Rapid Characterization of Triterpene Saponins from Zornia brasiliensis by HPLC-ESI-MS/MS
Abstract
1. Introduction
2. Results
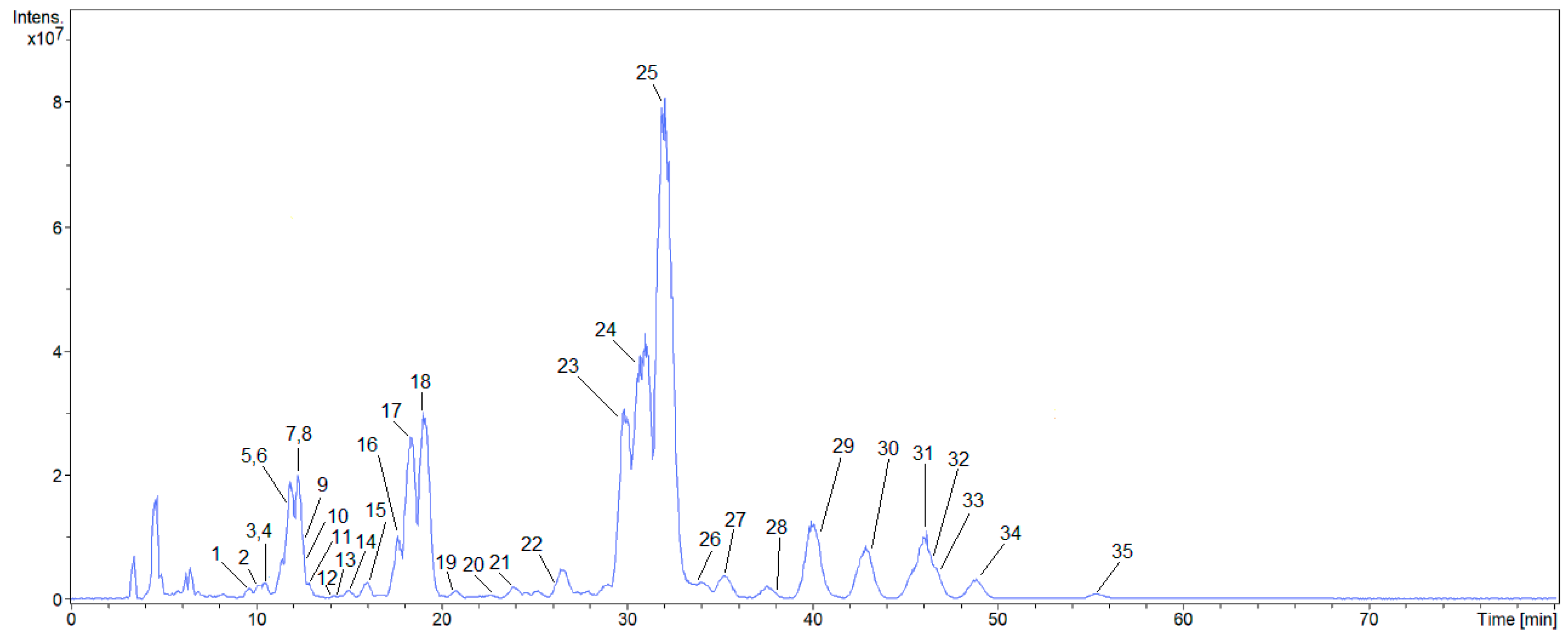
2.1. Characterization of the Compounds by HPLC-ESI-MS/MS
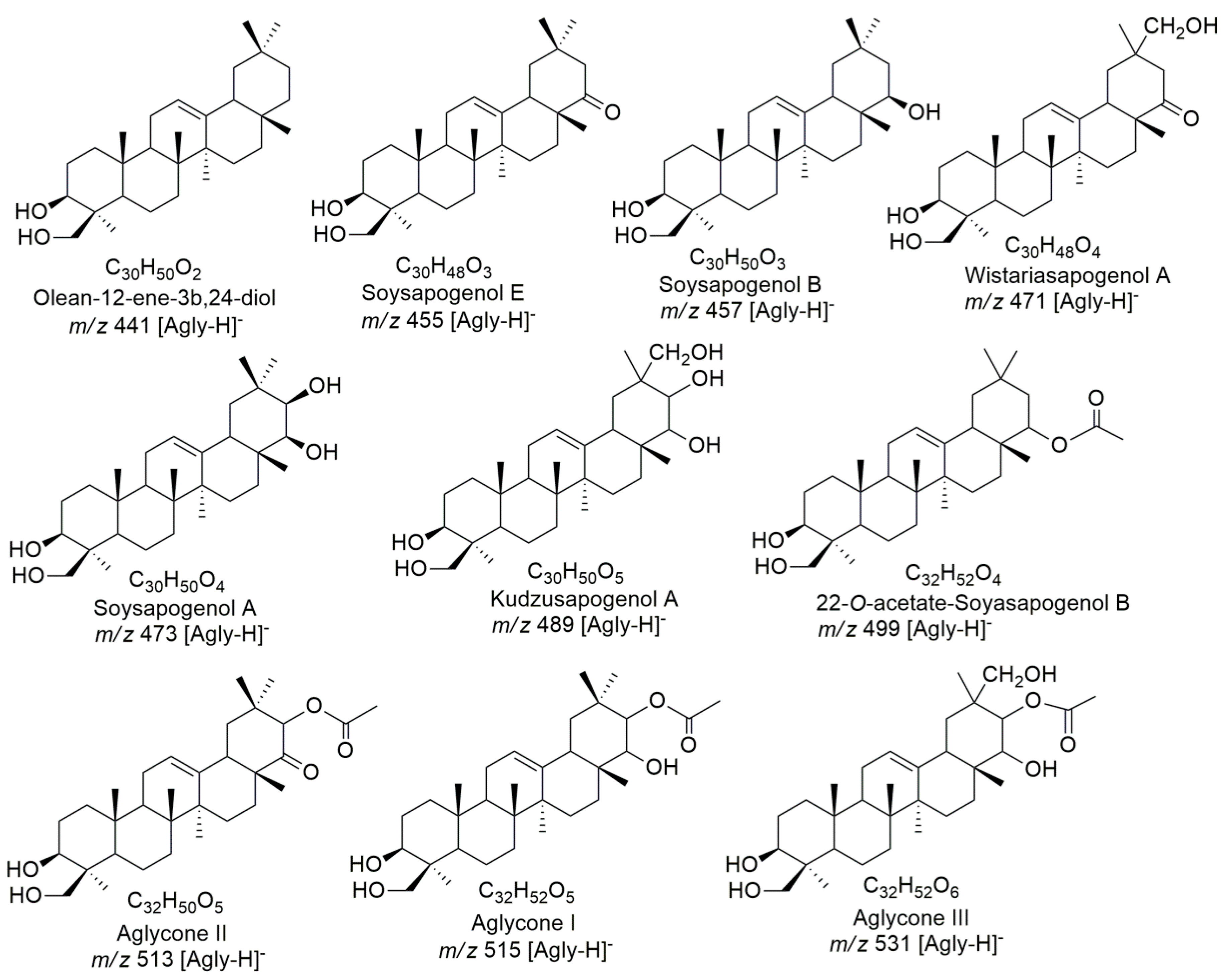
2.1.1. Aglycones of Saponins of Z. Brasiliensis
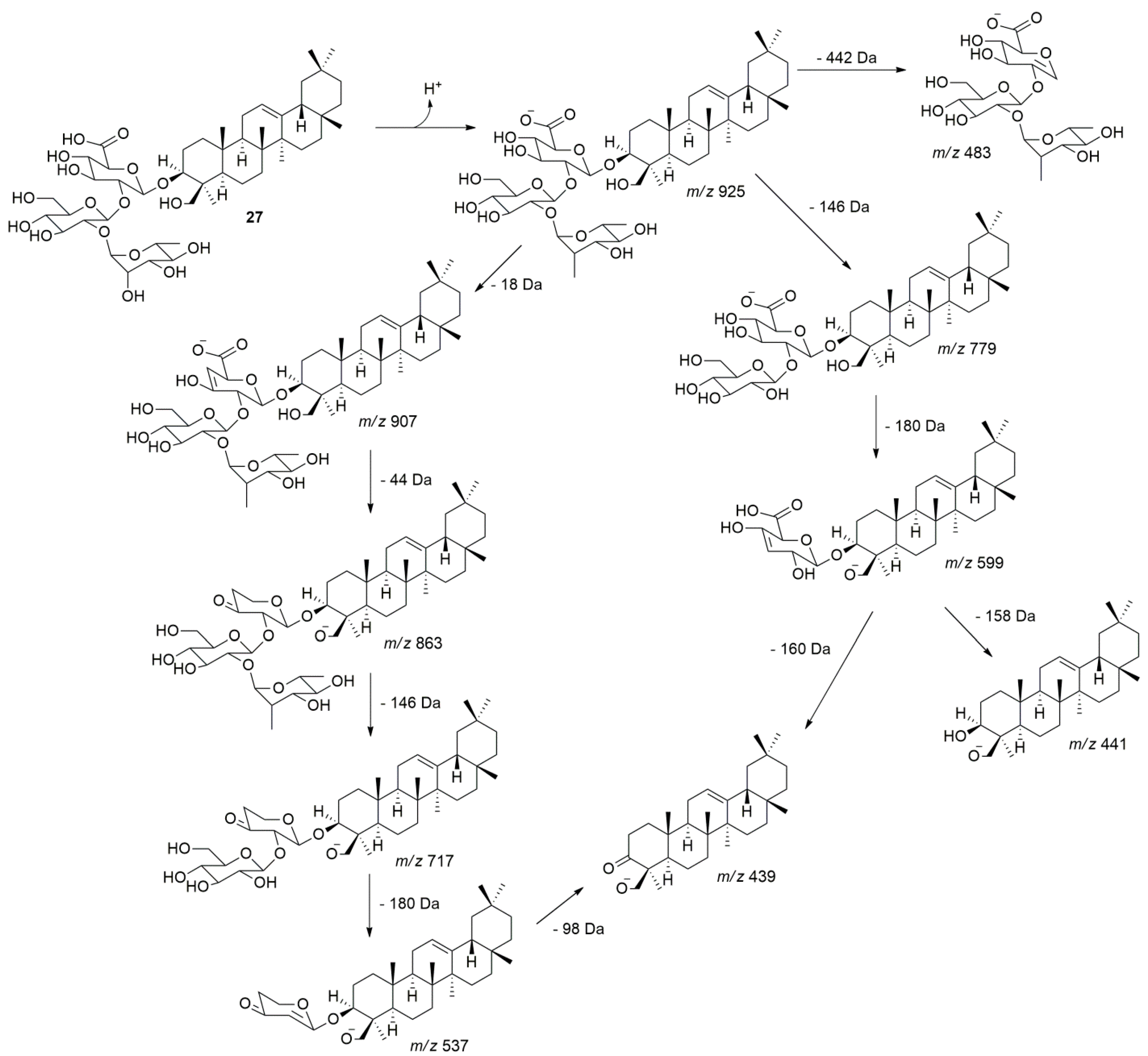
2.1.2. Saponins of Z. brasiliensis
2.2. NMR Identification of Compound 25
3. Materials and Methods
3.1. Reagents and Materials
3.2. Plant Material
3.3. Extraction and Isolation of Z. brasiliensis Constituents
3.4. HPLC-DAD Conditions
3.5. HPLC-ESI-MS/MS Conditions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Legume Phylogeny Working Group. A new subfamily classification of the Leguminosae based on a taxonomically comprehensive phylogeny. Taxon 2017, 66, 44–77. [Google Scholar] [CrossRef]
- Perez, A.P.F.; Lewis, G.P.; Queiroz, R.T.; Silva, J.S.; De Azevedo Tozzi, A.M.G.; Rodrigues, K.F. Fruit as diagnostic characteristic to recognize Brazilian species of Zornia (Leguminosae, Papilionoideae). Phytotaxa 2015, 219, 27–42. [Google Scholar] [CrossRef]
- Fortuna-Perez, A.P. O gênero Zornia J.F. Gmel. (Leguminosae, Papilionoideae, Dalbergieae): Revisão taxonômica das espécies ocorrentes no Brasil e filogenia. Ph.D. Thesis, Universidade Estadual de Campinas—Campinas-SP, Campinas, Brazil, 2009. [Google Scholar]
- Belcavello, L.; Cunha, M.R.H.; Andrade, M.A.; Batitucci, M.C.P. Citotoxicidade e danos ao DNA induzidos pelo extrato de Zornia diphylla, uma planta medicinal. Nat. Line 2012, 10, 140–145. [Google Scholar]
- Rojas, A.; Bah, M.; Rojas, J.I.; Serrano, V.; Pacheco, S. Spasmolytic activity of some plants used by the otomi indians of Queretaro (Mexico) for the treatment of gastrointestinal disorders. Phytomedicine 1999, 6, 367–371. [Google Scholar] [CrossRef]
- Greetha, K.M.; Shilpa, S.; Murugan, V. Anticonvulsant Activity of the Methanolic Extract of Whole Plant of Zornia diphylla (Linn) Pers. J. Pharm. Res. 2012, 5, 3670–3672. [Google Scholar]
- Arunkumar, R.; Nair, S.A.; Subramoniam, A. Induction of cell-specific apoptosis and protection of mice from cancer challenge by a steroid positive compound from Zornia diphylla (L.) Pers. J. Pharmacol. Pharmacother 2012, 3, 233–241. [Google Scholar]
- Obi, C.L.; Ramalivhana, J.; Samie, A.; Igumbor, E.O. Prevalence, pathogenesis, antbiotic susceptibility profiles, and in-vitro activity of selected medicinal plants against Aeromonas isolated from stool samples of patients in the Venda region of South Africa. J. Health Popul. Nutr. 2007, 25, 428–435. [Google Scholar] [PubMed]
- Laxane, S.N.; Swarnkar, S.K.; Setty, M.M. Antioxidant studies on the ethanolic extract of Zornia gibbosa. Pharmacol. Online 2008, 1, 319–330. [Google Scholar]
- Laxane, S.N.; Swarnkar, S.K.; Zanwar, S.B.; Setty, M.M. Anti-inflammatory studies of the alcoholic extract of Zornia gibbosa. Pharmacol. Online 2011, 1, 67–76. [Google Scholar]
- Brahmachari, G.; Ghosh, S.; Mondal, S.; Jash, S.K.; Mandal, L.C.; Mondal, A. Cyclic voltammetric studies with plant extracts of some traditionally used Indian medicinal plants to evaluate their antioxidant potential. Biochem. Indian J. 2009, 3, 32–35. [Google Scholar]
- Arunkumar, R.; Nair, S.A.; Rameshkumar, K.B.; Subramoniam, A. The essential oil constituents of Zornia diphylla (L.) Pers., and anti-inflammatory and antimicrobial activities of the oil (Article). Rec. Nat. Prod. 2014, 8, 385–393. [Google Scholar]
- Leuner, O.; Havlik, J.; Prokudina, E.; Novy, P.; Kokoska, L. Distribution of isoflavones and coumestrol in neglected tropical and subtropical legumes. J. Sci. Food Agric. 2013, 93, 575–579. [Google Scholar] [CrossRef] [PubMed]
- Ren, F.Z.; Gao, Y.Q.; Cheng, X.X.; Li, L.H.; Chen, S.H.; Zhang, Y.L. Study on Chemical Constituents of Zornia diphylla. Chin. Pharm. J. 2012, 47, 179–181. [Google Scholar]
- Agra, M.F.; Freitas, P.F.; Barbosa-Filho, J.M. Synopsis of the plants know as medicinal and poisonous in Northeast of Brazil. Rev. Bras. Farmacogn. 2007, 17, 114–140. [Google Scholar] [CrossRef]
- BFG—The Brazil Flora Group. Growing knowledge: An overview of Seed Plantdiversity in Brazil. Rodriguésia 2015, 6, 1085–1113. [Google Scholar]
- Mohlenbrock, R.H. A monograph of the leguminous genus Zornia. Webbia 1961, 16, 1–141. [Google Scholar] [CrossRef]
- Missouri Botanical Garden. 2017. Available online: http://www.tropicos.org/Name/13035232 (accessed on 22 October 2018).
- Da Silva, A.D.S.; Cavalcante-Silva, L.H.A.; Da Matta, C.B.B.; Silva, D.D.F.; Araújo, M.V.D.; Tavares, J.F.; Alexandre-Moreira, M.S. Antinociceptive effect of 7-methoxyflavone isolated from Zornia brasiliensis. Nat. Prod. Res. 2013, 27, 1695–1699. [Google Scholar] [CrossRef] [PubMed]
- Costa, E.V.; Menezes, L.R.; Rocha, S.L.; Baliza, I.R.; Dias, R.B.; Rocha, C.A.; Soares, M.B.; Bezerra, D.P. Antitumor properties of the leaf essential oil of Zornia brasiliensis. Planta Med. 2015, 81, 563–567. [Google Scholar] [CrossRef]
- Nascimento, Y.M.; Abreu, L.S.; Lima, R.L.; Silva, A.D.S.; Costa, V.C.O.; Melo, J.I.M. Zornioside, a dihydrochalcone C-glycoside, and other compounds from Zornia brasiliensis. Rev. Bras. Farmacogn. 2017, 28, 192–197. [Google Scholar] [CrossRef]
- Huhman, D.V.; Sumner, L.W. Metabolic profiling of saponins in Medicago sativa and Medicago truncatula using HPLC coupled to an electrospray ion-trap mass spectrometer. Phytochemistry 2002, 59, 347–360. [Google Scholar] [CrossRef]
- Ling, Y.; Lin, Z.; Zha, W.; Lian, T.; You, S. Rapid Detection and Characterisation of Triterpene Saponins from the Root of Pulsatilla chinensis (Bunge) Regel by HPLC-ESI-QTOFMS/MS. Phytochem. Anal. 2016, 27, 174–183. [Google Scholar] [CrossRef] [PubMed]
- Pollier, J.; Morreel, K.; Geelen, D.; Goossens, A. Metabolite Profiling of Triterpene Saponins in Medicago truncatula Hairy Roots by Liquid Chromatography Fourier Transform Ion Cyclotron Resonance Mass Spectrometry. J. Nat. Prod. 2011, 74, 1462–1476. [Google Scholar] [CrossRef] [PubMed]
- Negri, G.; Tabach, R. Saponins, tannins and flavonols found in hydroethanolic extract from Periandra dulcis roots. Rev. Bras. Farmacogn. 2013, 23, 851–860. [Google Scholar] [CrossRef]
- Perret, C.; Wolfender, J.L.; Hostettmann, K. LC/ES-MS Analysis of Triterpene Glycosides: Rapid Estimation of the Saponin Content of Dried Berries of Phytolacca dodecandra. Phytochem. Anal. 1999, 10, 272–278. [Google Scholar] [CrossRef]
- Zhang, W.; Popovich, D.G. Chemical and Biological Characterization of Oleanane Triterpenoids from Soy. Molecules 2009, 14, 2959–2975. [Google Scholar] [CrossRef]
- Berhow, M.A.; Kong, S.B.; Vermillion, K.E.; Duval, S.M. Complete Quantification of Group A and Group B Soyasaponins in Soybeans. J. Agric. Food Chem. 2006, 54, 2035–2044. [Google Scholar] [CrossRef]
- Jin, M.; Yang, Y.; Su, B.; Ren, Q. Rapid quantification and characterization of soyasaponins by high-performance liquid chromatography coupled with electrospray mass spectrometry. J. Chromatogr. A 2006, 1108, 31–37. [Google Scholar] [CrossRef]
- Takada, Y.; Sasama, H.; Sayama, T.; Kikuchi, A.; Kato, S.; Ishimoto, M.; Tsukamoto, C. Genetic and chemical analysis of a key biosynthetic step for soyasapogenol A, an aglycone of group A saponins that influence soymilk flavor. Theor. Appl. Genet. 2013, 126, 721–731. [Google Scholar] [CrossRef]
- Konoshima, T.; Kozuka, M.; Haruna, M.; Ito, K.; Kimura, T. The structures of New Triterpenoids from Wistaria brachybotrys Sieb. et Zucc. Chem. Pharm. Bull. 1989, 37, 1550–1553. [Google Scholar] [CrossRef]
- Takeshita, T.; Hamada, S.; Nohara, T. New triterpenoid sapogenols from Abrus cantoniensis (I). Chem. Pharm. Bull. 1989, 37, 846–848. [Google Scholar] [CrossRef]
- Arao, T.; Idzu, T.; Kinjo, J.; Nohara, T.; Isobe, R. Oleanene-type triterpene glycosides from puerariae radix. III. Three new saponins from Pueraria thomsonii. Chem. Pharm. Bull. 1996, 44, 1970–1972. [Google Scholar] [CrossRef]
- Kitagawa, I.; Wang, H.K.; Taniyama, T.; Yoshikawa, M. Reinvestigation of the Structures os Soyasapogenols A, B and E, Oleanene-Sapogenols from Soybean. Structures od Soyasaponins I, II, e III. Chem. Pharm. Bull. 1988, 36, 153–161. [Google Scholar] [CrossRef]
- Price, K.R.; Johnson, I.T.; Fenwick, G.R.; Malinow, M.R. The chemistry and biological significance of saponins in foods and feedingstuffs. Crit. Rev. Food Sci. Nutr. 1987, 26, 27–135. [Google Scholar] [CrossRef] [PubMed]
- Konoshima, T.; Kozuka, M.; Haruna, M.; Ito, K.; Kimura, T.; Tokuda, H. Studies on the constituents of leguminous plants. XII. The structures of new triterpenoid saponins from Wistaria brachybotrys Sieb. et Zucc. Chem. Pharm. Bull. 1989, 37, 2731–2735. [Google Scholar] [CrossRef][Green Version]
- Ding, Y.; Takeshita, T.; Yokoyama, K.; Kinjo, J.; Nohara, T. Triterpenoid Glycosides from Sophorae Subprostratae Radix. Chem. Pharm. Bull. 1992, 40, 139–142. [Google Scholar] [CrossRef]
- Curl, C.L.; Price, K.R.; Fenwick, G.R. Soyasaponin A3, a New Monodesmosidic Saponin Isolated from the Seeds of Glycine max. J. Nat. Prod. 1988, 51, 122–124. [Google Scholar] [CrossRef]
- Woldemichael, G.M.; Montenegro, G.; Timmermann, B.N. Triterpenoidal lupin saponins from the Chilean legume Lupinus oreophilus Phil. Phytochemistry 2003, 63, 853–857. [Google Scholar] [CrossRef]
- Giuliana, B.; Raffaella, P.; Cecilia, F.C.; Maria, A.A.; Cataldi, T.R.; Philippe, S.K.; Alessandro, B.; Daniela, R.; Luigi, M. Determination of soyasaponins in Fagioli di Sarconi beans (Phaseolus vulgaris L.) by LC-ESI-FTICR-MS and evaluation of their hypoglycemic activity. Anal. Bioanal. Chem. 2018, 410, 1561–1569. [Google Scholar]
- Decroos, K.; Vincken, J.P.; Heng, L.; Bakker, R.; Gruppen, H.; Verstraete, W. Simultaneous quantification of differently glycosylated, acetylated, and 2,3-dihydro-2,5-dihydroxy-6-methyl-4H-pyran-4-one-conjugated soyasaponins using reversed-phase high-performance liquid chromatography with evaporative light scattering detection. J. Chromatogr. A 2005, 1072, 185–193. [Google Scholar] [CrossRef]
- Udayama, M.; Ohkawa, M.; Yoshida, N.; Kinjo, J.; Nohara, T. Structures of three new oleanene glucuronides isolated from Lathyrus palustres var. Pilosus and hepatoprotective activity. Chem. Pharm. Bull. 1998, 46, 1412–1415. [Google Scholar] [CrossRef][Green Version]
- Sakamoto, S.; Kuroyanagi, M.; Ueno, A.; Sekuta, S. Triterpenoid saponins from Sophora subprostrata. Phytochemistry 1992, 31, 1339–1342. [Google Scholar] [CrossRef]
- Rupasinghe, H.V.; Jackson, C.J.C.; Poysa, V.; Di Berardo, C.; Bewley, J.D.; Jenkinson, J. Soyasapogenol A and B Distribution in Soybean (Glycine max L. Merr.) in Relation to Seed Physiology; Genetic Variability, and Growing Location. J. Agric. Food Chem. 2003, 51, 5888–5894. [Google Scholar] [CrossRef] [PubMed]
- Kudou, S.; Tonomura, M.; Tsukamoto, C.; Uchida, T.; Sakabe, T.; Tamura, N.; Okubo, K. Isolation and Structural Elucidation of DDMP-Conjugated Soyasaponins as Genuine Saponins from Soybean Seeds. Biosci. Biotechnol. Biochem. 1993, 57, 546–550. [Google Scholar] [CrossRef]
- Dalluge, J.J.; Eliason, E.; Frazer, S. Simultaneous Identification of Soyasaponins and Isoflavones and Quantification of Soyasaponin B in Soy Products, Using Liquid Chromatography/Electrospray Ionization-Mass Spectrometry. J. Agric. Food Chem. 2003, 51, 3520–3524. [Google Scholar] [CrossRef] [PubMed]
- Güçlü-Ustündağ, O.; Mazza, G. Saponins: Properties, applications and processing. Crit. Rev. Food Sci. Nutr. 2007, 47, 231–258. [Google Scholar] [CrossRef] [PubMed]
- Barbosa, A.P.; Silva, B.P.; Parente, J.P. A new complex triterpenoid saponin from Samanea saman with haemolytic activity and adjuvant effect. Phytochem. Lett. 2012, 5, 626–631. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are not available from the authors. |
 ) indicates some key HMBC correlations for compound 25.
) indicates some key HMBC correlations for compound 25.
 ) indicates some key HMBC correlations for compound 25.
) indicates some key HMBC correlations for compound 25.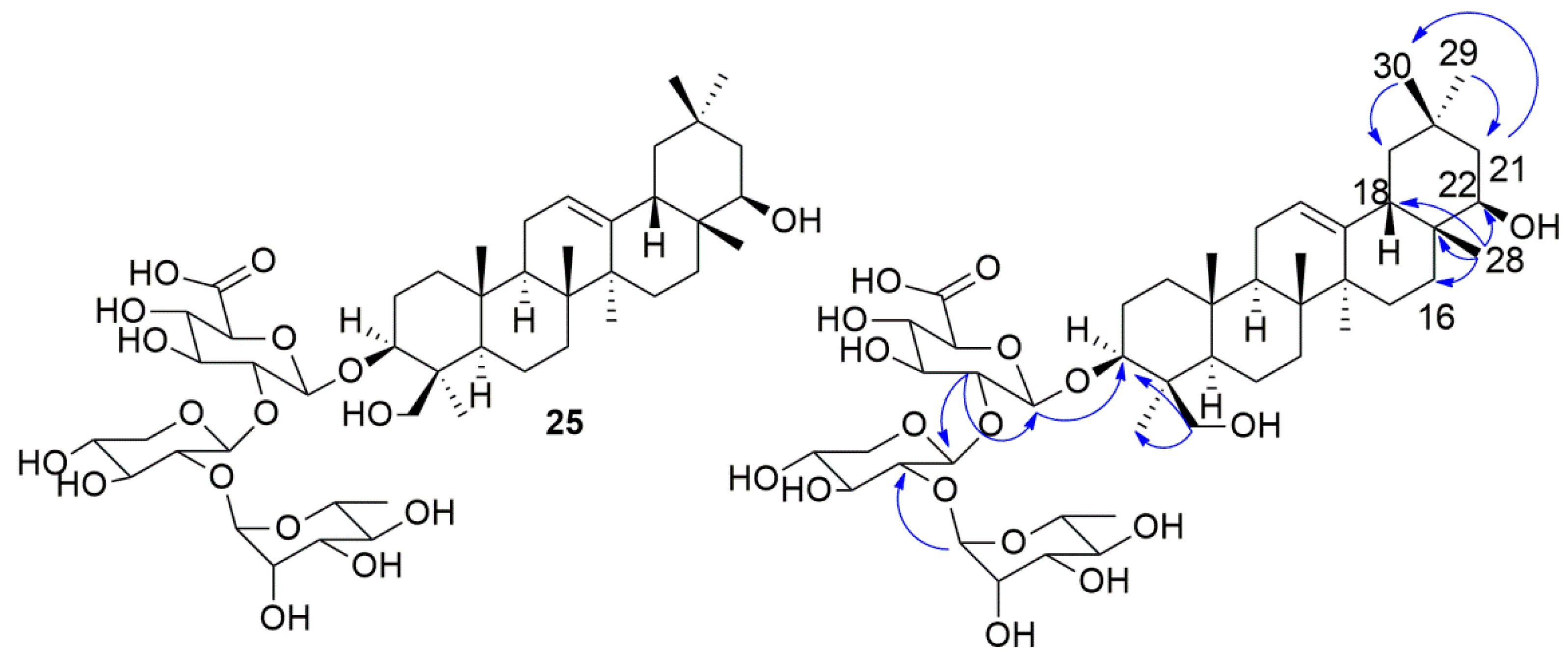
| Compound | TR (min) | [M − H]− | [M − H]− Calculated | Molecular Formula | Error (ppm) | Fragments (m/z) | Tentative Identification | Reference |
|---|---|---|---|---|---|---|---|---|
| 1 | 10.5 | 925.4800 | 925.4802 | C47H73O18 | 0.2 | 907, 863, 779, 717, 629, 567, 471, 453, 439, 407 | Wistariasaponin A | [36] |
| 2 | 11.1 | 779.4239 | 779.4223 | C41H63O14 | 2.0 | 735, 629, 585, 567, 471, 439, 407 | Pen-HexA-Wistariasapogenol A | - |
| 3 | 11.2 | 797.4310 | 797.4329 | C41H65O15 | 2.4 | 753, 647, 621, 603, 585, 489, 457, 307 | Pen-HexA-Kudzusapogenol A | - |
| 4 | 943.4912 | 943.4908 | C47H75O19 | −0.4 | 925, 881, 797, 735, 647, 585, 489, 457 | Subproside II | [37] | |
| 5 | 12.5 | 957.5056 | 957.5065 | C48H77O19 | 0.9 | 939, 895, 811, 749, 631, 569, 483, 473 | Soyasaponin A3 | [28,38] |
| 6 | 927.4943 | 927.4959 | C47H75O18 | 1.8 | 909, 865, 781, 719, 631, 569, 473, 453 | dHex-Pen-HexA-Soyasapogenol A | - | |
| 7 | 13.0 | 781.4350 | 781.4380 | C41H65O14 | 3.9 | 763, 631, 613, 569, 473, 307 | Pen-HexA-Soyasapogenol A | - |
| 8 | 811.4459 | 811.4485 | C42H67O15 | 3.2 | 793, 631, 587, 473, 337 | Hex-HexA-Soyasapogenol A | [39] | |
| 9 | 13.2 | 985.4961 | 985.5014 | C49H77O20 | 5.3 | 967, 839, 821, 777, 689, 627, 531, 489, 457, 453 | dHex-Pen-HexA-Aglycone III | - |
| 10 | 13.7 | 839.4404 | 839.4435 | C43H67O16 | 3.6 | 821, 689, 671, 627, 531, 489, 457, 307 | Pen-HexA-Aglycone III | - |
| 11 | 14.2 | 997.5022 | 997.5014 | C50H77O20 | −0.8 | 979, 935, 851, 789, 671, 609, 513, 483, 471 | dHex-Hex-HexA-Aglycone II | - |
| 12 | 15.1 | 851.4412 | 851.4435 | C44H67O16 | 2.7 | 807, 671, 513, 471 | Hex-HexA-Aglycone II | - |
| 13 | 15.6 | 967.4901 | 967.4908 | C49H75O19 | 0.7 | 949, 905, 821, 671, 653, 609, 513, 471, 453 | dHex-Pen-HexA-Aglycone II | - |
| 14 | 16.9 | 649.3933 | 649.3957 | C36H57O10 | 3.8 | 631, 473 | HexA-Soyasapogenol A | - |
| 15 | 17.7 | 821.4303 | 821.4329 | C43H65O15 | 3.2 | 689, 671, 653, 609, 513, 471, 307 | Pen-HexA-Aglycone II | - |
| 16 | 18.3 | 999.5135 | 999.5170 | C50H79O20 | 3.6 | 981, 937, 853, 791, 673, 611, 515, 483, 473 | dHex-Hex-HexA-Aglycone I | - |
| 17 | 19.5 | 969.5074 | 969.5065 | C49H77O19 | −1.0 | 951, 907, 823, 761, 673, 611, 515, 473, 453 | dHex-Pen-HexA-Aglycone I | - |
| 18 | 19.9 | 853.4559 | 853.4591 | C44H69O17 | 3.8 | 835, 673, 655, 611, 515, 473, 337 | Hex-HexA-Aglycone I | - |
| 19 | 20.2 | 823.4463 | 823.4485 | C43H67O15 | 2.7 | 805, 673, 629, 611, 515, 473, 307 | Pen-HexA-Aglycone I | - |
| 20 | 23.9 | 837.4603 | 837.4642 | C44H69O15 | 2.0 | 657,639, 595, 499, 497, 457, 455, 453 | Hex-HexA-22-O-acetate-Soyasapogenol B | - |
| 21 | 24.8 | 939.4935 | 939.4959 | C48H75O18 | 2.6 | 921, 877, 793, 731, 613, 551, 483, 455 | Soyasaponin Be | [22,24,25,27,29,40,41] |
| 22 | 26.2 | 691.4042 | 691.4063 | C38H59O11 | 3.1 | 673, 515, 473 | HexA-Aglycone I | - |
| 23 | 31.0 | 941.5123 | 941.5115 | C48H77O18 | −0.8 | 923, 879, 795, 733, 615, 553, | Soyasaponin I | [22,24,27,28,29,40,41] |
| 24 | 32.6 | 795.4520 | 795.4536 | C42H67O14 | 2.1 | 777, 733, 615, 553, 457, 337 | Soyasaponin III | [27,28,29,41] |
| 25 | 33.0 | 911.5006 | 911.5010 | C47H75O17 | 0.5 | 893, 849, 765, 703, 615, 553, | Soyasaponin IIa | [27,28,29,41] |
| 26 | 34.0 | 765.4418 | 765.4431 | C41H65O13 | 1.6 | 747, 615, 597, 553, 457, 307 | Soyasaponin IV | [27,28,29,41] |
| 27 | 36.9 | 925.5168 | 925.5166 | C48H77O17 | −0.2 | 907, 863, 779, 717, 599, 537, 483, 441, 439 | dHex-Hex-HexA-Olean-12-ene-3β,24-diol | - |
| 28 | 39.3 | 779.4562 | 779.4587 | C42H67O13 | 3.2 | 599, 441, 439, 307 | Hex-HexA-Olean-12-ene-3β,24-diol | - |
| 29 | 41.3 | 633.3998 | 633.4008 | C36H57O9 | 1.5 | 615, 457 | Soyasapogenol B monoglucuronide | [28,42] |
| 30 | 44.1 | 895.5043 | 895.5061 | C47H75O16 | 1.9 | 877, 833, 749, 687, 599, 537, 453, 441, 439, 337 | dHex-Pen-HexA-Olean-12-ene-3β,24-diol | - |
| 31 | 47.8 | 793.4348 | 793.4380 | C42H65O14 | 4.0 | 775, 731, 613, 551, 455, 337 | Soyasaponin Be' | [24,30] |
| 32 | 48.0 | 909.4843 | 909.4853 | C47H73O17 | 1.1 | 891, 847, 763, 701, 613, 551, 455, 453 | Soyasaponin Bg | [25,30] |
| 33 | 48.7 | 763.4286 | 763.4274 | C41H63O13 | −1.5 | 719, 613, 569, 455, 307 | Soyasaponin Bg' | [30] |
| 34 | 51.1 | 749.4485 | 749.4482 | C41H65O12 | −0.5 | 599, 537, 441, 439, 307 | Pen-HexA-Olean-12-ene-3β,24-diol | - |
| 35 | 58.8 | 631.3856 | 631.3852 | C36H55O9 | −0.7 | 613, 455 | HexA-Soyasapogenol E | - |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nascimento, Y.M.; Abreu, L.S.; Lima, R.L.; Costa, V.C.O.; Melo, J.I.M.d.; Braz-Filho, R.; Silva, M.S.; Tavares, J.F. Rapid Characterization of Triterpene Saponins from Zornia brasiliensis by HPLC-ESI-MS/MS. Molecules 2019, 24, 2519. https://doi.org/10.3390/molecules24142519
Nascimento YM, Abreu LS, Lima RL, Costa VCO, Melo JIMd, Braz-Filho R, Silva MS, Tavares JF. Rapid Characterization of Triterpene Saponins from Zornia brasiliensis by HPLC-ESI-MS/MS. Molecules. 2019; 24(14):2519. https://doi.org/10.3390/molecules24142519
Chicago/Turabian StyleNascimento, Yuri Mangueira, Lucas Silva Abreu, Ramon Leal Lima, Vicente Carlos O. Costa, José Iranildo Miranda de Melo, Raimundo Braz-Filho, Marcelo Sobral Silva, and Josean Fechine Tavares. 2019. "Rapid Characterization of Triterpene Saponins from Zornia brasiliensis by HPLC-ESI-MS/MS" Molecules 24, no. 14: 2519. https://doi.org/10.3390/molecules24142519
APA StyleNascimento, Y. M., Abreu, L. S., Lima, R. L., Costa, V. C. O., Melo, J. I. M. d., Braz-Filho, R., Silva, M. S., & Tavares, J. F. (2019). Rapid Characterization of Triterpene Saponins from Zornia brasiliensis by HPLC-ESI-MS/MS. Molecules, 24(14), 2519. https://doi.org/10.3390/molecules24142519








