The Anticonvulsant Activity of a Flavonoid-Rich Extract from Orange Juice Involves both NMDA and GABA-Benzodiazepine Receptor Complexes
Abstract
:1. Introduction
2. Results
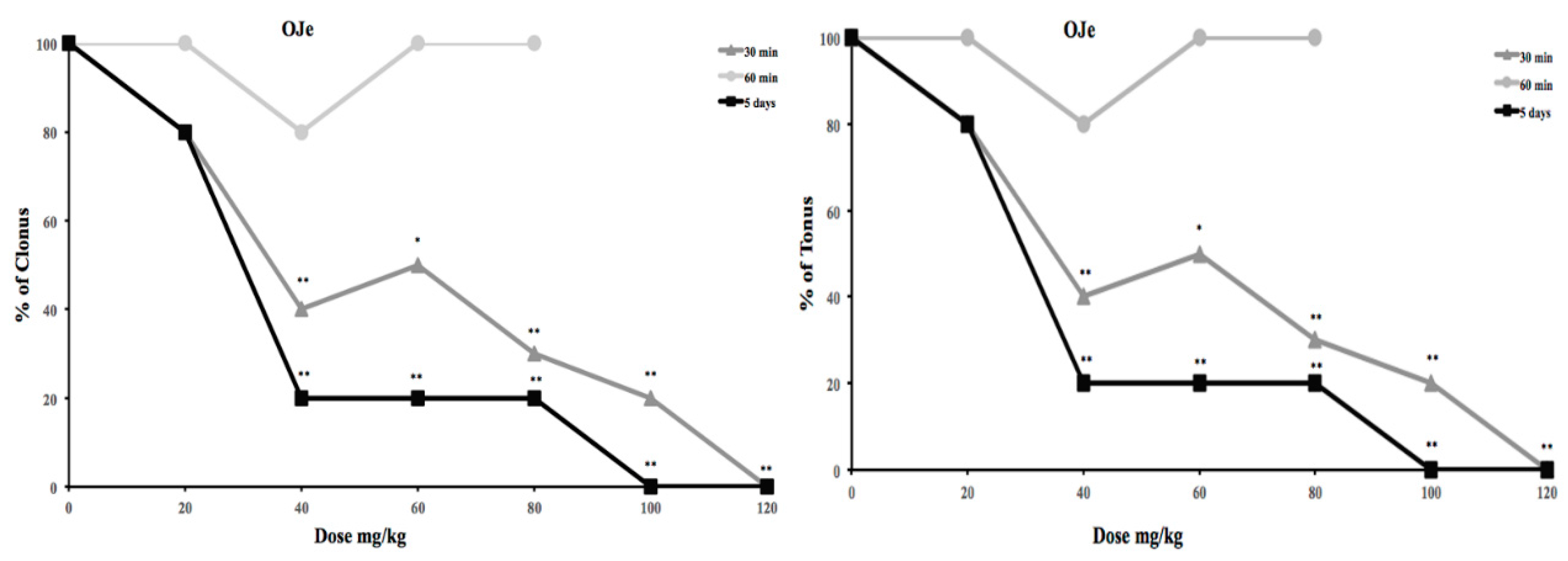
2.1. OJe HES Mitigates Pentylenetetrazole (PTZ)-Induced Seizures
2.2. Effects of OJe, HES or NRTN in Audiogenic Seizure Prone DBA/2 Mice
2.2.1. Interactions between NMDA Antagonists (CPPene, d-Cycloserine, Felbamate) and OJe against Audiogenic Seizures in DBA/2 Mice
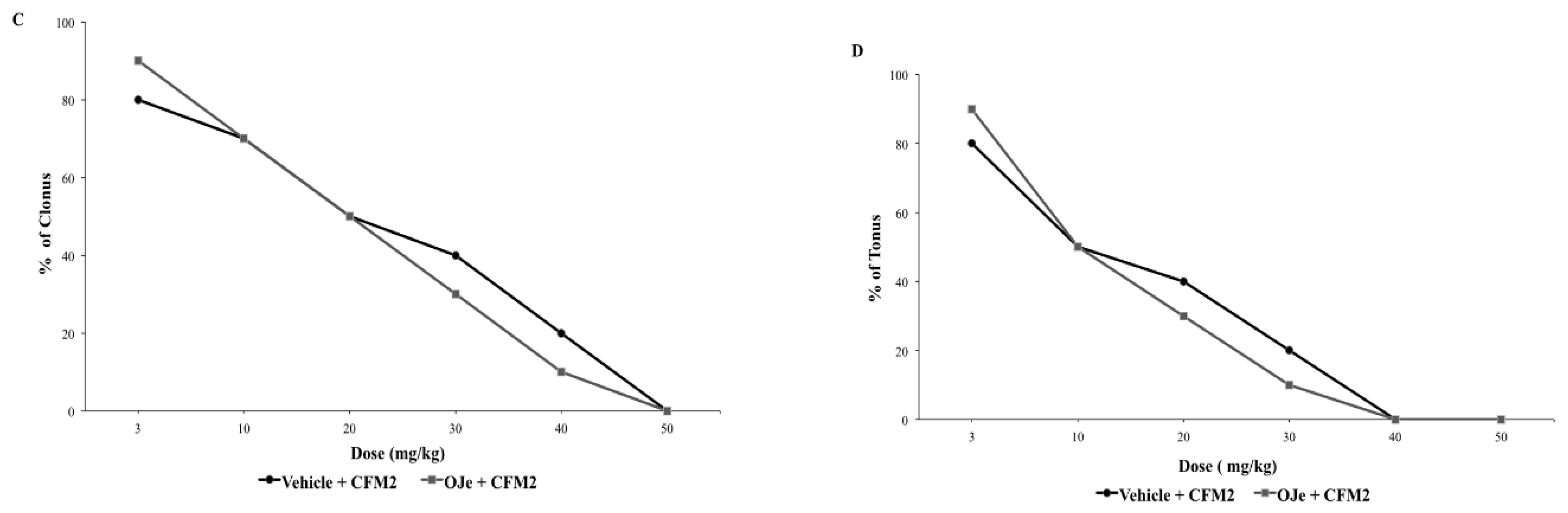
2.2.2. Interaction between NBQX and CFM-2, Two AMPA Receptor Antagonists and OJe
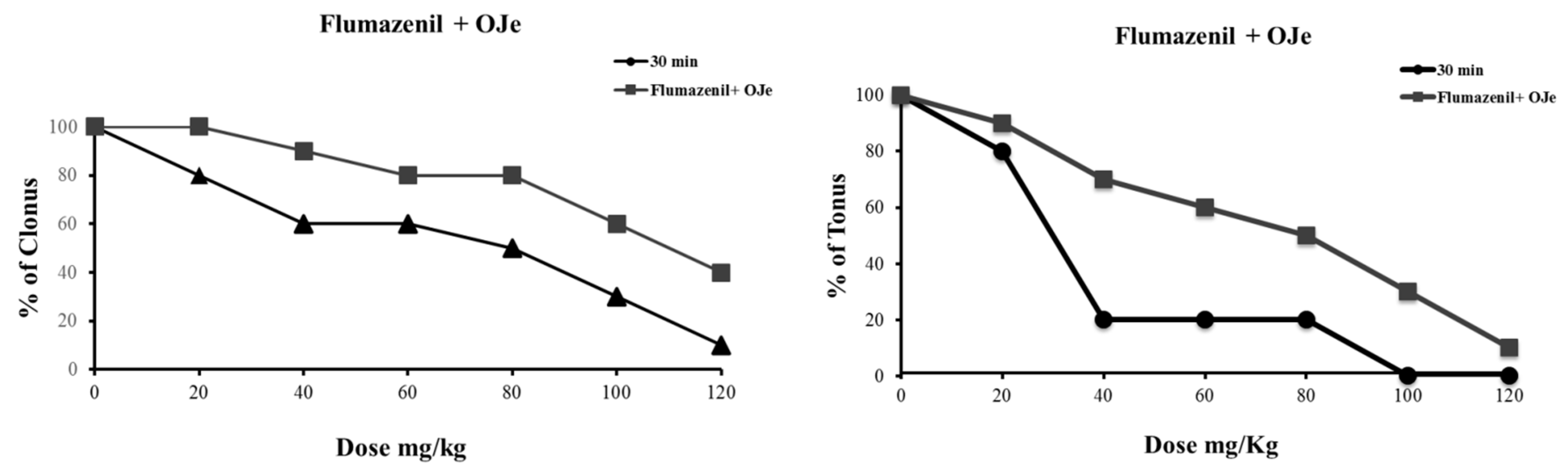
2.2.3. Treatment with Flumazenil
2.3. Effects of OJe on Absence Seizures in WAG/Rij Rats
3. Discussion
4. Materials and Methods
4.1. Animals
4.2. Drugs
4.3. Pentylenetetrazole (PTZ)-Induced Seizures in CD-1 Mice
4.3.1. Experiment 1 (Acute Treatment)
4.3.2. Experiment 2 (Subchronic Treatment)
4.4. Audiogenic Seizures in DBA/2 Mice
4.4.1. Administration of NMDA and AMPA Receptor Antagonists with OJe in DBA/2 Mice
4.4.2. Co-Administration of Flumazenil with OJe in DBA/2 Mice
4.5. Experiments in WAG/Rij Rats
4.6. Statistical Analysis
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Sierra-Paredes, G.; Sierra-Marcuno, G. Extrasynaptic GABA and glutamate receptors in epilepsy. CNS Neurol. Disorders Drug Targets 2007, 6, 288–300. [Google Scholar] [CrossRef]
- Costello, D.J.; Delanty, N. Oxidative injury in epilepsy: Potential for antioxidant therapy? Exp. Rev. Neurother. 2004, 4, 541–553. [Google Scholar] [CrossRef] [PubMed]
- Obay, B.D.; Tasdemir, E.; Tumer, C.; Bilgin, H.; Atmaca, M. Dose dependent effects of ghrelin on pentylenetetrazole-induced oxidative stress in a rat seizure model. Peptides 2008, 29, 448–455. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.; Xu, Q.; Zhang, L.; Kong, D.; Ma, R.; Wang, L. Protective effect of resveratrol against kainate-induced temporal lobe epilepsy in rats. Neurochem. Res. 2009, 34, 1393–1400. [Google Scholar] [CrossRef] [PubMed]
- Aguiar, C.C.; Almeida, A.B.; Araujo, P.V.; de Abreu, R.N.; Chaves, E.M.; do Vale, O.C.; Macedo, D.S.; Woods, D.J.; Fonteles, M.M.; Vasconcelos, S.M. Oxidative stress and epilepsy: Literature review. Oxid. Med. Cell. Longev. 2012, 2012, 795259. [Google Scholar] [CrossRef] [PubMed]
- Xu, K.; Stringer, J.L. Antioxidants and free radical scavengers do not consistently delay seizure onset in animal models of acute seizures. Epilepsy Behav. 2008, 13, 77–82. [Google Scholar] [CrossRef] [PubMed]
- Tome, A.R.; Feng, D.; Freitas, R.M. The effects of alpha-tocopherol on hippocampal oxidative stress prior to in pilocarpine-induced seizures. Neurochem. Res. 2010, 35, 580–587. [Google Scholar] [CrossRef] [PubMed]
- Hou, Y.; Aboukhatwa, M.A.; Lei, D.L.; Manaye, K.; Khan, I.; Luo, Y. Anti-depressant natural flavonols modulate bdnf and beta amyloid in neurons and hippocampus of double tgad mice. Neuropharmacology 2010, 58, 911–920. [Google Scholar] [CrossRef] [PubMed]
- Hanrahan, J.R.; Chebib, M.; Johnston, G.A. Flavonoid modulation of GABA(A) receptors. Br. J. Pharmacol. 2011, 163, 234–245. [Google Scholar] [CrossRef] [PubMed]
- Yao, L.H.; Jiang, Y.M.; Shi, J.; Tomas-Barberan, F.A.; Datta, N.; Singanusong, R.; Chen, S.S. Flavonoids in food and their health benefits. Plant Foods Hum. Nutr. 2004, 59, 113–122. [Google Scholar] [CrossRef] [PubMed]
- Benavente-Garcia, O.; Castillo, J. Update on uses and properties of Citrus flavonoids: New findings in anticancer, cardiovascular, and anti-inflammatory activity. J. Agric. Food Chem. 2008, 56, 6185–6205. [Google Scholar] [CrossRef] [PubMed]
- Grosso, G.; Galvano, F.; Mistretta, A.; Marventano, S.; Nolfo, F.; Calabrese, G.; Buscemi, S.; Drago, F.; Veronesi, U.; Scuderi, A. Red orange: Experimental models and epidemiological evidence of its benefits on human health. Oxida. Med. Cell. Longev. 2013, 2013, 157240. [Google Scholar] [CrossRef] [PubMed]
- Ferlazzo, N.; Visalli, G.; Smeriglio, A.; Cirmi, S.; Lombardo, G.E.; Campiglia, P.; Di Pietro, A.; Navarra, M. Flavonoid fraction of orange and bergamot juices protect human lung epithelial cells from hydrogen peroxide-induced oxidative stress. Evid. Based Compl. Alt. 2015, 2015, 957031. [Google Scholar] [CrossRef] [PubMed]
- Ferlazzo, N.; Visalli, G.; Cirmi, S.; Lombardo, G.E.; Lagana, P.; Di Pietro, A.; Navarra, M. Natural iron chelators: Protective role in a549 cells of flavonoids-rich extracts of citrus juices in Fe3+-induced oxidative stress. Environ. Toxicol. Pharmacol. 2016, 43, 248–256. [Google Scholar] [CrossRef] [PubMed]
- Celano, M.; Maggisano, V.; De Rose, R.F.; Bulotta, S.; Maiuolo, J.; Navarra, M.; Russo, D. Flavonoid fraction of Citrus reticulata juice reduces proliferation and migration of anaplastic thyroid carcinoma cells. Nutr. Cancer 2015, 67, 1183–1190. [Google Scholar] [CrossRef] [PubMed]
- Delle Monache, S.; Sanità, P.; Trapasso, E.; Ursino, M.R.; Dugo, P.; Russo, M.; Ferlazzo, N.; Calapai, G.; Angelucci, A.; Navarra, M. Mechanisms underlying the anti-tumoral effects of Citrus bergamia juice. PLoS ONE 2013, 8, e61484. [Google Scholar] [CrossRef] [PubMed]
- Ferlazzo, N.; Cirmi, S.; Russo, M.; Trapasso, E.; Ursino, M.R.; Lombardo, G.E.; Gangemi, S.; Calapai, G.; Navarra, M. NF-κB mediates the antiproliferative and proapoptotic effects of bergamot juice in HepG2 cells. Life Sci. 2016, 146, 81–91. [Google Scholar] [CrossRef] [PubMed]
- Navarra, M.; Ursino, M.R.; Ferlazzo, N.; Russo, M.; Schumacher, U.; Valentiner, U. Effect of Citrus bergamia juice on human neuroblastoma cells in vitro and in metastatic xenograft models. Fitoterapia 2014, 95, 83–92. [Google Scholar] [CrossRef] [PubMed]
- Visalli, G.; Ferlazzo, N.; Cirmi, S.; Campiglia, P.; Gangemi, S.; Di Pietro, A.; Calapai, G.; Navarra, M. Bergamot juice extract inhibits proliferation by inducing apoptosis in human colon cancer cells. Anti-Cancer Agents Med. Chem. 2014, 14, 1402–1413. [Google Scholar] [CrossRef]
- Risitano, R.; Currò, M.; Cirmi, S.; Ferlazzo, N.; Campiglia, P.; Caccamo, D.; Ientile, R.; Navarra, M. Flavonoid fraction of bergamot juice reduces LPS-induced inflammatory response through SIRT1-mediated NF-κB inhibition in THP-1 monocytes. PloS ONE 2014, 9, e107431. [Google Scholar] [CrossRef]
- Currò, M.; Risitano, R.; Ferlazzo, N.; Cirmi, S.; Gangemi, C.; Caccamo, D.; Ientile, R.; Navarra, M. Citrus bergamia juice extract attenuates β-amyloid-induced pro-inflammatory activation of THP-1 cells through MAPK and AP-1 pathways. Sci. Rep. 2016, 6, 20809. [Google Scholar] [CrossRef] [PubMed]
- Impellizzeri, D.; Bruschetta, G.; Di Paola, R.; Ahmad, A.; Campolo, M.; Cuzzocrea, S.; Esposito, E.; Navarra, M. The anti-inflammatory and antioxidant effects of bergamot juice extract (BJe) in an experimental model of inflammatory bowel disease. Clin. Nutr. 2015, 34, 1146–1154. [Google Scholar] [CrossRef] [PubMed]
- Impellizzeri, D.; Cordaro, M.; Campolo, M.; Gugliandolo, E.; Esposito, E.; Benedetto, F.; Cuzzocrea, S.; Navarra, M. Anti-inflammatory and antioxidant effects of flavonoid-rich fraction of bergamot juice (BJe) in a mouse model of intestinal ischemia/reperfusion injury. Front. Pharmacol. 2016, 7, 203. [Google Scholar] [CrossRef] [PubMed]
- Cirmi, S.; Bisignano, C.; Mandalari, G.; Navarra, M. Anti-infective potential of citrus bergamia risso et poiteau (bergamot) derivatives: A systematic review. Phytother. Res. 2016, 30, 1404–1411. [Google Scholar] [CrossRef] [PubMed]
- Marino, A.; Paterniti, I.; Cordaro, M.; Morabito, R.; Campolo, M.; Navarra, M.; Esposito, E.; Cuzzocrea, S. Role of natural antioxidants and potential use of bergamot in treating rheumatoid arthritis. Pharma. Nutr. 2015, 3, 53–59. [Google Scholar] [CrossRef]
- Kumar, A.; Lalitha, S.; Mishra, J. Possible nitric oxide mechanism in the protective effect of hesperidin against pentylenetetrazole (PTZ)-induced kindling and associated cognitive dysfunction in mice. Epilepsy Behav. 2013, 29, 103–111. [Google Scholar] [CrossRef] [PubMed]
- Meldrum, B.S.; Rogawski, M.A. Molecular targets for antiepileptic drug development. Neurotherapeutics 2007, 4, 18–61. [Google Scholar] [CrossRef] [PubMed]
- De Sarro, G.; Carotti, A.; Campagna, F.; McKernan, R.; Rizzo, M.; Falconi, U.; Palluotto, F.; Giusti, P.; Rettore, C.; De Sarro, A. Benzodiazepine receptor affinities, behavioral, and anticonvulsant activity of 2-aryl-2,5-dihydropyridazino[4,3-b]indol-3(3H)-ones in mice. Pharmacol. Biochem. Behav. 2000, 65, 475–487. [Google Scholar] [CrossRef]
- Dingledine, R. Glutamatergic Mechanisms Related to Epilepsy: Ionotropic Receptors. In Jasper’s Basic Mechanisms of the Epilepsies, 4th ed.; Noebels, J.L., Avoli, M., Rogawski, M.A., Olsen, R.W., Delgado-Escueta, A.V., Eds.; National Center for Biotechnology Information: Bethesda, MD, USA, 2012. [Google Scholar]
- Citraro, R.; Russo, E.; Gratteri, S.; Di Paola, E.D.; Ibbadu, G.F.; Curinga, C.; Gitto, R.; Chimirri, A.; Donato, G.; De Sarro, G. Effects of non-competitive AMPA receptor antagonists injected into some brain areas of WAG/Rij rats, an animal model of generalized absence epilepsy. Neuropharmacology 2006, 51, 1058–1067. [Google Scholar] [CrossRef] [PubMed]
- Russo, E.; Gitto, R.; Citraro, R.; Chimirri, A.; De Sarro, G. New AMPA antagonists in epilepsy. Exp. Opin. Investig. Drugs 2012, 21, 1371–1389. [Google Scholar] [CrossRef] [PubMed]
- De Sarro, G.; Chimirri, A.; Meldrum, B.S. Group III mGlu receptor agonists potentiate the anticonvulsant effect of AMPA and NMDA receptor block. Eur. J. Pharmacol. 2002, 451, 55–61. [Google Scholar] [CrossRef]
- De Sarro, G.; Gratteri, S.; Naccari, F.; Pasculli, M.P.; De Sarro, A. Influence of d-cycloserine on the anticonvulsant activity of some antiepileptic drugs against audiogenic seizures in DBA/2 mice. Epilepsy Res. 2000, 40, 109–121. [Google Scholar] [CrossRef]
- Ferreri, G.; Chimirri, A.; Russo, E.; Gitto, R.; Gareri, P.; De Sarro, A.; De Sarro, G. Comparative anticonvulsant activity of N-acetyl-1-aryl-6,7-dimethoxy-1,2,3,4-tetrahydroisoquinoline derivatives in rodents. Pharmacol. Biochem. Behav. 2004, 77, 85–94. [Google Scholar] [CrossRef] [PubMed]
- Watson, G.B.; Bolanowski, M.A.; Baganoff, M.P.; Deppeler, C.L.; Lanthorn, T.H. d-cycloserine acts as a partial agonist at the glycine modulatory site of the NMDA receptor expressed in xenopus oocytes. Brain Res. 1990, 510, 158–160. [Google Scholar] [CrossRef]
- Russo, E.; Scicchitano, F.; Citraro, R.; Aiello, R.; Camastra, C.; Mainardi, P.; Chimirri, S.; Perucca, E.; Donato, G.; De Sarro, G. Protective activity of α-lactoalbumin (ALAC), a whey protein rich in tryptophan, in rodent models of epileptogenesis. Neuroscience 2012, 226, 282–288. [Google Scholar] [CrossRef] [PubMed]
- De Sarro, G.; Ibbadu, G.F.; Marra, R.; Rotiroti, D.; Loiacono, A.; Donato Di Paola, E.; Russo, E. Seizure susceptibility to various convulsant stimuli in dystrophin-deficient mdx mice. Neurosci. Res. 2004, 50, 37–44. [Google Scholar] [CrossRef] [PubMed]
- Russo, E.; Donato di Paola, E.; Gareri, P.; Siniscalchi, A.; Labate, A.; Gallelli, L.; Citraro, R.; De Sarro, G. Pharmacodynamic potentiation of antiepileptic drugs’ effects by some hmg-coa reductase inhibitors against audiogenic seizures in DBA/2 mice. Pharmacol. Res. 2013, 70, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Citraro, R.; Russo, E.; Ngomba, R.T.; Nicoletti, F.; Scicchitano, F.; Whalley, B.J.; Calignano, A.; De Sarro, G. CB1 agonists, locally applied to the cortico-thalamic circuit of rats with genetic absence epilepsy, reduce epileptic manifestations. Epilepsy Res. 2013, 106, 74–82. [Google Scholar] [CrossRef] [PubMed]
- Citraro, R.; Chimirri, S.; Aiello, R.; Gallelli, L.; Trimboli, F.; Britti, D.; De Sarro, G.; Russo, E. Protective effects of some statins on epileptogenesis and depressive-like behavior in WAG/Rij rats, a genetic animal model of absence epilepsy. Epilepsia 2014, 55, 1284–1291. [Google Scholar] [CrossRef] [PubMed]
- Citraro, R.; Leo, A.; de Fazio, P.; De Sarro, G.; Russo, E. Antidepressants but not antipsychotics have antiepileptogenic effects with limited effects on comorbid depressive-like behavior in the wag/rij rat model of absence epilepsy. Br. J. Pharmacol. 2015, 172, 3177–3188. [Google Scholar] [CrossRef] [PubMed]
- Litchfield, J.T., Jr.; Wilcoxon, F. A simplified method of evaluating dose-effect experiments. J. Pharmacol. Exp. Ther. 1949, 96, 99–113. [Google Scholar] [PubMed]
- De Sarro, G.; Paola, E.D.; Gratteri, S.; Gareri, P.; Rispoli, V.; Siniscalchi, A.; Tripepi, G.; Gallelli, L.; Citraro, R.; Russo, E. Fosinopril and zofenopril, two angiotensin-converting enzyme (ACE) inhibitors, potentiate the anticonvulsant activity of antiepileptic drugs against audiogenic seizures in dba/2 mice. Pharmacol. Res. 2012, 65, 285–296. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Samples of the flavonoid-rich extract from orange juice used in this studycompounds are available from the authors at the Department of Chemical, Biological, Pharmaceutical and Environmental Sciences, University of Messina, Messina, Italy.



| Treatment (mg/kg; i.p.) | Time(min) | Seizure Phase (%) | Number of Mice | |||
|---|---|---|---|---|---|---|
| Clonus | Tonus | Death | ||||
| Vehicle | Saline | 30 | 100 | 100 | 75 | 8 |
| OJe | 20 | 30 | 100 | 87.5 | 75 | 8 |
| 40 | 30 | 75 | 37.5 ** | 37.5 ** | 8 | |
| 80 | 30 | 75 | 50 * | 75 | 8 | |
| 100 | 30 | 50 * | 25 ** | 25 ** | 8 | |
| 120 | 30 | 25 ** | 0 ** | 0 ** | 8 | |
| 40 | 60 | 100 | 75 | 75 | 8 | |
| 40 | 120 | 100 | 100 | 100 | 8 | |
| HES | 40 | 30 | 100 | 87.5 | 75 | 8 |
| 80 | 30 | 100 | 50 * | 50 * | 8 | |
| 100 | 30 | 62.5 | 25 ** | 25 ** | 8 | |
| 120 | 30 | 25 ** | 12.5 ** | 0 ** | 8 | |
| 40 | 60 | 100 | 100 | 87.5 | 8 | |
| 40 | 120 | 100 | 100 | 87.5 | 8 | |
| NRTN | 40 | 30 | 100 | 100 | 75 | 8 |
| 80 | 30 | 100 | 75 | 75 | 8 | |
| 100 | 30 | 87.5 | 50 * | 50 * | 8 | |
| 120 | 30 | 62.5 | 25 ** | 37.5 ** | 8 | |
| 40 | 60 | 100 | 100 | 100 | 8 | |
| 40 | 120 | 100 | 100 | 100 | 8 | |
| HES + NRTN | 120 + 120 | 30 | 37.5 ** | 12.5 ** | 12.5 ** | 8 |
| Treatment (mg/kg; os) | Time (Days) | Seizure Phase (%) | Number of Mice | |||
|---|---|---|---|---|---|---|
| Clonus | Tonus | Death | ||||
| Vehicle | Saline | 5 | 100 | 100 | 100 | 10 |
| OJe | 20 | 5 | 100 | 60 * | 40 ** | 10 |
| 40 | 5 | 80 | 20 ** | 20 ** | 10 | |
| HES | 40 | 5 | 100 | 75 | 50 * | 8 |
| 80 | 5 | 90 | 40 ** | 30 ** | 10 | |
| NRTN | 40 | 5 | 100 | 100 | 80 | 10 |
| 80 | 5 | 100 | 100 | 75 | 8 | |
| Treatment (mg/kg; i.p.) | Latency (s) | |||
|---|---|---|---|---|
| Clonus | Tonus | Death | ||
| Vehicle | Saline | 164 (152–177) | 681 (618–750) | 706 (586–851) |
| OJe | 20 | 174 (158–192) | 712 (584–868) | 776 (645–934) |
| 40 | 194 (176–214) | 852 (695–1044) * | 918 (746–1130) * | |
| 80 | 211 (192–230) | 868 (684–1102) * | 909 (684–1208) * | |
| HES | 40 | 176 (156–198) | 734 (592–910) | 804 (672–962) |
| 80 | 184 (160–211.6) | 845 (683–1110) * | 878 (724–1064.8) * | |
| NRTN | 40 | 168 (150–201) | 756 (564–900) | 801 (644–966) |
| 80 | 179 (155–206.7) | 784 (598–1027.8) | 834 (696–999.4) | |
| Treatment (mg/kg; 5 Days; os) | Latency (s) | |||
|---|---|---|---|---|
| Clonus | Tonus | Death | ||
| Vehicle | Saline | 158 (140–178) | 664 (598–737) | 712 (604–839) |
| OJe | 20 | 160 (140–180) | 670 (494–697) | 788 (602–946) |
| 40 | 224 (198–253) | 928 (715–1204) * | 998 (776–1284) * | |
| HES | 40 | 168 (150–201) | 756 (564–900) | 801 (644–966) |
| 80 | 218 (200–248) | 899 (709–1198) * | 995 (788–1226) * | |
| NRTN | 40 | 155 (146–198) | 744 (526–897) | 798 (625–978) |
| 80 | 196 (150–201) | 767 (602–916) | 802 (627–974) | |
| Treatment | Time (min) | ED50 Values | |
|---|---|---|---|
| Clonus | Tonus | ||
| OJe | 30 | 71.89 (56.75–91.08) ** | 36.34 (25.49–51.82) ** |
| HES | 30 | 112.05 (81.85–153.40) | 57.54 (47.33–69.95) |
| NRTN | 30 | >120 | 66.65 (48.49–91.22) |
| Treatment | ED50 Values | |||
|---|---|---|---|---|
| Clonus | Tonus | |||
| CPPene | plus | Saline | 1.76 (1.21–2.26) | 0.79 (0.44–1.43) |
| plus | OJe | 2.69 (2.30–3.15) ** | 2.61 (2.04–3.33) ** | |
| d-cycloserine | plus | Saline | 27.6 (17.7–43.2) | 14.4 (7.8–26.5) |
| plus | OJe | 58.7 (37.4–92.3) ** | 28.3 (20.1–39.8) ** | |
| Felbamate | plus | Saline | 48.8 (35.4–67.2) | 23.1 (12.1–44.0) |
| plus | OJe | 105.6 (64.9–171.7) ** | 65.9 (38.5–112.8) ** | |
| NBQX | plus | Saline | 4.9 (3.2–7.5) | 2.2 (1.4–3.6) |
| plus | OJe | 4.1 (2.6–6.49) | 2.8 (2.05–3.73) | |
| CFM–2 | plus | Saline | 10.04 (11.3–13.1) | 9.42 (7.34–12.09) |
| plus | OJe | 15.9 (11.3–22.46) * | 10.78 (7.59–15.30) | |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Citraro, R.; Navarra, M.; Leo, A.; Donato Di Paola, E.; Santangelo, E.; Lippiello, P.; Aiello, R.; Russo, E.; De Sarro, G. The Anticonvulsant Activity of a Flavonoid-Rich Extract from Orange Juice Involves both NMDA and GABA-Benzodiazepine Receptor Complexes. Molecules 2016, 21, 1261. https://doi.org/10.3390/molecules21091261
Citraro R, Navarra M, Leo A, Donato Di Paola E, Santangelo E, Lippiello P, Aiello R, Russo E, De Sarro G. The Anticonvulsant Activity of a Flavonoid-Rich Extract from Orange Juice Involves both NMDA and GABA-Benzodiazepine Receptor Complexes. Molecules. 2016; 21(9):1261. https://doi.org/10.3390/molecules21091261
Chicago/Turabian StyleCitraro, Rita, Michele Navarra, Antonio Leo, Eugenio Donato Di Paola, Ermenegildo Santangelo, Pellegrino Lippiello, Rossana Aiello, Emilio Russo, and Giovambattista De Sarro. 2016. "The Anticonvulsant Activity of a Flavonoid-Rich Extract from Orange Juice Involves both NMDA and GABA-Benzodiazepine Receptor Complexes" Molecules 21, no. 9: 1261. https://doi.org/10.3390/molecules21091261
APA StyleCitraro, R., Navarra, M., Leo, A., Donato Di Paola, E., Santangelo, E., Lippiello, P., Aiello, R., Russo, E., & De Sarro, G. (2016). The Anticonvulsant Activity of a Flavonoid-Rich Extract from Orange Juice Involves both NMDA and GABA-Benzodiazepine Receptor Complexes. Molecules, 21(9), 1261. https://doi.org/10.3390/molecules21091261