Activated Integrated Stress Response Induced by Salubrinal Promotes Cisplatin Resistance in Human Gastric Cancer Cells via Enhanced xCT Expression and Glutathione Biosynthesis
Abstract
1. Introduction
2. Results
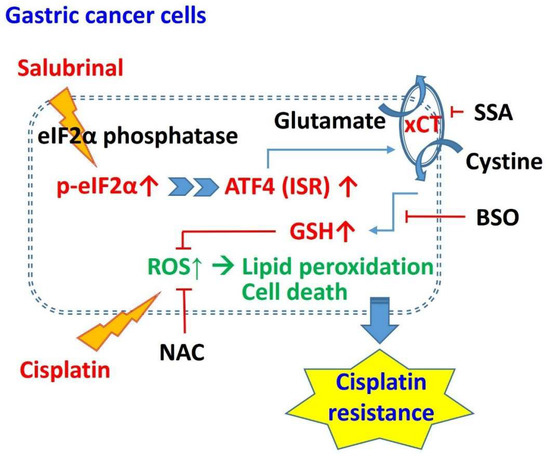
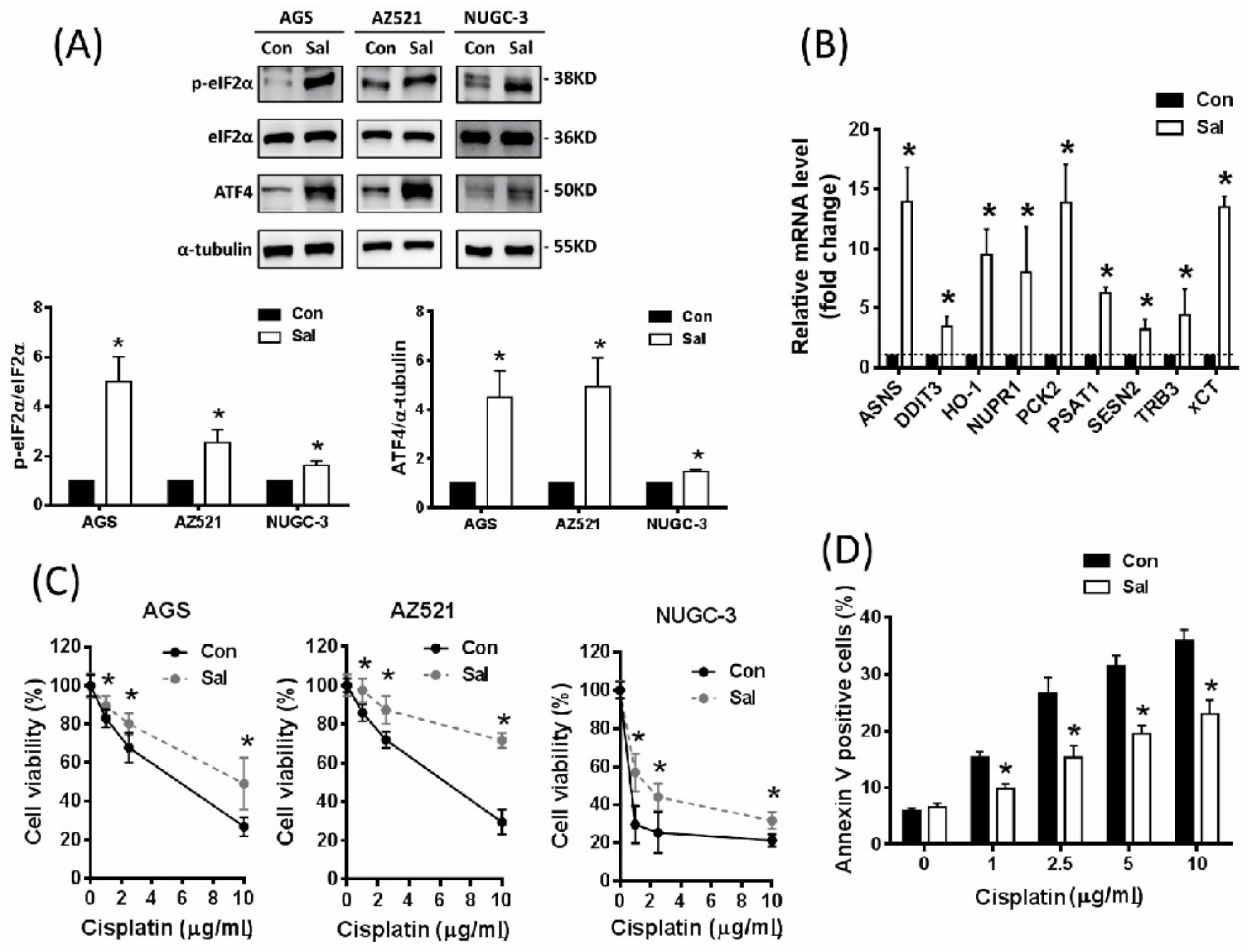
2.1. Salubrinal Activates ISR and Induces Cisplatin Resistance in Gastric Cancer Cells
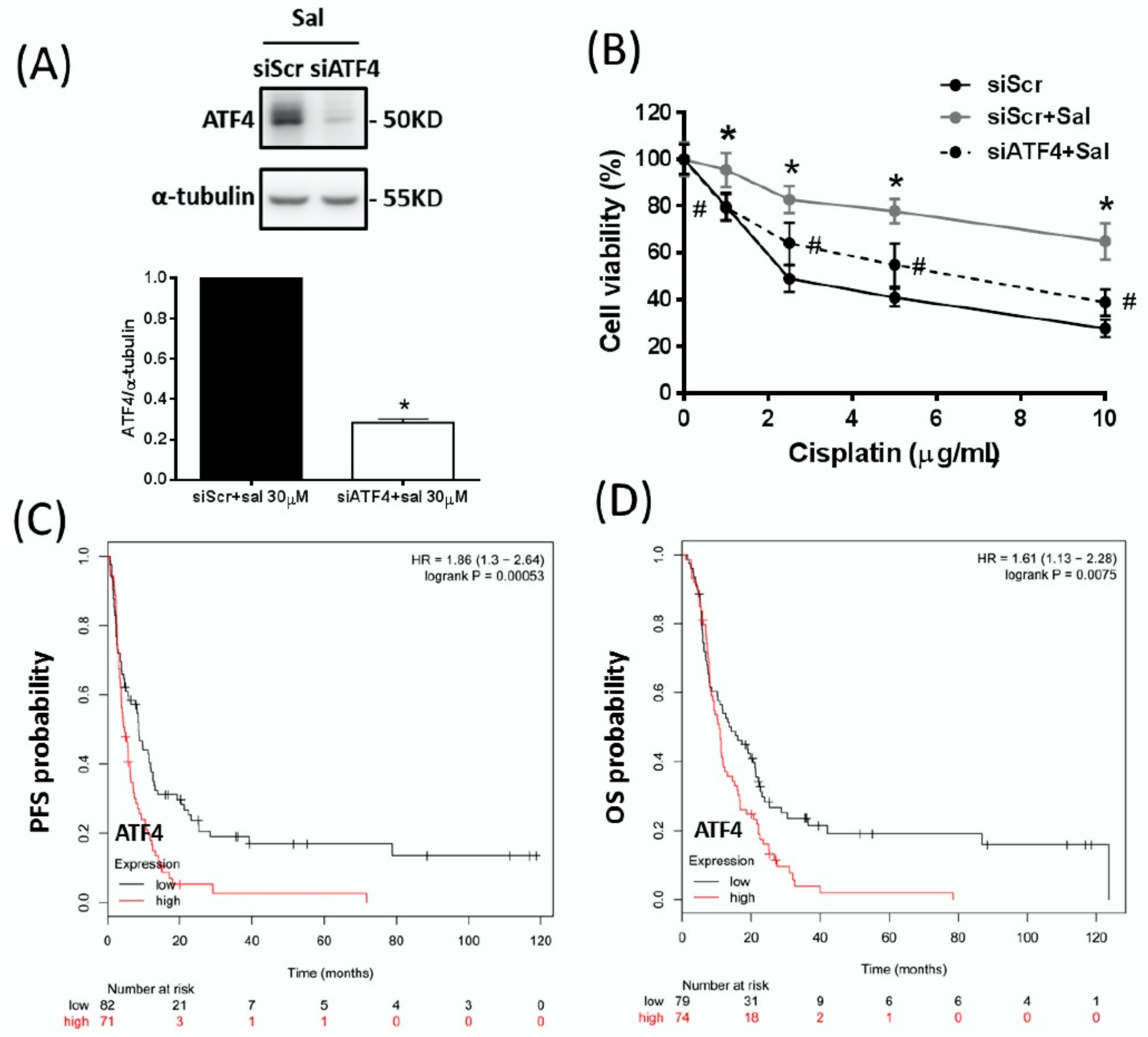
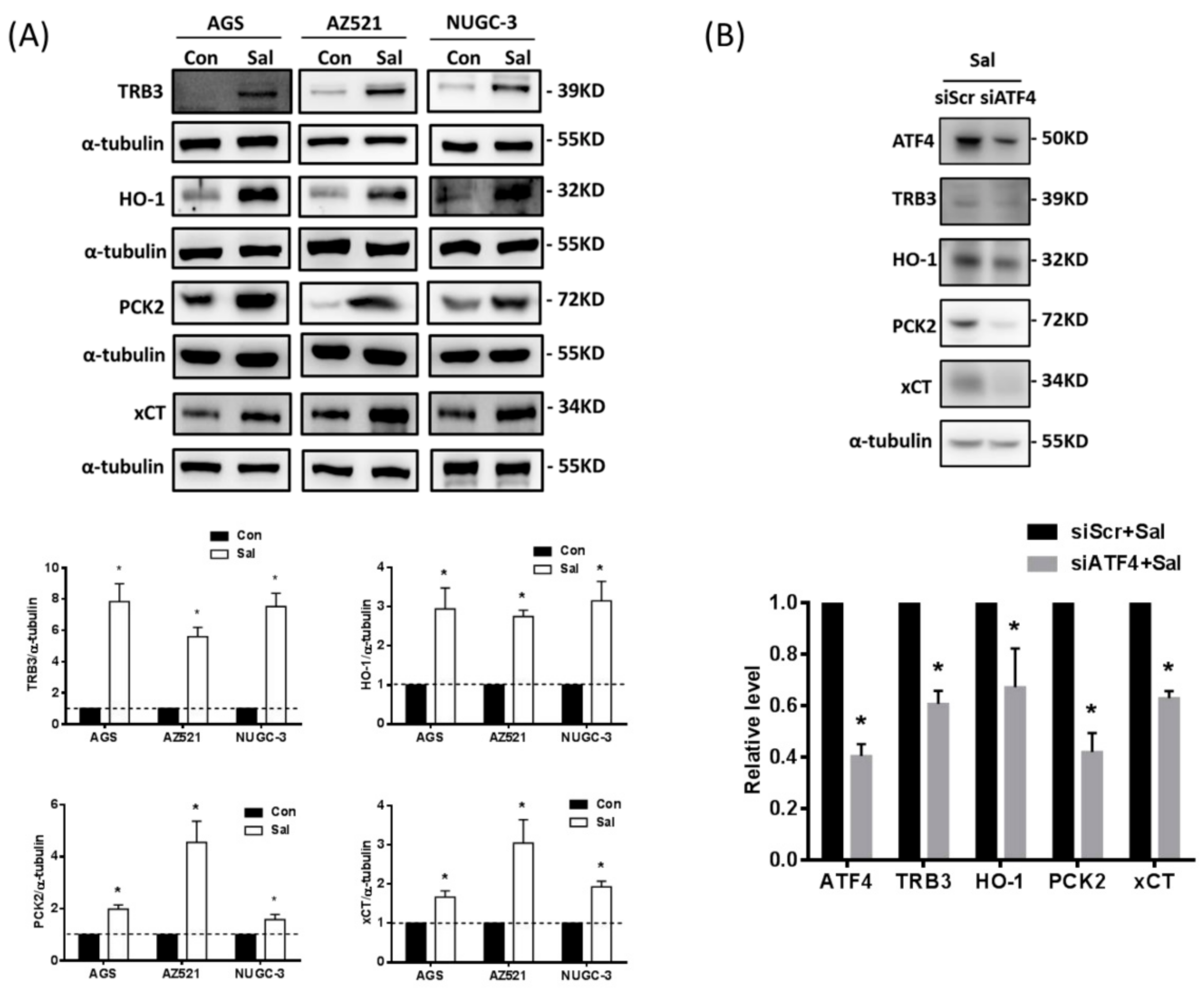
2.2. ATF4 Plays an Essential Role in Salubrinal-Induced Cisplatin Resistance in Gastric Cancer Cells
2.3. Some of the ATF4-Regulated Genes, TRB3, HO-1, PCK2, and xCT, are Associated with Gastric Cancer Patients with Poor Prognosis after Adjuvant Chemotherapy
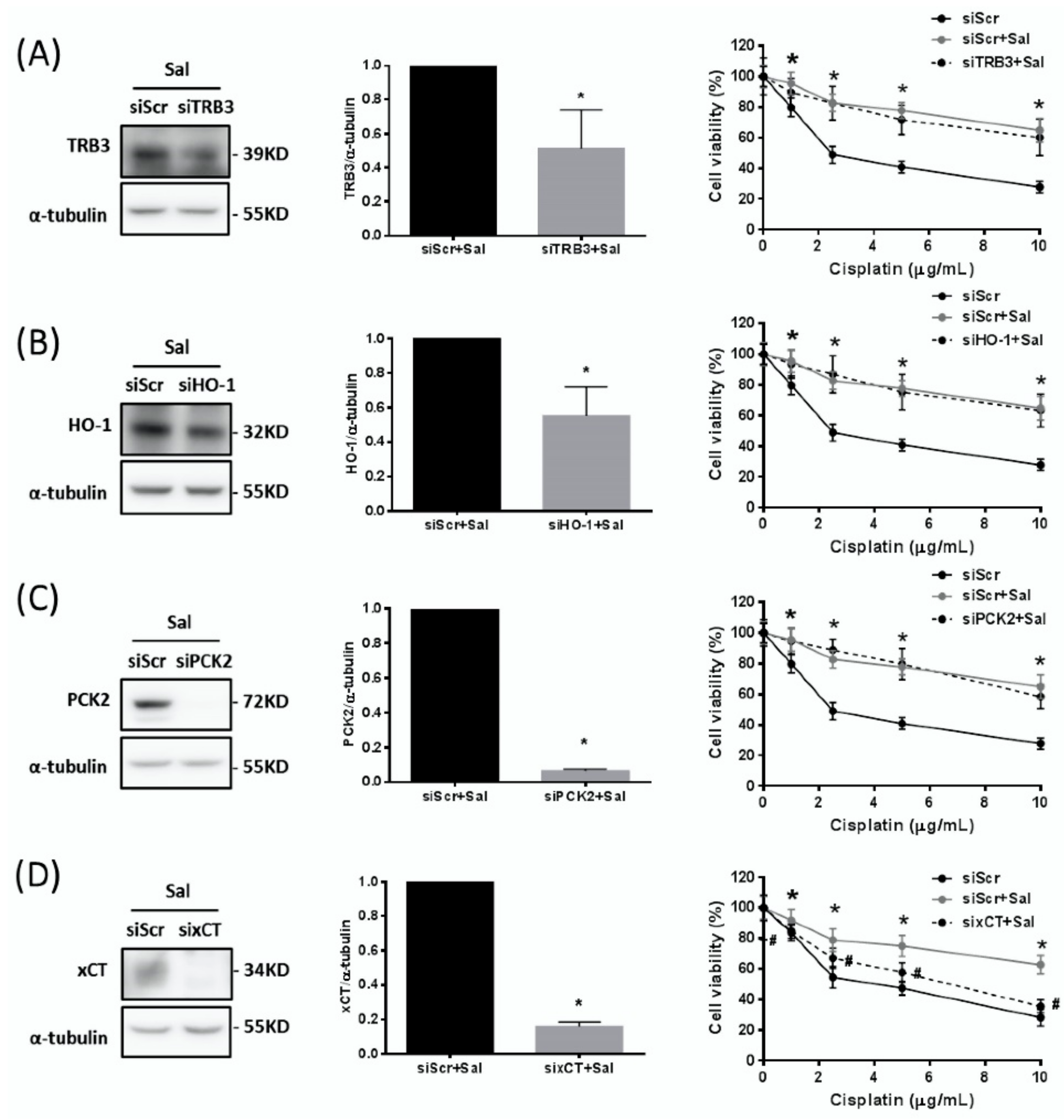
2.4. Up-Regulated Expression of xCT, but not of TRB3, HO-1, or PCK2, Contributes to the Salubrinal-Induced Cisplatin Resistance
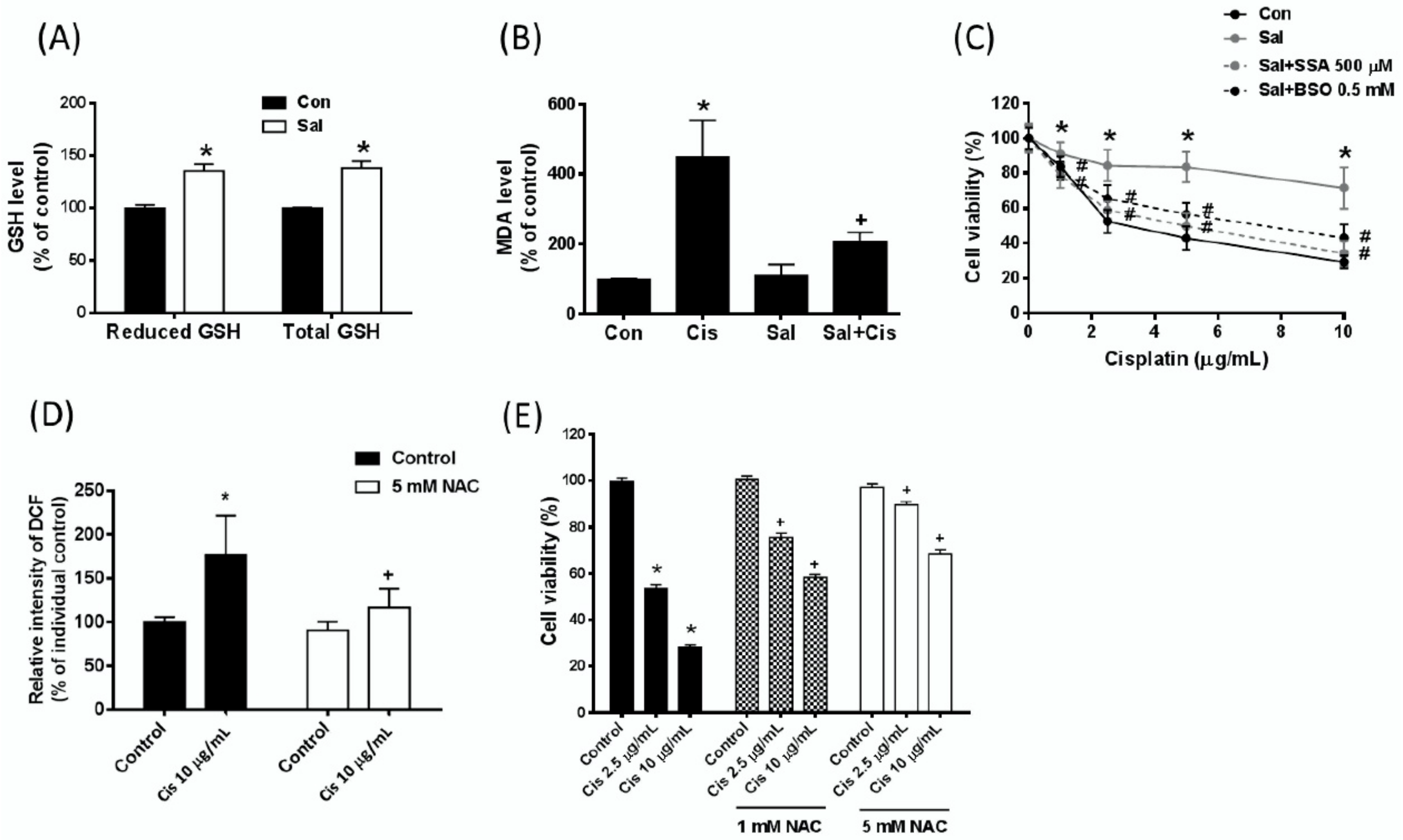
2.5. Salubrinal-Induced xCT Expression is Associated with Increased Intracellular Glutathione (GSH) Biosynthesis and Decreased Cisplatin-Induced Oxidative Stress
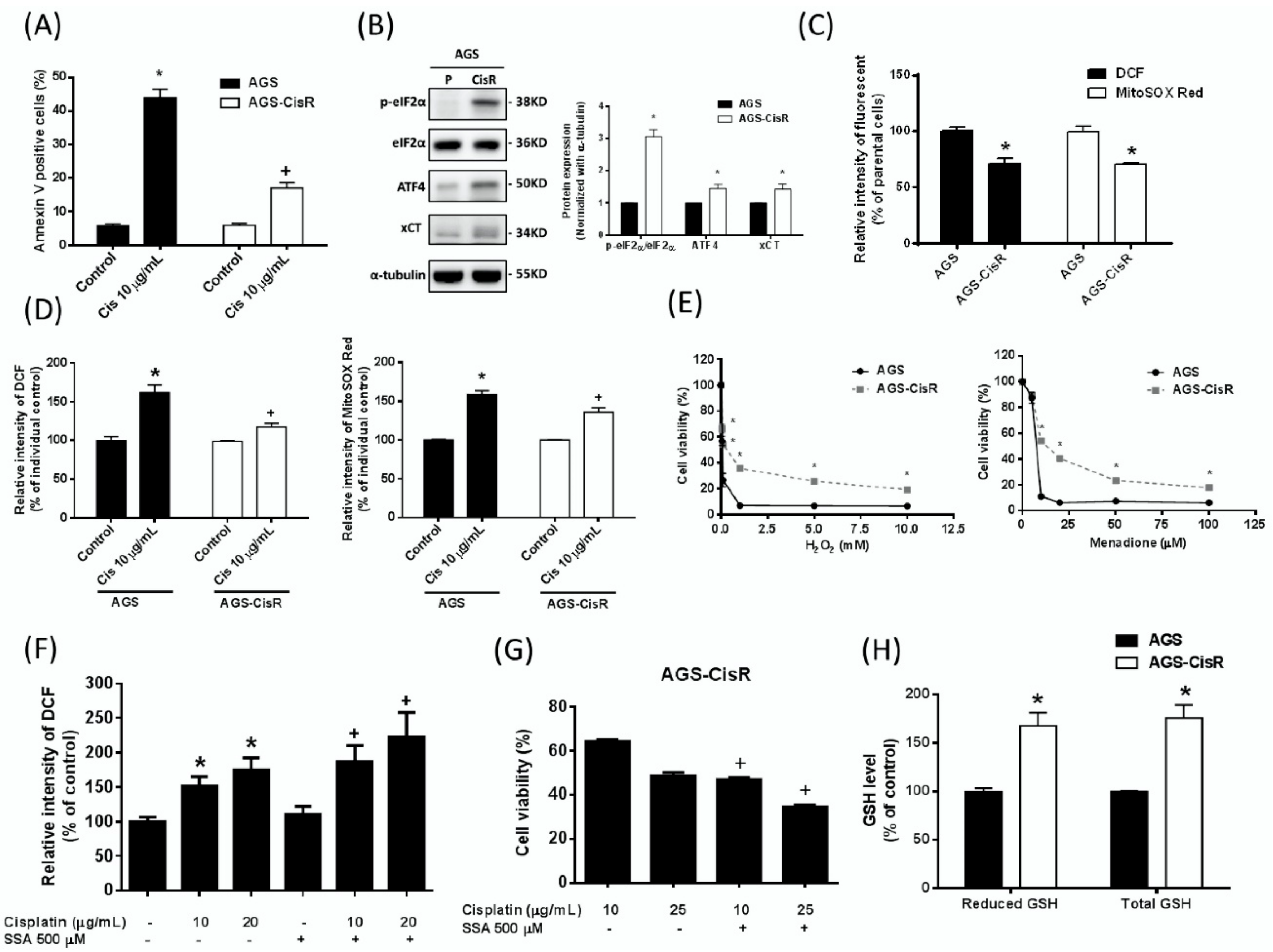
2.6. High xCT Expression Contributes to Cisplatin Resistance of the Cisplatin-Resistant Gastric Cancer Cells
3. Discussion
4. Materials and Methods
4.1. Cell Culture
4.2. Determination of Cell Viability
4.3. Annexin V Staining Assay
4.4. Detection of the Levels of Intracellular ROS and Mitochondrial ROS
4.5. Kaplan–Meier Plotter Analysis
4.6. Western Blot Analysis
4.7. Small Interfering RNA (siRNA)-Mediated Genetic Knockdown
4.8. Quantitative Real-Time Reverse Transcription (RT)-Polymerase Chain Reaction (q-RT PCR)
4.9. Microarray Analysis and Ingenuity Pathway Analysis (IPA)
4.10. Glutathione (GSH) Detection
4.11. Lipid Peroxidation Assay
4.12. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| 5-FU | 5-Fluorouracil |
| AARE | amino acid response element |
| ATF4 | activating transcription factor-4 |
| ATP7B | copper-transporting ATPase |
| BSA | bovine serum albumin |
| BSO | buthioninesulfoximine |
| CO | carbon monoxide |
| CTR1 | copper transporter 1 |
| EDTA | ethylenediaminetetraacetic acid |
| eIF2α | eukaryotic translation initiation factor 2α |
| ERCC1 | excision repair cross-complementing rodent repair deficiency, complementation group 1 |
| FBS | foetal bovine serum |
| GAPDH | glyceraldehyde 3-phosphate dehydrogenase |
| GCN2 | general control nonderepressible 2 |
| GSH | glutathione |
| H2O2 | hydrogen peroxide |
| HO-1(HMOX1) | heme oxygenase 1 |
| HRI | heme-regulated eIF2α kinase |
| IPA | Ingenuity pathway analysis |
| ISR | Integrated stress response |
| KM | Kaplan-Meier |
| MDA | Malondialdehyde |
| MRP2 | multidrug resistance-associated protein 2 |
| mtDNA | mitochondrial DNA |
| Na3VO4 | sodium orthovanadate |
| NAC | N-acetylcysteine |
| NaCl | sodium chloride |
| NADPH | nicotinamide adenine dinucleotide phosphate |
| OS | overall survival |
| P/S | penicillin/streptomycin |
| PAGE | polyacrylamide gel electrophoresis |
| PCK2 | phosphoenolpyruvate carboxykinase 2 |
| PEPCK | phosphoenolpyruvate carboxykinase |
| PERK | PKR-like endoplasmic reticulum kinase |
| PFS | progression-free survival |
| PKR | protein kinase R |
| PMSF | phenylmethanesulfonyl fluoride |
| PVDF | polyvinylidene difluoride membrane |
| q-RTPCR | quantitative real-time reverse transcription-polymerase chain reaction |
| ROS | reactive oxygen species |
| RT | reverse transcription |
| SDS | sodium dodecyl sulfate |
| SRB | Sulforhodamine B |
| SSA | sulfasalazine |
| TCA | trichloroacetic acid |
| TRB3 | tribbles-related protein 3 |
| VRACs | volume-regulated anion channels |
References
- Ajani, J.A.; Lee, J.; Sano, T.; Janjigian, Y.Y.; Fan, D.; Song, S. Gastric adenocarcinoma. Nat. Rev. Dis. Primers 2017, 3, 17036. [Google Scholar] [CrossRef] [PubMed]
- Wagner, A.D.; Groth, W.; Haerting, J.; Kleber, G.; Grothey, A.; Fleig, W.E. Chemotherapy in advanced gastric cancer: A systematic review and meta-analysis based on aggregate data. J. Clin. Oncol. 2006, 24, 2903–2909. [Google Scholar] [CrossRef] [PubMed]
- Webb, A.; Cunningham, D.; Scarffe, J.H.; Harper, P.; Norman, A.; Joffe, J.K.; Hughes, M.; Mansi, J.; Findlay, M.; Hill, A.; et al. Randomized trial comparing epirubicin, cisplatin, and fluorouracil versus fluorouracil, doxorubicin, and methotrexate in advanced esophagogastric cancer. J. Clin. Oncol. 1997, 15, 261–267. [Google Scholar] [CrossRef] [PubMed]
- Sasako, M.; Sakuramoto, S.; Katai, H.; Kinoshita, T.; Furukawa, H.; Yamaguchi, T.; Nashimoto, A.; Fujii, M.; Nakajima, T.; Ohashi, Y. Five-year outcomes of a randomized phase III trial comparing adjuvant chemotherapy with S-1 versus surgery alone in stage II or III gastric cancer. J. Clin. Oncol. 2011, 29, 4387–4393. [Google Scholar] [CrossRef] [PubMed]
- Noh, S.H.; Park, S.R.; Yang, H.K.; Chung, H.C.; Chung, I.J.; Kim, S.W.; Yu, W.; Lee, J.I.; Shin, D.B.; Ji, J.; et al. CLASSIC trial investigators, Adjuvant capecitabine plus oxaliplatin for gastric cancer after D2 gastrectomy (CLASSIC): 5-year follow-up of an open-label, randomised phase 3 trial. Lancet Oncol. 2014, 15, 1389–1396. [Google Scholar] [CrossRef]
- Kim, N.K.; Park, Y.S.; Heo, D.S.; Suh, C.; Kim, S.Y.; Park, K.C.; Kang, Y.K.; Shin, D.B.; Kim, H.T.; Kim, H.J.; et al. A phase III randomized study of 5-fluorouracil and cisplatin versus 5-fluorouracil, doxorubicin, and mitomycin C versus 5-fluorouracil alone in the treatment of advanced gastric cancer. Cancer 1993, 71, 3813–3818. [Google Scholar] [CrossRef]
- Cvitkovic, E. A historical perspective on oxaliplatin: Rethinking the role of platinum compounds and learning from near misses. Semin. Oncol. 1998, 25, 1–3. [Google Scholar] [PubMed]
- Andrews, P.A.; Howell, S.B. Cellular pharmacology of cisplatin: Perspectives on mechanisms of acquired resistance. Cancer Cells 1990, 2, 35–43. [Google Scholar] [PubMed]
- Saris, C.P.; van de Vaart, P.J.; Rietbroek, R.C.; Blommaert, F.A. In vitro formation of DNA adducts by cisplatin, lobaplatin and oxaliplatin in calf thymus DNA in solution and in cultured human cells. Carcinogenesis 1996, 17, 2763–2769. [Google Scholar] [CrossRef] [PubMed]
- Cullen, K.J.; Yang, Z.; Schumaker, L.; Gu, Z. Mitochondria as a critical target of the chemotheraputic agent cisplatin in head and neck cancer. J. Bioenerg. Biomembr. 2007, 39, 43–50. [Google Scholar] [CrossRef] [PubMed]
- Woynarowski, J.M.; Chapman, W.G.; Napier, C.; Herzig, M.C.; Juniewicz, P. Sequence- and region-specificity of oxaliplatin adducts in naked and cellular DNA. Mol. Pharmacol. 1998, 54, 770–777. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Schumaker, L.M.; Egorin, M.J.; Zuhowski, E.G.; Guo, Z.; Cullen, K.J. Cisplatin preferentially binds mitochondrial DNA and voltage-dependent anion channel protein in the mitochondrial membrane of head and neck squamous cell carcinoma: Possible role in apoptosis. Clin. Cancer Res. 2006, 12, 5817–5825. [Google Scholar] [CrossRef] [PubMed]
- Murata, T.; Hibasami, H.; Maekawa, S.; Tagawa, T.; Nakashima, K. Preferential binding of cisplatin to mitochondrial DNA and suppression of ATP generation in human malignant melanoma cells. Biochem. Int. 1990, 20, 949–955. [Google Scholar] [PubMed]
- Galluzzi, L.; Vitale, I.; Michels, J.; Brenner, C.; Szabadkai, G.; Harel-Bellan, A.; Castedo, M.; Kroemer, G. Systems biology of cisplatin resistance: Past, present and future. Cell Death Dis. 2014, 5, e1257. [Google Scholar] [CrossRef] [PubMed]
- Ishida, S.; Lee, J.; Thiele, D.J.; Herskowitz, I. Uptake of the anticancer drug cisplatin mediated by the copper transporter Ctr1 in yeast and mammals. Proc. Natl. Acad. Sci. USA 2002, 99, 14298–14302. [Google Scholar] [CrossRef] [PubMed]
- Nakayama, K.; Kanzaki, A.; Ogawa, K.; Miyazaki, K.; Neamati, N.; Takebayashi, Y. Copper-transporting P-type adenosine triphosphatase (ATP7B) as a cisplatin based chemoresistance marker in ovarian carcinoma: Comparative analysis with expression of MDR1, MRP1, MRP2, LRP and BCRP. Int. J. Cancer 2002, 101, 488–495. [Google Scholar] [CrossRef] [PubMed]
- Yamasaki, M.; Makino, T.; Masuzawa, T.; Kurokawa, Y.; Miyata, H.; Takiguchi, S.; Nakajima, K.; Fujiwara, Y.; Matsuura, N.; Mori, M.; et al. Role of multidrug resistance protein 2 (MRP2) in chemoresistance and clinical outcome in oesophageal squamous cell carcinoma. Br. J. Cancer 2011, 104, 707–713. [Google Scholar] [CrossRef] [PubMed]
- Planells-Cases, R.; Lutter, D.; Guyader, C.; Gerhards, N.M.; Ullrich, F.; Elger, D.A.; Kucukosmanoglu, A.; Xu, G.; Voss, F.K.; Reincke, S.M.; et al. Subunit composition of VRAC channels determines substrate specificity and cellular resistance to Pt-based anti-cancer drugs. EMBO J. 2015, 34, 2993–3008. [Google Scholar] [CrossRef] [PubMed]
- Britten, R.A.; Liu, D.; Tessier, A.; Hutchison, M.J.; Murray, D. ERCC1 expression as a molecular marker of cisplatin resistance in human cervical tumor cells. Int. J. Cancer 2000, 89, 453–457. [Google Scholar] [CrossRef]
- Perego, P.; Giarola, M.; Righetti, S.C.; Supino, R.; Caserini, C.; Delia, D.; Pierotti, M.A.; Miyashita, T.; Reed, J.C.; Zunino, F. Association between cisplatin resistance and mutation of p53 gene and reduced bax expression in ovarian carcinoma cell systems. Cancer Res. 1996, 56, 556–562. [Google Scholar] [PubMed]
- Cho, H.J.; Kim, J.K.; Kim, K.D.; Yoon, H.K.; Cho, M.Y.; Park, Y.P.; Jeon, J.H.; Lee, E.S.; Byun, S.S.; Lim, H.M.; et al. Upregulation of Bcl-2 is associated with cisplatin-resistance via inhibition of Bax translocation in human bladder cancer cells. Cancer Lett. 2006, 237, 56–66. [Google Scholar] [CrossRef] [PubMed]
- Fraser, M.; Bai, T.; Tsang, B.K. Akt promotes cisplatin resistance in human ovarian cancer cells through inhibition of p53 phosphorylation and nuclear function. Int. J. Cancer 2008, 122, 534–546. [Google Scholar] [CrossRef] [PubMed]
- Harding, H.P.; Zhang, Y.; Zeng, H.; Novoa, I.; Lu, P.D.; Calfon, M.; Sadri, N.; Yun, C.; Popko, B.; Paules, R.; et al. An integrated stress response regulates amino acid metabolism and resistance to oxidative stress. Mol. Cell 2003, 11, 619–633. [Google Scholar] [CrossRef]
- Pakos-Zebrucka, K.; Koryga, I.; Mnich, K.; Ljujic, M.; Samali, A.; Gorman, A.M. The integrated stress response. EMBO Rep. 2016, 17, 1374–1395. [Google Scholar] [CrossRef] [PubMed]
- B’Chir, W.; Maurin, A.C.; Carraro, V.; Averous, J.; Jousse, C.; Muranishi, Y.; Parry, L.; Stepien, G.; Fafournoux, P.; Bruhat, A. The eIF2alpha/ATF4 pathway is essential for stress-induced autophagy gene expression. Nucleic Acids Res. 2013, 41, 7683–7699. [Google Scholar] [CrossRef] [PubMed]
- Garcia, M.A.; Meurs, E.F.; Esteban, M. The dsRNA protein kinase PKR: Virus and cell control. Biochimie 2007, 89, 799–811. [Google Scholar] [CrossRef] [PubMed]
- Han, A.P.; Yu, C.; Lu, L.; Fujiwara, Y.; Browne, C.; Chin, G.; Fleming, M.; Leboulch, P.; Orkin, S.H.; Chen, J.J. Heme-regulated eIF2alpha kinase (HRI) is required for translational regulation and survival of erythroid precursors in iron deficiency. EMBO J. 2001, 20, 6909–6918. [Google Scholar] [CrossRef] [PubMed]
- Walter, P.; Ron, D. The unfolded protein response: From stress pathway to homeostatic regulation. Science 2011, 334, 1081–1086. [Google Scholar] [CrossRef] [PubMed]
- Wek, R.C.; Jiang, H.Y.; Anthony, T.G. Coping with stress: EIF2 kinases and translational control. Biochem. Soc. Trans. 2006, 34, 7–11. [Google Scholar] [CrossRef] [PubMed]
- Wek, R.C.; Staschke, K.A. How do tumours adapt to nutrient stress? EMBO J. 2010, 29, 1946–1947. [Google Scholar] [CrossRef] [PubMed]
- Lewerenz, J.; Maher, P. Basal levels of eIF2alpha phosphorylation determine cellular antioxidant status by regulating ATF4 and xCT expression. J. Biol. Chem. 2009, 284, 1106–1115. [Google Scholar] [CrossRef] [PubMed]
- Bi, M.; Naczki, C.; Koritzinsky, M.; Fels, D.; Blais, J.; Hu, N.; Harding, H.; Novoa, I.; Varia, M.; Raleigh, J.; et al. ER stress-regulated translation increases tolerance to extreme hypoxia and promotes tumor growth. EMBO J. 2005, 24, 3470–3481. [Google Scholar] [CrossRef] [PubMed]
- Dey, S.; Sayers, C.M.; Verginadis, I.I.; Lehman, S.L.; Cheng, Y.; Cerniglia, G.J.; Tuttle, S.W.; Feldman, M.D.; Zhang, P.J.; Fuchs, S.Y.; et al. ATF4-dependent induction of heme oxygenase 1 prevents anoikis and promotes metastasis. J. Clin. Investig. 2015, 125, 2592–2608. [Google Scholar] [CrossRef] [PubMed]
- Igarashi, T.; Izumi, H.; Uchiumi, T.; Nishio, K.; Arao, T.; Tanabe, M.; Uramoto, H.; Sugio, K.; Yasumoto, K.; Sasaguri, Y.; et al. Clock and ATF4 transcription system regulates drug resistance in human cancer cell lines. Oncogene 2007, 26, 4749–4760. [Google Scholar] [CrossRef] [PubMed]
- Palam, L.R.; Gore, J.; Craven, K.E.; Wilson, J.L.; Korc, M. Integrated stress response is critical for gemcitabine resistance in pancreatic ductal adenocarcinoma. Cell Death Dis. 2015, 6, e1913. [Google Scholar] [CrossRef] [PubMed]
- Zhu, H.; Chen, X.; Chen, B.; Chen, B.; Fan, J.; Song, W.; Xie, Z.; Jiang, D.; Li, Q.; Zhou, M.; et al. Activating transcription factor 4 mediates a multidrug resistance phenotype of esophageal squamous cell carcinoma cells through transactivation of STAT3 expression. Cancer Lett. 2014, 354, 142–152. [Google Scholar] [CrossRef] [PubMed]
- Baker, B.M.; Nargund, A.M.; Sun, T.; Haynes, C.M. Protective coupling of mitochondrial function and protein synthesis via the eIF2alpha kinase GCN-2. PLoS Genet. 2012, 8, e1002760. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Reyes, I.; Sanchez-Arago, M.J.; Cuezva, M. AMPK and GCN2-ATF4 signal the repression of mitochondria in colon cancer cells. Biochem. J. 2012, 444, 249–259. [Google Scholar] [CrossRef] [PubMed]
- Silva, J.M.; Wong, A.; Carelli, V.; Cortopassi, G.A. Inhibition of mitochondrial function induces an integrated stress response in oligodendroglia. Neurobiol. Dis. 2009, 34, 357–365. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.F.; Chen, M.S.; Chou, Y.C.; Ueng, Y.F.; Yin, P.H.; Yeh, T.S.; Lee, H.C. Mitochondrial dysfunction enhances cisplatin resistance in human gastric cancer cells via the ROS-activated GCN2-eIF2alpha-ATF4-xCT pathway. Oncotarget 2016, 7, 74132–74151. [Google Scholar] [PubMed]
- Boyce, M.; Bryant, K.F.; Jousse, C.; Long, K.; Harding, H.P.; Scheuner, D.; Kaufman, R.J.; Ma, D.; Coen, D.M.; Ron, D.; et al. A selective inhibitor of eIF2alpha dephosphorylation protects cells from ER stress. Science 2005, 307, 935–939. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Chen, X.A.; Hu, J.; Jiang, J.K.; Li, Y.; Chan-Salis, K.Y.; Gu, Y.; Chen, G.; Thomas, C.; Pugh, B.F.; et al. ATF4 gene network mediates cellular response to the anticancer PAD inhibitor YW3-56 in triple-negative breast cancer cells. Mol. Cancer Ther. 2015, 14, 877–888. [Google Scholar] [CrossRef] [PubMed]
- Cunard, R. Mammalian tribbles homologs at the crossroads of endoplasmic reticulum stress and mammalian target of rapamycin pathways. Scientifica (Cairo) 2013, 2013, 750871. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Lv, S.; Qin, Y.; Shu, F.; Xu, Y.; Chen, J.; Xu, B.E.; Sun, X.; Wu, J. TRB3 interacts with CtIP and is overexpressed in certain cancers. Biochim. Biophys. Acta 2007, 1770, 273–278. [Google Scholar] [CrossRef] [PubMed]
- Hegedus, Z.; Czibula, A.; Kiss-Toth, E. Tribbles: A family of kinase-like proteins with potent signalling regulatory function. Cell Signal. 2007, 19, 238–250. [Google Scholar] [CrossRef] [PubMed]
- Yokoyama, T.; Nakamura, T. Tribbles in disease: Signaling pathways important for cellular function and neoplastic transformation. Cancer Sci. 2011, 102, 1115–1122. [Google Scholar] [CrossRef] [PubMed]
- Ohoka, N.; Yoshii, S.; Hattori, T.; Onozaki, K.; Hayashi, H. TRB3, a novel ER stress-inducible gene, is induced via ATF4-CHOP pathway and is involved in cell death. EMBO J. 2005, 24, 1243–1255. [Google Scholar] [CrossRef] [PubMed]
- Maines, M.D. Heme oxygenase: Function, multiplicity, regulatory mechanisms, and clinical applications. FASEB J. 1988, 2, 2557–2568. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, T.; Takano, N.; Ishiwata, K.; Ohmura, M.; Nagahata, Y.; Matsuura, T.; Kamata, A.; Sakamoto, K.; Nakanishi, T.; Kubo, A.; et al. Reduced methylation of PFKFB3 in cancer cells shunts glucose towards the pentose phosphate pathway. Nat. Commun. 2014, 5, 3480. [Google Scholar] [CrossRef] [PubMed]
- Hjortso, M.D.; Andersen, M.H. The expression, function and targeting of haem oxygenase-1 in cancer. Curr. Cancer Drug Targets 2014, 14, 337–347. [Google Scholar] [CrossRef] [PubMed]
- Chau, L.Y. Heme oxygenase-1: Emerging target of cancer therapy. J. Biomed. Sci. 2015, 22, 22. [Google Scholar] [CrossRef] [PubMed]
- Kongpetch, S.; Kukongviriyapan, V.; Prawan, A.; Senggunprai, L.; Kukongviriyapan, U.; Buranrat, B. Crucial role of heme oxygenase-1 on the sensitivity of cholangiocarcinoma cells to chemotherapeutic agents. PLoS ONE 2012, 7, e34994. [Google Scholar] [CrossRef] [PubMed]
- Berberat, P.O.; Dambrauskas, Z.; Gulbinas, A.; Giese, T.; Giese, N.; Kunzli, B.; Autschbach, F.; Meuer, S.; Büchler, M.W.; Friess, H. Inhibition of heme oxygenase-1 increases responsiveness of pancreatic cancer cells to anticancer treatment. Clin. Cancer Res. 2005, 11, 3790–3798. [Google Scholar] [CrossRef] [PubMed]
- Balsa-Martinez, E.; Puigserver, P. Cancer cells hijack gluconeogenic enzymes to fuel cell growth. Mol. Cell 2015, 60, 509–511. [Google Scholar] [CrossRef] [PubMed]
- Leithner, K. PEPCK in cancer cell starvation. Oncoscience 2015, 2, 805–806. [Google Scholar] [PubMed]
- Leithner, K.; Hrzenjak, A.; Trotzmuller, M.; Moustafa, T.; Kofeler, H.C.; Wohlkoenig, C.; Stacher, E.; Lindenmann, J.; Harris, A.L.; Olschewski, A.; et al. PCK2 activation mediates an adaptive response to glucose depletion in lung cancer. Oncogene 2015, 34, 1044–1050. [Google Scholar] [CrossRef] [PubMed]
- Matassa, D.S.; Amoroso, M.R.; Lu, H.; Avolio, R.; Arzeni, D.; Procaccini, C.; Faicchia, D.; Maddalena, F.; Simeon, V.; Agliarulo, I.; et al. Oxidative metabolism drives inflammation-induced platinum resistance in human ovarian cancer. Cell Death Differ. 2016, 23, 1542–1554. [Google Scholar] [CrossRef] [PubMed]
- Chillaron, J.; Roca, R.; Valencia, A.; Zorzano, A.; Palacin, M. Heteromeric amino acid transporters: Biochemistry, genetics, and physiology. Am. J. Physiol. Renal Physiol. 2001, 281, F995–F1018. [Google Scholar] [CrossRef] [PubMed]
- Bannai, S. Exchange of cystine and glutamate across plasma membrane of human fibroblasts. J. Biol. Chem. 1986, 261, 2256–2263. [Google Scholar] [PubMed]
- Sato, H.; Nomura, S.; Maebara, K.; Sato, K.; Tamba, M.; Bannai, S. Transcriptional control of cystine/glutamate transporter gene by amino acid deprivation. Biochem. Biophys. Res. Commun. 2004, 325, 109–116. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.H.; Kuo, M.T. Role of glutathione in the regulation of cisplatin resistance in cancer chemotherapy. Met. Based Drugs 2010, 2010, 430939. [Google Scholar] [CrossRef] [PubMed]
- Ishikawa, T.; Wright, C.D.; Ishizuka, H. GS-X pump is functionally overexpressed in cis-diamminedichloroplatinum (II)-resistant human leukemia HL-60 cells and down-regulated by cell differentiation. J. Biol. Chem. 1994, 269, 29085–29093. [Google Scholar] [PubMed]
- Jansen, B.A.; Brouwer, J.; Reedijk, J. Glutathione induces cellular resistance against cationic dinuclear platinum anticancer drugs. J. Inorg. Biochem. 2002, 89, 197–202. [Google Scholar] [CrossRef]
- Meijer, C.; Mulder, N.H.; Timmer-Bosscha, H.; Sluiter, W.J.; Meersma, G.J.; de Vries, E.G. Relationship of cellular glutathione to the cytotoxicity and resistance of seven platinum compounds. Cancer Res. 1992, 52, 6885–6889. [Google Scholar] [PubMed]
- Lopes-Coelho, F.; Gouveia-Fernandes, S.; Gonçalves, L.G.; Nunes, C.; Faustino, I.; Silva, F.; Félix, A.; Pereira, S.A.; Serpa, J. HNF1β drives glutathione (GSH) synthesis underlying intrinsic carboplatin resistance of ovarian clear cell carcinoma (OCCC). Tumour Biol. 2016, 37, 4813–4829. [Google Scholar] [CrossRef] [PubMed]
- Szász, A.M.; Lánczky, A.; Nagy, Á.; Förster, S.; Hark, K.; Green, J.E.; Boussioutas, A.; Busuttil, R.; Szabó, A.; Győrffy, B. Cross-validation of survival associated biomarkers in gastric cancer using transcriptomic data of 1065 patients. Oncotarget 2016, 7, 49322–49333. [Google Scholar] [CrossRef] [PubMed]
| ATF4-Regulated Genes | Progression Free Survival | Overall Survival |
|---|---|---|
| High vs. Low Expression | High vs. Low Expression | |
| (Hazard Ratio, HR) | (Hazard Ratio, HR) | |
| TRB3 | 2.57 (1.69–3.9) | 2.55 (1.67–3.89) |
| Logrank p = 5.3 × 10−6 | Logrank p = 8.9 × 10−6 | |
| (n = 110 vs. 43) | (n = 110 vs. 43) | |
| HO-1 | 1.55 (1.04–2.3) | 1.74 (1.15–2.63) |
| Logrank p = 0.028 | Logrank p = 0.008 | |
| (n = 111 vs. 42) | (n = 111 vs. 42) | |
| PCK2 | 1.67 (1.15–2.42) | 1.84 (1.26–2.69) |
| Logrank p = 0.0066 | Logrank p = 0.0013 | |
| (n = 102 vs. 51) | (n = 43 vs. 110) | |
| xCT | 1.43 (1.01–2.02) | 1.48 (1.04–2.11) |
| Logrank p = 0.043 | Logrank p = 0.027 | |
| (n = 71 vs. 82) | (n = 82 vs. 71) |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, S.-F.; Wung, C.-H.; Chen, M.-S.; Chen, C.-F.; Yin, P.-H.; Yeh, T.-S.; Chang, Y.-L.; Chou, Y.-C.; Hung, H.-H.; Lee, H.-C. Activated Integrated Stress Response Induced by Salubrinal Promotes Cisplatin Resistance in Human Gastric Cancer Cells via Enhanced xCT Expression and Glutathione Biosynthesis. Int. J. Mol. Sci. 2018, 19, 3389. https://doi.org/10.3390/ijms19113389
Wang S-F, Wung C-H, Chen M-S, Chen C-F, Yin P-H, Yeh T-S, Chang Y-L, Chou Y-C, Hung H-H, Lee H-C. Activated Integrated Stress Response Induced by Salubrinal Promotes Cisplatin Resistance in Human Gastric Cancer Cells via Enhanced xCT Expression and Glutathione Biosynthesis. International Journal of Molecular Sciences. 2018; 19(11):3389. https://doi.org/10.3390/ijms19113389
Chicago/Turabian StyleWang, Sheng-Fan, Chih-Hsuan Wung, Meng-Shian Chen, Chian-Feng Chen, Pen-Hui Yin, Tien-Shun Yeh, Yuh-Lih Chang, Yueh-Ching Chou, Hung-Hsu Hung, and Hsin-Chen Lee. 2018. "Activated Integrated Stress Response Induced by Salubrinal Promotes Cisplatin Resistance in Human Gastric Cancer Cells via Enhanced xCT Expression and Glutathione Biosynthesis" International Journal of Molecular Sciences 19, no. 11: 3389. https://doi.org/10.3390/ijms19113389
APA StyleWang, S.-F., Wung, C.-H., Chen, M.-S., Chen, C.-F., Yin, P.-H., Yeh, T.-S., Chang, Y.-L., Chou, Y.-C., Hung, H.-H., & Lee, H.-C. (2018). Activated Integrated Stress Response Induced by Salubrinal Promotes Cisplatin Resistance in Human Gastric Cancer Cells via Enhanced xCT Expression and Glutathione Biosynthesis. International Journal of Molecular Sciences, 19(11), 3389. https://doi.org/10.3390/ijms19113389