Intratumoral Distribution and pH-Dependent Drug Release of High Molecular Weight HPMA Copolymer Drug Conjugates Strongly Depend on Specific Tumor Substructure and Microenvironment
Abstract
1. Introduction
2. Results
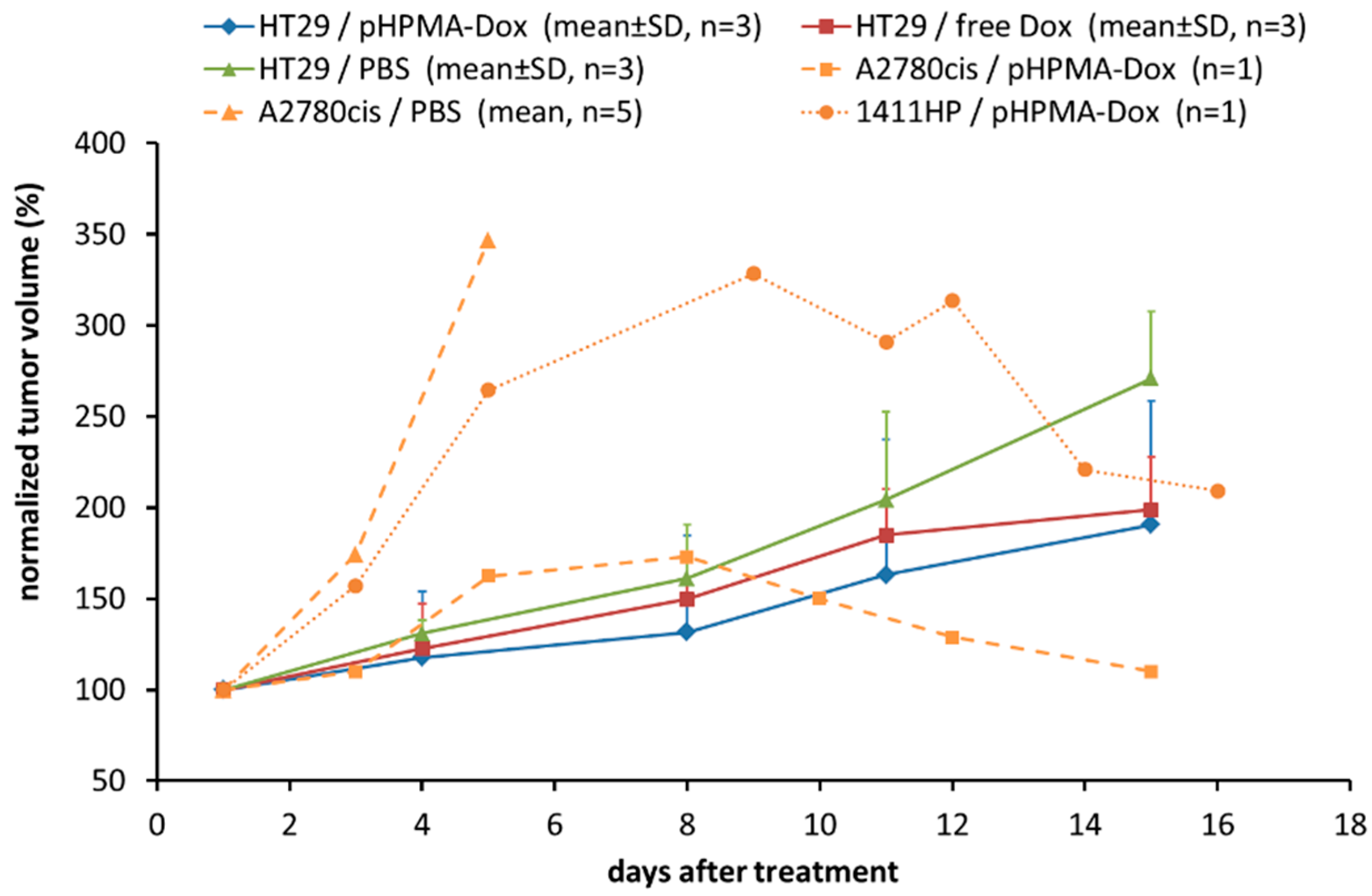
2.1. Testing of the pHPMA-Dox Conjugate in the HT29 Colorectal Carcinoma Xenograft Model
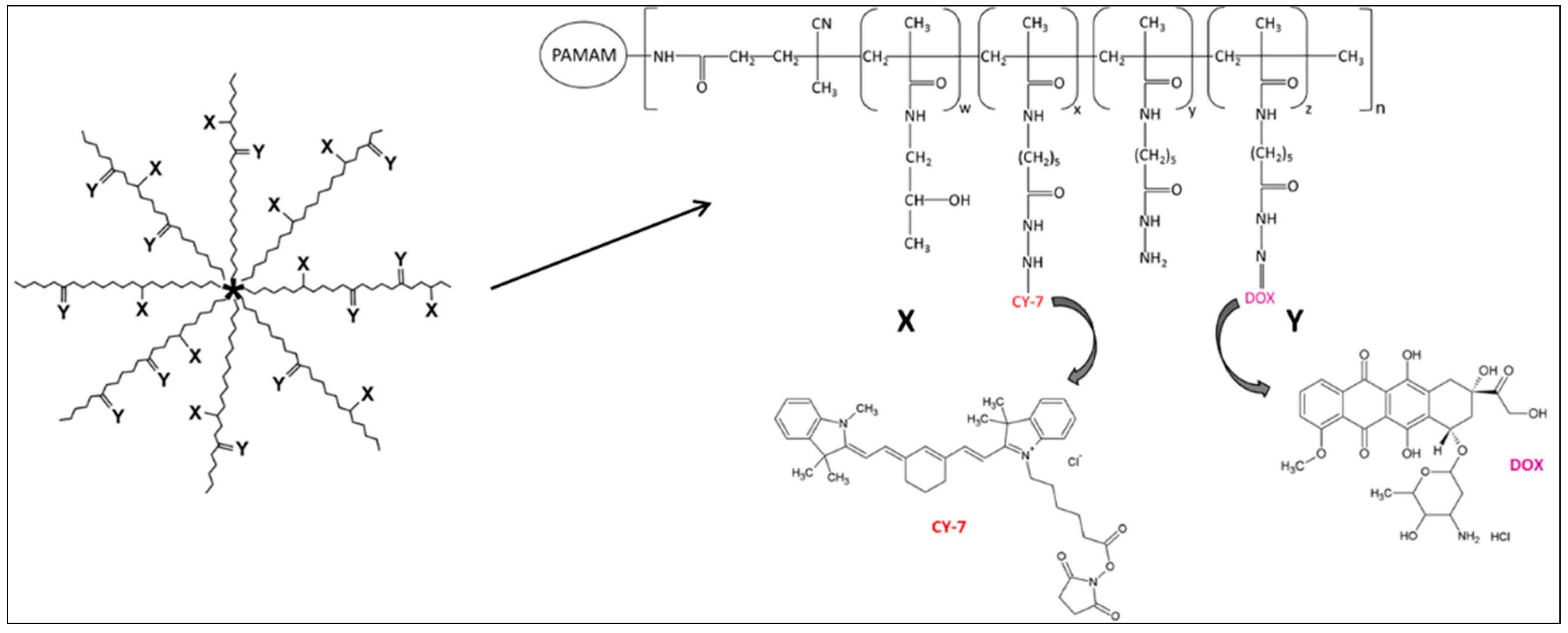
2.2. Synthesis and Physico-Chemical Characterization of Cy7-Labeled Variant of the pHPMA-Dox Conjugate
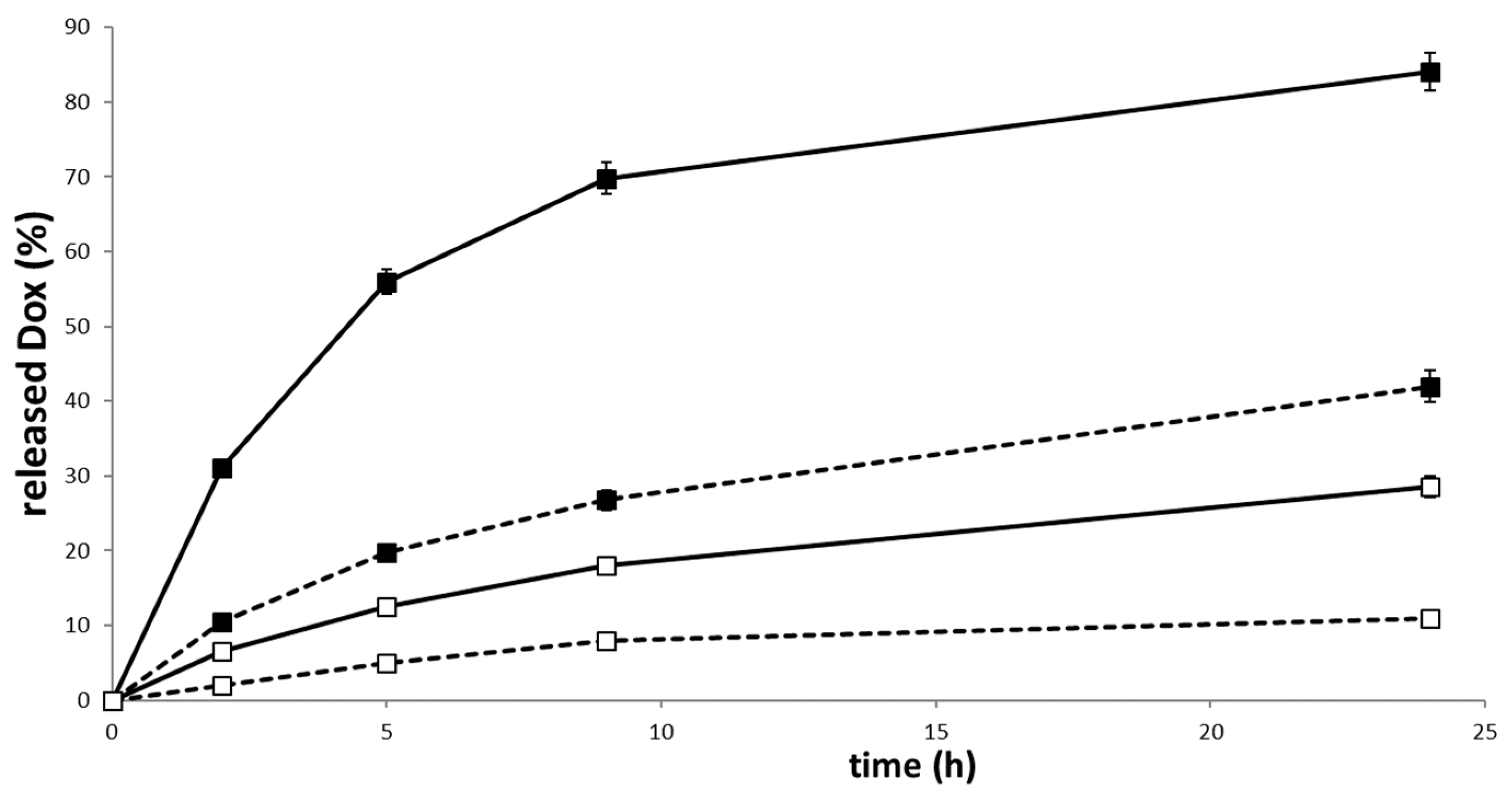
2.3. Analysis of pH-Dependent Drug Relaease and Cellular Uptake of the pHPMA-Dox-Cy7 Conjugate
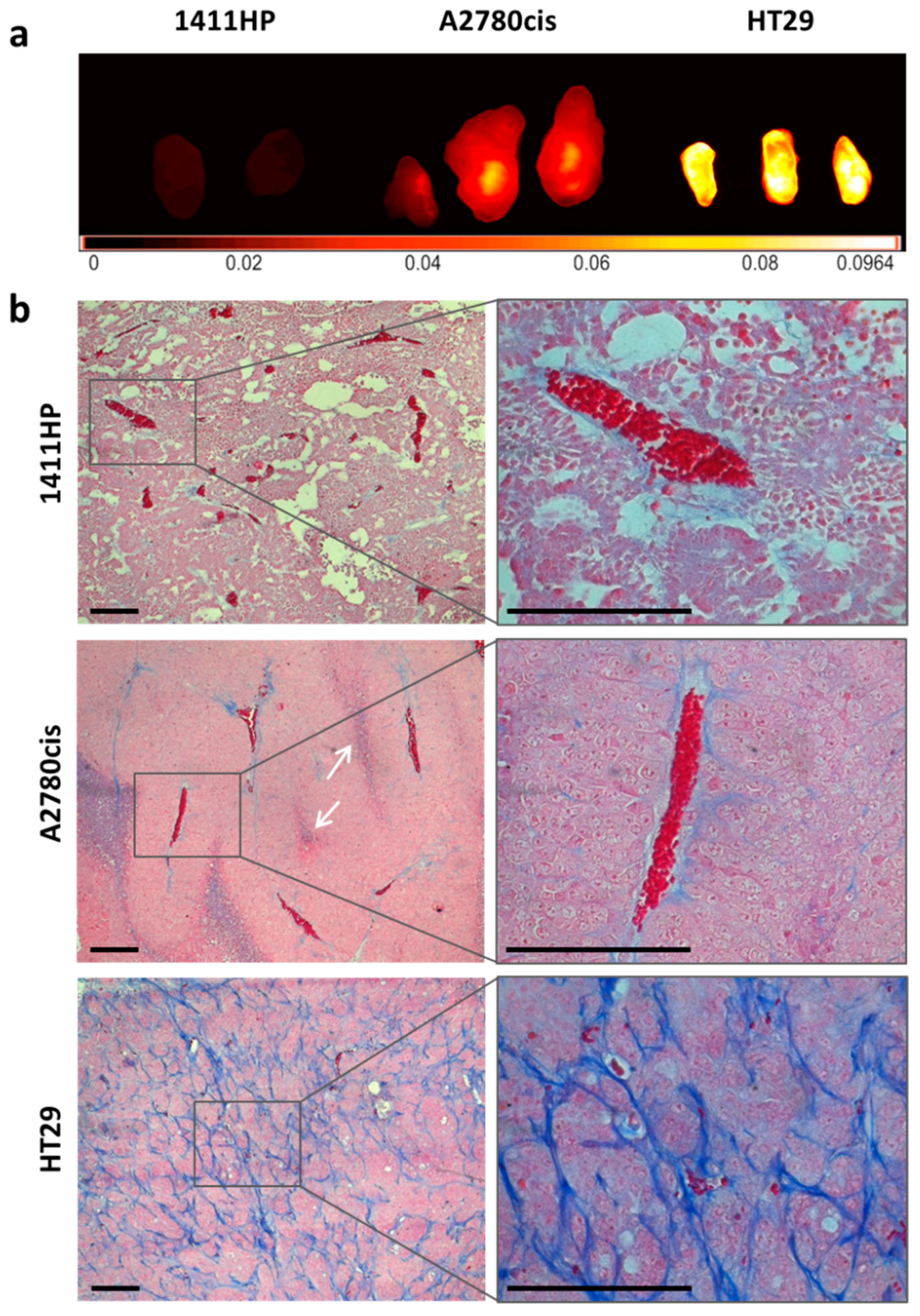
2.4. Characterization of Cell Line Derived Xenograft Tumors
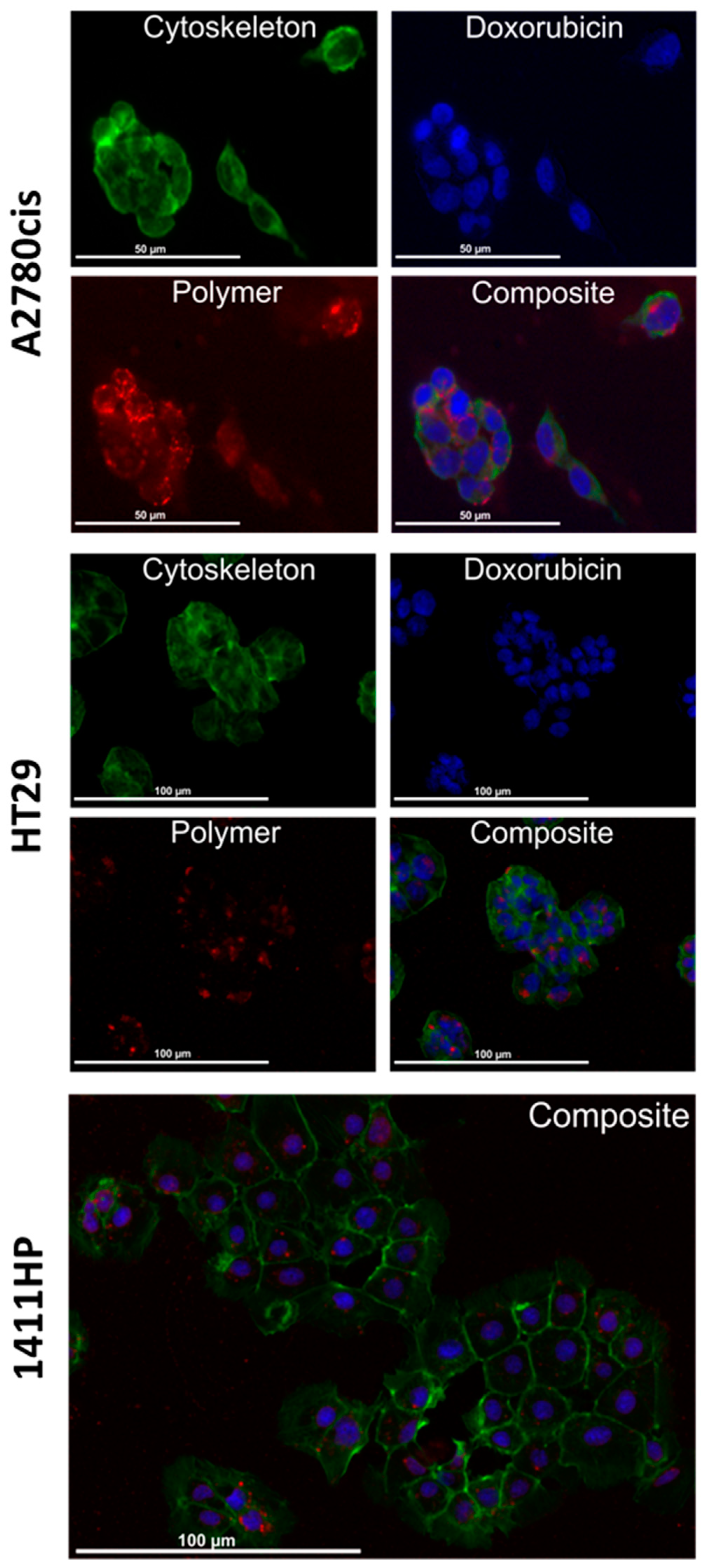
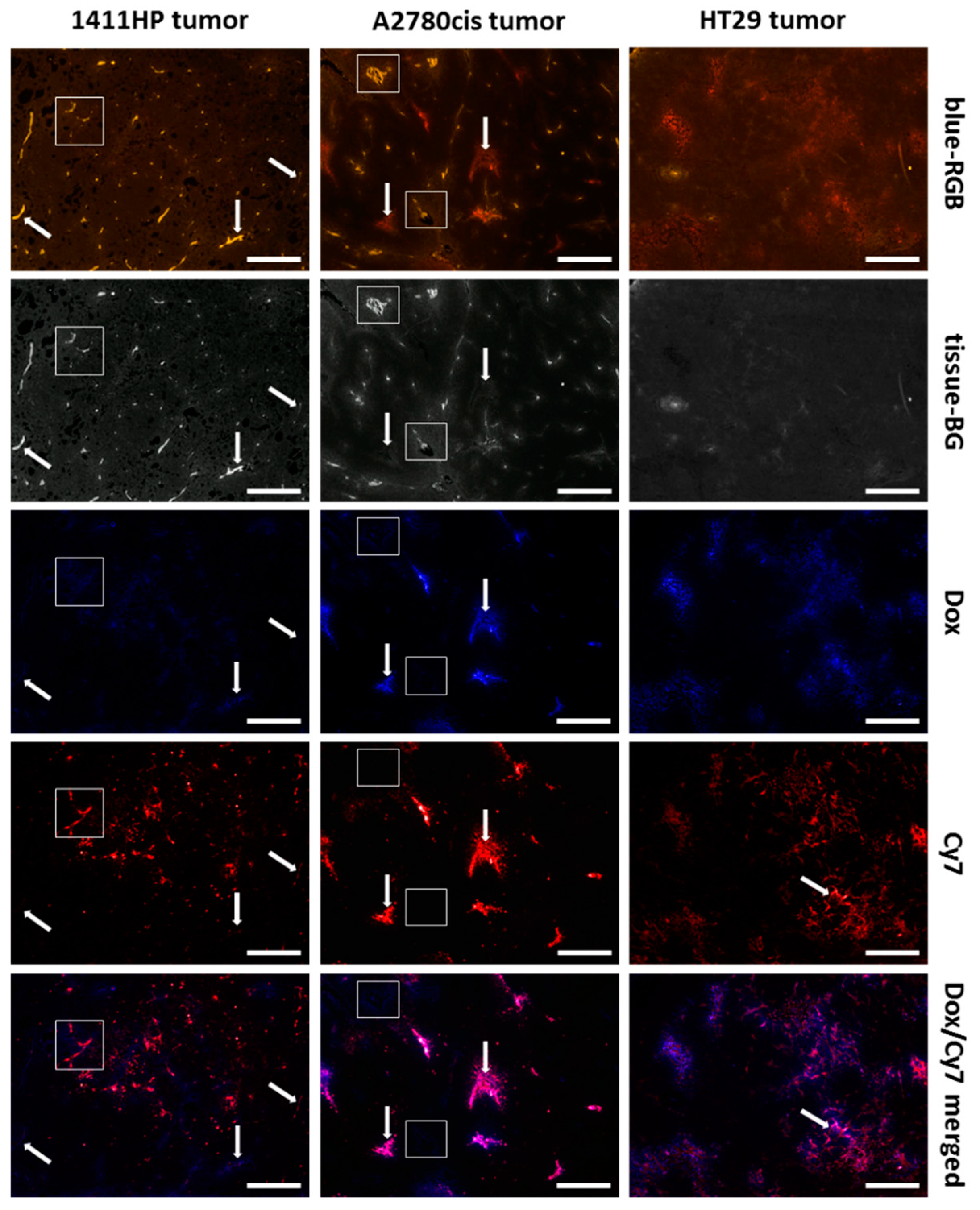
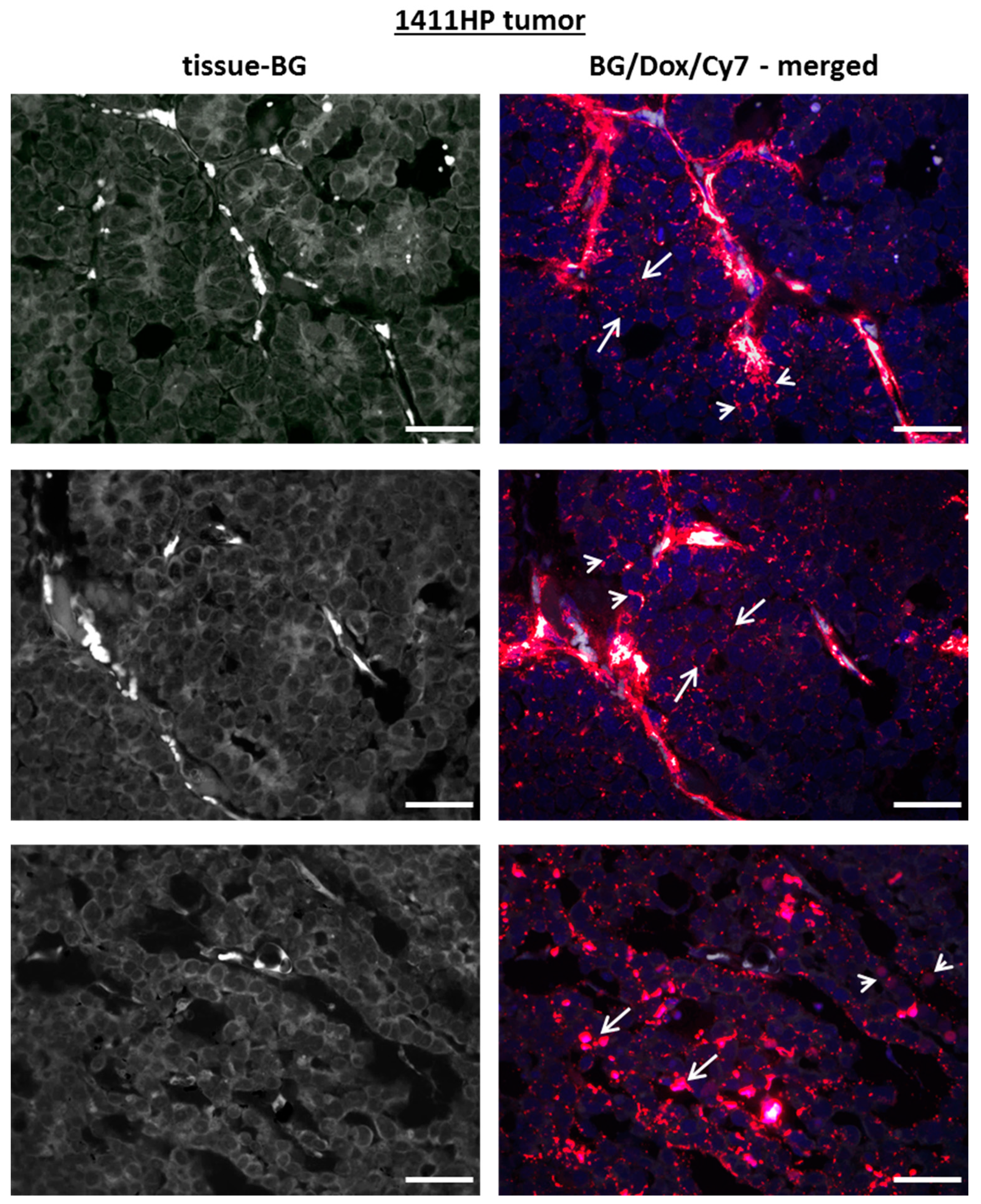
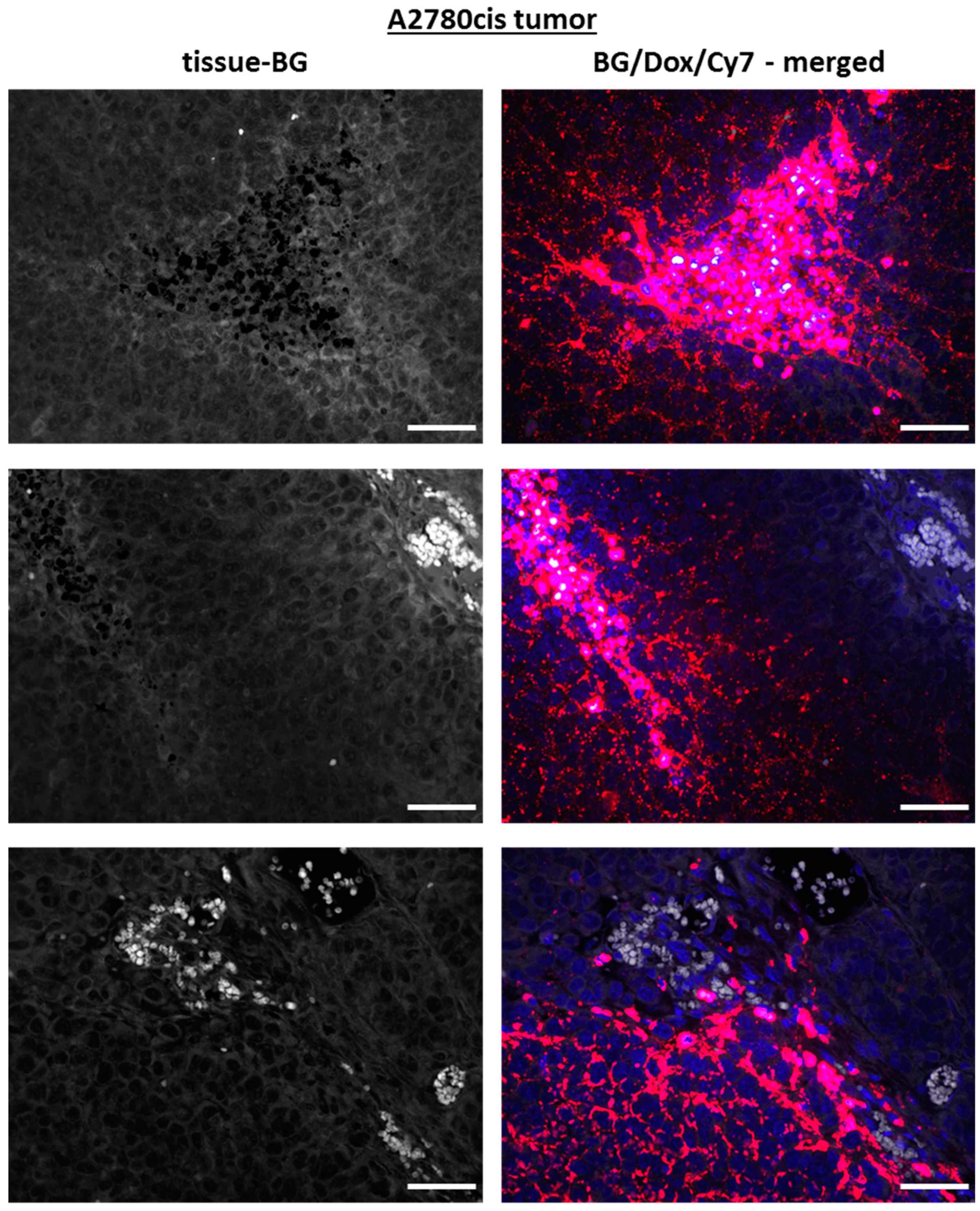
2.5. Intratumoral Distribution and Drug Release of pHPMA-Dox-Cy7 in Xenograft Tumors
3. Discussion
4. Conclusions
5. Materials and Methods
5.1. Synthesis and Physico-Chemical Characterization of Star-Shaped Polymer Drug Conjugate pHPMA-Dox-Cy7
5.2. Cell Culture, SRB Cytotoxicity Assay, Preparation of Cell Monolayers
5.3. Animal Care and Treatment, Preparation of Tumor Probes and Slides
5.4. Multispectral Fluorescence Microscopy of Fixed Monolayer Cells and Tumor Tissue Slides
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| MDPI | Multidisciplinary Digital Publishing Institute |
| DOAJ | Directory of Open Access Journals |
| TLA | Three letter acronym |
| LD | Linear dichroism |
References
- Maeda, H. The enhanced permeability and retention (EPR) effect in tumor vasculature: The key role of tumor-selective macromolecular drug targeting. Adv. Enzym. Regul. 2001, 41, 189–207. [Google Scholar] [CrossRef]
- Danhier, F. To exploit the tumor microenvironment: Since the EPR effect fails in the clinic, what is the future of nanomedicine? J. Control. Release 2016, 244, 108–121. [Google Scholar] [CrossRef] [PubMed]
- Golombek, S.K.; May, J.N.; Theek, B.; Appold, L.; Drude, N.; Kiessling, F.; Lammers, T. Tumor targeting via EPR: Strategies to enhance patient responses. Adv. Drug Deliv. Rev. 2018, 130, 17–38. [Google Scholar] [CrossRef] [PubMed]
- Nichols, J.W.; Bae, Y.H. EPR: Evidence and fallacy. J. Control. Release 2014, 190, 451–464. [Google Scholar] [CrossRef]
- Nakamura, Y.; Mochida, A.; Choyke, P.L.; Kobayashi, H. Nanodrug Delivery: Is the Enhanced Permeability and Retention Effect Sufficient for Curing Cancer? Bioconjug. Chem. 2016, 27, 2225–2238. [Google Scholar] [CrossRef]
- Maeda, H. Toward a full understanding of the EPR effect in primary and metastatic tumors as well as issues related to its heterogeneity. Adv. Drug Deliv. Rev. 2015, 91, 3–6. [Google Scholar] [CrossRef] [PubMed]
- Ojha, T.; Pathak, V.; Shi, Y.; Hennink, W.E.; Moonen, C.T.W.; Storm, G.; Kiessling, F.; Lammers, T. Pharmacological and physical vessel modulation strategies to improve EPR-mediated drug targeting to tumors. Adv. Drug Deliv. Rev. 2017, 119, 44–60. [Google Scholar] [CrossRef] [PubMed]
- Nichols, J.W.; Sakurai, Y.; Harashima, H.; Bae, Y.H. Nano-sized drug carriers: Extravasation, intratumoral distribution, and their modeling. J. Control. Release 2017, 267, 31–46. [Google Scholar] [CrossRef] [PubMed]
- Chytil, P.; Koziolova, E.; Etrych, T.; Ulbrich, K. HPMA Copolymer-Drug Conjugates with Controlled Tumor-Specific Drug Release. Macromol. Biosci. 2018, 18. [Google Scholar] [CrossRef] [PubMed]
- Duncan, R. Development of HPMA copolymer-anticancer conjugates: Clinical experience and lessons learnt. Adv. Drug Deliv. Rev. 2009, 61, 1131–1148. [Google Scholar] [CrossRef] [PubMed]
- Kopecek, J.; Kopeckova, P. HPMA copolymers: Origins, early developments, present, and future. Adv. Drug Deliv. Rev. 2010, 62, 122–149. [Google Scholar] [CrossRef]
- Lammers, T.; Ulbrich, K. HPMA copolymers: 30 years of advances. Adv. Drug Deliv. Rev. 2010, 62, 119–121. [Google Scholar] [CrossRef] [PubMed]
- Heinrich, A.K.; Lucas, H.; Schindler, L.; Chytil, P.; Etrych, T.; Mader, K.; Mueller, T. Improved Tumor-Specific Drug Accumulation by Polymer Therapeutics with pH-Sensitive Drug Release Overcomes Chemotherapy Resistance. Mol. Cancer Ther. 2016, 15, 998–1007. [Google Scholar] [CrossRef] [PubMed]
- Callahan, J.; Kopeckov, P.; Kopecek, J. Intracellular trafficking and subcellular distribution of a large array of HPMA copolymers. Biomacromolecules 2009, 10, 1704–1714. [Google Scholar] [CrossRef] [PubMed]
- Jensen, K.D.; Nori, A.; Tijerina, M.; Kopeckova, P.; Kopecek, J. Cytoplasmic delivery and nuclear targeting of synthetic macromolecules. J. Control. Release 2003, 87, 89–105. [Google Scholar] [CrossRef]
- Chytil, P.; Hoffmann, S.; Schindler, L.; Kostka, L.; Ulbrich, K.; Caysa, H.; Mueller, T.; Mader, K.; Etrych, T. Dual fluorescent HPMA copolymers for passive tumor targeting with pH-sensitive drug release II: Impact of release rate on biodistribution. J. Control. Release 2013, 172, 504–512. [Google Scholar] [CrossRef]
- Hoffmann, S.; Vystrcilova, L.; Ulbrich, K.; Etrych, T.; Caysa, H.; Mueller, T.; Mader, K. Dual fluorescent HPMA copolymers for passive tumor targeting with pH-sensitive drug release: Synthesis and characterization of distribution and tumor accumulation in mice by noninvasive multispectral optical imaging. Biomacromolecules 2012, 13, 652–663. [Google Scholar] [CrossRef]
- Matsumoto, Y.; Nichols, J.W.; Toh, K.; Nomoto, T.; Cabral, H.; Miura, Y.; Christie, R.J.; Yamada, N.; Ogura, T.; Kano, M.R.; et al. Vascular bursts enhance permeability of tumour blood vessels and improve nanoparticle delivery. Nat. Nanotechnol. 2016, 11, 533–538. [Google Scholar] [CrossRef]
- Adiseshaiah, P.P.; Crist, R.M.; Hook, S.S.; McNeil, S.E. Nanomedicine strategies to overcome the pathophysiological barriers of pancreatic cancer. Nat. Rev. Clin. Oncol. 2016, 13, 750–765. [Google Scholar] [CrossRef]
- Han, X.; Xu, Y.; Geranpayehvaghei, M.; Anderson, G.J.; Li, Y.; Nie, G. Emerging nanomedicines for anti-stromal therapy against desmoplastic tumors. Biomaterials 2020, 232, 119745. [Google Scholar] [CrossRef]
- Miao, L.; Lin, C.M.; Huang, L. Stromal barriers and strategies for the delivery of nanomedicine to desmoplastic tumors. J. Control. Release 2015, 219, 192–204. [Google Scholar] [CrossRef]
- Chan, T.S.; Shaked, Y.; Tsai, K.K. Targeting the Interplay Between Cancer Fibroblasts, Mesenchymal Stem Cells, and Cancer Stem Cells in Desmoplastic Cancers. Front. Oncol. 2019, 9, 688. [Google Scholar] [CrossRef] [PubMed]
- Etrych, T.; Strohalm, J.; Chytil, P.; Cernoch, P.; Starovoytova, L.; Pechar, M.; Ulbrich, K. Biodegradable star HPMA polymer conjugates of doxorubicin for passive tumor targeting. Eur. J. Pharm. Sci. 2011, 42, 527–539. [Google Scholar] [CrossRef] [PubMed]
- Etrych, T.; Daumova, L.; Pokorna, E.; Tuskova, D.; Lidicky, O.; Kolarova, V.; Pankrac, J.; Sefc, L.; Chytil, P.; Klener, P. Effective doxorubicin-based nano-therapeutics for simultaneous malignant lymphoma treatment and lymphoma growth imaging. J. Control. Release 2018, 289, 44–55. [Google Scholar] [CrossRef] [PubMed]
- Ulbrich, K.; Etrych, T.; Chytil, P.; Jelinkova, M.; Rihova, B. HPMA copolymers with pH-controlled release of doxorubicin: In vitro cytotoxicity and in vivo antitumor activity. J. Control. Release 2003, 87, 33–47. [Google Scholar] [CrossRef]


| Polymer | Mw | Ð | Rh (nm) | Hydrazide Content (mol.%) | Number of Arms b | Dox (wt.%) | Cy7 (wt.%) |
|---|---|---|---|---|---|---|---|
| 1 a | 23,700 | 1.6 | 3.0 | - | n.a. | - | - |
| 2 a | 165,000 | 1.7 | 9.4 | 5.2 | 7 | - | - |
| 3 | ~180,000 c | ~1.8 c | 10.8 | - | 7 | 10.6 | 0.76 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Noack, A.-K.; Lucas, H.; Chytil, P.; Etrych, T.; Mäder, K.; Mueller, T. Intratumoral Distribution and pH-Dependent Drug Release of High Molecular Weight HPMA Copolymer Drug Conjugates Strongly Depend on Specific Tumor Substructure and Microenvironment. Int. J. Mol. Sci. 2020, 21, 6029. https://doi.org/10.3390/ijms21176029
Noack A-K, Lucas H, Chytil P, Etrych T, Mäder K, Mueller T. Intratumoral Distribution and pH-Dependent Drug Release of High Molecular Weight HPMA Copolymer Drug Conjugates Strongly Depend on Specific Tumor Substructure and Microenvironment. International Journal of Molecular Sciences. 2020; 21(17):6029. https://doi.org/10.3390/ijms21176029
Chicago/Turabian StyleNoack, Anne-Kathrin, Henrike Lucas, Petr Chytil, Tomáš Etrych, Karsten Mäder, and Thomas Mueller. 2020. "Intratumoral Distribution and pH-Dependent Drug Release of High Molecular Weight HPMA Copolymer Drug Conjugates Strongly Depend on Specific Tumor Substructure and Microenvironment" International Journal of Molecular Sciences 21, no. 17: 6029. https://doi.org/10.3390/ijms21176029
APA StyleNoack, A.-K., Lucas, H., Chytil, P., Etrych, T., Mäder, K., & Mueller, T. (2020). Intratumoral Distribution and pH-Dependent Drug Release of High Molecular Weight HPMA Copolymer Drug Conjugates Strongly Depend on Specific Tumor Substructure and Microenvironment. International Journal of Molecular Sciences, 21(17), 6029. https://doi.org/10.3390/ijms21176029








