The Potential of Phytomelatonin as a Nutraceutical
Abstract
:1. Introduction
2. Phytomelatonin in Plants
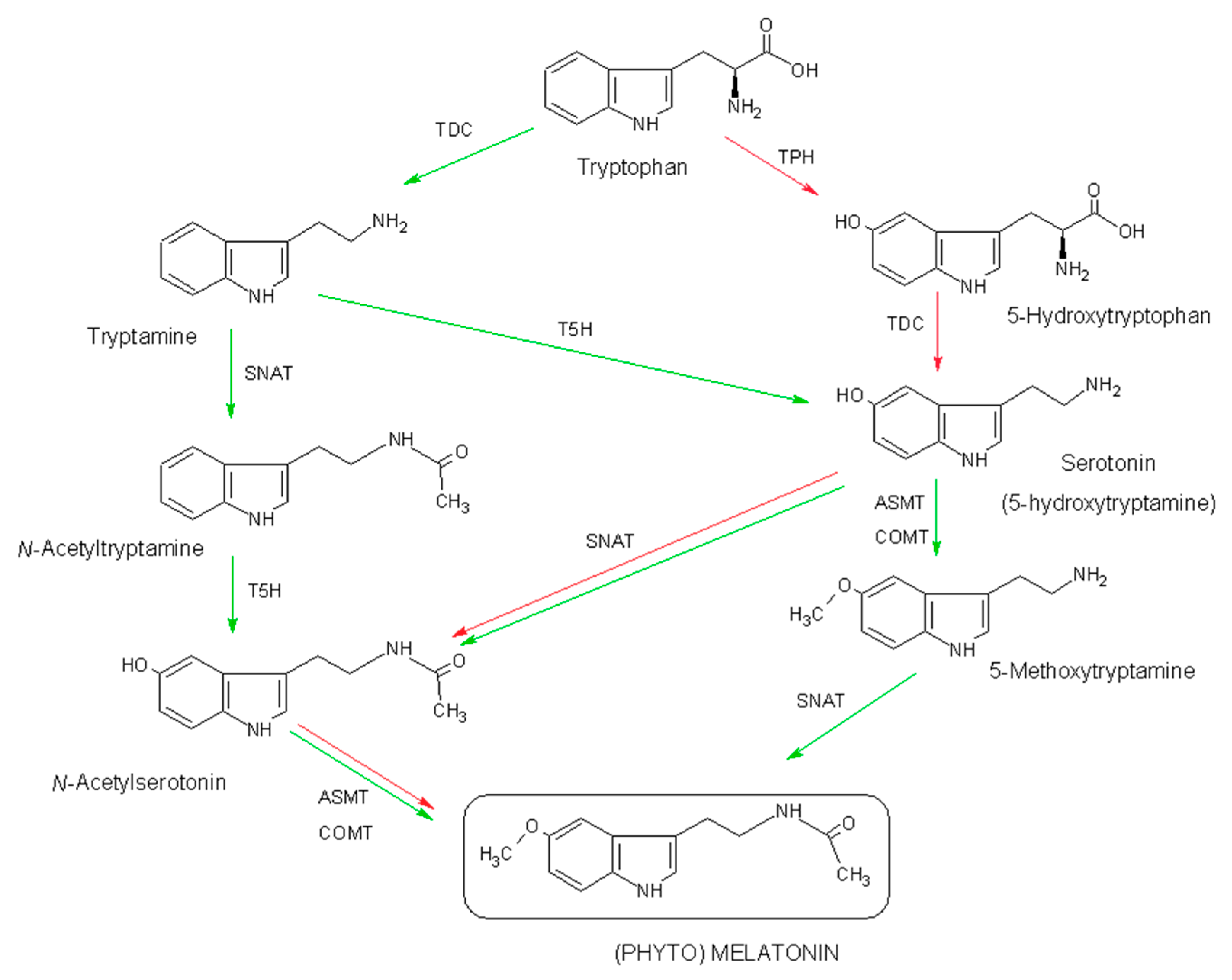
2.1. Biosynthesis of Phytomelatonin
2.2. Levels of Phytomelatonin
3. Phytomelatonin as a Nutraceutical
- 2000 B.C.—Here, eat this root.
- 1000 A.D.—That root is heathen. Here, say this prayer.
- 1850 A.D.—That prayer is superstition. Here, drink this potion.
- 1940 A.D.—That potion is snake oil. Here, swallow this pill.
- 1985 A.D.—That pill is ineffective. Here, take this antibiotic.
- 2000 A.D.—That antibiotic is artificial. Here, eat this root.
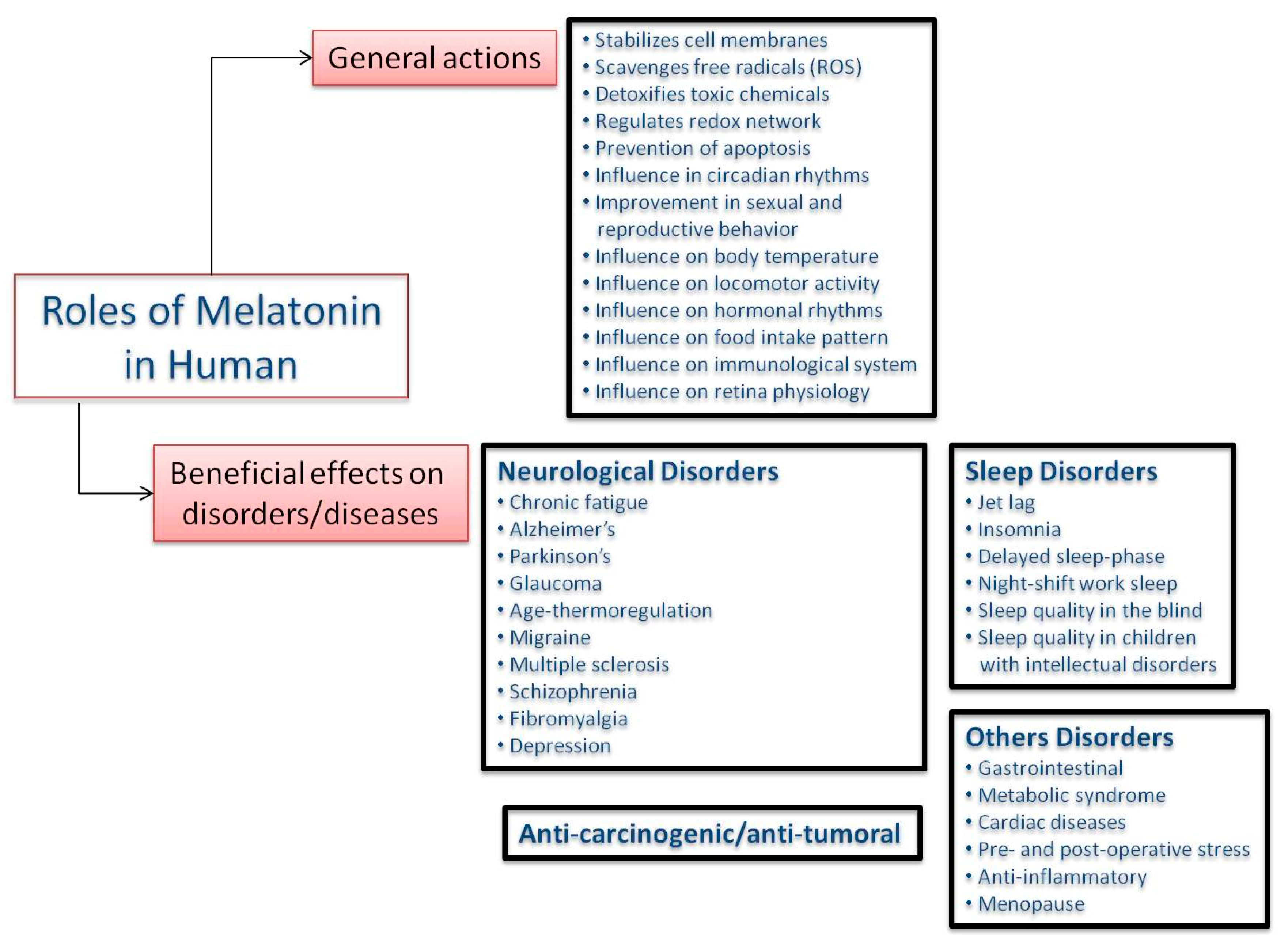
3.1. Melatonin in Humans
3.2. Phytomelatonin as an Antioxidant
3.3. Phytomelatonin as Anti-Carcinogen
3.4. Phytomelatonin as Gastrointestinal Tract Protector
3.5. Phytomelatonin as Sleep-Quality Enhancer and Jet Lag Remedy
4. Sources of Melatonin and Phytomelatonin
5. Availability of Melatonin and Phytomelatonin Supplements
6. Concluding Remarks
- The presence of phytomelatonin in all plant species analyzed to date has opened the door to its use as a nutraceutical compound.
- Aromatic and medicinal plants present higher phytomelatonin levels than ordinary vegetables. Such botanical herbs are optimal candidates for use in future melatonin supplements.
- The search for and study of phytomelatonin-rich species and varieties should be regarded as a priority.
- The control of growth conditions might help to obtain phytomelatonin-rich plants. A greater number of studies with respect to more plants and varieties is necessary.
- Phytomelatonin has great potential as a therapy for many diseases both at physiological and pharmacological doses.
- Its antioxidant, anti-carcinogenic, and rhythm-synchronizing properties represent an excellent area in which its use as a nutraceutical could have wide beneficial applications.
- The absence of undesirable contaminants generated during the chemical synthesis of melatonin is the key factor in choosing phytomelatonin as a “100% natural” supplement.
- New preparations containing phytomelatonin will inevitably appear, with synthetic supplements almost certainly being displaced by natural substances.
- It is to be hoped that such products will contain natural substances whose origin and possible contamination will be strictly controlled.
- However, the possible harmful effects of phytochemicals (other than phytomelatonin) of either vegetable origin (such as alkaloids), or of non-natural origin (such as pesticides), with a possible presence in plant extracts, should be taken into account and studied in depth.
Conflicts of Interest
References
- Herxheimer, A. Jet lag. Clin. Evid. 2005, 13, 2178–2183. [Google Scholar]
- Reiter, R.J. The melatonin rhythm: Both a clock and a calendar. Experientia 1993, 49, 654–664. [Google Scholar] [CrossRef] [PubMed]
- Saper, C.B.; Scammell, T.E.; Lu, J. Hypothalamic regulation of sleep and circadian rhythms. Nature 2005, 437, 1257–1263. [Google Scholar] [CrossRef] [PubMed]
- Jan, J.E.; Reiter, R.J.; Wasdell, M.B.; Bax, M. The role of the thalamus in sleep, pineal melatonin production, and circadian rhythm sleep disorders. J. Pineal Res. 2009, 46, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Paredes, S.; Marchena, A.; Bejarano, I.; Espino, J.; Barriga, C.; Rial, R.; Reiter, R.; Rodriguez, A. Melatonin and tryptophan affect the activity-rest rhythm, core and peripheral temperatures, and interleukin levels in the ringdove: Changes with age. J. Gerontol. Biol. Sci. 2009, 64, 340–350. [Google Scholar] [CrossRef] [PubMed]
- Carrillo-Vico, A.; Lardone, P.J.; Alvarez-Sanchez, N.; Rodríguez-Rodriguez, A.; Guerrero, J.M. Melatonin: Buffering the immune system. Int. J. Mol. Sci. 2013, 14, 8638–8683. [Google Scholar] [CrossRef] [PubMed]
- Marková, M.; Adámeková, E.; Kubatka, P.; Bojková, B.; Ahlersová, E.; Ahlers, I. Effect of prolonged melatonin administration on metabolic parameters and organ weights in young male and female Sprague-Dawley rats. Acta Vet. Brno 2003, 72, 163–173. [Google Scholar] [CrossRef]
- Reiter, R.J.; Tan, D.; Poeggeler, B.; Menendez-Pelaez, A.; Chen, L.; Saarela, S. Melatonin as a free-radical scavenger. Implications for aging and age-related diseases. Ann. N. Y. Acad. Sci. 1994, 714, 1–12. [Google Scholar] [CrossRef]
- Reiter, R.J.; Tan, D.X.; Qi, W.; Manchester, L.C.; Karbownik, M.; Calvo, J.R. Pharmacology and physiology of melatonin in the reduction of oxidative stress in vivo. Neurosignals 2000, 9, 160–171. [Google Scholar] [CrossRef] [PubMed]
- Catalá, A. The ability of melatonin to counteract lipid peroxidation in biological membranes. Curr. Mol. Med. 2007, 7, 638–649. [Google Scholar] [CrossRef] [PubMed]
- Hardeland, R. Melatonin in aging and disease. Multiple consequences of reduced secretion, options and limits of treatment. Aging Dis. 2012, 3, 194–225. [Google Scholar] [PubMed]
- Witt-Enderby, P.A.; Radio, N.M.; Doctor, J.S.; Davies, V.L. Therapeutics treatments potentially mediated by melatonin receptors: Potential clinics uses in the prevention of osteoporosis, cancer and as an adjuvant therapy. J. Pineal Res. 2006, 41, 297–305. [Google Scholar] [CrossRef] [PubMed]
- Di Bella, G.; Mascia, F.; Gualano, L.; Di Bella, L. Melatonin anticancer effects: Review. Int. J. Mol. Sci. 2013, 14, 2410–2430. [Google Scholar] [CrossRef] [PubMed]
- Seely, D.; Wu, P.; Fritz, H.; Kennedy, D.A.; Tsui, T.; Seely, A.J.E.; Mills, E. Melatonin as adjuvant cancer care with and without chemotherapy: A systematic review and meta-analysis of randomized trials. Integr. Cancer Ther. 2012, 11, 293–303. [Google Scholar] [CrossRef] [PubMed]
- Mociková, K.; Mníchová, M.; Kubatka, P.; Bojková, B.; Ahlers, I.; Ahlersová, E. Mammary carcinogenesis induced in Wistarhan rats by the combination of ionizing radiation and dimethylbenz(a)anthracene: Prevention with melatonin. Neoplasma 2000, 47, 227–229. [Google Scholar]
- Blask, D.E.; Dauchy, R.T.; Sauer, L.A.; Krause, J.A. Melatonin uptake and growth prevention in rat hepatoma 7288CTC in response to dietary melatonin: Melatonin receptor-mediated inhibition of tumor linoleic acid metabolism to the growth signaling molecule 13-hydroxyoctadecadienoic acid and the potential role of phytomelatonin. Carcinogenesis 2004, 25, 951–960. [Google Scholar] [PubMed]
- Van Tassel, D.L.; O`Neill, S.D. Melatonin: Identification of a potential dark signal in plants. Plant Physiol. 1993, 102, 659. [Google Scholar]
- van Tassel, D.L.; Roberts, N.; O`Neill, S.D. Melatonin from higher plants: Isolation and identification of N-acetyl-5-methoxytryptamine. Plant Physiol. 1995, 108, 101. [Google Scholar]
- Dubbels, R.; Reiter, R.J.; Klenke, E.; Goebel, A.; Schnakenberg, E.; Ehlers, C.; Schiwara, H.W.; Schloot, W. Melatonin in edible plants identified by radioimmunoassay and by HPLC-MS. J. Pineal Res. 1995, 18, 28–31. [Google Scholar] [CrossRef] [PubMed]
- Hattori, A.; Migitaka, H.; Iigo, M.; Yamamoto, K.; Ohtani-Kaneko, R.; Hara, M.; Suzuki, T.; Reiter, R.J. Identification of melatonin in plants and its effects on plasma melatonin levels and binding to melatonin receptors in vertebrates. Biochem. Mol. Biol. Int. 1995, 35, 627–634. [Google Scholar] [PubMed]
- Kolar, J.; Machackova, I.; Illnerova, H.; Prinsen, E.; van Dongen, W.; van Onckelen, H. Melatonin in higher plant determined by radioimmunoassay and liquid chromatography-mass spectrometry. Biol. Rhythm Res. 1995, 26, 406–409. [Google Scholar]
- Arnao, M.B.; Hernández-Ruiz, J. The physiological function of melatonin in plants. Plant Signal. Behav. 2006, 1, 89–95. [Google Scholar] [CrossRef] [PubMed]
- Back, K.; Tan, D.X.; Reiter, R.J. Melatonin biosynthesis in plants: Multiple pathways catalyze tryptophan to melatonin in the cytoplasm or chloroplasts. J. Pineal Res. 2016, 61, 426–437. [Google Scholar] [CrossRef] [PubMed]
- Tan, D.X.; Manchester, C.L.; Esteban-Zubero, E.; Zhou, Z.; Reiter, J.R. Melatonin as a potent and inducible endogenous antioxidant: Synthesis and metabolism. Molecules 2015, 20, 18886–18906. [Google Scholar] [CrossRef] [PubMed]
- Byeon, Y.; Lee, H.Y.; Lee, K.; Back, K. Caffeic acid O-methyltransferase is involved in the synthesis of melatonin by methylating N-acetylserotonin in Arabidopsis. J. Pineal Res. 2014, 57, 219–227. [Google Scholar] [CrossRef] [PubMed]
- Park, S.; Lee, K.; Kim, Y.S.; Back, K. Tryptamine 5-hydroxylase-deficient Sekiguchi rice induces synthesis of 5-hydroxytryptophan and N-acetyltryptamine but decreases melatonin biosynthesis during senescence process of detached leaves. J. Pineal Res. 2012, 52, 211–216. [Google Scholar] [CrossRef] [PubMed]
- Kang, S.; Kang, K.; Lee, K.; Back, K. Characterization of tryptamine 5-hydroxylase and serotonin synthesis in rice plants. Plant Cell Rep. 2007, 26, 2009–2015. [Google Scholar] [CrossRef] [PubMed]
- Arnao, M.B.; Hernández-Ruiz, J. Melatonin: Plant growth regulator and/or biostimulator during stress? Trends Plant Sci. 2014, 19, 789–797. [Google Scholar] [CrossRef] [PubMed]
- Tan, D.X.; Hardeland, R.; Back, K.; Manchester, L.C.; Latorre-Jimenez, M.A.; Reiter, R.J. On the significance of an alternate pathway of melatonin synthesis via 5-methoxytryptamine: Comparisons across species. J. Pineal Res. 2016, 61, 27–40. [Google Scholar] [CrossRef] [PubMed]
- Byeon, Y.; Tan, D.X.; Reiter, R.J.; Back, K. Predominance of 2-hydroxymelatonin over melatonin in plants. J. Pineal Res. 2015, 59, 448–454. [Google Scholar] [CrossRef] [PubMed]
- Paredes, S.D.; Korkmaz, A.; Manchester, L.C.; Tan, D.X.; Reiter, R.J. Phytomelatonin: A review. J. Exp. Bot. 2009, 60, 57–69. [Google Scholar] [CrossRef] [PubMed]
- Arnao, M.B. Phytomelatonin: Discovery, content, and role in plants. Adv. Bot. 2014, 2014, e815769. [Google Scholar] [CrossRef]
- Feng, X.; Wang, M.; Zhao, Y.; Han, P.; Dai, Y. Melatonin from different fruit sources, functional sources, and analytical methods. Trends Food Sci. Technol. 2014, 37, 21–31. [Google Scholar] [CrossRef]
- Ramakrishna, A.; Giridhar, P.; Sankar, K.; Ravishankar, G. Melatonin and serotonin profiles in beans of Coffea species. J. Pineal Res. 2012, 52, 470–476. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.; Huo, Y.; Tan, D.X.; Liang, Z.; Zhang, W.; Zhang, Y. Melatonin in chinese medicinal herbs. Life Sci. 2003, 73, 19–26. [Google Scholar] [CrossRef]
- Manchester, L.C.; Tan, D.X.; Reiter, R.J.; Park, W.; Monis, K.; Qi, W. High levels of melatonin in the seeds of edible plants. Possible function in germ tissue protection. Life Sci. 2000, 67, 3023–3029. [Google Scholar] [CrossRef]
- Aguilera, Y.; Rebollo-Hernanz, M.; Herrera, T.; Cayuelas, L.T.; Rodriguez-Rodriguez, P.; de Pablo, A.L.L.; Arribas, S.M.; Martin-Cabrejas, M.A. Intake of bean sprouts influences melatonin and antioxidant capacity biomarker levels in rats. Food Funct. 2016, 7, 1438–1445. [Google Scholar] [CrossRef] [PubMed]
- Lei, Q.; Wang, L.; Tan, D.X.; Zhao, Y.; Zheng, X.D.; Chen, H.; Li, Q.T.; Zuo, B.X.; Kong, J. Identification of genes for melatonin synthetic enzymes in Red Fuji apple (Malus domestica Borkh. cv. Red) and their expression and melatonin production during fruit development. J. Pineal Res. 2013, 55, 443–451. [Google Scholar] [PubMed]
- Zhao, Y.; Tan, D.X.; Lei, Q.; Chen, H.; Wang, L.; Li, Q.T.; Gao, Y.; Kong, J. Melatonin and its potential biological functions in the fruits of sweet cherry. J. Pineal Res. 2013, 55, 79–88. [Google Scholar] [CrossRef] [PubMed]
- González-Gómez, D.; Lozano, M.; Fernández-León, M.; Ayuso, M.; Bernalte, M.; Rodríguez, A. Detection and quantification of melatonin and serotonin in eight sweet cherry cultivars (Prunus avium L.). Eur. Food Res. Technol. 2009, 229, 223–229. [Google Scholar] [CrossRef]
- Sturtz, M.; Cerezo, A.; Cantos-Villar, E.; Garcia-Parrilla, M. Determination of the melatonin content of different varieties of tomatoes (Lycopersicon esculentum) and strawberries (Fragaria ananassa). Food Chem. 2011, 127, 1329–1334. [Google Scholar] [CrossRef] [PubMed]
- Korkmaz, A.; Deger, O.; Cuci, Y. Profiling the melatonin content in organs of the pepper plant during different growth stages. Sci. Hortic. 2014, 172, 242–247. [Google Scholar] [CrossRef]
- Stege, P.W.; Sombra, L.L.; Messina, G.; Martinez, L.D.; Silva, M.F. Determination of melatonin in wine and plant extracts by capillary electrochromatography with immobilized carboxylic multi-walled carbon nanotubes as stationary phase. Electrophoresis 2010, 31, 2242–2248. [Google Scholar] [CrossRef] [PubMed]
- Vitalini, S.; Gardana, C.; Zanzotto, A.; Simonetti, P.; Faoro, F.; Fico, G.; Iriti, M. The presence of melatonin in grapevine (Vitis vinifera L.) berry tissues. J. Pineal Res. 2011, 51, 331–337. [Google Scholar] [CrossRef] [PubMed]
- Afreen, F.; Zobayed, S.M.A.; Kozai, T. Melatonin in Glycyrrhiza uralensis: Response of plant roots to spectral quality of light and UV-B radiation. J. Pineal Res. 2006, 41, 108–115. [Google Scholar] [CrossRef] [PubMed]
- Murch, S.J.; Saxena, P.K. A melatonin-rich germplasm line of St John’s wort (Hypericum perforatum L.). J. Pineal Res. 2006, 41, 284–287. [Google Scholar] [CrossRef] [PubMed]
- Murch, S.J.; Simmons, C.B.; Saxena, P.K. Melatonin in feverfew and other medicinal plants. Lancet 1997, 350, 1598–1599. [Google Scholar] [CrossRef]
- Marioni, F.; Bertoli, A.; Pistelli, L. A straightforward procedure to biosynthesise melatonin using freshly chopped Achillea millefolium L. as reagent. Phytochem. Lett. 2008, 1, 107–110. [Google Scholar] [CrossRef]
- Arnao, M.B.; Hernández-Ruiz, J. Growth conditions influence the melatonin content of tomato plants. Food Chem. 2013, 138, 1212–1214. [Google Scholar] [CrossRef] [PubMed]
- Arnao, M.B.; Hernández-Ruiz, J. Functions of melatonin in plants: A review. J. Pineal Res. 2015, 59, 133–150. [Google Scholar] [CrossRef] [PubMed]
- Arnao, M.B.; Hernández-Ruiz, J. Melatonin in its relationship to plant hormones. Ann. Bot. 2018, 121. [Google Scholar] [CrossRef] [PubMed]
- Arnao, M.B.; Hernández-Ruiz, J. Growth activity, rooting capacity, and tropism: Three auxinic precepts fulfilled by melatonin. Acta Physiol. Plant 2017, 39, 127. [Google Scholar] [CrossRef]
- DeFelice, S.L. The nutraceutical revolution: Its impact on food industry R&D. Trends Food Sci. Technol. 1995, 6, 59–61. [Google Scholar]
- Yu, P.; Hegeman, A.D.; Cohen, J.D. A facile means for the identification of indolic compounds from plant tissues. Plant J. 2014, 79, 1065–1075. [Google Scholar] [CrossRef] [PubMed]
- Pandey, M.; Verma, R.K.; Saraf, S.A. Nutraceuticals: New era of medicine and health. Asian J. Pharm. Clin. Res. 2010, 3, 11–15. [Google Scholar]
- Singh, J.; Sinha, S. Classification, Regulatory Acts and Applications of Nutraceuticals for Health. Int. J. Pharma Biosci. 2012, 2, 177–187. [Google Scholar]
- Espín, J.C.; García-Conesa, M.T.; Tomás-Barberán, F.A. Nutraceuticals: Facts and fiction. Phytochemistry 2007, 68, 2986–3008. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, S.; Sharma, P.K.; Kumara, S. Nutraceuticals: A review. J. Chronother. Drug Deliv. 2015, 6, 1–10. [Google Scholar]
- Nasri, H.; Baradaran, A.; Shirzad, H.; Kopaei, M.R. New concepts in nutraceuticals as alternative for pharmaceuticals. Int. J. Prev. Med. 2014, 5, 1487–1499. [Google Scholar] [PubMed]
- Rowe, S.B. What is a Nutrient? Defining the Food-Drug Continuum. In Round Table Forum, Moderator’s Comments; Georgetown University: Washington, DC, USA, 1999. [Google Scholar]
- Maronde, E.; Stehle, J.H. The mammalian pineal gland: Known facts, unknown facets. Trends Endocrinol. Metab. 2007, 18, 142–149. [Google Scholar] [CrossRef] [PubMed]
- Claustrat, B.; Brun, J.; Chazot, G. The basic physiology and pathophysiology of melatonin. Sleep Med. Rev. 2005, 9, 11–24. [Google Scholar] [CrossRef] [PubMed]
- Srinivasan, V.; Pandi-Perumal, S.R.; Maestroni, G.J.M.; Esquifino, A.I.; Hardeland, R.; Cardinali, D.P. Role of melatonin in neurodegenerative diseases. Neurotoxicol. Res. 2005, 7, 293–318. [Google Scholar] [CrossRef]
- Waterhouse, J.; Reilly, T.; Atkinson, G. Jet lag. Lancet 1997, 350, 1611–1616. [Google Scholar] [CrossRef]
- Korkmaz, A.; Reiter, R.J.; Tan, D.X.; Manchester, L.C. Melatonin; from pineal gland to healthy foods. Spatula DD 2011, 1, 33–36. [Google Scholar] [CrossRef]
- Reiter, R.J.; Tan, D.X.; Burkhardt, S.; Manchester, L.C. Melatonin in plants. Nutr. Rev. 2001, 59, 286–290. [Google Scholar] [CrossRef] [PubMed]
- Reiter, R.J.; Tan, D.X.; Manchester, L.C.; Simopoulos, A.P.; Maldonado, M.D.; Flores, L.J.; Terron, M.P. Melatonin in edible plants (phytomelatonin): Identification, concentrations, bioavailability and proposed functions. World Rev. Nutr. Diet. 2007, 97, 211–230. [Google Scholar] [PubMed]
- Tan, D.X.; Reiter, R.J.; Manchester, L.C. Melatonin in walnuts: Influence on levels of melatonin and total antioxidant capacity of blood. Nutrition 2005, 21, 920–924. [Google Scholar]
- Gonzalez-Flores, D.; Gamero, E.; Garrido, M.; Ramirez, R.; Moreno, D.; Delgado, J.; Valdes, E.; Barriga, C.; Rodriguez, A.B.; Paredes, S.D. Urinary 6-sulfatoxymelatonin and total antioxidant capacity increase after the intake of a grape juice cv. Tempranillo stabilized with HHP. Food Funct. 2012, 3, 34–39. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Flores, D.; Velardo, B.; Garrido, M.; González-Gómez, D.; Lozano, M.; Ayuso, M.; Barriga, C.; Paredes, S.D.; Rodriguez, A.B. Ingestion of Japanese plums (Prunus salicina Lindl. cv. Crimson Globe) increases the urinary 6-sulfatoxymelatonin and total antioxidant capacity levels in young, middle-aged and elderly humans: Nutritional and functional characterization of their content. J. Food Nutr. Res. 2011, 50, 229. [Google Scholar]
- Garrido, M.; Paredes, S.D.; Cubero, J.; Lozano, M.; Toribio-Delgado, A.F.; Muñoz, J.L.; Reiter, R.J.; Barriga, C.; Rodríguez, A.B. Jerte valley cherry-enriched diets improve nocturnal rest and increase 6-sulfatoxymelatonin and total antioxidant capacity in the urine of middle-aged and elderly humans. J. Gerontol. A Biol. Sci. Med. Sci. 2010, 65, 909–914. [Google Scholar] [CrossRef] [PubMed]
- Garrido, M.; Espino, J.; González-Gómez, D.; Lozano, M.; Cubero, J.; Toribio-Delgado, A.F.; Maynar-Mariño, J.I.; Terrón, M.P.; Muñoz, J.L.; Pariente, J.A.; et al. A nutraceutical product based on Jerte Valley cherries improves sleep and augments the antioxidant status in humans. Eur. e-J. Clin. Nutr. Metab. 2009, 4, e321–e323. [Google Scholar] [CrossRef]
- Maldonado, M.D.; Moreno, H.; Calvo, J.R. Melatonin present in beer contributes to increase the levels of melatonin and antioxidant capacity of the human serum. Clin. Nutr. 2009, 28, 188–191. [Google Scholar] [CrossRef] [PubMed]
- Sae-Teaw, M.; Johns, J.; Johns, N.P.; Subongkot, S. Serum melatonin levels and antioxidant capacities after consumption of pineapple, orange, or banana by healthy male volunteers. J. Pineal Res. 2013, 55, 58–64. [Google Scholar] [CrossRef] [PubMed]
- Johns, N.P.; Johns, J.; Porasupthana, S.; Plaimee, P.; Sae-Teaw, M. Dietary intake of melatonin from tropical fruit altered urinary excretion of 6-sulfatoxymelatonin in healthy volunteers. J. Agric. Food Chem. 2013, 61, 913–919. [Google Scholar] [CrossRef] [PubMed]
- Tan, D.X.; Manchester, L.C.; Burkhardt, S.; Sainz, R.S.; Mayo, J.C.; Kohen, R.; Shohami, E.; Huo, Y.; Hardeland, R.; Reiter, R.J. AFMK, a biogenic amine and melatonin metabolite, functions as a potent antioxidant. FASEB J. 2001, 15, 2294–2296. [Google Scholar] [CrossRef] [PubMed]
- Gitto, E.; Tan, D.X.; Reiter, R.J.; Karbownik, M.; Manchester, L.; Cuzzocrea, S.; Fulia, F.; Barberi, I. Individual and synergistic antioxidative actions of melatonin: Studies with vitamin E, vitamin C, glutathione and desferoxamine in rat liver homogenates. J. Pharm. Pharmacol. 2001, 53, 1393–1401. [Google Scholar] [CrossRef] [PubMed]
- Galano, A.; Tan, D.; Reiter, R. Melatonin as a natural ally against oxidative stress: A physicochemical examination. J. Pineal Res. 2011, 51, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Morreale, M.; Livrea, M.A. Synergistic effect of glycolic acid on the antioxidant activity of a-tocopherol and melatonin in lipid bilayers and in human skin homogenates. Biochem. Mol. Biol. Int. 1997, 42, 1093–1102. [Google Scholar] [CrossRef] [PubMed]
- Benot, S.; Goberna, R.; Reiter, R.J.; Garcia-Mauriño, S.; Osuna, C.; Guerrero, J.M. Physiological levels of melatonin contribute to the antioxidant capacity of human serum. J. Pineal Res. 1999, 27, 59–64. [Google Scholar] [CrossRef] [PubMed]
- Blask, D.E.; Sauer, L.A.; Dauchy, R.T. Melatonin as a chronobiotic/anticancer agent: Cellular, biochemical and molecular mechanisms of action and their implications for circadian-based cancer therapy. Curr. Top. Med. Chem. 2002, 2, 113–132. [Google Scholar] [CrossRef] [PubMed]
- Reiter, R.J. Mechanisms of cancer inhibition by melatonin. J. Pineal Res. 2004, 37, 213–214. [Google Scholar] [CrossRef] [PubMed]
- Kamdar, B.B.; Tergas, A.; Mateen, F.J.; Bhayani, N.H.; Oh, J. Night-shift work and risk of breast cancer: A systematic review and meta-analysis. Breast Cancer Res. Treat. 2013, 138, 291–301. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez, C.; Martin, V.; Herrera, F.; García-Santos, G.; Rodríguez-Blanco, J.; Casado-Zapico, S.; Sanchez-Sanchez, A.M.; Suárez, S.; Puente-Moncada, N.; Anítua, M.; Antolín, I. Mechanisms involved in the pro-apoptotic effect of melatonin in cancer cells. Int. J. Mol. Sci. 2013, 14, 6597–6613. [Google Scholar] [CrossRef] [PubMed]
- Anisimov, V.; Popovich, I.; Zabezhinski, M.; Anisimov, S.; Vesnushkin, G.; Vinogradova, I. Melatonin as antioxidant, geroprotector and anticarcinogen. Biochim. Biophys. Acta 2006, 1757, 573–589. [Google Scholar] [CrossRef] [PubMed]
- Pandi-Perumal, S.R.; Trakht, I.; Srinivasan, V.; Spence, D.W.; Maestroni, G.J.M.; Zisapel, N.; Cardinali, D.P. Physiological effects of melatonin: Role of melatonin receptors and signal transduction pathways. Progr. Neurobiol. 2008, 85, 335–353. [Google Scholar] [CrossRef] [PubMed]
- Miller, S.C.; Pandi-Perumal, S.R.; Esquifino, A.I.; Cardinali, D.P.; Maestroni, G.J.M. The role of melatonin in inmmuno-enhancement: Potential application in cancer. Int. J. Exp. Pathol. 2006, 87, 81–87. [Google Scholar] [CrossRef] [PubMed]
- Jung-Hynes, B.; Reiter, R.J.; Ahmad, N. Sirtuins, melatonin and circadian rhythms: Building a bridge between aging and cancer. J. Pineal Res. 2010, 48, 9–19. [Google Scholar] [CrossRef] [PubMed]
- Venegas, C.; García, J.A.; Escames, G.; Ortiz, F.; López, A.; Doerrier, C.; García-Corzo, L.; López, L.C.; Reiter, R.J.; Acuña-Castroviejo, D. Extrapineal melatonin: Analysis of its subcellular distribution and daily fluctuations. J. Pineal Res. 2012, 52, 217–227. [Google Scholar] [CrossRef] [PubMed]
- Slominski, A.; Tobin, D.J.; Zmijewski, M.A.; Wortsman, J.; Paus, R. Melatonin in the skin: Synthesis, metabolism and functions. Trends Endocrinol. Metab. 2008, 19, 17–24. [Google Scholar] [CrossRef] [PubMed]
- Tan, D.X.; Manchester, L.C.; Reiter, R.J.; Qi, W.B.; Zhang, M.; Weintraub, S.T.; Cabrera, J.; Saínz, R.M.; Mayo, J.C. Identification of highly elevated levels of melatonin in bone marrow: Its origin and significance. Biochim. Biophys. Acta 1999, 1472, 206–214. [Google Scholar] [CrossRef]
- Raikhlin, N.T.; Kvetnoy, I.M.; Tolkachev, V.N. Melatonin may be synthesized in enterochromafinne cells. Nature 1975, 255, 344–345. [Google Scholar] [CrossRef] [PubMed]
- Bubenik, G. Gastrointestinal melatonin: Localization, function, and clinical revelance. Dig. Dis. Sci. 2002, 47, 2336–2348. [Google Scholar] [CrossRef] [PubMed]
- Huether, G. Melatonin synthesis in the gastrointestinal tract and the impact of nutritional factors on circulating melatonin. Ann. N. Y. Acad. Sci. 1994, 719, 146–158. [Google Scholar] [CrossRef] [PubMed]
- Konturek, S.J.; Konturek, P.C.; Brzozowski, T.; Bubenik, G.A. Role of melatonin in upper gastrointestinal tract. J. Physiol. Pharmacol. 2007, 58, 23–52. [Google Scholar] [PubMed]
- Hardeland, R.; Pandi-Perumal, S.R. Melatonin, a potent agent in antioxidative defense: Actions as a natural food constituent, gastrointestinal factor, drug and prodrug. Nutr. Metab. 2005, 2, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Oba, S.; Nakamura, K.; Sahashi, Y.; Hattori, A.; Nagata, C. Consumption of vegetables alters morning urinary 6-sulfatoxymelatonin concentration. J. Pineal Res. 2008, 45, 17–23. [Google Scholar] [CrossRef] [PubMed]
- Humbert, W.; Pévet, P. The decrease of pineal melatonin production with age. Ann. N. Y. Acad. Sci. 1994, 719, 43–61. [Google Scholar] [CrossRef] [PubMed]
- Poeggeler, B. Melatonin, aging, and age-related diseases: Perspectives for prevention, intervention, and therapy. Endocrine 2005, 27, 201–212. [Google Scholar] [CrossRef]
- Hardeland, R.; Madrid, J.A.; Tan, D.X.; Reiter, R.J. Melatonin, the circadian multioscillator system and health: The need for detailed analysis of peripheral melatonin signal. J. Pineal Res. 2012, 52, 139–166. [Google Scholar] [CrossRef] [PubMed]
- Dahlitz, M.; Alvarez, B.; Vignau, J.; English, J.; Arendt, J.; Parkes, J. Delayed sleep phase syndrome response to melatonin. Lancet 1991, 337, 1121–1124. [Google Scholar] [CrossRef]
- Dollins, A.; Zhdanova, I.; Wurtman, R.; Lynch, H.; Deng, M. Effect of inducing nocturnal serum melatonin concentrations in daytime on sleep, mood, body temperature, and performance. Proc. Natl. Acad. Sci. USA 1994, 91, 1824–1828. [Google Scholar] [CrossRef] [PubMed]
- Bonnefont-Rousselot, D.; Collin, F. Melatonin: Action as antioxidant and potential applications in human disease and aging. Toxicology 2010, 278, 55–67. [Google Scholar] [CrossRef] [PubMed]
- Seabra, M.; Bignotto, M.; Pinto, L., Jr.; Tufik, S. Randomized double blind clinical trial, controlled with placebo, of the toxicology of chronic melatonin treatment. J. Pineal Res. 2000, 29, 193–200. [Google Scholar] [CrossRef] [PubMed]
- Jan, J.; Hamilton, D.; Seward, N.; Fast, D.; Freeman, R.; Laudon, M. Clinical trial of controlled-release melatonin in children with sleep-wake disorders. J. Pineal Res. 2000, 29, 34–39. [Google Scholar] [CrossRef] [PubMed]
- Nordlund, J.J.; Lerner, A.B. The effects of oral melatonin on skin color and on the release of pituitary hormones. J. Clin. Endocrinol. Metab. 1977, 45, 768–774. [Google Scholar] [CrossRef] [PubMed]
- Hugel, H.M.; Kennaway, D.J. Synthesis and chemistry of melatonin and of related compounds. A review. Org. Prep. Proced. Int. 1995, 27, 1–31. [Google Scholar] [CrossRef]
- Prabhakar, C.; Vasanth-Kumar, N.; Ravikanth-Reddy, M.; Sarma, R.M.; Om-Reddy, G. Process research and development of melatonin. Org. Process Res. Dev. 1999, 3, 155–160. [Google Scholar] [CrossRef]
- Thomson, D.W.; Commeureuc, A.G.J.; Berlin, S.; Murphy, J.A. Efficient route to the pineal hormone melatonin by radical-based indole synthesis. Synth. Commun. 2003, 33, 3631–3641. [Google Scholar] [CrossRef]
- Lerner, A.B.; Case, J.D.; Takahashi, Y.; Lee, T.H.; Mori, W. Isolation of melatonin, a pineal factor that lightens melanocytes. J. Am. Chem. Soc. 1958, 80, 2587. [Google Scholar] [CrossRef]
- Lerner, A.; Case, J.; Heinzelmann, R. Structure of melatonin. J. Am. Chem. Soc. 1959, 81, 6084–6085. [Google Scholar] [CrossRef]
- Williamson, B.L.; Tomlinson, A.J.; Naylor, S.; Gleich, G.J. Contaminats in commercial preparations of melatonin. Mayo Clin. Proc. 1997, 72, 1094–1095. [Google Scholar] [CrossRef]
- Naylor, S.; Johnson, K.L.; Williamson, B.L.; Klarskov, K.; Gleich, G.J. Structural characterization of contaminants in commercial preparations of melatonin by on-line HPLC-electrospray ionization-tandem mass spectrometry. Adv. Exp. Med. Biol. 1999, 467, 769–777. [Google Scholar] [PubMed]
- Williamson, B.L.; Kenneth, L.J.; Tomlinson, A.J.; Gleich, G.J.; Naylor, S. On-line HPLC-tandem mass spectrometry structural characterization of case associated contaminants of l-tryptophan implicated with the onset of eosinophilia-myalgia syndrome. Toxicol. Lett. 1988, 99, 139–150. [Google Scholar] [CrossRef]
- Williamson, B.L.; Tomlinson, A.J.; Mishra, P.K.; Gleich, G.J.; Naylor, S. Structural characterization of contaminants found in commercial preparations of melatonin: Similarities to case-related compounds from l-tryptophan associated with Eosinophilia-Myalgia Syndrome. Chem. Res. Toxicol. 1998, 11, 234–240. [Google Scholar] [CrossRef] [PubMed]
- OECD-Organization for Economic Co-operation & Development. Screening Information DataSet (SIDS); Initial Assessment Report on Phthalimide; OECD-Organization for Economic Co-operation & Development: Paris, France, 2006. [Google Scholar]
- Reiter, R.J.; Mayo, J.C.; Tan, D.X.; Sainz, R.M.; Alatorre-Jimenez, M.; Qin, L. Melatonin as an antioxidant: Under promises but over delivers. J. Pineal Res. 2016, 61, 253–278. [Google Scholar] [CrossRef] [PubMed]
- Shahidi, F.; Ambigaipalan, P. Phenolics and polyphenolics in foods, beverages and spices: Antioxidant activity and health effects. A review. J. Funct. Foods 2015, 18, 820–897. [Google Scholar] [CrossRef]
- Pisoschi, A.M.; Pop, A. The role of antioxidants in the chemistry of oxidative stress: A review. Eur. J. Med. Chem. 2015, 97, 55–74. [Google Scholar] [CrossRef] [PubMed]
- Oroian, M.; Escriche, I. Antioxidants: Characterization, natural sources, extraction and analysis. Food Res. Int. 2015, 74, 10–36. [Google Scholar] [CrossRef] [PubMed]
- Kaulmann, A.; Bohn, T. Carotenoids, inflammation, and oxidative stress-implications of cellular signaling pathways and relation to chronic disease prevention. Nutr. Res. 2014, 34, 907–929. [Google Scholar] [CrossRef] [PubMed]
- Capello, C.; Fischer, U.; Hungerbühler, K. What is a green solvent? A comprehensive framework for the environmental assesment of solvents. Green Chem. 2007, 9, 927–934. [Google Scholar] [CrossRef]
- FDA (Food and Drug Administration) (Ed.) Drugs-FDA Glossary of Terms; Food and Drug Administration: Silver Spring, MD, USA, 2012; Volume 2016.
- Finley, J.W.; Finley, J.W.; Ellwood, K.; Hoadley, J. Launching a new food product or dietary supplement in the United States: Industrial, regulatory, and nutritional considerations. Annu. Rev. Nutr. 2014, 34, 421–447. [Google Scholar] [CrossRef] [PubMed]
- Coppens, P.; da Silva, M.F.; Pettman, S. European regulations on nutraceuticals, dietary supplements and functional foods: A framework based on safety. Toxicology 2006, 221, 59–74. [Google Scholar] [CrossRef] [PubMed]
- EFSA (European Food Safety Authority). Regulation (EC) Number 178/2002 of the European Parlament and of the Council laying down the general principles and requirements of food law, establishing the European Food Safety Authority and laying down procedures in matters of food safety. Off. J. Eur. Commun. 2002, L31, 1–24. [Google Scholar]
Sample Availability: Samples of the compounds are not available from the authors. |

| Common Name/Species | Phytomelatonin Content (ng·g−1 DW or FW) a | References |
|---|---|---|
| Coffee beans/Coffea sp. | 5800–6500 DW | [34] |
| Goji berry/Lycium barbarum L. | 530–103 DW | [35,36] |
| Kidney bean sprouts/Phaseolus vulgaris L. | 529 DW | [37] |
| White radish/Raphanus sativus L. | 485–0.6 DW | [20,35] |
| Jujube/Ziziphus jujube Lam. | 256 DW | [35] |
| White mustard/Brassica hirta L. | 189 DW | [36] |
| Apple/Malus domestica Borkh. | 134–0.4 FW | [20,38] |
| Black mustard/Brassica nigra L. | 129 DW | [36] |
| Sweet cherry/Prunus avium L. | 120–8.0 FW | [39,40] |
| Tomato/Solanum lycopersicum L. | 114–0.3 FW | [20,41] |
| Fenugreek/Trigonella foecum-graecum L. | 43 DW | [36] |
| Bellpepper/Capsicum annuum L. | 42–9.0 FW | [42] |
| Almond/Prunus amygdalus Batsch. | 39 DW | [36] |
| Sunflower/Helianthus annuus L. | 29 DW | [36] |
| Fennel/Foeniculum vulgare Mill. | 28 DW | [36] |
| Grape/Vitis vinifera L. | 18–1.2 FW | [43,44] |
| Alfalfa/Medicago sativa L. | 16 DW | [36] |
| Cardamom/Elettaria cardamomum L. | 15 DW | [36] |
| Strawberry/Fragaria ananassa Duch. | 11.2–1.4 FW | [41] |
| Common Name/Species | Phytomelatonin Content (ng·g−1 DW) | References |
|---|---|---|
| Thyme/Thymus vulgaris L. | 38,000 | [43] |
| Chinese liquorice/Glycyrrhiza uralensis Fisch. | 34,000 | [45] |
| Sage/Salvia officinalis L. | 29,000 | [43] |
| St. John’s wort/Hypericum perforatum L. | 23,000 | [46] |
| Peppermint/Mentha piperita | 19,500 | [35] |
| Cat’s claw herb/Uncaria rhynchophylla Miq. | 2460 | [35] |
| Tokyo violet/Viola philipica Cav. | 2360 | [35] |
| Feverfew/Tanacetum parthenium L. | 1700 | [47] |
| Mulberry/Morus alba L. | 1510 | [35] |
| Aloe/Aloe vera L. | 516 | [35] |
| Clove/Syzygium aromaticum L. | 446 | [35] |
| Yarrow/Achillea millefolium L. | 340 | [48] |
| Figwort/Scrophularia ningpoensis Hemsl. | 342 | [35] |
| Korean mint/Agastache rugosa Kuntz. | 300 | [35] |
| Qin Jiao/Gentiana macrophylla Pall. | 180 | [35] |
| Scullcap/Scutellaria amoena C.H. Wright | 178 | [35] |
| Japanese honeysuckle/Lonicea japonica Thunb | 140 | [35] |
| Curcuma/Curcuma aeruginosa Roxb. | 120 | [35] |
| Chemical Synthesis Route | Initial Structure (Relevant Precursor) | Reference |
|---|---|---|
| 1 | 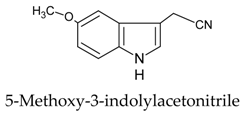 | [107] |
| 2 | 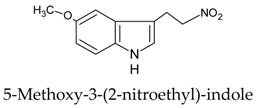 | [107] |
| 3 | 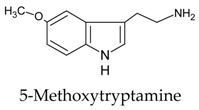 | [107,108] |
| 4 | 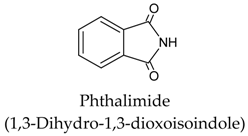 | [108,109] |
| Contaminant Compounds |
|---|
| 1,2,3,4-tetrahydro-β-carboline-3-carboxylic acid |
| 3-(phenylamino)alanine |
| 1,1′-ethylidenebis-(tryptophan) (so-called peak E) |
| 2-(3-indolylmethyl)-tryptophan |
| formaldehyde-melatonin |
| formaldehyde-melatonin condensation products |
| hydroxymelatonin isomers |
| 5-hydroxy-tryptamine derivatives |
| 5-methoxy-tryptamine derivatives |
| N-acetyl- and diacetyl-indole derivatives |
| 1,3-diphthalimidopropane |
| hydroxy-bromo-propylphthalimide |
| chloropropylphthalimide |
| Formulation | Trademark | Origin | Dosis | Form (units) | Brand, Country |
|---|---|---|---|---|---|
| #1 | HerbatoninPRO | Rice extract | 0.3 mg | Capsules (120) | Natural Health Int., USA |
| #2 | Sleep Support | Tart cherry skins Tart cherry juice | 15 µg 3 µg | Capsules (60) Sachets (liquid) | Tru2U, New Zealand |
| #3 | Melatonin 10 mg | Vegetarian | 10 mg | Sublingual tablets (180) | Bioclinic Naturals, Canada |
| #4 | Curaderm system | Diverse plants | - | Body Cream | Curapharm, USA |
| #5 | Fitomelatonina | Diverse plants | - | Cosmetic Cream | Effegilab, Italy |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
B. Arnao, M.; Hernández-Ruiz, J. The Potential of Phytomelatonin as a Nutraceutical. Molecules 2018, 23, 238. https://doi.org/10.3390/molecules23010238
B. Arnao M, Hernández-Ruiz J. The Potential of Phytomelatonin as a Nutraceutical. Molecules. 2018; 23(1):238. https://doi.org/10.3390/molecules23010238
Chicago/Turabian StyleB. Arnao, Marino, and Josefa Hernández-Ruiz. 2018. "The Potential of Phytomelatonin as a Nutraceutical" Molecules 23, no. 1: 238. https://doi.org/10.3390/molecules23010238
APA StyleB. Arnao, M., & Hernández-Ruiz, J. (2018). The Potential of Phytomelatonin as a Nutraceutical. Molecules, 23(1), 238. https://doi.org/10.3390/molecules23010238






