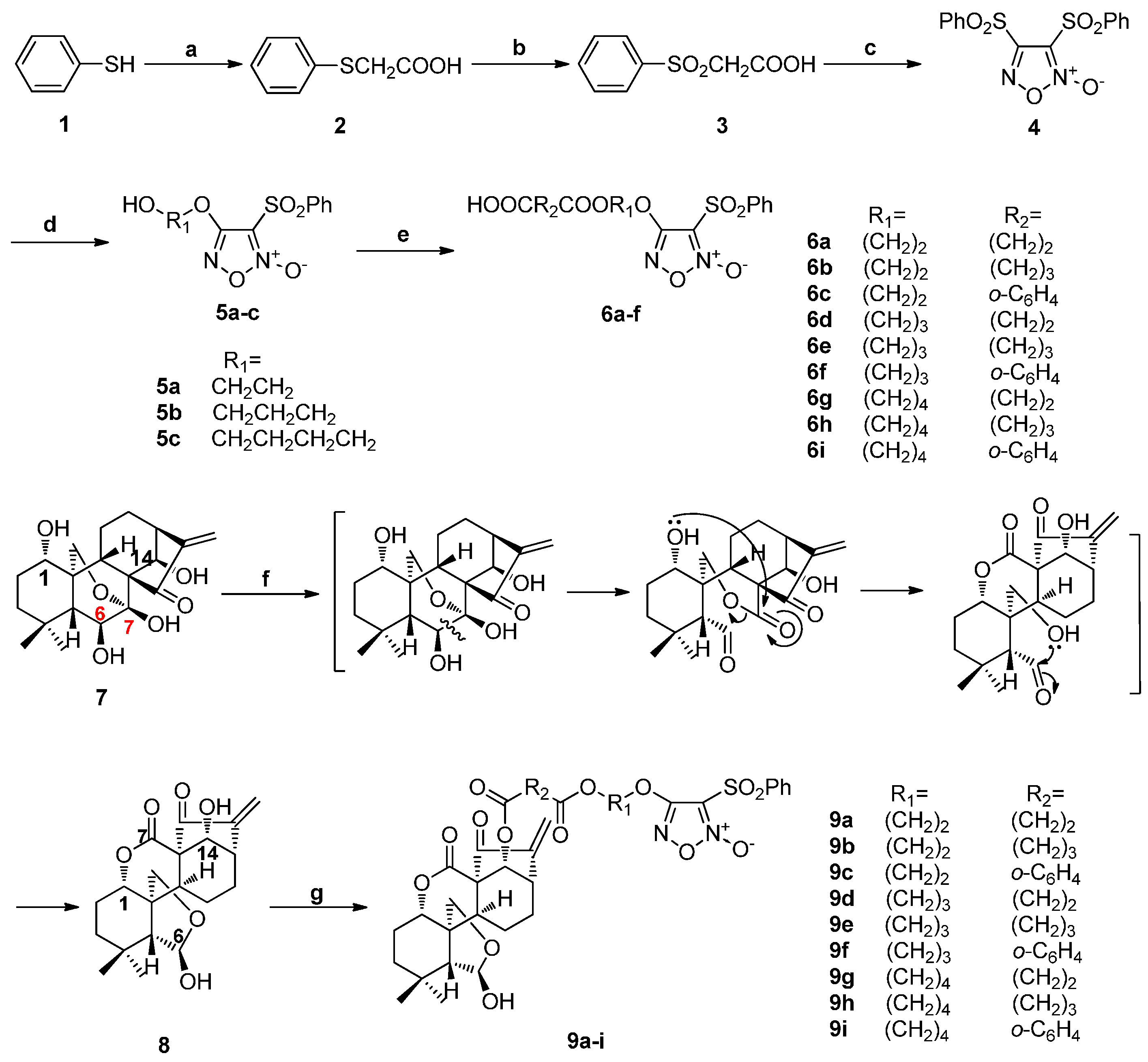Abstract
Herein, we reported on a series of synthetic nitric oxide-releasing enmein-type diterpenoid hybrids (9a–i). All the target compounds showed potent antibacterial activity against selected Gram-positive bacteria S. aureus and B. subtilis. The antiproliferative activity against human tumor K562, MGC-803, CaEs-17 and Bel-7402 cells, and human normal liver cells L-02 was tested and the structure activity relationships (SARs) were also concluded. Compounds 9b and 9d showed the best activity against S. aureus and B. subtilis with the same minimal inhibitory concentrations (MICs) of 4 and 2 μg/mL, respectively. The derivative 9f displayed IC50 values of 1.68, 1.11, 3.60 and 0.72 μM against the four cancer cell lines above and 18.80 μM against normal liver cells L-02; meanwhile, 9f also released a high level of NO at the time point of 60 min of 22.24 μmol/L. Furthermore, it was also found that 9f induced apoptosis via the mitochondria-related pathway and arrested cell cycle of Bel-7402 cells at S phase. These findings might be important to explore new chemical entities for the main causes of in-hospital mortality of S. aureus infection, combined with a solid tumor.
1. Introduction
Bacterial infections, causing deadly diseases, have increased at an alarming rate [1,2]. The development of drug resistance among the infectious bacterial strains and absence of effective preventive measures have become the two major driving forces for design and identify suitable active new chemical entities. On the other hand, cancer, uncontrolled, rapidly proliferating, abnormal cells, is now the second leading cause of human deaths, and the high mortality rate engages with the rising number of diverse cancer types [3]. These have triggered research aiming to find new lead structures that may be beneficial in the design of novel antitumor agents [4,5]. Patients with neoplastic disorders are subjected to chemotherapeutic treatment, and susceptible to microbial infections due to the poor immunity [6]. For example, Staphylococcus aureus is a pathogen responsible for the main causes of in-hospital mortality [7]. Therefore, the discovery of new novel potent antibacterial and antitumor chemical entities is challenging work for medicinal chemists [8,9,10,11].
A potential solution is to explore innovative natural scaffolds from natural sources [12,13]. Among numerous exploited medicinal plants, the Isodon species are traditionally used as antitumor and antibacterial folk medicine and have many complex and bioactive diterpenoids [14,15]. They have recently received great attention from researchers from many research fields [16,17,18]. In this context, enmein-type ent-kauranoids, one of the important and main classes of diterpenoids from the plants of Isodon species with antitumor and antibacterial activities, are worthy of intensive investigation.
Nitric oxide (NO) is an endogenous, small and reactive molecule, which has various physiological and biological properties [19,20,21]. High concentration of NO is cytotoxic to tumor cells by inducing tumor cell apoptosis, sensitizing drug-resistant tumor cells, and inhibiting tumor metastasis, and so on [22,23]. NO also exhibits antimicrobial activity and kills bacteria through lipid peroxidation, DNA cleavage, and protein dysfunction. The multiple bactericidal pathways of NO make it a potent broad-spectrum antimicrobial agent with low risk [24,25,26]. Thus, NO-donating groups are good pharmacophores for both antitumor and antibacterial agents.
On the basis of the above, we developed a series of new enmein-type ent-kauranoid derivatives containing furozan-based NO-donors. The antibacterial potency, antiproliferative activity and NO releasing ability of target compounds were tested. Furthermore, the preliminary antitumor mechanisms were also disclosed.
2. Results and Discussion
2.1. Chemistry
Diphenylsulfonylfuroxan (4) was obtained in a three-step sequence starting from benzenethiol (1), and was then treated with corresponding diol to afford monophenylsulfonylfuroxans (5a–c). Furoxan-based NO donors intermediate 6a–i were obtained from the condensation of 5a–c with corresponding anhydride. Enmein-type ent-kauranoid derivative 8 was synthesized from oridonin (7) by treatment with sodium periodate in water. Target NO donor/enmein-type ent-kauranoid hybrids 9a–i were designed and synthesized from 8 with 6a–i in the presence of DMAP/EDCI in DCM. Flash chromatography was used only at the last step of the synthetic route of each target compound (Scheme 1) [22,27].
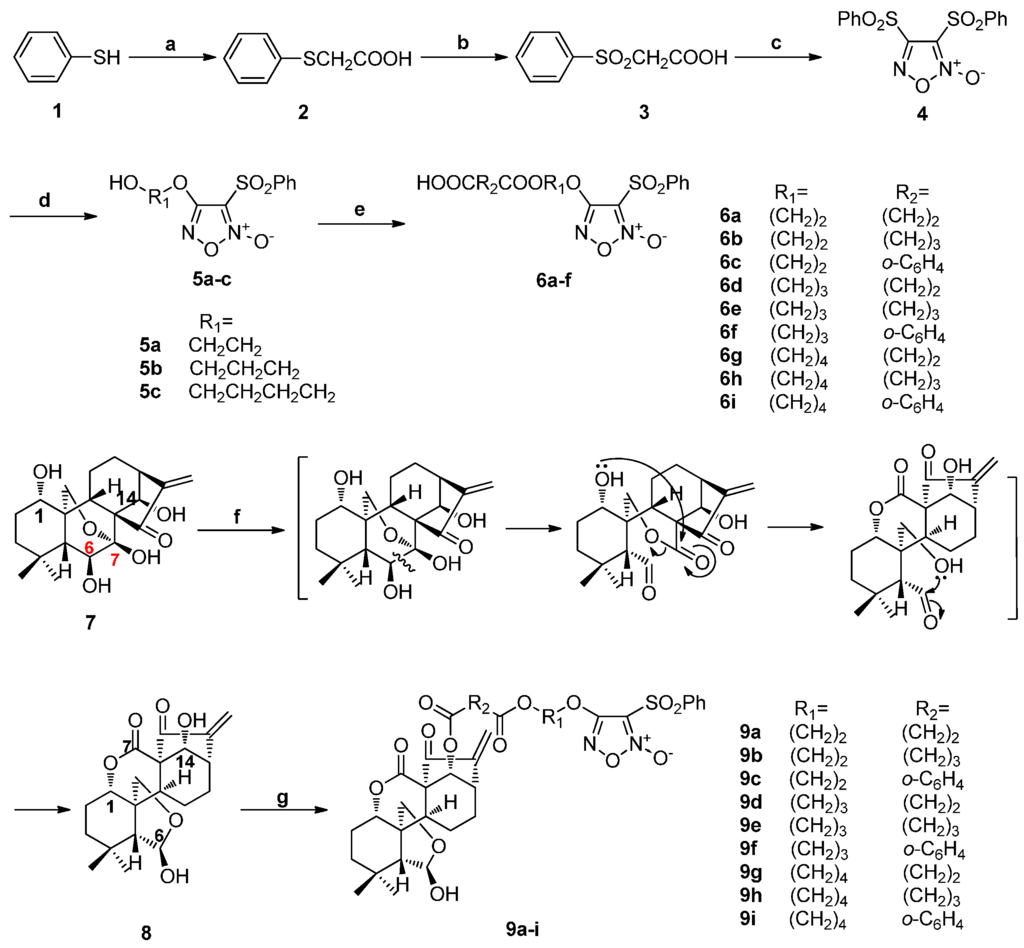
Scheme 1.
Synthesis of NO donor/enmein-type ent-kauranoid hybrids 9a–i. Reagents and conditions: (a) ClCH2COOH, NaOH(aq), 140 °C, 2 h; (b) 30% H2O2, AcOH, rt, 3 h; (c) fuming HNO3, 90 °C, 4 h; (d) diol, THF, 30% NaOH, 0 °C, 4–8 h; (e) triethylamine, succinic anhydride, DMAP, rt, 1 h; (f) NaIO4, H2O, rt, 6 h; (g) 6, EDCI, DMAP, rt, 12 h.
2.2. Antimicrobial Activity
The antibacterial activity of NO donor/enmein-type ent-kauranoid hybrids 9a–i against the Gram-negative bacterium Escherichia coli (ATCC 25922), Gram-positive bacterium Staphylococcus aureus (ATCC 29213) and Bacillus subtilis (CMCC 63501), and the fungus Monilia albicans (ATCC 10231) was first disclosed and summarized in Table 1. Parent compound enmein-type 6,7-seco-ent-kauranoid 8 showed the minimum inhibitory concentration (MIC) above 100 μg/mL and derivatives 9a–i exhibited antibacterial activity against gram-positive bacterium S. aureus and B. subtilis to some extent. In the S. aureus strain, 9a–i exhibited similar or stronger antibacterial activity than 7, which meant that structural modifications introduced going in the right direction, improving its biological properties. In the B. subtilis strain, 9b–d and 9i were even more potent than positive control chloromycetin. Thus, the introduction of NO-donor substituent groups at the 14-position of enmein-type 6,7-seco-ent-kauranoid could improve antimicrobial activity against S. aureus and B. subtilis. However, almost all of them were inactive (MIC > 100 μg/mL) against E. coli and M. albicans, and target compounds selectively inhibited Gram-positive bacteria S. aureus and B. Subtilis.

Table 1.
The antimicrobial activity of the synthesized compounds (MIC μg/mL).
The most promising compounds 9b and 9d exhibited the strongest antibacterial activity among 9a–i with MICs of 4 and 2 μg/mL against S. aureus and B. subtilis. The substituents (R1 and R2) of 9b were (CH2)2 and (CH2)3, respectively, while the substituents were (CH2)3 and (CH2)2 of 9d. Coincidentally, two of the most active compounds, 9b and 9d, had the same total linkage length of five carbons (CH2)5. The linkages between NO donor and parent compound always affected the biological activity. Different lead compounds had their own favorable linker [22,23,28,29].
2.3. Antiproliferative Activity
The antiproliferative activity of 9a–i against four human cancer cell lines K562 leukemia cell line, MGC-803 gastric cancer cell line, CaEs-17 esophageal cancer cell line, and Bel-7402 hepatoma cell line, and human normal liver cells L-02 was evaluated by MTT assay (Table 2). All the target molecules 9a–i were stronger than oridonin 7 with IC50 values below 1.33 μM and lower than Taxol (1.89 μM) against Bel-7402 cell line. The most potent 9f with IC50 of 0.72 μM was 1-fold stronger than Taxol. While, target compounds were not so sensitive to CaEs-17 cells with IC50 ranging from 3.60 to 5.13 μM. IC50 values against MGC-803 cell line were a little weaker than Taxol, between 1.11 to 1.83 μM. For K562 cell line, the antiproliferative activity of all the derivatives was weaker than Taxol. Most NO donor derivatives showed cytotoxic selectivity between tumor and normal liver cells to some extent, with IC50 values ranging from 10.36 to 19.87 μM against L-02 normal liver cells. These results confirmed previous reports [28,29] that some NO donating derivatives showed less cytotoxicity to normal cells.

Table 2.
Antiproliferative activity of the synthesized compounds (IC50 μM).
In most cases against the selected tumor cell lines, among the derivatives 9a–i, when the substitution R1 was the same (9a–c, 9d–f and 9g–i) and R2 was changed among (CH2)2, (CH2)3, and o-C6H4 in each group, compounds (9c, 9f and 9i) with R2 of aromatic group of o-C6H4 showed stronger activity than corresponding ones with alkyl groups (9a, 9b, 9d, 9e, 9g and 9h). Between alkyl substituents (CH2)2 and (CH2)3 in R2, the latter one (9b, 9e and 9h) was favorable. For example, 9i with the same R1 of (CH2)4 as 9g and 9h, and R2 of o-C6H4, showed slightly lower IC50 values of 1.25, 1.86, 3.82 and 0.78 μM against K562, MGC-803, CaEs-17 and Bel-7402 tumor cells, respectively. When the substitution R2 was the same and R1 was changed among (CH2)2, (CH2)3, and (CH2)4, compounds showed statistically equal IC50 values.
2.4. NO-Releasing Ability
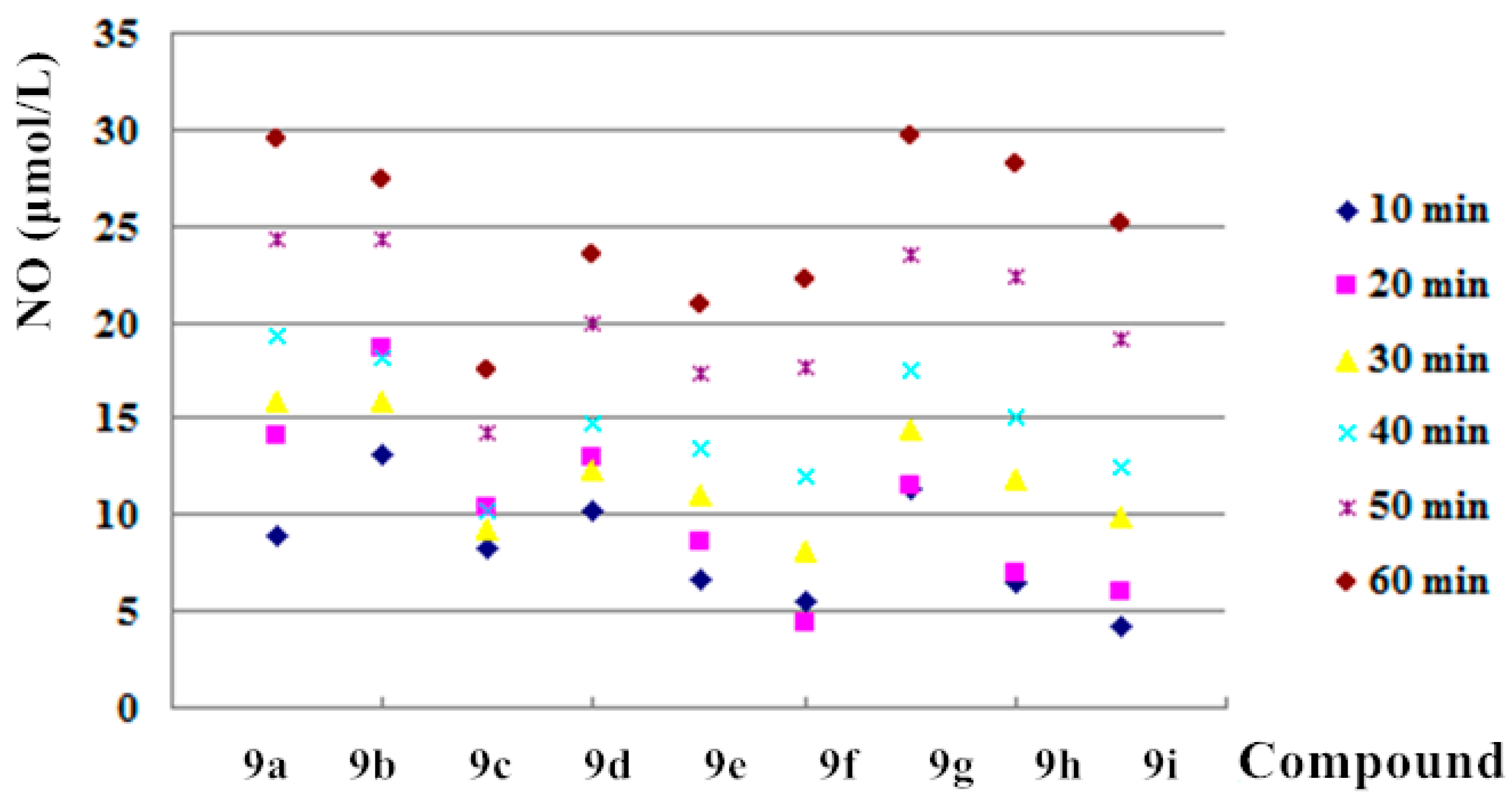
The NO-releasing ability of 9a–i was determined by Griess assay at 100 μM and measured at the time point of 10, 20, 30, 40, 50 and 60 min (Figure 1). The concentrations of released NO for all synthetic hybrids increased with time and were more than 20 μmol/L (except 9c) at the time point of 60 min. High NO releasing ability might, at least to a certain extent, contribute to antiproliferative and antibacterial activities [23,26]. The most promising NO-releasing enmein-type 6,7-seco-ent-kauranoid derivative was 9f with the highest selectivity index (SI) value of 26.1. It was chosen for intensive mechanism study on hepatoma Bel-7402 cell line.
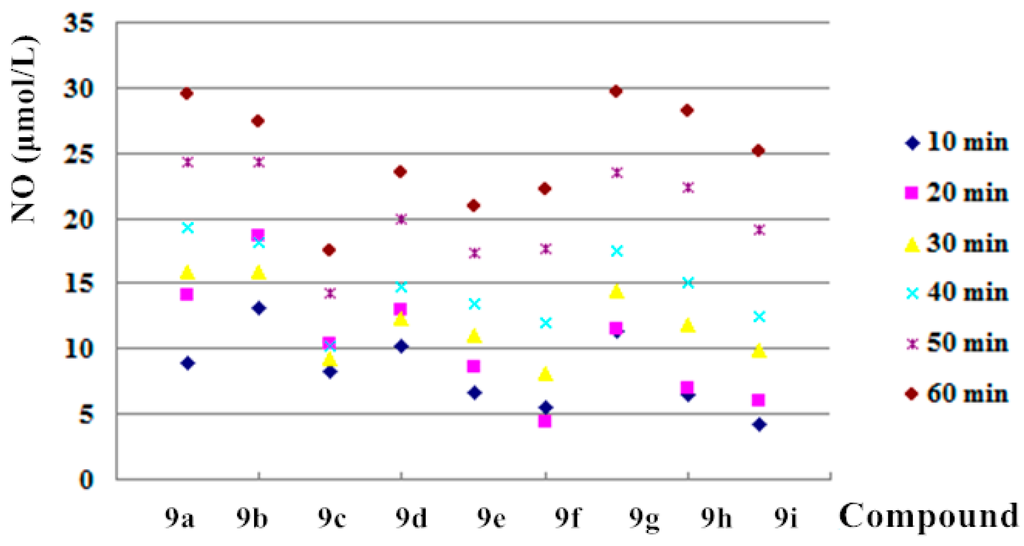
Figure 1.
NO-releasing ability of compounds 9a–i.
2.5. Influence of 9f on the Bel-7402 Cell Cycle
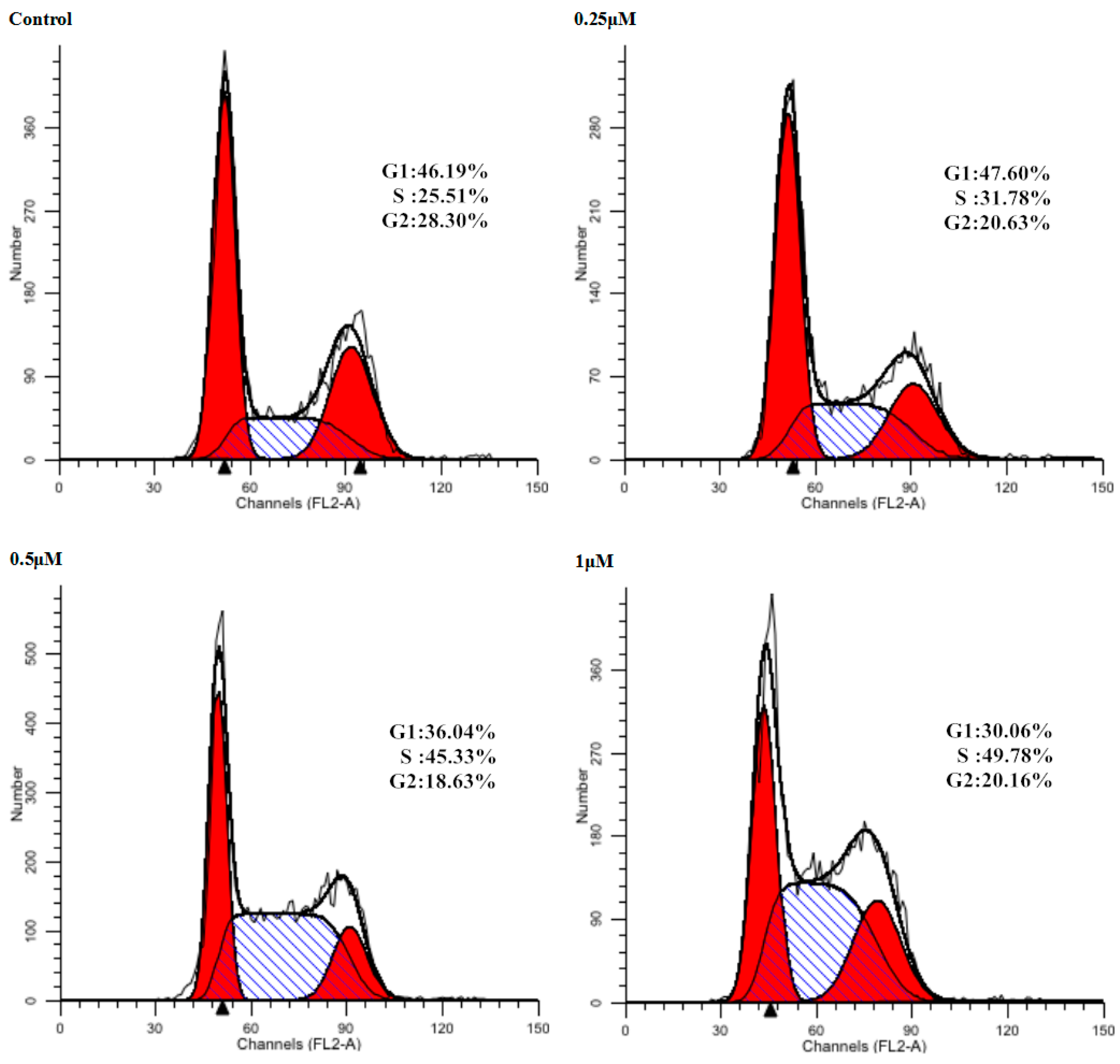
Cell cycle arrest is an important sign for inhibition of proliferation and the series of events that take place in a cell leading to its division and replication. Some NO donating hybrids exhibited cell cycle arrest properties [28]. As the inhibitory effect of 9f on cell proliferation was observed, we next assessed the effect on the cell cycle distribution of Bel-7402 cells by flow cytometry (Figure 2). Treatment of Bel-7402 cells with 9f at 0.25, 0.5 and 1 μM resulted in a remarkable increase in the percentage of cells in S/G2 phase from 25.51% of control group to 31.78%, 45.33%, and 49.78%, respectively. Compound 9f could influence Bel-7402 cell cycle progression at low micromolar concentrations in a dose-dependent manner.
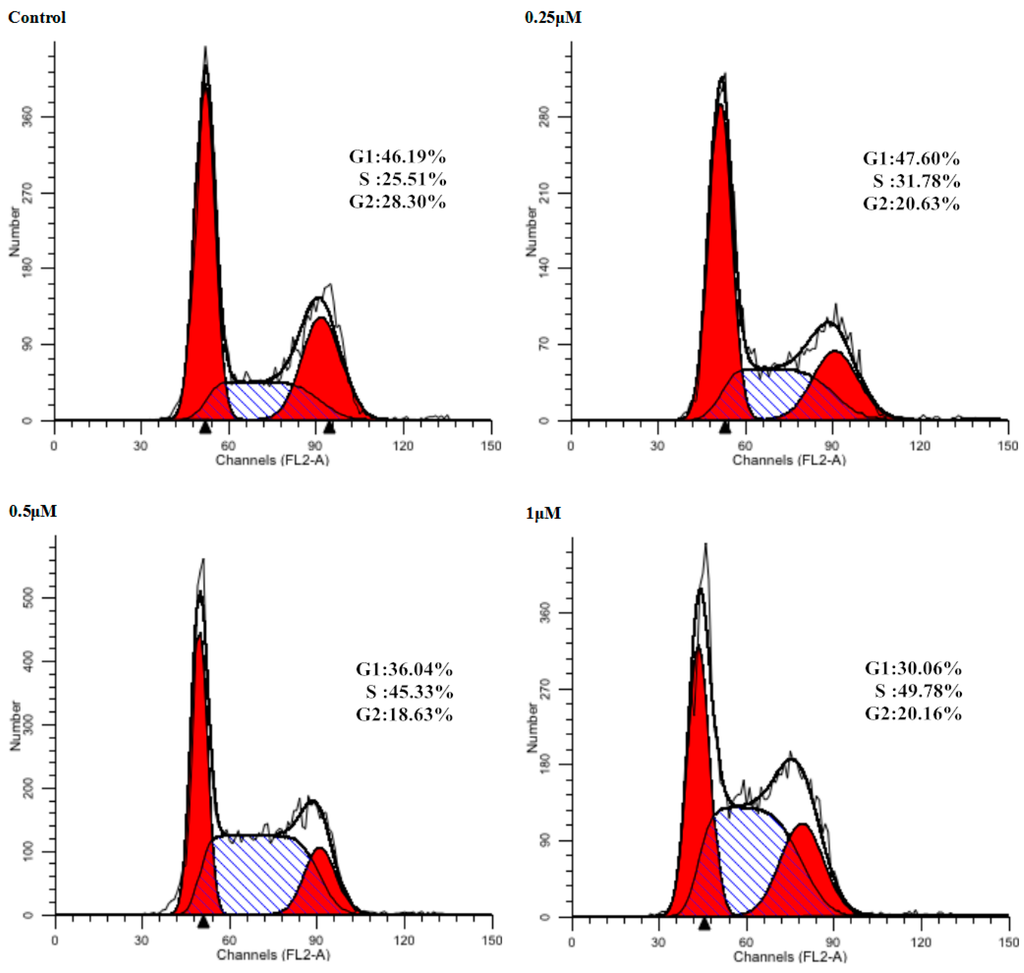
Figure 2.
Influence of Bel-7402 cell cycle by 9f. Left red part: cells in G1 phase; Right red part: cells in G2 phase; Oblique line part: cells in S phase; White part: total cells.
2.6. Induction of Apoptosis by 9f
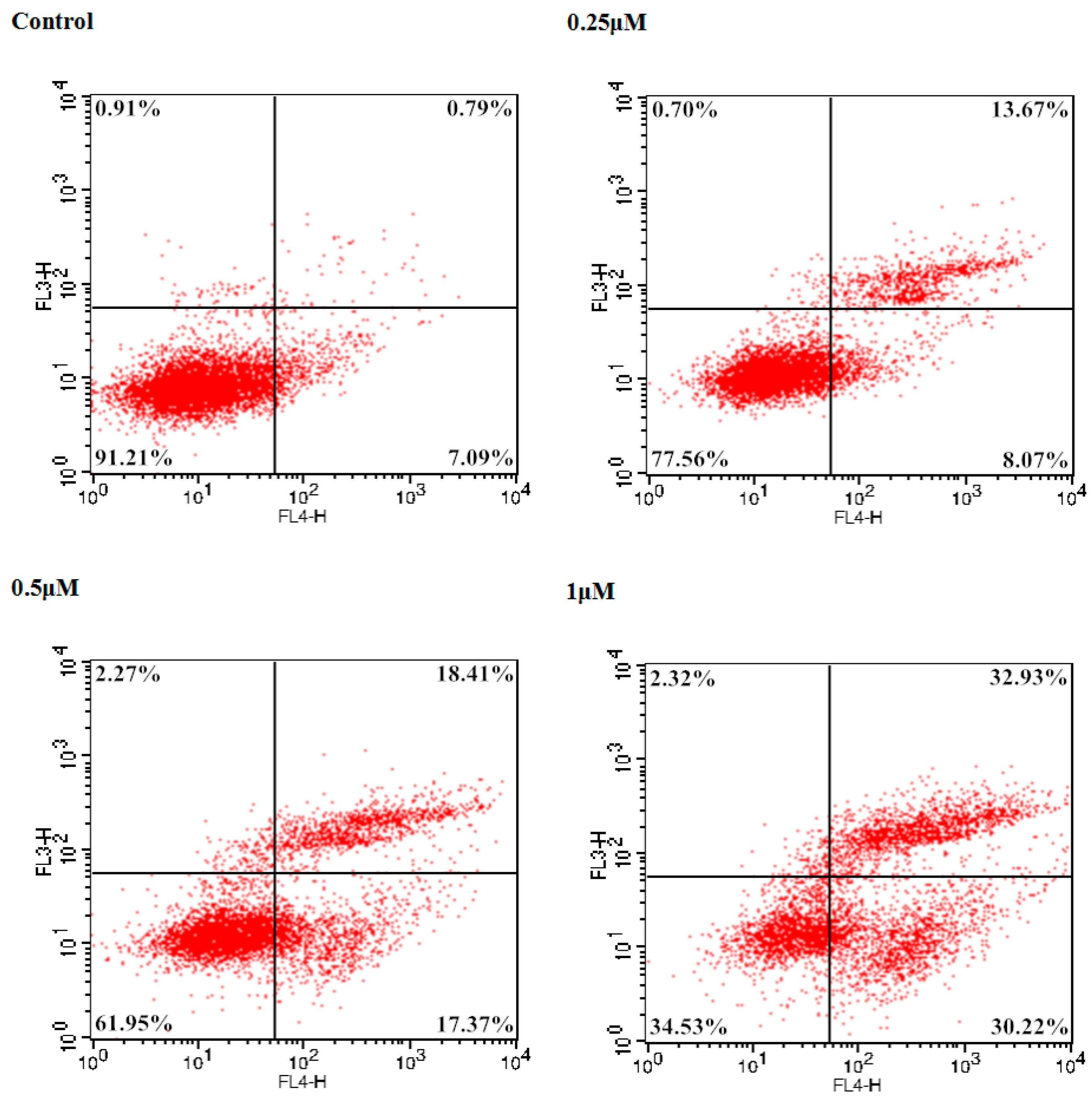
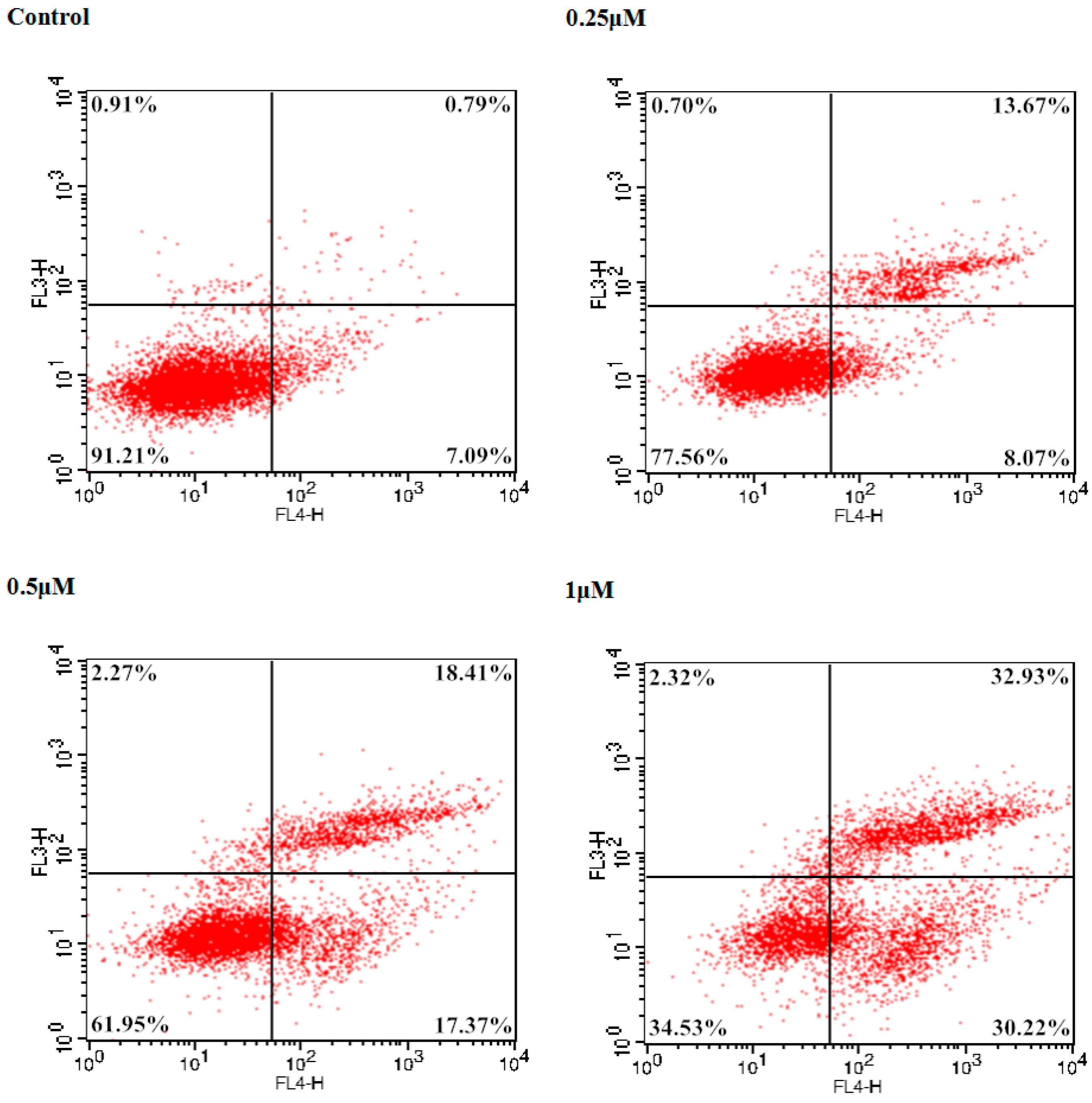
To examine the potent cancer cell antiproliferative effect of 9f on Bel-7402 cells, the number of apoptotic cells was monitored using flow cytometry. The total percentage of apoptotic cells (early and late, Q2 + Q4) was 7.88% treated with a vehicle alone. In comparison with the control group, 2.76-, 4.54-, and 8.01-fold percentages of apoptotic cells were observed when different concentrations (0.25, 0.5 and 1 μM) of 9f were added to Bel-7402 cells. As shown in Figure 3, compound 9f caused significant induction of apoptosis in a dose-dependent manner in Bel-7402 cells and resulted in 21.74%, 35.78% and 63.15% apoptotic cells, respectively.
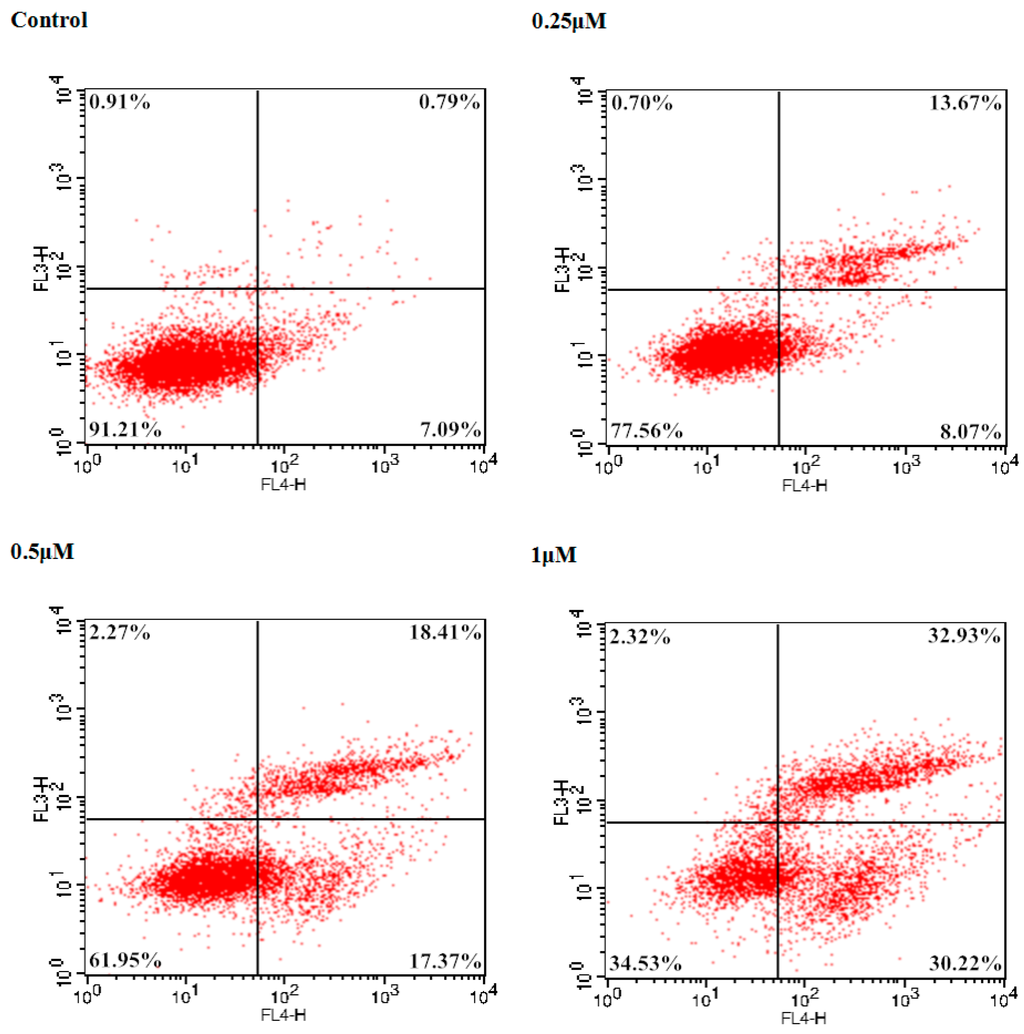
Figure 3.
Induction of apoptosis by 9f in Bel-7402 cells.
2.7. 9f Induced Mitochondrial Depolarization
In the field of cancer, cellular apoptosis plays a very important role. The loss of mitochondrial membrane potential is a key event during drug-induced apoptosis. In order to investigate the mitochondria related effect of 9f, the fluorescent probe JC-1 (Keygen, KGA601, Nanjing, China) was used to detect the changes on mitochondrial membrane potential. Before Bel-7402 cells were stained with JC-1, cells were incubated with 0, 0.15, 0.3, and 0.6 μM of 9f. Then, the flow cytometry analysis was carried out to determine the cell numbers with collapsed mitochondria in different cell groups (Figure 4). The percentage of apoptotic cells increased in a dose-dependent fashion and showed 4.62%, 8.81%, 21.82% and 25.98%, respectively. These results together with Annexin-V and PI double stain apoptosis assay confirmed that 9f could induce apoptosis in a Bel-7402 cells mitochondria-related pathway.
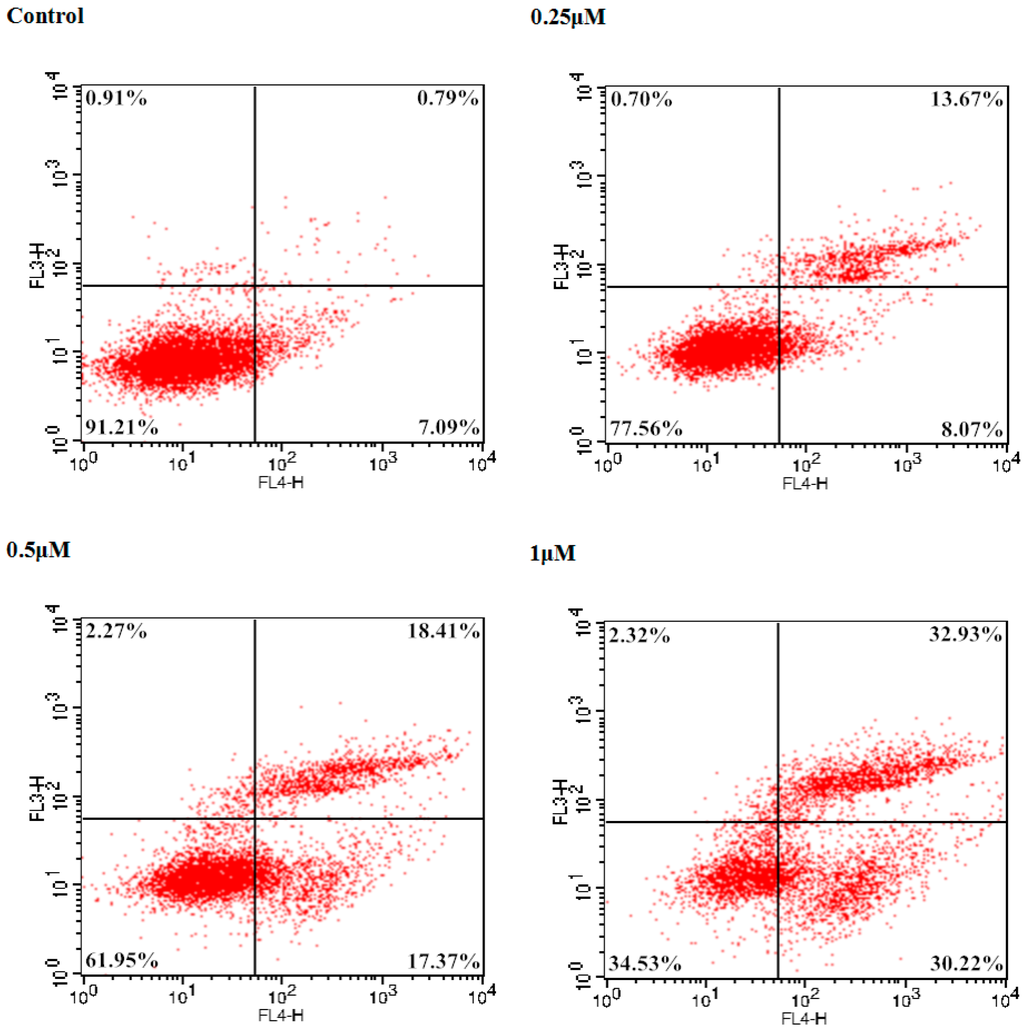
Figure 4.
9f induced mitochondrial depolarization in Bel-7402 cells.
3. Materials and Methods
3.1. Chemistry
3.1.1. General
All commercially available solvents and reagents were purchased from a local commercial supplier (Yuwang Chemical Industries, Ltd., Shenyang, China) and used directly. Melting points (m.p.) were tested using micro melting point apparatus XT-4 (Taike, Beijing, China) and not corrected. Infrared (IR) spectra were recorded on a Nicolet Impact 410 instrument (Thermo Fisher Scientific, Waltham, MA, USA) using a KBr pellet. 1H and 13C NMR spectra were taken (TMS as internal standard) on a Bruker AV-300 (Bruker Corp., Karlsruhe, Germany) or ACF 500 spectrometer (Bruker Corp., Karlsruhe, Germany) in CDCl3. Low resolution mass spectra (MS) were carried out using FTMS-2000 (Thermo Fisher Scientific, Waltham, MA, USA). High resolution mass spectra (HR-MS) were obtained with Agilent QTOF 6520 (Agilent Technologies China, Beijing, China).
3.1.2. General Procedure to Synthesize 9a–i
Compound 8 (72 mg, 0.2 mmol) was dissolved in 15 mL of dichloromethane and mixed with 6a–i (0.24 mmol), EDCI and DMAP, stirring at room temperature for 8 h. When the reaction was finished (monitored by TLC), the reaction mixture was poured into 10% HCl (about 15 mL), and then extracted three times (each 10 mL) with dichloromethane. The organic phases were combined together, washed with water (about 15 mL) and saturated brine (about 15 mL), sequentially, then dried over anhydrous Na2SO4, and evaporated to dryness in reduced pressure. At the last step, the product was purified by flash column chromatography (CH2Cl2/MeOH v/v 300:1) to obtain the target molecules.
Compound 9a: white solid, 48% yield: m.p. 92 °C–94 °C; IR (KBr) υmax 3439, 2959, 2025, 1723, 1615, 1553, 1450, 1371, 734, 685 cm−1; 13C NMR (CDCl3, 100 MHz), δ (ppm) 198.07, 172.91, 172.69, 167.22, 158.99, 147.86, 138.18, 135.83, 129.89 (×2), 128.68 (×2), 120.53, 110.61, 101.90, 76.18, 74.67, 74.09, 68.13, 61.46, 60.34, 53.93, 49.96, 48.60, 40.77, 37.16, 33.12, 33.03, 31.72, 31.13, 29.89, 23.33, 23.16, 19.74; 1H NMR (CDCl3, 300 MHz), δ (ppm) 8.07 (2H, d, J = 7.5 Hz, 2′,6′-Ar–H), 7.78 (1H, d, J = 7.5 Hz, 4′-Ar–H), 7.64 (2H, t, J = 7.5 Hz, 3′,5′-Ar–H), 6.20 (1H, s, 6-OH), 5.74 (1H, s, 17-CH2), 5.62 (1H, s, 17-CH2), 5.32 (1H, s, 6-CH), 4.47~4.65 (6H, m, overlapped, 1-CH, 14-CH, 25,26-CH2), 4.05, 3.92 (each 1H, dd, JA = JB = 9.6 Hz, 20-CH2), 3.16 (1H, d, J = 9.3 Hz, 13-CH), 2.53~2.90 (4H, m, 22,23-CH2), 2.17 (2H, m, 12-CH2), 1.97 (1H, m, 5-CH), 1.61~1.74 (5H, m, 3,11-CH2, 9-CH), 1.45 (2H, m, 2-CH2), 1.01 (3H, s, 18-CH3), 0.95 (3H, s, 19-CH3); MS (ESI) m/z: 748.1 [M + NH4]+, 731.0 [M + H]+, 765.3 [M + Cl]−; HR-MS (ESI, M + NH4) m/z: calcd for C34H42N3O14S: 748.2382, found: 748.2388.
Compound 9b: white solid, 46% yield: m.p. 88 °C–89 °C; IR (KBr) υmax 3442, 2954, 2025,1746, 1618, 1553, 1452, 1358, 732, 685 cm−1; 13C NMR (CDCl3, 100 MHz), δ (ppm) 198.14, 172.73, 172.65, 167.27, 158.79, 147.85, 138.12, 135.87, 129.87 (×2), 128.71 (×2), 120.54, 110.52, 101.87, 76.17, 74.59, 74.12, 69.04, 61.23, 60.35, 53.90, 49.93, 48.58, 40.73, 37.12, 33.00 (×2), 32.89, 31.10, 29.85, 23.29, 23.13, 19.87, 19.61; 1H NMR (CDCl3, 500 M Hz), δ (ppm) 8.00 (2H, d, J = 7.5 Hz, 2′,6′-Ar–H), 7.89 (1H, d, J = 7.5 Hz, 4′-Ar–H), 7.74 (2H, t, J = 7.5 Hz, 3′,5′-Ar–H), 6.18 (1H, s, 6-OH), 5.73 (1H, s, 17-CH2), 5.55 (1H, s, 17-CH2), 5.31 (1H, s, 6-CH), 4.48~4.63 (6H, m, 1-CH, 14-CH, 26,27-CH2), 4.05, 3.92 (each 1H, dd, JA = JB = 9.0 Hz, 20-CH2), 3.14 (1H, d, J = 9.0 Hz, 13-CH), 2.38~2.73 (6H, m, overlapped, 22,23,24-CH2), 2.00 (2H, m, 12-CH2), 1.95 (1H, m, 5-CH), 1.61~1.74 (5H, m, 3,11-CH2, 9-CH), 1.45 (2H, m, 2-CH2), 1.02 (3H, s, 18-CH3), 0.95 (3H, s, 19-CH3); MS (ESI) m/z: 762.3 [M + NH4]+, 745.2 [M + H]+, 779.4 [M + Cl]−; HR-MS (ESI, M + NH4) m/z: calcd for C35H44N3O14S: 762.2539, found: 762.2532.
Compound 9c: white solid, 45% yield: m.p. 137 °C–139 °C; IR (KBr) υmax 3443, 2954, 2025, 1757, 1727, 1617, 1553, 1450, 735, 685 cm−1; 13C NMR (CDCl3, 100 MHz), δ (ppm) 198.21, 167.15, 167.06, 166.58, 158.87, 147.65, 137.95, 135.74, 131.89, 131.71, 131.19, 130.92, 130.14, 129.76 (×2), 128.87, 128.55 (×2); 120.72, 110.49, 101.80, 76.24, 75.27, 74.58, 68.88, 62.45, 60.21, 53.87, 50.14, 48.76, 40.74, 37.00, 32.93, 30.97, 31.08, 29.86, 23.29, 23.07, 19.76; 1H NMR (CDCl3, 300 M Hz), δ (ppm) 8.00 (2H, d, J = 7.5 Hz, 2′,6′-Ar–H), 7.81 (1H, t, J = 4.5 Hz, 4′-Ar–H), 7.69 (1H, t, J = 4.5 Hz, 22-Ar–H), 7.56 (3H, m, 21,23,24-Ar–H), 7.41 (2H, t, J = 7.5 Hz, 3′,5′-Ar–H), 6.24 (1H, s, 6-OH), 5.87 (1H, s, 17-CH2), 5.52 (1H, s, 17-CH2), 5.33 (1H, s, 6-CH), 4.40~4.76 (6H, m, 1-CH, 14-CH, 29,30-CH2), 4.07, 3.96 (each 1H, dd, JA = JB = 9.3 Hz, 20-CH2), 3.29 (1H, d, J = 9.6 Hz, 13-CH), 2.17 (2H, m, 12-CH2), 1.97 (1H, m, 5-CH), 1.52~1.80 (5H, m, 3,11-CH2, 9-CH), 1.41 (2H, m, 2-CH2), 1.02 (3H, s, 18-CH3), 0.98 (3H, s, 19-CH3); MS (ESI) m/z: 801.3 [M + Na]+; HR-MS (ESI, M + NH4) m/z: calcd for C38H42N3O14S: 796.2382, found: 796.2376.
Compound 9d: white solid, 44% yield: m.p. 110 °C–112 °C; IR (KBr) υmax 3459, 2955, 2025, 1754, 1615, 1554, 1451, 1355, 732, 685 cm−1; 13C NMR (CDCl3, 100 MHz), δ (ppm) 197.92, 172.08, 171.73, 167.13, 158.90, 147.69, 138.01, 135.67, 129.73 (×2), 128.60 (×2); 120.46, 110.56, 101.76, 76.04, 74.52, 74.18, 67.91, 60.35, 60.16, 53.79, 49.79, 48.47, 40.59, 36.98, 32.88, 30.99, 29.74, 29.05, 28.65, 27.81, 23.18, 23.03, 19.70; 1H NMR (CDCl3, 300 M Hz), δ (ppm) 8.03 (2H, d, J = 9.0 Hz, 2′,6′-Ar–H), 7.91 (1H, t, J = 8.1 Hz, 4′-Ar–H), 7.75 (2H, t, J = 9.0 Hz, 3′,5′-Ar–H), 6.04 (1H, s, 6-OH), 5.74 (1H, s, 17-CH2), 5.69 (1H, s, 17-CH2), 5.19 (1H, s, 6-CH), 4.68 (2H, m, 1,14-CH), 4.45 (2H, t, J = 6.0 Hz, 25-CH2), 4.14 (2H, t, J = 6.0 Hz, 27-CH2), 3.90, 3.54 (each 1H, dd, JA = JB = 9.0 Hz, 20-CH2), 3.14 (1H, d, J = 5.4 Hz, 13-CH), 2.36~2.63 (6H, m, 22,23,26-CH2), 2.08 (2H, m, 12-CH2), 1.38~1.76 (6H, m, 3,11-CH2, 5,9-CH), 1.43 (2H, m, 2-CH2), 0.93 (3H, s, 18-CH3), 0.85 (3H, s, 19-CH3); MS (ESI) m/z: 767.3 [M + Na]+; HR-MS (ESI, M + NH4) m/z: calcd for C35H44N3O14S: 762.2539, found: 762.2551.
Compound 9e: white solid, 42% yield: m.p. 82 °C–84 °C; IR (KBr) υmax 3458, 2955, 2025, 1738, 1615, 1554, 1451, 1358, 732, 685 cm−1; 13C NMR (CDCl3, 100 MHz), δ (ppm) 197.96, 172.78, 172.56, 167.11, 158.85, 147.73, 138.02, 135.71, 129.76 (×2), 128.53 (×2), 120.40, 110.47, 101.75, 76.05, 74.49, 73.96, 68.00, 61.33, 60.16, 53.79, 49.82, 48.46, 40.63, 37.01, 32.97, 32.89, 31.57, 30.99, 29.74, 27.88, 23.19, 23.03, 19.75, 19.60; 1H NMR (CDCl3, 300 M Hz), δ (ppm) 8.02 (2H, d, J = 8.4 Hz, 2′,6′-Ar–H), 7.90 (1H, t, J = 7.5 Hz, 4′-Ar–H), 7.74 (2H, t, J = 8.4 Hz, 3′,5′-Ar–H), 6.25 (1H, d, J = 2.2 Hz, 6-OH), 6.01 (1H, s, 17-CH2), 5.77 (1H, s, 14-CH), 5.68 (1H, s, 17-CH2), 5.19 (1H, d, J = 2.4 Hz, 6-CH), 4.71 (1H, m, 1-CH), 4.55 (2H, t, J = 6.0 Hz, 26-CH2), 4.12 (2H, t, J = 6.0 Hz, 28-CH2), 3.88, 3.35 (each 1H, dd, JA = JB = 9.0 Hz, 20-CH2), 3.17 (1H, d, J = 9.3 Hz, 13-CH), 2.21~2.51 (8H, m, overlapped, 22,23,24,27-CH2), 2.07 (2H, m, 12-CH2), 1.67~1.78 (6H, m, overlapped, 3,11-CH2, 5,9-CH), 1.24 (2H, m, 2-CH2), 0.94 (3H, s, 18-CH3), 0.88 (3H, s, 19-CH3); MS (ESI) m/z: 781.4 [M + Na]+; HR-MS (ESI, M + NH4) m/z: calcd for C36H46N3O14S: 776.2695, found: 776.2687.
Compound 9f: white solid, 28% yield: m.p. 128 °C–130 °C; IR (KBr) υmax 3445, 2956, 2025, 1758, 1723, 1616, 1554, 1451, 735, 685 cm−1; 13C NMR (CDCl3, 100 MHz), δ (ppm) 198.23, 167.13, 167.02, 166.55, 158.86, 147.67, 137.94, 135.77, 131.91, 131.67, 131.18, 130.94, 130.15, 129.76 (×2), 128.85, 128.54 (×2); 120.75, 110.51, 101.84, 76.27, 75.29, 74.54, 68.91, 62.48, 60.23, 53.91, 50.09, 48.78, 40.72, 37.02, 32.97, 30.99, 31.07, 29.86, 29.79, 23.30, 23.12, 19.75; 1H NMR (CDCl3, 300 M Hz), δ (ppm) 8.07 (2H, d, J = 7.0 Hz, 2′,6′-Ar–H), 7.75 (2H, m, 22,4′-Ar–H), 7.64 (3H, m, 21,23,24-Ar–H), 7.57 (2H, m, 3′,5′-Ar–H), 6.21 (1H, s, 6-OH), 5.92 (1H, s, 17-CH2), 5.56 (1H, s, 17-CH2), 5.32 (1H, s, 6-CH), 4.31~4.60 (6H, m, overlapped, 1-CH, 14-CH, 29,31-CH2), 4.08, 3.97 (each 1H, dd, JA = JB = 9.0 Hz, 20-CH2), 3.26 (1H, d, J = 8.7 Hz, 13-CH), 2.57~2.78 (2H, m, 30-CH2), 2.35 (2H, m, 12-CH2), 1.95 (1H, m, 5-CH), 1.42~1.78 (5H, m, 3,11-CH2, 9-CH), 1.24 (2H, m, 2-CH2), 1.03 (3H, s, 18-CH3), 0.96 (3H, s, 19-CH3); MS (ESI) m/z: 815.4 [M + Na]+; HR-MS (ESI, M + NH4) m/z: calcd for C39H44N3O14S: 810.2539, found: 810.2530.
Compound 9g: white solid, 37% yield: m.p. 85 °C–87 °C; IR (KBr) υmax 3442, 2956, 2025, 1755, 1616, 1554, 1452, 1360, 733, 685 cm−1; 13C NMR (CDCl3, 100 MHz), δ (ppm) 197.92, 172.21, 172.00, 167.09, 158.93, 147.69, 138.01, 135.67, 129.72 (×2), 128.56 (×2); 120.45, 110.47, 101.76, 76.03, 74.55, 74.16, 70.98, 63.86, 60.14, 53.78, 49.80, 48.46, 40.59, 36.99, 32.88, 30.99, 29.74, 29.09, 28.74, 25.18, 24.95, 23.19, 23.02, 19.73; 1H NMR (CDCl3, 300 M Hz), δ (ppm) 8.06 (2H, d, J = 7.8 Hz, 2′,6′-Ar–H), 7.90 (1H, t, J = 7.2 Hz, 4′-Ar–H), 7.65 (2H, t, J = 7.8 Hz, 3′,5′-Ar–H), 6.25 (1H, s, 6-OH), 6.03 (1H, s, 17-CH2), 5.77 (1H, s, 14-CH), 5.68 (1H, s, 17-CH2), 5.19 (1H, s, 6-CH), 4.62 (1H, m, 1-CH), 4.42 (2H, m, 25-CH2), 4.07 (2H, m, 28-CH2), 3.90, 3.54 (each 1H, dd, JA = JB = 10.5 Hz, 20-CH2), 3.14 (1H, d, J = 9.6 Hz, 13-CH), 2.47~2.54 (8H, m, overlapped, 22,23,26,27-CH2), 1.66~1.81 (8H, m, overlapped, 3,11,12-CH2, 5,9-CH), 1.24 (2H, m, 2-CH2), 0.93 (3H, s, 18-CH3), 0.88 (3H, s, 19-CH3); MS (ESI) m/z: 776.3 [M + NH4]+, 793.2 [M + Cl]−; HR-MS (ESI, M + NH4) m/z: calcd for C36H46N3O14S: 776.2695, found: 776.2693.
Compound 9h: white solid, 42% yield: m.p. 78 °C–80 °C; IR (KBr) υmax 3444, 2960, 2025, 1755, 1616, 1554, 1452, 1365, 732, 685 cm−1; 13C NMR (CDCl3, 100 MHz), δ (ppm) 198.10, 172.34, 172.11, 167.26, 159.04, 147.69, 138.16, 135.81, 129.85 (×2), 128.66 (×2); 120.58, 110.58, 101.85, 76.18, 74.57, 74.30, 71.11, 63.98, 60.28, 53.90, 49.92, 48.57, 40.71, 37.09, 32.98, 31.09, 29.84, 29.20, 28.86, 25.29, 25.05, 23.30, 23.13, 22.76, 19.83; 1H NMR (CDCl3, 300 M Hz), δ (ppm) 8.06 (2H, d, J = 7.2 Hz, 2′,6′-Ar–H), 7.77 (1H, t, J = 7.2 Hz, 4′-Ar–H), 7.63 (2H, t, J = 7.2 Hz, 3′,5′-Ar–H), 6.21 (1H, s, 6-OH), 5.73 (1H, s, 17-CH2), 5.57 (1H, s, 17-CH2), 5.32 (1H, m, 6-CH), 4.57 (2H, m, 1,14-CH), 4.45 (2H, t, J = 6.3 Hz, 26-CH2), 4.14 (2H, t, J = 6.0 Hz, 29-CH2), 4.06, 3.93 (each 1H, dd, JA = JB = 9.3 Hz, 20-CH2), 3.13 (1H, d, J = 9.6 Hz, 13-CH), 2.45~2.50 (10H, m, overlapped, 22,23,24,27,28-CH2), 2.35 (2H, m, 12-CH2), 1.58~1.98 (6H, m, overlapped, 3,11-CH2, 5,9-CH), 1.24 (2H, m, 2-CH2), 0.93 (3H, s, 18-CH3), 0.87 (3H, s, 19-CH3); MS (ESI) m/z: 795.4 [M + Na]+; HR-MS (ESI, M + NH4) m/z: calcd for C37H48N3O14S: 790.2852, found: 790.2841.
Compound 9i: white solid, 43% yield: m.p. 126 °C–128 °C; IR (KBr) υmax 3443, 2955, 2025, 1758, 1723, 1615, 1553, 1451, 734, 685 cm−1; 13C NMR (CDCl3, 100 MHz), δ (ppm) 198.12, 167.09, 166.90, 166.51, 158.96, 147.64, 137.97, 135.72, 131.71, 131.41, 131.22, 130.89, 130.16, 129.75 (×2), 128.64, 128.53 (×2); 120.58, 110.49, 101.76, 76.13, 74.98, 74.56, 70.94, 64.72, 60.15, 53.79, 49.97, 48.69, 40.61, 36.97, 32.88, 31.00, 29.80, 25.26, 24.95, 23.23, 23.02, 19.76; 1H NMR (CDCl3, 300 M Hz), δ (ppm) 8.07 (2H, d, J = 7.8 Hz, 2′,6′-Ar–H), 7.73 (3H, m, 23,25,26-Ar–H), 7.60 (2H, m, 24,4′-Ar–H), 7.50 (2H, t, J = 7.8 Hz, 3′,5′-Ar–H), 6.21 (1H, s, 6-OH), 5.93 (1H, s, 17-CH2), 5.56 (1H, s, 17-CH2), 5.34 (1H, m, 6-CH), 4.68 (2H, m, 1,14-CH), 4.40~4.62 (4H, m, 29,32-CH2), 4.08, 3.97 (each 1H, dd, JA = JB = 9.0 Hz, 20-CH2), 3.30 (1H, d, J = 9.0 Hz, 13-CH), 2.79 (2H, m, 30-CH2), 2.62 (2H, m, 31-CH2), 2.42 (2H, m, 12-CH2), 2.03 (1H, m, 5-CH), 1.97 (1H, m, 9-CH), 1.70~1.81 (4H, m, 3,11-CH2), 1.24(2H, m, 2-CH2), 1.03 (3H, s, 18-CH3), 0.96 (3H, s, 19-CH3); MS (ESI) m/z: 829.4 [M + Na]+; HR-MS (ESI, M + NH4) m/z: calcd for C40H46N3O14S: 824.2695, found: 824.2697.
3.2. Biology
3.2.1. Antibacterial Assay
The minimal inhibitory concentrations (MICs) were carried out by microbroth dilution method described by the Clinical Laboratory Standards Institute [30,31]. A stock solution of all the target compounds was prepared with DMSO-medium (1:2), and then graded quantities of the test compounds were diluted with medium. The specified concentration suspension of fungus and bacterium contained approximately 103, and 105 CFU/mL was added into microtitration plates. The inoculated 96-well plates were incubated at 35 °C for 18 h. MIC was defined as the lowest concentrations that prevented visible growth of the bacteria.
3.2.2. MTT Assay
MTT assay was developed to monitor mammalian cell survival and proliferation in vitro [32]. CaEs-17 cells (K562, Bel-7402 and MGC-803 cells) from American Type Culture Collection (ATCC, Manassas, VA, USA) were cultivated in RPMI-1640 medium supplemented with 5% fetal bovine serum (v/v), 100 U/mL penicillin, and 50 mg/mL streptomycin. Cells (5 × 104 cell/mL) at the log phase of their growth cycle were added to each well of a 96-well plate (100 µL/well) and incubated for 24 h at 37 °C in a humidified atmosphere of 5% CO2. Then, cells were treated with or without test compounds in different concentrations. After 72 h, 5 mg/mL MTT solution (20 µL per well) was added. Cells were incubated at 37 °C. After 4 h, MTT solution was removed and DMSO was added to each well (150 µL). Ten minutes later at room temperature, the optical density (OD) values were measured at the wavelength of 490 nm on a Microplate Reader NO. 550 (BIO-RAD Instruments Inc., Hercules, CA, USA). In this experiment, 10 µg/mL of Taxol was used as the positive control and 0.1% DMSO was used as the negative reference.
3.2.3. Griess Assay
NO produced by the compounds was determined by assaying the levels of NO2 using the Griess reagent [33]. The levels of NO released from 100 μM of each compound were tested by nitrate/nitrite in triplicate. The lysates were reacted with Griess reagent (40 min), centrifugalized (10 min), and then measured at 540 nm. This assay was carried out according to the manufacturer’s instructions for 60 min (Beyotime, Nanjing, China).
3.2.4. Cell Cycle Arrest
Bel-7402 cells (5.0 × 103 cells/well) were plated in 6-well plates and incubated for 24 h, and then incubated with different concentrations (0.25, 0.5 and 1 μM) of 9f at 37 °C for 48 h. After 48 h treatment, cells were treated with 70% ethanol and RNase, sequentially, and stained with PI [34]. Cellular DNA content was measured by a flow cytometer for cell cycle distribution analysis.
3.2.5. Cellular Apoptosis
Bel-7402 cells were incubated with different concentrations (0.25, 0.5 and 1 μM) of 9f for 72 h. Then, Annexin-V and PI double staining was used to detect apoptotic cells to analyze apoptosis according to the manufacturer’s instructions by flow cytometry [35].
3.2.6. Mitochondrial Membrane Potential
Bel-7402 cells were cultured in six-well plates after treatment with 9f or vehicles for 48 h. The cells were cells were collected by trypsinization, washed with PBS (Sigma Chemical Co., Shanghai, China) and stained according to the manufacturer’s instruction (Keygen, KGA601, Nanjing, China) with the lipophilic cationic dye JC-1. The flow cytometry analysis was carried out to monitor the percentage of cells with healthy or collapsed mitochondrial membrane potentials [36].
4. Conclusions
In summary, a series of NO donor/enmein-type diterpenoid derivatives (9a–i) were synthesized from relevant commercial available resources. All of the target compounds showed antibacterial activity against Gram-positive bacteria S. aureus and B. subtilis and antiproliferative activity against the K562 leukemia cell line, the MGC-803 gastric cancer cell line, the CaEs-17 esophageal cancer cell line, the Bel-7402 hepatoma cell line, and human normal liver cells L-02 to some extent. It was found that 9b and 9d with the same total linkage length of five carbons were the most active against S. aureus and B. subtilis with MICs of 4 and 2 μg/mL, respectively. Compound 9f displayed IC50 values of 1.68, 1.11, 3.60 and 0.72 μM against four tested cancer cell lines, respectively, and 18.80 μM against normal liver cells L-02. The SI value was 26.1 between L-02 normal liver and Bel-7402 hepatoma cell lines. The preliminary SARs were also concluded based on the obtained biological evaluation data. Furthermore, it was also found that 9f induced apoptosis via the mitochondria-related pathway and arrested cell cycle of Bel-7402 cells at the S stage. These findings were important to explore new chemical entities with innovative natural scaffolds for the treatment of infection combined with solid tumors.
Supplementary Materials
Supplementary materials can be found at http://www.mdpi.com/1422-0067/17/6/747/s1, including 1H NMR and H-H COSY of 8, 1H and 13C NMR of methyl ester of 6a, and 1H and 13C NMR of 9f.
Acknowledgments
This work was financially supported by the National Natural Science Foundation of China (81373280, 21502121), the Project Funded by the China Post Doctoral Science Foundation (2015M570258), General Scientific Research Projects of the Department of Education in Liaoning Province (L2014382), the Key Laboratory for the Chemistry and Molecular Engineering of Medicinal Resources (Guangxi Normal University), the Ministry of the Education of China (CMEMR2015-B07), the Jiangsu Key Laboratory of New Drug Research and Clinical Pharmacy (KF-XY-201401) and the Career Development Support Plan for Young and Middle-aged Teachers in Shenyang Pharmaceutical University.
Author Contributions
Dahong Li, Huiming Hua and Jinyi Xu conceived and designed the experiments; Dahong Li, Tong Han, Shengtao Xu, Tingting Zhou and Keguang Cheng performed the experiments; Dahong Li, Keguang Cheng and Zhanlin Li analyzed the data; Dahong Li, Zhenzhong Wang, Wei Xiao and Jinyi Xu wrote the paper. Xu Hu revised the manuscript and did the corresponding experiments according to the reviewers’ suggestions.
Conflicts of Interest
The authors declare no conflict of interest.
Abbreviations
| NO | Nitric Oxide |
| SAR | Structure Activity Relationship |
| DMAP | 4-Dimethylaminopyridine |
| EDCI | 1-Ethyl-3-(3-dimethylaminopropyl)carbodiimide Hydrochloride |
| rt | Room Temperature |
| THF | Tetrahydrofuran |
| SI | Selectivity Index |
| MTT | 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide |
| TLC | Thin Layer Chromatograph |
| FBS | Fetal Bovine Serum |
| TMS | Tetramethylsilane |
| 1H NMR | Proton Nuclear Magnetic Resonance |
| TMS | Tetramethlysilane |
| ESI MS | Electrospray Ionization-Mass Spectrometry |
| HR-MS | High Resolution Mass Spectrum |
| DCM | Dichloromethane |
| MIC | Minimal Inhibitory Concentration |
| PI | Propidium Iodide |
| NT | Not Test |
References
- Pfeltz, R.F.; Wilkinson, B.J. The escalating challenge of vancomycin resistance in Staphylococcus aureus. Curr. Drug Targets Infect. Disord. 2004, 4, 273–294. [Google Scholar] [CrossRef] [PubMed]
- Roberts, M.C. Distribution of macrolide, lincosamide, streptogramin, ketolide and oxazolidinone (MLSKO) resistance genes in Gram-negative bacteria. Curr. Drug Targets Infect. Disord. 2004, 4, 207–215. [Google Scholar] [CrossRef] [PubMed]
- Hoadley, K.A.; Yau, C.; Wolf, D.M.; Cherniack, A.D.; Tamborero, D.; Ng, S.; Leiserson, M.D.; Niu, B.; McLellan, M.D.; Uzunangelov, V.; et al. Multiplatform analysis of 12 cancer types reveals molecular classification within and across tissues of origin. Cell 2014, 158, 929–944. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.B.; Di, Y.T.; Bao, Y.; Yuan, C.M.; Chen, G.; Li, D.H.; Bai, J.; He, H.P.; Hao, X.J.; Pei, Y.H.; et al. Peganumine A, a β-carboline dimer with a new octacyclic scaffold from Peganum harmala. Org. Lett. 2014, 16, 4028–4031. [Google Scholar] [CrossRef] [PubMed]
- Sai, C.M.; Li, D.H.; Xue, C.M.; Wang, K.B.; Hu, P.; Pei, Y.H.; Bai, J.; Jing, Y.K.; Li, Z.L.; Hua, H.M. Two pairs of enantiomeric alkaloid dimers from Macleaya cordata. Org. Lett. 2015, 17, 4102–4105. [Google Scholar] [CrossRef] [PubMed]
- Rostom, S.A.; Faidallah, H.M.; Radwan, M.F.; Badr, M.H. Bifunctional ethyl 2-amino-4-methylthiazole-5-carboxylate derivatives: Synthesis and in vitro biological evaluation as antimicrobial and anticancer agents. Eur. J. Med. Chem. 2014, 76, 170–181. [Google Scholar] [CrossRef] [PubMed]
- Climo, M.W.; Yokoe, D.S.; Warren, D.K.; Perl, T.M.; Bolon, M.; Herwaldt, L.A.; Weinstein, R.A.; Sepkowitz, K.A.; Jernigan, J.A.; Sanogo, K.; et al. Effect of daily chlorhexidine bathing on hospital-acquired infection. N. Engl. J. Med. 2013, 368, 533–542. [Google Scholar] [CrossRef] [PubMed]
- Vinšová, J.; Kozic, J.; Krátký, M.; Stolaříková, J.; Mandíková, J.; Trejtnar, F.; Buchta, V. Salicylanilide diethyl phosphates: Synthesis, antimicrobial activity and cytotoxicity. Bioorg. Med. Chem. 2014, 22, 728–737. [Google Scholar] [CrossRef] [PubMed]
- Huczyński, A.; Rutkowski, J.; Popiel, K.; Maj, E.; Wietrzyk, J.; Stefańska, J.; Majcher, U.; Bartl, F. Synthesis, antiproliferative and antibacterial evaluation of C-ring modified colchicine analogues. Eur. J. Med. Chem. 2015, 90, 296–301. [Google Scholar] [CrossRef] [PubMed]
- Emirdağ-Öztürk, S.; Karayıldırım, T.; Çapcı-Karagöz, A.; Alankuş-Çalışkan, Ö.; Özmen, A.; Poyrazoğlu-Çoban, E. Synthesis, antimicrobial and cytotoxic activities, and structure-activity relationships of gypsogenin derivatives against human cancer cells. Eur. J. Med. Chem. 2014, 82, 565–573. [Google Scholar] [CrossRef] [PubMed]
- Gu, W.; Hao, Y.; Zhang, G.; Wang, S.F.; Miao, T.T.; Zhang, K.P. Synthesis, in vitro antimicrobial and cytotoxic activities of new carbazole derivatives of ursolic acid. Bioorg. Med. Chem. Lett. 2015, 25, 554–557. [Google Scholar] [CrossRef] [PubMed]
- Harvey, A.L. Natural products in drug discovery. Drug Discov. Today 2008, 13, 894–901. [Google Scholar] [CrossRef] [PubMed]
- Newman, D.J.; Cragg, G.M. Natural products as sources of new drugs over the 30 years from 1981 to 2010. J. Nat. Prod. 2012, 75, 311–335. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.D.; Huang, S.X.; Han, Q.B. Diterpenoids from Isodon species and their biological activities. Nat. Prod. Rep. 2006, 23, 673–698. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Li, D.H.; Wang, C.L.; Zhang, Y.H.; Xu, J.Y. Recent progress in the development of natural ent-kaurane diterpenoids with anti-tumor activity. Mini Rev. Med. Chem. 2011, 11, 910–919. [Google Scholar] [CrossRef] [PubMed]
- Cha, J.Y.; Yeoman, J.T.S.; Reisman, S.E. A concise total synthesis of (−)-maoecrystal Z. J. Am. Chem. Soc. 2011, 133, 14964–14967. [Google Scholar] [CrossRef] [PubMed]
- Gong, J.X.; Lin, G.A.; Sun, W.B.; Li, C.C.; Yang, Z. Total synthesis of (±) maoecrystal V. J. Am. Chem. Soc. 2010, 132, 16745–16746. [Google Scholar] [CrossRef] [PubMed]
- Pan, Z.; Zheng, C.; Wang, H.; Chen, Y.; Li, Y.; Cheng, B.; Zhai, H. Total synthesis of (±)-sculponeatin N. Org. Lett. 2014, 16, 216–219. [Google Scholar] [CrossRef] [PubMed]
- Carpenter, A.W.; Schoenfisch, M.H. Nitric oxide release: Part II. Therapeutic applications. Chem. Soc. Rev. 2012, 41, 3742–3752. [Google Scholar] [CrossRef] [PubMed]
- Bogdan, C. Nitric oxide and the immune response. Nat. Immunol. 2001, 2, 907–916. [Google Scholar] [CrossRef] [PubMed]
- Mocellin, S. Nitric oxide: Cancer target or anticancer agent? Curr. Cancer Drug Targets 2009, 9, 214–236. [Google Scholar] [CrossRef] [PubMed]
- Han, C.; Huang, Z.J.; Zheng, C.; Wan, L.D.; Zhang, L.W.; Peng, S.X.; Ding, K.; Ji, H.B.; Tian, J.D.; Zhang, Y.H. Novel hybrids of (phenylsulfonyl)furoxan and anilinopyrimidine as potent and selective epidermal growth factor receptor inhibitors for intervention of non-small-cell lung cancer. J. Med. Chem. 2013, 56, 4738–4748. [Google Scholar] [CrossRef] [PubMed]
- Ai, Y.; Kang, F.; Huang, Z.; Xue, X.; Lai, Y.; Peng, S.; Tian, J.; Zhang, Y. Synthesis of CDDO-amino acid-nitric oxide donor trihybrids as potential antitumor agents against both drug-sensitive and drug-resistant colon cancer. J. Med. Chem. 2015, 58, 2452–2464. [Google Scholar] [CrossRef] [PubMed]
- Fang, F.C. Antimicrobial reactive oxygen and nitrogen species: Concepts and controversies. Nat. Rev. Microbiol. 2004, 2, 820–832. [Google Scholar] [CrossRef] [PubMed]
- Privett, B.J.; Broadnax, A.D.; Bauman, S.J.; Riccio, D.A.; Schoenfisch, M.H. Examination of bacterial resistance to exogenous nitric oxide. Nitric Oxide 2012, 26, 169–173. [Google Scholar] [CrossRef] [PubMed]
- Backlund, C.J.; Worley, B.V.; Schoenfisch, M.H. Anti-biofilm action of nitric oxide-releasing alkyl-modified poly(amidoamine) dendrimers against Streptococcus mutans. Acta Biomater. 2016, 29, 198–205. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Li, D.; Xu, S.; Cai, H.; Yao, H.; Zhang, Y.; Jiang, J.; Xu, J. The conversion of oridonin to spirolactone-type or enmein-type diterpenoid: Synthesis and biological evaluation of ent-6,7-seco-oridonin derivatives as novel potential anticancer agents. Eur. J. Med. Chem. 2012, 52, 242–250. [Google Scholar] [CrossRef] [PubMed]
- Fu, J.; Liu, L.; Huang, Z.; Lai, Y.; Ji, H.; Peng, S.; Tian, J.; Zhang, Y. Hybrid molecule from O2-(2,4-dinitrophenyl)diazeniumdiolate and oleanolic acid: A glutathione S-transferase π-activated nitric oxide prodrug with selective anti-human hepatocellular carcinoma activity and improved stability. J. Med. Chem. 2013, 56, 4641–4655. [Google Scholar] [CrossRef] [PubMed]
- Fang, L.; Appenroth, D.; Decker, M.; Kiehntopf, M.; Lupp, A.; Peng, S.; Fleck, C.; Zhang, Y.; Lehmann, J. NO-donating tacrine hybrid compounds improve scopolamine-induced cognition impairment and show less hepatotoxicity. J. Med. Chem. 2008, 51, 7666–7669. [Google Scholar] [CrossRef]
- Zhang, X.M.; Guo, H.; Li, Z.S.; Song, F.H.; Wang, W.M.; Dai, H.Q.; Zhang, L.X.; Wang, J.G. Synthesis and evaluation of isatin-β-thiosemicarbazones as novel agents against antibiotic-resistant Gram-positive bacterial species. Eur. J. Med. Chem. 2015, 101, 419–430. [Google Scholar]
- Liao, J.; Yang, F.; Zhang, L.; Chai, X.; Zhao, Q.; Yu, S.; Zou, Y.; Meng, Q.; Wu, Q. Synthesis and biological evaluation of novel fluconazole analogues bearing 1,3,4-oxadiazole moiety as potent antifungal agents. Arch. Pharm. Res. 2015, 38, 470–479. [Google Scholar]
- Chen, T.C.; Wu, C.L.; Lee, C.C.; Chen, C.L.; Yu, D.S.; Huang, H.S. Structure-based hybridization, synthesis and biological evaluation of novel tetracyclic heterocyclic azathioxanthone analogues as potential antitumor agents. Eur. J. Med. Chem. 2015, 103, 615–627. [Google Scholar]
- Bi, Y.; Yang, X.; Zhang, T.; Liu, Z.; Zhang, X.; Lu, J.; Cheng, K.; Xu, J.; Wang, H.; Lv, G.; et al. Design, synthesis, nitric oxide release and antibacterial evaluation of novel nitrated ocotillol-type derivatives. Eur. J. Med. Chem. 2015, 101, 71–80. [Google Scholar]
- Shen, T.; Li, W.; Wang, Y.Y.; Zhong, Q.Q.; Wang, S.Q.; Wang, X.N.; Ren, D.M.; Lou, H.X. Antiproliferative activities of Garcinia bracteata extract and its active ingredient, isobractatin, against human tumor cell lines. Arch. Pharm. Res. 2014, 37, 412–420. [Google Scholar]
- Lepiarczyk, M.; Kałuża, Z.; Bielawska, A.; Czarnomysy, R.; Gornowicz, A.; Bielawski, K. Cytotoxic activity of octahydropyrazin[2,1-a:5,4-a′]diisoquinoline derivatives in human breast cancer cells. Arch. Pharm. Res. 2015, 38, 628–641. [Google Scholar]
- Yugandhar, D.; Nayak, V.L.; Archana, S.; Shekar, K.C.; Srivastava, A.K. Design, synthesis and anticancer properties of novel oxa/azaspiro[4,5]trienones as potent apoptosis inducers through mitochondrial disruption. Eur. J. Med. Chem. 2015, 101, 348–357. [Google Scholar]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).