Opinion on the Hurdles and Potential Health Benefits in Value-Added Use of Plant Food Processing By-Products as Sources of Phenolic Compounds
Abstract
1. Introduction
2. Role of Genetics on the Distribution and Biosynthesis of Plant Phenolics
3. Microbiological Safety and Decontamination
4. Characterization of Phenolic Compounds
4.1. Sample Preparation and Phenolic Extraction
4.2. Estimation of Total Phenolic Content (TPC)
4.3. Identification and Quantification of Polyphenols
5. Potential Health Benefits
5.1. Antioxidant Potential
5.2. Neutralization of Metal Ions
5.3. Bioavailability of Phenolics
5.4. Cardiovascular Diseases
5.5. Phenolics as Adjuvants in Cancer Prevention and Treatment
5.6. Type 2 Diabetes and Obesity
5.7. Anti-Inflammatory Effects
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Shahidi, F.; Ambigaipalan, P. Phenolics and polyphenolics in foods, beverages and spices: Antioxidant activity and health effects—A review. J. Funct. Foods 2015, 18, 820–897. [Google Scholar] [CrossRef]
- Zhang, H.; Tsao, R. Dietary polyphenols, oxidative stress and antioxidant and anti-inflammatory effects. Curr. Opin. Food Sci. 2016, 8, 33–42. [Google Scholar] [CrossRef]
- Soares, S.; Kohl, S.; Thalmann, S.; Mateus, N.; Meyerhof, W.; de Freitas, V. Different phenolic compounds activate distinct human bitter taste receptors. J. Agric. Food Chem. 2013, 61, 1525–1533. [Google Scholar] [CrossRef] [PubMed]
- Atanasova-Penichon, V.; Barreau, C.; Richard-Forget, F. Antioxidant secondary metabolites in cereals: Potential involvement in resistance to Fusarium and mycotoxin accumulation. Front. Microbiol. 2016, 7, 566. [Google Scholar] [CrossRef] [PubMed]
- Pradeep, P.M.; Sreerama, Y.N. Soluble and bound phenolics of two different millet genera and their milled fractions: Comparative evaluation of antioxidant properties and inhibitory effects on starch hydrolysing enzyme activities. J. Funct. Foods 2017, 35, 682–693. [Google Scholar] [CrossRef]
- Sun, S.; Kadouh, H.C.; Zhu, W.; Zhou, K. Bioactivity-guided isolation and purification of α-glucosidase inhibitor, 6-O-d-glycosides, from Tinta Cão grape pomace. J. Funct. Foods 2016, 23, 573–579. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Ma, Y.; Gao, F.; Zhao, Y.; Cai, S.; Pang, M. The free, esterified, and insoluble-bound phenolic profiles of Rhus chinensis Mill. fruits and their pancreatic lipase inhibitory activities with molecular docking analysis. J. Funct. Foods 2018, 40, 729–735. [Google Scholar] [CrossRef]
- Melo, P.S.; Massarioli, A.P.; Denny, C.; dos Santos, L.F.; Franchin, M.; Pereira, G.E.; Vieira, T.M.; Rosalen, P.L.; de Alencar, S.M. Winery by-products: Extraction optimization, phenolic composition and cytotoxic evaluation to act as a new source of scavenging of reactive oxygen species. Food Chem. 2015, 181, 160–169. [Google Scholar] [CrossRef] [PubMed]
- Do Valle Calomeni, A.; de Souza, V.B.; Tulini, F.L.; Thomazini, M.; Ostroschi, L.C.; de Alencar, S.M.; Massarioli, A.P.; de Carvalho Balieiro, J.C.; de Carvalho, R.A.; Favaro-Trindade, C.S. Characterization of antioxidant and antimicrobial properties of spray-dried extracts from peanut skins. Food Bioprod. Process. 2017, 105, 215–223. [Google Scholar] [CrossRef]
- Morzelle, M.C.; Salgado, J.M.; Telles, M.; Mourelle, D.; Bachiega, P.; Buck, H.S.; Viel, T.A. Neuroprotective effects of pomegranate peel extract after chronic infusion with amyloid-β peptide in mice. PLoS ONE 2016, 11, e0166123. [Google Scholar] [CrossRef] [PubMed]
- Tremocoldi, M.A.; Rosalen, P.L.; Franchin, M.; Massarioli, A.P.; Denny, C.; Daiuto, É.R.; Paschoal, J.A.R.; Melo, P.S.; de Alencar, S.M. Exploration of avocado by-products as natural sources of bioactive compounds. PLoS ONE 2018, 13, e0192577. [Google Scholar] [CrossRef] [PubMed]
- De Camargo, A.C.; Regitano-d’Arce, M.A.B.; Rasera, G.B.; Canniatti-Brazaca, S.G.; do Prado Silva, L.; Alvarenga, V.O.; Sant’Ana, A.S.; Shahidi, F. Phenolic acids and flavonoids of peanut by-products: Antioxidant capacity and antimicrobial effects. Food Chem. 2017, 237, 538–544. [Google Scholar] [CrossRef] [PubMed]
- Chang, S.K.; Alasalvar, C.; Shahidi, F. Review of dried fruits: Phytochemicals, antioxidant efficacies, and health benefits. J. Funct. Foods 2016, 21, 113–132. [Google Scholar] [CrossRef]
- Alvarez-Parrilla, E.; Urrea-López, R.; de la Rosa, L.A. Bioactive components and health effects of pecan nuts and their byproducts: A review. J. Food Bioact. 2018, 1, 56–92. [Google Scholar]
- De Oliveira, W.P.; Biasoto, A.C.T.; Marques, V.F.; Dos Santos, I.M.; Magalhaes, K.; Correa, L.C.; Negro-Dellacqua, M.; Miranda, M.S.; de Camargo, A.C.; Shahidi, F. Phenolics from winemaking by-products better decrease VLDL-cholesterol and triacylglycerol levels than those of red wine in Wistar rats. J. Food Sci. 2017, 82, 2432–2437. [Google Scholar] [CrossRef] [PubMed]
- De Camargo, A.C.; Regitano-d’Arce, M.A.B.; Biasoto, A.C.T.; Shahidi, F. Low molecular weight phenolics of grape juice and winemaking byproducts: Antioxidant activities and inhibition of oxidation of human low-density lipoprotein cholesterol and DNA strand breakage. J. Agric. Food Chem. 2014, 62, 12159–12171. [Google Scholar] [CrossRef] [PubMed]
- De Camargo, A.C.; Regitano-d’Arce, M.A.B.; Gallo, C.R.; Shahidi, F. Gamma-irradiation induced changes in microbiological status, phenolic profile and antioxidant activity of peanut skin. J. Funct. Foods 2015, 12, 129–143. [Google Scholar] [CrossRef]
- Oldoni, T.L.C.; Melo, P.S.; Massarioli, A.P.; Moreno, I.A.M.; Bezerra, R.M.N.; Rosalen, P.L.; da Silva, G.V.J.; Nascimento, A.M.; Alencar, S.M. Bioassay-guided isolation of proanthocyanidins with antioxidant activity from peanut (Arachis hypogaea) skin by combination of chromatography techniques. Food Chem. 2016, 192, 306–312. [Google Scholar] [CrossRef] [PubMed]
- Ambigaipalan, P.; de Camargo, A.C.; Shahidi, F. Identification of phenolic antioxidants and bioactives of pomegranate seeds following juice extraction using HPLC-DAD-ESI-MSn. Food Chem. 2017, 221, 1883–1894. [Google Scholar] [CrossRef] [PubMed]
- García-Villalba, R.; Espín, J.C.; Aaby, K.; Alasalvar, C.; Heinonen, M.; Jacobs, G.; Voorspoels, S.; Koivumäki, T.; Kroon, P.A.; Pelvan, E.; et al. Validated method for the characterization and quantification of extractable and nonextractable ellagitannins after acid hydrolysis in pomegranate fruits, juices, and extracts. J. Agric. Food Chem. 2015, 63, 6555–6566. [Google Scholar] [CrossRef] [PubMed]
- Ambigaipalan, P.; de Camargo, A.C.; Shahidi, F. Phenolic compounds of pomegranate byproducts (outer skin, mesocarp, divider membrane) and their antioxidant activities. J. Agric. Food Chem. 2016, 64, 6584–6604. [Google Scholar] [CrossRef] [PubMed]
- Molina-Calle, M.; Priego-Capote, F.; de Castro, M.D.L. Development and application of a quantitative method for determination of flavonoids in orange peel: Influence of sample pretreatment on composition. Talanta 2015, 144, 349–355. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Wang, X.; Zhao, C.; Tian, G.; Zhang, H.; Xiao, H.; He, L.; Zheng, J. Chemical mapping of essential oils, flavonoids and carotenoids in citrus peels by Raman microscopy. J. Food Sci. 2017, 82, 2840–2846. [Google Scholar] [CrossRef] [PubMed]
- Ayoub, M.; de Camargo, A.C.; Shahidi, F. Antioxidants and bioactivities of free, esterified and insoluble-bound phenolics from berry seed meals. Food Chem. 2016, 197, 221–232. [Google Scholar] [CrossRef] [PubMed]
- Leite-Legatti, A.V.; Batista, Â.G.; Dragano, N.R.V.; Marques, A.C.; Malta, L.G.; Riccio, M.F.; Eberlin, M.N.; Machado, A.R.T.; de Carvalho-Silva, L.B.; Ruiz, A.L.T.G.; et al. Jaboticaba peel: Antioxidant compounds, antiproliferative and antimutagenic activities. Food Res. Int. 2012, 49, 596–603. [Google Scholar] [CrossRef]
- He, B.; Zhang, L.-L.; Yue, X.-Y.; Liang, J.; Jiang, J.; Gao, X.-L.; Yue, P.-X. Optimization of ultrasound-assisted extraction of phenolic compounds and anthocyanins from blueberry (Vaccinium ashei) wine pomace. Food Chem. 2016, 204, 70–76. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Mendoza, M.P.; Espinosa-Pardo, F.A.; Baseggio, A.M.; Barbero, G.F.; Maróstica Junior, M.R.; Rostagno, M.A.; Martínez, J. Extraction of phenolic compounds and anthocyanins from juçara (Euterpe edulis Mart.) residues using pressurized liquids and supercritical fluids. J. Supercrit. Fluid 2017, 119, 9–16. [Google Scholar] [CrossRef]
- Martini, D.; D’Egidio, M.G.; Nicoletti, I.; Corradini, D.; Taddei, F. Effects of durum wheat debranning on total antioxidant capacity and on content and profile of phenolic acids. J. Funct. Foods 2015, 17, 83–92. [Google Scholar] [CrossRef]
- Hitayezu, R.; Baakdah, M.M.; Kinnin, J.; Henderson, K.; Tsopmo, A. Antioxidant activity, avenanthramide and phenolic acid contents of oat milling fractions. J. Cereal Sci. 2015, 63, 35–40. [Google Scholar] [CrossRef]
- De Camargo, A.C.; Regitano-d’Arce, M.A.B.; Biasoto, A.C.T.; Shahidi, F. Enzyme-assisted extraction of phenolics from winemaking by-products: Antioxidant potential and inhibition of α-glucosidase and lipase activities. Food Chem. 2016, 212, 395–402. [Google Scholar] [CrossRef] [PubMed]
- De Camargo, A.C.; Regitano-d’Arce, M.A.B.; Shahidi, F. Phenolic profile of peanut by-products: Antioxidant potential and inhibition of α-glucosidase and lipase activities. J. Am. Oil Chem. Soc. 2017, 94, 959–971. [Google Scholar] [CrossRef]
- Volstatova, T.; Marsik, P.; Rada, V.; Geigerova, M.; Havlik, J. Effect of apple extracts and selective polyphenols on the adhesion of potential probiotic strains of Lactobacillus gasseri R and Lactobacillus casei FMP. J. Funct. Foods 2017, 35, 391–397. [Google Scholar] [CrossRef]
- De Toledo, N.M.V.; Nunes, L.P.; da Silva, P.P.M.; Spoto, M.H.F.; Canniatti-Brazaca, S.G. Influence of pineapple, apple and melon by-products on cookies: Physicochemical and sensory aspects. Int. J. Food Sci. Technol. 2017, 52, 1185–1192. [Google Scholar] [CrossRef]
- Selani, M.M.; Brazaca, S.G.C.; dos Santos Dias, C.T.; Ratnayake, W.S.; Flores, R.A.; Bianchini, A. Characterisation and potential application of pineapple pomace in an extruded product for fibre enhancement. Food Chem. 2014, 163, 23–30. [Google Scholar] [CrossRef] [PubMed]
- Deng, Q.; Penner, M.H.; Zhao, Y. Chemical composition of dietary fiber and polyphenols of five different varieties of wine grape pomace skins. Food Res. Int. 2011, 44, 2712–2720. [Google Scholar] [CrossRef]
- De Toledo, N.; de Camargo, A.; Ramos, P.; Button, D.; Granato, D.; Canniatti-Brazaca, S. Potentials and pitfalls on the use of passion fruit by-products in drinkable yogurt: Physicochemical, technological, microbiological, and sensory aspects. Beverages 2018, 4, 47. [Google Scholar] [CrossRef]
- Weidner, S.; Powałka, A.; Karamać, M.; Amarowicz, R. Extracts of phenolic compounds from seeds of three wild grapevines—Comparison of their antioxidant activities and the content of phenolic compounds. Int. J. Mol. Sci. 2012, 13, 3444–3457. [Google Scholar] [CrossRef] [PubMed]
- Selani, M.M.; Bianchini, A.; Ratnayake, W.S.; Flores, R.A.; Massarioli, A.P.; de Alencar, S.M.; Canniatti Brazaca, S.G. Physicochemical, functional and antioxidant properties of tropical fruits co-products. Plant Foods Hum. Nutr. 2016, 71, 137–144. [Google Scholar] [CrossRef] [PubMed]
- Black, M.; Bewley, J.D.; Halmer, P. The Encyclopedia of Seeds: Science, Technology and Uses; CAB International: Wallingford, UK, 2006. [Google Scholar]
- Peng, H.; Li, W.; Li, H.; Deng, Z.; Zhang, B. Extractable and non-extractable bound phenolic compositions and their antioxidant properties in seed coat and cotyledon of black soybean (Glycinemax (L.) merr). J. Funct. Foods 2017, 32, 296–312. [Google Scholar] [CrossRef]
- Liyana-Pathirana, C.; Dexter, J.; Shahidi, F. Antioxidant properties of wheat as affected by pearling. J. Agric. Food Chem. 2006, 54, 6177–6184. [Google Scholar] [CrossRef] [PubMed]
- Liyana-Pathirana, C.; Shahidi, F. Optimization of extraction of phenolic compounds from wheat using response surface methodology. Food Chem. 2005, 93, 47–56. [Google Scholar] [CrossRef]
- Liyana-Pathirana, C.M.; Shahidi, F. Importance of insoluble-bound phenolics to antioxidant properties of wheat. J. Agric. Food Chem. 2006, 54, 1256–1264. [Google Scholar] [CrossRef] [PubMed]
- Liyana-Pathirana, C.M.; Shahidi, F. Antioxidant and free radical scavenging activities of whole wheat and milling fractions. Food Chem. 2007, 101, 1151–1157. [Google Scholar] [CrossRef]
- Madhujith, T.; Shahidi, F. Antioxidant and antiproliferative potential of pearled barley (Hordeum vulgarae.). Pharm. Biol. 2008, 46, 88–95. [Google Scholar] [CrossRef]
- Araujo, L.; Bispo, W.M.S.; Rios, J.A.; Fernandes, S.A.; Rodrigues, F.Á. Alkaloids and phenolics biosynthesis increases mango resistance to infection by Ceratocystis fimbriata. Bragantia 2016, 75, 199–211. [Google Scholar] [CrossRef]
- Blum, U. Allelopathic interactions involving phenolic acids. J. Nematol. 1996, 28, 259–267. [Google Scholar] [PubMed]
- Dixit, G.; Praveen, A.; Tripathi, T.; Yadav, V.K.; Verma, P.C. Herbivore-responsive cotton phenolics and their impact on insect performance and biochemistry. J. Asia Pac. Entomol. 2017, 20, 341–351. [Google Scholar] [CrossRef]
- Li, Z.H.; Wang, Q.; Ruan, X.; Pan, C.D.; Jiang, D.A. Phenolics and plant allelopathy. Molecules 2010, 15, 8933–8952. [Google Scholar] [CrossRef] [PubMed]
- Mandal, S.M.; Chakraborty, D.; Dey, S. Phenolic acids act as signaling molecules in plant-microbe symbioses. Plant Signal. Behav. 2010, 5, 359–368. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Guo, X.; Harivandi, M.A. Allelopathic effects of phenolic acids detected in buffalograss (Buchloe dactyloides) clippings on growth of annual bluegrass (Poa annua) and buffalograss seedlings. Environ. Exp. Bot. 1998, 39, 159–167. [Google Scholar] [CrossRef]
- Rani, P.U.; Pratyusha, S. Defensive role of Gossypium hirsutum L. anti-oxidative enzymes and phenolic acids in response to Spodoptera litura F. feeding. J. Asia Pac. Entomol. 2013, 16, 131–136. [Google Scholar] [CrossRef]
- Catalá, R.; Medina, J.; Salinas, J. Integration of low temperature and light signaling during cold acclimation response in Arabidopsis. Proc. Natl. Acad. Sci. USA 2011, 108, 16475. [Google Scholar] [CrossRef] [PubMed]
- Gould, K.S. Nature’s Swiss army knife: The diverse protective roles of anthocyanins in leaves. J. Biomed. Biotechnol. 2004, 2004, 314–320. [Google Scholar] [CrossRef] [PubMed]
- Hernández, I.; Alegre, L.; Van Breusegem, F.; Munné-Bosch, S. How relevant are flavonoids as antioxidants in plants? Trends Plant Sci. 2009, 14, 125–132. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Tikunov, Y.; Schouten, R.E.; Marcelis, L.F.M.; Visser, R.G.F.; Bovy, A. Anthocyanin biosynthesis and degradation mechanisms in Solanaceous vegetables: A Review. Front. Chem. 2018, 6, 52. [Google Scholar] [CrossRef] [PubMed]
- Passeri, V.; Koes, R.; Quattrocchio, F.M. New challenges for the design of high value plant products: Stabilization of anthocyanins in plant vacuoles. Front. Plant Sci. 2016, 7, 153. [Google Scholar] [CrossRef] [PubMed]
- Owens, D.K.; Crosby, K.C.; Runac, J.; Howard, B.A.; Winkel, B.S.J. Biochemical and genetic characterization of Arabidopsis flavanone 3β-hydroxylase. Plant Physiol. Biochem. 2008, 46, 833–843. [Google Scholar] [CrossRef] [PubMed]
- Pollastri, S.; Tattini, M. Flavonols: Old compounds for old roles. Ann. Bot. 2011, 108, 1225–1233. [Google Scholar] [CrossRef] [PubMed]
- Dakora, F.D.; Phillips, D.A. Diverse functions of isoflavonoids in legumes transcend anti-microbial definitions of phytoalexins. Physiol. Mol. Plant Pathol. 1996, 49, 1–20. [Google Scholar] [CrossRef]
- Dixon, R.A.; Pasinetti, G.M. Flavonoids and isoflavonoids: From plant biology to agriculture and neuroscience. Plant. Physiol. 2010, 154, 453–457. [Google Scholar] [CrossRef] [PubMed]
- Samanta, A.; Das, G.; Das, S. Roles of flavonoids in plants. Int. J. Pharm. Sci. Technol. 2011, 6, 12–35. [Google Scholar]
- Wang, X. Structure, function, and engineering of enzymes in isoflavonoid biosynthesis. Funct. Integr. Genom. 2011, 11, 13–22. [Google Scholar] [CrossRef] [PubMed]
- Barbehenn, R.V.; Peter Constabel, C. Tannins in plant–herbivore interactions. Phytochemistry 2011, 72, 1551–1565. [Google Scholar] [CrossRef] [PubMed]
- De Colmenares, N.G.; Ramírez-Martínez, J.R.; Aldana, J.O.; Ramos-Niño, M.E.; Clifford, M.N.; Pékerar, S.; Méndez, B. Isolation, characterisation and determination of biological activity of coffee proanthocyanidins. J. Sci. Food Agric. 1999, 77, 368–372. [Google Scholar] [CrossRef]
- Tuominen, A.; Toivonen, E.; Mutikainen, P.; Salminen, J.-P. Defensive strategies in Geranium sylvaticum. Part 1: Organ-specific distribution of water-soluble tannins, flavonoids and phenolic acids. Phytochemistry 2013, 95, 394–407. [Google Scholar] [CrossRef] [PubMed]
- Barbehenn, R.V.; Jaros, A.; Lee, G.; Mozola, C.; Weir, Q.; Salminen, J.-P. Hydrolyzable tannins as “quantitative defenses”: Limited impact against Lymantria dispar caterpillars on hybrid poplar. J. Insect Physiol. 2009, 55, 297–304. [Google Scholar] [CrossRef] [PubMed]
- Salminen, J.-P.; Karonen, M. Chemical ecology of tannins and other phenolics: We need a change in approach. Funct. Ecol. 2011, 25, 325–338. [Google Scholar] [CrossRef]
- Salminen, J.-P.; Ossipov, V.; Haukioja, E.; Pihlaja, K. Seasonal variation in the content of hydrolysable tannins in leaves of Betula pubescens. Phytochemistry 2001, 57, 15–22. [Google Scholar] [CrossRef]
- Tuominen, A. Defensive strategies in Geranium sylvaticum, Part 2: Roles of water-soluble tannins, flavonoids and phenolic acids against natural enemies. Phytochemistry 2013, 95, 408–420. [Google Scholar] [CrossRef] [PubMed]
- Boerjan, W.; Ralph, J.; Baucher, M. Lignin Biosynthesis. Annu. Rev. Plant Biol. 2003, 54, 519–546. [Google Scholar] [CrossRef] [PubMed]
- Humphreys, J.M.; Chapple, C. Rewriting the lignin roadmap. Curr. Opin. Plant Biol. 2002, 5, 224–229. [Google Scholar] [CrossRef]
- Liu, Q.; Luo, L.; Zheng, L. Lignins: Biosynthesis and biological functions in plants. Int. J. Mol. Sci. 2018, 19, 335. [Google Scholar] [CrossRef] [PubMed]
- Malavasi, U.C.; Davis, A.S.; Malavasi, M.M. Lignin in woody plants under water stress: A review. Floresta Ambient. 2016, 23, 589–597. [Google Scholar] [CrossRef]
- Monties, B. Plant cell walls as fibrous lignocellulosic composites: Relations with lignin structure and function. Anim. Feed Sci. Technol. 1991, 32, 159–175. [Google Scholar] [CrossRef]
- Bagniewska-Zadworna, A.; Barakat, A.; Łakomy, P.; Smoliński, D.J.; Zadworny, M. Lignin and lignans in plant defence: Insight from expression profiling of cinnamyl alcohol dehydrogenase genes during development and following fungal infection in Populus. Plant Sci. 2014, 229, 111–121. [Google Scholar] [CrossRef] [PubMed]
- Harmatha, J.; Dinan, L. Biological activities of lignans and stilbenoids associated with plant-insect chemical interactions. Phytochem. Rev. 2003, 2, 321–330. [Google Scholar] [CrossRef]
- Cunha, W.; Andrade e Silva, M.; Cassio, R.; Ambrosio, S.; Kenupp, J. Lignans: Chemical and biological properties. In Phytochemicals—A Global Perspective of Their Role in Nutrition and Health; Rao, V., Ed.; InTech: Rijeka, Croatia, 2012. [Google Scholar]
- Lewis, N.G.; Kato, M.J.; Lopes, N.; Davin, L.B. Lignans: Diversity, biosynthesis, and function. In Chemistry of the Amazon; American Chemical Society: Washington, DC, USA, 1995; Volume 588, pp. 135–167. [Google Scholar]
- John, J.A.; Shahidi, F. Phenolic compounds and antioxidant activity of Brazil nut (Bertholletia excelsa). J. Funct. Foods 2010, 2, 196–209. [Google Scholar] [CrossRef]
- Arruda, H.S.; Pereira, G.A.; de Morais, D.R.; Eberlin, M.N.; Pastore, G.M. Determination of free, esterified, glycosylated and insoluble-bound phenolics composition in the edible part of araticum fruit (Annona crassiflora Mart.) and its by-products by HPLC-ESI-MS/MS. Food Chem. 2018, 245, 738–749. [Google Scholar] [CrossRef] [PubMed]
- Kammerer, D.; Claus, A.; Carle, R.; Schieber, A. Polyphenol screening of pomace from red and white grape varieties (Vitis vinifera L.) by HPLC-DAD-MS/MS. J. Agric. Food Chem. 2004, 52, 4360–4367. [Google Scholar] [CrossRef] [PubMed]
- Levin, D. Plant phenolics: An ecological perspective. Am. Nat. 1971, 105, 157–181. [Google Scholar] [CrossRef]
- Fraser, C.M.; Chapple, C. The phenylpropanoid pathway in Arabidopsis. In The Arabidopsis Book; American Society of Plant Biologists: Rockville, MD, USA, 2011. [Google Scholar]
- Ghasemzadeh, A.; Ghasemzadeh, N. Flavonoids and phenolic acids: Role and biochemical activity in plants and human. J. Med. Plant Res. 2011, 5, 6697–6703. [Google Scholar] [CrossRef]
- Grundhöfer, P.; Niemetz, R.; Schilling, G.; Gross, G.G. Biosynthesis and subcellular distribution of hydrolyzable tannins. Phytochemistry 2001, 57, 915–927. [Google Scholar] [CrossRef]
- Haslam, E. Vegetable tannins—Lessons of a phytochemical lifetime. Phytochemistry 2007, 68, 2713–2721. [Google Scholar] [CrossRef] [PubMed]
- Ossipov, V.; Salminen, J.-P.; Ossipova, S.; Haukioja, E.; Pihlaja, K. Gallic acid and hydrolysable tannins are formed in birch leaves from an intermediate compound of the shikimate pathway. Biochem. Syst. Ecol. 2003, 31, 3–16. [Google Scholar] [CrossRef]
- Winkel-Shirley, B. Flavonoid biosynthesis. A colorful model for genetics, biochemistry, cell biology, and biotechnology. Plant Physiol. 2001, 126, 485–493. [Google Scholar] [CrossRef] [PubMed]
- Vogt, T. Phenylpropanoid biosynthesis. Mol. Plant. 2010, 3, 2–20. [Google Scholar] [CrossRef] [PubMed]
- Tsao, R. Chemistry and biochemistry of dietary polyphenols. Nutrients 2010, 2, 1231–1246. [Google Scholar] [CrossRef] [PubMed]
- Holton, T.A.; Cornish, E.C. Genetics and biochemistry of anthocyanin biosynthesis. Plant Cell 1995, 7, 1071–1083. [Google Scholar] [CrossRef] [PubMed]
- Shoeva, O.Y.; Mock, H.-P.; Kukoeva, T.V.; Börner, A.; Khlestkina, E.K. Regulation of the flavonoid biosynthesis pathway genes in purple and black grains of Hordeum vulgare. PLoS ONE 2016, 11, e0163782. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.-S.; Huang, Y.; Wang, F.; Song, X.; Wang, G.-L.; Xiong, A.-S. Transcript profiling of structural genes involved in cyanidin-based anthocyanin biosynthesis between purple and non-purple carrot (Daucus carota L.) cultivars reveals distinct patterns. BMC Plant Biol. 2014, 14, 262. [Google Scholar] [CrossRef] [PubMed]
- Yildiz, M.; Willis, D.K.; Cavagnaro, P.F.; Iorizzo, M.; Abak, K.; Simon, P.W. Expression and mapping of anthocyanin biosynthesis genes in carrot. Theor. Appl. Genet. 2013, 126, 1689–1702. [Google Scholar] [CrossRef] [PubMed]
- Koornneef, M. Mutations affecting the testa colour in Arabidopsis. Arabinose Inf. Serv. 1990, 27, 1–4. [Google Scholar]
- Shirley, B.W. Flavonoids in seeds and grains: Physiological function, agronomic importance and the genetics of biosynthesis. Seed Sci. Res. 1998, 8, 415–422. [Google Scholar] [CrossRef]
- Mol, J.; Grotewold, E.; Koes, R. How genes paint flowers and seeds. Trends Plant Sci. 1998, 3, 212–217. [Google Scholar] [CrossRef]
- Quattrocchio, F.; Baudry, A.; Lepiniec, L.; Grotewold, E. The regulation of flavonoid biosynthesis, in The Science of Flavonoids; Springer: New York, NY, USA, 2006; pp. 97–122. [Google Scholar]
- Heim, M.A.; Jakoby, M.; Werber, M.; Martin, C.; Weisshaar, B.; Bailey, P.C. The basic helix-loop-helix transcription factor family in plants: A genome-wide study of protein structure and functional diversity. Mol. Biol. Evol. 2003, 20, 735–747. [Google Scholar] [CrossRef] [PubMed]
- Kranz, H.D.; Denekamp, M.; Greco, R.; Jin, H.; Leyva, A.; Meissner, R.C.; Petroni, K.; Urzainqui, A.; Bevan, M.; Martin, C.; et al. Towards functional characterisation of the members of the R2R3-MYB gene family from Arabidopsis thaliana. Plant J. 1998, 16, 263–276. [Google Scholar] [CrossRef] [PubMed]
- Stracke, R.; Werber, M.; Weisshaar, B. The R2R3-MYB gene family in Arabidopsis thaliana. Curr. Opin. Plant Biol. 2001, 4, 447–456. [Google Scholar] [CrossRef]
- Wang, L.; Ran, L.; Hou, Y.; Tian, Q.; Li, C.; Liu, R.; Fan, D.; Luo, K. The transcription factor MYB115 contributes to the regulation of proanthocyanidin biosynthesis and enhances fungal resistance in poplar. New Phytol. 2017, 215, 351–367. [Google Scholar] [CrossRef] [PubMed]
- De Vetten, N.; Quattrocchio, F.; Mol, J.; Koes, R. The an11 locus controlling flower pigmentation in petunia encodes a novel WD-repeat protein conserved in yeast, plants, and animals. Genes Dev. 1997, 11, 1422–1434. [Google Scholar] [CrossRef] [PubMed]
- Sompornpailin, K.; Makita, Y.; Yamazaki, M.; Saito, K. A WD-repeat-containing putative regulatory protein in anthocyanin biosynthesis in Perilla frutescens. Plant Mol. Biol. 2002, 50, 485–495. [Google Scholar] [CrossRef] [PubMed]
- Carey, C.C.; Strahle, J.T.; Selinger, D.A.; Chandler, V.L. Mutations in the pale aleurone color1 regulatory gene of the Zea mays anthocyanin pathway have distinct phenotypes relative to the functionally similar TRANSPARENT TESTA GLABRA1 gene in Arabidopsis thaliana. Plant Cell 2004, 16, 450–464. [Google Scholar] [CrossRef] [PubMed]
- Kiferle, C.; Fantini, E.; Bassolino, L.; Povero, G.; Spelt, C.; Buti, S.; Giuliano, G.; Quattrocchio, F.; Koes, R.; Perata, P.; et al. Tomato R2R3-MYB proteins SlANT1 and SlAN2: Same protein activity, different roles. PLoS ONE 2015, 10, e0136365. [Google Scholar] [CrossRef] [PubMed]
- Nesi, N.; Jond, C.; Debeaujon, I.; Caboche, M.; Lepiniec, L. The Arabidopsis TT2 gene encodes an R2R3 MYB domain protein that acts as a key determinant for proanthocyanidin accumulation in developing seed. Plant Cell 2001, 13, 2099–2114. [Google Scholar] [PubMed]
- Spelt, C.; Quattrocchio, F.; Mol, J.N.; Koes, R. Anthocyanin1 of petunia encodes a basic helix-loop-helix protein that directly activates transcription of structural anthocyanin genes. Plant Cell 2000, 12, 1619–1632. [Google Scholar] [CrossRef] [PubMed]
- Smeriglio, A.; Barreca, D.; Bellocco, E.; Trombetta, D. Chemistry, pharmacology and health benefits of anthocyanins. Phytother. Res 2016, 30, 1265–1286. [Google Scholar] [CrossRef] [PubMed]
- Francisco, R.M.; Regalado, A.; Ageorges, A.; Burla, B.J.; Bassin, B.; Eisenach, C.; Zarrouk, O.; Vialet, S.; Marlin, T.; Chaves, M.M.; et al. ABCC1, an ATP binding cassette protein from grape berry, transports anthocyanidin 3-O-Glucosides. Plant Cell 2013, 25, 1840–1854. [Google Scholar] [CrossRef] [PubMed]
- Hu, D.G.; Sun, C.H.; Ma, Q.J.; You, C.X.; Cheng, L.; Hao, Y.J. MdMYB1 regulates anthocyanin and malate accumulation by directly facilitating their transport into vacuoles in apples. Plant Physiol. 2016, 170, 1315–1330. [Google Scholar] [PubMed]
- Xu, W.; Dubos, C.; Lepiniec, L. Transcriptional control of flavonoid biosynthesis by MYB–bHLH–WDR complexes. Trends Plant Sci. 2015, 20, 176–185. [Google Scholar] [CrossRef] [PubMed]
- Lepiniec, L.; Debeaujon, I.; Routaboul, J.M.; Baudry, A.; Pourcel, L.; Nesi, N.; Caboche, M. Genetics and biochemistry of seed flavonoids. Annu. Rev. Plant Biol. 2006, 57, 405–430. [Google Scholar] [CrossRef] [PubMed]
- Xu, W.; Bobet, S.; Le Gourrierec, J.; Grain, D.; De Vos, D.; Berger, A.; Salsac, F.; Kelemen, Z.; Boucherez, J.; Rolland, A.; et al. Transparent testa 16 and 15 act through different mechanisms to control proanthocyanidin accumulation in Arabidopsis testa. J. Exp. Bot. 2017, 68, 2859–2870. [Google Scholar] [CrossRef] [PubMed]
- Stracke, R.; Jahns, O.; Keck, M.; Tohge, T.; Niehaus, K.; Fernie, A.R.; Weisshaar, B. Analysis of production of flavonol glycosides-dependent flavonol glycoside accumulation in Arabidopsis thaliana plants reveals MYB11-, MYB12- and MYB111-independent flavonol glycoside accumulation. New Phytol. 2010, 188, 985–1000. [Google Scholar] [CrossRef] [PubMed]
- Muleke, E.M.; Fan, L.; Wang, Y.; Xu, L.; Zhu, X.; Zhang, W.; Cao, Y.; Karanja, B.K.; Liu, L. Coordinated regulation of anthocyanin biosynthesis genes confers varied phenotypic and spatial-temporal anthocyanin accumulation in radish (Raphanus sativus L.). Front. Plant Sci. 2017, 8, 1243. [Google Scholar] [CrossRef] [PubMed]
- Barba-Espín, G.; Glied, S.; Crocoll, C.; Dzhanfezova, T.; Joernsgaard, B.; Okkels, F.; Lütken, H.; Müller, R. Foliar-applied ethephon enhances the content of anthocyanin of black carrot roots (Daucus carota ssp. sativus var. atrorubens Alef.). BMC Plant Biol. 2017, 17, 70. [Google Scholar]
- Mano, H.; Ogasawara, F.; Sato, K.; Higo, H.; Minobe, Y. Isolation of a regulatory gene of anthocyanin biosynthesis in tuberous roots of purple-fleshed sweet potato. Plant Physiol. 2007, 143, 1252–1268. [Google Scholar] [CrossRef] [PubMed]
- Ben-Simhon, Z.; Judeinstein, S.; Trainin, T.; Harel-Beja, R.; Bar-Ya’akov, I.; Borochov-Neori, H.; Holland, D. A “white” anthocyanin-less pomegranate (Punica granatum L.) caused by an insertion in the coding region of the leucoanthocyanidin dioxygenase (LDOX; ANS) gene. PLoS ONE 2015, 10, e0142777. [Google Scholar] [CrossRef] [PubMed]
- Vanderauwera, S.; Zimmermann, P.; Rombauts, S.; Vandenabeele, S.; Langebartels, C.; Gruissem, W.; Inze, D.; Van Breusegem, F. Genome-wide analysis of hydrogen peroxide-regulated gene expression in Arabidopsis reveals a high light-induced transcriptional cluster involved in anthocyanin biosynthesis. Plant Physiol. 2005, 139, 806–821. [Google Scholar] [CrossRef] [PubMed]
- Catola, S.; Castagna, A.; Santin, M.; Calvenzani, V.; Petroni, K.; Mazzucato, A.; Ranieri, A. The dominant allele Aft induces a shift from flavonol to anthocyanin production in response to UV-B radiation in tomato fruit. Planta 2017, 246, 263–275. [Google Scholar] [CrossRef] [PubMed]
- Ilk, N.; Ding, J.; Ihnatowicz, A.; Koornneef, M.; Reymond, M. Natural variation for anthocyanin accumulation under high-light and low-temperature stress is attributable to the enhancer of AG-4 2 (HUA2) locus in combination with production of anthocyanin pigment1 (PAP1) and PAP2. New Phytol. 2015, 206, 422–435. [Google Scholar] [CrossRef] [PubMed]
- Pourcel, L.; Routaboul, J.-M.; Kerhoas, L.; Caboche, M.; Lepiniec, L.; Debeaujon, I. Transparent testa10 encodes a laccase-like enzyme involved in oxidative polymerization of flavonoids in Arabidopsis seed coat. Plant Cell 2005, 17, 2966–2980. [Google Scholar] [CrossRef] [PubMed]
- Bueso, E.; Munoz-Bertomeu, J.; Campos, F.; Brunaud, V.; Martinez, L.; Sayas, E.; Ballester, P.; Yenush, L.; Serrano, R. ARABIDOPSIS THALIANA HOMEOBOX25 uncovers a role for Gibberellins in seed longevity. Plant Physiol. 2014, 164, 999–1010. [Google Scholar] [CrossRef] [PubMed]
- Debeaujon, I.; Leon-Kloosterziel, K.M.; Koornneef, M. Influence of the testa on seed dormancy, germination, and longevity in Arabidopsis. Plant Physiol. 2000, 122, 403–414. [Google Scholar] [CrossRef] [PubMed]
- Windsor, J.B.; Symonds, V.V.; Mendenhall, J.; Lloyd, A.M. Arabidopsis seed coat development: Morphological differentiation of the outer integument. Plant J. 2000, 22, 483–493. [Google Scholar] [CrossRef] [PubMed]
- Smýkal, P.; Vernoud, V.; Blair, M.W.; Soukup, A.; Thompson, R.D. The role of the testa during development and in establishment of dormancy of the legume seed. Front. Plant Sci. 2014, 5, 351. [Google Scholar] [PubMed]
- Dixon, R.A.; Xie, D.Y.; Sharma, S.B. Proanthocyanidins—A final frontier in flavonoid research? New Phytol. 2005, 165, 9–28. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.-F.; Doligez, A.; Fournier-Level, A.; Le Cunff, L.; Bertrand, Y.; Canaguier, A.; Morel, C.; Miralles, V.; Veran, F.; Souquet, J.-M.; et al. Dissecting genetic architecture of grape proanthocyanidin composition through quantitative trait locus mapping. BMC Plant Biol. 2012, 12, 30. [Google Scholar] [CrossRef] [PubMed]
- Paolocci, F.; Robbins, M.P.; Passeri, V.; Hauck, B.; Morris, P.; Rubini, A.; Arcioni, S.; Damiani, F. The strawberry transcription factor FaMYB1 inhibits the biosynthesis of proanthocyanidins in Lotus corniculatus leaves. J. Exp. Bot. 2011, 62, 1189–1200. [Google Scholar] [CrossRef] [PubMed]
- Baudry, A.; Heim, M.A.; Dubreucq, B.; Caboche, M.; Weisshaar, B.; Lepiniec, L. TT2, TT8, and TTG1 synergistically specify the expression of BANYULS and proanthocyanidin biosynthesis in Arabidopsis thaliana. Plant J. 2004, 39, 366–380. [Google Scholar] [CrossRef] [PubMed]
- Escaray, F.J.; Passeri, V.; Babuin, F.M.; Marco, F.; Carrasco, P.; Damiani, F.; Pieckenstain, F.L.; Paolocci, F.; Ruiz, O.A. Lotus tenuis × L. corniculatus interspecific hybridization as a means to breed bloat-safe pastures and gain insight into the genetic control of proanthocyanidin biosynthesis in legumes. BMC Plant Biol. 2014, 14, 40. [Google Scholar] [CrossRef] [PubMed]
- Jende-Strid, B. Genetic control of flavonoid biosynthesis in barley. Hereditas 1993, 119, 187–204. [Google Scholar] [CrossRef]
- Aastrup, S.; Outtrup, H.; Erdal, K. Location of the proanthocyanidins in the barley grain. Carlsberg Res. Commun. 1984, 49, 105–109. [Google Scholar] [CrossRef]
- Harlan, H. Some Distinctions in Our Cultivated Barleys with Reference to their Use in Plant Breeding; Department of Agriculture Press: Washington, DC, USA, 1914. [Google Scholar]
- Naz, A.A.; Reinert, S.; Bostanci, C.; Seperi, B.; Leon, J.; Böttger, C.; Südekum, K.-H.; Frei, M. Mining the global diversity for bioenergy traits of barley straw: Genomewide association study under varying plant water status. GCB Bioenergy 2017, 9, 1356–1369. [Google Scholar] [CrossRef]
- Robards, K.; Antolovich, M. Analytical chemistry of fruit bioflavonoids. A Review. Analyst 1997, 122, 11R–34R. [Google Scholar] [CrossRef]
- Perez-Gregorio, M.R.; Regueiro, J.; Simal-Gandara, J.; Rodrigues, A.S.; Almeida, D.P. Increasing the added-value of onions as a source of antioxidant flavonoids: A critical review. Crit. Rev. Food Sci. Nutr. 2014, 54, 1050–1062. [Google Scholar] [CrossRef] [PubMed]
- Ciancolini, A.; Alignan, M.; Pagnotta, M.A.; Miquel, J.; Vilarem, G.; Crinò, P. Morphological characterization, biomass and pharmaceutical compounds in Italian globe artichoke genotypes. Ind. Crops Prod. 2013, 49, 326–333. [Google Scholar] [CrossRef]
- Jeffery, E.H.; Brown, A.F.; Kurilich, A.C.; Keck, A.S.; Matusheski, N.; Klein, B.P.; Juvik, J.A. Variation in content of bioactive components in broccoli. J. Food Compos. Anal. 2003, 16, 323–330. [Google Scholar] [CrossRef]
- Martinez, M.; Poirrier, P.; Chamy, R.; Prüfer, D.; Schulze-Gronover, C.; Jorquera, L.; Ruiz, G. Taraxacum officinale and related species—An ethnopharmacological review and its potential as a commercial medicinal plant. J. Ethnopharmacol. 2015, 169, 244–262. [Google Scholar] [CrossRef] [PubMed]
- Paredes-Lopez, O.; Cervantes-Ceja, M.L.; Vigna-Perez, M.; Hernandez-Perez, T. Berries: Improving human health and healthy aging, and promoting quality life—A review. Plant Foods Hum. Nutr. 2010, 65, 299–308. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Kosińska-Cagnazzo, A.; Kerr, W.L.; Amarowicz, R.; Swanson, R.B.; Pegg, R.B. Separation and characterization of phenolic compounds from dry-blanched peanut skins by liquid chromatography–electrospray ionization mass spectrometry. J. Chromatogr. A 2014, 1356, 64–81. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.Y.; Kerr, W.L.; Swanson, R.B.; Hargrove, J.L.; Pegg, R.B. Peanut skins-fortified peanut butters: Effect of processing on the phenolics content, fibre content and antioxidant activity. Food Chem. 2014, 145, 883–891. [Google Scholar] [CrossRef] [PubMed]
- De Camargo, A.C.; Vieira, T.M.F.S.; Regitano-d’Arce, M.A.B.; de Alencar, S.M.; Calori-Domingues, M.A.; Spoto, M.H.F.; Canniatti-Brazaca, S.G. Gamma irradiation of in-shell and blanched peanuts protects against mycotoxic fungi and retains their nutraceutical components during long-term storage. Int. J. Mol. Sci. 2012, 13, 10935–10958. [Google Scholar] [CrossRef] [PubMed]
- Pacheco, A.M.; Scussel, V.M. Selenium and aflatoxin levels in raw Brazil nuts from the Amazon basin. J. Agric. Food Chem. 2007, 55, 11087–11092. [Google Scholar] [CrossRef] [PubMed]
- Belluco, B.; de Camargo, A.C.; da Gloria, E.M.; Dias, C.T.D.S.; Button, D.C.; Calori-Domingues, M.A. Deoxynivalenol in wheat milling fractions: A critical evaluation regarding ongoing and new legislation limits. J. Cereal Sci. 2017, 77, 284–290. [Google Scholar] [CrossRef]
- Baert, K.; De Meulenaer, B.; Kamala, A.; Kasase, C.; Devlieghere, F. Occurrence of patulin in organic, conventional, and handcrafted apple juices marketed in Belgium. J. Food Prot. 2006, 69, 1371–1378. [Google Scholar] [CrossRef] [PubMed]
- Piemontese, L.; Solfrizzo, M.; Visconti, A. Occurrence of patulin in conventional and organic fruit products in Italy and subsequent exposure assessment. Food Addit. Contam. 2005, 22, 437–442. [Google Scholar] [CrossRef] [PubMed]
- Sekhon-Loodu, S.; Warnakulasuriya, S.N.; Rupasinghe, H.P.V.; Shahidi, F. Antioxidant ability of fractionated apple peel phenolics to inhibit fish oil oxidation. Food Chem. 2013, 140, 189–196. [Google Scholar] [CrossRef] [PubMed]
- Wilson-Kakashita, G.; Gerdes, D.L.; Hall, W.R. The effect of gamma irradiation on the quality of English walnuts (Junglans regia). LWT Food Sci. Technol. 1995, 28, 17–20. [Google Scholar] [CrossRef]
- Grant, I.R.; Nixon, C.R.; Patterson, M.F. Effect of low-dose irradiation on growth of and toxin production by Staphylococcus aureus and Bacillus cereus in roast beef and gravy. Int. J. Food Microbiol. 1993, 18, 25–36. [Google Scholar] [CrossRef]
- Aouidi, F.; Ayari, S.; Ferhi, H.; Roussos, S.; Hamdi, M. Gamma irradiation of air-dried olive leaves: Effective decontamination and impact on the antioxidative properties and on phenolic compounds. Food Chem. 2011, 127, 1105–1113. [Google Scholar] [CrossRef] [PubMed]
- De Camargo, A.C.; Vieira, T.M.F.S.; Regitano-d’Arce, M.A.B.; de Alencar, S.M.; Calori-Domingues, M.A.; Canniatti-Brazaca, S.G. Gamma radiation induced oxidation and tocopherols decrease in in-shell, peeled and blanched peanuts. Int. J. Mol. Sci. 2012, 13, 2827–2845. [Google Scholar] [CrossRef] [PubMed]
- Kilcast, D. Effect of irradiation on vitamins. Food Chem. 1994, 49, 157–164. [Google Scholar] [CrossRef]
- Koike, A.; Barreira, J.C.M.; Barros, L.; Santos-Buelga, C.; Villavicencio, A.L.C.H.; Ferreira, I.C.F.R. Irradiation as a novel approach to improve quality of Tropaeolum majus L. flowers. Innov. Food Sci. Emerg. Technol. 2015, 30, 138–144. [Google Scholar] [CrossRef]
- Marszałek, K.; Mitek, M.; Skąpska, S. The effect of thermal pasteurization and high pressure processing at cold and mild temperatures on the chemical composition, microbial and enzyme activity in strawberry purée. Innov. Food Sci. Emerg. Technol. 2015, 27, 48–56. [Google Scholar] [CrossRef]
- Halpin-Dohnalek, M.I.; Marth, E.H. Staphylococcus aureus: Production of extracellular compounds and behavior in foods—A review. J. Food Prot. 1989, 52, 267–282. [Google Scholar] [CrossRef]
- Santos Alexandre, A.P.; Vela-Paredes, R.S.; Santos, A.S.; Costa, N.S.; Canniatti-Brazaca, S.G.; Calori-Domingues, M.A.; Augusto, P.E.D. Ozone treatment to reduce deoxynivalenol (DON) and zearalenone (ZEN) contamination in wheat bran and its impact on nutritional quality. Food Addit. Contam. A 2018, 1189–1199. [Google Scholar] [CrossRef] [PubMed]
- Pinela, J.; Ferreira, I.C.F.R. Nonthermal physical technologies to decontaminate and extend the shelf-life of fruits and vegetables: Trends aiming at quality and safety. Crit. Rev. Food Sci. Nutr. 2017, 57, 2095–2111. [Google Scholar] [CrossRef] [PubMed]
- Barcia, M.T.; Pertuzatti, P.B.; Gómez-Alonso, S.; Godoy, H.T.; Hermosín-Gutiérrez, I. Phenolic composition of grape and winemaking by-products of Brazilian hybrid cultivars BRS Violeta and BRS Lorena. Food Chem. 2014, 159, 95–105. [Google Scholar] [CrossRef] [PubMed]
- De Camargo, A.C.; Vidal, C.M.M.; Canniatti-Brazaca, S.G.; Shahidi, F. Fortification of cookies with peanut skins: Effects on the composition, polyphenols, antioxidant properties, and sensory quality. J. Agric. Food Chem. 2014, 62, 11228–11235. [Google Scholar] [CrossRef] [PubMed]
- Shahidi, F.; de Camargo, A.C. Tocopherols and tocotrienols in common and emerging dietary sources: Occurrence, applications, and health benefits. Int. J. Mol. Sci. 2016, 17, 1745. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, A.C.; Jorge, N. Bioactive compounds of the lipid fractions of agro-industrial waste. Food Res. Int. 2014, 66, 493–500. [Google Scholar] [CrossRef]
- Arranz, S.; Cert, R.; Perez-Jimenez, J.; Cert, A.; Saura-Calixto, F. Comparison between free radical scavenging capacity and oxidative stability of nut oils. Food Chem. 2008, 110, 985–990. [Google Scholar] [CrossRef] [PubMed]
- De Camargo, A.C.; Canniatti-Brazaca, S.G.; Vieira, T.M.F.S.; Regitano-d’Arce, M.A.B.; Calori-Domingues, M.A. Gamma radiation effects on peanut skin antioxidants. In Proceedings of the 2011 International Nuclear Atlantic Conference, Belo Horizonte, Brazil, 24–28 October 2011; pp. 1–10. [Google Scholar]
- Krygier, K.; Sosulski, F.; Hogge, L. Free, esterified, and insoluble-bound phenolic acids. 2. Composition of phenolic acids in rapeseed flour and hulls. J. Agric. Food Chem. 1982, 30, 334–336. [Google Scholar] [CrossRef]
- Naczk, M.; Shahidi, F. The effect of methanol-ammonia-water treatment on the content of phenolic acids of canola. Food Chem. 1989, 31, 159–164. [Google Scholar] [CrossRef]
- Chen, G.-L.; Zhang, X.; Chen, S.-G.; Han, M.-D.; Gao, Y.-Q. Antioxidant activities and contents of free, esterified and insoluble-bound phenolics in 14 subtropical fruit leaves collected from the south of China. J. Funct. Foods 2017, 30, 290–302. [Google Scholar] [CrossRef]
- Chandrasekara, A.; Shahidi, F. Bioactivities and antiradical properties of millet grains and hulls. J. Agric. Food Chem. 2011, 59, 9563–9571. [Google Scholar] [CrossRef] [PubMed]
- Yoshiara, L.Y.; Madeira, T.B.; Delaroza, F.; Da Silva, J.B.; Ida, E.I. Optimization of soy isoflavone extraction with different solvents using the simplex-centroid mixture design. Int. J. Food Sci. Nutr. 2012, 63, 978–986. [Google Scholar] [CrossRef] [PubMed]
- Handa, C.L.; de Lima, F.S.; Guelfi, M.F.G.; Georgetti, S.R.; Ida, E.I. Multi-response optimisation of the extraction solvent system for phenolics and antioxidant activities from fermented soy flour using a simplex-centroid design. Food Chem. 2016, 197, 175–184. [Google Scholar] [CrossRef] [PubMed]
- Chandrasekara, A.; Rasek, O.A.; John, J.A.; Chandrasekara, N.; Shahidi, F. Solvent and extraction conditions control the assayable phenolic content and antioxidant activities of seeds of black beans, canola and millet. J. Am. Oil Chem. Soc. 2016, 93, 275–283. [Google Scholar] [CrossRef]
- Ksibi, I.E.; Slama, R.B.; Faidi, K.; Ticha, M.B.; M’henni, M.F. Mixture approach for optimizing the recovery of colored phenolics from red pepper (Capsicum annum L.) by-products as potential source of natural dye and assessment of its antimicrobial activity. Ind. Crops Prod. 2015, 70, 34–40. [Google Scholar] [CrossRef]
- Xu, D.P.; Li, Y.; Meng, X.; Zhou, T.; Zhou, Y.; Zheng, J.; Zhang, J.J.; Li, H.B. Natural antioxidants in foods and medicinal plants: Extraction, assessment and resources. Int. J. Mol. Sci. 2017, 18, 96. [Google Scholar] [CrossRef] [PubMed]
- Dinkova, R.; Heffels, P.; Shikov, V.; Weber, F.; Schieber, A.; Mihalev, K. Effect of enzyme-assisted extraction on the chilled storage stability of bilberry (Vaccinium myrtillus L.) anthocyanins in skin extracts and freshly pressed juices. Food Res. Int. 2014, 65, 35–41. [Google Scholar] [CrossRef]
- Sarnoski, P.J.; Johnson, J.V.; Reed, K.A.; Tanko, J.M.; O’Keefe, S.F. Separation and characterisation of proanthocyanidins in Virginia type peanut skins by LC-MSn. Food Chem. 2012, 131, 927–939. [Google Scholar] [CrossRef]
- Aaby, K.; Grimmer, S.; Holtung, L. Extraction of phenolic compounds from bilberry (Vaccinium myrtillus L.) press residue: Effects on phenolic composition and cell proliferation. LWT Food Sci. Technol. 2013, 54, 257–264. [Google Scholar] [CrossRef]
- Zhang, Y.; Wong, A.I.C.; Wu, J.; Abdul Karim, N.B.; Huang, D. Lepisanthes alata (Malay cherry) leaves are potent inhibitors of starch hydrolases due to proanthocyanidins with high degree of polymerization. J. Funct. Foods 2016, 25, 568–578. [Google Scholar] [CrossRef]
- Huynh, N.T.; Smagghe, G.; Gonzales, G.B.; Van Camp, J.; Raes, K. Enzyme-assisted extraction enhancing the phenolic release from cauliflower (Brassica oleracea L. var. botrytis) outer leaves. J. Agric. Food Chem. 2014, 62, 7468–7476. [Google Scholar] [CrossRef] [PubMed]
- Esposito, F.; Arlotti, G.; Maria Bonifati, A.; Napolitano, A.; Vitale, D.; Fogliano, V. Antioxidant activity and dietary fibre in durum wheat bran by-products. Food Res. Int. 2005, 38, 1167–1173. [Google Scholar] [CrossRef]
- Barba, F.J.; Zhu, Z.; Koubaa, M.; Sant’Ana, A.S.; Orlien, V. Green alternative methods for the extraction of antioxidant bioactive compounds from winery wastes and by-products: A review. Trends Food Sci. Technol. 2016, 49, 96–109. [Google Scholar] [CrossRef]
- Casazza, A.A.; Aliakbarian, B.; De Faveri, D.; Fiori, L.; Perego, P. Antioxidants from winemaking wastes: A study on extraction parameters using response surface methodology. J. Food Biochem. 2012, 36, 28–37. [Google Scholar] [CrossRef]
- De Camargo, A.C.; de Souza, T.M.F.; Rasera, G.B.; Canniatti-Brazaca, S.G.; de Alencar, S.M.; Regitano-d’Arce, M.A.B. Lower solvent concentration and time for extraction of peanut skin antioxidants at optimized conditions. In Peanuts: Production, Nutritional Content and Health Implications; Nova Publishers: Hauppauge, NY, USA, 2014; pp. 31–50. [Google Scholar]
- Ferreres, F.; Grosso, C.; Gil-Izquierdo, A.; Valentão, P.; Mota, A.T.; Andrade, P.B. Optimization of the recovery of high-value compounds from pitaya fruit by-products using microwave-assisted extraction. Food Chem. 2017, 230, 463–474. [Google Scholar] [CrossRef] [PubMed]
- Falcão, H.G.; Handa, C.L.; Ramos, M.B.; Silva de, A.C.; Camargo Shahidi, F.; Kurozawa, L.E.; Ida, E.I. Soybean ultrasound pre-treatment prior to soaking affects β-glucosidase activity, isoflavone profile and soaking time. Food Chem. 2018, 269, 404–412. [Google Scholar]
- Swain, T.; Hillis, W.E. The phenolic constituents of Prunus domestica. I.—The quantitative analysis of phenolic constituents. J. Sci. Food Agric. 1959, 10, 63–68. [Google Scholar] [CrossRef]
- Singleton, V.L.; Orthofer, R.; Lamuela-Raventós, R.M. Analysis of total phenols and other oxidation substrates and antioxidants by means of folin-ciocalteu reagent. In Methods in Enzymology; Academic Press: Cambridge, MA, USA, 1999; pp. 152–178. [Google Scholar]
- Shinde, T.S.; Brooks, J.D.; Sun-Waterhouse, D. Preparation and use of apple skin polyphenol extracts in milk: Enhancement of the viability and adhesion of probiotic Lactobacillus acidophilus (ATCC 1643) bacteria. Int. J. Food Sci. Technol. 2015, 50, 1303–1310. [Google Scholar] [CrossRef]
- Garrido, I.; Monagas, M.; Gómez-Cordovés, C.; Bartolomé, B. Polyphenols and antioxidant properties of almond skins: Influence of industrial processing. J. Food Sci. 2008, 73, C106–C115. [Google Scholar] [CrossRef] [PubMed]
- Contini, M.; Baccelloni, S.; Massantini, R.; Anelli, G. Extraction of natural antioxidants from hazelnut (Corylus avellana L.) shell and skin wastes by long maceration at room temperature. Food Chem. 2008, 110, 659–669. [Google Scholar] [CrossRef]
- Vadivel, V.; Brindha, P. Antioxidant property of solvent extract and acid/alkali hydrolysates from rice hulls. Food Biosci. 2015, 11, 85–91. [Google Scholar] [CrossRef]
- Abutheraa, R.; Hettiarachchy, N.; Kumar-Phillips, G.; Horax, R.; Chen, P.; Morawicki, R.; Kwon, Y.M. Antimicrobial activities of phenolic extracts derived from seed coats of selected soybean varieties. J. Food Sci. 2017, 82, 731–737. [Google Scholar] [CrossRef] [PubMed]
- Amarowicz, R.; Naczk, M.; Shahidi, F. Antioxidant activity of crude tannins of canola and rapeseed hulls. J. Am. Oil Chem. Soc. 2000, 77, 957–961. [Google Scholar] [CrossRef]
- Makris, D.P.; Boskou, G.; Andrikopoulos, N.K. Polyphenolic content and in vitro antioxidant characteristics of wine industry and other agri-food solid waste extracts. J. Food Compost. Anal. 2007, 20, 125–132. [Google Scholar] [CrossRef]
- Alshikh, N.; de Camargo, A.C.; Shahidi, F. Phenolics of selected lentil cultivars: Antioxidant activities and inhibition of low-density lipoprotein and DNA damage. J. Funct. Foods 2015, 18, 1022–1038. [Google Scholar] [CrossRef]
- Ma, Y.; Kosińska-Cagnazzo, A.; Kerr, W.L.; Amarowicz, R.; Swanson, R.B.; Pegg, R.B. Separation and characterization of soluble esterified and glycoside-bound phenolic compounds in dry-blanched peanut skins by liquid chromatography–electrospray ionization mass spectrometry. J. Agric. Food Chem. 2014, 62, 11488–11504. [Google Scholar] [CrossRef] [PubMed]
- Reed, J.D.; Krueger, C.G.; Vestling, M.M. MALDI-TOF mass spectrometry of oligomeric food polyphenols. Phytochemistry 2005, 66, 2248–2263. [Google Scholar] [CrossRef] [PubMed]
- Aguilar-Hernández, I.; Afseth, N.K.; López-Luke, T.; Contreras-Torres, F.F.; Wold, J.P.; Ornelas-Soto, N. Surface enhanced Raman spectroscopy of phenolic antioxidants: A systematic evaluation of ferulic acid, p-coumaric acid, caffeic acid and sinapic acid. Vib. Spectrosc. 2017, 89, 113–122. [Google Scholar] [CrossRef]
- Mignolet, A.; Mathieu, V.; Goormaghtigh, E. HTS-FTIR spectroscopy allows the classification of polyphenols according to their differential effects on the MDA-MB-231 breast cancer cell line. Analyst 2017, 142, 1244–1257. [Google Scholar] [CrossRef] [PubMed]
- Ricci, A.; Lagel, M.C.; Parpinello, G.P.; Pizzi, A.; Kilmartin, P.A.; Versari, A. Spectroscopy analysis of phenolic and sugar patterns in a food grade chestnut tannin. Food Chem. 2016, 203, 425–429. [Google Scholar] [CrossRef] [PubMed]
- Truong, V.-L.; Bak, M.-J.; Jun, M.; Kong, A.-N.T.; Ho, C.-T.; Jeong, W.-S. Antioxidant defense and hepatoprotection by procyanidins from almond (Prunus amygdalus) skins. J. Agric. Food Chem. 2014, 62, 8668–8678. [Google Scholar] [CrossRef] [PubMed]
- Madhujith, T.; Izydorczyk, M.; Shahidi, F. Antioxidant properties of pearled barley fractions. J. Agric. Food Chem. 2006, 54, 3283–3289. [Google Scholar] [CrossRef] [PubMed]
- Park, M.; Cho, H.; Jung, H.; Lee, H.; Hwang, K.T. Antioxidant and anti-inflammatory activities of tannin fraction of the extract from black raspberry seeds compared to grape seeds. J. Food Biochem. 2013, 38, 259–270. [Google Scholar] [CrossRef]
- Rahman, J.; de Camargo, A.C.; Shahidi, F. Phenolic profiles and antioxidant activity of defatted camelina and sophia seeds. Food Chem. 2017, 240, 917–925. [Google Scholar] [CrossRef] [PubMed]
- Rahman, M.J.; de Camargo, A.C.; Shahidi, F. Phenolic and polyphenolic profiles of chia seeds and their in vitro biological activities. J. Funct. Foods 2017, 35, 622–634. [Google Scholar] [CrossRef]
- Guo, J.; Tao, H.; Cao, Y.; Ho, C.-T.; Jin, S.; Huang, Q. Prevention of obesity and type 2 diabetes with aged citrus peel (chenpi) extract. J. Agric. Food Chem. 2016, 64, 2053–2061. [Google Scholar] [CrossRef] [PubMed]
- Denny, C.; Lazarini, J.G.; Franchin, M.; Melo, P.S.; Pereira, G.E.; Massarioli, A.P.; Moreno, I.A.M.; Paschoal, J.A.R.; Alencar, S.M.; Rosalen, P.L. Bioprospection of Petit Verdot grape pomace as a source of anti-inflammatory compounds. J. Funct. Foods 2014, 8, 292–300. [Google Scholar] [CrossRef]
- Lingua, M.S.; Fabani, M.P.; Wunderlin, D.A.; Baroni, M.V. In vivo antioxidant activity of grape, pomace and wine from three red varieties grown in Argentina: Its relationship to phenolic profile. J. Funct. Foods 2016, 20, 332–345. [Google Scholar] [CrossRef]
- Albishi, T.; John, J.A.; Al-Khalifa, A.S.; Shahidi, F. Antioxidative phenolic constituents of skins of onion varieties and their activities. J. Funct. Foods 2013, 5, 1191–1203. [Google Scholar] [CrossRef]
- Li, S.; Lo, C.Y.; Ho, C.T. Hydroxylated polymethoxyflavones and methylated flavonoids in sweet orange (Citrus sinensis) peel. J. Agric. Food Chem. 2006, 54, 4176–4185. [Google Scholar] [CrossRef] [PubMed]
- Tatsuno, T.; Jinno, M.; Arima, Y.; Kawabata, T.; Hasegawa, T.; Yahagi, N.; Takano, F.; Ohta, T. Anti-inflammatory and anti-melanogenic proanthocyanidin oligomers from peanut skin. Biol. Pharm. Bull. 2012, 35, 909–916. [Google Scholar] [CrossRef] [PubMed]
- Park, S.; Seok, J.K.; Kwak, J.Y.; Suh, H.-J.; Kim, Y.M.; Boo, Y.C. Anti-inflammatory effects of pomegranate peel extract in THP-1 cells exposed to particulate matter PM10. Evid.-Based Complement. Altern. Med. 2016. [Google Scholar] [CrossRef] [PubMed]
- Muliterno, M.M.; Rodrigues, D.; de Lima, F.S.; Ida, E.I.; Kurozawa, L.E. Conversion/degradation of isoflavones and color alterations during the drying of okara. LWT Food Sci. Technol. 2017, 75, 512–519. [Google Scholar] [CrossRef]
- Smith, L.L. Cytokine hypothesis of overtraining: A physiological adaptation to excessive stress? Med. Sci. Sports Exerc. 2000, 32, 317–331. [Google Scholar] [CrossRef] [PubMed]
- Toscano, L.T.; Silva, A.S.; Toscano, L.T.; Tavares, R.L.; Biasoto, A.C.T.; de Camargo, A.C.; da Silva, C.S.O.; Gonçalves, M.C.R.; Shahidi, F. Phenolics from purple grape juice increase serum antioxidant status and improve lipid profile and blood pressure in healthy adults under intense physical training. J. Funct. Foods 2017, 33, 419–424. [Google Scholar] [CrossRef]
- Leopoldini, M.; Marino, T.; Russo, N.; Toscano, M. Antioxidant properties of phenolic compounds: H-atom versus electron transfer mechanism. J. Phys. Chem. A 2004, 108, 4916–4922. [Google Scholar] [CrossRef]
- Granato, D.; Shahidi, F.; Wrolstad, R.; Kilmartin, P.; Melton, L.D.; Hidalgo, F.J.; Miyashita, K.; Camp, J.V.; Alasalvar, C.; Ismail, A.B.; et al. Antioxidant activity, total phenolics and flavonoids contents: Should we ban in vitro screening methods? Food Chem. 2018, 264, 471–475. [Google Scholar] [CrossRef] [PubMed]
- Koroleva, O.; Torkova, A.; Nikolaev, I.; Khrameeva, E.; Fedorova, T.; Tsentalovich, M.; Amarowicz, R. Evaluation of the antiradical properties of phenolic acids. Int. J. Mol. Sci. 2014, 15, 16351. [Google Scholar] [CrossRef] [PubMed]
- Prior, R.L. Oxygen radical absorbance capacity (ORAC): New horizons in relating dietary antioxidants/bioactives and health benefits. J. Funct. Foods 2015, 18, 797–810. [Google Scholar] [CrossRef]
- Shahidi, F.; Zhong, Y. Measurement of antioxidant activity. J. Funct. Foods 2015, 18, 757–781. [Google Scholar] [CrossRef]
- Rahman, M.J.; Ambigaipalan, P.; Shahidi, F. Biological activities of camelina and sophia seeds phenolics: Inhibition of LDL oxidation, DNA damage, and pancreatic lipase and α-glucosidase activities. J. Food Sci. 2018, 83, 237–245. [Google Scholar] [CrossRef] [PubMed]
- Iora, S.R.F.; Maciel, G.M.; Zielinski, A.A.F.; da Silva, M.V.; Pontes, P.V.A.; Haminiuk, C.W.I.; Granato, D. Evaluation of the bioactive compounds and the antioxidant capacity of grape pomace. Int. J. Food Sci. Technol. 2015, 50, 62–69. [Google Scholar] [CrossRef]
- Chu, H.; Tang, Q.; Huang, H.; Hao, W.; Wei, X. Grape-seed proanthocyanidins inhibit the lipopolysaccharide-induced inflammatory mediator expression in RAW264.7 macrophages by suppressing MAPK and NF-κB signal pathways. Environ. Toxicol. Pharmacol. 2016, 41, 159–166. [Google Scholar] [CrossRef] [PubMed]
- Park, J.S.; Park, M.K.; Oh, H.J.; Woo, Y.J.; Lim, M.A.; Lee, J.H.; Ju, J.H.; Jung, Y.O.; Lee, Z.H.; Park, S.H.; et al. Grape-seed proanthocyanidin extract as suppressors of bone destruction in inflammatory autoimmune arthritis. PLoS ONE 2012, 7, e51377. [Google Scholar] [CrossRef] [PubMed]
- Bao, L.; Zhang, Z.; Dai, X.; Ding, Y.; Jiang, Y.; Li, Y. Effects of grape seed proanthocyanidin extract on renal injury in type 2 diabetic rats. Mol. Med. Rep. 2015, 11, 645–652. [Google Scholar] [CrossRef] [PubMed]
- Denny, C.; Melo, P.S.; Franchin, M.; Massarioli, A.P.; Bergamaschi, K.B.; de Alencar, S.M.; Rosalen, P.L. Guava pomace: A new source of anti-inflammatory and analgesic bioactives. BMC Complement. Altern. Med. 2013, 13, 235. [Google Scholar] [CrossRef] [PubMed]
- Albishi, T.; John, J.A.; Al-Khalifa, A.S.; Shahidi, F. Antioxidant, anti-inflammatory and DNA scission inhibitory activities of phenolic compounds in selected onion and potato varieties. J. Funct. Foods 2013, 5, 930–939. [Google Scholar] [CrossRef]
- Singh, R.P.; Murthy, K.N.C.; Jayaprakasha, G.K. Studies on the antioxidant activity of pomegranate (Punica granatum) peel and seed extracts using in vitro models. J. Agric. Food Chem. 2002, 50, 81–86. [Google Scholar] [CrossRef] [PubMed]
- Braughler, J.M.; Duncan, L.A.; Chase, R.L. The involvement of iron in lipid peroxidation. Importance of ferric to ferrous ratios in initiation. J. Biol. Chem. 1986, 261, 10282–10289. [Google Scholar] [PubMed]
- Troncoso, J.C.; Costello, A.C.; Kim, J.H.; Johnson, G.V.W. Metal-catalyzed oxidation of bovine neurofilaments in vitro. Free Radic. Biol. Med. 1995, 18, 891–899. [Google Scholar] [CrossRef]
- Tournour, H.H.; Segundo, M.A.; Magalhães, L.M.; Barreiros, L.; Queiroz, J.; Cunha, L.M. Valorization of grape pomace: Extraction of bioactive phenolics with antioxidant properties. Ind. Crops Prod. 2015, 74, 397–406. [Google Scholar] [CrossRef]
- Andjelković, M.; Van Camp, J.; De Meulenaer, B.; Depaemelaere, G.; Socaciu, C.; Verloo, M.; Verhe, R. Iron-chelation properties of phenolic acids bearing catechol and galloyl groups. Food Chem. 2006, 98, 23–31. [Google Scholar] [CrossRef]
- Zhang, L.; Wang, Y.; Li, D.; Ho, C.T.; Li, J.; Wan, X. The absorption, distribution, metabolism and excretion of procyanidins. Food Funct. 2016, 7, 1273–1281. [Google Scholar] [CrossRef] [PubMed]
- Ou, K.; Gu, L. Absorption and metabolism of proanthocyanidins. J. Funct. Foods 2014, 7, 43–53. [Google Scholar] [CrossRef]
- Landete, J.M. Ellagitannins, ellagic acid and their derived metabolites: A review about source, metabolism, functions and health. Food Res. Int. 2011, 44, 1150–1160. [Google Scholar] [CrossRef]
- Batista, Â.G.; Lenquiste, S.A.; Cazarin, C.B.B.; da Silva, J.K.; Luiz-Ferreira, A.; Bogusz, S., Jr.; Wang Hantao, L.; de Souza, R.N.; Augusto, F.; Prado, M.A.; et al. Intake of jaboticaba peel attenuates oxidative stress in tissues and reduces circulating saturated lipids of rats with high-fat diet-induced obesity. J. Funct. Foods 2014, 6, 450–461. [Google Scholar] [CrossRef]
- Cazarin, C.B.; da Silva, J.K.; Colomeu, T.C.; Batista, A.G.; Vilella, C.A.; Ferreira, A.L.; Junior, S.B.; Fukuda, K.; Augusto, F.; de Meirelles, L.R.; et al. Passiflora edulis peel intake and ulcerative colitis: Approaches for prevention and treatment. Exp. Biol. Med. 2014, 239, 542–551. [Google Scholar] [CrossRef] [PubMed]
- Lenquiste, S.A.; Marineli, R.S.; Moraes, É.A.; Dionísio, A.P.; Brito, E.S.; Maróstica Junior, M.R. Jaboticaba peel and jaboticaba peel aqueous extract shows in vitro and in vivo antioxidant properties in obesity model. Food Res. Int. 2015, 77, 162–170. [Google Scholar] [CrossRef]
- Cazarin, C.B.B.; da Silva, J.K.; Colomeu, T.C.; Batista, Â.G.; Meletti, L.M.M.; Paschoal, J.A.R.; Bogusz Junior, S.; Braga, P.A.C.; Reyes, F.G.R.; Augusto, F.; et al. Intake of Passiflora edulis leaf extract improves antioxidant and anti-inflammatory status in rats with 2,4,6-trinitrobenzenesulphonic acid induced colitis. J. Funct. Foods 2015, 17, 575–586. [Google Scholar] [CrossRef]
- Liu, H.Y.; Tayyari, F.; Edison, A.S.; Su, Z.H.; Gu, L.W. NMR-based metabolomics reveals urinary metabolome modifications in female Sprague-Dawley rats by cranberry procyanidins. J. Nutr. Biochem. 2016, 34, 136–145. [Google Scholar] [CrossRef] [PubMed]
- Sano, A.; Yamakoshi, J.; Tokutake, S.; Tobe, K.; Kubota, Y.; Kikuchi, M. Procyanidin B1 is detected in human serum after intake of proanthocyanidin-rich grape seed extract. Biosci. Biotechnol. Biochem. 2003, 67, 1140–1143. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Sarrias, A.; Gimenez-Bastida, J.A.; Garcia-Conesa, M.T.; Gomez-Sanchez, M.B.; Garcia-Talavera, N.V.; Gil-Izquierdo, A.; Sanchez-Alvarez, C.; Fontana-Compiano, L.O.; Morga-Egea, J.P.; Pastor-Quirante, F.A.; et al. Occurrence of urolithins, gut microbiota ellagic acid metabolites and proliferation markers expression response in the human prostate gland upon consumption of walnuts and pomegranate juice. Mol. Nutr. Food Res. 2010, 54, 311–322. [Google Scholar] [CrossRef] [PubMed]
- Manach, C.; Williamson, G.; Morand, C.; Scalbert, A.; Remesy, C. Bioavailability and bioefficacy of polyphenols in humans. I. Review of 97 bioavailability studies. Am. J. Clin. Nutr. 2005, 81, 230S–242S. [Google Scholar] [CrossRef] [PubMed]
- Deprez, S.; Mila, I.; Huneau, J.F.; Tome, D.; Scalbert, A. Transport of proanthocyanidin dimer, trimer, and polymer across monolayers of human intestinal epithelial Caco-2 cells. Antioxid. Redox Signal. 2001, 3, 957–967. [Google Scholar] [CrossRef] [PubMed]
- Ou, K.Q.; Percival, S.S.; Zou, T.; Khoo, C.; Gu, L.W. Transport of cranberry A-type procyanidin dimers, trimers, and tetramers across monolayers of human intestinal epithelial Caco-2 cells. J. Agric. Food Chem. 2012, 60, 1390–1396. [Google Scholar] [CrossRef] [PubMed]
- Tomás-Barberán, F.A.; García-Villalba, R.; González-Sarrías, A.; Selma, M.V.; Espín, J.C. Ellagic acid metabolism by human gut microbiota: Consistent observation of three urolithin phenotypes in intervention trials, independent of food source, age, and health status. J. Agric. Food Chem. 2014, 62, 6535–6538. [Google Scholar] [CrossRef] [PubMed]
- Dall’Asta, M.; Calani, L.; Tedeschi, M.; Jechiu, L.; Brighenti, F.; Del Rio, D. Identification of microbial metabolites derived from in vitro fecal fermentation of different polyphenolic food sources. Nutrition 2012, 28, 197–203. [Google Scholar] [CrossRef] [PubMed]
- Pekkanen, J.; Linn, S.; Heiss, G.; Suchindran, C.M.; Leon, A.; Rifkind, B.M.; Tyroler, H.A. Ten-year mortality from cardiovascular disease in relation to cholesterol level among men with and without preexisting cardiovascular disease. N. Engl. J. Med. 1990, 322, 1700–1707. [Google Scholar] [CrossRef] [PubMed]
- Martín-Carrón, N.; Goñi, I.; Larrauri, J.A.; García-Alonso, A.; Saura-Calixto, F. Reduction in serum total and LDL cholesterol concentrations by a dietary fiber and polyphenol-rich grape product in hypercholesterolemic rats. Nutr. Res. 1999, 19, 1371–1381. [Google Scholar] [CrossRef]
- Aviram, M.; Volkova, N.; Coleman, R.; Dreher, M.; Reddy, M.K.; Ferreira, D.; Rosenblat, M. Pomegranate phenolics from the peels, arils, and flowers are antiatherogenic: Studies in vivo in atherosclerotic apolipoprotein e-deficient (E0) mice and in vitro in cultured macrophages and lipoproteins. J. Agric. Food Chem. 2008, 56, 1148–1157. [Google Scholar] [CrossRef] [PubMed]
- Chung, K.T.; Wong, T.Y.; Wei, C.I.; Huang, Y.W.; Lin, Y. Tannins and human health: A review. Crit. Rev. Food Sci. Nutr. 1998, 38, 421–464. [Google Scholar] [CrossRef] [PubMed]
- Amarowicz, R. Natural phenolic compounds protect LDL against oxidation. Eur. J. Lipid Sci. Technol. 2016, 118, 677–679. [Google Scholar] [CrossRef]
- Salah, N.; Miller, N.J.; Paganga, G.; Tijburg, L.; Bolwell, G.P.; Riceevans, C. Polyphenolic flavanols as scavengers of aqueous phase radicals and as chain-breaking antioxidants. Arch. Biochem. Biophys. 1995, 322, 339–346. [Google Scholar] [CrossRef] [PubMed]
- Zhong, Y.; Shahidi, F. Lipophilised epigallocatechin gallate (EGCG) derivatives and their antioxidant potential in food and biological systems. Food Chem. 2012, 131, 22–30. [Google Scholar] [CrossRef]
- Perera, N.; Ambigaipalan, P.; Shahidi, F. Epigallocatechin gallate (EGCG) esters with different chain lengths fatty acids and their antioxidant activity in food and biologicalsystems. J. Food Bioact. 2018, 1, 124–133. [Google Scholar]
- Pan, M.H.; Lai, C.S.; Wu, J.C.; Ho, C.T. Molecular mechanisms for chemoprevention of colorectal cancer by natural dietary compounds. Mol. Nutr. Food Res. 2011, 55, 32–45. [Google Scholar] [CrossRef] [PubMed]
- Pan, M.H.; Ho, C.T. Chemopreventive effects of natural dietary compounds on cancer development. Chem. Soc. Rev. 2008, 37, 2558–2574. [Google Scholar] [CrossRef] [PubMed]
- Khanna, K.K.; Jackson, S.P. DNA double-strand breaks: Signaling, repair and the cancer connection. Nat. Genet. 2001, 27, 247–254. [Google Scholar] [CrossRef] [PubMed]
- Abid-Essefi, S.; Baudrimont, I.; Hassen, W.; Ouanes, Z.; Mobio, T.A.; Anane, R.; Creppy, E.E.; Bacha, H. DNA fragmentation, apoptosis and cell cycle arrest induced by zearalenone in cultured DOK, Vero and Caco-2 cells: Prevention by Vitamin E. Toxicology 2003, 192, 237–248. [Google Scholar] [CrossRef]
- Miranda, D.D.C.; Arçari, D.P.; Ladeira, M.S.P.; Calori-Domingues, M.A.; Romero, A.C.; Salvadori, D.M.F.; Gloria, E.M.; Pedrazzoli, J., Jr.; Ribeiro, M.L. Analysis of DNA damage induced by aflatoxin B1 in Dunkin–Hartley guinea pigs. Mycopathologia 2007, 163, 275–280. [Google Scholar] [CrossRef] [PubMed]
- Calori-Domingues, M.A.; Bernardi, C.M.G.; Nardin, M.S.; de Souza, G.V.; dos Santos, F.G.R.; Stein, M.A.; da Gloria, E.M.; Dias, C.T.S.; de Camargo, A.C. Co-occurrence and distribution of deoxynivalenol, nivalenol and zearalenone in wheat from Brazil. Food Addit. Contam. Part B Surveill. 2016, 9, 142–151. [Google Scholar] [CrossRef] [PubMed]
- Santili, A.B.N.; de Camargo, A.C.; Nunes, R.S.R.; da Gloria, E.M.; Machado, P.F.; Cassoli, L.D.; Dias, C.T.S.; Calori-Domingues, M.A. Aflatoxin M1 in raw milk from different regions of São Paulo state–Brazil. Food Addit. Contam. Part B Surveill. 2015, 8, 207–214. [Google Scholar] [CrossRef] [PubMed]
- Calori-Domingues, M.A.; Iwahashi, P.M.R.; Ponce, G.H.; da Gloria, E.M.; Dias, C.T.S.; Button, D.C.; de Camargo, A.C. Aflatoxin B1 and zearalenone in soybeans: Occurrence and distribution in whole and defective kernels. Food Addit. Contam. Part B Surveill. 2018. [Google Scholar] [CrossRef] [PubMed]
- Long, M.; Yang, S.-H.; Han, J.-X.; Li, P.; Zhang, Y.; Dong, S.; Chen, X.; Guo, J.; Wang, J.; He, J.-B. The protective effect of grape-seed proanthocyanidin extract on oxidative damage induced by zearalenone in Kunming mice liver. Int. J. Mol. Sci. 2016, 17, 808. [Google Scholar] [CrossRef] [PubMed]
- Long, M.; Yang, S.; Zhang, Y.; Li, P.; Han, J.; Dong, S.; Chen, X.; He, J. Proanthocyanidin protects against acute zearalenone-induced testicular oxidative damage in male mice. Environ. Sci. Pollut. Res. Int. 2017, 24, 938–946. [Google Scholar] [CrossRef] [PubMed]
- Ben Salem, I.; Prola, A.; Boussabbeh, M.; Guilbert, A.; Bacha, H.; Lemaire, C.; Abid-Essefi, S. Activation of ER stress and apoptosis by α- and β-zearalenol in HCT116 cells, protective role of quercetin. Neurotoxicology 2016, 53, 334–342. [Google Scholar] [CrossRef] [PubMed]
- Ben Salem, I.; Prola, A.; Boussabbeh, M.; Guilbert, A.; Bacha, H.; Abid-Essefi, S.; Lemaire, C. Crocin and quercetin protect HCT116 and HEK293 cells from Zearalenone-induced apoptosis by reducing endoplasmic reticulum stress. Cell Stress Chaperones 2015, 20, 927–938. [Google Scholar] [CrossRef] [PubMed]
- Engelbrecht, A.M.; Mattheyse, M.; Ellis, B.; Loos, B.; Thomas, M.; Smith, R.; Peters, S.; Smith, C.; Myburgh, K. Proanthocyanidin from grape seeds inactivates the PI3-kinase/PKB pathway and induces apoptosis in a colon cancer cell line. Cancer Lett. 2007, 258, 144–153. [Google Scholar] [CrossRef] [PubMed]
- Lai, C.S.; Li, S.; Miyauchi, Y.; Suzawa, M.; Ho, C.T.; Pan, M.H. Potent anti-cancer effects of citrus peel flavonoids in human prostate xenograft tumors. Food Funct. 2013, 4, 944–949. [Google Scholar] [CrossRef] [PubMed]
- Cheung-Ong, K.; Giaever, G.; Nislow, C. DNA-damaging agents in cancer chemotherapy: Serendipity and chemical biology. Chem. Biol. 2013, 20, 648–659. [Google Scholar] [CrossRef] [PubMed]
- Simunek, T.; Sterba, M.; Popelova, O.; Adamcova, M.; Hrdina, R.; Gersl, V. Anthracycline-induced cardiotoxicity: Overview of studies examining the roles of oxidative stress and free cellular iron. Pharmacol. Rep. 2009, 61, 154–171. [Google Scholar] [CrossRef]
- Kalender, Y.; Yel, M.; Kalender, S. Doxorubicin hepatotoxicity and hepatic free radical metabolism in rats: The effects of vitamin E and catechin. Toxicology 2005, 209, 39–45. [Google Scholar] [CrossRef] [PubMed]
- Singh, N.P.; Danner, D.B.; Tice, R.R.; Brant, L.; Schneider, E.L. DNA damage and repair with age in individual human lymphocytes. Mutat. Res. 1990, 237, 123–130. [Google Scholar] [CrossRef]
- Mayer, P.J.; Lange, C.S.; Bradley, M.O.; Nichols, W.W. Age-dependent decline in rejoining of X-ray-induced DNA double-strand breaks in normal human lymphocytes. Mutat. Res. 1989, 219, 95–100. [Google Scholar] [CrossRef]
- Aprotosoaie, A.C.; Trifan, A.; Gille, E.; Petreus, T.; Bordeianu, G.; Miron, A. Can phytochemicals be a bridge to develop new radioprotective agents? Phytochem. Rev. 2014, 14, 555–566. [Google Scholar] [CrossRef]
- Zhang, Y.; Chen, S.; Wei, C.; Chen, J.; Ye, X. Proanthocyanidins from Chinese bayberry (Myrica rubra Sieb. et Zucc.) leaves regulate lipid metabolism and glucose consumption by activating AMPK pathway in HepG2 cells. J. Funct. Foods 2017, 29, 217–225. [Google Scholar] [CrossRef]
- Kumar, S.; Narwal, S.; Kumar, V.; Prakash, O. α-Glucosidase inhibitors from plants: A natural approach to treat diabetes. Pharmacogn. Rev. 2011, 5, 19–29. [Google Scholar] [CrossRef] [PubMed]
- Yoshikawa, M.; Nishida, N.; Shimoda, H.; Takada, M.; Kawahara, Y.; Matsuda, H. Polyphenol constituents from Salacia species: Quantitative analysis of mangiferin with α-glucosidase and aldose reductase inhibitory activities. Yakugaku Zasshi 2001, 121, 371–378. [Google Scholar] [CrossRef] [PubMed]
- Bustanji, Y.; Issa, A.; Mohammad, M.; Hudaib, M.; Tawah, K.; Alkhatib, H.; Almasri, I.; Al-Khalidi, B. Inhibition of hormone sensitive lipase and pancreatic lipase by Rosmarinus officinalis extract and selected phenolic constituents. J. Med. Plant Res. 2010, 4, 2235–2242. [Google Scholar]
- Kalyanaraman, B.; Premovic, P.I.; Sealy, R.C. Semiquinone anion radicals from addition of amino acids, peptides, and proteins to quinones derived from oxidation of catechols and catecholamines. An ESR spin stabilization study. J. Biol. Chem. 1987, 262, 11080–11087. [Google Scholar] [PubMed]
- Naczk, M.; Towsend, M.; Zadernowski, R.; Shahidi, F. Protein-binding and antioxidant potential of phenolics of mangosteen fruit (Garcinia mangostana). Food Chem. 2011, 128, 292–298. [Google Scholar] [CrossRef] [PubMed]
- De Freitas, V.; Mateus, N. Structural features of procyanidin interactions with salivary proteins. J. Agric. Food Chem. 2001, 49, 940–945. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.A.; Cho, E.J.; Tanaka, T.; Yokozawa, T. Inhibitory activities of proanthocyanidins from persimmon against oxidative stress and digestive enzymes related to diabetes. J. Nutr. Sci. Vitaminol. 2007, 53, 287–292. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.-J.; Lee, S.-B.; Lee, H.-S.; Lee, S.-Y.; Baek, J.-S.; Kim, D.; Moon, T.-W.; Robyt, J.F.; Park, K.-H. Comparative study of the inhibition of α-glucosidase, α-amylase, and cyclomaltodextrin glucanosyltransferase by acarbose, isoacarbose, and acarviosine–glucose. Arch. Biochem. Biophys. 1999, 371, 277–283. [Google Scholar] [CrossRef] [PubMed]
- Kadouh, H.C.; Sun, S.; Zhu, W.; Zhou, K. α-Glucosidase inhibiting activity and bioactive compounds of six red wine grape pomace extracts. J. Funct. Foods 2016, 26, 577–584. [Google Scholar] [CrossRef] [PubMed]
- Ma, C.; Hu, L.; Kou, X.; Lv, W.; Lou, Z.; Wang, H. Rapid screening of potential α-amylase inhibitors from Rhodiola rosea by UPLC-DAD-TOF-MS/MS-based metabolomic method. J. Funct. Foods 2017, 36, 144–149. [Google Scholar] [CrossRef]
- Masumoto, S.; Terao, A.; Yamamoto, Y.; Mukai, T.; Miura, T.; Shoji, T. Non-absorbable apple procyanidins prevent obesity associated with gut microbial and metabolomic changes. Sci. Rep. 2016, 6, 31208. [Google Scholar] [CrossRef] [PubMed]
- Adisakwattana, S.; Charoenlertkul, P.; Yibchok-Anun, S. α-Glucosidase inhibitory activity of cyanidin-3-galactoside and synergistic effect with acarbose. J. Enzyme Inhib. Med. Chem. 2009, 24, 65–69. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Chen, S.; Ye, X. The anti-obesity properties of the proanthocyanidin extract from the leaves of Chinese bayberry (Myrica rubra Sieb.et Zucc.). Food Funct. 2017, 8, 3259–3270. [Google Scholar] [CrossRef] [PubMed]
- Vazquez-Flores, A.A.; Wong-Paz, J.E.; Lerma-Herrera, M.A.; Martinez-Gonzalez, A.I.; Olivas-Aguirre, F.J.; Aguilar, C.N.; Wall-Medrano, A.; Gonzalez-Aguilar, G.A.; Alvarez-Parrilla, E.; de la Rosa, L.A. Proanthocyanidins from the kernel and shell of pecan (Carya illinoinensis): Average degree of polymerization and effects on carbohydrate, lipid, and peptide hydrolysis in a simulated human digestive system. J. Funct. Foods 2017, 28, 227–234. [Google Scholar] [CrossRef]
- García-Conesa, M.-T. Dietary polyphenols against metabolic disorders: How far have we progressed in the understanding of the molecular mechanisms of action of these compounds? Crit. Rev. Food Sci. Nutr. 2017, 57, 1769–1786. [Google Scholar] [CrossRef] [PubMed]
- Batista, G.A.; Edilene, S.S.; Mendonça, M.C.P.; da Silva, J.K.; Dionísio, P.A.; Sartori, C.R.; da Cruz-Höfling, M.A.; Maróstica Júnior, M.R. Jaboticaba berry peel intake prevents insulin-resistance-induced tau phosphorylation in mice. Mol. Nutr. Food Res. 2017, 61, 1600952. [Google Scholar] [CrossRef] [PubMed]
- Plaza, M.; Batista, Â.G.; Cazarin, C.B.B.; Sandahl, M.; Turner, C.; Östman, E.; Maróstica Júnior, M.R. Characterization of antioxidant polyphenols from Myrciaria jaboticaba peel and their effects on glucose metabolism and antioxidant status: A pilot clinical study. Food Chem. 2016, 211, 185–197. [Google Scholar] [CrossRef] [PubMed]
- Marseglia, L.; Manti, S.; D’Angelo, G.; Nicotera, A.; Parisi, E.; Di Rosa, G.; Gitto, E.; Arrigo, T. Oxidative stress in obesity: A critical component in human diseases. Int. J. Mol. Sci. 2015, 16, 378–400. [Google Scholar] [CrossRef] [PubMed]
- Savini, I.; Catani, V.M.; Evangelista, D.; Gasperi, V.; Avigliano, L. Obesity-associated oxidative stress: Strategies finalized to improve redox state. Int. J. Mol. Sci. 2013, 14, 10497–10538. [Google Scholar] [CrossRef] [PubMed]
- Ma, Q. Role of Nrf2 in oxidative stress and toxicity. Ann. Rev. Pharmacol. Toxicol. 2013, 53, 401–426. [Google Scholar] [CrossRef] [PubMed]
- Ozdal, T.; Sela, D.A.; Xiao, J.; Boyacioglu, D.; Chen, F.; Capanoglu, E. The reciprocal interactions between polyphenols and gut microbiota and effects on bioaccessibility. Nutrients 2016, 8, 78. [Google Scholar] [CrossRef] [PubMed]
- Etxeberria, U.; Fernández-Quintela, A.; Milagro, F.I.; Aguirre, L.; Martínez, J.A.; Portillo, M.P. Impact of polyphenols and polyphenol-rich dietary sources on gut microbiota composition. J. Agric. Food Chem. 2013, 61, 9517–9533. [Google Scholar] [CrossRef] [PubMed]
- Danneskiold-Samsøe, N.B.; Barros, H.D.F.Q.; Santos, R.; Bicas, J.L.; Cazarin, C.B.B.; Madsen, L.; Kristiansen, K.; Pastore, G.M.; Brix, S.; Maróstica Júnior, M.R. Interplay between food and gut microbiota in health and disease. Food Res. Int. 2018. [Google Scholar] [CrossRef]
- Annuzzi, G.; Bozzetto, L.; Costabile, G.; Giacco, R.; Mangione, A.; Anniballi, G.; Vitale, M.; Vetrani, C.; Cipriano, P.; Corte, G.D.; et al. Diets naturally rich in polyphenols improve fasting and postprandial dyslipidemia and reduce oxidative stress: A randomized controlled trial. Am. J. Clin. Nutr. 2014, 99, 463–471. [Google Scholar] [CrossRef] [PubMed]
- Dragano, N.R.V.; Marques, A.Y.C.; Cintra, D.E.C.; Solon, C.; Morari, J.; Leite-Legatti, A.V.; Velloso, L.A.; Marostica, M.R. Freeze-dried jaboticaba peel powder improves insulin sensitivity in high-fat-fed mice. Br. J. Nutr. 2013, 110, 447–455. [Google Scholar] [CrossRef] [PubMed]
- Grivennikov, S.I.; Greten, F.R.; Karin, M. Immunity, inflammation, and cancer. Cell 2010, 140, 883–899. [Google Scholar] [CrossRef] [PubMed]
- Sadik, C.D.; Luster, A.D. Lipid-cytokine-chemokine cascades orchestrate leukocyte recruitment in inflammation. J. Leukoc. Biol. 2012, 91, 207–215. [Google Scholar] [CrossRef] [PubMed]
- Choy, E.H.; Panayi, G.S. Cytokine pathways and joint inflammation in rheumatoid arthritis. N. Engl. J. Med. 2001, 344, 907–916. [Google Scholar] [CrossRef] [PubMed]
- Donath, M.Y.; Shoelson, S.E. Type 2 diabetes as an inflammatory disease. Nat. Rev. Immunol. 2011, 11, 98–107. [Google Scholar] [CrossRef] [PubMed]
- Del Rio, D.; Rodriguez-Mateos, A.; Spencer, J.P.; Tognolini, M.; Borges, G.; Crozier, A. Dietary (poly)phenolics in human health: Structures, bioavailability, and evidence of protective effects against chronic diseases. Antioxid. Redox Signal. 2013, 18, 1818–1892. [Google Scholar] [CrossRef] [PubMed]
- Pan, M.H.; Lai, C.S.; Dushenkov, S.; Ho, C.T. Modulation of inflammatory genes by natural dietary bioactive compounds. J. Agric. Food Chem. 2009, 57, 4467–4477. [Google Scholar] [CrossRef] [PubMed]
- Pan, M.H.; Lai, C.S.; Ho, C.T. Anti-inflammatory activity of natural dietary flavonoids. Food Funct. 2010, 1, 15–31. [Google Scholar] [CrossRef] [PubMed]
- Gosslau, A.; Li, S.; Ho, C.T.; Chen, K.Y.; Rawson, N.E. The importance of natural product characterization in studies of their anti-inflammatory activity. Mol. Nutr. Food Res. 2011, 55, 74–82. [Google Scholar] [CrossRef] [PubMed]
- Zhong, Y.; Chiou, Y.-S.; Pan, M.-H.; Shahidi, F. Anti-inflammatory activity of lipophilic epigallocatechin gallate (EGCG) derivatives in LPS-stimulated murine macrophages. Food Chem. 2012, 134, 742–748. [Google Scholar] [CrossRef] [PubMed]
- Terra, X.; Valls, J.; Vitrac, X.; Merrillon, J.M.; Arola, L.; Ardevol, A.; Blade, C.; Fernandez-Larrea, J.; Pujadas, G.; Salvado, J.; et al. Grape-seed procyanidins act as antiinflammatory agents in endotoxin-stimulated RAW 264.7 macrophages by inhibiting NFkB signaling pathway. J. Agric. Food Chem. 2007, 55, 4357–4365. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.X.; Bai, M.; Zhou, L.; Lou, L.L.; Liu, Q.B.; Zhang, Y.; Li, L.Z.; Song, S.J. Food byproducts as a new and cheap source of bioactive compounds: Lignans with antioxidant and anti-inflammatory properties from Crataegus pinnatifida seeds. J. Agric. Food. Chem. 2015, 63, 7252–7260. [Google Scholar] [CrossRef] [PubMed]
- Nieto, J.A.; Jaime, L.; Arranz, E.; Reglero, G.; Santoyo, S. Winemaking by-products as anti-inflammatory food ingredients. Food Agric. Immunol. 2017, 28, 1507–1518. [Google Scholar] [CrossRef]
- Bachoual, R.; Talmoudi, W.; Boussetta, T.; Braut, F.; El-Benna, J. An aqueous pomegranate peel extract inhibits neutrophil myeloperoxidase in vitro and attenuates lung inflammation in mice. Food Chem. Toxicol. 2011, 49, 1224–1228. [Google Scholar] [CrossRef] [PubMed]
- Ismail, T.; Sestili, P.; Akhtar, S. Pomegranate peel and fruit extracts: A review of potential anti-inflammatory and anti-infective effects. J. Ethnopharmacol. 2012, 143, 397–405. [Google Scholar] [CrossRef] [PubMed]
- BenSaad, L.A.; Kim, K.H.; Quah, C.C.; Kim, W.R.; Shahimi, M. Anti-inflammatory potential of ellagic acid, gallic acid and punicalagin A&B isolated from Punica granatum. BMC Complement. Altern. Med. 2017, 17, 47. [Google Scholar]
- Xu, X.; Yin, P.; Wan, C.; Chong, X.; Liu, M.; Cheng, P.; Chen, J.; Liu, F.; Xu, J. Punicalagin inhibits inflammation in LPS-induced RAW264.7 macrophages via the suppression of TLR4-mediated MAPKs and NF-κB activation. Inflammation 2014, 37, 956–965. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.E.; Hwang, C.J.; Lee, H.P.; Kim, C.S.; Son, D.J.; Ham, Y.W.; Hellstrom, M.; Han, S.B.; Kim, H.S.; Park, E.K.; et al. Inhibitory effect of punicalagin on lipopolysaccharide-induced neuroinflammation, oxidative stress and memory impairment via inhibition of nuclear factor-κB. Neuropharmacology 2017, 117, 21–32. [Google Scholar] [CrossRef] [PubMed]
- Usta, C.; Ozdemir, S.; Schiariti, M.; Puddu, P.E. The pharmacological use of ellagic acid-rich pomegranate fruit. Int. J. Food Sci. Nutr. 2013, 64, 907–913. [Google Scholar] [CrossRef] [PubMed]
- Favarin, D.C.; Teixeira, M.M.; de Andrade, E.L.; Alves, C.F.; Chica, J.E.L.; Sorgi, C.A.; Faccioli, L.H.; Rogerio, A.P. Anti-inflammatory effects of ellagic acid on acute lung injury induced by acid in mice. Mediat. Inflamm. 2013, 2013, 164202. [Google Scholar]
- Marin, M.; Maria Giner, R.; Rios, J.L.; Recio, M.C. Intestinal anti-inflammatory activity of ellagic acid in the acute and chronic dextrane sulfate sodium models of mice colitis. J. Ethnopharmacol. 2013, 150, 925–934. [Google Scholar] [CrossRef] [PubMed]
- El-Shitany, N.A.; El-Bastawissy, E.A.; El-desoky, K. Ellagic acid protects against carrageenan-induced acute inflammation through inhibition of nuclear factor κB, inducible cyclooxygenase and proinflammatory cytokines and enhancement of interleukin-10 via an antioxidant mechanism. Int. Immunopharmacol. 2014, 19, 290–299. [Google Scholar] [CrossRef] [PubMed]
- Allam, G.; Mahdi, E.A.; Alzahrani, A.M.; Abuelsaad, A.S. Ellagic acid alleviates adjuvant induced arthritis by modulation of pro- and anti-inflammatory cytokines. Cent. Eur. J. Immunol. 2016, 41, 339–349. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.M.; Wang, Z.H.; Liu, C.H.; Chen, C.S. Ellagic acid inhibits IL-1beta-induced cell adhesion molecule expression in human umbilical vein endothelial cells. Br. J. Nutr. 2007, 97, 692–698. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Lo, C.-Y.; Dushenkov, S.; Ho, C.-T. Polymethoxyflavones: Chemistry, biological activity, and occurrence in orange peel. In Dietary Supplements; American Chemical Society: Washington, DC, USA, 2008; Volume 987, pp. 191–210. [Google Scholar]
- Xie, C.; Kang, J.; Ferguson, M.E.; Nagarajan, S.; Badger, T.M.; Wu, X. Blueberries reduce pro-inflammatory cytokine TNF-α and IL-6 production in mouse macrophages by inhibiting NF-κB activation and the MAPK pathway. Mol. Nutr. Food Res. 2011, 55, 1587–1591. [Google Scholar] [CrossRef] [PubMed]
- Joseph, S.V.; Edirisinghe, I.; Burton-Freeman, B.M. Berries: Anti-inflammatory effects in humans. J. Agric. Food Chem. 2014, 62, 3886–3903. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Wang, L.; Wu, Z.; Yao, L.; Wu, Y.; Huang, L.; Liu, K.; Zhou, X.; Gou, D. Anthocyanin-rich fractions from red raspberries attenuate inflammation in both RAW264.7 macrophages and a mouse model of colitis. Sci. Rep. 2014, 4, 6234. [Google Scholar] [CrossRef] [PubMed]

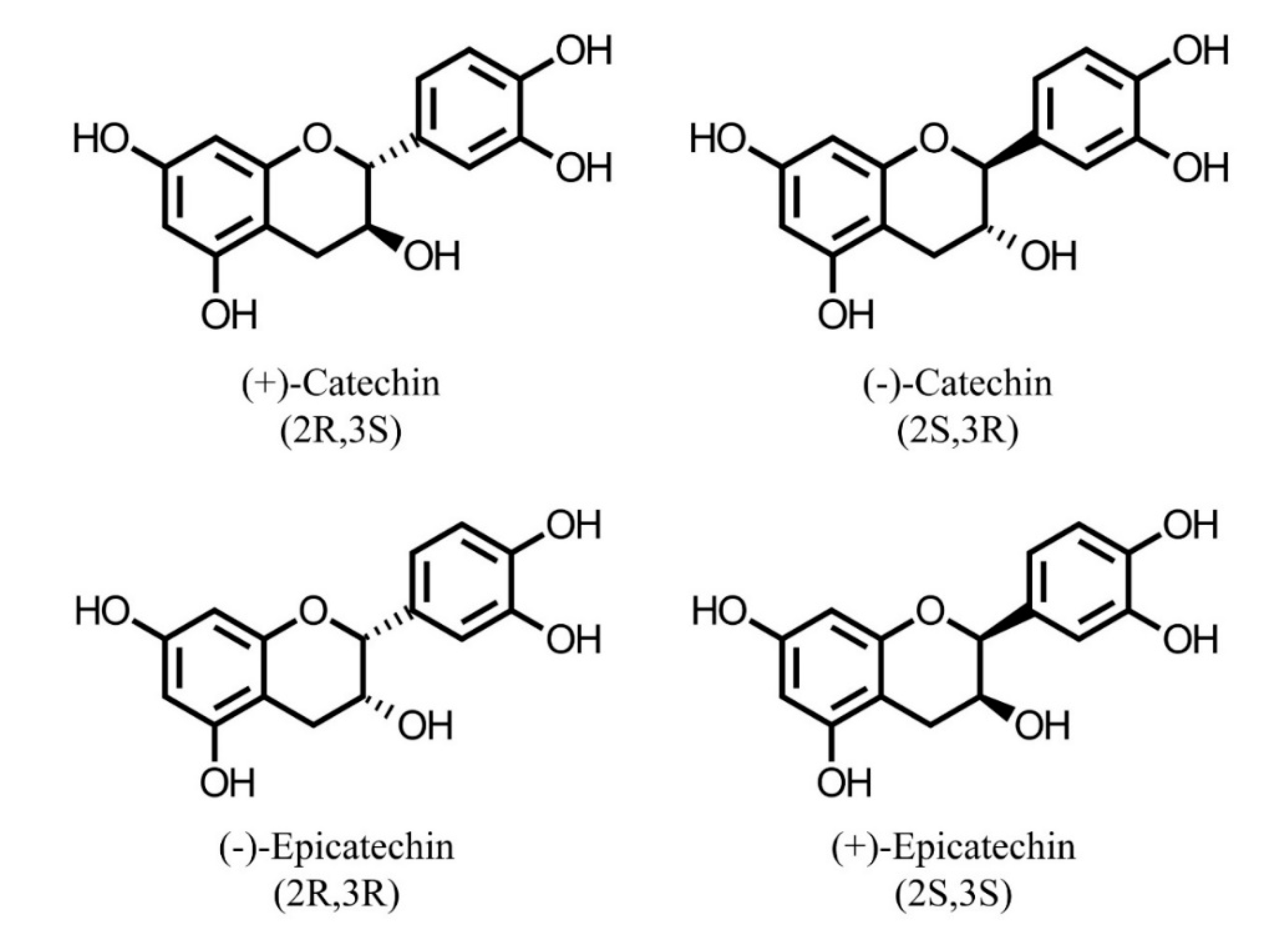
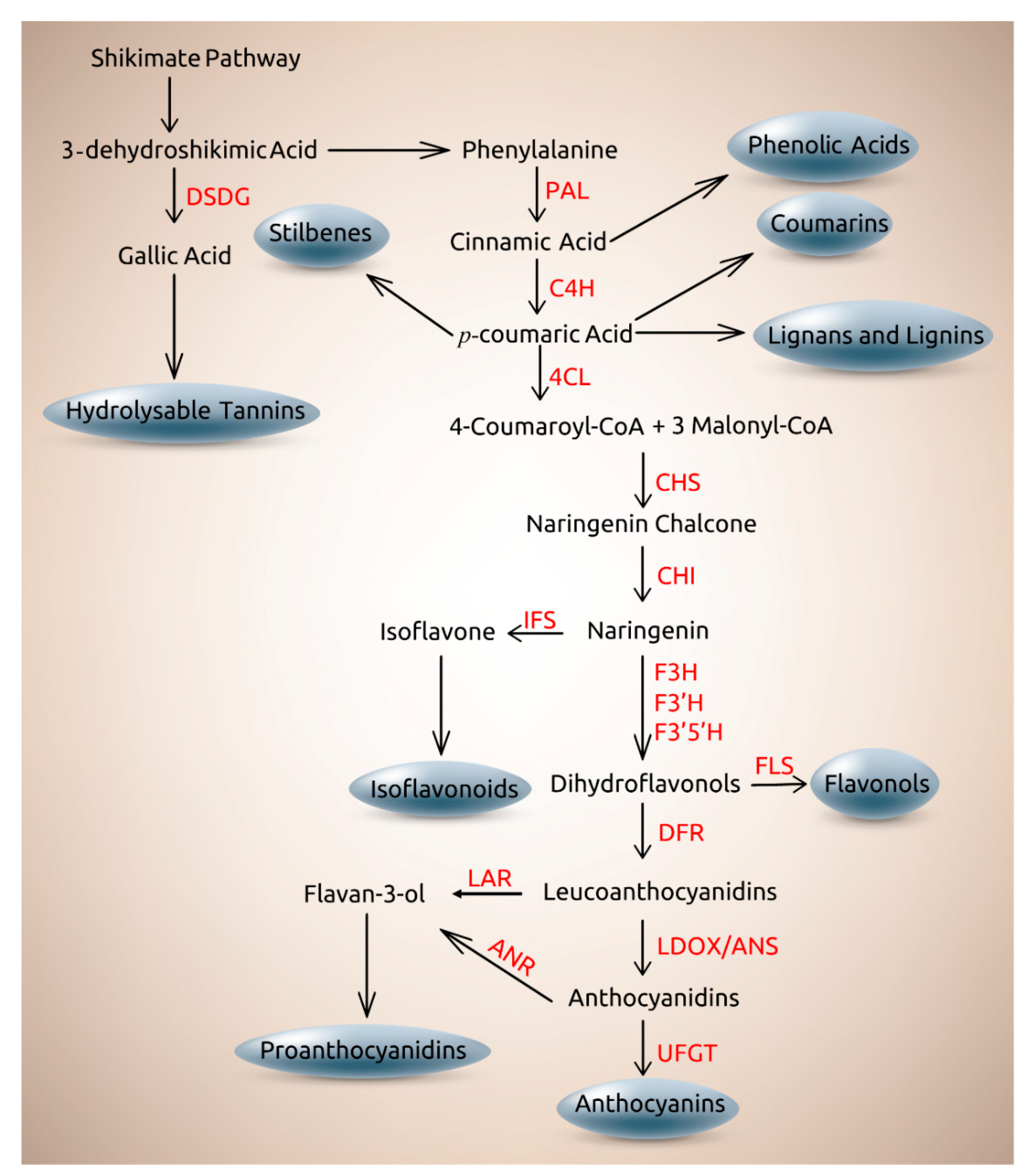
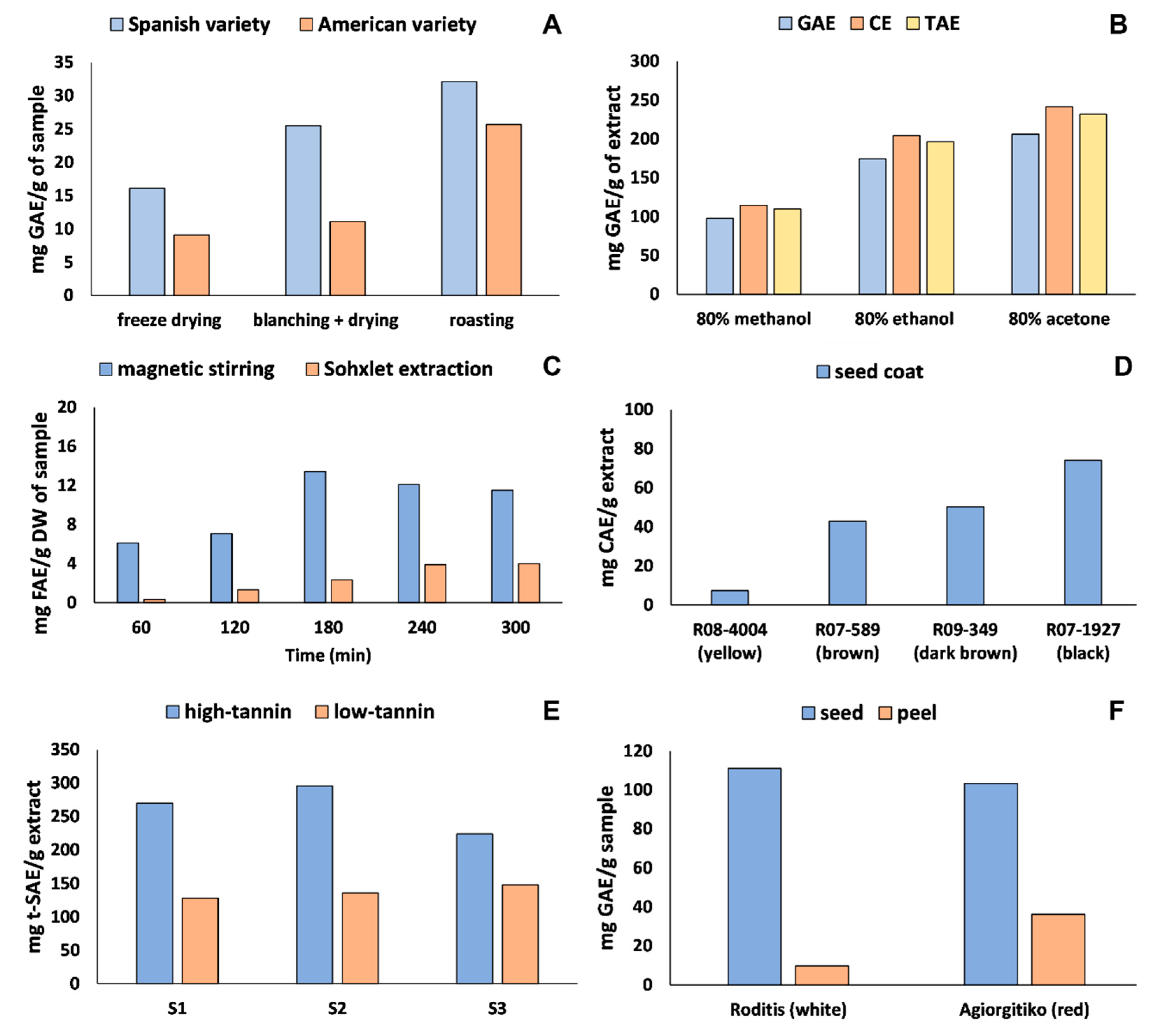
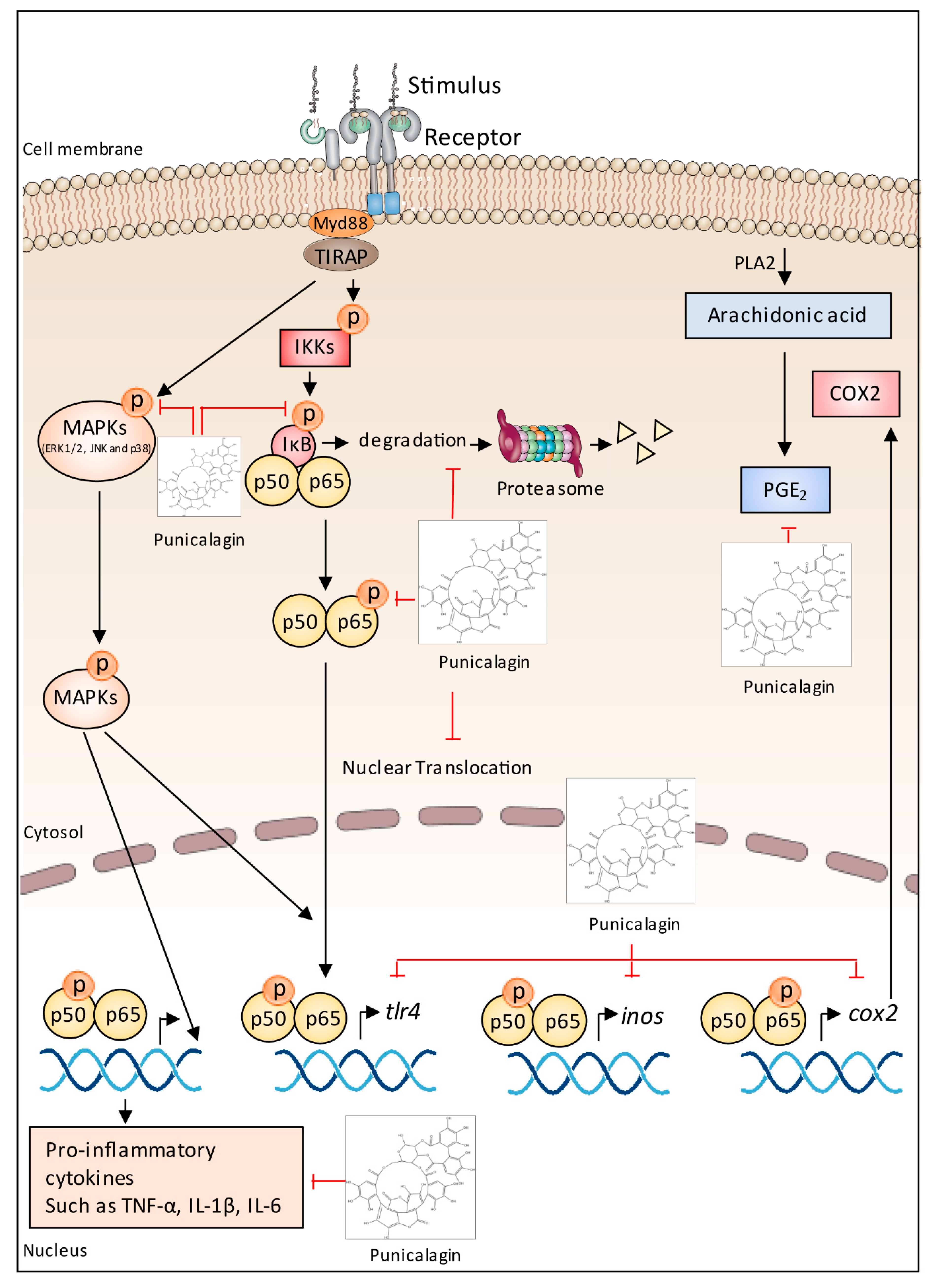
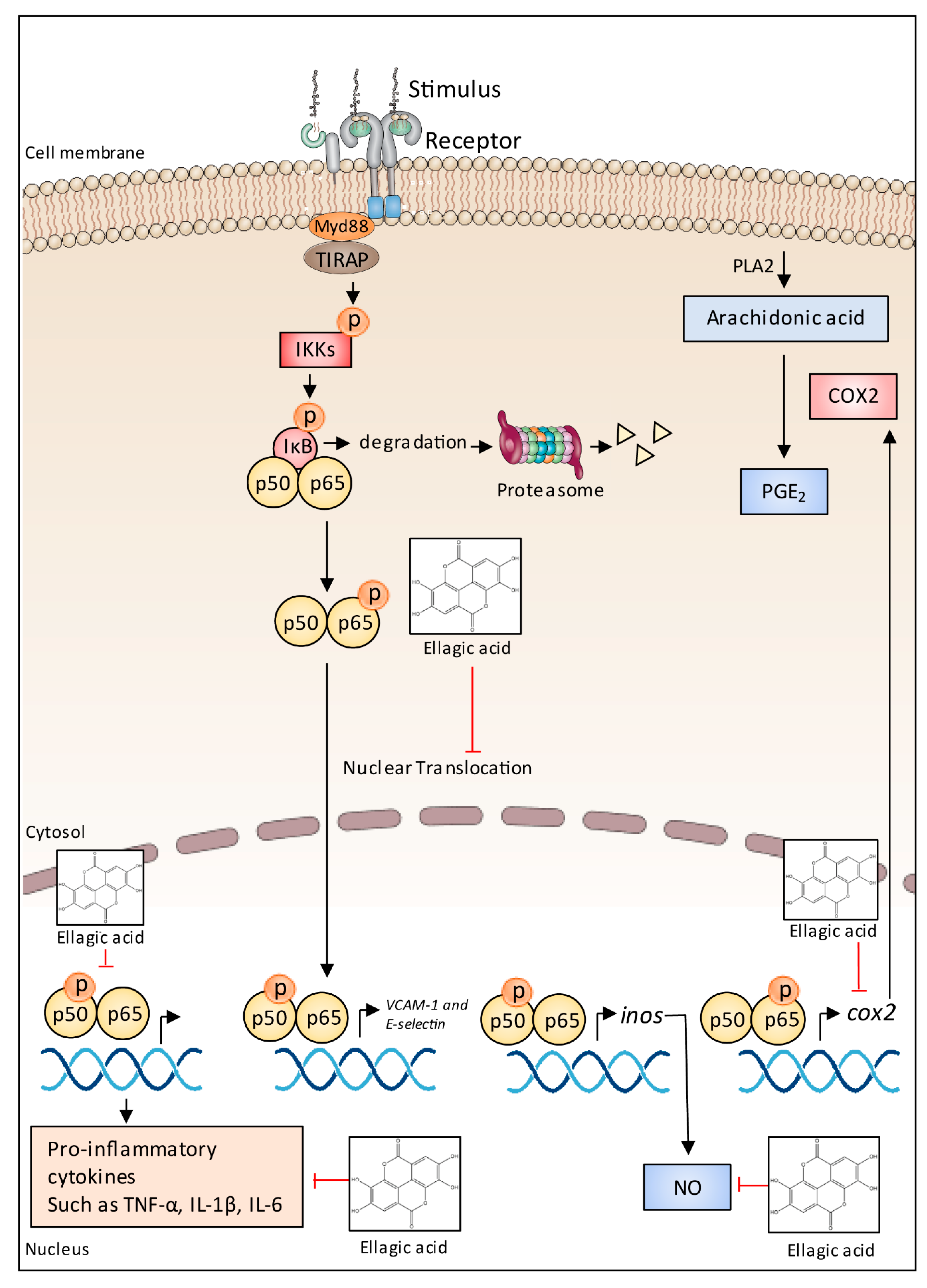
| Compounds | Function | References |
|---|---|---|
| Monomerics | ||
| Phenolic acids | Protection against infection of microbes, improvement of nutrient uptake, protection against insect depredation, signaling molecules in plant-microbes symbioses, involvement in plant allelopathy. | [46,47,48,49,50,51,52] |
| Flavonoids | Attract pollinators and seed dispersers, protection against oxidative stresses derived from UV, high light, and low temperatures, preventing photoinhibition and photobleaching, regulation of auxin transport, modulation of flower color, protection from high intensity light and UV, protection against DNA damage, involvement in plant allelopathy, antimicrobial activity, regulation of Rhizobium nodulation genes, protection against depredation by herbivores. | [53,54,55,56,57,58,59,60,61,62,63] |
| Dimerics, oligomerics, and polymerics | ||
| Proanthocyanidins | Protection against depredation by invertebrates and vertebrates, scavenging of reactive oxygen species, protection against microbes infection. | [64,65,66] |
| Hydrosable tannins | Protection against wounds and depredation by microbes and herbivores. | [67,68,69,70] |
| Cell wall materials | ||
| Lignins | Lodging resistance, involvement in plant fertility, mechanical barrier in seeds, biotic and abiotic stress resistance, involvement in plant growth and development. | [71,72,73,74,75] |
| Lignans | Scavenging of reactive oxygen species and antimicrobial activity, protection against insect depredation, involvement in plant allelopathy, phytohormone-like property. | [76,77,78,79] |
| Feedstock | Product Fraction | Phenolic Compounds | Method * | Ref. |
|---|---|---|---|---|
| Almond | Skin | Proanthocyanidins | HPLC-MS | [203] |
| Apple | Peel | Phenolic acids and monomeric flavonoids | UPLC-MS | [151] |
| Avocado | Peel and seed | Phenolic acids and flavonoids | HPLC-MS | [11] |
| Barley | Outermost milling fraction | Phenolic acids | HPLC | [204] |
| Blackberry | Seed meal | Phenolic acids, monomeric flavonoids, proanthocyanidins, and anthocyanins | HPLC-MS | [24] |
| Black raspberry | Seed meal | Phenolic acids, monomeric flavonoids, proanthocyanidins, and anthocyanins | HPLC-MS | [24] |
| Black raspberry | Seed | Ellagitannins and proanthocyanidins | HPLC-MS | [205] |
| Blueberry | Wine pomace | Anthocyanins | HPLC-MS | [26] |
| Blueberry | Seed meal | Phenolic acids, monomeric flavonoids, proanthocyanidins, and anthocyanins | HPLC-MS | [24] |
| Brazil nut | Skin | Phenolic acids, monomeric flavonoids, and proanthocyanidins | HPLC-MS | [80] |
| Camelina | Seed meal | Phenolic acids, monomeric flavonoids, and proanthocyanidins | HPLC-MS | [206] |
| Chia | Seed meal | Phenolic acids, monomeric flavonoids and proanthocyanidins | HPLC-MS | [207] |
| Citrus reticulata | Chempi (aged peel) | 5-demethylated polymethoxyflavones | HPLC | [208] |
| Grape | Pomace | Phenolic acids, monomeric flavonoids, proanthocyanidins, and anthocyanins | HPLC-MS | [209,210] |
| Grape | Pomace and rachi | Phenolic acids, monomeric flavonoids, proanthocyanidins, and anthocyanins | HPLC | [8] |
| Grape | Winemaking and grape juice by-products | Phenolic acids, monomeric flavonoids, and proanthocyanidins | HPLC-MS | [16] |
| Mango | Residual pulp | Phenolic acids and monomeric flavonoids | HPLC | [38] |
| Millet | Hull | Phenolic acids | HPLC-MS | [171] |
| Onion | Skin | Monomeric flavonoids | HPLC-MS | [211] |
| Orange | Peel | Flavonoids | HPLC-MS | [22] |
| Orange | Peel | Polymethoxyflavones | HPLC-MS | [212] |
| Passion fruit | Peel, albedo and seed | Phenolic acids and monomeric flavonoids | HPLC | [38] |
| Peanuts | Skin and meal | Phenolic acids, monomeric flavonoids, and proanthocyanidins | HPLC-MS | [12,31] |
| Peanuts | Skin | Phenolic acids, monomeric flavonoids, and proanthocyanidins | HPLC-MS | [17,144] |
| Peanuts | Skin | Proanthocyanidins | HPLC | [18,213] |
| Pineapple | Peel, and residual pulp | Phenolic acids and monomeric flavonoids | HPLC | [38] |
| Pomegranate | Peel and seed | Phenolic acids, monomeric flavonoids, anthocyanins, proanthocyanidins, and ellagitannins | HPLC-MS | [19,21] |
| Pomegranate | Peel | Punicalagin and ellagic acid | HPLC | [214] |
| Soybean | Okara | Isoflavones | UPLC | [215] |
| Soybean | Seed coat | Phenolic acids and flavonoids | HPLC-MS | [40] |
| Sophia | Seed meal | Phenolic acids, monomeric flavonoids, and proanthocyanidins | HPLC-MS | [206] |
| Wheat | Bran | Phenolic acids | HPLC | [28,41] |
| Feedstock | Product Fraction | Evaluation Purpose and/or Application | Ref. |
|---|---|---|---|
| Almond | Skin | Effects towards antioxidant enzymes using cell and animal models. | [203] |
| Apple | Peel | Scavenging activity against DPPH radical, and ferric reducing antioxidant power. Inhibition of fish oil oxidation. | [151] |
| Avocado | Peel and seed | Reducing power (FRAP) and antioxidant potential against ABTS radical cation, DPPH radical, and reactive oxygen species (peroxyl and superoxide radical and hypochlorous acid). Anti-inflammatory activity by inhibition TNF-α and nitric oxide in mouse macrophage RAW 264.7 cells. | [11] |
| Barley | Outermost milling fraction | Antioxidant potential against ABTS radical cation, DPPH, peroxyl and superoxide radical. Antioxidant potential using a photoinduced chemiluminescence technique. | [204] |
| Barley | Outermost milling fraction | Scavenging of peroxyl and hydroxyl radicals, metal chelation activity, inhibition of radical-induced supercoiled DNA breakage and antiproliferative activities using Caco-2 human adenocarcinoma cells. | [45] |
| Blackberry | Seed meal | Antioxidant activity (towards hydroxyl and peroxyl radicals), reducing power, chelation capacity, prevention of DNA damage, and LDL-cholesterol oxidation. | [24] |
| Black raspberry | Seed | Reducing power (FRAP) and antioxidant potential towards DPPH radical and ABTS radical cation. Anti-inflammatory activity by reduction of nitric oxide using RAW 264.7 cells. | [205] |
| Black raspberry | Seed meal | Antioxidant activity (towards hydroxyl and peroxyl radicals), reducing power, chelation capacity, prevention of DNA damage, and LDL-cholesterol oxidation. | [24] |
| Blueberry | Seed meal | Antioxidant activity (towards hydroxyl and peroxyl radicals), reducing power, chelation capacity, prevention of DNA damage, and LDL-cholesterol oxidation. | [24] |
| Brazil nut | Skin | Antioxidant potential towards ABTS radical cation, and DPPH, hydroxyl, and peroxyl radicals. | [80] |
| Camelina | Seed meal | Potential biological activities of camelina and sophia seed meals through inhibition of LDL-cholesterol oxidation, DNA damage as well as pancreatic lipase and α-glucosidase activities. | [223] |
| Camelina | Seed meal | Antioxidant potential towards ABTS radical cation, reducing power and metal chelation. | [206] |
| Canola | Hull | Antioxidant potential of crude tannins by β-carotene-linoleate model system, DPPH radical, and reducing power. | [195] |
| Chia | Seed meal | Antioxidant potential towards ABTS radical cation, DPPH and hydroxyl radical. Reducing power, chelation capacity and antioxidant capacity in beta-carotene linoleate model system. Inhibition of activities against pancreatic lipase, α-glucosidase, human LDL-cholesterol oxidation in vitro, DNA damage induced by peroxyl and hydroxyl radicals. | [207] |
| Citrus reticulata | Chempi (aged peel) | Prevention of obesity and type 2 diabetes in mouse model. | [208] |
| Grape | Pomace | Anti-inflammatory activity in mice (inhibition of TNF-α and IL-1β). | [209] |
| Grape | Pomace | Antioxidant capacity using yeast cells. | [210] |
| Grape | Pomace | Isolation and identification of phenolics bearing inhibition capacity towards α-glucosidase. | [6] |
| Grape | Pomace | Antioxidant potential towards DPPH radical and ABTS radical cation. | [224] |
| Grape | Pomace and rachi | Antioxidant activity (towards DPPH radical, ABTS radical cation, peroxyl radical, superoxide anion, hypochlorous acid) and anti-inflammatory effect by suppressing TNF-α liberation in vitro. | [8] |
| Grape | Seed | Anti-inflammatory activity (inhibition of cytokines and suppression of MAPK and NF-κB) in RAW264.7 macrophages. | [225] |
| Grape | Seed | Reduction of bone loss in the experimental arthritis. | [226] |
| Grape | Seed | Reduction of kidney injury in experimental type 2 diabetes. | [227] |
| Grape | Winemaking by-products | Antioxidant potential towards ABTS radical cation, DPPH and hydroxyl radical. Reducing power and inhibition of α-glucosidase and lipase activities. | [30] |
| Grape | Winemaking and grape juice by-products | Antioxidant activity (towards DPPH radical, ABTS radical cation, and hydrogen peroxide), reducing power, prevention of DNA damage, and LDL-cholesterol oxidation. | [16] |
| Grape | Winemaking by-products | Bioactivity using cardiometabolic biomarkers in Wistar rats. | [15] |
| Guava | Pomace | Anti-inflammatory activity through reduction of edema and neutrophil migration in mice models. | [228] |
| Mango | Residual pulp | Microbiological safety and antioxidant activity (towards DPPH radical, ABTS radical cation) | [38] |
| Millet | Hull | Hydroxyl and peroxyl radical inhibition, inhibition of DNA strand scission induced by both ROS, inhibition of liposome oxidation, and human colon adenocarcinoma cell proliferation inhibition. | [171] |
| Onion | Skin | Inhibition of peroxyl and hydroxyl radical induced supercoiled DNA strand scission, cupric ion induced human low-density lipoprotein peroxidation inhibition in vitro, inhibition of lipopolysaccharide stimulated cyclooxygenase-2 expression in mouse macrophage cell model. | [229] |
| Onion | Skin | Antioxidant potential (ABTS radical cation, DPPH radical, and reducing power). | [211] |
| Passion fruit | Peel, albedo and seed | Microbiological safety and antioxidant activity (towards DPPH radical, ABTS radical cation) | [38] |
| Peanuts | Skin | Gamma-irradiation induced changes and microbiological safety. Antioxidant potential (towards DPPH radical, ABTS radical cation, hydroxyl radical, and hydrogen peroxide), reducing power, prevention of DNA damage, and LDL-cholesterol oxidation. | [17] |
| Peanuts | Skin and meal | Antioxidant potential against ABTS radical cation, DPPH and hydroxyl radicals, and reducing power. Antioxidant capacity in gamma-irradiated fish model system. Antimicrobial activity against Gram-positive and Gram-negative bacteria. | [12] |
| Peanuts | Skin and meal | Antioxidant potential towards ABTS radical cation, DPPH and hydroxyl radicals, and reducing power. Inhibition of α-glucosidase and lipase activities. | [31] |
| Peanuts | Skin | Isolation, structural characterization of proanthocyanidins, and evaluation of their antioxidant activity towards DPPH radical, ABTS radical cation, and ferric reducing antioxidant power. | [18] |
| Peanuts | Skin | Isolation and identification of proanthocyanidins. Inhibition of TNF-α and IL-6 in cultured human monocytic THP-1 cells. | [213] |
| Pineapple | Peel, and residual pulp | Microbiological safety and antioxidant activity (towards DPPH radical, ABTS radical cation) | [38] |
| Pomegranate | Peel and seed | Scavenging of ABTS radical cation, DPPH and hydroxyl radicals, and metal chelation. Potential bioactivity towards inhibition of α-glucosidase and lipase activity, inhibition of human low-density lipoprotein (LDL) oxidation in vitro and inhibition of peroxyl and hydroxyl radical-induced DNA strand scission. | [19,21] |
| Pomegranate | Peel and seed | Antioxidant activity in beta-carotene-linoleate model system and against DPPH radical. Prevention of lipid peroxidation in albino rat liver homogenate in vitro, scavenging activity towards hydroxyl radical scavenging activity, and human low-density lipoprotein (LDL) oxidation in vitro. | [230] |
| Pomegranate | Peel | Anti-inflammatory activity through inhibition of expression of TNF-α, IL-1β, MCP-1 and ICAM-1 and adhesion of monocytes to endothelial cells. | [214] |
| Rapeseed | Hull | Antioxidant potential of crude tannins by β-carotene-linoleate model system, DPPH radical, and reducing power. | [195] |
| Sophia | Seed meal | Antioxidant potential towards ABTS radical cation, reducing power and metal chelation. | [206] |
| Sophia | Seed meal | Potential biological activities of camelina and sophia seed meals through inhibition of LDL-cholesterol oxidation, DNA damage as well as pancreatic lipase and α-glucosidase activities. | [223] |
| Soybean | Seed coat | Antioxidant potential towards ABTS radical cation and DPPH as well as reducing power (FRAP assay). | [40] |
| Wheat | Bran | Antioxidant potential against peroxyl radical and via photochemiluminescence method, antioxidant capacity in seal blubber oil (Rancimat test) and inhibition of oxidation of low-density lipoprotein and DNA in vitro. | [41] |
| Wheat | Bran | Antioxidant potential against ABTS radical cation. | [42] |
| Wheat | Bran | Antioxidant potential against ABTS radical cation, DPPH and peroxyl radicals, reducing power, inhibition of photochemilumenescence, and iron (II) chelation activity. Inhibition of oxidation of human low-density lipoprotein cholesterol and DNA in vitro. Oxidative stability using stripped corn oil in Rancimat test. | [43] |
| Wheat | Bran | Antioxidant potential towards ABTS radical cation, DPPH, superoxide radicals, hydroxyl radical, and scavenging of hydrogen peroxide. Reducing power and ferrous chelating activity. | [44] |
| Wheat | Bran fractions | Total antioxidant capacity towards ABTS radical cation as affected by debranning. | [28] |
| Compound | IC50 (µg/mL) | Ref. |
|---|---|---|
| A-amylase | [280] | |
| (−)-epicatechin | 140 | |
| Epigallocatechin | >300 | |
| (−)-4′-O-methylepigallocatechin | >300 | |
| (−)-epicatechin-(4b→8)-(−)-4′-O-methylepigallocatechin | >300 | |
| α-glucosidase | [280] | |
| (−)-epicatechin | 140 | |
| Epigallocatechin | >300 | |
| (−)-4′-O-Methylepigallocatechin | >300 | |
| (−)-epicatechin-(4b→8)-(−)-4′-O-methylepigallocatechin | >300 | |
| Lipase | [281] | |
| Rosmarinic acid | 125 | |
| Chlorogenic acid | 96.5 | |
| Caffeic acid | 32.6 | |
| Gallic acid | 10.1 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
De Camargo, A.C.; Schwember, A.R.; Parada, R.; Garcia, S.; Maróstica Júnior, M.R.; Franchin, M.; Regitano-d’Arce, M.A.B.; Shahidi, F. Opinion on the Hurdles and Potential Health Benefits in Value-Added Use of Plant Food Processing By-Products as Sources of Phenolic Compounds. Int. J. Mol. Sci. 2018, 19, 3498. https://doi.org/10.3390/ijms19113498
De Camargo AC, Schwember AR, Parada R, Garcia S, Maróstica Júnior MR, Franchin M, Regitano-d’Arce MAB, Shahidi F. Opinion on the Hurdles and Potential Health Benefits in Value-Added Use of Plant Food Processing By-Products as Sources of Phenolic Compounds. International Journal of Molecular Sciences. 2018; 19(11):3498. https://doi.org/10.3390/ijms19113498
Chicago/Turabian StyleDe Camargo, Adriano Costa, Andrés R. Schwember, Roberto Parada, Sandra Garcia, Mário Roberto Maróstica Júnior, Marcelo Franchin, Marisa Aparecida Bismara Regitano-d’Arce, and Fereidoon Shahidi. 2018. "Opinion on the Hurdles and Potential Health Benefits in Value-Added Use of Plant Food Processing By-Products as Sources of Phenolic Compounds" International Journal of Molecular Sciences 19, no. 11: 3498. https://doi.org/10.3390/ijms19113498
APA StyleDe Camargo, A. C., Schwember, A. R., Parada, R., Garcia, S., Maróstica Júnior, M. R., Franchin, M., Regitano-d’Arce, M. A. B., & Shahidi, F. (2018). Opinion on the Hurdles and Potential Health Benefits in Value-Added Use of Plant Food Processing By-Products as Sources of Phenolic Compounds. International Journal of Molecular Sciences, 19(11), 3498. https://doi.org/10.3390/ijms19113498








