Comparison of Physicochemical Properties of Starches from Flesh and Peel of Green Banana Fruit
Abstract
1. Introduction
2. Results and Discussion
2.1. Starch and Soluble Sugar Contents in Dry Flesh and Peel
2.2. Morphology and Granule Size of Starch
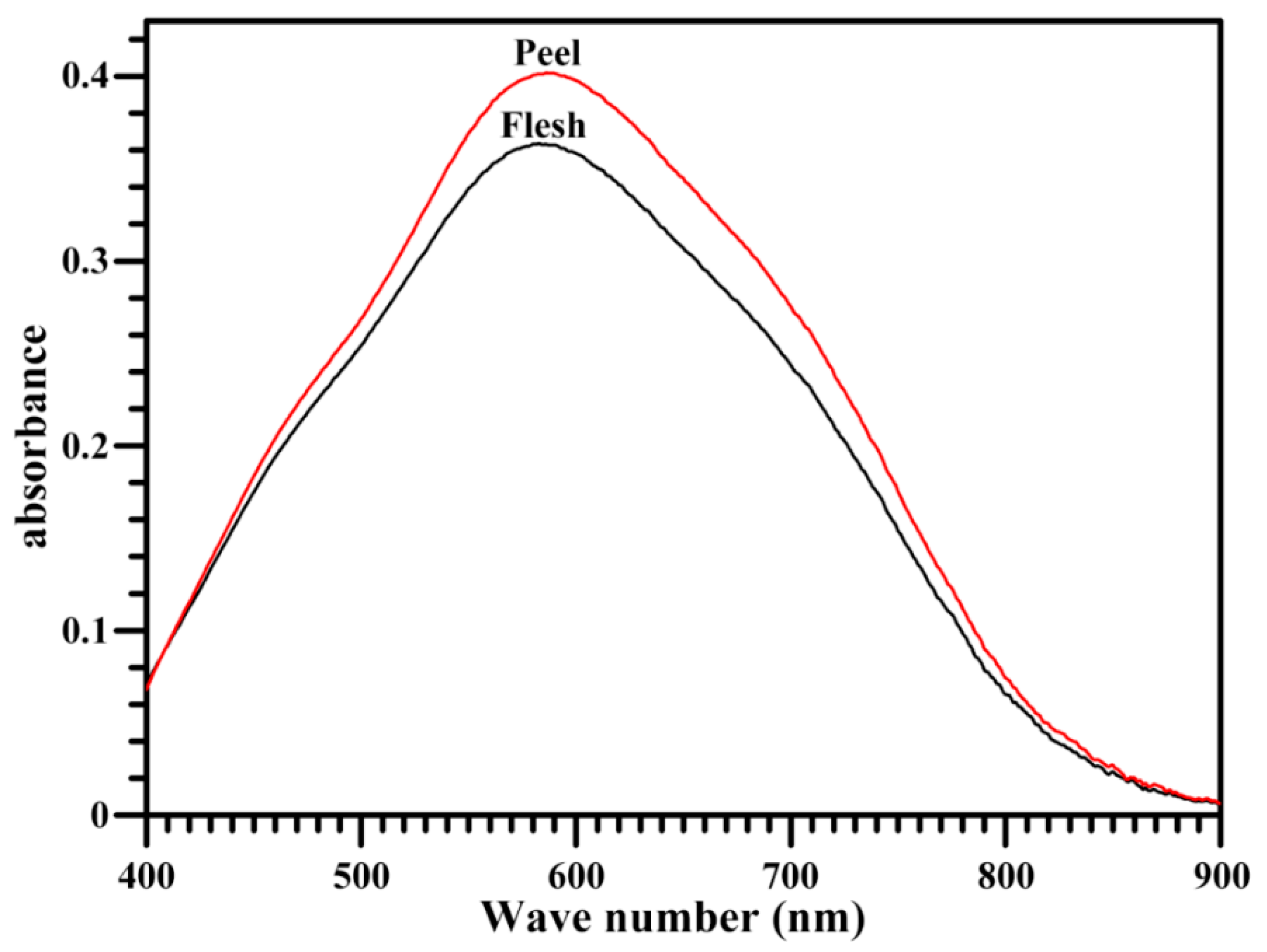
2.3. Iodine Absorption Spectrum and Apparent Amylose Content of Starch
2.4. Crystalline Structure of Starch
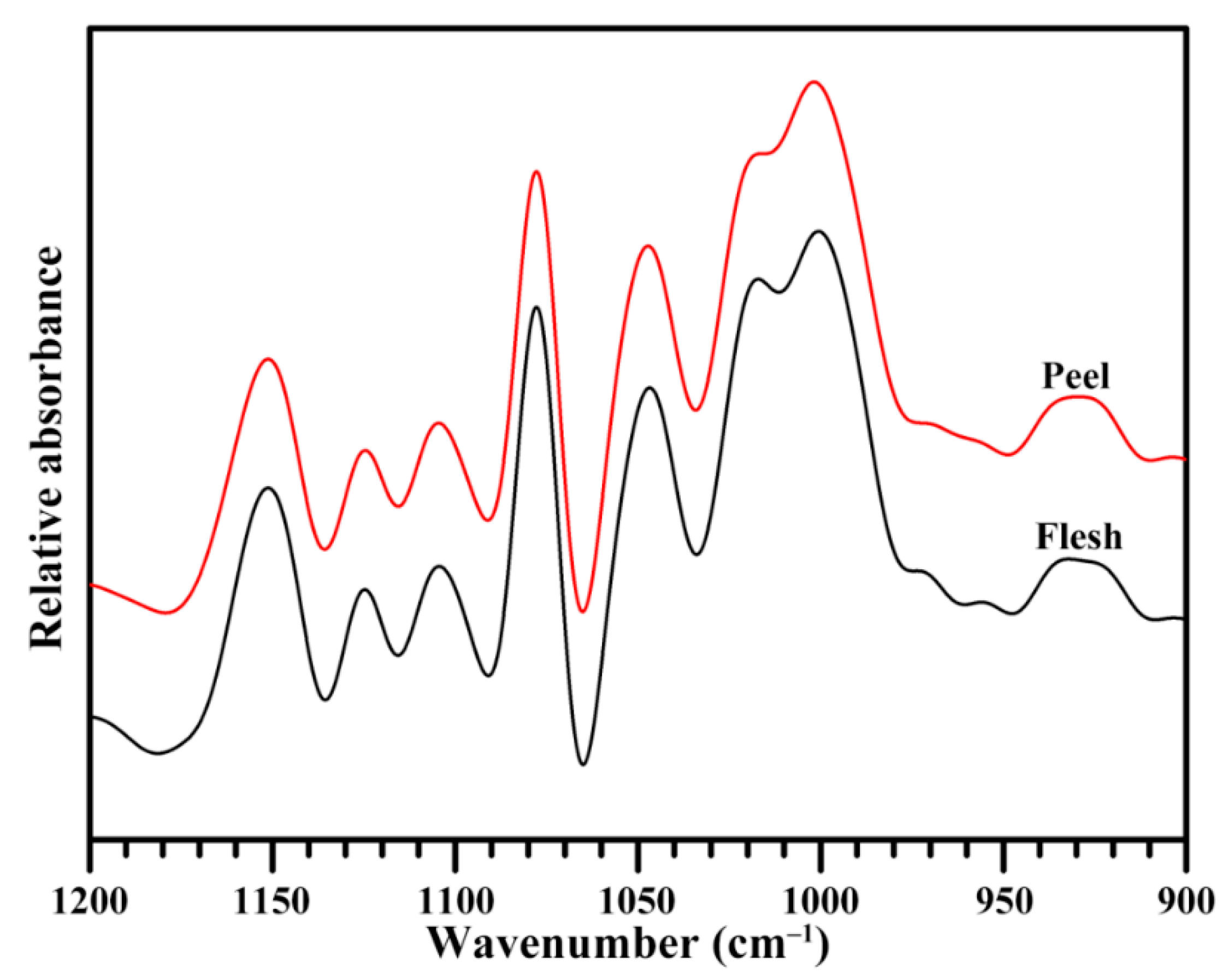
2.5. Short-Range Ordered Structure of Starch
2.6. Lamellar Structure of Starch
2.7. Thermal Properties of Starch
2.8. Swelling Power and Water Solubility of Starch
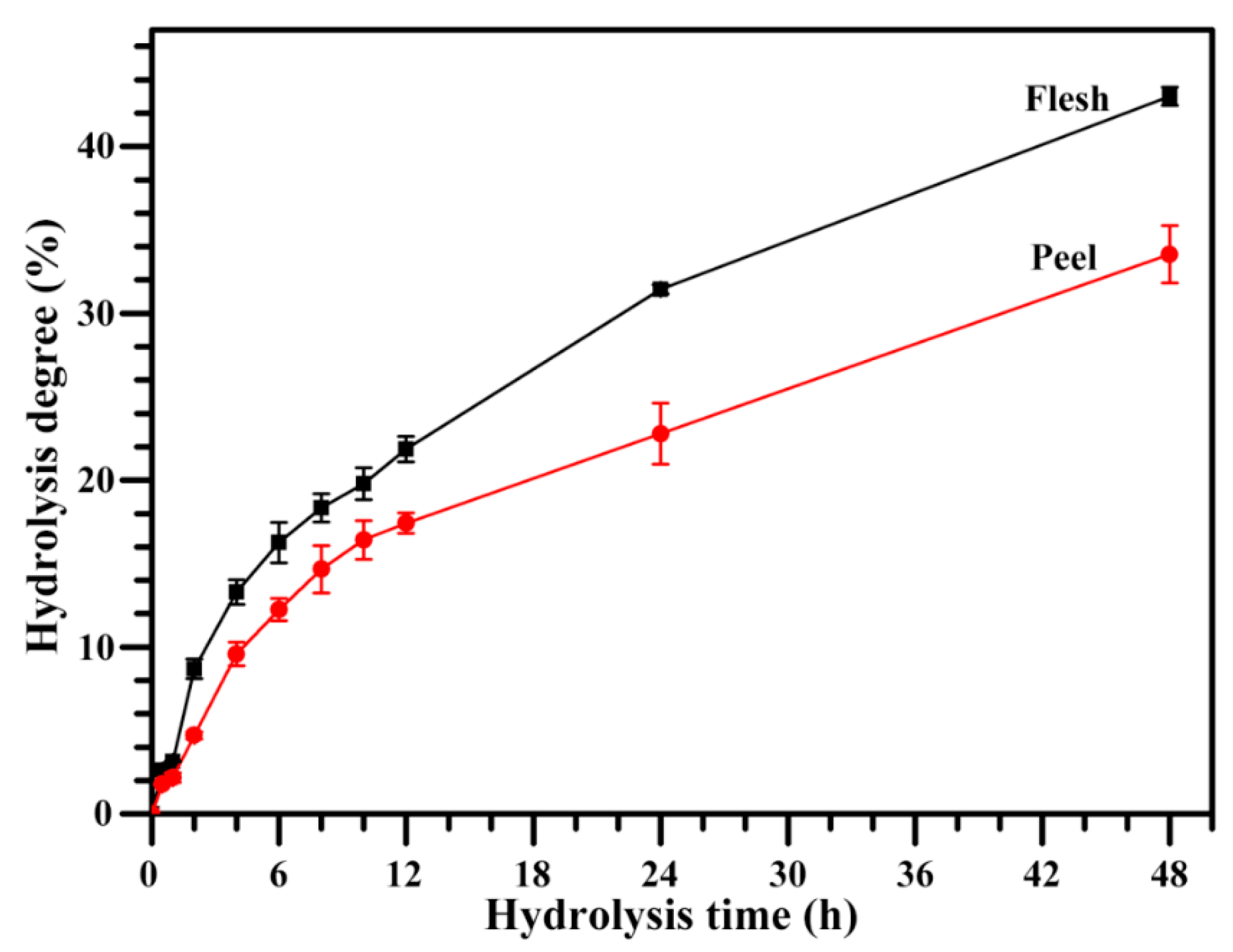
2.9. Hydrolysis of Starch
2.10. Digestion Properties of Starch
3. Materials and Methods
3.1. Plant Materials
3.2. Determination of Starch and Soluble Sugar Contents in Dry Flesh and Peel
3.3. Starch Isolation
3.4. Morphology Observation and Granule Size Analysis
3.5. Determination of Iodine Absorption Spectrum and AAC
3.6. Crystalline Structure Analysis
3.7. Short-Range Ordered Structure Analysis
3.8. Lamellar Structure Analysis
3.9. Determination of Swelling Power and Water Solubility
3.10. Determination of Thermal Properties
3.11. Determination of Hydrolysis Degree
3.12. Determination of Digestion Properties
3.13. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Emmambux, M.N.; Taylor, J.R.N. Morphology, physical, chemical, and functional properties of starches from cereals, legumes, and tubers cultivated in Afria: A review. Starch 2013, 65, 715–729. [Google Scholar] [CrossRef]
- García, N.L.; Famá, L.; Dufresne, A.; Aranguren, M.; Goyanes, S. A comparison between the physic-chemical properties of tuber and cereal starches. Food Res. Int. 2009, 42, 976–982. [Google Scholar] [CrossRef]
- Guo, K.; Lin, L.; Fan, X.; Zhang, L.; Wei, C. Comparison of structural and functional properties of starches from five fruit kernels. Food Chem. 2018, 257, 75–82. [Google Scholar] [CrossRef] [PubMed]
- Fan, X.; Zhang, S.; Lin, L.; Zhao, L.; Liu, A.; Wei, C. Properties of new starches from tubers of Arisaema elephas, yunnanense and erubescens. Food Hydrocoll. 2016, 61, 183–190. [Google Scholar] [CrossRef]
- Huang, J.; Zhao, L.; Man, J.; Wang, J.; Zhou, W.; Huai, H.; Wei, C. Comparison of physicochemical properties of B-type nontraditional starches from different sources. Int. J. Biol. Macromol. 2015, 78, 165–172. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Donner, E.; Yin, Y.; Huang, R.L.; Fan, M.Z. The physicochemical properties and in vitro digestibility of selected cereals, tubers and legumes grown in China. Food Chem. 2006, 99, 470–477. [Google Scholar] [CrossRef]
- Zhang, L.; Zhao, L.; Bian, X.; Guo, K.; Zhou, L.; Wei, C. Characterization and comparative study of starches from seven purple sweet potatoes. Food Hydrocoll. 2018, 80, 168–176. [Google Scholar] [CrossRef]
- Gao, H.; Cai, J.; Han, W.; Huai, H.; Chen, Y.; Wei, C. Comparison of starches isolated from three different Trapa species. Food Hydrocoll. 2014, 37, 174–181. [Google Scholar] [CrossRef]
- Huang, J.; Zhao, L.; Huai, H.; Li, E.; Zhang, F.; Wei, C. Structural and functional properties of starches from wild Trapa quadrispinosa, japonica, mammillifera and incise. Food Hydrocoll. 2015, 48, 117–126. [Google Scholar] [CrossRef]
- Singh, B.; Singh, J.P.; Kaur, A.; Singh, N. Bioactive compounds in banana and their associated health benefits: a review. Food Chem. 2016, 206, 1–11. [Google Scholar] [CrossRef] [PubMed]
- FAOSTAT. Available online: http://www.fao.org/faostat/en/#data/QC/visualize (accessed on 19 August 2018).
- Zhang, P.; Whistler, R.L.; BeMiller, J.N.; Hamaker, B.R. Banana starch: production, physicochemical properties, and digestibility: A review. Carbohydr. Polym. 2005, 59, 443–458. [Google Scholar] [CrossRef]
- Hernández-Carmona, F.; Morales-Matos, Y.; Lambis-Miranda, H.; Pasqualino, J. Starch extraction potential from plantain peel wastes. J. Environ. Chem. Eng. 2017, 5, 4980–4985. [Google Scholar] [CrossRef]
- Qamar, S.; Shaikh, A. Therapeutic potentials and compositional changes of valuable compounds from banana: A review. Trends Food Sci. Technol. 2018, 79, 1–9. [Google Scholar] [CrossRef]
- Aurore, G.; Parfait, B.; Fahrasmane, L. Bananas, raw materials for making processed food products. Trends Food Sci. Technol. 2009, 20, 78–91. [Google Scholar] [CrossRef]
- Subedi, P.P.; Walsh, K.B. Assessment of sugar and starch in intact banana and mango fruit by SWNIR spectroscopy. Postharvest Biol. Technol. 2011, 62, 238–245. [Google Scholar] [CrossRef]
- Jiang, H.; Zhang, Y.; Hong, Y.; Bi, Y.; Gu, Z.; Cheng, L.; Li, Z.; Li, C. Digestibility and changes to structural characteristics of green banana starch during in vitro digestion. Food Hydrocoll. 2015, 49, 192–199. [Google Scholar] [CrossRef]
- Savlak, N.; Türker, B.; Yeşilkanat, N. Effects of particle size distribution on some physical, chemical and functional properties of unripe banana flour. Food Chem. 2016, 213, 180–186. [Google Scholar] [CrossRef] [PubMed]
- Bi, Y.; Zhang, Y.; Jiang, H.; Hong, Y.; Gu, Z.; Cheng, L.; Li, Z.; Li, C. Molecular structure and digestibility of banana flour and starch. Food Hydrocoll. 2017, 72, 219–227. [Google Scholar] [CrossRef]
- Sarawong, C.; Schoenlechner, R.; Sekiguchi, K.; Berghofer, E.; Ng, P.K.G. Effect of extrusion cooking on the physicochemical properties, resistant starch, phenolic content and antioxidant capacities of green banana flour. Food Chem. 2014, 143, 33–39. [Google Scholar] [CrossRef] [PubMed]
- Anyasi, T.A.; Jideani, A.I.O.; Mchau, G.R.A. Effect of organic acid pretreatment on some physical, functional and antioxidant properties of flour obtained from three unripe banana cultivars. Food Chem. 2015, 172, 515–522. [Google Scholar] [CrossRef] [PubMed]
- Sardá, F.A.H.; de Lima, F.N.R.; Lopes, N.T.T.; Santos, A.O.; Tobaruela, E.C.; Kato, E.T.M.; Menezes, E.W. Identification of carbohydrate parameters in commercial unripe banana flour. Food Res. Int. 2016, 81, 203–209. [Google Scholar] [CrossRef]
- Rayo, L.M.; Carvalho, L.C.; Sardá, A.H.; Dacanal, G.C.; Menezes, E.W.; Tadini, C.C. Production of instant green banana flour (Musa cavendischii, var. Nanicao) by a pulsed-fluidized bed agglomeration. LWT-Food Sci. Technol. 2015, 63, 461–469. [Google Scholar] [CrossRef]
- Tribess, T.B.; Hernández-Uribe, J.P.; Méndez-Montealvo, M.G.C.; Menezes, E.W.; Bello-Perez, L.A.; Tadini, C.C. Thermal properties and resistant starch content of green banana flour (Musa cavendishii) produced at different drying conditions. LWT Food Sci. Technol. 2009, 42, 1022–1025. [Google Scholar] [CrossRef]
- Vatanasuchart, N.; Niyomwit, B.; Wongkrajang, K. Resistant starch content, in vitro starch digestibility and physico-chemical properties of flour and starch from Thai bananas. Maejo Int. J. Sci. Technol. 2012, 6, 259–271. [Google Scholar]
- Agama-Acevedo, E.; Nuñez-Santiago, M.C.; Alvarez-Ramirez, J.; Bello-Pérez, L.A. Physicochemical, digestibility and structural characteristics of starch isolated from banana cultivars. Carbohydr. Polym. 2015, 124, 17–24. [Google Scholar] [CrossRef] [PubMed]
- Peroni-Okita, F.H.G.; Simão, R.A.; Cardoso, M.B.; Soares, C.A.; Lajolo, F.M.; Cordenunsi, B.R. In vivo degradation of banana starch: Structural characterization of the degradation process. Carbohydr. Polym. 2010, 81, 291–299. [Google Scholar] [CrossRef]
- Espinosa-Solis, V.; Jane, J.L.; Bello-Perez, L.A. Physicochemical characteristics of starches from unripe fruits of mango and banana. Starch 2009, 61, 291–299. [Google Scholar] [CrossRef]
- Emaga, T.H.; Robert, C.; Ronkart, S.N.; Wathelet, B.; Paquot, M. Dietary fibre components and pectin chemical features of peels during ripening in banana and plantain varieties. Bioresour. Technol. 2008, 99, 4346–4354. [Google Scholar] [CrossRef] [PubMed]
- Oyewo, O.A.; Onyango, M.S.; Wolkersdorfer, C. Application of banana peels nanosorbent for the removal of radioactive minerals from real mine water. J. Environ. Radioact. 2016, 164, 369–376. [Google Scholar] [CrossRef] [PubMed]
- Rebello, L.P.G.; Ramos, A.M.; Pertuzatti, P.B.; Barcia, M.T.; Castillo-Muñoz, N.; Hermosín-Gutiérrez, I. Flour of banana (Musa AAA) peel as a source of antioxidant phenolic compounds. Food Res. Int. 2014, 55, 397–403. [Google Scholar] [CrossRef]
- Vu, H.T.; Scarlett, C.J.; Vuong, Q.V. Phenolic compounds within banana peel and their potential uses: A review. J. Funct. Foods 2018, 40, 238–248. [Google Scholar] [CrossRef]
- Eshak, N.S. Sensory evaluation and nutritional value of balady flat bread supplemented with banana peels as a natural source of dietary fiber. Ann. Agric. Sci. 2016, 61, 229–235. [Google Scholar] [CrossRef]
- Mahindrakar, K.V.; Rathod, V.K. Utilization of banana peels for removal of strontium (II) from water. Environ. Technol. Innov. 2018, 11, 371–383. [Google Scholar] [CrossRef]
- Alkarkhi, A.F.M.; Ramli, S.B.; Yong, Y.S.; Easa, A.M. Comparing physicochemical properties of banana pulp and peel flours prepared from green and ripe fruits. Food Chem. 2011, 129, 312–318. [Google Scholar] [CrossRef]
- Adão, R.C.; Glória, M.B.A. Bioactive amines and carbohydrate changes during ripening of ‘Prata’ banana (Musa acuminate × M. balbisiana). Food Chem. 2005, 90, 705–711. [Google Scholar] [CrossRef]
- Emaga, T.H.; Andrianaivo, R.H.; Wathelet, B.; Tchango, J.T.; Paquot, M. Effects of the stage of maturation and varieties on the chemical composition of banana and plantain peels. Food Chem. 2007, 103, 590–600. [Google Scholar] [CrossRef]
- Utrilla-Coello, R.G.; Rodríguez-Huezo, M.E.; Carrillo-Navas, H.; Hernández-Jaimes, C.; Vernon-Carter, E.J.; Alvarez-Ramirez, J. In vitro digestibility, physicochemical, thermal and rheological properties of banana starches. Carbohydr. Polym. 2014, 101, 154–162. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.; Cai, C.; Gilbert, R.G.; Li, E.; Wang, J.; Wei, C. Relationships between amylopectin molecular structures and functional properties of different-sized fractions of normal and high-amylose maize starches. Food Hydrocoll. 2016, 52, 359–368. [Google Scholar] [CrossRef]
- Lin, L.; Zhang, Q.; Zhang, L.; Wei, C. Evaluation of the molecular structural parameters of normal rice starch and their relationships with its thermal and digestion properties. Molecules 2017, 22, 1526. [Google Scholar] [CrossRef] [PubMed]
- He, W.; Wei, C. Progress in C-type starches from different plant sources. Food Hydrocoll. 2017, 73, 162–175. [Google Scholar] [CrossRef]
- Cai, J.; Man, J.; Huang, J.; Liu, Q.; Wei, W.; Wei, C. Relationship between structure and functional properties of normal rice starches with different amylose contents. Carbohydr. Polym. 2015, 125, 35–44. [Google Scholar] [CrossRef] [PubMed]
- Xu, B.; Man, J.M.; Wei, C.X. Methods for determining relative crystallinity of plant starch X-ray powder diffraction spectra. Chin. Bull. Bot. 2012, 47, 278–285. [Google Scholar]
- Sevenou, O.; Hill, S.E.; Farhat, I.A.; Mitchell, J.R. Organisation of the external region of the starch granule as determined by infrared spectroscopy. Int. J. Biol. Macromol. 2002, 31, 79–85. [Google Scholar] [CrossRef]
- Cai, C.; Cai, J.; Man, J.; Yang, Y.; Wang, Z.; Wei, C. Allomorph distribution and granule structure of lotus rhizome C-type starch during gelatinization. Food Chem. 2014, 142, 408–415. [Google Scholar] [CrossRef] [PubMed]
- Yuryev, V.P.; Krivandin, A.V.; Kiseleva, V.I.; Wasserman, L.A.; Genkina, N.K.; Fornal, J.; Blaszczak, W.; Schiraldi, A. Structural parameters of amylopectin clusters and semi-crystalline growth rings in wheat starches with different amylose content. Carbohydr. Res. 2004, 339, 2683–2691. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Fan, X.; Lin, L.; Zhao, L.; Liu, A.; Wei, C. Properties of starch from root tuber of Stephania epigaea in comparison with potato and maize starches. Int. J. Food Prop. 2017, 20, 1740–1750. [Google Scholar] [CrossRef]
- Englyst, H.N.; Kingman, S.M.; Cummings, J.H. Classification and measurement of nutritionally important starch fractions. Eur. J. Clin. Nutr. 1992, 45, S33–S50. [Google Scholar]
- Waliszewski, K.N.; Aparicio, M.A.; Bello, L.A.; Monroy, J.A. Changes of banana starch by chemical and physical modification. Carbohydr. Polym. 2003, 52, 237–242. [Google Scholar] [CrossRef]
- Cai, C.; Lin, L.; Man, J.; Zhao, L.; Wang, Z.; Wei, C. Different structural properties of high-amylose maize starch fractions varying in granule size. J. Agric. Food Chem. 2014, 62, 11711–11721. [Google Scholar] [CrossRef] [PubMed]
- Wei, C.; Qin, F.; Zhou, W.; Yu, H.; Xu, B.; Chen, C.; Zhu, L.; Wang, Y.; Gu, M.; Liu, Q. Granule structure and distribution of allomorphs in C-type high-amylose rice starch granule modified by antisense RNA inhibition of starch branching enzyme. J. Agric. Food Chem. 2010, 58, 11946–11954. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.; Guo, D.; Zhao, L.; Zhang, X.; Wang, J.; Zhang, F.; Wei, C. Comparative structure of starches from high-amylose maize inbred lines and their hybrids. Food Hydrocoll. 2016, 52, 19–28. [Google Scholar] [CrossRef]
Sample Availability: Samples of the starches are available from the authors. |
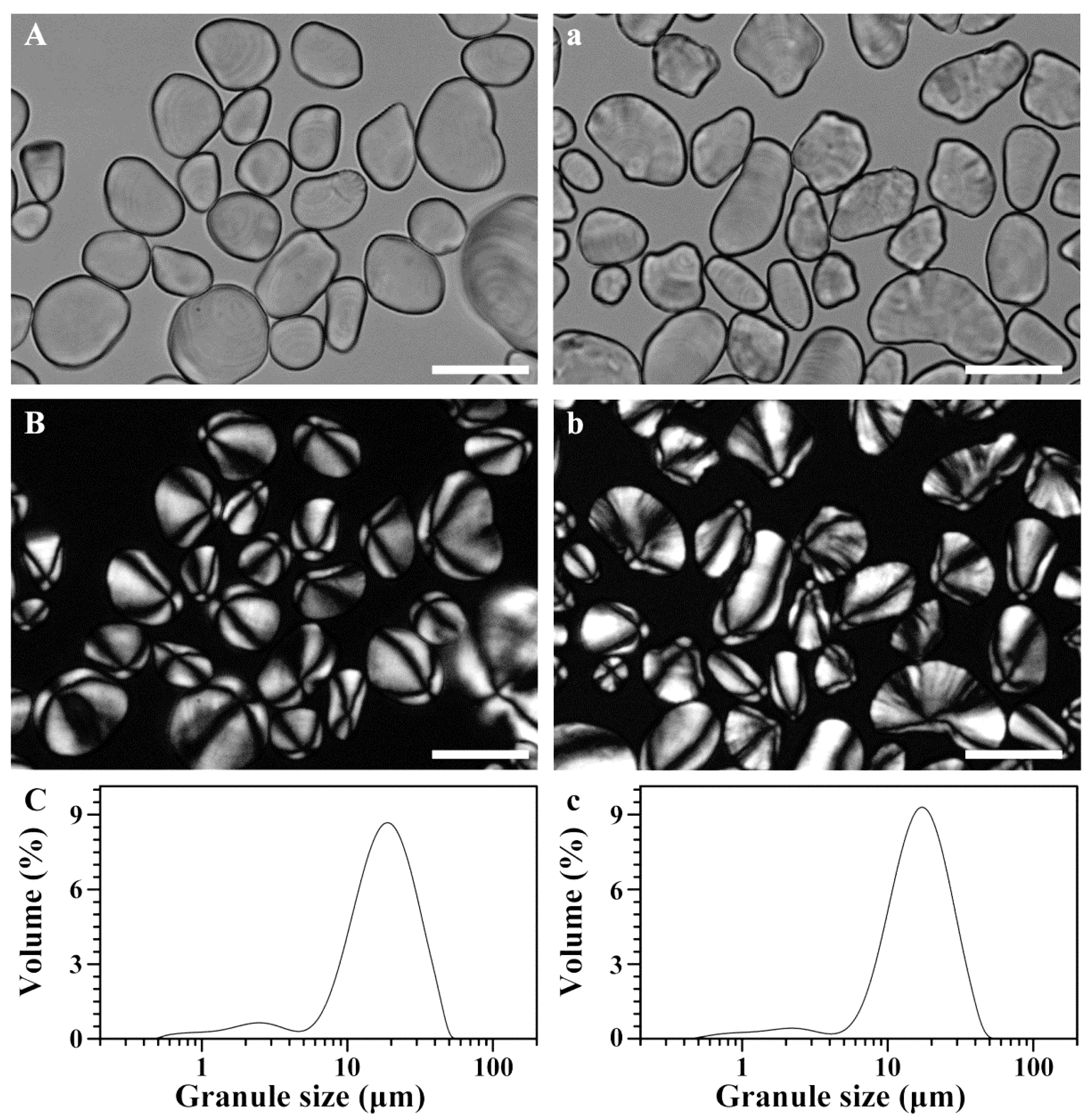




| Tissues | d(0.1) (μm) | d(0.5) (μm) | d(0.9) (μm) | D[4,3] (μm) |
|---|---|---|---|---|
| Flesh | 6.949 ± 0.001 | 16.439 ± 0.016 | 30.066 ± 0.034 | 17.496 ± 0.017 |
| Peel | 7.450 ± 0.002 *** | 15.355 ± 0.001 *** | 27.249 ± 0.005 *** | 16.353 ± 0.001 *** |
| Tissues | λmax (nm) | AAC (%) | RC (%) |
|---|---|---|---|
| Flesh | 584.5 ± 0.9 | 21.3 ± 0.3 | 28.0 ± 0.8 |
| Peel | 586.5 ± 0.5 * | 25.7 ± 0.3 *** | 26.1 ± 0.5 |
| Tissues | To (°C) | Tp (°C) | Tc (°C) | ΔT (°C) | ΔH (J·g−1) |
|---|---|---|---|---|---|
| Flesh | 66.4 ± 0.1 | 69.8 ± 0.1 | 74.0 ± 0.6 | 7.6 ± 0.5 | 15.8 ± 0.6 |
| Peel | 68.0 ± 0.1 *** | 71.6 ± 0.1 *** | 76.4 ± 0.3 ** | 8.4 ± 0.2 | 16.1 ± 0.8 |
| Tissues | Native Starch | Gelatinized Starch | ||||
|---|---|---|---|---|---|---|
| RDS (%) | SDS (%) | RS (%) | RDS (%) | SDS (%) | RS (%) | |
| Flesh | 1.7 ± 0.2 | 4.3 ± 0.3 | 94.1 ± 0.2 | 73.0 ± 1.1 | 5.1 ± 0.2 | 21.9 ± 1.2 |
| Peel | 1.4 ± 0.3 | 3.4 ± 0.2 * | 95.2 ± 0.3 * | 72.3 ± 0.7 | 4.5 ± 1.0 | 23.2 ± 0.5 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, Z.; Guo, K.; Lin, L.; He, W.; Zhang, L.; Wei, C. Comparison of Physicochemical Properties of Starches from Flesh and Peel of Green Banana Fruit. Molecules 2018, 23, 2312. https://doi.org/10.3390/molecules23092312
Li Z, Guo K, Lin L, He W, Zhang L, Wei C. Comparison of Physicochemical Properties of Starches from Flesh and Peel of Green Banana Fruit. Molecules. 2018; 23(9):2312. https://doi.org/10.3390/molecules23092312
Chicago/Turabian StyleLi, Zheng, Ke Guo, Lingshang Lin, Wei He, Long Zhang, and Cunxu Wei. 2018. "Comparison of Physicochemical Properties of Starches from Flesh and Peel of Green Banana Fruit" Molecules 23, no. 9: 2312. https://doi.org/10.3390/molecules23092312
APA StyleLi, Z., Guo, K., Lin, L., He, W., Zhang, L., & Wei, C. (2018). Comparison of Physicochemical Properties of Starches from Flesh and Peel of Green Banana Fruit. Molecules, 23(9), 2312. https://doi.org/10.3390/molecules23092312






