Recent Advances in AIEgens for Metal Ion Biosensing and Bioimaging
Abstract
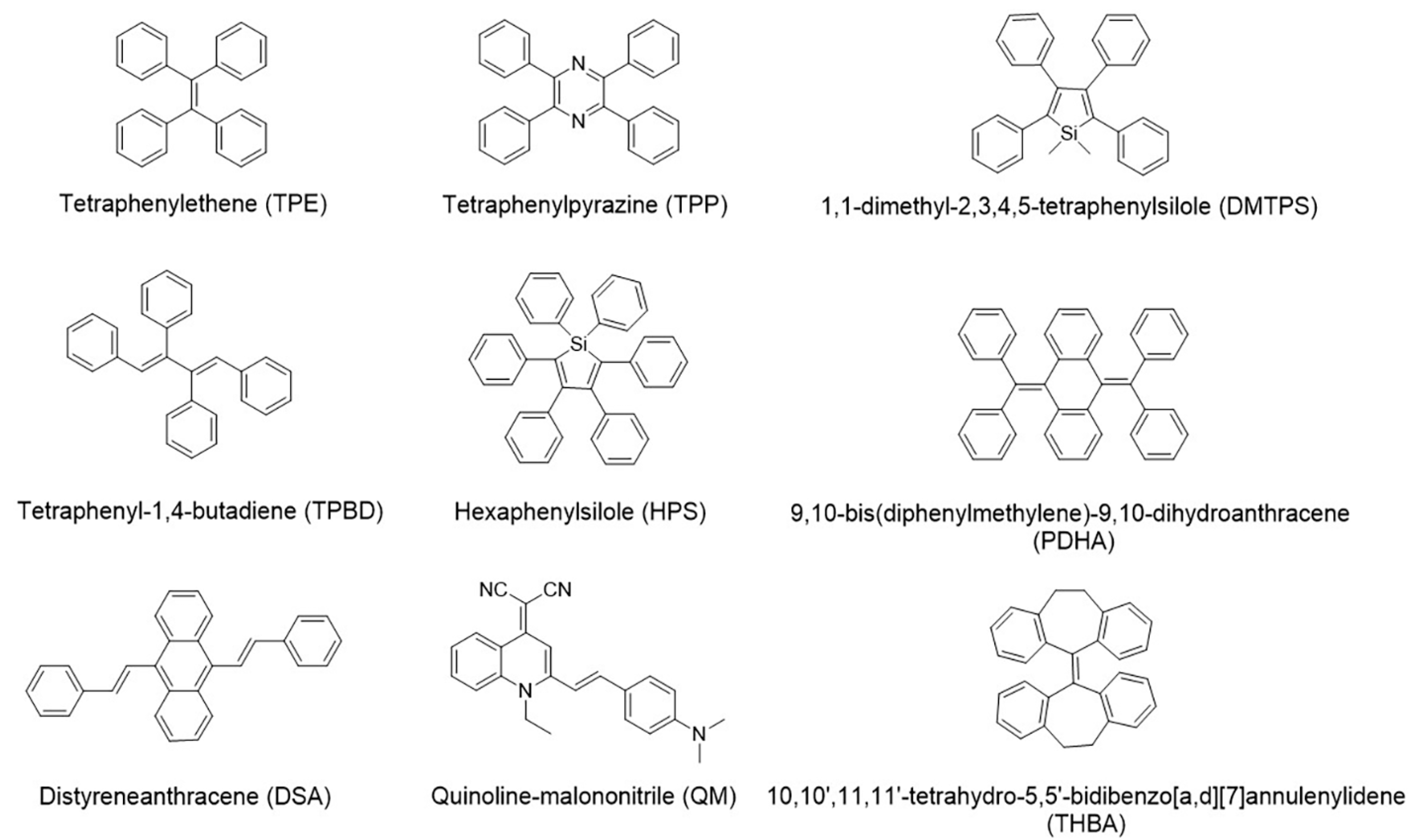
1. Introduction
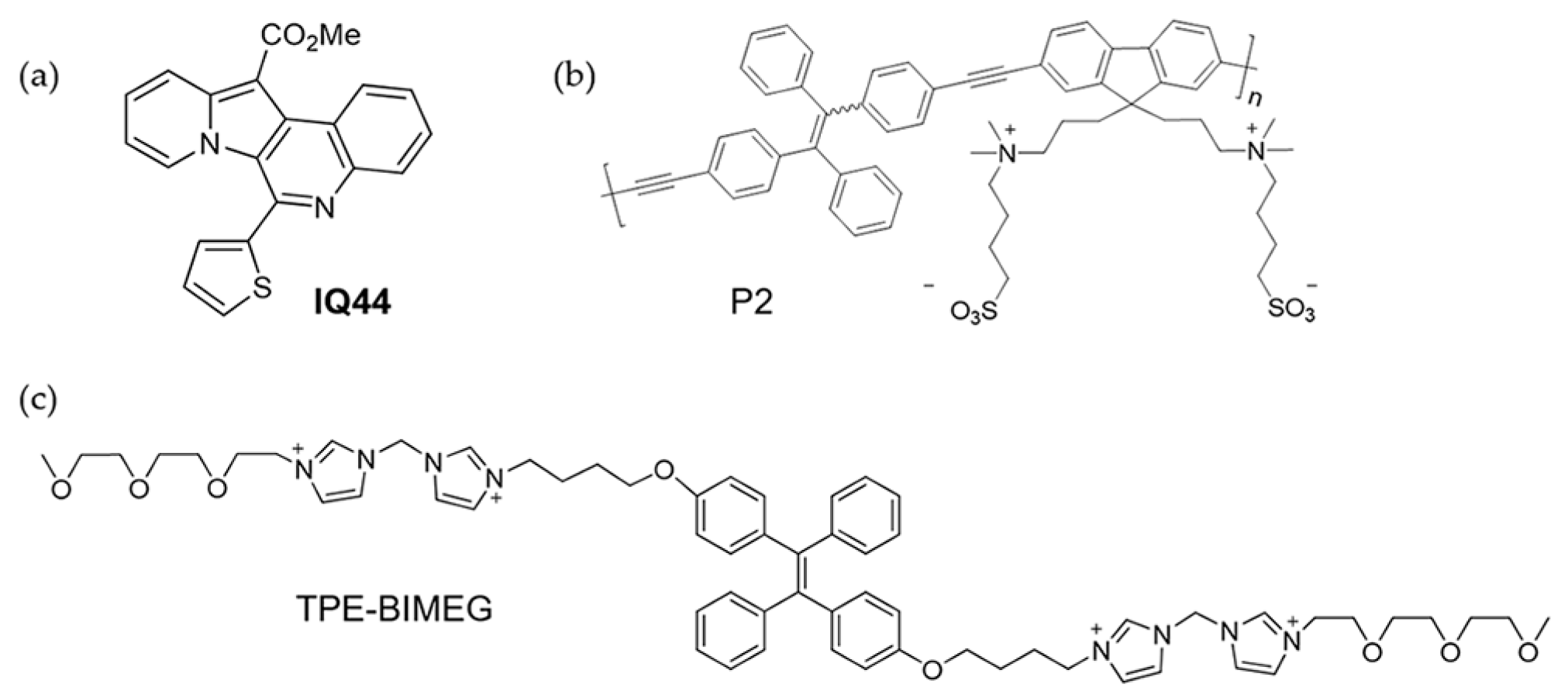
2. Alkali Metal Ions
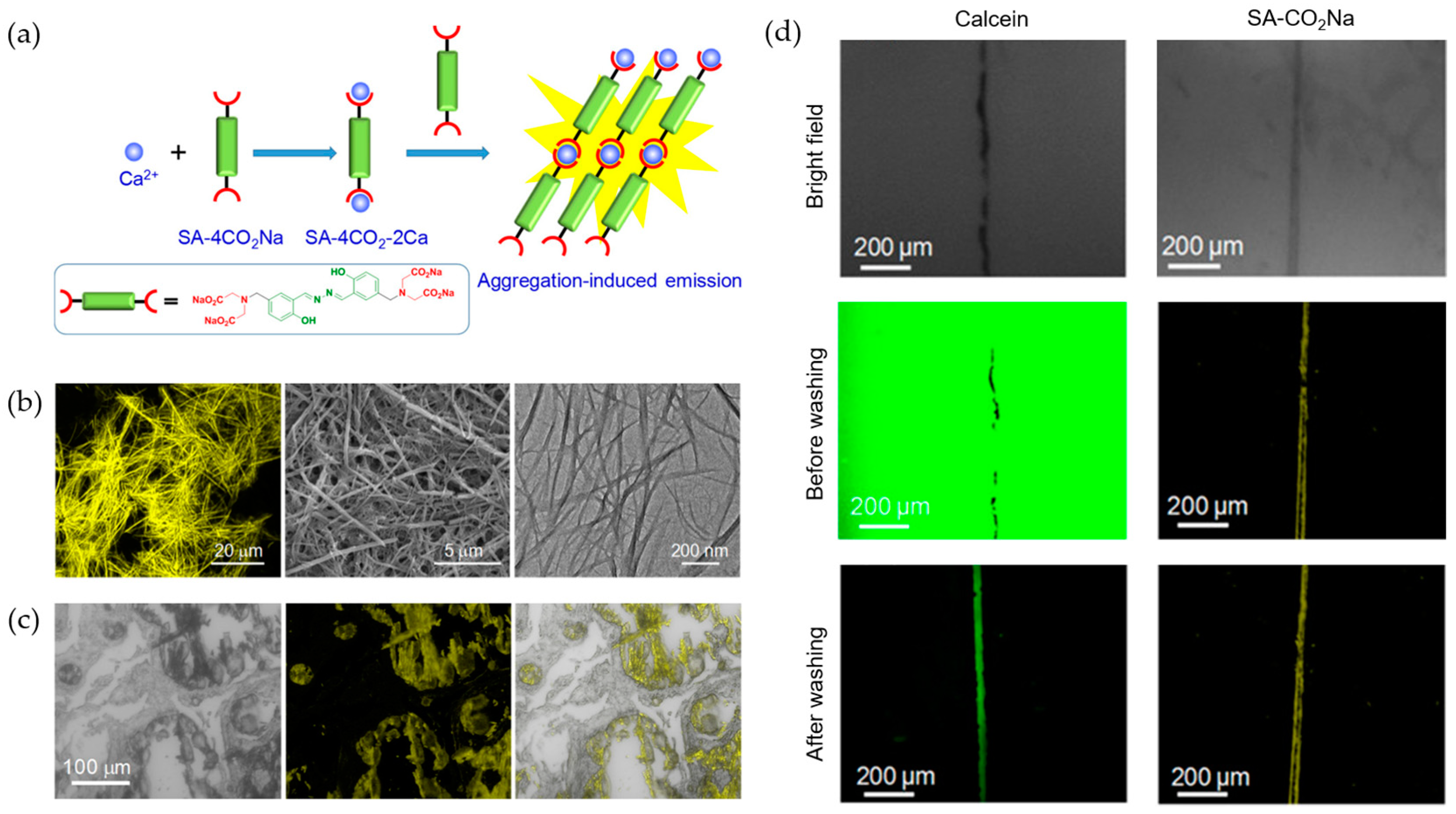
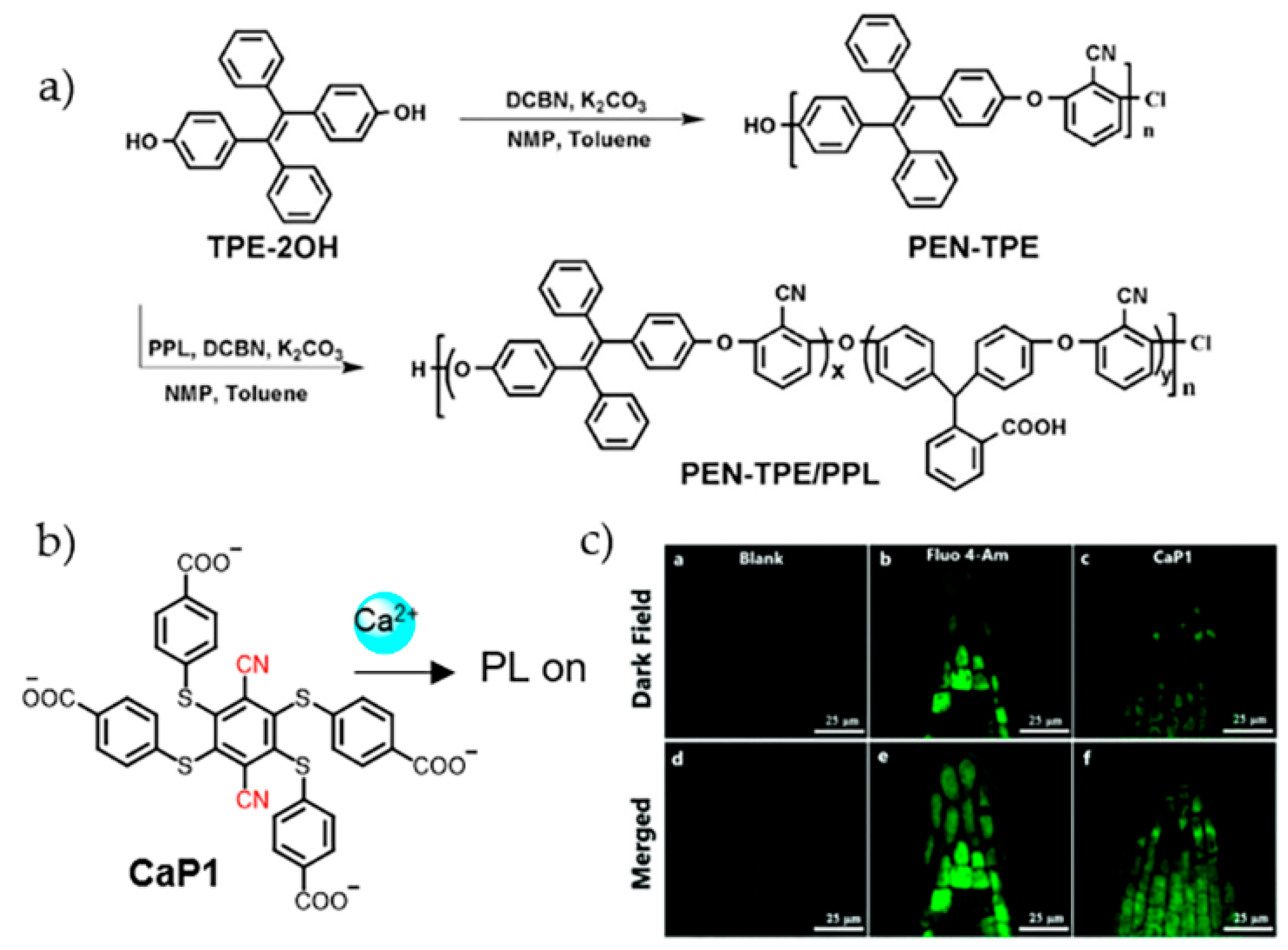
3. Alkaline Earth Metal Ions
4. Transition Metal Ions
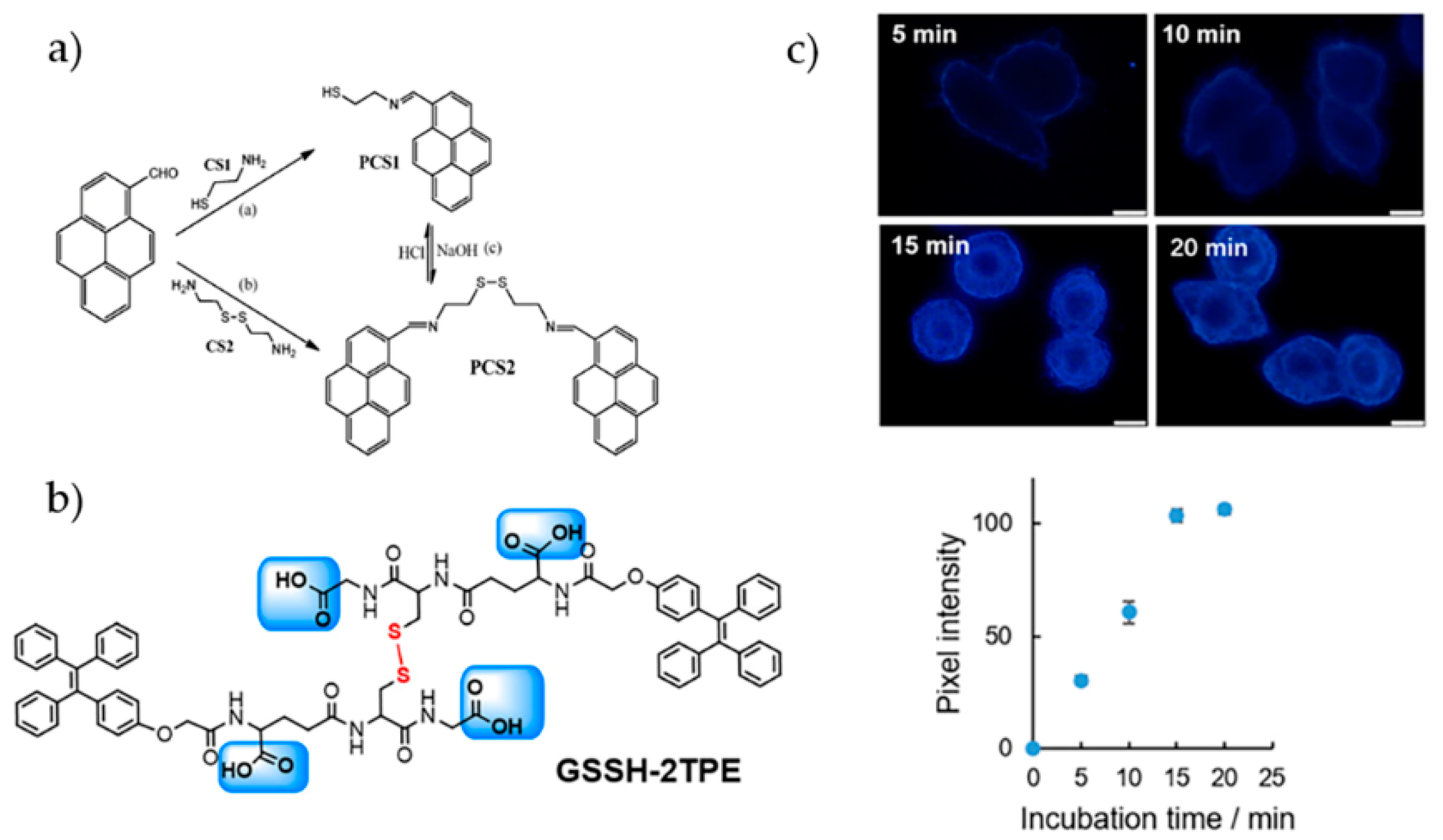
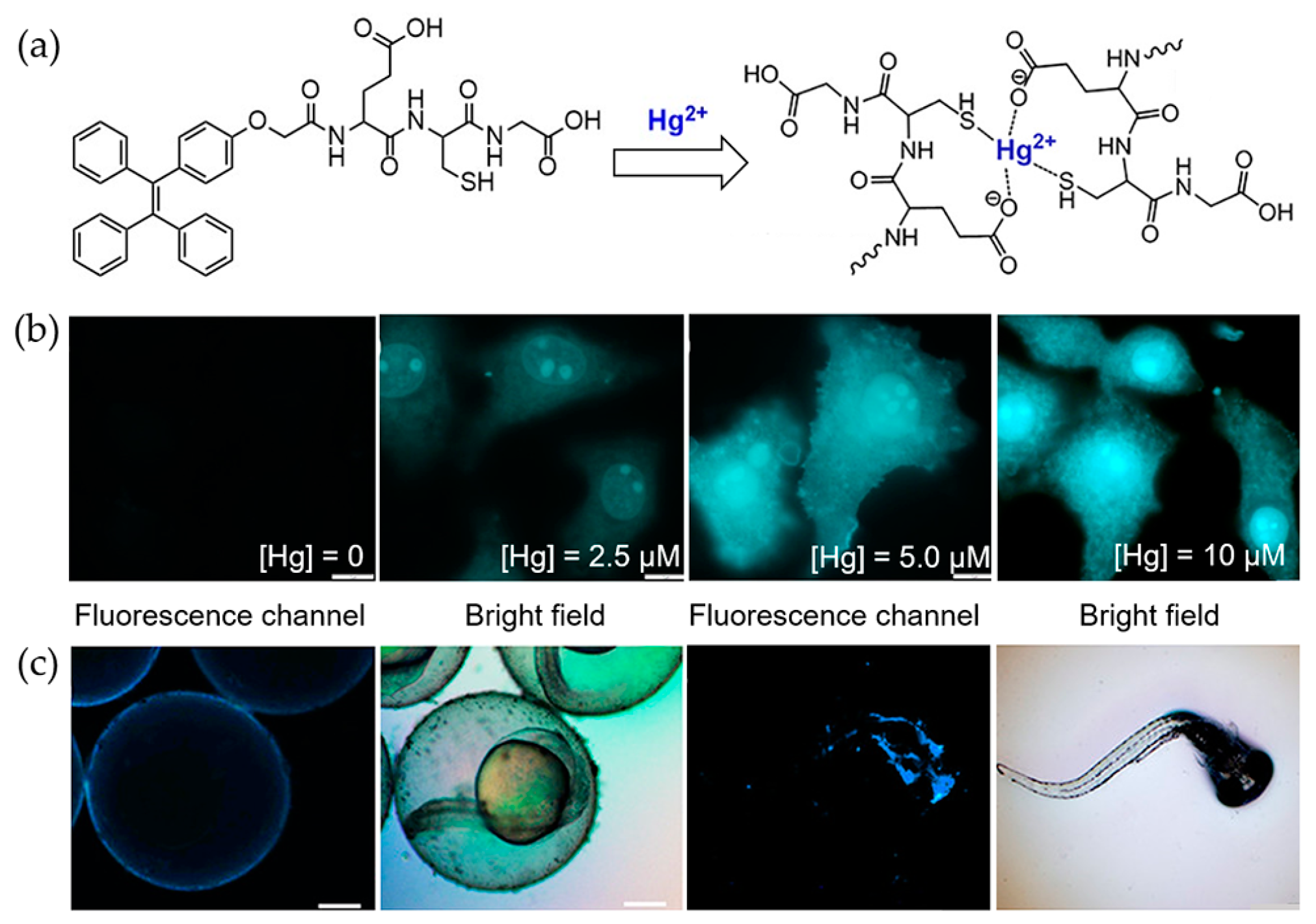
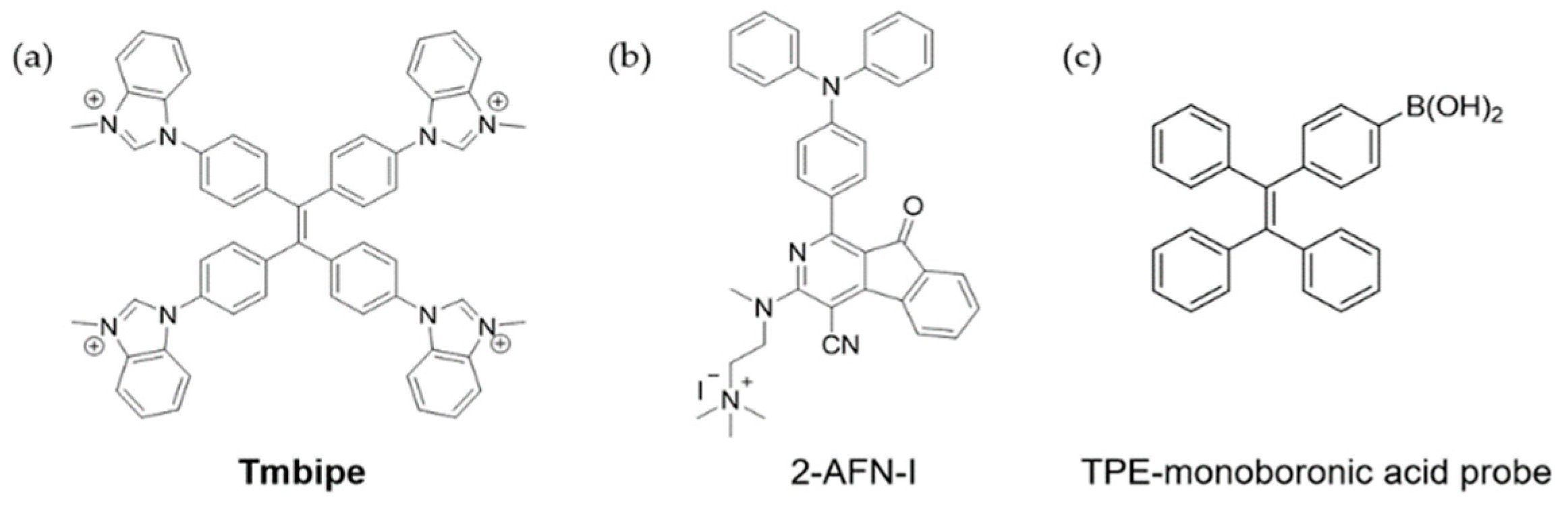
4.1. Mercury Ions
4.2. Cu2+
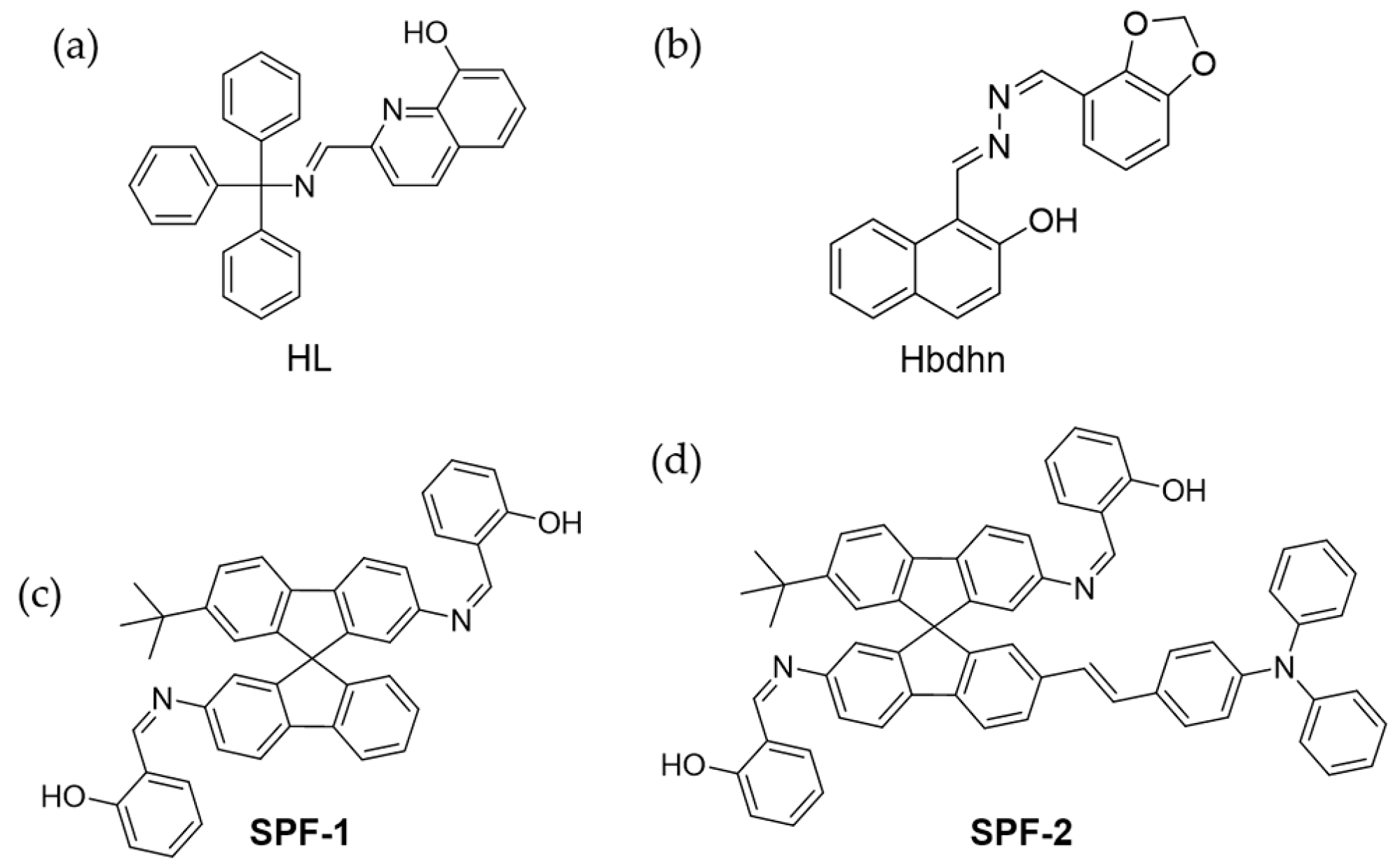
4.3. Zn2+
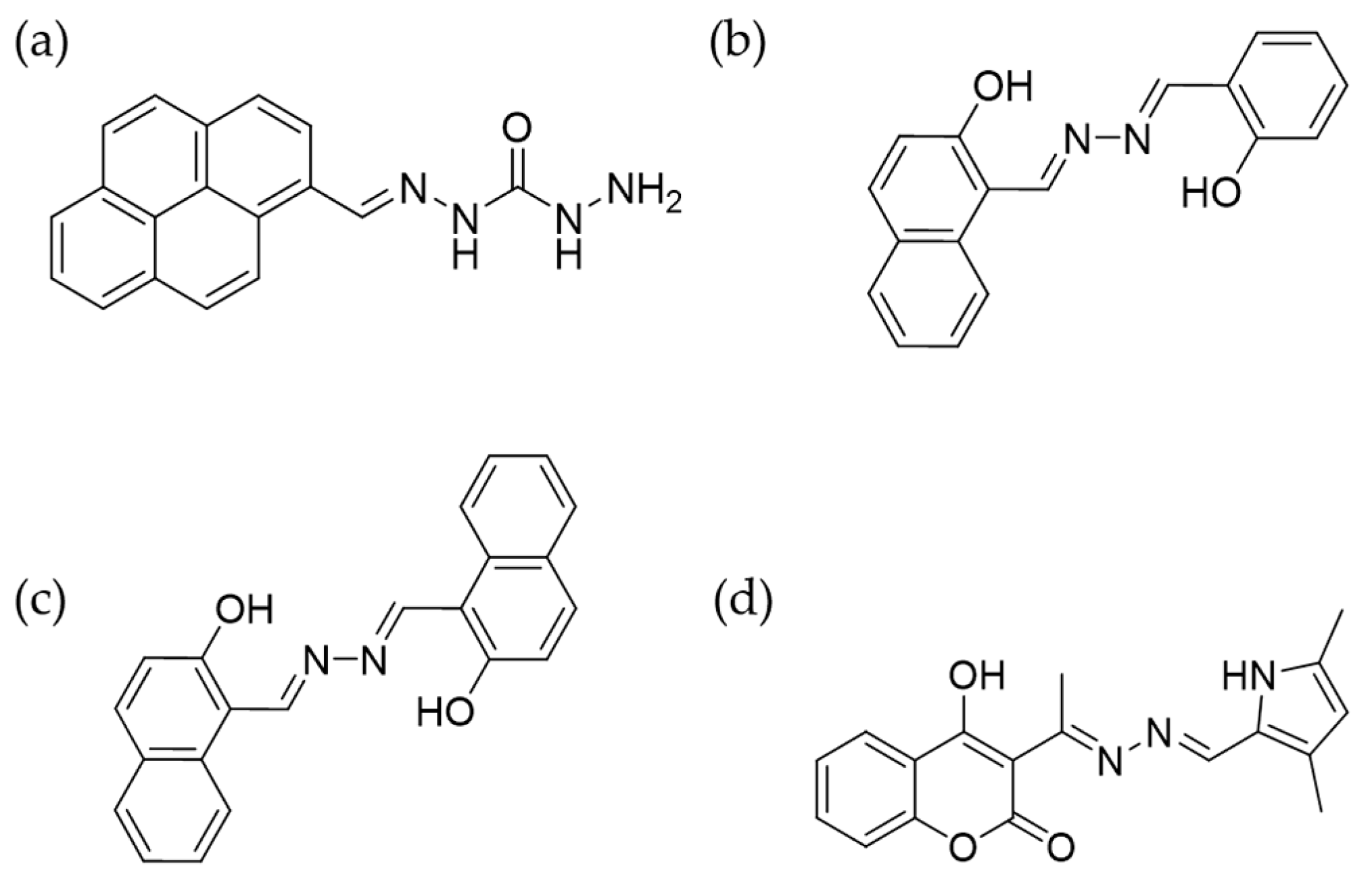
4.4. Fe3+
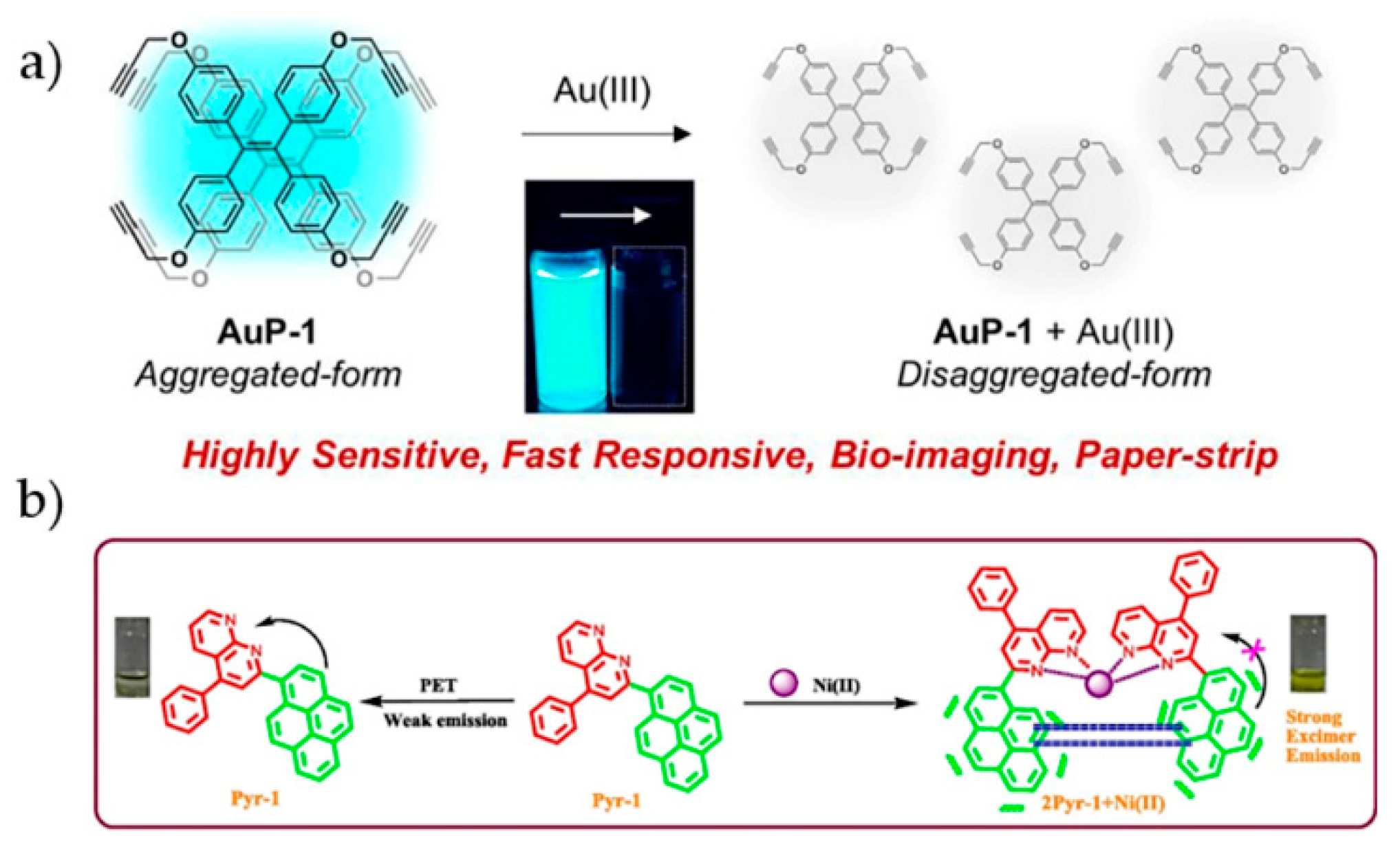
4.5. Other Transition Metal Ions
5. Other Metal Ions
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Carter, K.P.; Young, A.M.; Palmer, A.E. Fluorescent sensors for measuring metal ions in living systems. Chem. Rev. 2014, 114, 4564–4601. [Google Scholar] [CrossRef] [PubMed]
- Bear, M.F.; Connors, B.; Paradiso, M. Neuroscience: Exploring the Brain, 3rd ed.; Lippincott Williams & Wilkins: Baltimore, MD, USA, 2015. [Google Scholar]
- Bischof, H.; Burgstaller, S.; Waldeck-Weiermair, M.; Rauter, T.; Schinagl, M.; Ramadani-Muja, J.; Graier, W.F.; Malli, R. Live-cell imaging of physiologically relevant metal ions using genetically encoded FRET-based probes. Cells 2019, 8, 492. [Google Scholar] [CrossRef] [PubMed]
- Nelson, D.L.; Cox, M.M. Lehninger Principles of Biochemistry, 6th ed.; Worth Publishers: New York, NY, USA, 2012. [Google Scholar]
- Weaver, R.F. Molecular Biology, 5th ed.; McGraw-Hill: New York, NY, USA, 2013. [Google Scholar]
- Rauk, A. The chemistry of Alzheimer’s disease. Chem. Soc. Rev. 2009, 38, 2698–2715. [Google Scholar] [CrossRef] [PubMed]
- Qian, X.H.; Xu, Z.C. Fluorescence imaging of metal ions implicated in diseases. Chem. Soc. Rev. 2015, 44, 4487–4493. [Google Scholar] [CrossRef]
- Ratte, H.T. Bioaccumulation and toxicity of silver compounds: A review. Environ. Toxicol. Chem. 1999, 18, 89–108. [Google Scholar] [CrossRef]
- McLaughlin, M.J.; Parker, D.R.; Clarke, J.M. Metals and micronutrients-food safety issues. Field Crop. Res. 1999, 60, 143–163. [Google Scholar] [CrossRef]
- Waisberg, M.; Joseph, P.; Hale, B.; Beyersmann, D. Molecular and cellular mechanisms of cadmium carcinogenesis. Toxicology 2003, 192, 95–117. [Google Scholar] [CrossRef]
- de Silva, A.P.; Gunaratne, H.Q.N.; Gunnlaugsson, T.; Huxley, A.J.M.; McCoy, C.P.; Rademacher, J.T.; Rice, T.E. Signaling recognition events with fluorescent sensors and switches. Chem. Rev. 1997, 97, 1515–1566. [Google Scholar] [CrossRef]
- Lee, M.H.; Kim, J.S.; Sessler, J.L. Small molecule-based ratiometric fluorescence probes for cations, anions, and biomolecules. Chem. Soc. Rev. 2015, 44, 4185–4191. [Google Scholar] [CrossRef]
- Guo, Z.Q.; Park, S.; Yoon, J.; Shin, I. Recent progress in the development of near-infrared fluorescent probes for bioimaging applications. Chem. Soc. Rev. 2014, 43, 16–29. [Google Scholar] [CrossRef]
- Zhou, J.; Ma, H.M. Design principles of spectroscopic probes for biological applications. Chem. Sci. 2016, 7, 6309–6315. [Google Scholar] [CrossRef] [PubMed]
- Antina, E.V.; Bumagina, N.A.; V’yugin, A.I.; Solomonov, A.V. Fluorescent indicators of metal ions based on dipyrromethene platform. Dye. Pigment. 2017, 136, 368–381. [Google Scholar] [CrossRef]
- Chan, J.; Dodani, S.C.; Chang, C.J. Reaction-based small-molecule fluorescent probes for chemoselective bioimaging. Nat. Chem. 2012, 12, 973–984. [Google Scholar] [CrossRef] [PubMed]
- Jun, M.E.; Roy, B.; Ahn, K.H. “Turn-on” fluorescent sensing with “reactive” probes. Chem. Commun. 2011, 47, 7583–7601. [Google Scholar] [CrossRef] [PubMed]
- Johnson, I.; Spence, M.T.Z. The Molecular Probes Handbook, 11th ed.; Invitrogen Corp.: Carlsbad, CA, USA, 2010. [Google Scholar]
- Gao, M.; Tang, B.Z. Fluorescent sensors based on aggregation-induced emission: Recent advances and perspectives. ACS Sens. 2017, 2, 1382–1399. [Google Scholar] [CrossRef] [PubMed]
- Luo, J.; Xie, Z.; Lam, J.W.Y.; Cheng, L.; Chen, H.; Qiu, C.; Kwok, H.S.; Zhan, X.; Liu, Y.; Zhu, D.; et al. Aggregation-induced emission of 1-methyl-1,2,3,4,5-pentaphenylsilole. Chem. Commun. 2001, 21, 1740–1741. [Google Scholar] [CrossRef]
- Niu, G.L.; Zhang, R.Y.; Gu, Y.; Wang, J.G.; Ma, C.; Kwok, R.T.K.; Lam, J.W.Y.; Sung, H.H.Y.; Williams, I.D.; Wong, K.S.; et al. Highly photostable two-photo NIR AIEgens with tunable organelle specificity and deep tissue penetration. Biomaterials 2019, 208, 72–82. [Google Scholar] [CrossRef]
- Mei, J.; Leung, N.L.C.; Kwok, R.T.K.; Lam, J.W.Y.; Tang, B.Z. Aggregation-induced emission: Together we shine, united we soar! Chem. Rev. 2015, 115, 11718–11940. [Google Scholar] [CrossRef]
- Mei, J.; Hong, Y.; Lam, J.W.Y.; Qin, A.; Tang, Y.; Tang, B.Z. Aggregation-induced emission: The whole is more brilliant than the parts. Adv. Mater. 2014, 26, 5429–5479. [Google Scholar] [CrossRef]
- Chen, J.W.; Law, C.C.W.; Lam, J.W.Y.; Dong, Y.P.; Lo, S.M.F.; Williams, I.D.; Zhu, D.B.; Tang, B.Z. Synthesis, light emission, nanoaggregation, and restricted intramolecular rotation of 1,1-substituted 2,3,4,5-tetraphenylsiloles. Chem. Mater. 2003, 15, 1535–1546. [Google Scholar] [CrossRef]
- Cai, Y.J.; Du, L.L.; Samedov, K.; Gu, X.G.; Qi, F.; Sung, H.H.Y.; Patrick, B.O.; Yan, Z.P.; Jiang, X.F.; Zhang, H.K.; et al. Deciphering the working mechanism of aggregation-induced emission of tetraphenylethylene derivatives by ultrafast spectroscopy. Chem. Sci. 2018, 9, 4662–4670. [Google Scholar] [CrossRef] [PubMed]
- Zhuang, Y.; Huang, F.J.; Xu, Q.; Zhang, M.S.; Lou, X.D.; Xia, F. Facile, fast-responsive, and photostable imaging of telomerase activity in living cells with a fluorescence turn-on manner. Anal. Chem. 2016, 886, 3289–3294. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.Y.; Hu, F.; Zhao, R.; Zhang, G.X.; Yang, H.; Zhang, D.Q. Tetraphenylethylene conjugated with a specific peptide as a fluorescence turn-on bioprobe for the highly specific detection and tracing of tumor markers in live cancer cells. Anal. Chem. 2014, 86, 7987–7995. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.J.; Huang, J.; Li, J.J.; Yan, J.W.; Qin, J.G.; Li, Z. A graphene oxide-based AIE biosensor with high selectivity toward bovine serum albumin. Chem. Commun. 2011, 47, 12385–12387. [Google Scholar] [CrossRef]
- Shustova, N.B.; Ong, T.C.; Cozzolino, A.F.; Michaelis, V.K.; Griffin, R.G.; Dinca, M. Phenyl ring dynamics in a Tetraphenylethylene-bridged metal-organic framework: Implications for the mechanism of aggregation-induced emission. J. Am. Chem. Soc. 2012, 13436, 15061–15070. [Google Scholar] [CrossRef]
- Shi, H.B.; Liu, J.Z.; Geng, J.L.; Tang, B.Z.; Liu, B. Specific detection of integrin αvβ3 by light-up bioprobe with aggregation-induced emission characteristics. J. Am. Chem. Soc. 2012, 13423, 9569–9572. [Google Scholar] [CrossRef]
- Gu, X.G.; Kwok, R.T.K.; Lam, J.W.Y.; Tang, B.Z. AIEgens for biological process monitoring and disease theranostics. Biomaterials 2017, 146, 115–135. [Google Scholar] [CrossRef]
- Hu, F.; Huang, Y.Y.; Zhang, G.X.; Zhao, R.; Yang, H.; Zhang, D.Q. Targeted bioimaging and photodynamic therapy of cancer cells with an activatable red fluorescent bioprobe. Anal. Chem. 2014, 86, 7987–7995. [Google Scholar] [CrossRef]
- Xu, C.H.; Zou, H.; Zhao, Z.; Zhang, P.F.; Kwok, R.T.K.; Lam, J.W.Y.; Sung, H.H.Y.; Williams, I.D.; Tang, B.Z. A new strategy toward “simple” water-soluble AIE probes for hypoxia detection. Adv. Funct. Mater. 2019, 29, 1903278. [Google Scholar] [CrossRef]
- Zhang, R.Y.; Sung, S.H.P.; Feng, G.X.; Zhang, C.J.; Kenry; Tang, B.Z.; Liu, B. Aggregation-induced emission probe for specific turn-on quantification of soluble transferrin receptor: An important disease marker for iron deficiency anemia and kidney diseases. Anal. Chem. 2018, 90, 1154–1160. [Google Scholar] [CrossRef]
- Yin, J.; Hu, Y.; Yoon, J. Fluorescent probes and bioimaging: Alkali metals, alkaline earth metals and pH. Chem. Soc. Rev. 2015, 44, 4619–4644. [Google Scholar] [CrossRef] [PubMed]
- Lu, D.; He, L.; Wang, Y.; Xiong, M.; Hu, M.; Liang, H.; Huan, S.; Zhang, X.B.; Tan, W. Tetraphenylethene derivative modified DNA oligonucleotide for in situ potassium ion detection and imaging in living cells. Talanta 2017, 167, 550–556. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Yu, X.; Li, M.; Liao, N.X.; Bi, A.Y.; Jiang, Y.P.; Liu, S.; Gong, Z.C.; Zeng, W.B. Fluorescent probes for the detection of magnesium ions (Mg2+): From design to application. RSC Adv. 2018, 8, 12573–12587. [Google Scholar] [CrossRef]
- Carroll, M.F.; Schade, D.S. A practical approach to hypercalcemia. Am. Fam. Physician. 2003, 67, 1959–1966. [Google Scholar]
- Zaheer, A.; Murshed, M.; De Grand, A.M.; Morgan, T.G.; Karsenty, G.; Frangioni, J.V. Optical imaging of hydroxyapatite in the calcified vasculature of transgenic animals. Arterioscler. Thromb. Vasc. Biol. 2006, 26, 1132–1136. [Google Scholar] [CrossRef]
- Bian, Y.J.; Wang, L.Q.; Cao, F.X.; Tang, L.J. A simple fluorescence probe based on aggregation-induced emission (AIE) property for the detection of Mg2+ ions. J. Fluoresc. 2015, 26, 53–57. [Google Scholar] [CrossRef]
- Ishiwari, F.; Hasebe, H.; Matsumura, S.; Hajjaj, F.; Horii-Hayashi, N.; Nishi, M.; Someya, T.; Fukushima, T. Bioinspired design of a polymer gel sensor for the realization of extracellular Ca2+ imaging. Sci. Rep. 2016, 6, 24275. [Google Scholar] [CrossRef]
- Morishima, K.; Ishiwari, F.; Matsumura, S.; Fukushima, T.; Shibayama, M. Mesoscopic structural aspects of Ca2+-triggered polymer chain folding of a tetraphenylethene-appended poly(acrylic acid) in relation to its aggregation-induced emission behavior. Macromolecules 2017, 50, 5940–5945. [Google Scholar] [CrossRef]
- Gao, M.; Li, Y.X.; Chen, X.H.; Li, S.W.; Ren, L.; Tang, B.Z. Aggregation-induced emission probe for light-up and in situ detection of calcium ions at high concentration. ACS Appl. Mater. Interfaces 2018, 10, 14410–14417. [Google Scholar] [CrossRef]
- Zhang, J.D.; Yan, Z.; Wang, S.; She, M.Y.; Zhang, Z.; Cai, W.Z.; Liu, P.; Li, J.L. Water soluble chemosensor for Ca2+, based on aggregation-induced emission characteristics and its fluorescence imaging in living cells. Dye. Pigment. 2017, 150, 112–120. [Google Scholar] [CrossRef]
- Wang, P.; Jia, K.; Zhou, X.; Guan, X.; Wang, L.; Tian, Y.; Wu, C.; Liu, X. Ca2+ induced crosslinking of AIE-active polyarylene ether nitrile into fluorescent polymeric nanoparticles for cellular bioimaging. Macromol. Rapid Commun. 2017, 38, 1700360. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.L.; Zhou, Z.C.; Feng, H.; Zhang, C.Y.; Wang, Y.F.; Qian, Z.S.; Pan, J.W. An Aggregation-induced phosphorescence probe for calcium ion-specific detection and live-cell imaging in Arabidopsis Thaliana. Chem. Commun. 2019, 55, 4841–4844. [Google Scholar] [CrossRef] [PubMed]
- Rurack, K. Flipping the light switch ‘ON’-the design of sensor molecules that show cation-induced fluorescence enhancement with heavy and transition metal ions. Spectroc. Acta Pt. A-Molec. Biomolec. Spectr. 2001, 57, 2161–2195. [Google Scholar] [CrossRef]
- Aron, A.T.; Ramos-Torres, K.M.; Cotruvo, J.A.; Chang, C.J. Recognition- and reactivity-based fluorescent probes for studying transition metal signaling in living systems. Acc. Chem. Res. 2015, 48, 2434–2442. [Google Scholar] [CrossRef] [PubMed]
- Stein, E.D.; Cohen, Y.; Winer, A.M. Environmental distribution and transformation of mercury compounds. Crit. Rev. Environ. Sci. Technol. 1996, 26, 1–43. [Google Scholar] [CrossRef]
- Li, W.C.; Tse, H.F. Health risk and significance of mercury in the environment. Environ. Sci. Pollut. Res. 2015, 22, 192–201. [Google Scholar] [CrossRef]
- Wang, K.; Li, J.J.; Ji, S.M.; Li, L.J.; Qiu, Z.P.; Pan, C.Q.; Zhang, J.Y.; Huo, Y.P. Fluorescence probes based on AIE luminogen: Application for sensing Hg2+ in aqueous media and cellular imaging. New J. Chem. 2018, 42, 13836–13846. [Google Scholar] [CrossRef]
- Zhang, G.B.; Ding, A.X.; Zhang, Y.; Yang, L.M.; Kong, L.; Zhang, X.J.; Tao, X.T.; Tian, Y.P.; Yang, J.X. Schiff base modified α-cyanostilbene derivative with aggregation-induced emission enhancement characteristics for Hg2+ detection. Sens. Actuator B-Chem. 2014, 202, 209–216. [Google Scholar] [CrossRef]
- Zhang, G.B.; Zhang, X.J.; Zhang, Y.; Wang, H.; Kong, L.; Tian, Y.P.; Tao, X.T.; Hong, B.; Yang, J.X. Design of turn-on fluorescent probe for effective detection of Hg2+ by combination of AIEE-active fluorophore and binding site. Sens. Actuator B-Chem. 2015, 221, 730–739. [Google Scholar] [CrossRef]
- Fang, W.Y.; Zhang, G.B.; Chen, J.; Kong, L.; Yang, L.M.; Bi, H.; Yang, J.X. An AIE active probe for specific sensing of Hg2+ based on linear conjugated bis-Schiff base. Sens. Actuator B-Chem. 2016, 229, 338–346. [Google Scholar] [CrossRef]
- Gui, S.L.; Huang, Y.Y.; Hu, F.; Jin, Y.L.; Zhang, G.X.; Zhang, D.Q.; Zhao, R. Bioinspired peptide for imaging Hg2+ distribution in living cells and Zebrafish based on coordination-mediated supramolecular assembling. Anal. Chem. 2018, 90, 9708–9715. [Google Scholar] [CrossRef] [PubMed]
- Yuan, B.; Wang, D.X.; Zhu, L.N.; Lan, Y.L.; Cheng, M.; Zhang, L.M.; Chu, J.Q.; Li, X.Z.; Kong, D.M. Dinuclear HgII tetracarbene complex-triggered aggregation-induced emission for rapid and selective sensing of Hg2+ and organomercury species. Chem. Sci. 2019, 10, 4220–4226. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.T.; Li, S.W.; Ling, X.; Zhang, J.; Qin, A.J.; Zhuang, J.; Gao, M.; Tang, B.Z. Dual detection of bioaccumulated Hg2+ based on luminescent bacteria and aggregation-induced emission. Chem. Commun. 2019, 55, 7458–7461. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.Q.; Wen, X.Y.; Fan, Z.F. An AIE active pyrene based fluorescent probe for selective sensing Hg2+ and imaging in live cells. Spectroc. Acta Pt. A-Molec. Biomolec. Spectr. 2019, 223, 117315. [Google Scholar] [CrossRef] [PubMed]
- Bahta, M.; Ahmed, N. Naphthalimide-amino acid conjugates chemosensors for Hg2+ detection: Based on chelation mediated emission enhancement in aqueous solution. J. Photochem. Photobiol. A-Chem. 2019, 378, 85–93. [Google Scholar] [CrossRef]
- Gabr, M.T.; Pigge, F.C. A turn-on AIE active fluorescent sensor for Hg2+ by combination of 1,1-bis(2-pyridyl)ethylene and thiophene/bithiophene fragments. Mater. Chem. Front. 2017, 1, 1654–1661. [Google Scholar] [CrossRef]
- Jiang, Y.; Duan, Q.Y.; Zheng, G.S.; Yang, L.; Zhang, J.; Wang, Y.F.; Zhang, H.T.; He, J.; Sun, H.Y.; Ho, D. An ultra-sensitive and ratiometric fluorescent probe based on the DTBET process for Hg2+ detection and imaging applications. Analyst 2019, 144, 1353–1360. [Google Scholar] [CrossRef]
- Mukherjee, S.; Thilagar, P. Molecular flexibility tuned emission in “V” shaped naphthalimides: Hg(II) detection and aggregation induced emission enhancement (AIEE). Chem. Commun. 2013, 49, 7292–7294. [Google Scholar] [CrossRef]
- Wang, A.Z.; Yang, Y.X.; Yu, F.F.; Xue, L.W.; Hu, B.W.; Fan, W.P.; Dong, Y.J. A highly selective and sensitive fluorescent probe for quantitative detection of Hg2+ based on aggregation-induced emission features. Talanta 2015, 132, 864–870. [Google Scholar] [CrossRef]
- Chatterjee, A.; Banerjee, M.; Khandare, D.G.; Gawas, R.U.; Mascarenhas, S.C.; Ganguly, A.; Gupta, R.; Joshi, H. Aggregation-induced emission-based chemodosimeter approach for selective sensing and imaging of Hg(II) and methylmercury species. Anal. Chem. 2017, 89, 12698–12704. [Google Scholar] [CrossRef]
- Gao, T.; Huang, X.Y.; Huang, S.; Dong, J.; Yuan, K.; Feng, X.P.; Liu, T.T.; Yu, K.Q.; Zeng, W.B. Sensitive water-soluble fluorescent probe based on umpolung and aggregation-induced emission strategies for selective detection of Hg2+ in living cells and Zebrafish. J. Agric. Food Chem. 2019, 67, 2377–2383. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.; Gao, T.; Bi, A.Y.; Cao, X.Z.; Feng, B.; Liu, M.; Du, T.; Feng, X.P.; Zeng, W.B. Revealing aggregation-induced emission effect of imidazolium derivatives and application for detection of Hg2+. Dye Pigment. 2020, 172, 107830. [Google Scholar] [CrossRef]
- Chen, S.; Wang, W.J.; Yan, M.M.; Tu, Q.; Chen, S.W.; Li, T.B.; Yuan, M.S.; Wang, J.Y. 2-Hydroxy benzothiazole modified rhodol: Aggregation-induced emission and dual-channel fluorescence sensing of Hg2+ and Ag+ ions. Sens. Actuator B-Chem. 2018, 255, 2086–2094. [Google Scholar] [CrossRef]
- Niu, C.X.; Liu, Q.L.; Shang, Z.H.; Zhao, L.; Ouyang, J. Dual-emission fluorescent sensor based on AIE organic nanoparticles and Au nanoclusters for the detection of mercury and melamine. Nanoscale 2015, 7, 8457–8465. [Google Scholar] [CrossRef] [PubMed]
- Kepp, K.P.; Squitti, R. Copper imbalance in Alzheimer’s disease: Convergence of the chemistry and the clinic. Coord. Chem. Rev. 2019, 397, 168–187. [Google Scholar] [CrossRef]
- Dujols, V.; Ford, F.; Czarnik, A.W. A long-wavelength fluorescent chemodosimeter selective for Cu(II) ion in water. J. Am. Chem. Soc. 1997, 119, 7386–7387. [Google Scholar] [CrossRef]
- Xu, J.; Hou, Y.; Ma, Q.; Wu, X.; Feng, S.; Zhang, J.; Shen, Y. A highly selective fluorescent probe for Cu2+ based on rhodamine B derivative. Spectroc. Acta Pt. A-Molec. Biomolec. Spectr. 2014, 124, 416–422. [Google Scholar] [CrossRef]
- Wu, W.N.; Mao, P.D.; Wang, Y.; Mao, X.J.; Xu, Z.Q.; Xu, Z.H.; Zhao, X.L.; Fan, Y.C.; Hou, X.F. AEE active Schiff base-bearing pyrene unit and further Cu2+-induced self-assembly process. Sens. Actuator B-Chem. 2018, 258, 393–401. [Google Scholar] [CrossRef]
- Singh, A.; Singh, R.; Shellaiah, M.; Prakash, E.C.; Chang, H.C.; Raghunath, P.; Lin, M.C.; Lin, H.C.; Liu, B.; Zhou, H.L.; et al. Aggregation-induced emission activity and further Cu2+-induced self-assembly process of two Schiff compounds. Sens. Actuator B-Chem. 2017, 246, 554–562. [Google Scholar]
- Wang, Y.; Wu, H.; Wu, W.N.; Li, S.J.; Xu, Z.H.; Xu, Z.Q.; Fan, Y.C.; Zhao, X.L.; Liu, B.Z. An AIRE active Schiff base bearing coumarin and pyrrole unit: Cu2+ detection in either solution or aggregation states. Sens. Actuator B-Chem. 2018, 260, 106–115. [Google Scholar] [CrossRef]
- Ding, A.X.; Shi, Y.D.; Zhang, K.X.; Sun, W.; Tan, Z.L.; Lu, Z.L.; He, L. Self-assembled aggregation-induced emission micelle (AIE micelle) as interfacial fluorescence probe for sequential recognition of Cu2+ and ATP in water. Sens. Actuator B-Chem. 2018, 255, 440–447. [Google Scholar] [CrossRef]
- Maret, W. Crosstalk of the group IIa and IIb metals calcium and zinc in cellular signaling. Proc. Natl. Acad. Sci. USA 2001, 98, 12325–12327. [Google Scholar] [CrossRef] [PubMed]
- Frederickson, C.J.; Koh, J.Y.; Bush, A.I. The neurobiology of zinc in health and disease. Nat. Rev. Neurosci. 2005, 6, 449–462. [Google Scholar] [CrossRef] [PubMed]
- Arena, G.; Rizzarelli, E. Zn2+ interaction with Amyloid-B: Affinity and speciation. Molecules 2019, 24, 2796. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Li, S.M.; Zheng, J.Q.; Kong, D.Y.; Zheng, X.J.; Fang, D.C.; Jin, L.P. Coordination-directed stacking and aggregation-induced emission enhancement of the Zn(II) Schiff base complex. Inorg. Chem. 2017, 56, 984–990. [Google Scholar] [CrossRef] [PubMed]
- Wen, X.Y.; Wang, Q.; Fan, Z.F. An active fluorescent probe based on aggregation-induced emission for intracellular bioimaging of Zn2+ and tracking of interactions with single-stranded DNA. Anal. Chim. Acta 2018, 1013, 79–86. [Google Scholar] [CrossRef] [PubMed]
- Wan, J.Y.; Zhang, W.; Guo, H.D.; Liang, J.J.; Huang, D.Y.; Xiao, H.B. Two spirobifluorene-based fluorescent probes with aggregation-induced emission properties: Synthesis and application in the detection of Zn2+ and cell imaging. J. Mater. Chem. C 2019, 7, 2240–2249. [Google Scholar] [CrossRef]
- He, X.X.; Wang, X.M.; Zhang, L.; Fang, G.Z.; Liu, J.F.; Wang, S. Sensing and intracellular imaging of Zn2+ based on affinity peptide using an aggregation induced emission fluorescence “switch-on” probe. Sens. Actuator B-Chem. 2018, 271, 289–299. [Google Scholar] [CrossRef]
- Naskar, B.; Dhara, A.; Maiti, D.K.; Kukulka, M.; Mitoraj, M.P.; Srebro-Hooper, M.; Prodhan, C.; Chaudhuri, K.; Goswami, S. Aggregation-induced emission-based sensing platform for selective detection of Zn2+: Experimental and theoretical investigations. Chem. Phys. Chem. 2019, 20, 1630–1639. [Google Scholar] [CrossRef]
- Wang, J.X.; Lin, X.F.; Shu, T.; Su, L.; Liang, F.; Zhang, X.J. Self-assembly of metal nanoclusters for aggregation-induced emission. Int. J. Mol. Sci. 2019, 20, 1891. [Google Scholar] [CrossRef]
- Guo, Y.M.; Cao, F.P.; Lei, X.L.; Mang, L.H.; Cheng, S.J.; Song, J.T. Fluorescent copper nanoparticles: Recent advances in synthesis and applications for sensing metal ions. Nanoscale 2016, 8, 4852–4863. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.Y.; Hu, Y.F.; Zhang, L.L.; Huang, Y.; Zhao, S.L. Photoluminescence light-up detection of zinc ion and imaging in living cells based on the aggregation induced emission enhancement of glutathione-capped copper nanoclusters. Biosens. Bioelectron. 2017, 94, 523–529. [Google Scholar] [CrossRef] [PubMed]
- Jia, X.F.; Li, J.; Wang, E.K. Cu nanoclusters with aggregation induced emission enhancement. Small 2013, 9, 3873–3879. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Chen, Z.H.; Wan, Z.H.; Yang, T.Z.; Wang, H.; Mei, X.F. One-pot development of water-soluble copper nanoclusters with red emission and aggregation induced fluorescence enhancement. RSC Adv. 2016, 6, 34090–34095. [Google Scholar] [CrossRef]
- Rouault, T.A. The role of iron regulatory proteins in mammalian iron homeostasis and disease. Nat. Chem. Biol. 2006, 2, 406–414. [Google Scholar] [CrossRef]
- Gozzelin, R.; Arosio, P. Iron homeostasis in health and disease. Int. J. Mol. Sci. 2016, 17, 130. [Google Scholar] [CrossRef]
- Lim, B.; Baek, B.; Jang, K.; Lee, N.K.; Lee, J.H.; Lee, Y.; Kim, J.; Kang, S.W.; Park, J.; Kim, S.; et al. Novel turn-on fluorescent biosensors for selective detection of cellular Fe3+ in lysosomes: Thiophene as a selectivity-tuning handle for Fe3+ sensors. Dye. Pigment. 2019, 169, 51–59. [Google Scholar] [CrossRef]
- Yang, X.D.; Chen, X.L.; Lu, X.D.; Yan, C.G.; Xu, Y.K.; Hang, X.D.; Qu, J.Q.; Liu, R.Y. A highly selective and sensitive fluorescent chemosensor for detection of CN-, SO32- and Fe3+ based on aggregation-induced emission. J. Mater. Chem. C 2016, 4, 383–390. [Google Scholar] [CrossRef]
- Yang, D.L.; Li, F.; Luo, Z.M.; Bao, B.Q.; Hu, Y.L.; Weng, L.X.; Cheng, Y.X.; Wang, L.H. Conjugated polymer nanoparticles with aggregation induced emission characteristics for intracellular Fe3+ sensing. J. Polym. Sci. Part A Polym. Chem. 2016, 54, 1686–1693. [Google Scholar] [CrossRef]
- Yang, Y.; Wang, X.Y.; Cui, Q.L.; Cao, Q.; Li, L.D. Self-assembly of fluorescent organic nanoparticles for iron(III) sensing and cellular imaging. ACS Appl. Mater. Interfaces 2016, 8, 7440–7448. [Google Scholar] [CrossRef]
- Sinha, N.; Stegemann, L.; Tan, T.T.Y.; Doltsinis, N.L.; Strassert, C.A.; Hahn, F.E. Turn-on fluorescence in Tetra-NHC ligands by rigidification through metal complexation: An alternative to aggregation-induced emission. Angew. Chem. Int. Ed. 2017, 56, 2785–2789. [Google Scholar] [CrossRef] [PubMed]
- Kim, N.H.; Won, M.; Kim, J.S.; Huh, Y.; Kim, D. A highly sensitive and fast responsive fluorescent probe for detection of Gold(III) ions based on the AIEgen disaggregation. Dye Pigment 2019, 160, 647–653. [Google Scholar] [CrossRef]
- Khan, R.I.; Ramu, A.; Pitchumani, K. Design and one-pot synthesis of a novel pyrene based fluorescent sensor for selective “turn on”, naked eye detection of Ni2+ ions, and live cell imaging. Sens. Actuator B-Chem. 2018, 266, 429–437. [Google Scholar] [CrossRef]
- Gui, S.L.; Huang, Y.Y.; Hu, F.; Jin, Y.I.; Zhang, G.X.; Yan, L.S.; Zhang, D.Q.; Zhao, R. Fluorescence turn-on chemosensor for highly selective and sensitive detection and bioimaging of Al3+ in living cells based on ion-induced aggregation. Anal. Chem. 2015, 87, 1470–1474. [Google Scholar] [CrossRef] [PubMed]
- Xu, P.F.; Bao, Z.Y.; Yu, C.Y.; Qiu, Q.Q.; Wei, M.R.; Xi, W.B.; Qian, Z.S.; Feng, H. A water-soluble molecular probe with aggregation-induced emission for discriminative detection of Al3+ and Pb2+ and imaging in seedling root of Arabidopsis. Spectroc. Acta Pt. A-Molec. Biomolec. Spectr. 2019, 223, 117335. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.; Singh, R.; Shellaiah, M.; Prakash, E.C.; Chang, H.C.; Raghunath, P.; Lin, M.C.; Lin, H.C. A new pyrene-based aggregation induced ratiometric emission probe for selective detections of trivalent metal ions and its living cell application. Sens. Actuator B-Chem. 2015, 207, 338–345. [Google Scholar] [CrossRef]
- Simon, T.; Shellaiah, M.; Srinivasadesikan, V.; Lin, C.C.; Ko, F.H.; Sun, K.W.; Lin, M.C. A simple pyrene based AIEE active schiff base probe for selective naked eye and fluorescence off-on detection of trivalent cations with live cell application. Sens. Actuator B-Chem. 2016, 231, 18–29. [Google Scholar] [CrossRef]
- Shellaiah, M.; Simon, T.; Srinivasadesikan, V.; Lin, C.M.; Sun, K.W.; Ko, F.H.; Lin, M.C.; Lin, H.C. Novel pyrene containing monomeric and dimeric supramolecular AIEE active nano-probes utilized in selective ‘’off-on’’ trivalent metal and highly acidic pH sensing with live cell applications. J. Mater. Chem. C 2016, 4, 2056–2071. [Google Scholar] [CrossRef]
- Gui, S.L.; Huang, Y.Y.; Zhu, Y.Y.; Jin, Y.L.; Zhao, R. Biomimetic sensing system for tracing Pb2+ distribution in living cells based on the metal-peptide supramolecular assembly. ACS Appl. Mater. Interfaces 2019, 11, 5804–5811. [Google Scholar] [CrossRef]
- Chen, X.T.; Peng, L.; Feng, M.L.; Xiang, Y.; Tong, A.J.; He, L.F.; Liu, B.; Tang, Y.P. An aggregation induced emission enhancement-based ratiometric fluorescent sensor for detecting trace uranyl ion (UO22+) and the application in living cells imaging. J. Lumines. 2017, 186, 301–306. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are not available from the authors. |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, Y.; Zhong, H.; Huang, Y.; Zhao, R. Recent Advances in AIEgens for Metal Ion Biosensing and Bioimaging. Molecules 2019, 24, 4593. https://doi.org/10.3390/molecules24244593
Li Y, Zhong H, Huang Y, Zhao R. Recent Advances in AIEgens for Metal Ion Biosensing and Bioimaging. Molecules. 2019; 24(24):4593. https://doi.org/10.3390/molecules24244593
Chicago/Turabian StyleLi, Yongming, Huifei Zhong, Yanyan Huang, and Rui Zhao. 2019. "Recent Advances in AIEgens for Metal Ion Biosensing and Bioimaging" Molecules 24, no. 24: 4593. https://doi.org/10.3390/molecules24244593
APA StyleLi, Y., Zhong, H., Huang, Y., & Zhao, R. (2019). Recent Advances in AIEgens for Metal Ion Biosensing and Bioimaging. Molecules, 24(24), 4593. https://doi.org/10.3390/molecules24244593