Effects of a Lysine-Involved Maillard Reaction on the Structure and In Vitro Activities of Polysaccharides from Longan Pulp
Abstract
1. Introduction
2. Results
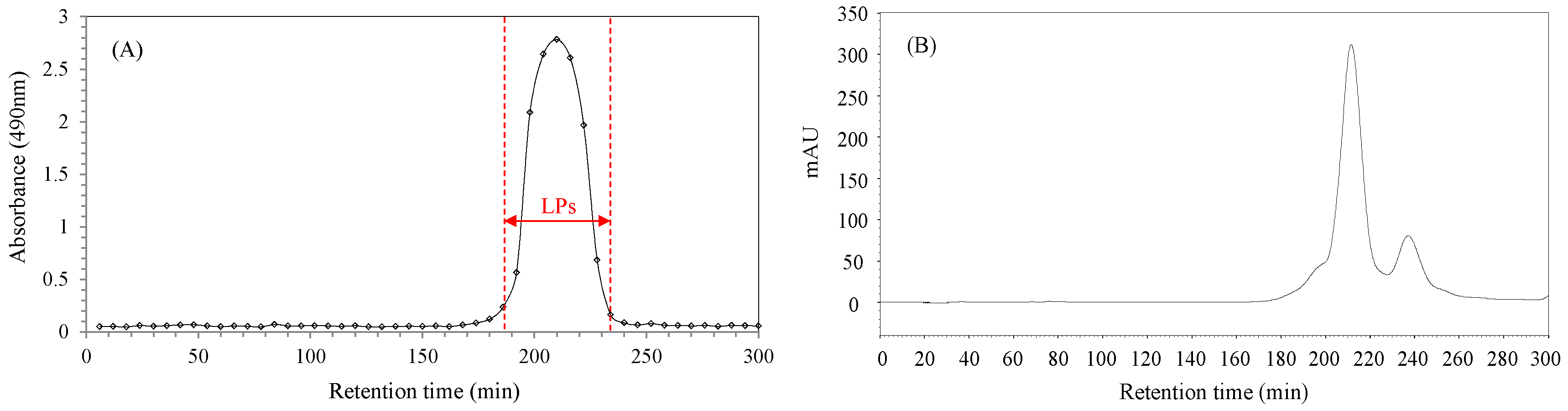
2.1. Purification of Longan Pulp Polysaccharides
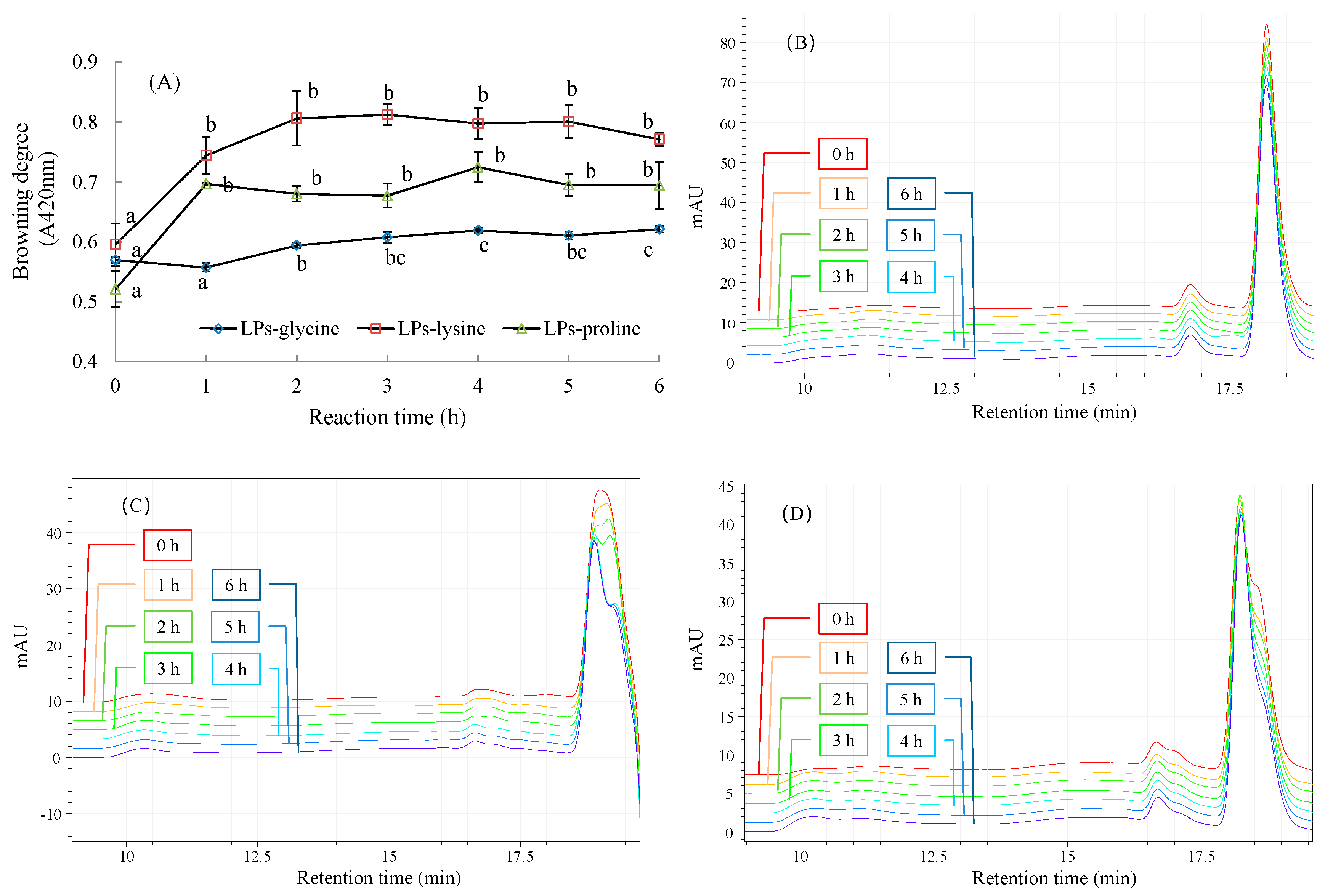
2.2. Physicochemical Changes during MR between LPs and Amino Acids
2.3. Chemical Compositions of LPs and MLPs
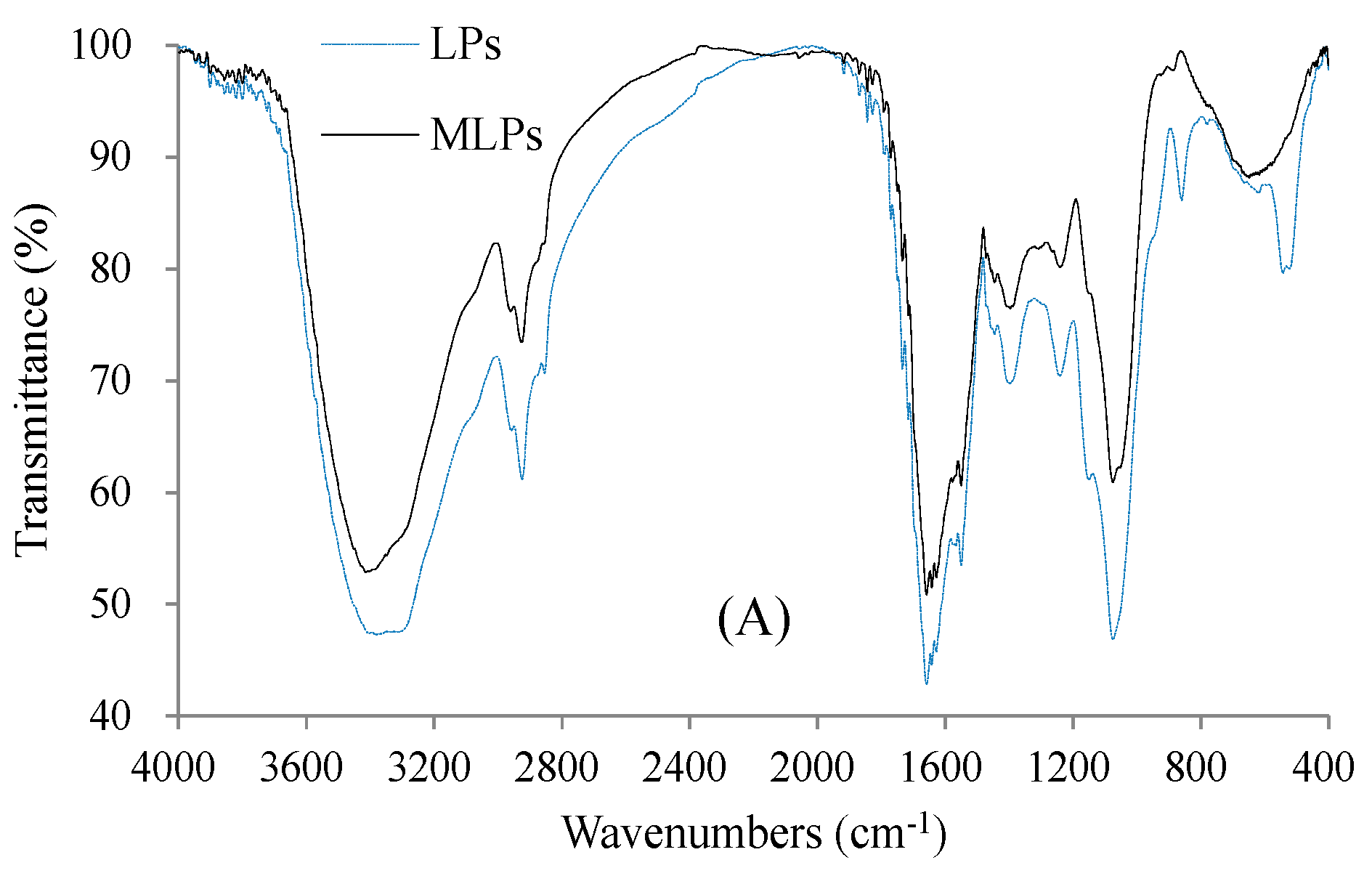
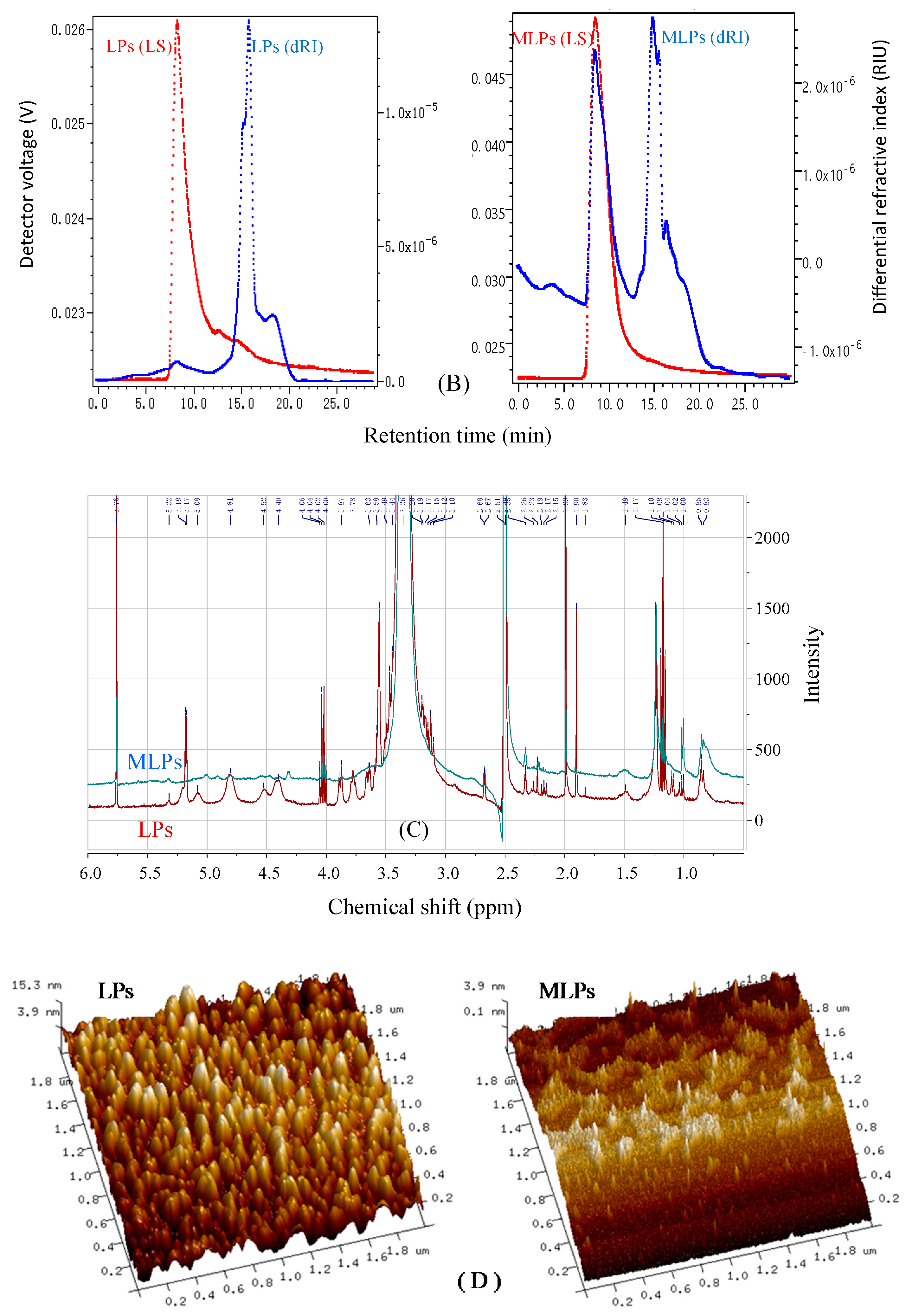
2.4. Structural Features of LPs and MLPs
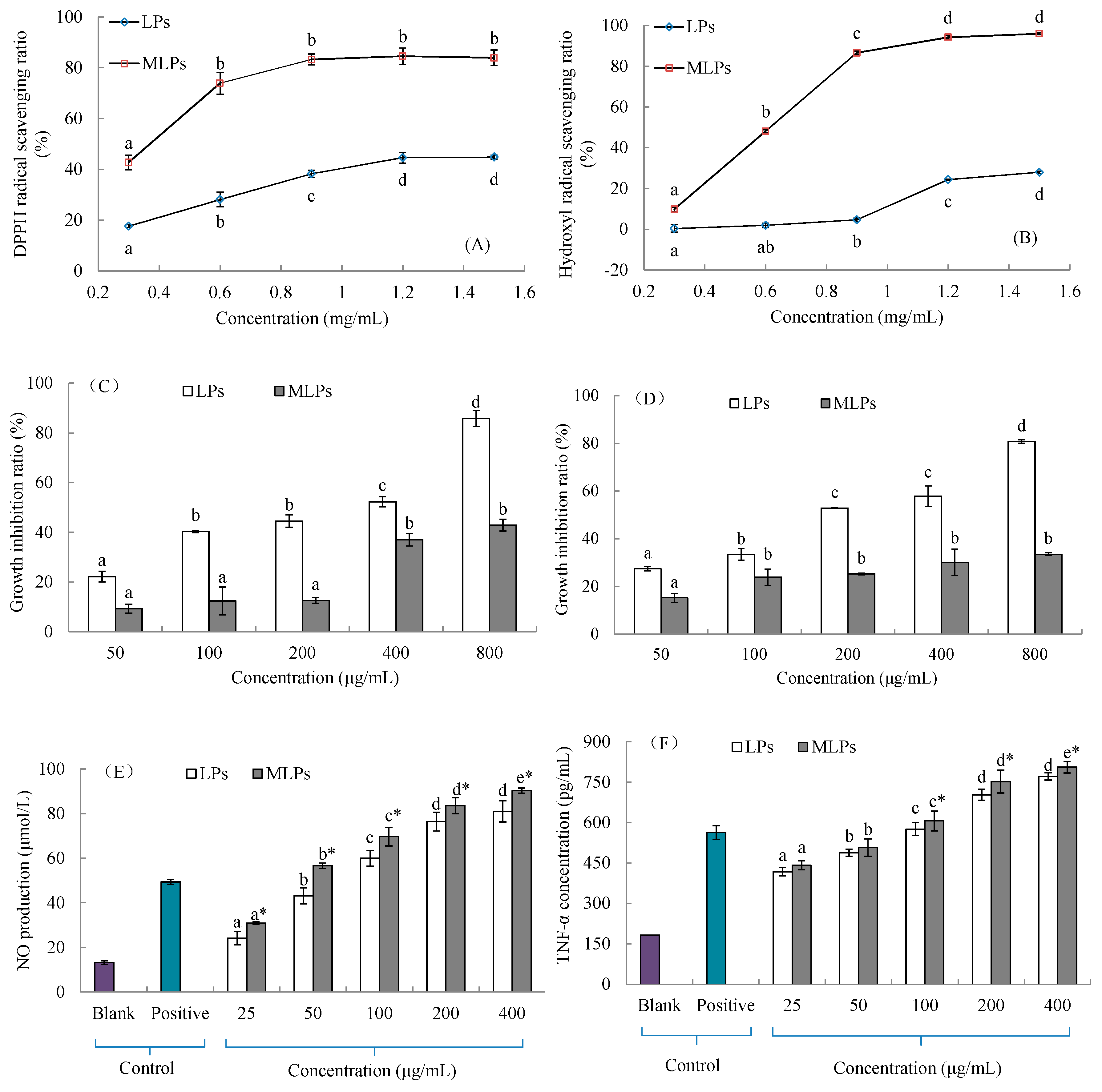
2.5. In Vitro Activities of LPs and MLPs
3. Discussion
3.1. Effects of Lys-Involved MR on the Structural Features of LPs
3.2. Effects of Lysine-Involved MR on the In Vitro Activities of LPs
4. Materials and Methods
4.1. Materials and Cells
4.2. Extraction and Isolation of Longan Pulp Polysaccharides
4.3. MR between LPs and Amino Acids
4.4. Preparation of MR-Modified LPs
4.5. Composition Analysis
4.6. Structural Analysis
4.7. Evaluation of In Vitro Activities
4.8. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Zhu, Q.; Jiang, Y.; Lin, S.; Wen, L.; Wu, D.; Zhao, M.; Chen, F.; Jia, Y.; Yang, B. Structural identification of (1→6)-α-D-Glucan, a key responsible for the health benefits of longan, and evaluation of anticancer activity. Biomacromolecules 2013, 14, 1999–2003. [Google Scholar] [CrossRef] [PubMed]
- Zhong, K.; Wang, Q.; He, Y.; He, X. Evaluation of radicals scavenging, immunity-modulatory and antitumor activities of longan polysaccharides with ultrasonic extraction on in S180 tumor mice models. Int. J. Biol. Macromol. 2010, 47, 356–360. [Google Scholar] [CrossRef] [PubMed]
- Yi, Y.; Wang, H.; Zhang, R.; Min, T.; Huang, F.; Liu, L.; Zhang, M. Characterization of polysaccharide from longan pulp as the macrophage stimulator. RSC Adv. 2015, 5, 97163–97170. [Google Scholar] [CrossRef]
- Han, M.-M.; Yi, Y.; Wang, H.-X.; Huang, F. Investigation of the Maillard reaction between polysaccharides and proteins from longan pulp and the improvement in activities. Molecules 2017, 22, 938. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Qi, C.; Guo, Y.; Zhou, W.; Zhang, Y. Toll-like receptor 4-related immunostimulatory polysaccharides: Primary structure, activity relationships, and possible interaction models. Carbohydr. Polym. 2016, 149, 186–206. [Google Scholar] [CrossRef] [PubMed]
- Jung, H.-J.; Lee, C.O.; Lee, K.-T.; Choi, J.; Park, H.-J. Structure-activity relationship of oleanane disaccharides isolated from Akebia quinata versus cytotoxicity against cancer cells and NO inhibition. Biol. Pharm. Bull. 2004, 27, 744–747. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.; Hu, J.; Wei, L.; Du, Y.; Shi, X.; Zhang, L. Antioxidant and antimicrobial activity of Maillard reaction products from xylan with chitosan/chitooligomer/glucosamine hydrochloride/taurine model systems. Food Chem. 2014, 148, 196–203. [Google Scholar] [CrossRef] [PubMed]
- Muppalla, S.R.; Sonavale, R.; Chawla, S.P.; Sharma, A. Functional properties of nisin–carbohydrate conjugates formed by radiation induced Maillard reaction. Radiat. Phys. Chem. 2012, 81, 1917–1922. [Google Scholar] [CrossRef]
- Lin, D.; Lu, W.; Kelly, A.L.; Zhang, L.; Zheng, B.; Miao, S. Interactions of vegetable proteins with other polymers: Structure-function relationships and applications in the food industry. Trends Food Sci. Technol. 2017, 68, 130–144. [Google Scholar] [CrossRef]
- De Oliveira, F.C.; Coimbra, J.S.; de Oliveira, E.B.; Zuñiga, A.D.G.; Rojas, E.E.G. Food protein-polysaccharide conjugates obtained via the Maillard reaction: A review. Crit. Rev. Food Sci. 2016, 56, 1108–1125. [Google Scholar] [CrossRef] [PubMed]
- Akhtar, M.; Ding, R. Covalently cross-linked proteins & polysaccharides: Formation, characterisation and potential applications. Curr. Opin. Colloid Interface Sci. 2017, 28, 31–36. [Google Scholar]
- Sun, T.; Xu, H.; Zhang, H.; Ding, H.; Cui, S.; Xie, J.; Xue, B.; Hua, X. Maillard reaction of oat β-glucan and the rheological property of its amino acid/peptide conjugates. Food Hydrocoll. 2018, 76, 30–34. [Google Scholar] [CrossRef]
- Han, M.-M.; Yao, J.; Yi, Y.; Huang, F.; Wang, L.-M. Changes of polysaccharides in longan pulp during drying: Physicochemical properties and biological activities. Food Sci. 2017, 38, 67–73. [Google Scholar]
- Zhou, C. The Research on Conversion Pumpkin Hexose to Pentose Using Bacillus subtilis; China Agricultural University: Beijing, China, 2014. [Google Scholar]
- Hu, H.; Liang, H.; Wu, Y. Isolation, purification and structural characterization of polysaccharide from Acanthopanax brachypus. Carbohydr. Polym. 2015, 127, 94–100. [Google Scholar] [CrossRef] [PubMed]
- Yi, Y.; Huang, F.; Zhang, M.-W.; Zhang, R.-F.; Deng, Y.-Y.; Wei, Z.-C.; He, J.-R. Solution properties and in vitro anti-tumor activities of polysaccharides from longan pulp. Molecules 2013, 18, 11601–11613. [Google Scholar] [CrossRef] [PubMed]
- Yi, Y.; Zhang, M.W.; Liao, S.T.; Zhang, R.F.; Deng, Y.Y.; Wei, Z.C.; Tang, X.J.; Zhang, Y. Effects of alkali dissociation on the molecular conformation and immunomodulatory activity of longan pulp polysaccharide LPI. Carbohydr. Polym. 2012, 87, 1311–1317. [Google Scholar] [CrossRef]
- Yang, W.-W.; Wang, L.-M.; Gong, L.-L.; Lu, Y.-M.; Pan, W.-J.; Wang, Y.; Zhang, W.-N.; Chen, Y. Structural characterization and antioxidant activities of a novel polysaccharide fraction from the fruiting bodies of Craterellus cornucopioides. Int. J. Biol. Macromol. 2018, 117, 473–482. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.; Sun, Q.; Xu, J.; Li, N.; Lin, J.; Chen, S.; Li, F. Purification, characterization, and bioactivities of a polysaccharide from mycelial fermentation of Bjerkandera fumosa. Carbohydr. Polym. 2017, 167, 115–122. [Google Scholar] [CrossRef] [PubMed]
- Zou, Y.-F.; Fu, Y.-P.; Chen, X.-F.; Austarheim, I.; Inngjerdingen, K.T.; Huang, C.; Lei, F.-Y.; Song, X.; Li, L.; Ye, G.; et al. Polysaccharides with immunomodulating activity from roots of Gentiana crassicaulis. Carbohydr. Polym. 2017, 172, 306–314. [Google Scholar] [CrossRef] [PubMed]
- Dong, C.-X.; Zhang, W.-S.; Sun, Q.-L.; Jiang, Y.-J.; Fu, Z.; Zhang, Y.; Du, J.; Sun, Y.-X.; Tao, N.; Yao, Z. Structural characterization of a pectin-type polysaccharide from Curcuma kwangsiensis and its effects on reversing MDSC-mediated T cell suppression. Int. J. Biol. Macromol. 2018, 115, 1233–1240. [Google Scholar] [CrossRef] [PubMed]
- Seedevi, P.; Moovendhan, M.; Sudharsan, S.; Sivasankar, P.; Sivakumar, L.; Vairamani, S.; Shanmugam, A. Isolation and chemical characteristics of rhamnose enriched polysaccharide from Grateloupia lithophila. Carbohydr. Polym. 2018, 195, 486–494. [Google Scholar] [CrossRef] [PubMed]
- Balnois, E.; Stoll, S.; Wilkinson, K.J.; Buffle, J.; Rinaudo, M.; Milas, M. Conformations of succinoglycan as observed by atomic force microscopy. Macromolecules 2000, 33, 7440–7447. [Google Scholar] [CrossRef]
- Ke, C.; Qiao, D.; Gan, D.; Sun, Y.; Ye, H.; Zeng, X. Antioxidant acitivity in vitro and in vivo of the capsule polysaccharides from Streptococcus equi subsp. zooepidemicus. Carbohydr. Polym. 2009, 75, 677–682. [Google Scholar] [CrossRef]
- Chen, W.; Zheng, R.; Zeng, H.; Zhang, S. The updated incidences and mortalities of major cancers in China, 2011. Chin. J. Cancer 2015, 34, 53. [Google Scholar] [CrossRef] [PubMed]
- Baxter, J.H. Free amino stability in reducing sugar systems. J. Food Sci. 1995, 60, 405–408. [Google Scholar] [CrossRef]
- Huang, D.; Li, Y.; Cui, F.; Chen, J.; Sun, J. Purification and characterization of a novel polysaccharide–peptide complex from Clinacanthus nutans Lindau leaves. Carbohydr. Polym. 2016, 137, 701–708. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.-R.; Naǵaraj, R.H.; Abraham, E.C. The Role of α- and ε-amino groups in the glycation-mediated cross-linking of γB-crystallin. J. Biol. Chem. 1997, 272, 14465–14469. [Google Scholar] [CrossRef] [PubMed]
- Ha, Y.-M.; Chun, S.-H.; Hong, S.-T.; Koo, Y.-C.; Choi, H.-D.; Lee, K.-W. Immune enhancing effect of a Maillard-type lysozyme-galactomannan conjugate via signaling pathways. Int. J. Biol. Macromol. 2013, 60, 399–404. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.-Y.; Qian, H.; Yao, W.-R. Melanoidins produced by the Maillard reaction: Structure and biological activity. Food Chem. 2011, 128, 573–584. [Google Scholar] [CrossRef]
- Yi, Y.; Lamikanra, O.; Sun, J.; Wang, L.-M.; Min, T.; Wang, H.-X. Activity diversity structure-activity relationship of polysaccharides from lotus root varieties. Carbohydr. Polym. 2018, 190, 67–76. [Google Scholar] [CrossRef] [PubMed]
- Leung, M.Y.K.; Liu, C.; Zhu, L.F.; Hui, Y.Z.; Yu, B.; Fung, K.P. Chemical and biological characterization of a polysaccharide biological response modifier from Aloe vera L. var. chinensis (Haw.) Berg. Glycobiology 2004, 14, 501–510. [Google Scholar] [PubMed]
- Dubois, M.; Gilles, K.A.; Hamilton, J.K.; Pebers, P.A.; Smith, F. Colorimetric method for determination of sugars and related substances. Anal. Chem. 1956, 28, 350–356. [Google Scholar] [CrossRef]
- Xue, F.; Li, C.; Zhu, X.; Wang, L.; Pan, S. Comparative studies on the physicochemical properties of soy protein isolate-maltodextrin and soy protein isolate-gum acacia conjugate prepared through Maillard reaction. Food Res. Int. 2013, 51, 490–495. [Google Scholar] [CrossRef]
- Wang, H.-X.; Yi, Y.; Sun, J.; Lamikanra, O.; Min, T. Fingerprint profiling of polysaccharides from different parts of lotus root varieties. RSC Adv. 2018, 8, 16574–16584. [Google Scholar] [CrossRef]
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C. Use of a free radical method to evaluate antioxidant activity. LWT-Food Sci. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Xiong, S.; Li, A.; Huang, N.; Lu, F.; Hou, D. Antioxidant and immunoregulatory activity of different polysaccharide fractions from tuber of Ophiopogon japonicus. Carbohydr. Polym. 2011, 86, 1273–1280. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds (LPs and MLPs) are available from the authors. |
| Composition | LPs | MLPs | |
|---|---|---|---|
| Polysaccharide content (g/100 g DW) * | 90.11 ± 0.15 | 66.08 ± 0.56 | |
| Protein content (g/100 g DW) | 6.54 ± 0.17 | 5.29 ± 0.10 | |
| Free amino group content (μmol/g DW) | 25.92 ± 0.14 | 16.35 ± 0.44 | |
| Molar ratio of monosaccharide composition | Mannose | 0.16 | 0.19 |
| Ribose | 0.05 | 0.01 | |
| Rhamnose | 0.15 | 0.17 | |
| Glucuronic acid | 0.12 | 0.13 | |
| Galacturonic acid | 0.04 | 0.03 | |
| Glucose | 1.00 | 1.00 | |
| Galactose | 0.21 | 0.24 | |
| Sample | Retention Time (min) | Weight-Average Molecular Weight (Da) | Mass Fraction (%) |
|---|---|---|---|
| LPs | 7.216–11.947 | 8.716 × 105 (±0.863%) | 3.6 |
| 12.005–13.833 | 1.174 × 105 (±1.593%) | 2.4 | |
| 13.862–15.343 | 7.068 × 103 (±2.736%) | 24.6 | |
| 15.372–16.590 | 2.130 × 103 (±4.084%) | 43.0 | |
| 16.619–17.432 | 5.678 × 103 (±4.028%) | 7.7 | |
| 17.461–20.131 | 4.529 × 103 (±4.060%) | 18.7 | |
| MLPs | 7.297–9.288 | 1.004 × 106 (±0.441%) | 23.0 |
| 9.349–12.637 | 6.861 × 105 (±0.414%) | 18.6 | |
| 12.667–13.692 | 2.641 × 105 (±0.565%) | 3.4 | |
| 13.692–15.261 | 4.056 × 104 (±0.867%) | 24.0 | |
| 15.291–15.925 | 2.782 × 104 (±1.096%) | 10.0 | |
| 15.955–16.920 | 4.337 × 104 (±0.984%) | 7.6 | |
| 16.950–17.674 | 4.231 × 104 (±0.908%) | 4.5 | |
| 17.705–20.842 | 4.958 × 104 (±1.361%) | 8.9 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yi, Y.; Han, M.-M.; Huang, F.; Wang, L.-M.; Min, T.; Wang, H.-X. Effects of a Lysine-Involved Maillard Reaction on the Structure and In Vitro Activities of Polysaccharides from Longan Pulp. Molecules 2019, 24, 972. https://doi.org/10.3390/molecules24050972
Yi Y, Han M-M, Huang F, Wang L-M, Min T, Wang H-X. Effects of a Lysine-Involved Maillard Reaction on the Structure and In Vitro Activities of Polysaccharides from Longan Pulp. Molecules. 2019; 24(5):972. https://doi.org/10.3390/molecules24050972
Chicago/Turabian StyleYi, Yang, Miao-Miao Han, Fei Huang, Li-Mei Wang, Ting Min, and Hong-Xun Wang. 2019. "Effects of a Lysine-Involved Maillard Reaction on the Structure and In Vitro Activities of Polysaccharides from Longan Pulp" Molecules 24, no. 5: 972. https://doi.org/10.3390/molecules24050972
APA StyleYi, Y., Han, M.-M., Huang, F., Wang, L.-M., Min, T., & Wang, H.-X. (2019). Effects of a Lysine-Involved Maillard Reaction on the Structure and In Vitro Activities of Polysaccharides from Longan Pulp. Molecules, 24(5), 972. https://doi.org/10.3390/molecules24050972




