Roles of Membrane Domains in Integrin-Mediated Cell Adhesion
Abstract
1. Introduction
2. Cytoskeletal Rearrangements
3. Integrin Structure
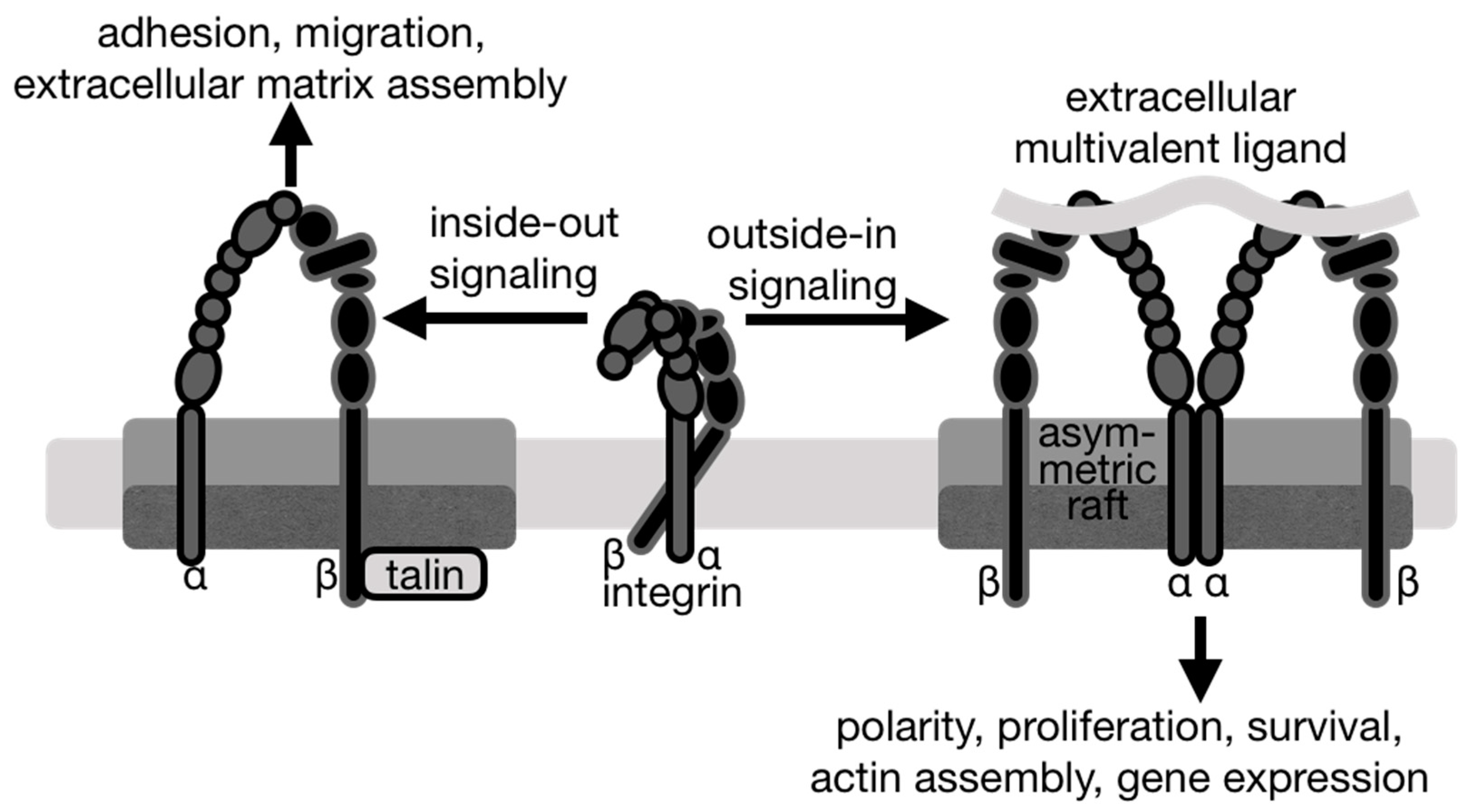
4. Integrin Signaling
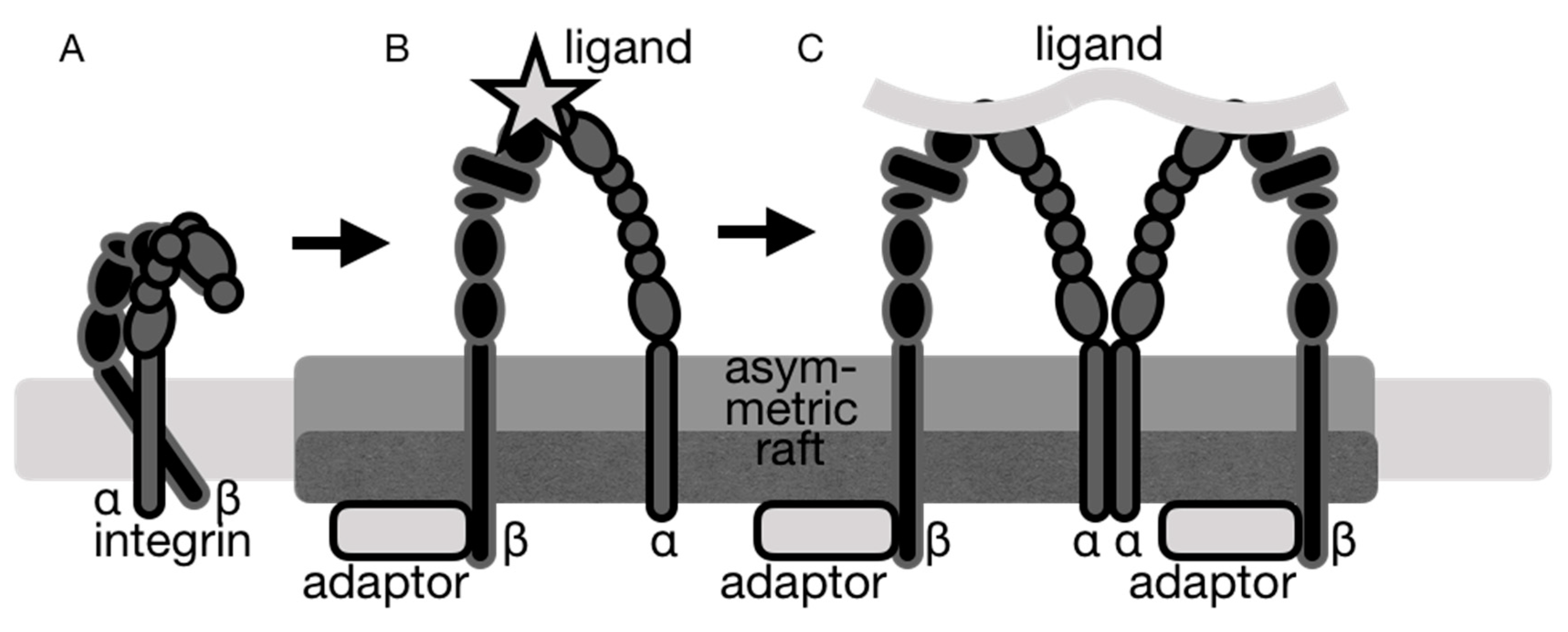
5. Lipid Rafts in Integrin Partitioning
6. Integrins and Lipid Raft Components
7. The Role of Cholesterol
8. Integrin-Glycosphingolipids Interactions
9. Role of Raft Thickness
10. Phosphatidylinositol 4,5-Bisphosphate (PI(4,5)P2) Lipid Microdomains
11. The Role of Integrin Acylation
12. Conclusions and Future Directions
Funding
Acknowledgments
Conflicts of Interest
References
- Danielli, J.F.; D’avson, H. A contribition to the theory of permeability of thin films. J. Cell. Comp. Physiol. 1935, 5, 495–508. [Google Scholar] [CrossRef]
- Singer, S.J.; Nicolson, G.L. The Fluid Mosaic Model of the Structure of Cell Membranes. Science 1972, 175, 720–731. [Google Scholar] [CrossRef]
- Engelman, N.M. Membranes are more mosaic than fluid. Nature 2005, 438, 578–580. [Google Scholar] [CrossRef]
- Goni, F.M. The basic structure and dynamics of cell membranes: An update of the Singer–Nicolson model. Biochim. Biophys. Acta (BBA) Biomembr. 2014, 1838, 1467–1476. [Google Scholar] [CrossRef]
- Walter, H.; Brooks, D.E. Phase separation in cytoplasm, due to macromolecular crowding, is the basis for microcompartmentation. FEBS Lett. 1995, 361, 135–139. [Google Scholar] [CrossRef]
- Simons, K.; Sampaio, J.L. Membrane Organization and Lipid Rafts. Cold Spring Harb. Perspect. Boil. 2011, 3, a004697. [Google Scholar] [CrossRef] [PubMed]
- Nicolson, G.L. The Fluid—Mosaic Model of Membrane Structure: Still relevant to understanding the structure, function and dynamics of biological membranes after more than 40years. Biochim. Biophys. Acta (BBA) Biomembr. 2014, 1838, 1451–1466. [Google Scholar] [CrossRef] [PubMed]
- Petersen, E.N.; Pavel, M.A.; Wang, H.; Hansen, S.B. Disruption of palmitate-mediated localization; a shared pathway of force and anesthetic activation of TREK-1 channels. Biochim. Biophys. Acta Biomembr. 2020, 1862, 183091. [Google Scholar] [CrossRef]
- Brown, D.A.; London, E. Structure of Detergent-Resistant Membrane Domains: Does Phase Separation Occur in Biological Membranes? Biochem. Biophys. Res. Commun. 1997, 240, 1–7. [Google Scholar] [CrossRef]
- Simons, K.; Ikonen, E. Functional rafts in cell membranes. Nature 1997, 387, 569–572. [Google Scholar] [CrossRef]
- Schroeder, R.J.; Ahmed, S.N.; Zhu, Y.; London, E.; Brown, D.A. Cholesterol and sphingolipid enhance the Triton X-100 insolubility of glycosylphosphatidylinositol-anchored proteins by promoting the formation of detergent-insoluble ordered membrane domains. J. Boil. Chem. 1998, 273, 1150–1157. [Google Scholar] [CrossRef]
- Brown, D.A.; London, E. Structure and Function of Sphingolipid- and Cholesterol-rich Membrane Rafts. J. Boil. Chem. 2000, 275, 17221–17224. [Google Scholar] [CrossRef] [PubMed]
- Simons, K.; Toomre, D. Lipid rafts and signal transduction. Nat. Rev. Mol. Cell Boil. 2000, 1, 31–39. [Google Scholar] [CrossRef] [PubMed]
- Kusumi, A.; Nakada, C.; Ritchie, K.; Murase, K.; Suzuki, K.; Murakoshi, H.; Kasai, R.S.; Kondo, J.; Fujiwara, T.K. Paradigm Shift of the Plasma Membrane Concept from the Two-Dimensional Continuum Fluid to the Partitioned Fluid: High-Speed Single-Molecule Tracking of Membrane Molecules. Annu. Rev. Biophys. Biomol. Struct. 2005, 34, 351–378. [Google Scholar] [CrossRef] [PubMed]
- Brown, D.A.; London, E. Functions of lipid rafts in biological membranes. Annu. Rev. Cell Dev. Boil. 1998, 14, 111–136. [Google Scholar] [CrossRef] [PubMed]
- Del Pozo, M.A.; Alderson, N.B.; Kiosses, W.B.; Chiang, H.-H.; Anderson, R.G.W.; Schwartz, M.A. Integrins Regulate Rac Targeting by Internalization of Membrane Domains. Science 2004, 303, 839–842. [Google Scholar] [CrossRef] [PubMed]
- Golub, T.; Wacha, S.; Caroni, P. Spatial and temporal control of signaling through lipid rafts. Curr. Opin. Neurobiol. 2004, 14, 542–550. [Google Scholar] [CrossRef]
- Jacobson, K.; Mouritsen, O.G.; Anderson, R.G.W. Lipid rafts: At a crossroad between cell biology and physics. Nature 2007, 9, 7–14. [Google Scholar] [CrossRef]
- Viola, A.; Gupta, N. Tether and trap: Regulation of membrane-raft dynamics by actin-binding proteins. Nat. Rev. Immunol. 2007, 7, 889–896. [Google Scholar] [CrossRef]
- Vassilieva, E.V.; Gerner-Smidt, K.; Ivanov, A.I.; Nusrat, A. Lipid rafts mediate internalization of beta1-integrin in migrating intestinal epithelial cells. Am. J. Physiol. Liver Physiol. 2008, 295, G965–G976. [Google Scholar] [CrossRef]
- Oneyama, C.; Hikita, T.; Enya, K.; Dobenecker, M.-W.; Saito, K.; Nada, S.; Tarakhovsky, A.; Okada, M. The Lipid Raft-Anchored Adaptor Protein Cbp Controls the Oncogenic Potential of c-Src. Mol. Cell 2008, 30, 426–436. [Google Scholar] [CrossRef]
- Lingwood, D.; Simons, K. Lipid Rafts as a Membrane-Organizing Principle. Science 2009, 327, 46–50. [Google Scholar] [CrossRef] [PubMed]
- Kanerva, K.; Uronen, R.-L.; Blom, T.; Li, S.; Bittman, R.; Lappalainen, P.; Peränen, J.; Raposo, G.; Ikonen, E.; Uronen, R.-L. LDL Cholesterol Recycles to the Plasma Membrane via a Rab8a-Myosin5b-Actin-Dependent Membrane Transport Route. Dev. Cell 2013, 27, 249–262. [Google Scholar] [CrossRef] [PubMed]
- Moissoglu, K.; Kiessling, V.; Wan, C.; Hoffman, B.D.; Norambuena, A.; Tamm, L.K.; Schwartz, M.A. Regulation of Rac1 translocation and activation by membrane domains and their boundaries. J. Cell Sci. 2014, 127, 2565–2576. [Google Scholar] [CrossRef] [PubMed]
- Fiore, V.F. Conformational coupling of integrin and Thy-1 regulates Fyn priming and fibroblast mechanotransduction. J. Cell Biol. 2015, 211, 173–190. [Google Scholar] [CrossRef]
- Krauss, K.; Altevogt, P. Integrin Leukocyte Function-associated Antigen-1-mediated Cell Binding Can Be Activated by Clustering of Membrane Rafts. J. Boil. Chem. 1999, 274, 36921–36927. [Google Scholar] [CrossRef]
- Green, J.M. Role of cholesterol in formation and function of a signaling complex involving alphavbeta3, integrin-associated protein (CD47), and heterotrimeric G proteins. J. Cell Biol. 1999, 146, 673–682. [Google Scholar] [CrossRef]
- Bodin, S.; Utton, M.A.; Noble, W.; Hill, J.E.; Anderton, B.H.; Hanger, D.P. Integrin-dependent interaction of lipid rafts with the actin cytoskeleton in activated human platelets. J. Cell Sci. 2005, 118, 759–769. [Google Scholar] [CrossRef][Green Version]
- Jeon, J.H.; Kim, S.K.; Kim, H.J.; Chang, J.; Ahn, C.M.; Chang, Y.S. Lipid raft modulation inhibits NSCLC cell migration through delocalization of the focal adhesion complex. Lung Cancer 2010, 69, 165–171. [Google Scholar] [CrossRef]
- Murai, T. The Role of Lipid Rafts in Cancer Cell Adhesion and Migration. Int. J. Cell Boil. 2012, 2012, 763283. [Google Scholar] [CrossRef]
- Wang, R. Lipid raft regulates the initial spreading of melanoma A375 cells by modulating beta1 integrin clustering. Int. J. Biochem. Cell Biol. 2013, 45, 1679–1689. [Google Scholar] [CrossRef]
- Orädd, G.; Shahedi, V.; Lindblom, G. Effect of sterol structure on the bending rigidity of lipid membranes: A 2H NMR transverse relaxation study. Biochim. Biophys. Acta Biomembr. 2009, 1788, 1762–1771. [Google Scholar] [CrossRef] [PubMed]
- Shah, A.D.; Inder, K.L.; Shah, A.; Cristino, A.S.; McKie, A.B.; Gabra, H.; Davis, M.J.; Hill, M.M. Integrative Analysis of Subcellular Quantitative Proteomics Studies Reveals Functional Cytoskeleton Membrane–Lipid Raft Interactions in Cancer. J. Proteome Res. 2016, 15, 3451–3462. [Google Scholar] [CrossRef] [PubMed]
- Stickney, J.T.; Bacon, W.C.; Rojas, M.; Ratner, N.; Ip, W. Activation of the tumor suppressor merlin modulates its interaction with lipid rafts. Cancer Res. 2004, 64, 2717–2724. [Google Scholar] [CrossRef] [PubMed][Green Version]
- McClatchey, A.I.; Giovannini, M. Membrane organization and tumorigenesis--the NF2 tumor suppressor, Merlin. Genes Dev. 2005, 19, 2265–2277. [Google Scholar] [CrossRef]
- Chinthalapudi, K.; Mandati, V.; Zheng, J.; Sharff, A.J.; Bricogne, G.; Griffin, P.R.; Kissil, J.; Izard, T. Lipid binding promotes the open conformation and tumor-suppressive activity of neurofibromin 2. Nat. Commun. 2018, 9, 1338. [Google Scholar] [CrossRef]
- Hynes, R.O. Integrins: Bidirectional, allosteric signaling machines. Cell 2002, 110, 673–687. [Google Scholar] [CrossRef]
- Banno, A.; Ginsberg, M.H. Integrin activation. Biochem. Soc. Trans. 2008, 36, 229–234. [Google Scholar] [CrossRef]
- Lee, J.-O.; Rieu, P.; Arnaout, M.; Liddington, R. Crystal structure of the A domain from the a subunit of integrin CR3 (CD11 b/CD18). Cell 1995, 80, 631–638. [Google Scholar] [CrossRef]
- Lu, C.; Takagi, J.; Springer, T.A. Association of the Membrane Proximal Regions of the α and β Subunit Cytoplasmic Domains Constrains an Integrin in the Inactive State. J. Boil. Chem. 2001, 276, 14642–14648. [Google Scholar] [CrossRef]
- Takagi, J.; Erickson, H.P.; Springer, T.A. C-terminal opening mimics ‘inside-out’ activation of integrin alpha5beta1. Nat. Genet. 2001, 8, 412–416. [Google Scholar] [CrossRef]
- Xiong, J.-P.; Stehle, T.; Diefenbach, B.; Zhang, R.; Dunker, R.; Scott, D.L.; Joachimiak, A.; Goodman, S.L.; Arnaout, M.A. Crystal Structure of the Extracellular Segment of Integrin alpha Vbeta 3. Science 2001, 294, 339–345. [Google Scholar] [CrossRef] [PubMed]
- Xiong, J.-P.; Stehle, T.; Zhang, R.; Joachimiak, A.; Frech, M.; Goodman, S.L.; Arnaout, M.A. Crystal Structure of the Extracellular Segment of Integrin alpha Vbeta 3 in Complex with an Arg-Gly-Asp Ligand. Science 2002, 296, 151–155. [Google Scholar] [CrossRef] [PubMed]
- Takagi, J.; Petre, B.M.; Walz, T.; Springer, T.A. Global Conformational Rearrangements in Integrin Extracellular Domains in Outside-In and Inside-Out Signaling. Cell 2002, 110, 599–611. [Google Scholar] [CrossRef]
- Arnaout, M.A. Integrin structure: New twists and turns in dynamic cell adhesion. Immunol. Rev. 2002, 186, 125–140. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.; Carman, C.V.; Springer, T.A. Bidirectional Transmembrane Signaling by Cytoplasmic Domain Separation in Integrins. Science 2003, 301, 1720–1725. [Google Scholar] [CrossRef] [PubMed]
- Carman, C.V.; Springer, T.A. Integrin avidity regulation: Are changes in affinity and conformation underemphasized? Curr. Opin. Cell Biol. 2003, 15, 547–556. [Google Scholar] [CrossRef]
- Xiong, J.-P.; Stehle, T.; Goodman, S.L.; Arnaout, M.A. A Novel Adaptation of the Integrin PSI Domain Revealed from Its Crystal Structure. J. Boil. Chem. 2004, 279, 40252–40254. [Google Scholar] [CrossRef]
- Luo, B.-H.; Springer, T.A.; Takagi, J. A Specific Interface between Integrin Transmembrane Helices and Affinity for Ligand. PLoS Boil. 2004, 2, e153. [Google Scholar] [CrossRef]
- Arnaout, M.A.; Mahalingam, B.; Xiong, J.-P. Integrin structure, allostery, and bidirectional signaling. Annu. Rev. Cell Dev. Boil. 2005, 21, 381–410. [Google Scholar] [CrossRef]
- Adair, B.D. Three-dimensional EM structure of the ectodomain of integrin αVβ3 in a complex with fibronectin. J. Cell Biol. 2005, 168, 1109–1118. [Google Scholar] [CrossRef]
- Luo, B.-H.; Carman, C.V.; Springer, T.A. Structural Basis of Integrin Regulation and Signaling. Annu. Rev. Immunol. 2007, 25, 619–647. [Google Scholar] [CrossRef] [PubMed]
- Arnaout, M.A.; Goodman, S.L.; Xiong, J.P. Structure and mechanics of integrin-based cell adhesion. Curr. Opin. Cell Biol. 2007, 19, 495–507. [Google Scholar] [CrossRef] [PubMed]
- Xiong, J.P. Crystal structure of the complete integrin alphaVbeta3 ectodomain plus an alpha/beta transmembrane fragment. J. Cell. Biol. 2009, 186, 589–600. [Google Scholar] [CrossRef] [PubMed]
- Eng, E.T. Intact alphaIIbbeta3 integrin is extended after activation as measured by solution X-ray scattering and electron microscopy. J. Biol. Chem. 2011, 286, 35218–35226. [Google Scholar] [CrossRef] [PubMed]
- Mahalingam, B.; Ajroud, K.; Alonso, J.L.; Anand, S.; Adair, B.D.; Horenstein, A.L.; Malavasi, F.; Xiong, J.-P.; Arnaout, M.A. Stable coordination of the inhibitory Ca2+ ion at the metal ion-dependent adhesion site in integrin CD11b/CD18 by an antibody-derived ligand aspartate: Implications for integrin regulation and structure-based drug design. J. Immunol. 2011, 187, 6393–6401. [Google Scholar] [CrossRef]
- Adair, B.D.; Xiong, J.-P.; Alonso, J.L.; Hyman, B.T.; Arnaout, M.A. EM Structure of the Ectodomain of Integrin CD11b/CD18 and Localization of Its Ligand-Binding Site Relative to the Plasma Membrane. PLoS ONE 2013, 8, e57951. [Google Scholar] [CrossRef]
- Dai, A.; Ye, F.; Taylor, D.W.; Hu, G.; Ginsberg, M.H.; Taylor, K.A. The Structure of a Full-length Membrane-embedded Integrin Bound to a Physiological Ligand. J. Boil. Chem. 2015, 290, 27168–27175. [Google Scholar] [CrossRef]
- Xu, X.P. Three-Dimensional Structures of Full-Length, Membrane-Embedded Human αIIbβ3 Integrin Complexes. Biophys. J. 2016, 110, 798–809. [Google Scholar] [CrossRef]
- Cormier, A. Cryo-EM structure of the alphavbeta8 integrin reveals a mechanism for stabilizing integrin extension. Nat. Struct. Mol. Biol. 2018, 25, 698–704. [Google Scholar] [CrossRef]
- Lau, T.-L.; Dua, V.; Ulmer, T.S. Structure of the Integrin αIIb Transmembrane Segment. J. Boil. Chem. 2008, 283, 16162–16168. [Google Scholar] [CrossRef]
- Kim, C.; Schmidt, T.; Cho, E.-G.; Ye, F.; Ulmer, T.S.; Ginsberg, M.H. Basic amino-acid side chains regulate transmembrane integrin signalling. Nature 2011, 481, 209–213. [Google Scholar] [CrossRef] [PubMed]
- Hughes, P.E.; Diaz-Gonzalez, F.; Leong, L.; Wu, C.; A McDonald, J.; Shattil, S.J.; Ginsberg, M.H. Breaking the integrin hinge. A defined structural constraint regulates integrin signaling. J. Boil. Chem. 1996, 271, 6571–6574. [Google Scholar] [CrossRef] [PubMed]
- Gottschalk, K.E. Transmembrane signal transduction of the alpha (IIb) beta (3) integrin. Protein Sci. 2002, 11, 1800–1812. [Google Scholar] [CrossRef] [PubMed]
- Armulik, A.; Nilsson, I.; Von Heijne, G.; Johansson, S. Determination of the Border between the Transmembrane and Cytoplasmic Domains of Human Integrin Subunits. J. Boil. Chem. 1999, 274, 37030–37034. [Google Scholar] [CrossRef] [PubMed]
- Lau, T.L. The structure of the integrin alphaIIbbeta3 transmembrane complex explains integrin transmembrane signalling. EMBO J. 2009, 28, 1351–1361. [Google Scholar] [CrossRef] [PubMed]
- Hanein, D.; Volkmann, N. Conformational Equilibrium of Human Platelet Integrin Investigated by Three-Dimensional Electron Cryo-Microscopy; Springer Science and Business Media LLC: Berlin, Germany, 2018; Volume 87, pp. 353–363. [Google Scholar]
- Van Zanten, T.S.; Cambi, A.; Koopman, M.; Joosten, B.; Figdor, C.; Garcia-Parajo, M.F. Hotspots of GPI-anchored proteins and integrin nanoclusters function as nucleation sites for cell adhesion. Proc. Natl. Acad. Sci. USA 2009, 106, 18557–18562. [Google Scholar] [CrossRef] [PubMed]
- Van Kooyk, Y.; Figdor, C. Avidity regulation of integrins: The driving force in leukocyte adhesion. Curr. Opin. Cell Boil. 2000, 12, 542–547. [Google Scholar] [CrossRef]
- Barczyk, M.; Carracedo, S.; Gullberg, D. Integrins. Cell Tissue Res. 2009, 339, 269–280. [Google Scholar] [CrossRef]
- Leitinger, B.; Hogg, N. The involvement of lipid rafts in the regulation of integrin function. J. Cell Sci. 2002, 115, 963–972. [Google Scholar]
- Howe, A.; Aplin, A.; Alahari, S.K.; Juliano, R.L. Integrin signaling and cell growth control. Curr. Opin. Cell Boil. 1998, 10, 220–231. [Google Scholar] [CrossRef]
- Rossi, F.; Yozgat, Y.; De Stanchina, E.; Veach, D.; Clarkson, B.; Manova, K.; Giancotti, F.G.; Antonescu, C.R.; Besmer, P. Imatinib upregulates compensatory integrin signaling in a mouse model of gastrointestinal stromal tumor and is more effective when combined with dasatinib. Mol. Cancer Res. 2010, 8, 1271–1283. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Hynes, R.O.; Naba, A. Overview of the Matrisome—An Inventory of Extracellular Matrix Constituents and Functions. Cold Spring Harb. Perspect. Boil. 2011, 4, a004903. [Google Scholar] [CrossRef] [PubMed]
- Sun, Z.; Guo, S.S.; Fässler, R. Integrin-mediated mechanotransduction. J. Cell Boil. 2016, 215, 445–456. [Google Scholar] [CrossRef] [PubMed]
- Nagasato, A.I.; Yamashita, H.; Matsuo, M.; Ueda, K.; Kioka, N. The distribution of vinculin to lipid rafts plays an important role in sensing stiffness of extracellular matrix. Biosci. Biotechnol. Biochem. 2017, 81, 1136–1147. [Google Scholar] [CrossRef]
- Shattil, S.J.; Kim, C.; Ginsberg, M.H. The final steps of integrin activation: The end game. Nat. Rev. Mol. Cell Boil. 2010, 11, 288–300. [Google Scholar] [CrossRef]
- Palazzo, A.F.; Eng, C.H.; Schlaepfer, D.D.; Marcantonio, E.E.; Gundersen, G.G. Localized Stabilization of Microtubules by Integrin- and FAK-Facilitated Rho Signaling. Science 2004, 303, 836–839. [Google Scholar] [CrossRef]
- Guan, J.-L. Cell biology: Integrins, Rafts, Rac, and Rho. Science 2004, 303, 773–774. [Google Scholar] [CrossRef]
- De Deyne, P.G.; O’Neill, A.; Resneck, W.G.; Dmytrenko, G.M.; Pumplin, D.W.; Bloch, R.J. The vitronectin receptor associates with clathrin-coated membrane domains via the cytoplasmic domain of its beta5 subunit. J. Cell Sci. 1998, 111, 2729–2740. [Google Scholar]
- Milner, R. Distinct roles for astrocyte alphavbeta5 and alphavbeta8 integrins in adhesion and migration. J. Cell Sci. 1999, 112, 4271–4279. [Google Scholar]
- Felsenfeld, D.P.; Schwartzberg, P.L.; Venegas, A.; Tse, R.; Sheetz, M.P. Selective regulation of integrin–cytoskeleton interactions by the tyrosine kinase Src. Nature 1999, 1, 200–206. [Google Scholar] [CrossRef]
- Decker, L.; Ffrench-Constant, C. Lipid Rafts and Integrin Activation Regulate Oligodendrocyte Survival. J. Neurosci. 2004, 24, 3816–3825. [Google Scholar] [CrossRef] [PubMed]
- Gagnoux-Palacios, L. Compartmentalization of integrin alpha6beta4 signaling in lipid rafts. J. Cell Biol. 2003, 162, 1189–1196. [Google Scholar] [CrossRef] [PubMed]
- Thorne, R.F. The integrins alpha3beta1 and alpha6beta1 physically and functionally associate with CD36 in human melanoma cells. Requirement for the extracellular domain OF CD36. J. Biol. Chem. 2000, 275, 35264–35275. [Google Scholar] [CrossRef] [PubMed]
- Claas, C.; Stipp, C.S.; Hemler, M.E. Evaluation of Prototype Transmembrane 4 Superfamily Protein Complexes and Their Relation to Lipid Rafts. J. Boil. Chem. 2000, 276, 7974–7984. [Google Scholar] [CrossRef] [PubMed]
- Baron, W.; Decker, L.; Colognato, H.; Ffrench-Constant, C. Regulation of Integrin Growth Factor Interactions in Oligodendrocytes by Lipid Raft Microdomains. Curr. Boil. 2003, 13, 151–155. [Google Scholar] [CrossRef]
- Gaul, V. The lateral diffusion and fibrinogen induced clustering of platelet integrin alphaIIbbeta3 reconstituted into physiologically mimetic GUVs. Integr. Biol. 2015, 7, 402–411. [Google Scholar] [CrossRef]
- Hussain, N.F.; Siegel, A.P.; Ge, Y.; Jordan, R.; Naumann, C.A. Bilayer Asymmetry Influences Integrin Sequestering in Raft-Mimicking Lipid Mixtures. Biophys. J. 2013, 104, 2212–2221. [Google Scholar] [CrossRef][Green Version]
- Sun, H. Carbon nanotubes enhance intercalated disc assembly in cardiac myocytes via the beta1-integrin-mediated signaling pathway. Biomaterials 2015, 55, 84–95. [Google Scholar] [CrossRef]
- Gopalakrishna, P.; Chaubey, S.K.; Manogaran, P.S.; Pande, G. Modulation of alpha5beta1 integrin functions by the phospholipid and cholesterol contents of cell membranes. J. Cell. Biochem. 2000, 77, 517–528. [Google Scholar] [CrossRef]
- Leitinger, N.; Huber, J.; Rizza, C.; Mechtcheriakova, D.; Bochkov, V.; Koshelnick, Y.; Berliner, J.A.; Binder, B.R.; Goepfert, T.M.; McCarthy, M.; et al. The isoprostane 8-iso-PGF 2α stimulates endothelial cells to bind monocytes: Difference to thromboxane-mediated endothelial activation. FASEB J. 2001, 15, 1254–1256. [Google Scholar] [CrossRef]
- Norambuena, A.; Schwartz, M.A. Effects of integrin-mediated cell adhesion on plasma membrane lipid raft components and signaling. Mol. Boil. Cell 2011, 22, 3456–3464. [Google Scholar] [CrossRef] [PubMed]
- Manes, S.; Del Real, G.; LaCalle, R.A.; Lucas, P.; Gómez-Moutón, C.; Sánchez-Palomino, S.; Delgado, R.; Alcamí, J.; Mira, E.; Martínez-A, C. Membrane raft microdomains mediate lateral assemblies required for HIV-1 infection. EMBO Rep. 2000, 1, 190–196. [Google Scholar] [CrossRef] [PubMed]
- Bi, J.; Wang, R.; Zeng, X. Lipid rafts regulate the lamellipodia formation of melanoma A375 cells via actin cytoskeleton-mediated recruitment of beta1 and beta3 integrin. Oncol. Lett. 2018, 16, 6540–6546. [Google Scholar] [PubMed]
- Salman, M.M.; Kitchen, P.; Woodroofe, M.N.; Bill, R.M.; Conner, A.C.; Heath, P.R.; Conner, M.T. Transcriptome Analysis of Gene Expression Provides New Insights into the Effect of Mild Therapeutic Hypothermia on Primary Human Cortical Astrocytes Cultured under Hypoxia. Front. Cell. Neurosci. 2017, 11, 386. [Google Scholar] [CrossRef]
- Lagos-Cabre, R. Alphavbeta3 Integrin regulates astrocyte reactivity. J. Neuroinflamm. 2017, 14, 194. [Google Scholar] [CrossRef]
- Maldonado, H. Astrocyte-to-neuron communication through integrin-engaged Thy-1/CBP/Csk/Src complex triggers neurite retraction via the RhoA/ROCK pathway. Biochim. Biophys. Acta Mol. Cell Res. 2017, 1864, 243–254. [Google Scholar] [CrossRef]
- Pande, G. The role of membrane lipids in regulation of integrin functions. Curr. Opin. Cell Boil. 2000, 12, 569–574. [Google Scholar] [CrossRef]
- Zhang, K.; Chen, J. The regulation of integrin function by divalent cations. Cell Adhes. Migr. 2012, 6, 20–29. [Google Scholar] [CrossRef]
- O’Donnell, V.B.; Murphy, R.C.; Watson, S.P. Platelet lipidomics: Modern day perspective on lipid discovery and characterization in platelets. Circ. Res. 2014, 114, 1185–1203. [Google Scholar] [CrossRef]
- Green, S.M.; Padula, M.P.; Marks, D.C.; Johnson, L. The Lipid Composition of Platelets and the Impact of Storage: An Overview. Transfus. Med. Rev. 2020, 34, 108–116. [Google Scholar] [CrossRef]
- Pokharel, S.M.; Shil, N.K.; Gc, J.B.; Colburn, Z.T.; Tsai, S.-Y.; Segovia, J.A.; Chang, T.-H.; Bandyopadhyay, S.; Natesan, S.; Jones, J.; et al. Integrin activation by the lipid molecule 25-hydroxycholesterol induces a proinflammatory response. Nat. Commun. 2019, 10, 1482. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.S. Netrin-1-Induced Stem Cell Bioactivity Contributes to the Regeneration of Injured Tissues via the Lipid Raft-Dependent Integrin alpha6beta4 Signaling Pathway. Sci. Rep. 2016, 6, 37526. [Google Scholar] [CrossRef] [PubMed]
- Gimpl, G.; Burger, K.; Fahrenholz, F. Cholesterol as Modulator of Receptor Function. Biochemistry 1997, 36, 10959–10974. [Google Scholar] [CrossRef]
- Su, J.; Gan-Schreier, H.; Goeppert, B.; Chamulitrat, W.; Stremmel, W.; Pathil, A. Bivalent Ligand UDCA-LPE Inhibits Pro-Fibrogenic Integrin Signalling by Inducing Lipid Raft-Mediated Internalization. Int. J. Mol. Sci. 2018, 19, 3254. [Google Scholar] [CrossRef] [PubMed]
- Babel, L. Lipid-rafts remain stable even after ionizing radiation induced disintegration of beta1 integrin containing focal adhesions. BMC Res. Notes 2017, 10, 697. [Google Scholar] [CrossRef] [PubMed]
- Gaus, K.; Le Lay, S.; Balasubramanian, N.; Schwartz, M.A. Integrin-mediated adhesion regulates membrane order. J. Cell Boil. 2006, 174, 725–734. [Google Scholar] [CrossRef]
- Linton, M.R.F. The Role of Lipids and Lipoproteins in Atherosclerosis. In Endotext; Feingold, K.R., Ed.; MDText. com, Inc.: South Dartmouth, MA, USA, 2000. [Google Scholar]
- Tall, A.R.; Yvan-Charvet, L. Cholesterol, inflammation and innate immunity. Nat. Rev. Immunol. 2015, 15, 104–116. [Google Scholar] [CrossRef]
- Wang, H.; Yuan, Z.; Pavel, M.A.; Hansen, S.B. The Role of High Cholesterol in Age-Related COVID19 Lethality. bioRxiv 2020. [Google Scholar] [CrossRef]
- Luo, J.; Yang, H.; Song, B.-L. Mechanisms and regulation of cholesterol homeostasis. Nat. Rev. Mol. Cell Boil. 2019, 21, 225–245. [Google Scholar] [CrossRef]
- Ge, Y.; Gao, J.; Jordan, R.; Naumann, C.A. Changes in Cholesterol Level Alter Integrin Sequestration in Raft-Mimicking Lipid Mixtures. Biophys. J. 2018, 114, 158–167. [Google Scholar] [CrossRef]
- Hancock, J.F. Lipid rafts: Contentious only from simplistic standpoints. Nat. Rev. Mol. Cell Boil. 2006, 7, 456–462. [Google Scholar] [CrossRef] [PubMed]
- Patla, I.; Volberg, T.; Elad, N.; Hirschfeld-Warneken, V.; Grashoff, C.; Fässler, R.; Spatz, J.P.; Geiger, B.; Medalia, O. Dissecting the molecular architecture of integrin adhesion sites by cryo-electron tomography. Nature 2010, 12, 909–915. [Google Scholar] [CrossRef]
- Shibata, A.C.E.; Chen, L.H.; Nagai, R.; Ishidate, F.; Chadda, R.; Miwa, Y.; Naruse, K.; Shirai, Y.M.; Fujiwara, T.K.; Kusumi, A.; et al. Rac1 recruitment to the archipelago structure of the focal adhesion through the fluid membrane as revealed by single-molecule analysis. Cytoskeleton 2013, 70, 161–177. [Google Scholar] [CrossRef] [PubMed]
- Geiger, C.; Agustar, H.K.; Compaoré, G.; Coulibaly, B.; Sié, A.; Becher, H.; Lanzer, M.; Jaenisch, T. Declining malaria parasite prevalence and trends of asymptomatic parasitaemia in a seasonal transmission setting in north-western Burkina Faso between 2000 and 2009–2012. Malar. J. 2013, 12, 27. [Google Scholar] [CrossRef]
- Yates, A.J.; Rampersaud, A. Sphingolipids as Receptor Modulators: An Overview. Ann. N. Y. Acad. Sci. 1998, 845, 57–71. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, J.S.; Brown, W.S.; Woodside, D.G.; Vanderslice, P.; McIntyre, B.W. Clustering T-cell GM1 lipid rafts increases cellular resistance to shear on fibronectin through changes in integrin affinity and cytoskeletal dynamics. Immunol. Cell Boil. 2009, 87, 324–336. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.; Fu, Y.; Gu, M.; Zhang, L.; Li, D.; Li, H.; Chien, S.; Shyy, J.Y.-J.; Zhu, Y. Activation of integrin α5 mediated by flow requires its translocation to membrane lipid rafts in vascular endothelial cells. Proc. Natl. Acad. Sci. USA 2016, 113, 769–774. [Google Scholar] [CrossRef]
- Zhang, C.; Zhou, T.; Chen, Z.; Yan, M.; Li, B.; Lv, H.; Wang, C.; Xiang, S.; Shi, L.; Zhu, Y.; et al. Coupling of Integrin alpha5 to Annexin A2 by Flow Drives Endothelial Activation. Circ. Res. 2020. [Google Scholar] [CrossRef]
- Simons, K.; Ehehalt, R. Cholesterol, lipid rafts, and disease. J. Clin. Investig. 2002, 110, 597–603. [Google Scholar] [CrossRef]
- Caiolfa, V.R.; Zamai, M.; Malengo, G.; Andolfo, A.; Madsen, C.D.; Sutin, J.; Digman, M.A.; Gratton, E.; Blasi, F.; Sidenius, N. Monomer–dimer dynamics and distribution of GPI-anchored uPAR are determined by cell surface protein assemblies. J. Cell Boil. 2007, 179, 1067–1082. [Google Scholar] [CrossRef]
- Ge, Y.; Siegel, A.P.; Jordan, R.; Naumann, C.A. Ligand Binding Alters Dimerization and Sequestering of Urokinase Receptors in Raft-Mimicking Lipid Mixtures. Biophys. J. 2014, 107, 2101–2111. [Google Scholar] [CrossRef] [PubMed]
- Drevot, P.; Langlet, C.; Guo, X.; Bernard, A.; Colard, O.; Chauvin, J.; Lasserre, R.; He, H.-T. TCR signal initiation machinery is pre-assembled and activated in a subset of membrane rafts. EMBO J. 2002, 21, 1899–1908. [Google Scholar] [CrossRef] [PubMed]
- Heberle, F.A.; Petruzielo, R.S.; Pan, J.; Drazba, P.; Kučerka, N.; Standaert, R.; Feigenson, G.W.; Katsaras, J. Bilayer Thickness Mismatch Controls Domain Size in Model Membranes. J. Am. Chem. Soc. 2013, 135, 6853–6859. [Google Scholar] [CrossRef]
- Sezgin, E.; Levental, I.; Mayor, S.; Eggeling, C. The mystery of membrane organization: Composition, regulation and roles of lipid rafts. Nat. Rev. Mol. Cell Boil. 2017, 18, 361–374. [Google Scholar] [CrossRef] [PubMed]
- Kučerka, N.; Pencer, J.; Nieh, M.-P.; Katsaras, J. Influence of cholesterol on the bilayer properties of monounsaturated phosphatidylcholine unilamellar vesicles. Eur. Phys. J. E 2007, 23, 247–254. [Google Scholar] [CrossRef]
- Spiess, M. Active and inactive beta1 integrins segregate into distinct nanoclusters in focal adhesions. J. Cell Biol. 2018, 217, 1929–1940. [Google Scholar] [CrossRef]
- Greisen, P.; Lum, K.; Ashrafuzzaman, M.; Greathouse, D.V.; Andersen, O.S.; Lundbaek, J.A.; Lundbæk, J.A. Linear rate-equilibrium relations arising from ion channel-bilayer energetic coupling. Proc. Natl. Acad. Sci. USA 2011, 108, 12717–12722. [Google Scholar] [CrossRef]
- Gandhavadi, M.; Allende, D.; Vidal, A.; Simon, S.; McIntosh, T. Structure, Composition, and Peptide Binding Properties of Detergent Soluble Bilayers and Detergent Resistant Rafts. Biophys. J. 2002, 82, 1469–1482. [Google Scholar] [CrossRef][Green Version]
- Soubias, O.; Teague, W.E.; Hines, K.G.; Gawrisch, K. Rhodopsin/Lipid Hydrophobic Matching—Rhodopsin Oligomerization and Function. Biophys. J. 2015, 108, 1125–1132. [Google Scholar] [CrossRef]
- Kalli, A.C.; Campbell, I.D.; Sansom, M.S. Multiscale simulations suggest a mechanism for integrin inside-out activation. Proc. Natl. Acad. Sci. USA 2011, 108, 11890–11895. [Google Scholar] [CrossRef]
- Hakanpaa, L. Targeting beta1-integrin inhibits vascular leakage in endotoxemia. Proc. Natl. Acad. Sci. USA 2018, 115, E6467–E6476. [Google Scholar] [CrossRef] [PubMed]
- Partridge, A.W. Transmembrane domain helix packing stabilizes integrin alphaIIbbeta 3 in the low affinity state. J. Biol. Chem. 2005, 280, 7294–7300. [Google Scholar] [CrossRef] [PubMed]
- Berger, B.W.; Kulp, D.W.; Span, L.M.; DeGrado, J.L.; Billings, P.C.; Senes, A.; Bennett, J.S.; DeGrado, W.F. Consensus motif for integrin transmembrane helix association. Proc. Natl. Acad. Sci. USA 2009, 107, 703–708. [Google Scholar] [CrossRef] [PubMed]
- Strandberg, E.; Killian, J. Snorkeling of lysine side chains in transmembrane helices: How easy can it get? FEBS Lett. 2003, 544, 69–73. [Google Scholar] [CrossRef]
- Lau, T.-L.; Partridge, A.W.; Ginsberg, M.H.; Ulmer, T.S. Structure of the Integrin β3 Transmembrane Segment in Phospholipid Bicelles and Detergent Micelles. Biochemistry 2008, 47, 4008–4016. [Google Scholar] [CrossRef]
- Lagarrigue, F.; Kim, C.; Ginsberg, M.H. The Rap1-RIAM-talin axis of integrin activation and blood cell function. Blood 2016, 128, 479–487. [Google Scholar] [CrossRef]
- Li, R.; Mitra, N.; Gratkowski, H.; Vilaire, G.; Litvinov, R.; Nagasami, C.; Weisel, J.W.; Lear, J.D.; DeGrado, W.F.; Bennett, J.S. Activation of Integrin alphaIIbbeta3 by Modulation of Transmembrane Helix Associations. Science 2003, 300, 795–798. [Google Scholar] [CrossRef]
- Li, R.; Gorelik, R.; Nanda, V.; Law, P.B.; Lear, J.D.; DeGrado, W.F.; Bennett, J.S. Dimerization of the Transmembrane Domain of Integrin αIIb Subunit in Cell Membranes. J. Boil. Chem. 2004, 279, 26666–26673. [Google Scholar] [CrossRef]
- Parthasarathy, K.; Lin, X.; Tan, S.M.; Law, S.A.; Torres, J. Transmembrane helices that form two opposite homodimeric interactions: An asparagine scan study of αM and β2 integrins. Protein Sci. 2008, 17, 930–938. [Google Scholar] [CrossRef]
- Li, R. Oligomerization of the integrin alphaIIbbeta3: Roles of the transmembrane and cytoplasmic domains. Proc. Natl. Acad. Sci. USA 2001, 98, 12462–12467. [Google Scholar] [CrossRef]
- Schneider, D.; Engelman, D.M. Involvement of transmembrane domain interactions in signal transduction by alpha/beta integrins. J. Biol. Chem. 2004, 279, 9840–9846. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Metcalf, U.G.; Gorelik, R.; Li, R.; Mitra, N.; Nanda, V.; Law, P.B.; Lear, J.D.; DeGrado, W.F.; Bennett, J.S. A push-pull mechanism for regulating integrin function. Proc. Natl. Acad. Sci. USA 2005, 102, 1424–1429. [Google Scholar] [CrossRef] [PubMed]
- Marsh, D. Energetics of Hydrophobic Matching in Lipid-Protein Interactions. Biophys. J. 2008, 94, 3996–4013. [Google Scholar] [CrossRef] [PubMed]
- Anbazhagan, V.; Schneider, D. The membrane environment modulates self-association of the human GpA TM domain—Implications for membrane protein folding and transmembrane signaling. Biochim. Biophys. Acta (BBA) Biomembr. 2010, 1798, 1899–1907. [Google Scholar] [CrossRef] [PubMed]
- Martel, V.; Racaud-Sultan, C.; Dupe, S.; Marie, C.; Paulhe, F.; Galmiche, A.; Block, M.R.; Albiges-Rizo, C. Conformation, Localization, and Integrin Binding of Talin Depend on Its Interaction with Phosphoinositides. J. Boil. Chem. 2001, 276, 21217–21227. [Google Scholar] [CrossRef] [PubMed]
- Saltel, F. New PI(4,5)P2- and membrane proximal integrin-binding motifs in the talin head control beta3-integrin clustering. J. Cell Biol. 2009, 187, 715–731. [Google Scholar] [CrossRef] [PubMed]
- Anthis, N.J.; Wegener, K.L.; Ye, F.; Kim, C.; Goult, B.T.; Lowe, E.D.; Vakonakis, I.; Bate, N.; Critchley, D.R.; Ginsberg, M.H.; et al. The structure of an integrin/talin complex reveals the basis of inside-out signal transduction. EMBO J. 2009, 28, 3623–3632. [Google Scholar] [CrossRef]
- Di Paolo, G. Recruitment and regulation of phosphatidylinositol phosphate kinase type 1 γ by the FERM domain of talin. Nature 2002, 420, 85–89. [Google Scholar] [CrossRef]
- Ling, K. Type I γ phosphatidylinositol phosphate kinase targets and regulates focal adhesions. Nature 2002, 420, 89–93. [Google Scholar] [CrossRef]
- De Pereda, J.M. Structural basis for phosphatidylinositol phosphate kinase type Igamma binding to talin at focal adhesions. J. Biol. Chem. 2005, 280, 8381–8386. [Google Scholar] [CrossRef]
- Kong, X. Structural basis for the phosphorylation-regulated focal adhesion targeting of type Igamma phosphatidylinositol phosphate kinase (PIPKIgamma) by talin. J. Mol. Biol. 2006, 359, 47–54. [Google Scholar] [CrossRef] [PubMed]
- Slochower, D.R.; Wang, Y.-H.; Tourdot, R.W.; Radhakrishnan, R.; Janmey, P.A. Counterion-mediated pattern formation in membranes containing anionic lipids. Adv. Colloid Interface Sci. 2014, 208, 177–188. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Brown, D.A. PIP2Clustering: From model membranes to cells. Chem. Phys. Lipids 2015, 192, 33–40. [Google Scholar] [CrossRef] [PubMed]
- Levental, I.; Christian, D.A.; Wang, Y.-H.; Madara, J.J.; Discher, D.; Janmey, P.A.; Ilya, L. Calcium-Dependent Lateral Organization in Phosphatidylinositol 4,5-Bisphosphate (PIP2)- and Cholesterol-Containing Monolayers. Biochemistry 2009, 48, 8241–8248. [Google Scholar] [CrossRef]
- James, D.J.; Khodthong, C.; Kowalchyk, J.A.; Martin, T.F.J. Phosphatidylinositol 4,5-bisphosphate regulates SNARE-dependent membrane fusion. J. Cell Boil. 2008, 182, 355–366. [Google Scholar] [CrossRef]
- Bogaart, G.V.D.; Meyenberg, K.; Risselada, H.J.; Amin, H.; Willig, K.I.; Hubrich, B.E.; Dier, M.; Hell, S.W.; Grubmüller, H.; Diederichsen, U.; et al. Membrane protein sequestering by ionic protein–lipid interactions. Nature 2011, 479, 552–555. [Google Scholar] [CrossRef]
- Pike, L.J.; Casey, L. Localization and Turnover of Phosphatidylinositol 4,5-Bisphosphate in Caveolin-enriched Membrane Domains. J. Boil. Chem. 1996, 271, 26453–26456. [Google Scholar] [CrossRef]
- Pike, L.J.; Miller, J.M. Cholesterol depletion delocalizes phosphatidylinositol bisphosphate and inhibits hormone-stimulated phosphatidylinositol turnover. J. Boil. Chem. 1998, 273, 22298–22304. [Google Scholar] [CrossRef]
- Johnson, C.M.; Chichili, G.R.; Rodgers, W. Compartmentalization of Phosphatidylinositol 4,5-Bisphosphate Signaling Evidenced Using Targeted Phosphatases. J. Boil. Chem. 2008, 283, 29920–29928. [Google Scholar] [CrossRef]
- Van Rheenen, J.; Achame, E.M.; Janssen, H.; Calafat, J.; Jalink, K. PIP2 signaling in lipid domains: A critical re-evaluation. EMBO J. 2005, 24, 1664–1673. [Google Scholar] [CrossRef]
- Graber, Z.T.; Gericke, A.; Kooijman, E.E. Phosphatidylinositol-4,5-bisphosphate ionization in the presence of cholesterol, calcium or magnesium ions. Chem. Phys. Lipids 2014, 182, 62–72. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Z.; Redfern, R.; Isler, Y.; Ross, A.H.; Gericke, A. Cholesterol stabilizes fluid phosphoinositide domains. Chem. Phys. Lipids 2014, 182, 52–61. [Google Scholar] [CrossRef] [PubMed]
- Wen, Y.; Vogt, V.M.; Feigenson, G.W. Multivalent Cation-Bridged PI(4,5)P2 Clusters Form at Very Low Concentrations. Biophys. J. 2018, 114, 2630–2639. [Google Scholar] [CrossRef] [PubMed]
- Kwik, J.; Boyle, S.; Fooksman, D.; Margolis, L.; Sheetz, M.P.; Edidin, M. Membrane cholesterol, lateral mobility, and the phosphatidylinositol 4,5-bisphosphate-dependent organization of cell actin. Proc. Natl. Acad. Sci. USA 2003, 100, 13964–13969. [Google Scholar] [CrossRef]
- Cai, X.; Lietha, D.; Ceccarelli, D.F.; Karginov, A.V.; Rajfur, Z.; Jacobson, K.; Hahn, K.M.; Eck, M.J.; Schaller, M.D. Spatial and Temporal Regulation of Focal Adhesion Kinase Activity in Living Cells. Mol. Cell. Boil. 2007, 28, 201–214. [Google Scholar] [CrossRef]
- Palmer, S.M. Lipid binding to the tail domain of vinculin: Specificity and the role of the N and C termini. J. Biol. Chem. 2009, 284, 7223–7231. [Google Scholar] [CrossRef]
- Chinthalapudi, K.; Rangarajan, E.S.; Patil, D.N.; George, E.M.; Brown, D.T.; Izard, T. Lipid binding promotes oligomerization and focal adhesion activity of vinculin. J. Cell Boil. 2014, 207, 643–656. [Google Scholar] [CrossRef]
- Chinthalapudi, K.; Patil, D.N.; Rangarajan, E.S.; Rader, C.; Izard, T. Lipid-Directed Vinculin Dimerization. Biochemistry 2015, 54, 2758–2768. [Google Scholar] [CrossRef]
- Izard, T.; Brown, D.T. Mechanisms and Functions of Vinculin Interactions with Phospholipids at Cell Adhesion Sites. J. Boil. Chem. 2016, 291, 2548–2555. [Google Scholar] [CrossRef]
- Chinthalapudi, K.; Rangarajan, E.S.; Brown, D.T.; Izard, T. Differential lipid binding of vinculin isoforms promotes quasi-equivalent dimerization. Proc. Natl. Acad. Sci. USA 2016, 113, 9539–9544. [Google Scholar] [CrossRef]
- Gilmore, A.P.; Burridge, K. Regulation of vinculin binding to talin and actin by phosphatidyl-inositol-4-5-bisphosphate. Nature 1996, 381, 531–535. [Google Scholar] [CrossRef] [PubMed]
- Goñi, G.M.; Epifano, C.; Boskovic, J.; Camacho-Artacho, M.; Zhou, J.; Bronowska, A.; Martín, M.T.; Eck, M.J.; Kremer, L.; Gräter, F.; et al. Phosphatidylinositol 4,5-bisphosphate triggers activation of focal adhesion kinase by inducing clustering and conformational changes. Proc. Natl. Acad. Sci. USA 2014, 111, E3177–E3186. [Google Scholar] [CrossRef] [PubMed]
- Acebrón, I.; Righetto, R.D.; Schoenherr, C.; de Buhr, S.; Redondo, R.; Culley, J.; Rodríguez, C.F.; Daday, C.; Biyani, N.; Llorca, O.; et al. Structural basis of Focal Adhesion Kinase activation on lipid membranes. EMBO J. 2020, in press. [Google Scholar]
- Chen, X.; Khajeh, J.A.; Ju, J.H.; Gupta, Y.K.; Stanley, C.B.; Do, C.; Heller, W.T.; Aggarwal, A.K.; Callaway, D.J.E.; Bu, Z. Phosphatidylinositol 4,5-Bisphosphate Clusters the Cell Adhesion Molecule CD44 and Assembles a Specific CD44-Ezrin Heterocomplex, as Revealed by Small Angle Neutron Scattering. J. Boil. Chem. 2015, 290, 6639–6652. [Google Scholar] [CrossRef] [PubMed]
- Anderluh, A.; Hofmaier, T.; Klotzsch, E.; Kudlacek, O.; Stockner, T.; Sitte, H.H.; Schütz, G.J. Direct PIP2 binding mediates stable oligomer formation of the serotonin transporter. Nat. Commun. 2017, 8, 14089. [Google Scholar] [CrossRef]
- Bussell, K. Destination lipid rafts. Nat. Rev. Mol. Cell Boil. 2002, 3, 399. [Google Scholar] [CrossRef]
- Zacharias, D.A.; Violin, J.D.; Newton, A.C.; Tsien, R.Y. Partitioning of Lipid-Modified Monomeric GFPs into Membrane Microdomains of Live Cells. Science 2002, 296, 913–916. [Google Scholar] [CrossRef]
- Resh, M.D. Covalent lipid modifications of proteins. Curr. Boil. 2013, 23, R431–R435. [Google Scholar] [CrossRef]
- Rabinovitz, I.; Tsomo, L.; Mercurio, A.M. Protein kinase C-alpha phosphorylation of specific serines in the connecting segment of the beta 4 integrin regulates the dynamics of type II hemidesmosomes. Mol. Cell. Boil. 2004, 24, 4351–4360. [Google Scholar] [CrossRef]
- Dutta, U.; Shaw, L.M. A key tyrosine (Y1494) in the beta4 integrin regulates multiple signaling pathways important for tumor development and progression. Cancer Res. 2008, 68, 8779–8787. [Google Scholar] [CrossRef]
- Germain, E.C.; Santos, T.M.; Rabinovitz, I. Phosphorylation of a novel site on the β4 integrin at the trailing edge of migrating cells promotes hemidesmosome disassembly. Mol. Biol. Cell 2009, 20, 56–67. [Google Scholar] [CrossRef]
- Yang, X.; Kovalenko, O.V.; Tang, W.; Claas, C.; Stipp, C.S.; Hemler, M.E. Palmitoylation supports assembly and function of integrin–tetraspanin complexes. J. Cell Boil. 2004, 167, 1231–1240. [Google Scholar] [CrossRef] [PubMed]
- Soung, Y.H.; Chung, J. Curcumin inhibition of the functional interaction between integrin alpha6beta4 and the epidermal growth factor receptor. Mol. Cancer Ther. 2011, 10, 883–891. [Google Scholar] [CrossRef] [PubMed]
- Anderson, A.M.; Ragan, M.A. Palmitoylation: A protein S-acylation with implications for breast cancer. NPJ Breast Cancer 2016, 2, 16028. [Google Scholar] [CrossRef] [PubMed]
- Hemler, M.E. Specific tetraspanin functions. J. Cell Boil. 2001, 155, 1103–1108. [Google Scholar] [CrossRef]
- Robinson, C.V.; Rohacs, T.; Hansen, S.B. Tools for Understanding Nanoscale Lipid Regulation of Ion Channels. Trends Biochem. Sci. 2019, 44, 795–806. [Google Scholar] [CrossRef]
- Stefansson, A.; Armulik, A.; Nilsson, I.; Von Heijne, G.; Johansson, S. Determination of N- and C-terminal Borders of the Transmembrane Domain of Integrin Subunits. J. Boil. Chem. 2004, 279, 21200–21205. [Google Scholar] [CrossRef]
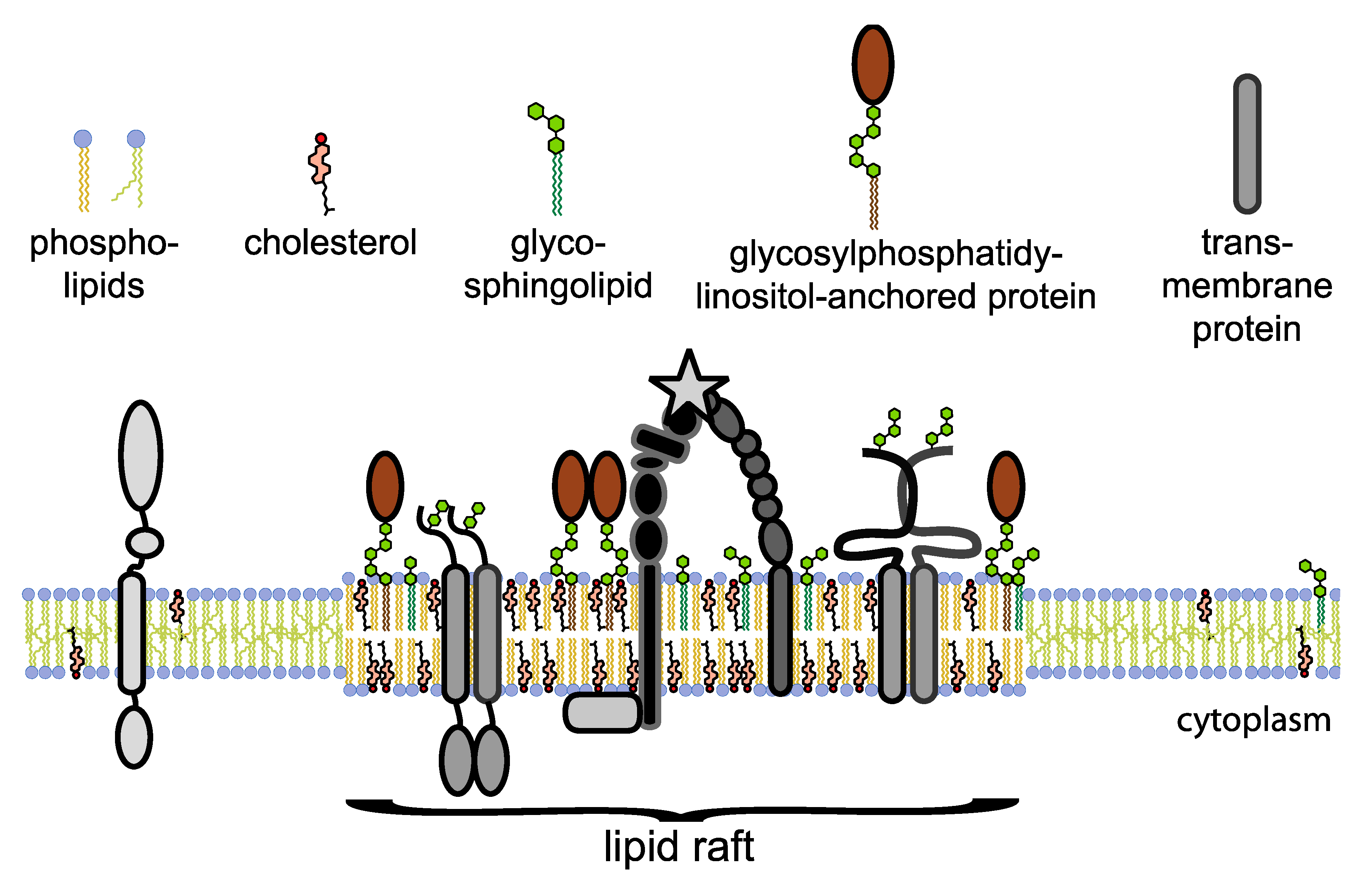

© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lietha, D.; Izard, T. Roles of Membrane Domains in Integrin-Mediated Cell Adhesion. Int. J. Mol. Sci. 2020, 21, 5531. https://doi.org/10.3390/ijms21155531
Lietha D, Izard T. Roles of Membrane Domains in Integrin-Mediated Cell Adhesion. International Journal of Molecular Sciences. 2020; 21(15):5531. https://doi.org/10.3390/ijms21155531
Chicago/Turabian StyleLietha, Daniel, and Tina Izard. 2020. "Roles of Membrane Domains in Integrin-Mediated Cell Adhesion" International Journal of Molecular Sciences 21, no. 15: 5531. https://doi.org/10.3390/ijms21155531
APA StyleLietha, D., & Izard, T. (2020). Roles of Membrane Domains in Integrin-Mediated Cell Adhesion. International Journal of Molecular Sciences, 21(15), 5531. https://doi.org/10.3390/ijms21155531



