Interferons and Dry Eye in Sjögren’s Syndrome
Abstract
1. Introduction
2. Interferons
2.1. Interferons
2.2. Involvement of Type I IFN and Type II IFN
2.3. IFN-γ in SS-Related Dry Eye in Animal Models
2.4. Severe Dry Eye Related to Sjögren’s Syndrome
2.5. The Microbiome in SS-Related Dry Eye and IFN-γ Level
2.6. Sjögren’s Syndrome-Related Dry Eye in Humans
2.7. Treatment for SS Related Dry Eye Patients
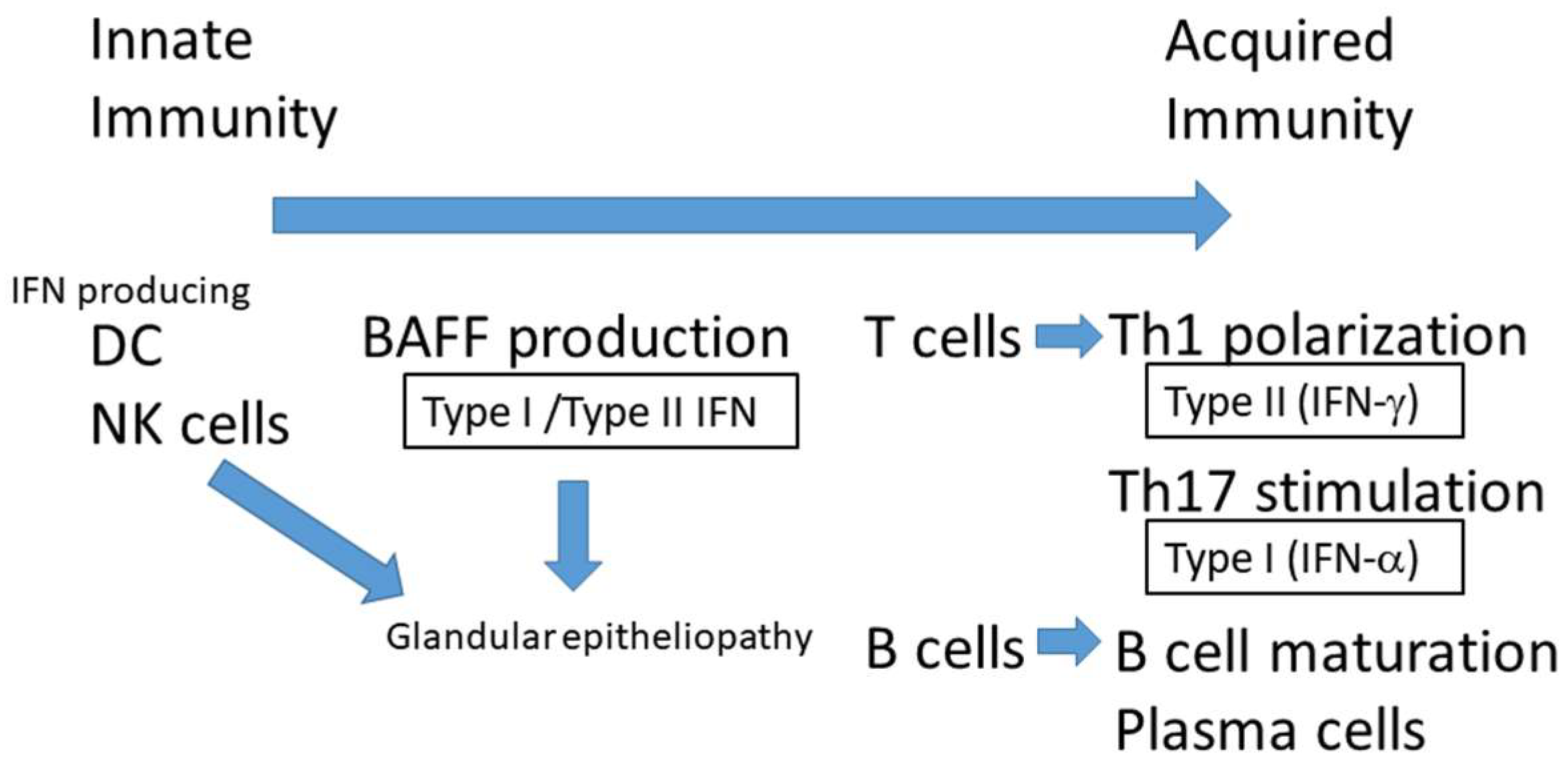
2.8. Hypothetical Pathogenic Processes of SS-Related Dry Eye
3. Future Directions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sumida, T.; Azuma, N.; Moriyama, M.; Takahashi, H.; Asashima, H.; Honda, F.; Abe, S.; Ono, Y.; Hirota, T.; Hirata, S.; et al. Clinical practice guideline for Sjogren’s syndrome 2017. Mod. Rheumatol. 2018, 28, 383–408. [Google Scholar] [CrossRef] [PubMed]
- Nocturne, G.; Mariette, X. Advances in understanding the pathogenesis of primary Sjögren’s syndrome. Nat. Rev. Rheumatol. 2013, 9, 544–556. [Google Scholar] [CrossRef] [PubMed]
- Tsuboi, H.; Hagiwara, S.; Asashima, H.; Takahashi, H.; Hirota, T.; Noma, H.; Umehara, H.; Kawakami, A.; Nakamura, H.; Sano, H.; et al. Comparison of performance of the 2016 ACR-EULAR classification criteria for primary Sjögren’s syndrome with other sets of criteria in Japanese patients. Ann. Rheum. Dis. 2017. [CrossRef] [PubMed]
- Shiboski, C.H.; Shiboski, S.C.; Seror, R.; Criswell, L.A.; Labetoulle, M.; Lietman, T.M.; Rasmussen, A.; Scofield, H.; Vitali, C.; Bowman, S.J.; et al. 2016 American College of Rheumatology/European League Against Rheumatism classification criteria for primary Sjögren’s syndrome: A consensus and data-driven methodology involving three international patient cohorts. Ann. Rheum. Dis. 2017, 76, 9–16. [Google Scholar] [CrossRef] [PubMed]
- Bron, A.J.; de Paiva, C.S.; Chauhan, S.K.; Bonini, S.; Gabison, E.E.; Jain, S.; Knop, E.; Markoulli, M.; Ogawa, Y.; Perez, V.; et al. TFOS DEWS II pathophysiology report. Ocul. Surf. 2017, 15, 438–510. [Google Scholar] [CrossRef] [PubMed]
- Youinou, P.; Pers, J.O. Disturbance of cytokine networks in Sjögren’s syndrome. Arthritis. Res. Ther. 2011, 13, 227. [Google Scholar] [CrossRef] [PubMed]
- Peck, A.B.; Nguyen, C.Q. What can Sjögren’s syndrome-like disease in mice contribute to human Sjögren’s syndrome? Clin. Immunol. 2017, 182, 14–23. [Google Scholar] [CrossRef] [PubMed]
- Schoenborn, J.R.; Wilson, C.B. Regulation of interferon-gamma during innate and adaptive immune responses. Adv. Immunol. 2007, 96, 41–101. [Google Scholar] [PubMed]
- Moore, P.A.; Belvedere, O.; Orr, A.; Pieri, K.; LaFleur, D.W.; Feng, P.; Soppet, D.; Charters, M.; Gentz, R.; Parmelee, D.; Li, Y.; et al. BLyS: Member of the tumor necrosis factor family and B lymphocyte stimulator. Science 1999, 285, 260–263. [Google Scholar] [CrossRef] [PubMed]
- Yoshimoto, K.; Tanaka, M.; Kojima, M.; Setoyama, Y.; Kameda, H.; Suzuki, K.; Tsuzaka, K.; Ogawa, Y.; Tsubota, K.; Abe, T.; et al. Regulatory mechanisms for the production of BAFF and IL-6 are impaired in monocytes of patients of primary Sjögren’s syndrome. Arthritis. Res. Ther. 2011, 13, R170. [Google Scholar] [CrossRef] [PubMed]
- Schneider, P.; MacKay, F.; Steiner, V.; Hofmann, K.; Bodmer, J.L.; Holler, N.; Ambrose, C.; Lawton, P.; Bixler, S.; Acha-Orbea, H.; et al. BAFF, a novel ligand of the tumor necrosis factor family, stimulates B. cell growth. J. Exp. Med. 1999, 189, 1747–1756. [Google Scholar] [CrossRef] [PubMed]
- Platanias, L.C. Mechanisms of type-I- and type-II-interferon-mediated signalling. Nat. Rev. Immunol. 2005, 5, 375–386. [Google Scholar] [CrossRef] [PubMed]
- Marchetti, M.; Monier, M.N.; Fradagrada, A.; Mitchell, K.; Baychelier, F.; Eid, P.; Johannes, L.; Lamaze, C. Stat-mediated signaling induced by type I and type II interferons (IFNs) is differentially controlled through lipid microdomain association and clathrin-dependent endocytosis of IFN receptors. Mol. Biol. Cell. 2006, 17, 2896–2909. [Google Scholar] [CrossRef] [PubMed]
- Lee, A.J.; Ashkar, A.A. The Dual Nature of Type I and Type II Interferons. Front Immunol. 2018, 9, 2061. [Google Scholar] [CrossRef] [PubMed]
- Nordmark, G.; Kristjansdottir, G.; Theander, E.; Eriksson, P.; Brun, J.G.; Wang, C.; Padyukov, L.; Truedsson, L.; Alm, G.; Eloranta, M.L.; et al. Additive effects of the major risk alleles of IRF5 and STAT4 in primary Sjögren’s syndrome. Genes. Immun. 2009, 10, 68–76. [Google Scholar] [CrossRef] [PubMed]
- Hall, J.C.; Baer, A.N.; Shah, A.A.; Criswell, L.A.; Shiboski, C.H.; Rosen, A.; Casciola-Rosen, L. Molecular Subsetting of Interferon Pathways in Sjögren’s Syndrome. Arthritis Rheumatol. 2015, 67, 2437–2446. [Google Scholar] [CrossRef] [PubMed]
- Nezos, A.; Gravani, F.; Tassidou, A.; Kapsogeorgou, E.K.; Voulgarelis, M.; Koutsilieris, M.; Crow, M.K.; Mavragani, C.P. Type I and II interferon signatures in Sjögren’s syndrome pathogenesis: Contributions in distinct clinical phenotypes and Sjögren’s related lymphomagenesis. J. Autoimmun. 2015, 63, 47–58. [Google Scholar] [CrossRef] [PubMed]
- Joung, J.; Konermann, S.; Gootenberg, J.S.; Abudayyeh, O.O.; Platt, R.J.; Brigham, M.D.; Sanjana, N.E.; Zhang, F. Genome-scale CRISPR-Cas9 knockout and transcriptional activation screening. Nat. Protoc. 2017, 12, 828–863. [Google Scholar] [CrossRef] [PubMed]
- Scott, D.A.; Zhang, F. Implications of human genetic variation in CRISPR-based therapeutic genome editing. Nat. Med. 2017, 23, 1095–1101. [Google Scholar] [CrossRef] [PubMed]
- Fox, R.I. Sjögren’s syndrome. Lancet 2005, 366, 321–331. [Google Scholar] [CrossRef]
- Tong, L.; Koh, V.; Thong, B.Y. Review of autoantigens in Sjögren’s syndrome: An update. J. Inflamm. Res. 2017, 10, 97–105. [Google Scholar] [CrossRef] [PubMed]
- Coursey, T.G.; Bohat, R.; Barbosa, F.L.; Pflugfelder, S.C.; de Paiva, C.S. Desiccating stress-induced chemokine expression in the epithelium is dependent on upregulation of NKG2D/RAE-1 and release of IFN-gamma in experimental dry eye. J. Immunol. 2014, 193, 5264–5272. [Google Scholar] [CrossRef] [PubMed]
- Reksten, T.R.; Lessard, C.J.; Sivils, K.L. Genetics in Sjögren Syndrome. Rheum. Dis. Clin. North. Am. 2016, 42, 435–447. [Google Scholar] [CrossRef] [PubMed]
- Bian, F.; Barbosa, F.L.; Corrales, R.M.; Pelegrino, F.S.; Volpe, E.A.; Pflugfelder, S.C.; de Paiva, C.S. Altered balance of interleukin-13/interferon-gamma contributes to lacrimal gland destruction and secretory dysfunction in CD25 knockout model of Sjögren’s syndrome. Arthritis. Res. Ther. 2015, 17, 53. [Google Scholar] [CrossRef] [PubMed]
- Sharma, R.; Zheng, L.; Guo, X.; Fu, S.M.; Ju, S.T.; Jarjour, W.N. Novel animal models for Sjögren’s syndrome: Expression and transfer of salivary gland dysfunction from regulatory T cell-deficient mice. J. Autoimmun. 2006, 27, 289–296. [Google Scholar] [CrossRef] [PubMed]
- Pelegrino, F.S.; Volpe, E.A.; Gandhi, N.B.; Li, D.Q.; Pflugfelder, S.C.; de Paiva, C.S. Deletion of interferon-gamma delays onset and severity of dacryoadenitis in CD25KO mice. Arthritis. Res. Ther. 2012, 14, R234. [Google Scholar] [CrossRef] [PubMed]
- Guimaraes de Souza, R.; Yu, Z.; Stern, M.E.; Pflugfelder, S.C.; de Paiva, C.S. Suppression of Th1-Mediated Keratoconjunctivitis Sicca by Lifitegrast. J. Ocul. Pharmacol. Ther. 2018, 34. [Google Scholar] [CrossRef] [PubMed]
- Meng, Z.; Klinngam, W.; Edman, M.C.; Hamm-Alvarez, S.F. Interferon-gamma treatment in vitro elicits some of the changes in cathepsin S and antigen presentation characteristic of lacrimal glands and corneas from the NOD mouse model of Sjögren’s Syndrome. Clin. Exp. Immunol 2017, 12, e0184781. [Google Scholar]
- Tahara, M.; Tsuboi, H.; Segawa, S.; Asashima, H.; Iizuka-Koga, M.; Hirota, T.; Takahashi, H.; Kondo, Y.; Matsui, M.; Matsumoto, I.; et al. RORgammat antagonist suppresses M3 muscarinic acetylcholine receptor-induced Sjögren’s syndrome-like sialadenitis. Clin. Exp. Immunol. 2017, 187, 213–224. [Google Scholar] [CrossRef] [PubMed]
- Asashima, H.; Tsuboi, H.; Takahashi, H.; Hirota, T.; Iizuka, M.; Kondo, Y.; Matsui, M.; Matsumoto, I.; Sumida, T. The anergy induction of M3 muscarinic acetylcholine receptor-reactive CD4+ T cells suppresses experimental sialadenitis-like Sjögren’s syndrome. Arthritis Rheumatol. 2015, 67, 2213–2225. [Google Scholar] [CrossRef] [PubMed]
- Jabs, D.A.; Prendergast, R.A.; Rorer, E.M.; Hudson, A.P.; Whittum-Hudson, J.A. Cytokines in autoimmune lacrimal gland disease in MRL/MpJ mice. Investig. Ophthalmol. Vis. Sci. 2001, 42, 2567–2571. [Google Scholar] [CrossRef] [PubMed]
- De Paiva, C.S.; Villarreal, A.L.; Corrales, R.M.; Rahman, H.T.; Chang, V.Y.; Farley, W.J.; Stern, M.E.; Niederkorn, J.Y.; Li, D.Q.; Pflugfelder, S.C. Dry eye-induced conjunctival epithelial squamous metaplasia is modulated by interferon-gamma. Investig. Ophthalmol. Vis. Sci. 2007, 48, 2553–2560. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, H.; Asano, K.; Nakamura, S.; Ishida-Yamamoto, A.; Iizuka, H. Interferon-gamma-dependent stimulation of human involucrin gene expression: STAT1 (signal transduction and activators of transcription 1) protein activates involucrin promoter activity. Biochem, J. 1999, 344, 797–802. [Google Scholar] [CrossRef]
- Kawasaki, S.; Kawamoto, S.; Yokoi, N.; Connon, C.; Minesaki, Y.; Kinoshita, S.; Okubo, K. Up-regulated gene expression in the conjunctival epithelium of patients with Sjögren’s syndrome. Exp. Eye. Res. 2003, 77, 17–26. [Google Scholar] [CrossRef]
- Tong, L.; Corrales, R.M.; Chen, Z.; Villarreal, A.L.; De Paiva, C.S.; Beuerman, R.; Li, D.Q.; Pflugfelder, S.C. Expression and regulation of cornified envelope proteins in human corneal epithelium. Invest. Ophthalmol Vis. Sci. 2006, 47, 1938–1946. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Chen, W.; De Paiva, C.S.; Corrales, R.M.; Volpe, E.A.; McClellan, A.J.; Farley, W.J.; Li, D.Q.; Pflugfelder, S.C. Interferon-gamma exacerbates dry eye-induced apoptosis in conjunctiva through dual apoptotic pathways. Investig. Ophthalmol. Vis. Sci. 2011, 52, 6279–6285. [Google Scholar] [CrossRef] [PubMed]
- Delaleu, N.; Madureira, A.C.; Immervoll, H.; Jonsson, R. Inhibition of experimental Sjögren’s syndrome through immunization with HSP60 and its peptide amino acids 437-460. Arthritis Rheum. 2008, 58, 2318–2328. [Google Scholar] [CrossRef] [PubMed]
- Hirai, N.; Kawasaki, S.; Tanioka, H.; Connon, C.J.; Yamasaki, K.; Yokoi, N.; Komuro, A.; Kinoshita, S. Pathological keratinisation in the conjunctival epithelium of Sjögren’s syndrome. Exp. Eye. Res. 2006, 82, 371–378. [Google Scholar] [CrossRef] [PubMed]
- Belizario, J.E.; Napolitano, M. Human microbiomes and their roles in dysbiosis, common diseases, and novel therapeutic approaches. Front. Microbiol. 2015, 6, 1050. [Google Scholar] [CrossRef] [PubMed]
- Coursey, T.G.; Bian, F.; Zaheer, M.; Pflugfelder, S.C.; Volpe, E.A.; de Paiva, C.S. Age-related spontaneous lacrimal keratoconjunctivitis is accompanied by dysfunctional T. regulatory cells. Mucosal. Immunol. 2017, 10, 743–756. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Zaheer, M.; Bian, F.; Quach, D.; Swennes, A.G.; Britton, R.A.; Pflugfelder, S.C.; de Paiva, C.S. Sjögren-Like Lacrimal Keratoconjunctivitis in Germ-Free Mice. Int. J. Mol. Sci. 2018, 19, 565. [Google Scholar] [CrossRef] [PubMed]
- Ogawa, Y.; Yamazaki, K.; Kuwana, M.; Mashima, Y.; Nakamura, Y.; Ishida, S.; Toda, I.; Oguchi, Y.; Tsubota, K.; Okamoto, S.; et al. A significant role of stromal fibroblasts in rapidly progressive dry eye in patients with chronic GVHD. Investig. Ophthalmol. Vis. Sci. 2001, 42, 111–119. [Google Scholar] [PubMed]
- Ogawa, Y.; Kuwana, M.; Yamazaki, K.; Mashima, Y.; Yamada, M.; Mori, T.; Okamoto, S.; Oguchi, Y.; Kawakami, Y. Periductal area as the primary site for T-cell activation in lacrimal gland chronic graft-versus-host disease. Investig. Ophthalmol. Vis. Sci. 2003, 44, 1888–1896. [Google Scholar] [CrossRef] [PubMed]
- Verstappen, G.M.; Corneth, O.B.J.; Bootsma, H.; Kroese, F.G.M. Th17 cells in primary Sjögren’s syndrome: Pathogenicity and plasticity. J. Autoimmun. 2018, 87, 16–25. [Google Scholar] [CrossRef] [PubMed]
- Brauner, S.; Folkersen, L.; Kvarnstrom, M.; Meisgen, S.; Petersen, S.; Franzen-Malmros, M.; Mofors, J.; Brokstad, K.A.; Klareskog, L.; Jonsson, R.; et al. H1N1 vaccination in Sjögren’s syndrome triggers polyclonal B cell activation and promotes autoantibody production. Ann. Rheum. Dis. 2017, 76, 1755–1763. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Reksten, T.R.; Ice, J.A.; Kelly, J.A.; Adrianto, I.; Rasmussen, A. Identification of a Sjögren’s syndrome susceptibility locus at OAS1 that influences isoform switching, protein expression, and responsiveness to type I. interferons. Ann. Rheum. Dis 2017, 13, e1006820. [Google Scholar] [CrossRef] [PubMed]
- Vitali, C.; Bombardieri, S.; Jonsson, R.; Moutsopoulos, H.M.; Alexander, E.L.; Carsons, S.E.; Daniels, T.E.; Fox, P.C.; Fox, R.I.; Kassan, S.S.; et al. Classification criteria for Sjögren’s syndrome: A revised version of the European criteria proposed by the American-European Consensus Group. Ann. Rheum. Dis. 2002, 61, 554–558. [Google Scholar] [CrossRef] [PubMed]
- Bodewes, I.L.A.; Al-Ali, S.; van Helden-Meeuwsen, C.G.; Maria, N.I.; Tarn, J.; Lendrem, D.W.; Schreurs, M.W.J.; Steenwijk, E.C.; van Daele, P.L.A.; Both, T.; et al. Systemic interferon type I and type II signatures in primary Sjögren’s syndrome reveal differences in biological disease activity. Rheumatology (Oxford). 2018, 57, 921–930. [Google Scholar] [CrossRef] [PubMed]
- Ogawa, Y. Sjögren’s Syndrome, Non-Sjögren’s Syndrome, and Graft-Versus-Host Disease Related Dry Eye. Investig. Ophthalmol. Vis. Sci. 2018, 61, 55–58. [Google Scholar]
- Arimoto, A.; Kitagawa, K.; Mita, N.; Takahashi, Y.; Shibuya, E.; Sasaki, H. Effect of rebamipide ophthalmic suspension on signs and symptoms of keratoconjunctivitis sicca in Sjögren syndrome patients with or without punctal occlusions. Cornea. Ann. Rheum. Dis. 2014, 33, 806–811. [Google Scholar] [CrossRef] [PubMed]
- Kinoshita, S.; Awamura, S.; Nakamichi, N.; Suzuki, H.; Oshiden, K.; Yokoi, N.; Rebamipide Ophthalmic Suspension Long-term Study, G. A. multicenter, open-label, 52-week study of 2% rebamipide (OPC-12759) ophthalmic suspension in patients with dry eye. Am. J. Ophthalmol. 2014, 157, 576–583. [Google Scholar] [CrossRef] [PubMed]
- Kinoshita, S.; Oshiden, K.; Awamura, S.; Suzuki, H.; Nakamichi, N.; Yokoi, N.; Rebamipide Ophthalmic Suspension Phase 3 Study, G. A. randomized, multicenter phase 3 study comparing 2% rebamipide (OPC-12759) with 0.1% sodium hyaluronate in the treatment of dry eye. Ophthalmology. Am. J. Ophthalmol 2013, 120, 1158–1165. [Google Scholar] [CrossRef] [PubMed]
- Koh, S.; Maeda, N.; Ikeda, C.; Oie, Y.; Soma, T.; Tsujikawa, M.; Watanabe, H.; Nishida, K. Effect of diquafosol ophthalmic solution on the optical quality of the eyes in patients with aqueous-deficient dry eye. Acta. Ophthalmol. 2014, 92, e671–e675. [Google Scholar] [CrossRef] [PubMed]
- Yokoi, N.; Kato, H.; Kinoshita, S. Facilitation of tear fluid secretion by 3% diquafosol ophthalmic solution in normal human eyes. Am. J. Ophthalmol. 2014, 157, 85–92.e81. [Google Scholar] [CrossRef] [PubMed]
- Cowlen, M.S.; Zhang, V.Z.; Warnock, L.; Moyer, C.F.; Peterson, W.M.; Yerxa, B.R. Localization of ocular P2Y2 receptor gene expression by in situ hybridization. Exp. Eye Res. 2003, 77, 77–84. [Google Scholar] [CrossRef]
- Nakamura, M.; Imanaka, T.; Sakamoto, A. Diquafosol ophthalmic solution for dry eye treatment. Adv. Ther. 2012, 29, 579–589. [Google Scholar] [CrossRef] [PubMed]
- Sugai, S. Sjögren’s syndrome associated with liver and neurological disorders, and malignant lymphoma. Intern. Med. 2000, 39, 193–194. [Google Scholar] [CrossRef] [PubMed]
- Jones, L.; Downie, L.E.; Korb, D.; Benitez-Del-Castillo, J.M.; Dana, R.; Deng, S.X.; Dong, P.N.; Geerling, G.; Hida, R.Y.; Liu, Y.; et al. TFOS DEWS II Management and Therapy Report. Ocul. Surf. 2017, 15, 575–628. [Google Scholar] [CrossRef] [PubMed]
- Baudouin, C.; de la Maza, M.S.; Amrane, M.; Garrigue, J.S.; Ismail, D.; Figueiredo, F.C.; Leonardi, A. One-year efficacy and safety of 0.1% cyclosporine A cationic emulsion in the treatment of severe dry eye disease. Eur. J. Ophthalmol. 2017, 27. [Google Scholar] [CrossRef] [PubMed]
- Lallemand, F.; Schmitt, M.; Bourges, J.L.; Gurny, R.; Benita, S.; Garrigue, J.S. Cyclosporine A delivery to the eye: A comprehensive review of academic and industrial efforts. Eur, J. Pharm Biopharm. 2017, 117, 14–28. [Google Scholar] [CrossRef] [PubMed]
- Lifitegrast (Xiidra) for Dry Eye Disease. JAMA 2017, 317, 1473–1474. [CrossRef] [PubMed]
- Foulks, G.N.; Forstot, S.L.; Donshik, P.C.; Forstot, J.Z.; Goldstein, M.H.; Lemp, M.A.; Nelson, J.D.; Nichols, K.K.; Pflugfelder, S.C.; Tanzer, J.M.; et al. Clinical guidelines for management of dry eye associated with Sjögren disease. Ocul. Surf. 2015, 13, 118–132. [Google Scholar] [CrossRef] [PubMed]
- Geerling, G.; Tauber, J.; Baudouin, C.; Goto, E.; Matsumoto, Y.; O’Brien, T.; Rolando, M.; Tsubota, K.; Nichols, K.K. The international workshop on meibomian gland dysfunction: Report of the subcommittee on management and treatment of meibomian gland dysfunction. Investig. Ophthalmol. Vis. Sci. 2011, 52, 2050–2064. [Google Scholar] [CrossRef] [PubMed]
- Kuklinski, E.; Asbell, P.A. Sjögren’s syndrome from the perspective of ophthalmology. Clin. Immunol. 2017, 182, 55–61. [Google Scholar] [CrossRef] [PubMed]
- Jackson, D.C.; Zeng, W.; Wong, C.Y.; Mifsud, E.J.; Williamson, N.A.; Ang, C.S.; Vingrys, A.J.; Downie, L.E. Tear Interferon-Gamma as a Biomarker for Evaporative Dry Eye Disease. Investig. Ophthalmol. Vis. Sci. 2016, 57, 4824–4830. [Google Scholar] [CrossRef] [PubMed]
- Triantafyllopoulou, A.; Moutsopoulos, H. Persistent viral infection in primary Sjögren’s syndrome: Review and perspectives. Clin. Rev. Allergy. Immunol. 2007, 32, 210–214. [Google Scholar] [CrossRef] [PubMed]
- Sumida, T.; Tsuboi, H.; Iizuka, M.; Hirota, T.; Asashima, H.; Matsumoto, I. The role of M3 muscarinic acetylcholine receptor reactive T cells in Sjögren’s syndrome: A critical review. J. Autoimmun. 2014, 51, 44–50. [Google Scholar] [CrossRef] [PubMed]

© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ogawa, Y.; Shimizu, E.; Tsubota, K. Interferons and Dry Eye in Sjögren’s Syndrome. Int. J. Mol. Sci. 2018, 19, 3548. https://doi.org/10.3390/ijms19113548
Ogawa Y, Shimizu E, Tsubota K. Interferons and Dry Eye in Sjögren’s Syndrome. International Journal of Molecular Sciences. 2018; 19(11):3548. https://doi.org/10.3390/ijms19113548
Chicago/Turabian StyleOgawa, Yoko, Eisuke Shimizu, and Kazuo Tsubota. 2018. "Interferons and Dry Eye in Sjögren’s Syndrome" International Journal of Molecular Sciences 19, no. 11: 3548. https://doi.org/10.3390/ijms19113548
APA StyleOgawa, Y., Shimizu, E., & Tsubota, K. (2018). Interferons and Dry Eye in Sjögren’s Syndrome. International Journal of Molecular Sciences, 19(11), 3548. https://doi.org/10.3390/ijms19113548






