Prodrug Strategies for Paclitaxel
Abstract
:1. Introduction
2. Structure and Activity Relationships
3. Small Molecule Paclitaxel Prodrug
3.1. Non-Targeting Modification
3.1.1. Hydrophilic Modification
Skeletal Migration
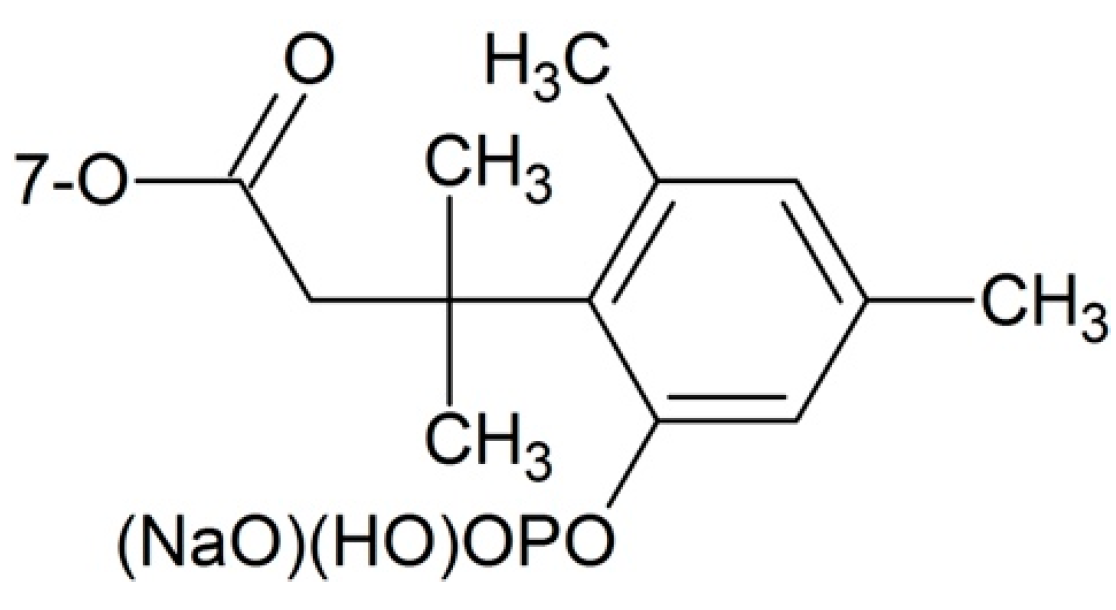
Phosphate Esters
Others
3.1.2. Hydrophobic Modification
Silicate Esters
Squalenoylation Technology
3.1.3. Mutual Drugs
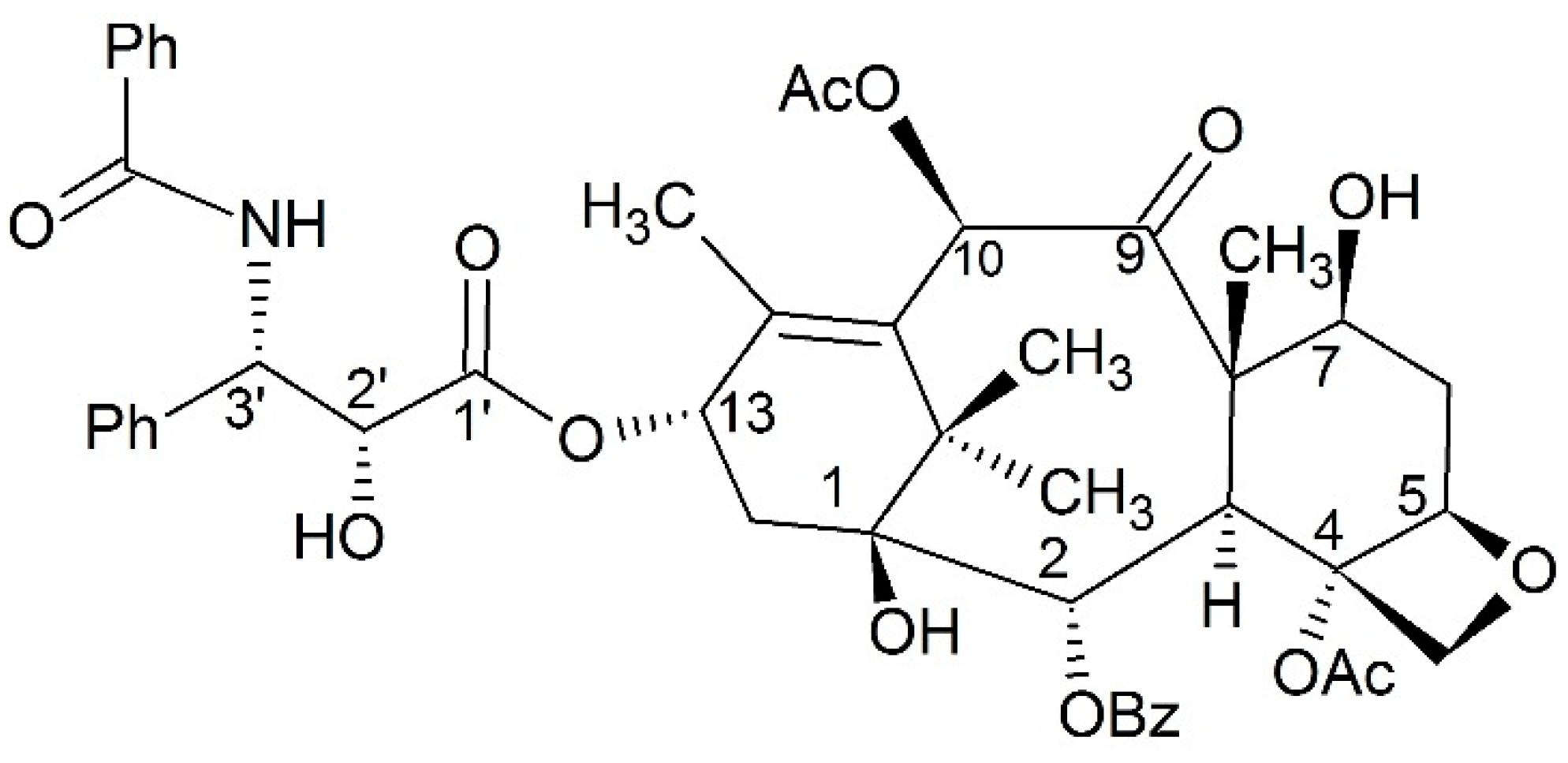
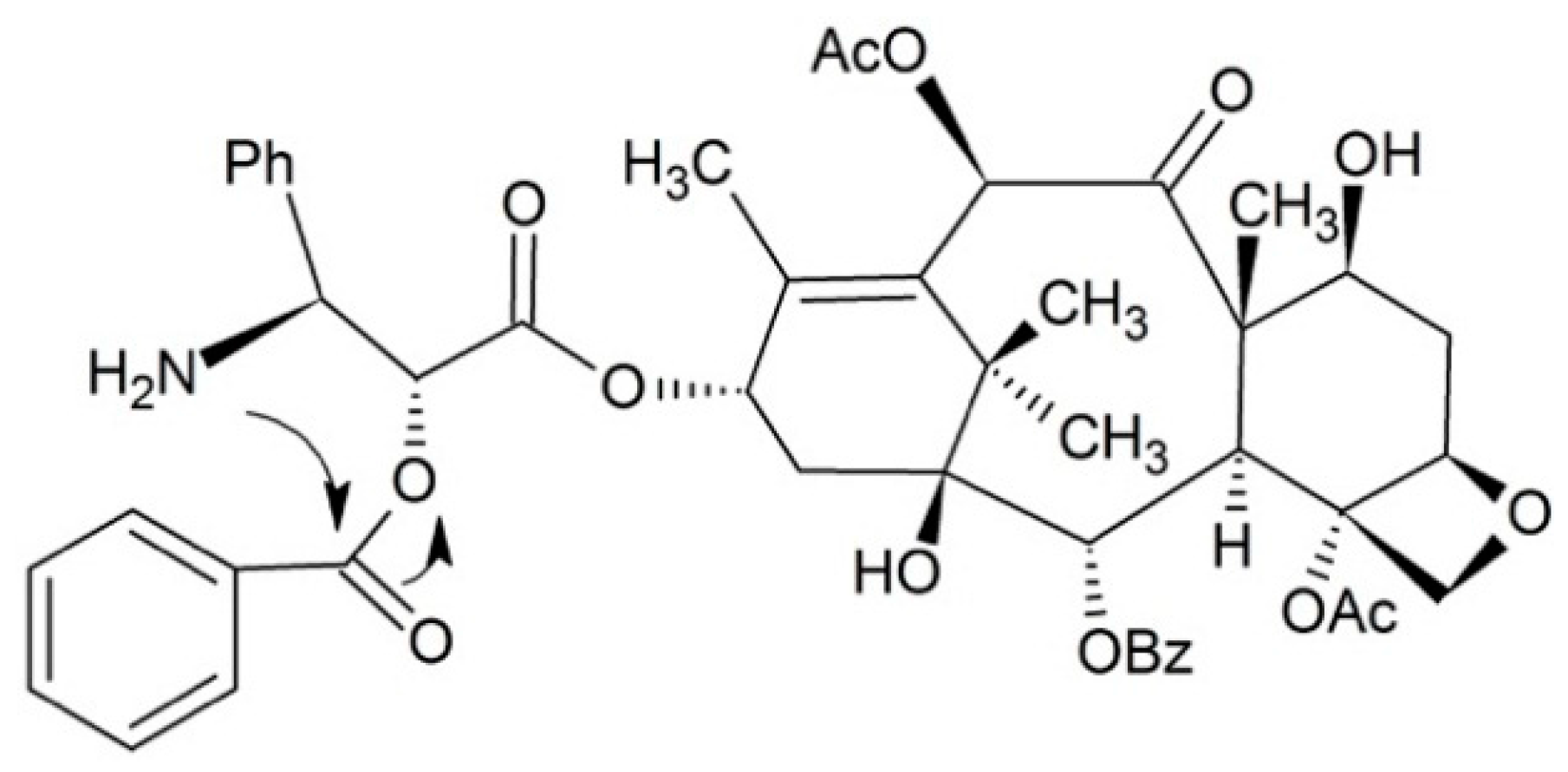
3.1.4. Dipeptide Prodrugs
3.1.5. Others
3.2. Targeting Modification
3.2.1. Targeting Tumor Overexpressed Enzymes
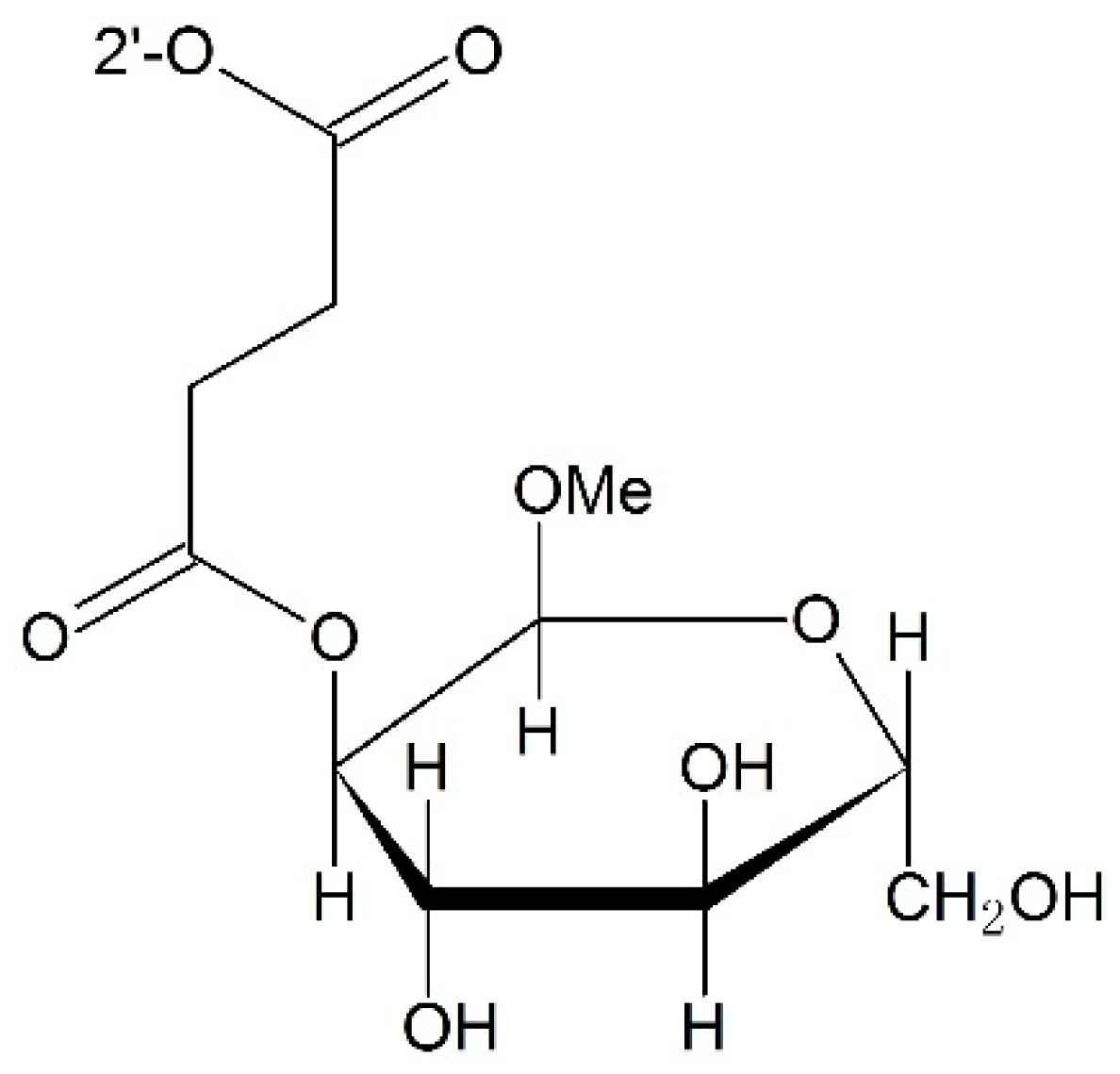
Targeting β-d-Glucuronidase
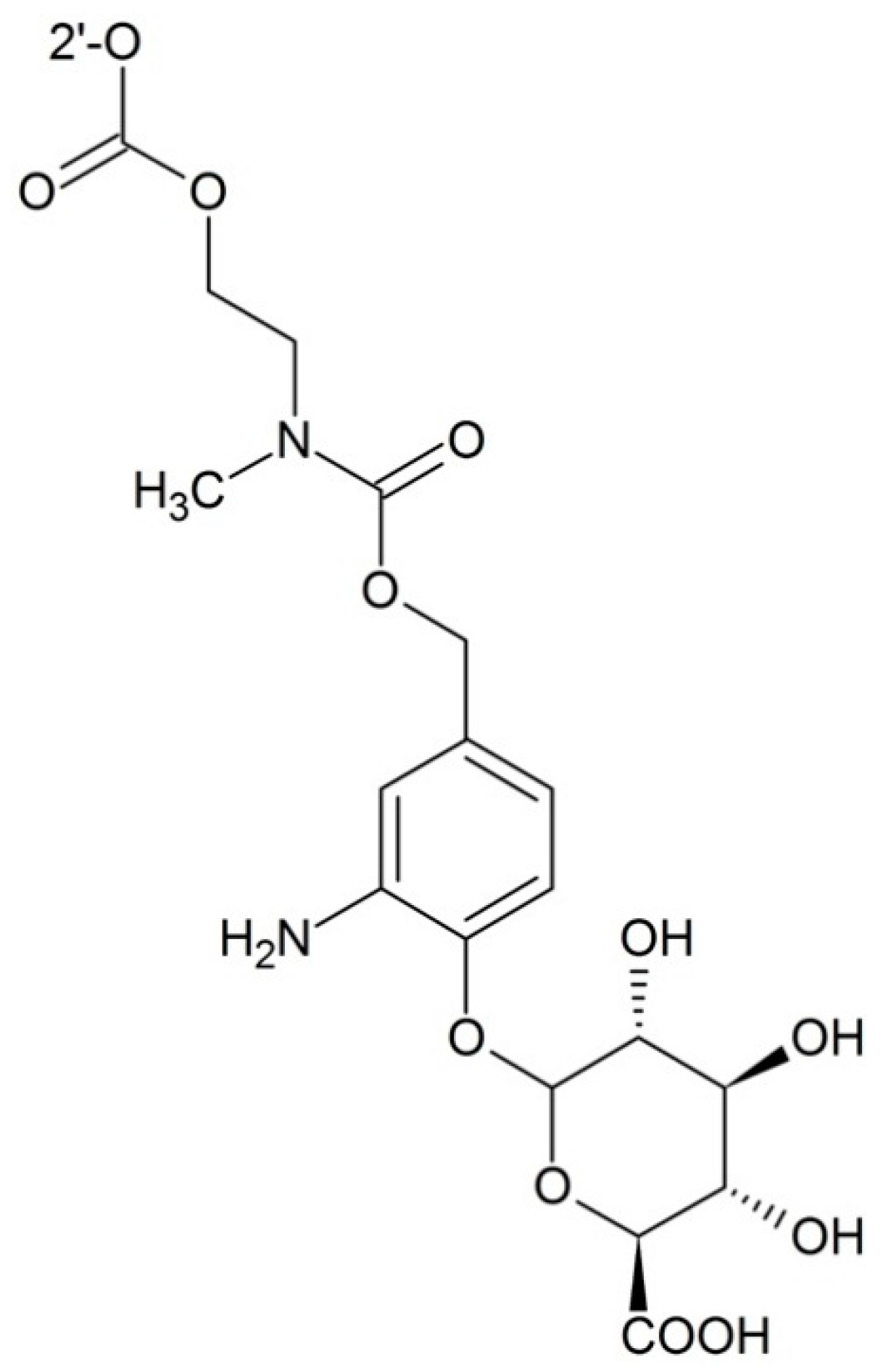
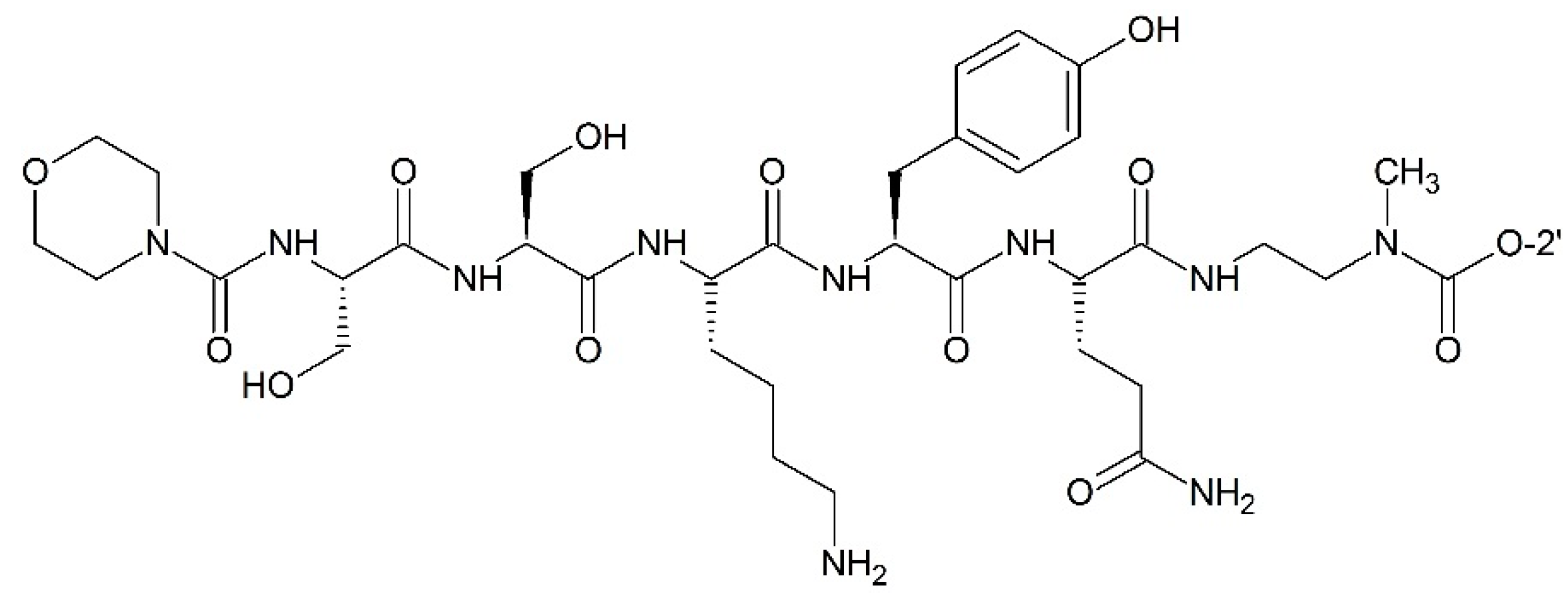
Targeting Prostate-Specific Antigen (PSA)
3.2.2. Targeting Transporters
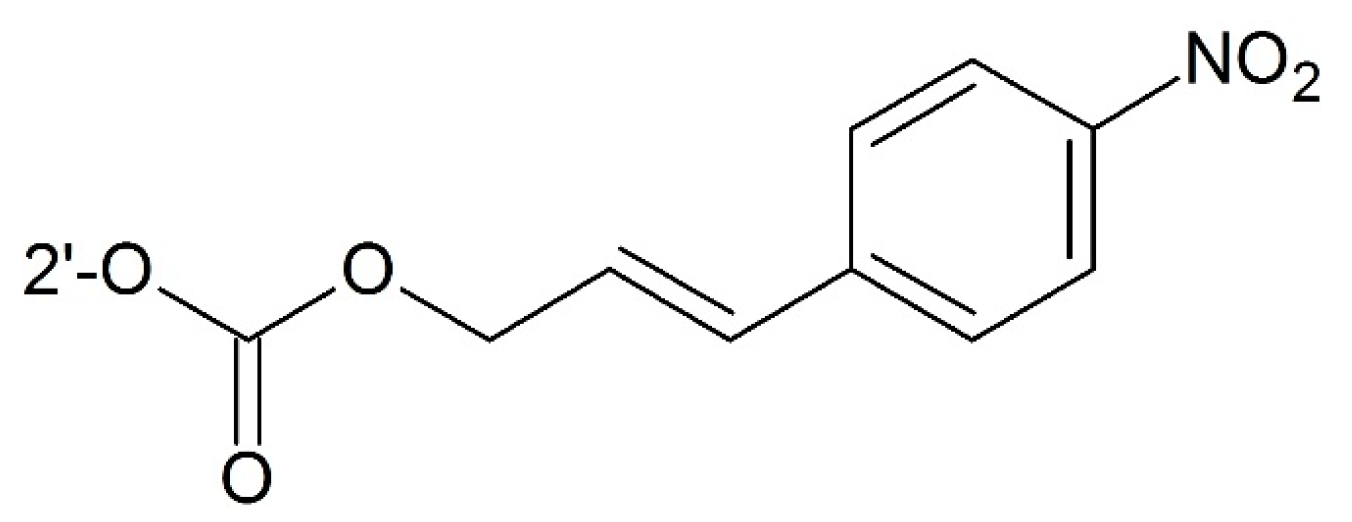
3.2.3. Targeting Hypoxia
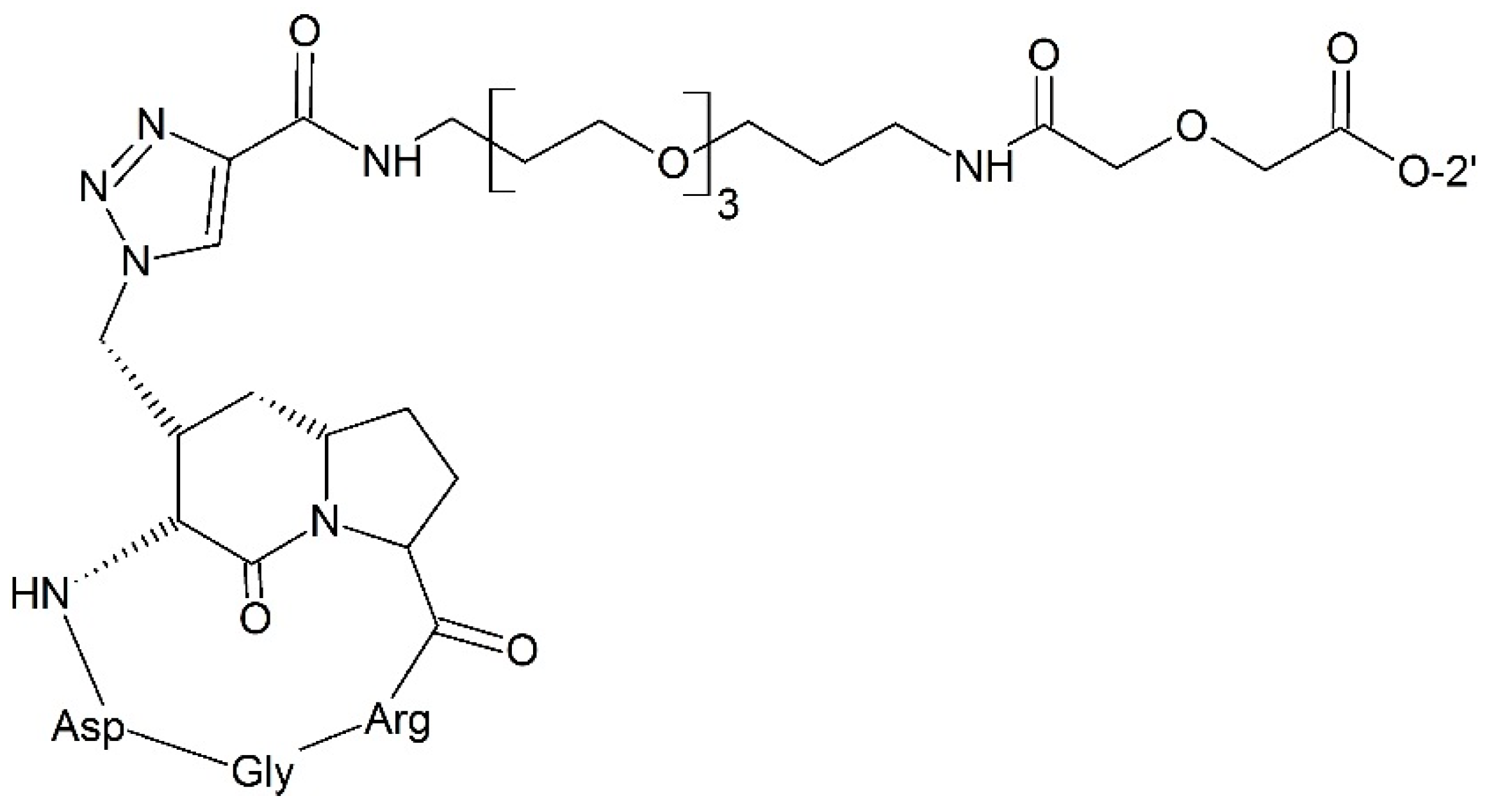
3.2.4. Targeting Integrin
3.2.5. Targeting Receptors
3.2.6. Targeting Glutathione
3.2.7. Prodrugs for Antibody Directed Enzyme Prodrug Therapy (ADEPT)
3.2.8. Prodrugs for Gene-Directed Enzyme Prodrug Therapy (GDEPT)
4. Macromolecular Prodrugs
4.1. Polyethylene Glycol (PEG)
4.1.1. PEG as Drug Carriers
4.1.2. PEG Copolymer Prodrugs
4.1.3. PEG Linker Prodrugs
4.2. Hyaluronic Acid (HA)
4.3. N-(2-Hydroxypropyl)methacrylamide (HPMA)
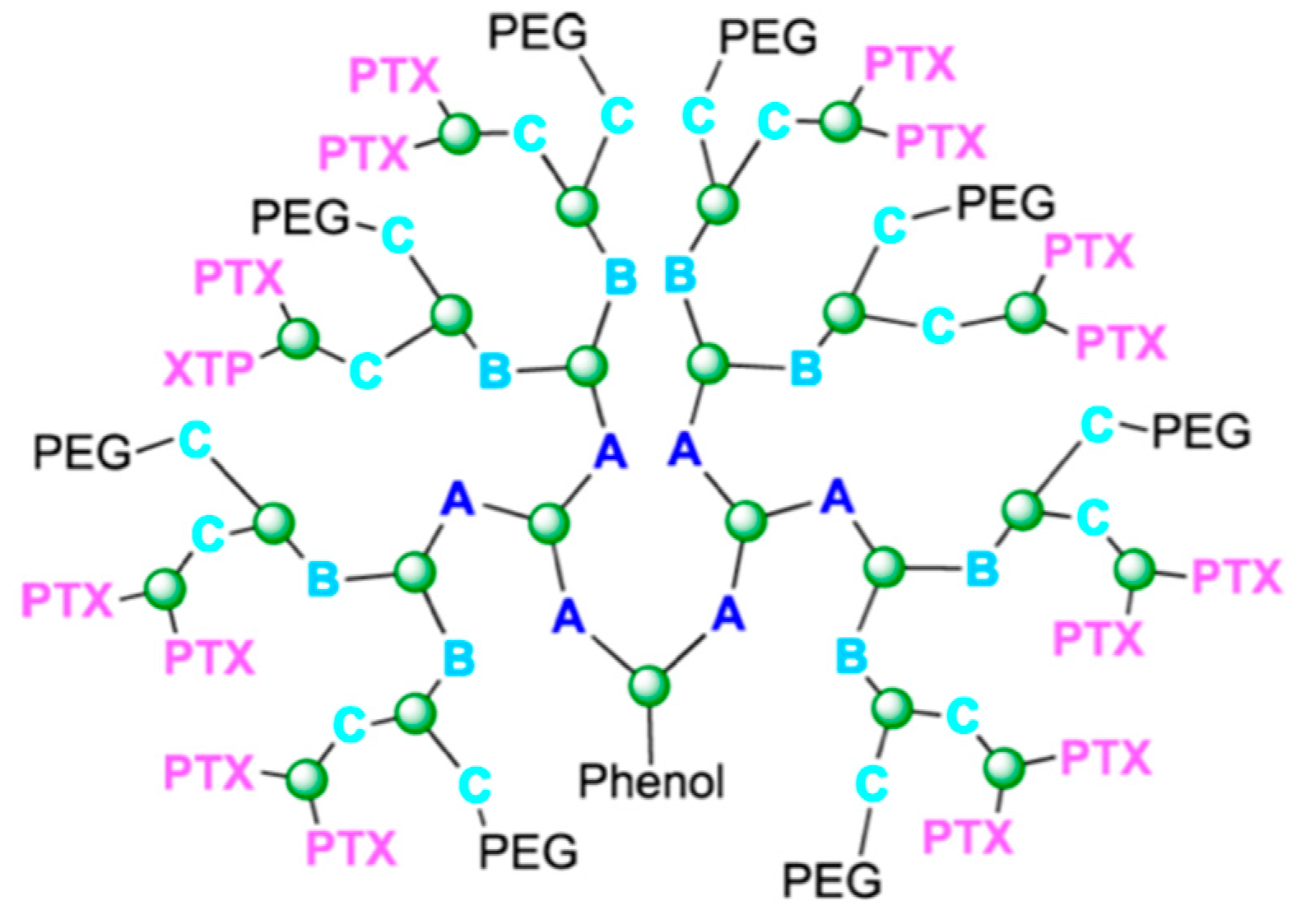
4.4. Dendrimers
4.5. Other Polymers
4.6. Proteins
5. Nanodevices
6. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Wani, M.C.; Taylor, H.L.; Wall, M.E.; Coggon, P.; McPhail, A.T. Plant antitumor agents. VI. Isolation and structure of taxol, a novel antileukemic and antitumor agent from Taxus brevifolia. J. Am. Chem. Soc. 1971, 93, 2325–2327. [Google Scholar] [CrossRef] [PubMed]
- De Furia, M.D. Paclitaxel (Taxol®): A new natural product with major anticancer activity. Phytomedicine 1997, 4, 273–282. [Google Scholar] [CrossRef]
- MacCari, G.; Mori, M.; Rodríguez-Salarichs, J.; Fang, W.; Díaz, J.F.; Botta, M. Free energy profile and kinetics studies of paclitaxel internalization from the outer to the inner wall of microtubules. J. Chem. Theory Comput. 2013, 9, 698–706. [Google Scholar] [CrossRef] [PubMed]
- Grover, S.; Rimoldis, J.M.; Molinero, A.A.; Chaudhary, A.G.; Kingston, D.G.; Hamel, E. Different effects of paclitaxel (Taxel) analogs modified at position C-2, C-7, and C-3′ on tubulin polymerization and polymer stabilization: Identification of a hyperactive paclitaxel derivative. Biochemistry 1995, 34, 3927–3934. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Edsall, R.; Jagtap, P.G.; Kingston, D.G.I.; Bane, S. Equilibrium studies of a fluorescent paclitaxel derivative binding to microtubules. Biochemistry 2000, 39, 616–623. [Google Scholar] [CrossRef] [PubMed]
- Javeed, A.; Ashraf, M.; Riaz, A.; Ghafoor, A.; Afzal, S.; Mahmood, M. Paclitaxel and immune system. Eur. J. Pharm. Sci. 2009, 38, 283–290. [Google Scholar] [CrossRef] [PubMed]
- Brunden, K.R.; Trojanowski, J.Q.; Smith, A.B.; Lee, V.M.Y.; Ballatore, C. Microtubule-stabilizing agents as potential therapeutics for neurodegenerative disease. Bioorg. Med. Chem. 2014, 22, 5040–5049. [Google Scholar] [CrossRef] [PubMed]
- Dadgar, S.; Ramjan, Z.; Floriano, W.B. Paclitaxel is an inhibitor and its boron dipyrromethene derivative is a fluorescent recognition agent for botulinum neurotoxin subtype A. J. Med. Chem. 2013, 56, 2791–2803. [Google Scholar] [CrossRef] [PubMed]
- Ooya, T.; Lee, J.; Park, K. Hydrotropic dendrimers of generations 4 and 5: Synthesis, characterization, and hydrotropic solubilization of paclitaxel. Bioconjug. Chem. 2004, 15, 1221–1229. [Google Scholar] [CrossRef] [PubMed]
- Menger, F.M.; Zhang, H.; de Joannis, J.; Kindt, J.T. Solubilization of Paclitaxel (taxol) by peptoad self-assemblies. Langmuir 2007, 23, 2308–2310. [Google Scholar] [CrossRef] [PubMed]
- Inada, A.; Oshima, T.; Baba, Y. Enhancing the water dispersibility of paclitaxel by complexation with hydrophobic peptides. Colloids Surf. B Biointerfaces 2015, 135, 408–415. [Google Scholar] [CrossRef] [PubMed]
- Nanda, R.; Sasmal, A.; Nayak, P.L. Preparation and characterization of chitosan–polylactide composites blended with Cloisite 30B for control release of the anticancer drug paclitaxel. Carbohydr. Polym. 2011, 83, 988–994. [Google Scholar] [CrossRef]
- Aleandri, S.; Bandera, D.; Mezzenga, R.; Landau, E.M. Biotinylated cubosomes: A versatile tool for active targeting and co-delivery of paclitaxel and fluorescein-based lipid dye. Langmuir 2015, 108, 12770–12776. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Chen, G.-S.; Chen, Y.; Cao, D.-X.; Ge, Z.-Q.; Yuan, Y.-J. Inclusion complexes of paclitaxel and oligo(ethylenediamino) bridged bis(β-cyclodextrin)s: Solubilization and antitumor activity. Bioorg. Med. Chem. 2004, 12, 5767–5775. [Google Scholar] [CrossRef] [PubMed]
- Boztas, A.O.; Karakuzu, O.; Galante, G.; Ugur, Z.; Kocabas, F.; Altuntas, C.Z.; Yazaydin, A.O. Synergistic interaction of paclitaxel and curcumin with cyclodextrin polymer complexation in human cancer cells. Mol. Pharm. 2013, 10, 2676–2683. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.V.; Batuwangala, M.; Mundra, R.; Mehta, K.; Patke, S.; Falletta, E.; Patil, R.; Gade, W.N. Biomineralized anisotropic gold microplate-macrophage interactions reveal frustrated phagocytosis-like phenomenon: A novel paclitaxel drug delivery vehicle. ACS Appl. Mater. Interfaces 2014, 6, 14679–14689. [Google Scholar] [CrossRef] [PubMed]
- Ren, F.; Bhana, S.; Norman, D.D.; Johnson, J.; Xu, L.; Baker, D.L.; Parrill, A.L.; Huang, X. Gold nanorods carrying paclitaxel for photothermal-chemotherapy of cancer. Bioconjug. Chem. 2013, 24, 376–386. [Google Scholar] [CrossRef] [PubMed]
- Joshi, N.; Saha, R.; Shanmugam, T.; Balakrishnan, B.; More, P.; Banerjee, R. Carboxymethyl-chitosan-tethered lipid vesicles: Hybrid nanoblanket for oral delivery of paclitaxel. Biomacromolecules 2013, 14, 2272–2282. [Google Scholar] [CrossRef] [PubMed]
- Kang, Y.; Wu, J.; Yin, G.; Huang, Z.; Liao, X.; Yao, Y.; Ouyang, P.; Wang, H.; Yang, Q. Characterization and biological evaluation of paclitaxel-loaded poly(l-lactic acid) microparticles prepared by supercritical CO2. Langmuir 2008, 24, 7432–7441. [Google Scholar] [CrossRef] [PubMed]
- Sahoo, S.K.; Labhasetwar, V. Enhanced antiproliferative activity of nanoparticles, transferrin-conjugated paclitaxel-loaded. Mol. Pharm. 2005, 2, 373–383. [Google Scholar] [CrossRef] [PubMed]
- Zhang, F.; Zhang, S.; Pollack, S.F.; Li, R.; Gonzalez, A.M.; Fan, J.; Zou, J.; Leininger, S.E.; Pavía-Sanders, A.; Johnson, R.; et al. Improving Paclitaxel Delivery: In Vitro and In Vivo characterization of PEGylated polyphosphoester-based nanocarriers. J. Am. Chem. Soc. 2015, 137, 2056–2066. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.; van Nostrum, C.F.; Hennink, W.E. Interfacially Hydrazone cross-linked thermosensitive polymeric micelles for acid-triggered release of paclitaxel. ACS Biomater. Sci. Eng. 2015, 1, 393–404. [Google Scholar] [CrossRef]
- Zhang, X.; Huang, Y.; Zhao, W.; Chen, Y.; Zhang, P.; Li, J.; Venkataramanan, R.; Li, S. PEG-farnesyl thiosalicylic acid telodendrimer micelles as an improved formulation for targeted delivery of paclitaxel. Mol. Pharm. 2014, 11, 2807–2814. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Lu, J.; Huang, Y.; Zhao, W.; Chen, Y.; Li, J.; Gao, X.; Venkataramanan, R.; Sun, M.; Stolz, D.B.; et al. PEG-farnesylthiosalicylate conjugate as a nanomicellar carrier for delivery of paclitaxel. Bioconjug. Chem. 2013, 24, 464–472. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Liu, K.; Huang, Y.; Xu, J.; Li, J.; Ma, X.; Li, S. Reduction-sensitive dual functional nanomicelles for improved delivery of Paclitaxel. Bioconjug. Chem. 2014, 25, 1689–1696. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Wang, X.; Shu, X.; Shen, Z.; Sun, R.C. Self-assembly and paclitaxel loading capacity of cellulose-grazft-poly(lactide) nanomicelles. J. Agric. Food Chem. 2012, 60, 3900–3908. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Mei, L.; Yu, Q.; Xu, C.; Qiu, Y.; Yang, Y.; Shi, K.; Zhang, Q.; Gao, H.; Zhang, Z.; et al. Multifunctional Tandem peptide modified paclitaxel-loaded liposomes for the treatment of vasculogenic mimicry and cancer stem cells in malignant glioma. ACS Appl. Mater. Interfaces 2015, 7, 16792–16801. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Huang, L.; Liu, F. Paclitaxel nanocrystals for overcoming multidrug resistance in cancer. Mol. Pharm. 2010, 7, 863–869. [Google Scholar] [CrossRef] [PubMed]
- Field, J.J.; Kanakkanthara, A.; Miller, J.H. Microtubule-targeting agents are clinically successful due to both mitotic and interphase impairment of microtubule function. Bioorg. Med. Chem. 2014, 22, 5050–5059. [Google Scholar] [CrossRef] [PubMed]
- Jiang, S.; Pan, A.W.; Lin, T.; Zhang, H.; Malfatti, M.; Turteltaub, K.; Henderson, P.T.; Pan, C. Paclitaxel enhances carboplatin-DNA adduct formation and cytotoxicity. Chem. Res. Toxicol. 2015, 28, 2250–2252. [Google Scholar] [CrossRef] [PubMed]
- Andreoli, M.; Persico, M.; Kumar, A.; Orteca, N.; Kumar, V.; Pepe, A.; Mahalingam, S.; Alegria, A.E.; Petrella, L.; Sevciunaite, L.; et al. Identification of the first inhibitor of the GBP1:PIM1 interaction. Implications for the development of a new class of anticancer agents against paclitaxel resistant cancer cells. J. Med. Chem. 2014, 57, 7916–7932. [Google Scholar] [CrossRef] [PubMed]
- Dai, X.; Fan, W.; Wang, Y.; Huang, L.; Jiang, Y.; Shi, L.; Mckinley, D.; Tan, W.; Tan, C. Combined delivery of Let-7b microRNA and Paclitaxel via biodegradable nanoassemblies for the treatment of KRAS mutant cancer. Mol. Pharm. 2016, 13, 520–533. [Google Scholar] [CrossRef] [PubMed]
- Shen, J.; Meng, Q.; Sui, H.; Yin, Q.; Zhang, Z.; Yu, H.; Li, Y. IRGD conjugated TPGS mediates codelivery of paclitaxel and survivin shRNA for the reversal of lung cancer resistance. Mol. Pharm. 2014, 11, 2579–2591. [Google Scholar] [CrossRef] [PubMed]
- Sun, T.-M.; Du, J.-Z.; Yao, Y.-D.; Mao, C.-Q.; Dou, S.; Huang, S.-Y.; Zhang, P.-Z.; Leong, K.W.; Song, E.-W.; Wang, J. Simultaneous delivery of siRNA and paclitaxel via a “Two-in-One” micelleplex promotes synergistic tumor suppression. ACS Nano 2011, 5, 1483–1494. [Google Scholar] [CrossRef] [PubMed]
- Damen, E.W.P.; Nevalainen, T.J.; van den Bergh, T.J.M.; de Groot, F.M.H.; Scheeren, H.W. Synthesis of novel paclitaxel prodrugs designed for bioreductive activation in hypoxic tumour tissue. Bioorg. Med. Chem. 2002, 10, 71–77. [Google Scholar] [CrossRef]
- Smith, A.B., III; Sugasawa, K.; Atasoylu, O.; Yang, C.P.H.; Horwitz, S.B. Design and synthesis of (+)-discodermolide-paclitaxel hybrids leading to enhanced biological activity. J. Med. Chem. 2011, 54, 6319–6327. [Google Scholar] [CrossRef] [PubMed]
- Wittman, M.D.; Altstadt, T.J.; Fairchild, C.; Hansel, S.; Johnston, K.; Kadow, J.F.; Long, B.H.; Rose, W.C.; Vyas, D.M.; Wu, M.J.; et al. Synthesis of metabolically blocked paclitaxel analogues. Bioorg. Med. Chem. Lett. 2001, 11, 809–810. [Google Scholar] [CrossRef]
- Ganesh, T.; Yang, C.; Norris, A.; Glass, T.; Bane, S.; Ravindra, R.; Banerjee, A.; Metaferia, B.; Thomas, S.L.; Giannakakou, P.; et al. Evaluation of the tubulin-bound paclitaxel conformation: Synthesis, biology, and SAR studies of C-4 to C-3′ bridged paclitaxel analogues. J. Med. Chem. 2007, 50, 713–725. [Google Scholar] [CrossRef] [PubMed]
- Tang, S.; Yang, C.; Brodie, P.; Bane, S.; Ravindra, R.; Sharma, S.; Jiang, Y.; Snyder, J.P.; Kingston, D.G. Bridging converts a noncytotoxic nor-paclitaxel derivative to a cytotoxic analogue by constraining it to the T-Taxol conformation. Org. Lett. 2006, 8, 3983–3986. [Google Scholar] [CrossRef] [PubMed]
- Shanker, N.; Kingston, D.G.I.; Ganesh, T.; Yang, C.; Alcaraz, A.A.; Geballe, M.T.; Banerjee, A.; McGee, D.; Snyder, J.P.; Bane, S. Enhanced microtubule binding and tubulin assembly properties of conformationally constrained paclitaxel derivatives. Biochemistry 2007, 46, 11514–11527. [Google Scholar] [CrossRef] [PubMed]
- Choy, N.; Shin, Y.; Nguyen, P.Q.; Curran, D.P.; Balachandran, R.; Madiraju, C.; Day, B.W. Simplified Discodermolide Analogues: Synthesis and Biological Evaluation of 4-epi-7-Dehydroxy-14,16-didemethyl-(+)-discodermolides as Microtubule-Stabilizing Agents. J. Med. Chem. 2003, 46, 2846–2864. [Google Scholar] [CrossRef] [PubMed]
- Podduturi, V.P.; Magaña, I.B.; O’Neal, D.P.; Derosa, P.A. Simulation of transport and extravasation of nanoparticles in tumors which exhibit enhanced permeability and retention effect. Comput. Methods Progr. Biomed. 2013, 112, 58–68. [Google Scholar] [CrossRef] [PubMed]
- Ballone, P.; Marchi, M. A density functional study of a new family of anticancer drugs: Paclitaxel, taxotere, epothilone, and discodermolide. J. Phys. Chem. A 1999, 103, 3097–3102. [Google Scholar] [CrossRef]
- Chaudhary, A.G.; Chordia, M.D.; Kingston, D.G.I. A novel benzoyl group migration: Synthesis and biological evaluation of 1-Benzoyl-2-des (benzoy1oxy)paclitaxe. J. Org. Chem. 1996, 60, 3260–3262. [Google Scholar] [CrossRef]
- Zhu, Q.; Huang, N.; Wang, M.; Chu, F. Comparative molecular field analysis of a series of paclitaxel analogs. J. Med. Chem. 1997, 40, 4319–4328. [Google Scholar] [CrossRef] [PubMed]
- Moyna, G.; Williams, H.J.; Scott, A.I.; Ringel, I.; Gorodetsky, R.; Swindell, C.S. Conformational studies of paclitaxel analogs modified at the C-2′ position in hydrophobic and hydrophilic solvent systems. J. Med. Chem. 1997, 40, 3305–3311. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.; Lagisetti, C.; Poliks, B.; Coates, R.M.; Kingston, D.G.I.; Bane, S. Dissecting paclitaxel—Microtubule association: Quantitative assessment of the 2′-OH group. Biochemistry 2013, 52, 2328–2336. [Google Scholar] [CrossRef] [PubMed]
- Milanesio, M.; Ugliengo, P.; Viterbo, D.; Appendino, G. Ab initio conformational study of the phenylisoserine side chain of paclitaxel. J. Med. Chem. 1999, 42, 291–299. [Google Scholar] [CrossRef] [PubMed]
- Altstadt, T.J.; Fairchild, C.R.; Golik, J.; Johnston, K.A.; Kadow, J.F.; Lee, F.Y.; Long, B.H.; Rose, W.C.; Vyas, D.M.; Wong, H.; et al. Synthesis and antitumor activity of novel C-7 paclitaxel ethers: Discovery of BMS-184476. J. Med. Chem. 2001, 44, 4577–4583. [Google Scholar] [CrossRef] [PubMed]
- Khmelnitsky, Y.L.; Budde, C.; Arnold, J.M.; Usyatinsky, A.; Clark, D.S.; Dordick, J.S. Synthesis of water-soluble paclitaxel derivatives by enzymatic acylation. J. Am. Chem. Soc. 1997, 119, 11554–11555. [Google Scholar] [CrossRef]
- Wrasidlo, W.; Gaedicke, G.; Guy, R.K.; Renaud, J.; Pitsinos, E.; Nicolaou, K.C.; Reisfeld, R.A.; Lode, H.N. A novel 2′-(N-methylpyridinium acetate) prodrug of paclitaxel induces superior antitumor responses in preclinical cancer models. Bioconjug. Chem. 2002, 13, 1093–9109. [Google Scholar] [CrossRef] [PubMed]
- Ali, S.M.; Hoemann, M.Z.; Aub, J.; Mitscher, L.A.; Georg, G.I. Novel cytotoxic 3′-(tert-Butyl) 3′-dephenyl analogs of paclitaxel and docetaxel. J. Med. Chem. 1996, 38, 3821–3828. [Google Scholar] [CrossRef]
- Kingston, D.G.; Chaudhary, A.G.; Chordia, M.D.; Gharpure, M.; Gunatilaka, A.L.; Higgs, P.I.; Rimoldi, J.M.; Samala, L.; Jagtap, P.G.; Giannakakou, P.; et al. Synthesis and biological evaluation of 2-acyl analogues of paclitaxel (Taxol). J. Med. Chem. 1998, 41, 3715–3726. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Kadow, J.F.; Farinat, V.; Fairchild, C.R.; Johnston, K.A.; Med, I. First Syntheses of novel paclitaxel(taxol) analogs modified at C4-position. J. Org. Chem. 1994, 59, 6156–6158. [Google Scholar] [CrossRef]
- Barboni, L.; Giarlo, G.; Ricciutelli, M.; Ballini, R.; Georg, G.I.; VanderVelde, D.G.; Himes, R.H.; Wang, M.; Lakdawala, A.; Snyder, J.P.; et al. Synthesis, modeling, and anti-tubulin activity of a d-Seco paclitaxel analogue. Org. Lett. 2004, 6, 461–464. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Fairchild, C.; Lon, B.H. Synthesis and biological evaluation of novel C-4 aziridine-bearing paclitaxel (Taxol) analogs. J. Med. Chem. 1995, 38, 2263–2267. [Google Scholar] [CrossRef] [PubMed]
- Walker, M.A.; Johnson, T.D.; Huang, S.; Vyas, D.M.; Kadow, J.F. Synthesis of a novel C-10 spiro-epoxide of paclitaxel. Bioorg. Med. Chem. Lett. 2001, 11, 1683–1685. [Google Scholar] [CrossRef]
- Chen, S.-H.; Farina, V.; Vyas, D.M.; Doyle, T.W.; Long, B.H.; Fairchild, C. Synthesis and biological evaluation of C-13 amide-linked paclitaxel (Taxol) analogs. J. Org. Chem. 1996, 61, 2065–2070. [Google Scholar] [CrossRef]
- Gunatilaka, A.A.L.; Ramdayal, F.D.; Sarragiotto, M.H.; Kingston, D.G.I.; Sackett, D.L.; Hamel, E. Synthesis and biological evaluation of novel paclitaxel (Taxol) d-ring modified analogues. J. Org. Chem. 1999, 64, 2694–2703. [Google Scholar] [CrossRef] [PubMed]
- Metaferia, B.B.; Hoch, J.; Glass, T.E.; Bane, S.L.; Chatterjee, S.K.; Snyder, J.P.; Lakdawala, A.; Cornett, B.; Kingston, D.G.I. Synthesis and biological evaluation of novel macrocyclic paclitaxel analogues. Org. Lett. 2001, 3, 2461–2464. [Google Scholar] [CrossRef] [PubMed]
- Hayashi, Y.; Skwarczynski, M.; Hamada, Y.; Sohma, Y.; Kimura, T.; Kiso, Y. A novel approach of water-soluble paclitaxel prodrug with no auxiliary and no byproduct: Design and synthesis of isotaxel. J. Med. Chem. 2003, 46, 3782–3784. [Google Scholar] [CrossRef] [PubMed]
- Golik, J.; Wong, H.S.L.; Chen, S.H.; Doyle, T.W.; Wright, J.J.K.; Knipe, J.; Rose, W.C.; Casazza, A.M.; Vyas, D.M. Synthesis and antitumor evaluation of paclitaxel phosphonooxymethyl ethers: A Novel class of water soluble paclitaxel pro-drugs. Bioorg. Med. Chem. Lett. 1996, 6, 1837–1842. [Google Scholar] [CrossRef]
- Ueda, Y.; Matiskella, J.D.; Mikkilinenit, A.B.; Farinal, V.; Knipe, J.O.; Roset, W.C.; Casazzat, A.M.; Vyas, D.M. Novel, water-soluble phosphate derivatives of 2′-ethoxy carbonylpaclitaxel as potential prodrugs of paclitaxel: Synthesis and antitumor evaluation. Bioorg. Med. Chem. Lett. 1995, 5, 247–252. [Google Scholar] [CrossRef]
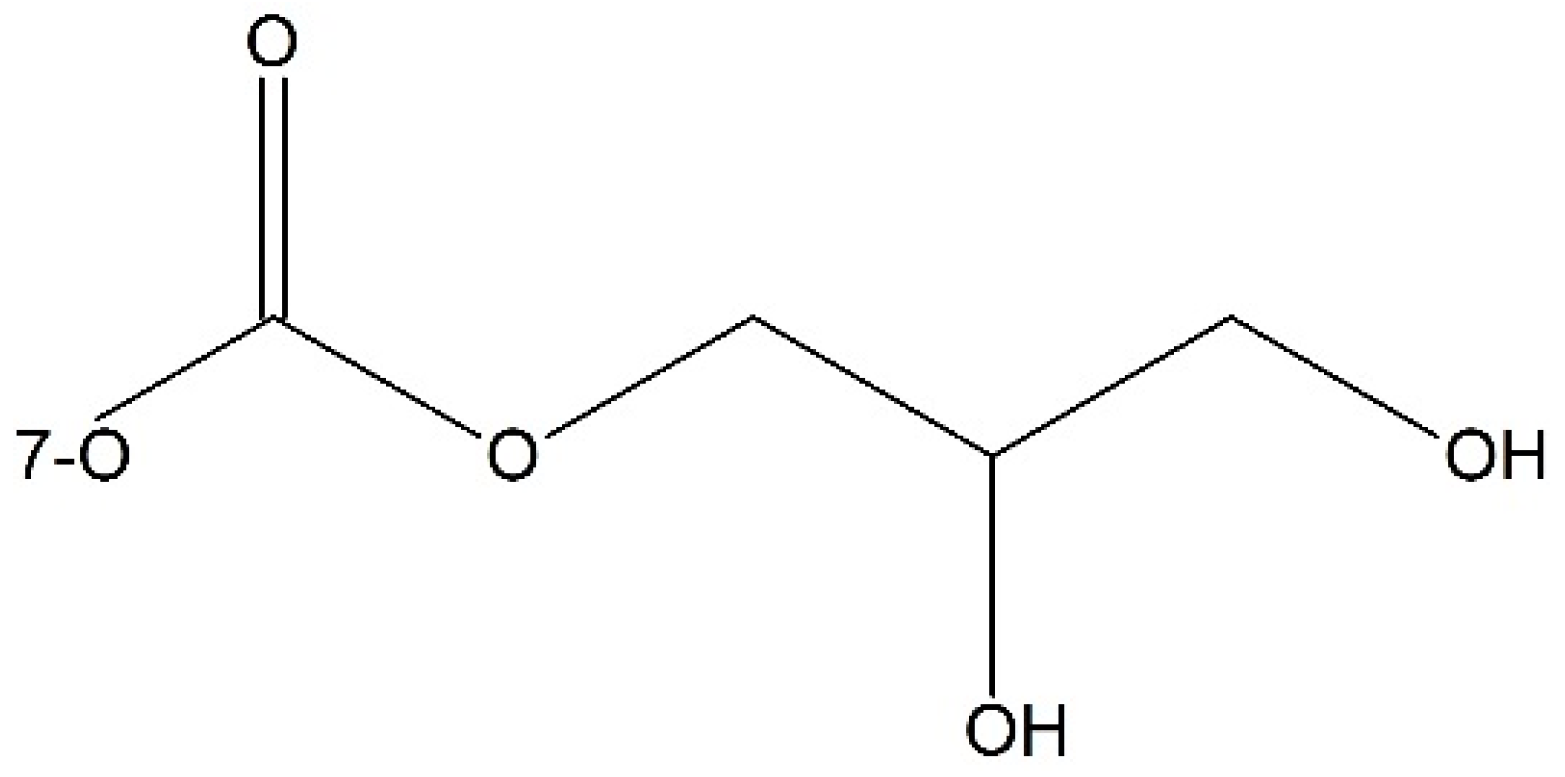
- Damen, E.W.P.; Wiegerinck, P.H.G.; Braamer, L.; Sperling, D.; de Vos, D.; Scheeren, H.W. Paclitaxel esters of malic acid as prodrugs with improved water solubility. Bioorg. Med. Chem. 2000, 8, 427–432. [Google Scholar] [CrossRef]
- Niethammer, A.; Gaedicke, G.; Lode, H.N.; Wrasidlo, W. Synthesis and preclinical characterization of a paclitaxel prodrug with improved antitumor activity and water solubility. Bioconjug. Chem. 2001, 12, 414–420. [Google Scholar] [CrossRef] [PubMed]
- Ansell, S.M.; Johnstone, S.A.; Tardi, P.G.; Lo, L.; Xie, S.; Shu, Y.; Harasym, T.O.; Harasym, N.L.; Williams, L.; Bermudes, D.; et al. Modulating the therapeutic activity of nanoparticle delivered paclitaxel by manipulating the hydrophobicity of prodrug conjugates. J. Med. Chem. 2008, 51, 3288–3296. [Google Scholar] [CrossRef] [PubMed]
- Wohl, A.R.; Michel, A.R.; Kalscheuer, S.; Macosko, C.W.; Panyam, J.; Hoye, T.R. Silicate esters of paclitaxel and docetaxel: Synthesis, hydrophobicity, hydrolytic stability, cytotoxicity, and prodrug potential. J. Med. Chem. 2014, 57, 2368–2379. [Google Scholar] [CrossRef] [PubMed]
- Han, J.; Michel, A.R.; Lee, H.S.; Kalscheuer, S.; Wohl, A.; Hoye, T.R.; McCormick, A.V.; Panyam, J.; Macosko, C.W. Nanoparticles containing high loads of paclitaxel-silicate prodrugs: Formulation, drug release, and anticancer efficacy. Mol. Pharm. 2015, 12, 4329–4335. [Google Scholar] [CrossRef] [PubMed]
- Dosio, F.; Reddy, L.H.; Ferrero, A.; Stella, B.; Cattel, L.; Couvreur, P. Novel nanoassemblies composed of squalenoyl-paclitaxel derivatives: Synthesis, characterization, and biological evaluation. Bioconjug. Chem. 2010, 21, 1349–1361. [Google Scholar] [CrossRef] [PubMed]
- Wittman, M.D.; Kadow, J.F.; Vyas, D.M.; Lee, F.L.; Rose, W.C.; Long, B.H.; Fairchild, C.; Johnston, K. Synthesis and antitumor activity of novel paclitaxel-chlorambucil hybrids. Bioorg. Med. Chem. Lett. 2001, 11, 811–814. [Google Scholar] [CrossRef]
- Li, X.; Yu, J.; Xu, S.; Wang, N.; Yang, H.; Yan, Z.; Cheng, G.; Liu, G. Chemical conjugation of muramyl dipeptide and paclitaxel to explore the combination of immunotherapy and chemotherapy for cancer. Glycoconj. J. 2008, 25, 415–425. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Zhao, N.; Liu, G. Conjugate (MTC-220) of muramyl dipeptide analogue and paclitaxel prevents both tumor growth and metastasis in mice. J. Med. Chem. 2011, 54, 2767–2777. [Google Scholar] [CrossRef] [PubMed]
- Caron, J.; Maksimenko, A.; Mougin, J.; Couvreur, P.; Dasmaele, D. Combined antitumoral therapy with nanoassemblies of bolaform polyisoprenoyl paclitaxel/gemcitabine prodrugs. Polym. Chem. 2014, 5, 1662. [Google Scholar] [CrossRef]
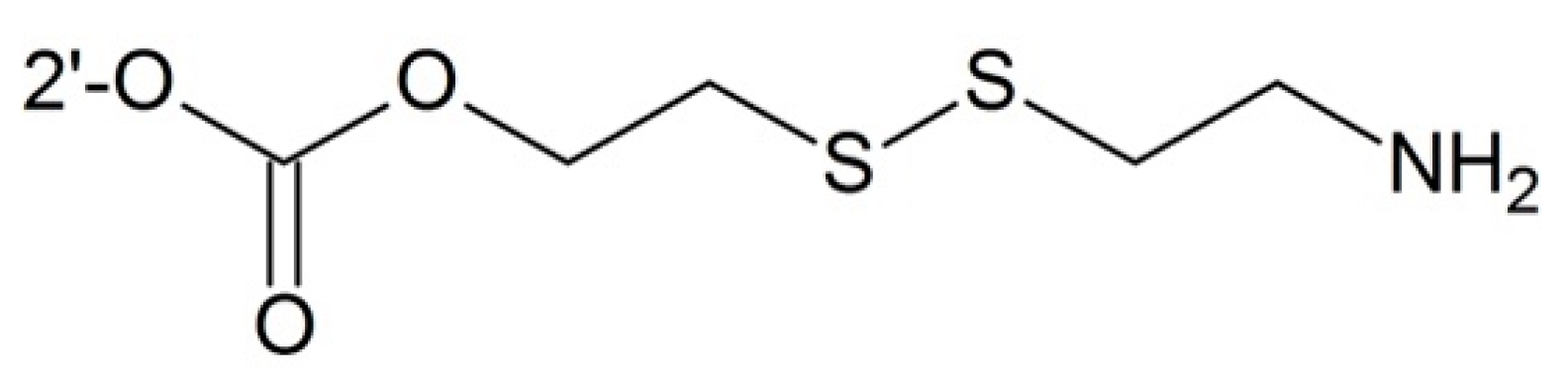
- Vrudhula, V.M.; MacMaster, J.F.; Li, Z.; Kerr, D.E.; Senter, P.D. Reductively activated disulfide prodrugs of paclitaxel. Bioorg. Med. Chem. Lett. 2002, 12, 3591–3594. [Google Scholar] [CrossRef]
- Trant, J.F.; McEachran, M.J.; Sran, I.; Turowec, B.A.; de Bruyn, J.R.; Gillies, E.R. Covalent polyisobutylene–paclitaxel conjugates for controlled release from potential vascular stent coatings. ACS Appl. Mater. Interfaces 2015, 7, 14506–14517. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.-E.; Garvey, D.S.; Janero, D.R.; Letts, L.G.; Marek, P.; Richardson, S.K.; Serebryanik, D.; Shumway, M.J.; Tam, S.W.; Trocha, A.M.; et al. V Combination of paclitaxel and nitric oxide as a novel treatment for the reduction of restenosis. J. Med. Chem. 2004, 47, 2276–2282. [Google Scholar] [CrossRef] [PubMed]
- Dubowchik, G.M.; Mosure, K.; Knipe, J.O.; Firestone, R.A. Cathepsin B-sensitive dipeptide prodrugs. 2. Models of anticancer drugs paclitaxel (Taxol), mitomycin C and doxorubicin. Bioorg. Med. Chem. Lett. 1998, 8, 3347–3352. [Google Scholar] [CrossRef]
- Ojima, I.; Duclos, O.; Dorman, G.; Simonot, B.; Prestwich, G.D.; Rao, S.; Lerro, K.A.; Horwitz, S.B. A New paclitaxel photoaffinity analog with a 3-(4-Benzoylphenyl)propanyl probe for characterization of drug-binding sites on tubulin and P-glycoprotein. J. Med. Chem. 1995, 38, 3891–3894. [Google Scholar] [CrossRef] [PubMed]
- Rice, A.; Liu, Y.; Michaelis, M.L.; Himes, R.H.; Georg, G.I.; Audus, K.L. Chemical modification of paclitaxel (Taxol) reduces P-glycoprotein interactions and increases permeation across the blood-brain barrier in vitro and in situ. J. Med. Chem. 2005, 48, 832–838. [Google Scholar] [CrossRef] [PubMed]
- Skwarczynski, M.; Noguchi, M.; Hirota, S.; Sohma, Y.; Kimura, T.; Hayashi, Y.; Kiso, Y. Development of first photoresponsive prodrug of paclitaxel. Bioorg. Med. Chem. Lett. 2006, 16, 4492–4496. [Google Scholar] [CrossRef] [PubMed]
- Noguchi, M.; Skwarczynski, M.; Prakash, H.; Hirota, S.; Kimura, T.; Hayashi, Y.; Kiso, Y. Development of novel water-soluble photocleavable protective group and its application for design of photoresponsive paclitaxel prodrugs. Bioorg. Med. Chem. 2008, 16, 5389–5397. [Google Scholar] [CrossRef] [PubMed]
- Bradleya, M.O.; Swindella, C.S.; Anthonya, F.H.; Witmana, P.A.; Devanesana, P.; Webb, N.L.; Baker, S.D.; Wolff, A.C.; Donehower, R.C. Tumor targeting by conjugation of DHA to paclitaxel. J. Control. Release 2001, 74, 233–236. [Google Scholar] [CrossRef]
- El Alaoui, A.; Saha, N.; Schmidt, F.; Monneret, C.; Florent, J.C. New Taxol® (paclitaxel) prodrugs designed for ADEPT and PMT strategies in cancer chemotherapy. Bioorg. Med. Chem. 2006, 14, 5012–5019. [Google Scholar] [CrossRef] [PubMed]
- Elsadek, B.; Graeser, R.; Esser, N.; Schäfer-Obodozie, C.; Ajaj, K.A.; Unger, C.; Warnecke, A.; Saleem, T.; El-Melegy, N.; Madkor, H.; et al. Development of a novel prodrug of paclitaxel that is cleaved by prostate-specific antigen: An in vitro and in vivo evaluation study. Eur. J. Cancer 2010, 46, 3434–3444. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.K.; Williams, S.A.; Isaacs, J.T.; Denmeade, S.R.; Khan, S.R. Modulating paclitaxel bioavailability for targeting prostate cancer. Bioorg. Med. Chem. 2007, 15, 4973–4984. [Google Scholar] [CrossRef] [PubMed]
- De Groot, F.M.H.; Van Berkom, L.W.A.; Scheeren, H.W. Synthesis and biological evaluation of 2′-carbamate-linked and 2′-carbonate-linked prodrugs of paclitaxel selective activation by the tumor-associated protease plasmin. J. Med. Chem. 2000, 43, 3093–3102. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.Z.; Sinchaikul, S.; Reddy, P.V.G.; Chang, M.Y.; Chen, S.T. Synthesis of 2′-paclitaxel methyl 2-glucopyranosyl succinate for specific targeted delivery to cancer cells. Bioorg. Med. Chem. Lett. 2007, 17, 617–620. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.-S.; Tungpradit, R.; Sinchaikul, S.; An, F.-M.; Liu, D.-Z.; Phutrakul, S.; Chen, S.-T. Targeting the delivery of glycan-based paclitaxel prodrugs to cancer cells via glucose transporters. J. Med. Chem. 2008, 51, 7428–7441. [Google Scholar] [CrossRef] [PubMed]
- Strese, S.; Fryknäs, M.; Larsson, R.; Gullbo, J. Effects of hypoxia on human cancer cell line chemosensitivity. BMC Cancer 2013, 13, 331. [Google Scholar] [CrossRef] [PubMed]
- Pilkington-Miksa, M.; Arosio, D.; Battistini, L.; Belvisi, L.; de Matteo, M.; Vasile, F.; Burreddu, P.; Carta, P.; Rassu, G.; Perego, P.; et al. Design, synthesis, and biological evaluation of novel cRGD-paclitaxel conjugates for integrin-assisted drug delivery. Bioconjug. Chem. 2012, 23, 1610–1622. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P.; Hu, L.; Yin, Q.; Feng, L.; Li, Y. Transferrin-modified c[RGDfK]-paclitaxel loaded hybrid micelle for sequential blood-brain barrier penetration and glioma targeting therapy. Mol. Pharm. 2012, 9, 1590–1598. [Google Scholar] [CrossRef] [PubMed]
- Colombo, R.; Mingozzi, M.; Belvisi, L.; Arosio, D.; Carenini, N.; Perego, P.; Zaffaroni, N.; Cesare, D.; Castiglioni, V.; Scanziani, E.; et al. Synthesis and biological evaluation (in vitro and in vivo) of cyclic RGD peptidomimetic—Paclitaxel conjugates targeting integrin αVβ3. J. Med. Chem. 2012, 55, 10460–10474. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Plasencia, C.; Hou, Y.; Neamati, N. Synthesis and biological evaluation of dimeric RGD peptide-paclitaxel conjugate as a model for integrin-targeted drug delivery. J. Med. Chem. 2005, 48, 1098–1106. [Google Scholar] [CrossRef] [PubMed]
- Shan, L.; Liu, M.; Wu, C.; Zhao, L.; Li, S.; Xu, L.; Cao, W.; Gao, G.; Gu, Y. Multi-small molecule conjugations as new targeted delivery carriers for tumor therapy. Int. J. Nanomed. 2015, 10, 5571–5591. [Google Scholar] [CrossRef] [PubMed]
- Ndungu, J.M.; Lu, Y.J.; Zhu, S.; Yang, C.; Wang, X.; Chen, G.; Shin, D.M.; Snyder, J.P.; Shoji, M.; Sun, A. Targeted delivery of paclitaxel to tumor cells: Synthesis and in vitro evaluation. J. Med. Chem. 2010, 53, 3127–3132. [Google Scholar] [CrossRef] [PubMed]
- Gund, M.; Khanna, A.; Dubash, N.; Damre, A.; Singh, K.S.; Satyam, A. Water-soluble prodrugs of paclitaxel containing self-immolative disulfide linkers. Bioorg. Med. Chem. Lett. 2015, 25, 122–127. [Google Scholar] [CrossRef] [PubMed]
- Vrudhula, V.M.; Kerr, D.E.; Siemers, N.O.; Dubowchik, G.M.; Senter, P.D. Cephalosporin prodrugs of paclitaxel for immunologically specific activation by l-49-sFv-β-Lactamase fusion protein. Bioorg. Med. Chem. Lett. 2003, 13, 539–542. [Google Scholar] [CrossRef]
- De Bont, D.B.; Leenders, R.G.; Haisma, H.J.; van der Meulen-Muileman, I.; Scheeren, H.W. Synthesis and biological activity of β-glucuronyl carbamate-based prodrugs of paclitaxel as potential candidates for ADEPT. Bioorg. Med. Chem. 1997, 5, 405–414. [Google Scholar] [CrossRef]
- Ishida, D.; Nawa, A.; Tanino, T.; Goshima, F.; Luo, C.H.; Iwaki, M.; Kajiyama, H.; Shibata, K.; Yamamoto, E.; Ino, K.; et al. Enhanced cytotoxicity with a novel system combining the paclitaxel-2′-ethylcarbonate prodrug and an HSV amplicon with an attenuated replication-competent virus, HF10 as a helper virus. Cancer Lett. 2010, 288, 17–27. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.-C.; Rissanen, S.; Stepniewski, M.; Cramariuc, O.; Róg, T.; Mirza, S.; Xhaard, H.; Wytrwal, M.; Kepczynski, M.; Bunker, A. Study of interaction between PEG carrier and three relevant drug molecules: Piroxicam, paclitaxel, and hematoporphyrin. J. Phys. Chem. B 2012, 116, 7334–7341. [Google Scholar] [CrossRef] [PubMed]
- Feng, X.; Yuan, Y.J.; Wu, J.C. Synthesis and evaluation of water-soluble paclitaxel prodrugs. Bioorg. Med. Chem. Lett. 2002, 12, 3301–3303. [Google Scholar] [CrossRef]
- Greenwald, R.B.; Gilbert, C.W.; Pendri, A.; Conover, C.D.; Xia, J.; Martinez, A. Drug delivery systems: Water soluble taxol 2′-poly(ethylene glycol) ester prodrugs-design and in vivo effectiveness. J. Med. Chem. 1996, 39, 424–431. [Google Scholar] [CrossRef] [PubMed]
- Liang, L.; Lin, S.W.; Dai, W.; Lu, J.K.; Yang, T.Y.; Xiang, Y.; Zhang, Y.; Li, R.T.; Zhang, Q. Novel cathepsin B-sensitive paclitaxel conjugate: Higher water solubility, better efficacy and lower toxicity. J. Control. Release 2012, 160, 618–629. [Google Scholar] [CrossRef] [PubMed]
- Gu, Y.; Zhong, Y.; Meng, F.; Cheng, R.; Deng, C.; Zhong, Z. Acetal-linked paclitaxel prodrug micellar nanoparticles as a versatile and potent platform for cancer therapy. Biomacromolecules 2013, 14, 2772–2780. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.; Zou, J.; Yu, L.; Ji, W.; Li, Y.; Law, W.; Cheng, C. Functional polylactide-g-paclitaxel–poly(ethylene glycol) by azide–alkyne click chemistry. Macromolecules 2011, 44, 4793–4800. [Google Scholar] [CrossRef]
- Lv, S.; Tang, Z.; Zhang, D.; Song, W.; Li, M.; Lin, J.; Liu, H.; Chen, X. Well-defined polymer-drug conjugate engineered with redox and pH-sensitive release mechanism for efficient delivery of paclitaxel. J. Control. Release 2014, 194, 220–227. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Shi, Y.; Feng, H.; Du, M.; Zhang, J.Z.; Hu, J.; Yang, D. Preparation of copolymer paclitaxel covalently linked via a disulfide bond and its application on controlled drug delivery. J. Phys. Chem. B 2012, 116, 9231–9237. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.; Wang, T.; Perche, F.; Taigind, A.; Torchilin, V.P. Enhanced anticancer activity of nanopreparation containing an MMP2-sensitive PEG-drug conjugate and cell-penetrating moiety. Proc. Natl. Acad. Sci. USA 2013, 110, 17047–17052. [Google Scholar] [CrossRef] [PubMed]
- Bao, Y.; Guo, Y.; Zhuang, X.; Li, D.; Cheng, B.; Tan, S.; Zhang, Z. Polyethylene glycol succinate-based redox-sensitive paclitaxel prodrug for overcoming multidrug resistance in cancer cells d-α-tocopherol. Mol. Pharm. 2014, 11, 3196–3209. [Google Scholar] [CrossRef] [PubMed]
- Safavy, A.; Raisch, K.P.; Khazaeli, M.B.; Buchsbaum, D.J.; Bonner, J.A. Paclitaxel derivatives for targeted therapy of cancer: Toward the development of smart taxanes. J. Med. Chem. 1999, 42, 4919–4924. [Google Scholar] [CrossRef] [PubMed]
- Safavy, A.; Raisch, K.P.; Matusiak, D.; Bhatnagar, S.; Helson, L. Single-drug multiligand conjugates: Synthesis and preliminary cytotoxicity evaluation of a paclitaxel-dipeptide “scorpion” molecule. Bioconjug. Chem. 2006, 17, 565–570. [Google Scholar] [CrossRef] [PubMed]
- Yin, T.; Wu, Q.; Wang, L.; Yin, L.; Zhou, J.; Huo, M. Well-defined redox-sensitive polyethene glycol–paclitaxel prodrug conjugate for tumor-specific delivery of paclitaxel using octreotide for tumor targeting. Mol. Pharm. 2015, 12, 3020–3031. [Google Scholar] [CrossRef] [PubMed]
- Yin, D.; Ge, Z.; Yang, W.; Liu, C.; Yuan, Y. Inhibition of tumor metastasis in vivo by combination of paclitaxel and hyaluronic acid. Cancer Lett. 2006, 243, 71–79. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Lee, K.; Tae, G.P. Hyaluronic acid-paclitaxel conjugate micelles: Synthesis, characterization, and antitumor activity. Bioconjug. Chem. 2008, 19, 1319–1325. [Google Scholar] [CrossRef] [PubMed]
- Yin, S.; Huai, J.; Chen, X.; Yang, Y.; Zhang, X.; Gan, Y.; Wang, G.; Gu, X.; Li, J. Intracellular delivery and antitumor effects of a redox-responsive polymeric paclitaxel conjugate based on hyaluronic acid. Acta Biomater. 2015, 26, 274–285. [Google Scholar] [CrossRef] [PubMed]
- Miller, K.; Eldar-Boock, A.; Polyak, D.; Segal, E.; Benayoun, L.; Shaked, Y.; Satchi-Fainaro, R. Antiangiogenic antitumor activity of HPMA copolymer-paclitaxel-alendronate conjugate on breast cancer bone metastasis mouse model. Mol. Pharm. 2011, 8, 1052–1062. [Google Scholar] [CrossRef] [PubMed]
- Erez, R.; Segal, E.; Miller, K.; Satchi-Fainaro, R.; Shabat, D. Enhanced cytotoxicity of a polymer-drug conjugate with triple payload of paclitaxel. Bioorg. Med. Chem. 2009, 17, 4327–4335. [Google Scholar] [CrossRef] [PubMed]
- Etrych, T.; Šírovaé, M.; Starovoytova, L.; Řiéhovaé, B.; Ulbrich, K. HPMA copolymer conjugates of paclitaxel and docetaxel with pH-controlled drug release. Mol. Pharm. 2010, 7, 1015–1026. [Google Scholar] [CrossRef] [PubMed]
- Cline, E.N.; Li, M.-H.; Choi, S.K.; Herbstman, J.F.; Kaul, N.; Meyhöfer, E.; Skiniotis, G.; Baker, J.R.; Larson, R.G.; Walter, N.G. Paclitaxel-conjugated PAMAM dendrimers adversely affect microtubule structure through two independent modes of action. Biomacromolecules 2013, 14, 654–664. [Google Scholar] [CrossRef] [PubMed]
- Lim, J.; Chouai, A.; Lo, S.-T.; Liu, W.; Sun, X.; Simanek, E.E. Design, synthesis, characterization, and biological evaluation of triazine dendrimers bearing paclitaxel using ester and ester/disulfide linkages. Bioconjug. Chem. 2009, 20, 2154–2161. [Google Scholar] [CrossRef] [PubMed]
- Lim, J.; Lo, S.T.; Hill, S.; Pavan, G.M.; Sun, X.K.; Simanek, E.E. Antitumor activity and molecular dynamics simulations of paclitaxel-laden triazine dendrimers. Mol. Pharm. 2012, 9, 404–412. [Google Scholar] [CrossRef] [PubMed]
- Lim, J.; Simanek, E.E. Synthesis of water-soluble dendrimers based on melamine bearing 16 paclitaxel groups. Org. Lett. 2008, 10, 201–204. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.; Lo, S.-T.; Lim, J.; da Costa, V.C.P.; Ramezani, S.; Öz, O.K.; Pavan, G.M.; Annunziata, O.; Sun, X.; Simanek, E.E. Design, synthesis and biological assessment of a triazine dendrimer with approximately 16 Paclitaxel groups and 8 PEG groups. Mol. Pharm. 2013, 10, 4452–4461. [Google Scholar] [CrossRef] [PubMed]
- Kala, S.; Mak, A.S.C.; Liu, X.; Posocco, P.; Pricl, S.; Peng, L.; Wong, A.S.T. Combination of dendrimer-nanovector-mediated small interfering RNA delivery to target Akt with the clinical anticancer drug paclitaxel for effective and potent anticancer activity in treating ovarian cancer. J. Med. Chem. 2014, 57, 2634–2642. [Google Scholar] [CrossRef] [PubMed]
- Satsangi, A.; Roy, S.S.; Satsangi, R.K.; Vadlamudi, R.K.; Ong, J.L. Design of a paclitaxel prodrug conjugate for active targeting of an enzyme upregulated in breast cancer cells. Mol. Pharm. 2014, 11, 1906–1918. [Google Scholar] [CrossRef] [PubMed]
- Clementi, C.; Miller, K.; Mero, A.; Satchi-Fainaro, R.; Pasut, G. Dendritic poly(ethylene glycol) bearing paclitaxel and alendronate for targeting bone neoplasms. Mol. Pharm. 2011, 8, 1063–1072. [Google Scholar] [CrossRef] [PubMed]
- Lee, E.; Lee, J.; Lee, I.H.; Yu, M.; Kim, H.; Chae, S.Y.; Jon, S. Conjugated chitosan as a novel platform for oral delivery of paclitaxel. J. Med. Chem. 2008, 51, 6442–6449. [Google Scholar] [CrossRef] [PubMed]
- Liebmann, J.E.; Cook, J.A.; Lipschultz, C.; Teague, D.; Fisher, J.; Mitchell, J.B. Cytotoxic studies of paclitaxel (Taxol) in human tumour cell lines. Br. J. Cancer 1993, 68, 1104–1109. [Google Scholar] [CrossRef] [PubMed]
- Cavallaro, G.; Licciardi, M.; Caliceti, P.; Salmaso, S.; Giammona, G. Synthesis, physico-chemical and biological characterization of a paclitaxel macromolecular prodrug. Eur. J. Pharm. Biopharm. 2004, 58, 151–159. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.; Chen, C.; Law, W.; Mok, J.; Zou, J.; Prasad, P.N.; Cheng, C. Well-defined degradable brush polymer-drug conjugates for sustained delivery of paclitaxel. Mol. Pharm. 2013, 10, 867–874. [Google Scholar] [CrossRef] [PubMed]
- Dalela, M.; Shrivastav, T.G.; Kharbanda, S.; Singh, H. pH-sensitive biocompatible nanoparticles of paclitaxel-conjugated poly(styrene-co- maleic acid) for anticancer drug delivery in solid tumor of syngeneic mice. ACS Appl. Mater. Interfaces 2015, 7, 26530–26548. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Xin, D.; Liu, K.; Zhu, M.; Xiang, J. Heparin-Paclitaxel Conjugates as Drug Delivery System: Synthesis, Self-Assembly Property, Drug Release, and Antitumor Activity. Bioconjug. Chem. 2009, 20, 2214–2221. [Google Scholar] [CrossRef] [PubMed]
- Dosio, F.; Brusa, P.; Crosasso, P.; Arpicco, S.; Cattel, L. Preparation, characterization and properties in vitro and in vivo of a paclitaxel-albumin conjugate. J. Control. Release 1997, 47, 293–304. [Google Scholar] [CrossRef]
- Safavy, A.; Bonner, J.A.; Waksal, H.W.; Buchsbaum, D.J.; Gillespie, G.Y.; Khazaeli, M.B.; Arani, R.; Chen, D.T.; Carpenter, M.; Raisch, K.P. Synthesis and biological evaluation of paclitaxel-C225 conjugate as a model for targeted drug delivery. Bioconjug. Chem. 2003, 14, 302–310. [Google Scholar] [CrossRef] [PubMed]
- Yuan, L.; Chen, W.; Hu, J.; Zhang, J.Z.; Yang, D. Mechanistic study of the covalent loading of paclitaxel via disulfide linkers for controlled drug release. Langmuir 2013, 29, 734–743. [Google Scholar] [CrossRef] [PubMed]
- Lai, C.-H.; Chang, T.-C.; Chuang, Y.-J.; Tzou, D.-L.; Lin, C.-C. Stepwise orthogonal click chemistry toward fabrication of paclitaxel/galactose functionalized fluorescent nanoparticles for HepG2 cell targeting and delivery. Bioconjug. Chem. 2013, 24, 1698–1709. [Google Scholar] [CrossRef] [PubMed]
- Ding, Y.; Zhou, Y.-Y.; Chen, H.; Geng, D.-D.; Wu, D.-Y.; Hong, J.; Shen, W.-B.; Hang, T.-J.; Zhang, C. The performance of thiol-terminated PEG-paclitaxel-conjugated gold nanoparticles. Biomaterials 2013, 34, 10217–10227. [Google Scholar] [CrossRef] [PubMed]
- Gibson, J.D.; Khanal, B.P.; Zubarev, E.R. Paclitaxel-functionalized gold nanoparticles. J. Am. Chem. Soc. 2007, 129, 11653–11661. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Zhu, S.; Wang, M.; Li, Y.; Shi, P.; Huang, X. Delivery of paclitaxel using PEGylated graphene oxide as a nanocarrier. ACS Appl. Mater. Interfaces 2015, 7, 1355–1363. [Google Scholar] [CrossRef] [PubMed]
- Zakharian, T.Y.; Seryshev, A.; Sitharaman, B.; Gilbert, B.E.; Knight, V.; Wilson, L.J. A fullerene-paclitaxel chemotherapeutic: Synthesis, characterization, and study of biological activity in tissue culture. J. Am. Chem. Soc. 2005, 127, 12508–12509. [Google Scholar] [CrossRef] [PubMed]
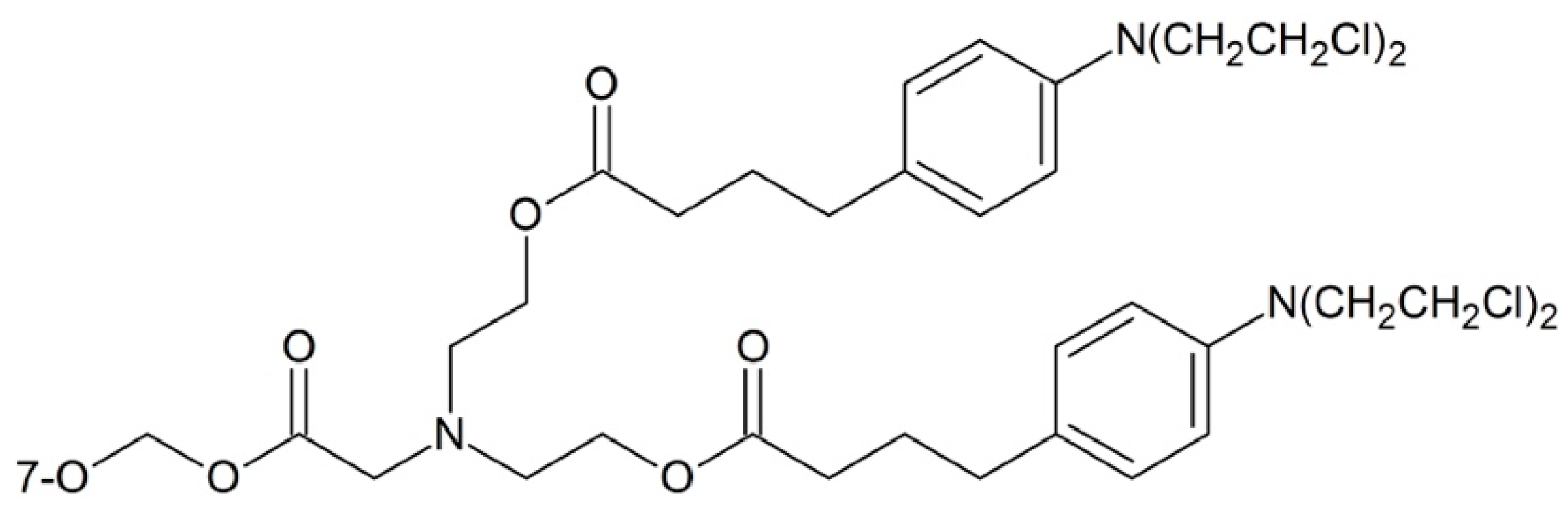
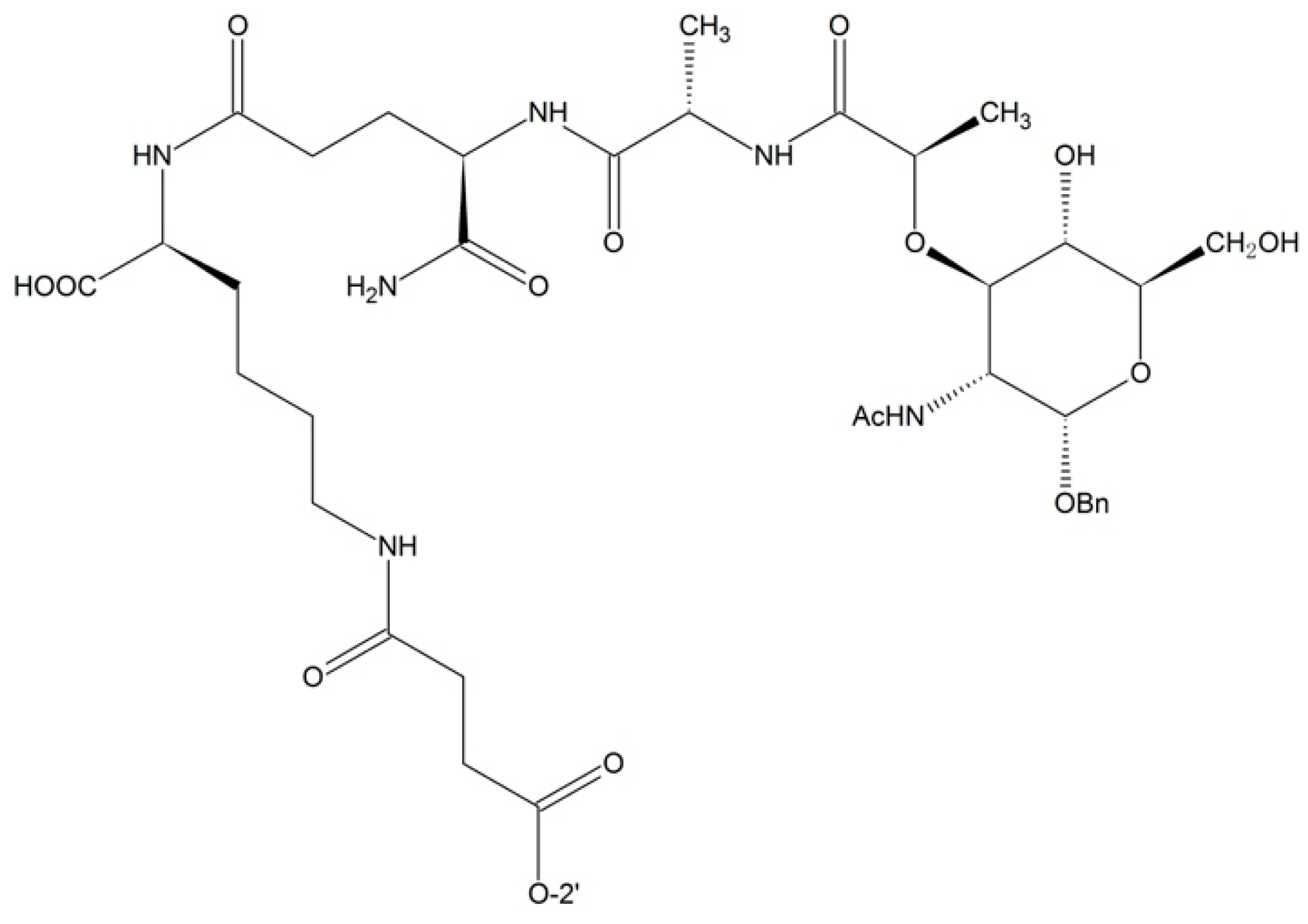
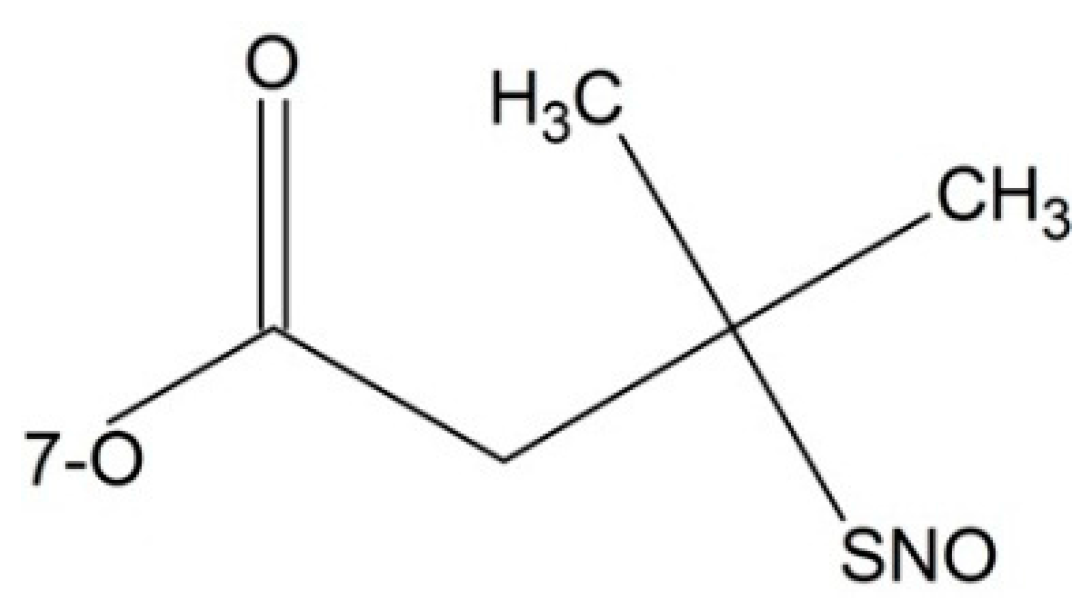
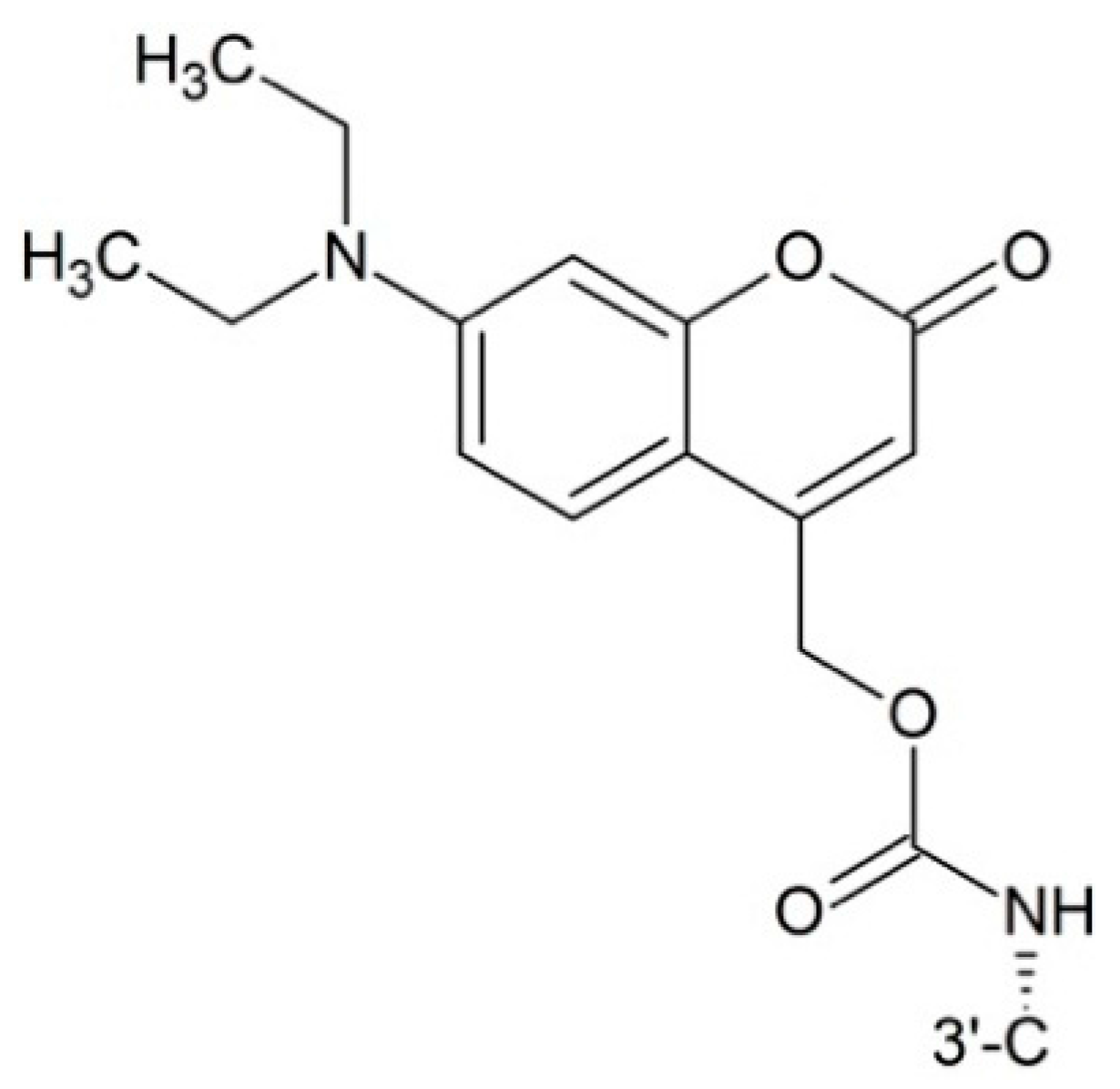
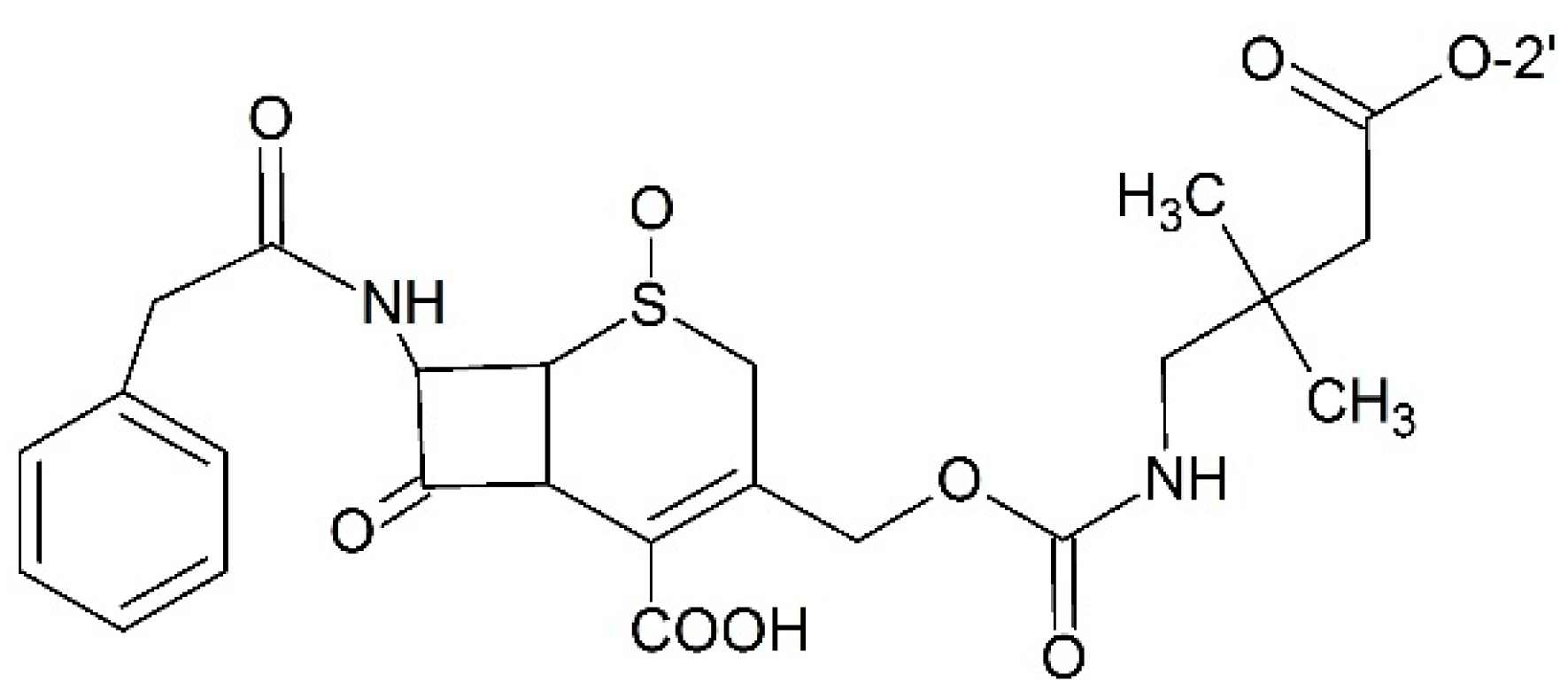
| 2′-O | 7-O | cLogP | T1/2 (min) |
|---|---|---|---|
| H | H | 3.2 | N/A |
| Si(OEt)3 | H | 5.0 | 3.7 |
| Si(On-Oct)3 | H | 7.7 | 12 |
| Si(Oi-Pr) | H | 5.6 | 120 |
| Si(Ot-Bu)2(OEt) | H | 5.8 | 12,000 |
| Si(Omenthyl)3 | H | 7.4 | 69,000 |
| H | Si(OEt)3 | 5.1 | 30 |
| H | Si(On-Oct)3 | 7.8 | 150 |
| Si(OEt)3 | Si(OEt)3 | 6.3 | 4.6 (2′-O) 33 (7-O) |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Meng, Z.; Lv, Q.; Lu, J.; Yao, H.; Lv, X.; Jiang, F.; Lu, A.; Zhang, G. Prodrug Strategies for Paclitaxel. Int. J. Mol. Sci. 2016, 17, 796. https://doi.org/10.3390/ijms17050796
Meng Z, Lv Q, Lu J, Yao H, Lv X, Jiang F, Lu A, Zhang G. Prodrug Strategies for Paclitaxel. International Journal of Molecular Sciences. 2016; 17(5):796. https://doi.org/10.3390/ijms17050796
Chicago/Turabian StyleMeng, Ziyuan, Quanxia Lv, Jun Lu, Houzong Yao, Xiaoqing Lv, Feng Jiang, Aiping Lu, and Ge Zhang. 2016. "Prodrug Strategies for Paclitaxel" International Journal of Molecular Sciences 17, no. 5: 796. https://doi.org/10.3390/ijms17050796
APA StyleMeng, Z., Lv, Q., Lu, J., Yao, H., Lv, X., Jiang, F., Lu, A., & Zhang, G. (2016). Prodrug Strategies for Paclitaxel. International Journal of Molecular Sciences, 17(5), 796. https://doi.org/10.3390/ijms17050796







