]
- Xu, H.; Barnes, G.T.; Yang, Q.; Tan, G.; Yang, D.; Chou, C.J.; Sole, J.; Nichols, A.; Ross, J.S.; Tartaglia, L.A.; et al. Chronic inflammation in fat plays a crucial role in the development of obesity-related insulin resistance. J. Clin. Investig. 2003, 112, 1821–1830. [Google Scholar] [CrossRef]
- Tanaka, M.; Ikeda, K.; Suganami, T.; Komiya, C.; Ochi, K.; Shirakawa, I.; Hamaguchi, M.; Nishimura, S.; Manabe, I.; Matsuda, T.; et al. Macrophage-inducible C-type lectin underlies obesity-induced adipose tissue fibrosis. Nat. Commun. 2014, 5, 4982. [Google Scholar] [CrossRef] [PubMed]
- Murano, I.; Barbatelli, G.; Parisani, V.; Latini, C.; Muzzonigro, G.; Castellucci, M.; Cinti, S. Dead adipocytes, detected as crown-like structures, are prevalent in visceral fat depots of genetically obese mice. J. Lipid Res. 2008, 49, 1562–1568. [Google Scholar] [CrossRef] [PubMed]
- Keophiphath, M.; Achard, V.; Henegar, C.; Rouault, C.; Clément, K.; Lacasa, D. Macrophage-secreted factors promote a profibrotic phenotype in human preadipocytes. Mol. Endocrinol. 2009, 23, 11–24. [Google Scholar] [CrossRef] [PubMed]
- Lin, D.; Chun, T.H.; Kang, L. Adipose extracellular matrix remodelling in obesity and insulin resistance. Biochem. Pharmacol. 2016, 119, 8–16. [Google Scholar] [CrossRef] [PubMed]
- Hopps, E.; Canino, B.; Montana, M.; Calandrino, V.; Urso, C.; Presti, R.L.; Caimi, G. Gelatinases and their tissue inhibitors in a group of subjects with obstructive sleep apnea syndrome. Clin. Hemorheol. Microcirc. 2016, 62, 27–34. [Google Scholar] [CrossRef] [PubMed]
- Berg, G.; Barchuk, M.; Miksztowicz, V. Behavior of metalloproteinases in adipose tissue, liver and arterial wall: An update of extracellular matrix remodeling. Cells 2019, 8, 158. [Google Scholar] [CrossRef]
- Gurung, P.; Moussa, K.; Adams-Huet, B.; Devaraj, S.; Jialal, I. Increased mast cell abundance in adipose tissue of metabolic syndrome: Relevance to the proinflammatory state and increased adipose tissue fibrosis. Am. J. Physiol. Metab. 2019, 316, E504–E509. [Google Scholar] [CrossRef]
- Hirai, S.; Ohyane, C.; Kim, Y.I.; Lin, S.; Goto, T.; Takahashi, N.; Kim, C.S.; Kang, J.; Yu, R.; Kawada, T. Involvement of mast cells in adipose tissue fibrosis. Am. J. Physiol. Metab. 2014, 306, E247–E255. [Google Scholar] [CrossRef]
- Frungieri, M.B.; Weidinger, S.; Meineke, V.; Köhn, F.M.; Mayerhofer, A. Proliferative action of mast-cell tryptase is mediated by PAR2, COX2, prostaglandins, and PPAR: Possible relevance to human fibrotic disorders. Proc. Natl. Acad. Sci. USA 2002, 99, 15072–15077. [Google Scholar] [CrossRef]
- Mukai, K.; Tsai, M.; Saito, H.; Galli, S.J. Mast cells as sources of cytokines, chemokines, and growth factors. Immunol. Rev. 2018, 282, 121–150. [Google Scholar] [CrossRef]
- Divoux, A.; Moutel, S.; Poitou, C.; Lacasa, D.; Veyrie, N.; Aissat, A.; Arock, M.; Guerre-Millo, M.; Clément, K. Mast cells in human adipose tissue: Link with morbid obesity, inflammatory status, and Diabetes. J. Clin. Endocrinol. Metab. 2012, 97, E1677–E1685. [Google Scholar] [CrossRef] [PubMed]
- Rajbhandari, P.; Arneson, D.; Hart, S.K.; Ahn, I.S.; Diamante, G.; Santos, L.C.; Zaghari, N.; Feng, A.C.; Thomas, B.J.; Vergnes, L.; et al. Single cell analysis reveals immune cell–adipocyte crosstalk regulating the transcription of thermogenic adipocytes. eLife 2019, 8, 8. [Google Scholar] [CrossRef]
- Merrick, D.; Sakers, A.; Irgebay, Z.; Okada, C.; Calvert, C.; Morley, M.; Percec, I.; Seale, P. Identification of a mesenchymal progenitor cell hierarchy in adipose tissue. Science 2019, 364, eaav2501. [Google Scholar] [CrossRef] [PubMed]
- Schwalie, P.C.; Dong, H.; Zachara, M.; Russeil, J.; Alpern, D.; Akchiche, N.; Caprara, C.; Sun, W.; Schlaudraff, K.U.; Soldati, G.; et al. A stromal cell population that inhibits adipogenesis in mammalian fat depots. Nature 2018, 559, 103–108. [Google Scholar] [CrossRef]
- Hepler, C.; Shan, B.; Zhang, Q.; Henry, G.H.; Shao, M.; Vishvanath, L.; Ghaben, A.L.; Mobley, A.B.; Strand, D.W.; Hon, G.C.; et al. Identification of functionally distinct fibro-inflammatory and adipogenic stromal subpopulations in visceral adipose tissue of adult mice. eLife 2018, 7, 7. [Google Scholar] [CrossRef] [PubMed]
- Burl, R.B.; Ramseyer, V.D.; Rondini, E.A.; Pique-Regi, R.; Lee, Y.H.; Granneman, J.G. Deconstructing adipogenesis induced by beta3-adrenergic receptor activation with single-cell expression profiling. Cell Metab. 2018, 28, 300–309. [Google Scholar] [CrossRef]
- Vijay, J.; Gauthier, M.F.; Biswell, R.L.; Louiselle, D.A.; Johnston, J.; Cheung, W.A.; Belden, B.; Pramatarova, A.; Biertho, L.; Gibson, M.; et al. Single-cell analysis of human adipose tissue identifies depot- and disease-specific cell types. Nat. Metab. 2019, 2, 97–109. [Google Scholar] [CrossRef]
- Cho, D.S.; Lee, B.; Doles, J.D. Refining the adipose progenitor cell landscape in healthy and obese visceral adipose tissue using single-cell gene expression profiling. Life Sci. Alliance 2019, 2, e201900561. [Google Scholar] [CrossRef]
- Zhang, H.; Kumar, S.; Barnett, A.H.; Eggo, M.C. Ceiling culture of mature human adipocytes: Use in studies of adipocyte functions. J. Endocrinol. 2000, 164, 119–128. [Google Scholar] [CrossRef]
- Sugihara, H.; Yonemitsu, N.; Miyabara, S.; Toda, S. Proliferation of unilocular fat cells in the primary culture. J. Lipid Res. 1987, 28, 1038–1045. [Google Scholar]
- Sugihara, H.; Yonemitsu, N.; Miyabara, S.; Yun, K. Primary cultures of unilocular fat cells: Characteristics of growth in vitro and changes in differentiation properties. Differentiation 1986, 31, 42–49. [Google Scholar] [CrossRef] [PubMed]
- Hwang, C.S.; Loftus, T.M.; Mandrup, S.; Lane, M.D. Adipocyte differentiation and leptin expression. Annu. Rev. Cell Dev. Biol. 1997, 13, 231–259. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.; Song, K.; Ahn, K.S. Alantolactone improves palmitate-induced glucose intolerance and inflammation in both lean and obese states in vitro: Adipocyte and adipocyte-macrophage co-culture system. Int. Immunopharmacol. 2017, 49, 187–194. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.F.; Craig, C.M.; Tolentino, L.L.; Choi, O.; Morton, J.; Rivas, H.; Cushman, S.W.; Engleman, E.G.; McLaughlin, T. Adipose tissue macrophages impair preadipocyte differentiation in humans. PLoS ONE 2017, 12, e0170728. [Google Scholar] [CrossRef] [PubMed]
- Suganami, T.; Nishida, J.; Ogawa, Y. A paracrine loop between adipocytes and macrophages aggravates inflammatory changes. Arter. Thromb. Vasc. Biol. 2005, 25, 2062–2068. [Google Scholar] [CrossRef]
- Yamashita, A.; Soga, Y.; Iwamoto, Y.; Yoshizawa, S.; Iwata, H.; Kokeguchi, S.; Takashiba, S.; Nishimura, F. Macrophage-adipocyte interaction: Marked interleukin-6 production by Lipopolysaccharide**. Obesity 2007, 15, 2549–2552. [Google Scholar] [CrossRef]
- Lacasa, D.; Taleb, S.; Keophiphath, M.; Miranville, A.; Clément, K. Macrophage-secreted factors impair human adipogenesis: Involvement of proinflammatory state in preadipocytes. Endocrinology 2007, 148, 868–877. [Google Scholar] [CrossRef]
- Isakson, P.; Hammarstedt, A.; Gustafson, B.; Smith, U. Impaired preadipocyte differentiation in human abdominal obesity. Diabetes 2009, 58, 1550–1557. [Google Scholar] [CrossRef]
- Chabot, K.; Gauthier, M.S.; Garneau, P.; Rabasa-Lhoret, R. Evolution of subcutaneous adipose tissue fibrosis after bariatric surgery. Diabetes Metab. 2017, 43, 125–133. [Google Scholar] [CrossRef]
- Baker, N.A.; Muir, L.A.; Washabaugh, A.R.; Neeley, C.K.; Chen, Y.-P.; Flesher, C.G.; Vorwald, J.; Finks, J.F.; Ghaferi, A.A.; Mulholland, M.W.; et al. Diabetes-specific regulation of adipocyte metabolism by the adipose tissue extracellular matrix. J. Clin. Endocrinol. Metab. 2017, 102, 1032–1043. [Google Scholar] [CrossRef]
- Di Caprio, N.; Bellas, E. Collagen stiffness and architecture regulate fibrotic gene expression in engineered adipose tissue. Adv. Biosyst. 2020, 4, e1900286. [Google Scholar] [CrossRef] [PubMed]
- Rajangam, T.; Park, M.H.; Kim, S.H. 3D Human adipose-derived stem cell clusters as a model for in vitro fibrosis. Tissue Eng. Part. C Methods 2016, 22, 679–690. [Google Scholar] [CrossRef] [PubMed]
- Patrick, C.W.; Chauvin, P.; Hobley, J.; Reece, G. Preadipocyte seeded PLGA scaffolds for adipose tissue engineering. Tissue Eng. 1999, 5, 139–151. [Google Scholar] [CrossRef] [PubMed]
- Patrick, C.W.; Zheng, B.; Johnston, C.; Reece, G.P. Long-term implantation of preadipocyte-seeded PLGA scaffolds. Tissue Eng. 2002, 8, 283–293. [Google Scholar] [CrossRef] [PubMed]
- Choi, Y.S.; Cha, S.M.; Lee, Y.Y.; Kwon, S.W.; Park, C.; Kim, M. Adipogenic differentiation of adipose tissue derived adult stem cells in nude mouse. Biochem. Biophys. Res. Commun. 2006, 345, 631–637. [Google Scholar] [CrossRef]
- Neubauer, M.; Hacker, M.; Bauer-Kreisel, P.; Weiser, B.; Fischbach, C.; Schulz, M.B.; Goepferich, A.; Blunk, T. Adipose tissue engineering based on mesenchymal stem cells and basic fibroblast growth factor in vitro. Tissue Eng. 2005, 11, 1840–1851. [Google Scholar] [CrossRef]
- Halbleib, M.; Skurk, T.; De Luca, C.; Von Heimburg, D.; Hauner, H. Tissue engineering of white adipose tissue using hyaluronic acid-based scaffolds. I: In vitro differentiation of human adipocyte precursor cells on scaffolds. Biomaterials 2003, 24, 3125–3132. [Google Scholar] [CrossRef]
- Kessler, L.; Gehrke, S.; Winnefeld, M.; Huber, B.; Hoch, E.; Walter, T.; Wyrwa, R.; Schnabelrauch, M.; Schmidt, M.; Kückelhaus, M.; et al. Methacrylated gelatin/hyaluronan-based hydrogels for soft tissue engineering. J. Tissue Eng. 2017, 8, 2041731417744157. [Google Scholar] [CrossRef]
- Kang, X.; Xie, Y.; Powell, H.M.; Lee, L.J.; Belury, M.A.; Lannutti, J.J.; Kniss, D.A. Adipogenesis of murine embryonic stem cells in a three-dimensional culture system using electrospun polymer scaffolds. Biomaterials 2007, 28, 450–458. [Google Scholar] [CrossRef]
- Brännmark, C.; Paúl, A.; Ribeiro, D.; Magnusson, B.; Brolén, G.; Enejder, A.; Forslöw, A. Increased adipogenesis of human adipose-derived stem cells on polycaprolactone fiber matrices. PLoS ONE 2014, 9, e113620. [Google Scholar] [CrossRef]
- Krontiras, P.; Gatenholm, P.; Hägg, D.A. Adipogenic differentiation of stem cells in three-dimensional porous bacterial nanocellulose scaffolds. J. Biomed. Mater. Res. Part. B Appl. Biomater. 2014, 103, 195–203. [Google Scholar] [CrossRef] [PubMed]
- Volz, A.C.; Hack, L.; Kluger, P.J. A cellulose-based material for vascularized adipose tissue engineering. J. Biomed. Mater. Res. Part. B Appl. Biomater. 2018, 107, 1431–1439. [Google Scholar] [CrossRef] [PubMed]
- Mauney, J.R.; Nguyen, T.; Gillen, K.M.; Kirker-Head, C.; Gimble, J.M.; Kaplan, D.L. Engineering adipose-like tissue in vitro and in vivo utilizing human bone marrow and adipose-derived mesenchymal stem cells with silk fibroin 3D scaffolds. Biomaterials 2007, 28, 5280–5290. [Google Scholar] [CrossRef] [PubMed]
- Abbott, R.D.; Wang, R.Y.; Reagan, M.R.; Chen, Y.; Borowsky, F.E.; Zieba, A.; Marra, K.G.; Rubin, J.P.; Ghobrial, I.M.; Kaplan, D.L. The use of silk as a scaffold for mature, sustainable unilocular adipose 3D tissue engineered systems. Adv. Health Mater. 2016, 5, 1667–1677. [Google Scholar] [CrossRef]
- Abbott, R.D.; Borowsky, F.E.; Alonzo, C.A.; Zieba, A.; Georgakoudi, I.; Kaplan, D.L. Variability in responses observed in human white adipose tissue models. J. Tissue Eng. Regen. Med. 2017, 12, 840–847. [Google Scholar] [CrossRef]
- Tytgat, L.; Kollert, M.R.; Van Damme, L.; Thienpont, H.; Ottevaere, H.; Duda, G.N.; Geissler, S.; Dubruel, P.; Van Vlierberghe, S.; Qazi, T.H. Evaluation of 3D printed gelatin-based scaffolds with varying pore size for MSC-based adipose tissue engineering. Macromol. Biosci. 2020, 20, e1900364. [Google Scholar] [CrossRef]
- Song, M.; Liu, Y.; Hui, L. Preparation and characterization of acellular adipose tissue matrix using a combination of physical and chemical treatments. Mol. Med. Rep. 2017, 17, 138–146. [Google Scholar] [CrossRef]
- Flynn, L.E. The use of decellularized adipose tissue to provide an inductive microenvironment for the adipogenic differentiation of human adipose-derived stem cells. Biomaterials 2010, 31, 4715–4724. [Google Scholar] [CrossRef]
- Choi, J.S.; Yang, H.-J.; Kim, B.G.; Kim, J.D.; Lee, S.H.; Lee, E.K.; Park, K.; Cho, Y.W.; Lee, H.Y. Fabrication of porous extracellular matrix scaffolds from human adipose tissue. Tissue Eng. Part. C Methods 2010, 16, 387–396. [Google Scholar] [CrossRef]
- Yuan, Y.; Gao, J.; Ogawa, R. Mechanobiology and mechanotherapy of adipose tissue-effect of mechanical force on fat tissue engineering. Plast. Reconstr. Surg. Glob. Open 2015, 3, e578. [Google Scholar] [CrossRef]
- Zhang, T.; Lin, S.; Shao, X.; Shi, S.; Zhang, Q.; Xue, C.; Lin, Y.; Zhu, B.; Cai, X. Regulating osteogenesis and adipogenesis in adipose-derived stem cells by controlling underlying substrate stiffness. J. Cell. Physiol. 2017, 233, 3418–3428. [Google Scholar] [CrossRef] [PubMed]
- Pope, B.D.; Warren, C.R.; Parker, K.K.; Cowan, C.A. Microenvironmental control of adipocyte fate and function. Trends Cell Biol. 2016, 26, 745–755. [Google Scholar] [CrossRef] [PubMed]
- Van, R.L.A.; Roncari, D. Complete differentiation of adipocyte precursors. A culture system for studying the cellular nature of adipose tissue. Cell Tissue Res. 1978, 195, 317–329. [Google Scholar]
- Or-Tzadikario, S.; Sopher, R.; Gefen, A. Quantitative monitoring of lipid accumulation over time in cultured adipocytes as function of culture conditions: Toward controlled adipose tissue engineering. Tissue Eng. Part. C Methods 2010, 16, 1167–1181. [Google Scholar] [CrossRef]
- Veilleux, A.; Caron-Jobin, M.; Noël, S.; Laberge, P.Y.; Tchernof, A. Visceral Adipocyte hypertrophy is associated with dyslipidemia independent of body composition and fat distribution in women. Diabetes 2011, 60, 1504–1511. [Google Scholar] [CrossRef]
- Anand, S.S.; Tarnopolsky, M.A.; Rashid, S.; Schulze, K.M.; Desai, D.; Mente, A.; Rao, S.; Yusuf, S.; Gerstein, H.; Sharma, A.M. Adipocyte hypertrophy, fatty liver and metabolic risk factors in South Asians: The molecular study of health and risk in ethnic groups (mol-SHARE). PLoS ONE 2011, 6, e22112. [Google Scholar] [CrossRef]
- Notbohm, J.; Lesman, A.; Tirrell, D.A.; Ravichandran, G. Quantifying cell-induced matrix deformation in three dimensions based on imaging matrix fibers. Integr. Biol. 2015, 7, 1186–1195. [Google Scholar] [CrossRef]
- Petroll, W.M.; Cavanagh, H.D.; Jester, J.V. Dynamic three-dimensional visualization of collagen matrix remodeling and cytoskeletal organization in living corneal fibroblasts. Scanning 2004, 26, 1–10. [Google Scholar] [CrossRef]
- Mohammadi, H.; Arora, P.D.; Simmons, C.A.; Janmey, P.A.; McCulloch, C.A. Inelastic behaviour of collagen networks in cell–matrix interactions and mechanosensation. J. R. Soc. Interface 2015, 12, 20141074. [Google Scholar] [CrossRef]
- Ban, E.; Franklin, J.M.; Nam, S.; Smith, L.R.; Wang, H.; Wells, R.G.; Chaudhuri, O.; Liphardt, J.T.; Shenoy, V.B. Mechanisms of plastic deformation in collagen networks induced by cellular forces. Biophys. J. 2018, 114, 450–461. [Google Scholar] [CrossRef]
- Godfrey, C.; Bremer, A.; Alba, D.; Apovian, C.; Koethe, J.R.; Koliwad, S.; Lewis, D.; Lo, J.A.; McComsey, G.; Eckard, A.; et al. Obesity and fat metabolism in human immunodeficiency virus–infected individuals: Immunopathogenic mechanisms and clinical implications. J. Infect. Dis. 2019, 220, 420–431. [Google Scholar] [CrossRef] [PubMed]
- Davidson, M.D.; Burdick, J.A.; Wells, R.G. Engineered biomaterial platforms to study fibrosis. Adv. Health Mater. 2020, 9, e1901682. [Google Scholar] [CrossRef]
- Loh, Q.L.; Choong, C. Three-dimensional scaffolds for tissue engineering applications: Role of porosity and pore size. Tissue Eng. Part. B Rev. 2013, 19, 485–502. [Google Scholar] [CrossRef] [PubMed]
- Van Grunsven, L.A. 3D in vitro models of liver fibrosis. Adv. Drug Deliv. Rev. 2017, 121, 133–146. [Google Scholar] [CrossRef] [PubMed]
- Sacchi, M.; Bansal, R.; Rouwkema, J. Bioengineered 3D models to recapitulate tissue fibrosis. Trends Biotechnol. 2020, 38, 623–636. [Google Scholar] [CrossRef]
- Shoham, N.; Gottlieb, R.; Sharabani-Yosef, O.; Zaretsky, U.; Benayahu, D.; Gefen, A. Static mechanical stretching accelerates lipid production in 3T3-L1 adipocytes by activating the MEK signaling pathway. Am. J. Physiol. Physiol. 2012, 302, C429–C441. [Google Scholar] [CrossRef]
- Lutz, T.; Woods, S.C. Overview of animal models of obesity. Curr. Protoc. Pharmacol. 2012, 58, 5–61. [Google Scholar] [CrossRef]
- Kanasaki, K.; Koya, D. Biology of obesity: Lessons from animal models of obesity. J. Biomed. Biotechnol. 2011, 2011, 1–11. [Google Scholar] [CrossRef]
- Kleinert, M.; Clemmensen, C.; Hofmann, S.M.; Moore, M.C.; Renner, S.; Woods, S.C.; Huypens, P.; Beckers, J.; De Angelis, M.H.; Schurmann, A.; et al. Animal models of obesity and diabetes mellitus. Nat. Rev. Endocrinol. 2018, 14, 140–162. [Google Scholar] [CrossRef]
- Vickers, S.P.; Jackson, H.C.; Cheetham, S.C. The utility of animal models to evaluate novel anti-obesity agents. Br. J. Pharmacol. 2011, 164, 1248–1262. [Google Scholar] [CrossRef]
- Chusyd, D.E.; Wang, D.; Huffman, D.M.; Nagy, T.R. Relationships between rodent white adipose fat pads and human white adipose fat depots. Front. Nutr. 2016, 3, 10. [Google Scholar] [CrossRef] [PubMed]
- Weyer, C.; Tataranni, P.A.; Snitker, S.; Danforth, E.; Ravussin, E. Increase in insulin action and fat oxidation after treatment with CL 316,243, a highly selective beta3-adrenoceptor agonist in humans. Diabetes 1998, 47, 1555–1561. [Google Scholar] [CrossRef] [PubMed]
- Arch, J.R.; Wilson, S. Prospects for beta 3-adrenoceptor agonists in the treatment of obesity and diabetes. Int. J. Obes. Relat. Metab. Disord. 1996, 20, 191–199. [Google Scholar] [PubMed]
- Tavernier, G.; Barbe, P.; Galitzky, J.; Berlan, M.; Caput, D.; Lafontan, M.; Langin, D. Expression of beta3-adrenoceptors with low lipolytic action in human subcutaneous white adipocytes. J. Lipid Res. 1996, 37, 87–97. [Google Scholar]
- Coleman, D.L. Obese and diabetes: Two mutant genes causing diabetes-obesity syndromes in mice. Diabetologia 1978, 14, 141–148. [Google Scholar] [CrossRef]
- Mayer, J.; Bates, M.W.; Dickie, M.M. Hereditary diabetes in genetically obese mice. Science 1951, 113, 746–747. [Google Scholar] [CrossRef]
- Zhang, Y.; Proença, R.; Maffei, M.; Barone, M.; Leopold, L.; Friedman, J.M. Positional cloning of the mouse obese gene and its human homologue. Nature 1994, 372, 425–432. [Google Scholar] [CrossRef]
- Chua, S.C.; Chung, W.K.; Wu-Peng, X.S.; Zhang, Y.; Liu, S.-M.; Tartaglia, L.; Leibel, R.L. Phenotypes of mouse diabetes and rat fatty due to mutations in the OB (leptin) receptor. Science 1996, 271, 994–996. [Google Scholar] [CrossRef]
- Bates, S.H.; Kulkarni, R.N.; Seifert, M.; Myers, M.G. Roles for leptin receptor/STAT3-dependent and -independent signals in the regulation of glucose homeostasis. Cell Metab. 2005, 1, 169–178. [Google Scholar] [CrossRef]
- Bates, S.H.; Stearns, W.H.; Dundon, T.A.; Schubert, M.; Tso, A.W.K.; Wang, Y.; Banks, A.S.; Lavery, H.J.; Haq, A.K.; Maratos-Flier, E.; et al. STAT3 signalling is required for leptin regulation of energy balance but not reproduction. Nature 2003, 421, 856–859. [Google Scholar] [CrossRef]
- Bray, G.A. The Zucker-fatty rat: A review. Fed. Proc. 1977, 36, 148–153. [Google Scholar] [PubMed]
- Bray, G.A.; York, D.A. Hypothalamic and genetic obesity in experimental animals: An autonomic and endocrine hypothesis. Physiol. Rev. 1979, 59, 719–809. [Google Scholar] [CrossRef] [PubMed]
- Challis, B.G.; Coll, A.P.; Yeo, G.S.H.; Pinnock, S.B.; Dickson, S.L.; Thresher, R.R.; Dixon, J.; Zahn, D.; Rochford, J.J.; White, A.; et al. Mice lacking pro-opiomelanocortin are sensitive to high-fat feeding but respond normally to the acute anorectic effects of peptide-YY3-36. Proc. Natl. Acad. Sci. USA 2004, 101, 4695–4700. [Google Scholar] [CrossRef] [PubMed]
- Yaswen, L.; Diehl, N.; Brennan, M.B.; Hochgeschwender, U. Obesity in the mouse model of pro-opiomelanocortin deficiency responds to peripheral melanocortin. Nat. Med. 1999, 5, 1066–1070. [Google Scholar] [CrossRef]
- Corander, M.P.; Rimmington, D.; Challis, B.G.; O’Rahilly, S.; Coll, A.P. Loss of agouti-related peptide does not significantly impact the phenotype of murine POMC deficiency. Endocrinology 2011, 152, 1819–1828. [Google Scholar] [CrossRef]
- Hariri, N.; Thibault, L. High-fat diet-induced obesity in animal models. Nutr. Res. Rev. 2010, 23, 270–299. [Google Scholar] [CrossRef]
- Becerril, S.; Rodríguez, A.; Catalán, V.; Méndez-Giménez, L.; Ramírez, B.; Sainz, N.; Llorente, M.; Unamuno, X.; Gomez-Ambrosi, J.; Frühbeck, G. Targeted disruption of the iNOS gene improves adipose tissue inflammation and fibrosis in leptin-deficient ob/ob mice: Role of tenascin C. Int. J. Obes. 2018, 42, 1458–1470. [Google Scholar] [CrossRef]
- Matsuda, A.; Makino, N.; Tozawa, T.; Shirahata, N.; Honda, T.; Ikeda, Y.; Sato, H.; Ito, M.; Kakizaki, Y.; Akamatsu, M.; et al. Pancreatic fat accumulation, fibrosis, and acinar cell injury in the zucker diabetic fatty rat fed a chronic high-fat diet. Pancreas 2014, 43, 735–743. [Google Scholar] [CrossRef]
- Lasker, S.; Rahman, M.; Parvez, F.; Zamila, M.; Miah, P.; Nahar, K.; Kabir, F.; Sharmin, S.B.; Subhan, N.; Ahsan, G.U.; et al. High-fat diet-induced metabolic syndrome and oxidative stress in obese rats are ameliorated by yogurt supplementation. Sci. Rep. 2019, 9, 1–15. [Google Scholar] [CrossRef]
- Nakazeki, F.; Nishiga, M.; Horie, T.; Nishi, H.; Nakashima, Y.; Baba, O.; Kuwabara, Y.; Nishino, T.; Nakao, T.; Ide, Y.; et al. Loss of periostin ameliorates adipose tissue inflammation and fibrosis in vivo. Sci. Rep. 2018, 8, 8553. [Google Scholar] [CrossRef]
- Vila, I.K.; Badin, P.M.; Marques, M.A.; Monbrun, L.; Lefort, C.; Mir, L.; Louche, K.; Bourlier, V.; Roussel, B.; Gui, P.; et al. Immune cell toll-like receptor 4 mediates the development of obesity- and endotoxemia-associated adipose tissue fibrosis. Cell Rep. 2014, 7, 1116–1129. [Google Scholar] [CrossRef] [PubMed]
- Payette, C.; Blackburn, P.; Lamarche, B.; Tremblay, A.; Bergeron, J.; Lemieux, I.; Després, J.-P.; Couillard, C. Sex differences in postprandial plasma tumor necrosis factor–α, interleukin-6, and C-reactive protein concentrations. Metabolism 2009, 58, 1593–1601. [Google Scholar] [CrossRef]
- Grove, K.L.; Fried, S.K.; Greenberg, A.S.; Xiao, X.; Clegg, D.J. A microarray analysis of sexual dimorphism of adipose tissues in high-fat-diet-induced obese mice. Int. J. Obes. 2010, 34, 989–1000. [Google Scholar] [CrossRef] [PubMed]
- Macotela, Y.; Boucher, J.; Tran, T.T.; Kahn, C.R. Sex and depot differences in adipocyte insulin sensitivity and glucose metabolism. Diabetes 2009, 58, 803–812. [Google Scholar] [CrossRef] [PubMed]
- Davis, K.E.; Neinast, M.D.; Sun, K.; Skiles, W.M.; Bills, J.D.; Zehr, J.A.; Zeve, D.; Hahner, L.D.; Cox, D.W.; Gent, L.M.; et al. The sexually dimorphic role of adipose and adipocyte estrogen receptors in modulating adipose tissue expansion, inflammation, and fibrosis. Mol. Metab. 2013, 2, 227–242. [Google Scholar] [CrossRef]
- Hilton, C.; Neville, M.J.; Karpe, F. MicroRNAs in adipose tissue: Their role in adipogenesis and obesity. Int. J. Obes. 2012, 37, 325–332. [Google Scholar] [CrossRef]
- Abente, E.; Subramanian, M.; Ramachandran, V.; Najafi-Shoushtari, H.; Najafi-Shoushtari, H. MicroRNAs in obesity-associated disorders. Arch. Biochem. Biophys. 2016, 589, 108–119. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Yang, Y.; Wu, J. TNFalpha-induced up-regulation of miR-155 inhibits adipogenesis by down-regulating early adipogenic transcription factors. Biochem. Biophys. Res. Commun. 2011, 414, 618–624. [Google Scholar] [CrossRef]
- Chen, Y.; Siegel, F.; Kipschull, S.; Haas, B.; Fröhlich, H.; Meister, G.; Pfeifer, A. miR-155 regulates differentiation of brown and beige adipocytes via a bistable circuit. Nat. Commun. 2013, 4, 1769. [Google Scholar] [CrossRef]
- Velázquez, K.T.; Enos, R.T.; Carson, M.S.; Cranford, T.L.; Bader, J.E.; Sougiannis, A.T.; Pritchett, C.; Fan, D.; Carson, J.A.; Murphy, E. miR155 deficiency aggravates high-fat diet-induced adipose tissue fibrosis in male mice. Physiol. Rep. 2017, 5, e13412. [Google Scholar] [CrossRef]
- Essandoh, K.; Li, Y.; Huo, J.; Fan, G.C. MiRNA-mediated macrophage polarization and its potential role in the regulation of inflammatory response. Shock 2016, 46, 122–131. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Zhang, Y.; Zhong, M.; Suo, Q.; Lv, K. Expression profiles of miRNAs in polarized macrophages. Int. J. Mol. Med. 2013, 31, 797–802. [Google Scholar] [CrossRef] [PubMed]
- Jablonski, K.A.; Gaudet, A.D.; Amici, S.A.; Popovich, P.G.; Guerau-De-Arellano, M. Control of the inflammatory macrophage transcriptional signature by miR-155. PLoS ONE 2016, 11, e0159724. [Google Scholar] [CrossRef] [PubMed]
- Gaudet, A.D.; Fonken, L.K.; Gushchina, L.V.; Aubrecht, T.G.; Maurya, S.K.; Periasamy, M.; Nelson, R.J.; Popovich, P.G. miR-155 deletion in female mice prevents diet-induced obesity. Sci. Rep. 2016, 6, 22862. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, Y.; Nagai, Y.; Honda, H.; Okamoto, N.; Yamamoto, S.; Hamashima, T.; Ishii, Y.; Tanaka, M.; Suganami, T.; Sasahara, M.; et al. Isoliquiritigenin attenuates adipose tissue inflammation in vitro and adipose tissue fibrosis through inhibition of innate immune responses in mice. Sci. Rep. 2016, 6, 23097. [Google Scholar] [CrossRef]
- Henninger, A.M.J.; Eliasson, B.; Jenndahl, L.E.; Hammarstedt, A. Adipocyte hypertrophy, inflammation and fibrosis characterize subcutaneous adipose tissue of healthy, non-obese subjects predisposed to type 2 diabetes. PLoS ONE 2014, 9, e105262. [Google Scholar] [CrossRef]
- Alba, D.; Farooq, J.A.; Lin, M.Y.C.; Schafer, A.L.; Shepherd, J.; Koliwad, S.K. Subcutaneous fat fibrosis links obesity to insulin resistance in chinese americans. J. Clin. Endocrinol. Metab. 2018, 103, 3194–3204. [Google Scholar] [CrossRef]
- Gu, D.; He, J.; Duan, X.; Reynolds, K.; Wu, X.; Chen, J.; Huang, G.; Whelton, P.K. Body weight and mortality among men and women in China. JAMA 2006, 295, 776–783. [Google Scholar] [CrossRef]
- Jan, V.; Cervera, P.; Maachi, M.; Baudrimont, M.; Kim, M.; Vidal, H.; Girard, P.-M.; LeVan, P.; Rozenbaum, W.; Lombès, A.; et al. Altered fat differentiation and adipocytokine expression are inter-related and linked to morphological changes and insulin resistance in HIV-1-infected lipodystrophic patients. Antivir. Ther. 2004, 9, 555–564. [Google Scholar]
- Lassen, P.B.; Liu, Y.; Tordjman, J.; Poitou, C.; Bouillot, J.-L.; Genser, L.; Zucker, J.-D.; Sokolovska, N.; Aron-Wisnewsky, J.; Charlotte, F.; et al. The FAT score, a fibrosis score of adipose tissue: Predicting weight-loss outcome after gastric bypass. J. Clin. Endocrinol. Metab. 2017, 102, 2443–2453. [Google Scholar] [CrossRef]
- Zuk, P.A.; Zhu, M.; Mizuno, H.; Huang, J.; Futrell, J.W.; Katz, A.J.; Benhaim, P.; Lorenz, H.P.; Hedrick, M.H. Multilineage cells from human adipose tissue: Implications for cell-based therapies. Tissue Eng. 2001, 7, 211–228. [Google Scholar] [CrossRef] [PubMed]
- Pawitan, J.A. Prospect of stem cell conditioned medium in regenerative medicine. Biomed. Res. Int. 2014, 2014, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Maria, A.T.J.; Toupet, K.; Maumus, M.; Fonteneau, G.; Le Quellec, A.; Jorgensen, C.; Guilpain, P.; Noel, D. Human adipose mesenchymal stem cells as potent anti-fibrosis therapy for systemic sclerosis. J. Autoimmun. 2016, 70, 31–39. [Google Scholar] [CrossRef] [PubMed]
- Borovikova, A.A.; Ziegler, M.E.; Banyard, D.A.; Wirth, G.A.; Paydar, K.Z.; Evans, G.R.D.; Widgerow, A.D. Adipose-derived tissue in the treatment of dermal fibrosis. Ann. Plast. Surg. 2018, 80, 297–307. [Google Scholar] [CrossRef] [PubMed]
- Lou, G.; Yang, Y.; Liu, F.; Ye, B.; Chen, Z.; Zheng, M.; Liu, Y. MiR-122 modification enhances the therapeutic efficacy of adipose tissue-derived mesenchymal stem cells against liver fibrosis. J. Cell. Mol. Med. 2017, 21, 2963–2973. [Google Scholar] [CrossRef]
- Paik, K.Y.; Kim, K.H.; Park, J.H.; Lee, J.I.; Kim, O.H.; Hong, H.E.; Seo, H.; Choi, H.J.; Ahn, J.; Lee, T.Y.; et al. A novel antifibrotic strategy utilizing conditioned media obtained from miR-150-transfected adipose-derived stem cells: Validation of an animal model of liver fibrosis. Exp. Mol. Med. 2020, 52, 438–449. [Google Scholar] [CrossRef]
- Lee, T.M.; Harn, H.-J.; Chiou, T.W.; Chuang, M.H.; Chen, C.H.; Chuang, C.-H.; Lin, P.C.; Lin, S.Z. Preconditioned adipose-derived stem cells ameliorate cardiac fibrosis by regulating macrophage polarization in infarcted rat hearts through the PI3K/STAT3 pathway. Lab. Investig. 2019, 99, 634–647. [Google Scholar] [CrossRef]
- Song, Y.; Peng, C.; Lv, S.; Cheng, J.; Liu, S.; Wen, Q.; Guan, G.; Liu, G. Adipose-derived stem cells ameliorate renal interstitial fibrosis through inhibition of EMT and inflammatory response via TGF-beta1 signaling pathway. Int. Immunopharmacol. 2017, 44, 115–122. [Google Scholar] [CrossRef]
- Rivera-Valdés, J.J.; García-Bañuelos, J.; Salazar-Montes, A.; García-Benavides, L.; Rosales-Dominguez, A.; Armendáriz-Borunda, J.; Sandoval-Rodríguez, A. Human adipose derived stem cells regress fibrosis in a chronic renal fibrotic model induced by adenine. PLoS ONE 2017, 12, e0187907. [Google Scholar] [CrossRef]
- Wang, Z.; Sun, N. Adipose-derived mesenchymal stem cells: A new tool for the treatment of renal fibrosis. Stem Cells Dev. 2018, 27, 1406–1411. [Google Scholar] [CrossRef]
- Sun, W.; Ni, X.; Sun, S.; Cai, L.; Yu, J.; Wang, J.; Nie, B.; Sun, Z.; Ni, X.; Cao, X. Adipose-derived stem cells alleviate radiation-induced muscular fibrosis by suppressing the expression of TGF-beta1. Stem Cells Int. 2016, 2016, 5638204. [Google Scholar] [CrossRef] [PubMed]
- Rathinasabapathy, A.; Bruce, E.; Espejo, A.; Horowitz, A.; Sudhan, D.; Nair, A.; Guzzo, D.; Francis, J.; Raizada, M.K.; Shenoy, V.; et al. Therapeutic potential of adipose stem cell-derived conditioned medium against pulmonary hypertension and lung fibrosis. Br. J. Pharmacol. 2016, 173, 2859–2879. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.H.; Lee, E.J.; Lee, S.Y.; Kim, J.H.; Shim, J.J.; Shin, C.; In, K.H.; Kang, K.H.; Uhm, C.S.; Kim, H.K.; et al. The effect of adipose stem cell therapy on pulmonary fibrosis induced by repetitive intratracheal bleomycin in mice. Exp. Lung Res. 2014, 40, 117–125. [Google Scholar] [CrossRef] [PubMed]
- Han, H.S.; Lee, H.; You, D.; Nguyen, V.Q.; Song, D.-G.; Oh, B.H.; Shin, S.; Choi, J.S.; Kim, J.D.; Pan, C.H.; et al. Human adipose stem cell-derived extracellular nanovesicles for treatment of chronic liver fibrosis. J. Control. Release 2020, 320, 328–336. [Google Scholar] [CrossRef]
- Qu, Y.; Zhang, Q.; Cai, X.; Li, F.; Ma, Z.; Xu, M.; Lu, L.G. Exosomes derived from miR-181-5p-modified adipose-derived mesenchymal stem cells prevent liver fibrosis via autophagy activation. J. Cell. Mol. Med. 2017, 21, 2491–2502. [Google Scholar] [CrossRef]
- Bai, X.; Yan, Y.; Song, Y.-H.; Seidensticker, M.; Rabinovich, B.; Metzele, R.; Bankson, J.A.; Vykoukal, D.; Alt, E.U. Both cultured and freshly isolated adipose tissue-derived stem cells enhance cardiac function after acute myocardial infarction. Eur. Heart J. 2009, 31, 489–501. [Google Scholar] [CrossRef]
- Eitta, R.S.A.; Ismail, A.A.; Abdelmaksoud, R.A.; Ghezlan, N.A.; Mehanna, R. Evaluation of autologous adipose-derived stem cells vs. fractional carbon dioxide laser in the treatment of post acne scars: A split-face study. Int. J. Dermatol. 2019, 58, 1212–1222. [Google Scholar] [CrossRef]
- Lee, J.W.; Park, S.H.; Lee, S.J.; Kim, S.H.; Suh, I.S.; Jeong, H.S. Clinical impact of highly condensed stromal vascular fraction injection in surgical management of depressed and contracted scars. Aesthetic Plast. Surg. 2018, 42, 1689–1698. [Google Scholar] [CrossRef]
- Gentile, P.; De Angelis, B.; Pasin, M.; Cervelli, G.; Curcio, C.B.; Floris, M.; Di Pasquali, C.; Bocchini, I.; Balzani, A.; Nicoli, F.; et al. Adipose-derived stromal vascular fraction cells and platelet-rich plasma. J. Craniofa. Surg. 2014, 25, 267–272. [Google Scholar] [CrossRef]
- Williams, R.M.; Zipfel, W.R.; Webb, W.W. Interpreting second-harmonic generation images of collagen i fibrils. Biophys. J. 2004, 88, 1377–1386. [Google Scholar] [CrossRef]
- Cox, G.; Kable, E.; Jones, A.; Fraser, I.; Manconi, F.; Gorrell, M.D. 3-Dimensional imaging of collagen using second harmonic generation. J. Struct. Biol. 2003, 141, 53–62. [Google Scholar] [CrossRef]
- Chen, X.; Nadiarynkh, O.; Plotnikov, S.; Campagnola, P.J. Second harmonic generation microscopy for quantitative analysis of collagen fibrillar structure. Nat. Protoc. 2012, 7, 654–669. [Google Scholar] [CrossRef] [PubMed]
- Alkhouli, N.; Mansfield, J.; Green, E.; Bell, J.S.; Knight, B.; Liversedge, N.; Tham, J.C.; Welbourn, R.; Shore, A.C.; Kos, K.; et al. The mechanical properties of human adipose tissues and their relationships to the structure and composition of the extracellular matrix. Am. J. Physiol. Metab. 2013, 305, E1427–E1435. [Google Scholar] [CrossRef] [PubMed]
- Sundarakrishnan, A.; Chen, Y.; Black, L.D.; Aldridge, B.B.; Kaplan, D.L. Engineered cell and tissue models of pulmonary fibrosis. Adv. Drug Deliv. Rev. 2018, 129, 78–94. [Google Scholar] [CrossRef] [PubMed]
- Huang, D.; Huang, Y.; Yang, X.; Yang, X.; Lin, H.; Feng, G.; Zhu, X.; Zhang, X.D. Viscoelasticity in natural tissues and engineered scaffolds for tissue reconstruction. Acta Biomater. 2019, 97, 74–92. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).