New Oral Formulation and in Vitro Evaluation of Docetaxel-Loaded Nanomicelles
Abstract
:1. Introduction
2. Results
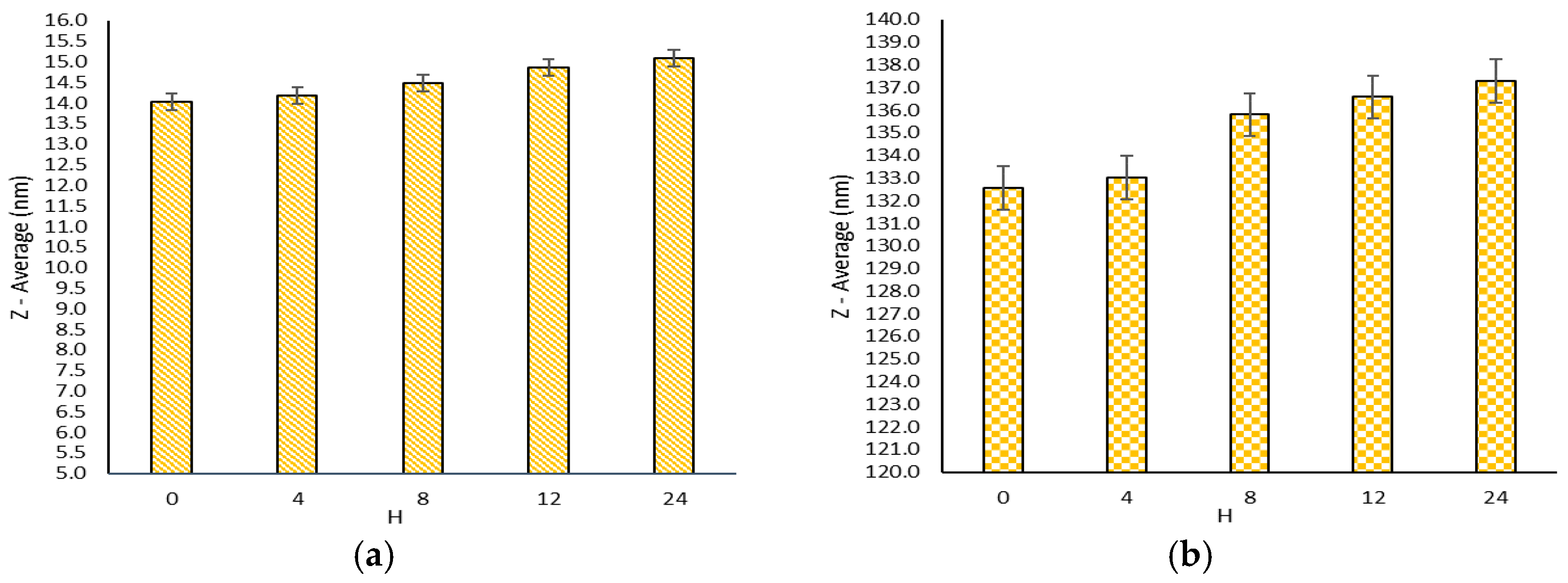
2.1. Nanomicelles Evaluation
2.2. Determination of Encapsulation Efficacy
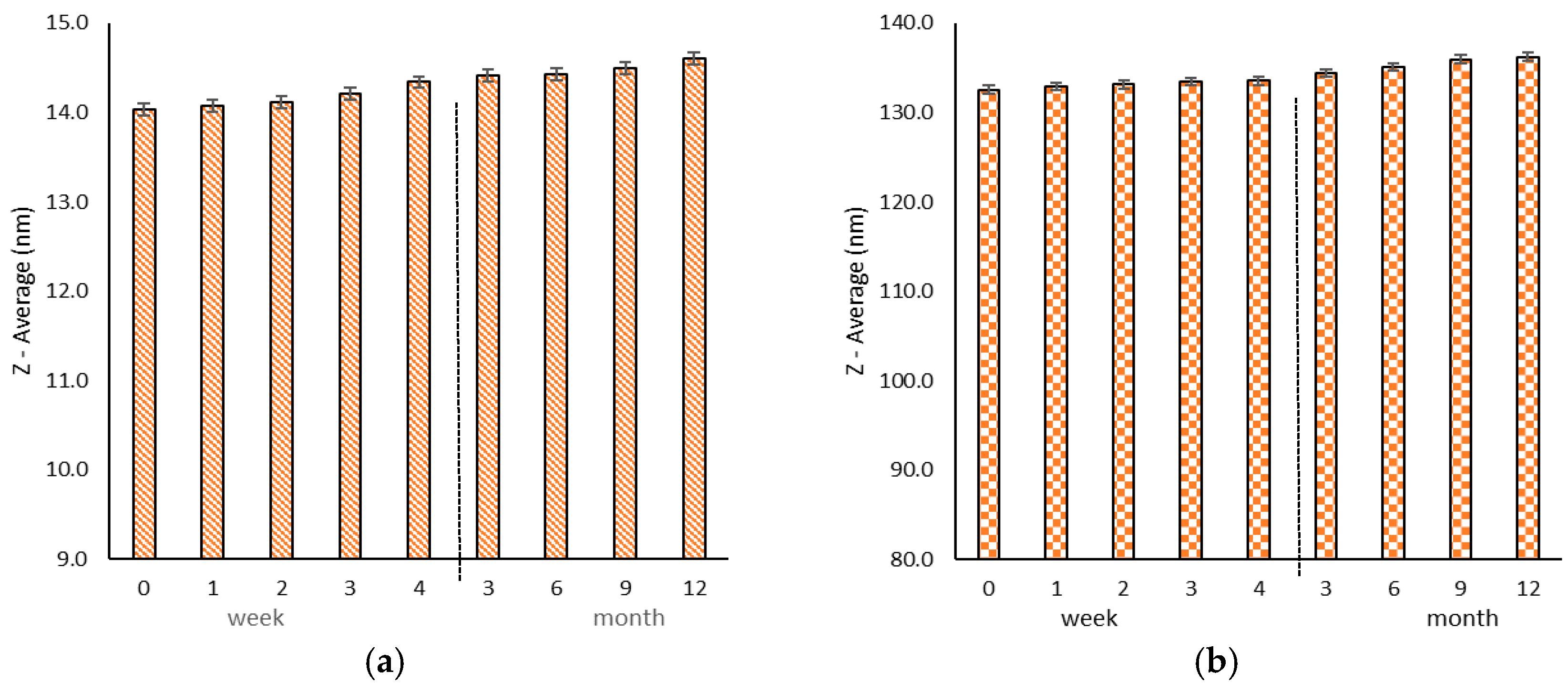
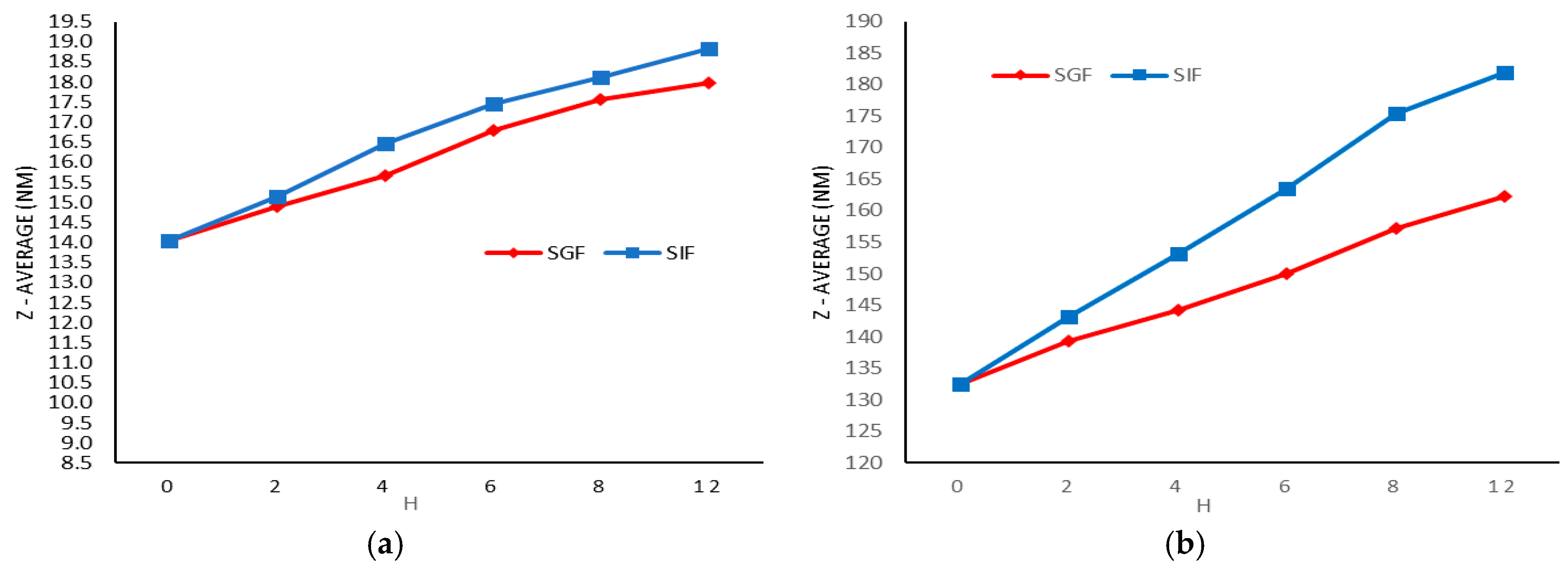
2.3. Stability Studies
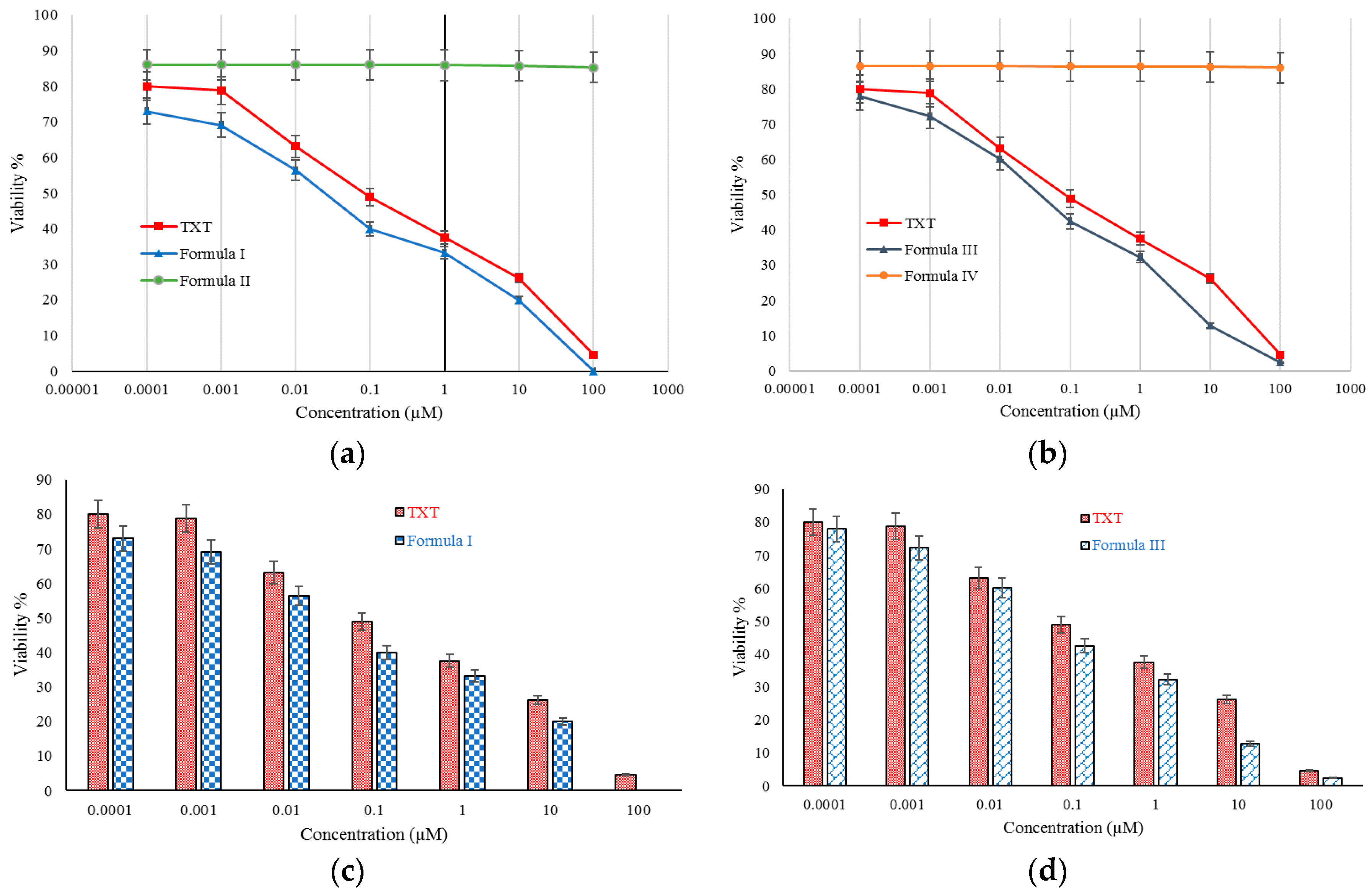
2.4. Cellular Toxicity Studies
2.5. IC50
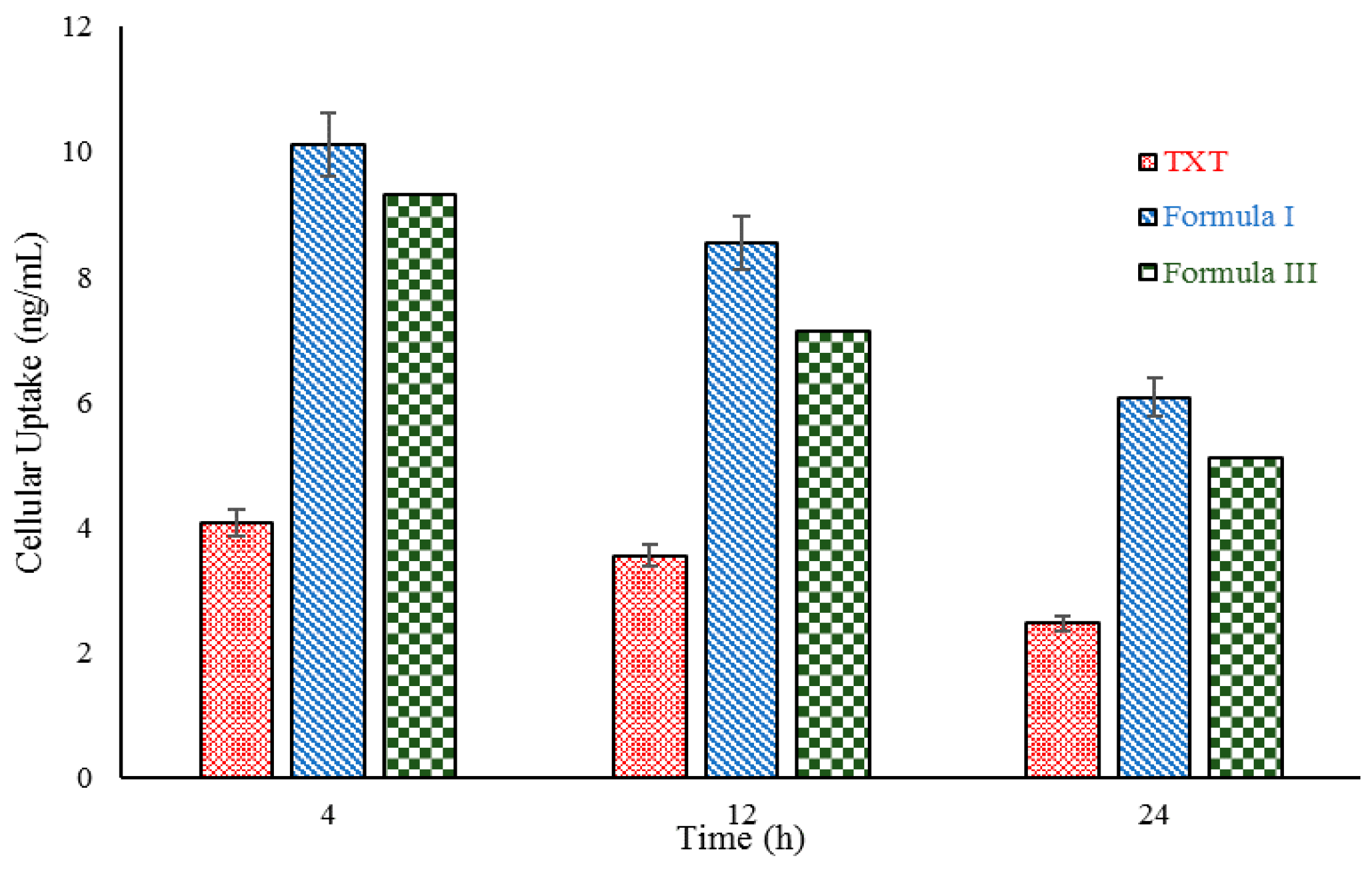
2.6. In Vitro Cell Uptake
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. DTX-Loaded Nanomicelles Preparation
4.3. Nanomicelles Characterization
4.4. Determination of Encapsulation Efficiency of DTX
4.5. Stability Studies
4.6. In Vitro Cells Toxicity Studies
4.7. Cellular Uptake
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- NIOSH List of Antineoplastic and Other Hazardous Drugs in Healthcare Settings: Proposed Additions to the NIOSH Hazardous Drug List 2016. CDC. Available online: http://www.cdc.gov/niosh/docket/review/docket233a/default.html (accessed on 20 September 2016).
- Zhao, P.; Astruc, D. Docetaxel Nanotechnology in Anticancer Therapy. ChemMedChem 2012, 7, 952–972. [Google Scholar] [CrossRef] [PubMed]
- Mosallaei, N.; Jaafari, M.R.; Hanafi, M.Y.; Golmohammadzadeh, S.H.; Malaekeh-Nikouei, B. Docetaxel-loaded solid lipid nanoparticles: Preparation, characterization, in vitro, and in vivo evaluations. J. Pharm. Sci. 2013, 102, 1994–2004. [Google Scholar] [CrossRef] [PubMed]
- Song, H.; Geng, H.; Ruan, J.; Wang, K.; Bao, C.; Wang, J.; Peng, X.; Zhang, X.; Cui, D. Development of Polysorbate 80/Phospholipid mixed micellar formation for docetaxel and assessment of its in vivo distribution in animal models. Nanoscale Res. Lett. 2011, 6, 354. [Google Scholar] [CrossRef] [PubMed]
- Cuppone, F.; Bria, E.; Carlini, P.; Milella, M.; Felici, A.; Sperduti, I.; Nistico, C.; Terzoli, E.; Cognetti, F.; Giannarelli, D. Taxanes as primary chemotherapy for early breast cancer: Meta-analysis of randomized trials. Cancer 2008, 113, 238–246. [Google Scholar] [CrossRef] [PubMed]
- Chin, D.L.; Lum, B.L.; Sikic, B.I. Rapid determination of PEGylated liposomal doxorubicin and its major metabolite in human plasma by ultraviolet-visible high-performance liquid chromatography. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2002, 779, 259–269. [Google Scholar] [CrossRef]
- Yanasarn, N.; Sloat, B.R.; Cui, Z. Nanoparticles engineered from lecithin-in water emulsions as a potential delivery system for docetaxel. Int. J. Pharm 2009, 379, 174–180. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Zhao, Y.; Geng, L.; Li, X.; Wang, X.; Liu, Z.; Wang, D.; Bi, K.; Chen, X. Pharmacokinetics and tissue distribution of docetaxel by liquid chromatography-mass spectrometry: Evaluation of folate receptor-targeting amphiphilic copolymer modified nanostructured lipid carrier. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2011, 879, 3721–3727. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.W.; Ramasamy, T.; Choi, J.Y.; Kim, J.H.; Yong, C.S.; Kim, J.O.; Choi, H.G. The influence of bile salt on the chemotherapeutic response of docetaxel-loaded thermosensitive nanomicelles. Int. J. Nanomed. 2014, 9, 3815–3824. [Google Scholar] [CrossRef] [PubMed]
- Dou, J.; Zhang, H.; Liu, X.; Zhang, M.; Zhai, G. Preparation and evaluation in vitro and in vivo of docetaxel loaded mixed micelles for oral administration. Colloids Surf. B Biointerfaces 2014, 114, 20–27. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Wang, J.; Zhang, X.; Lu, W.; Zhang, Q. A novel mixed micelle gel with thermo-sensitive property for the local delivery of docetaxel. J. Control. Release 2009, 135, 175–182. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Chen, L.; Gu, W.; Gao, Y.; Lin, L.; Zhang, Z.; Xi, Y.; Li, Y. The performance of docetaxel-loaded solid lipid nanoparticles targeted to hepatocellular carcinoma. Biomaterials 2009, 30, 226–232. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.; Liu, Z.; Wang, L.; Zhang, C.; Zhang, N. Nanostructured lipid carriers as novel carrier for parenteral delivery of docetaxel. Colloids Surf. B Biointerfaces 2011, 85, 262–269. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Liu, Z.; Liu, D.; Liu, C.; Juan, Z.; Zhang, N. Docetaxel loaded-lipid-based-nanosuspensions (DTX-LNS): Preparation, pharmacokinetics, tissue distribution and antitumor activity. Int. J. Pharm. 2011, 413, 194–201. [Google Scholar] [CrossRef] [PubMed]
- Yin, Y.M.; Cui, F.D.; Mu, C.F.; Choi, M.K.; Kim, J.S.; Chung, S.J.; Shim, C.K.; Kim, D.D. Docetaxel micro emulsion for enhanced oral bioavailability: Preparation and in vitro and in vivo evaluation. J. Control. Release 2009, 140, 86–94. [Google Scholar] [CrossRef] [PubMed]
- Leonhard, V.; Alasino, R.V.; Bianco, I.D.; Garro, A.G.; Heredia, V.; Beltramo, D.M. Self-assembled micelles of monosialogangliosides as nanodelivery vehicles for taxanes. J. Controlled Release 2012, 162, 619–627. [Google Scholar] [CrossRef] [PubMed]
- Baker, J.; Ajani, J.; Scotte, F.; Winther, D.; Martin, M.; Aapro, M.S.; von Minckwitz, G. Docetaxel-related side effects and their management. Eur. J. Oncol. Nurs. 2008, 12, 253–268. [Google Scholar] [CrossRef] [PubMed]
- Winther, D.; Saunte, D.M.; Knap, M.; Haahr, V.; Jensen, A.B. Nail changes due to docetaxel—A neglected side effect and nuisance for the patient. Support. Care Cancer 2007, 15, 1191–1197. [Google Scholar] [CrossRef] [PubMed]
- Taymouri, S.; Hassanzadeh, F.; Javanmard, S.; Mahzouni, P.; Varshosaz, J. Pharmacokinetics, Organ Toxicity and Antitumor Activity of Docetaxel Loaded in Folate Targeted Cholesterol Based Micelles. Curr. Drug Deliv. 2016, 13, 545–556. [Google Scholar] [CrossRef] [PubMed]
- Lee, E.; Kim, H.; Lee, I.H.; Jon, S. In vivo antitumor effects of chitosan-conjugated docetaxel after oral administration. J. Control. Release 2009, 140, 79–85. [Google Scholar] [CrossRef] [PubMed]
- Moesa, J.J.; Koolena, S.L.; Huitemaa, A.D.; Schellens, J.H.; Beijnen, J.H.; Nuijena, B. Pharmaceutical development and preliminary clinical testing of an oral solid dispersion formulation of docetaxel. Int. J. Pharm 2011, 420, 244–250. [Google Scholar] [CrossRef] [PubMed]
- Qiao, H.; Li, J.; Wang, Y.; Ping, Q.; Wang, G.; Gu, X. Synthesis and characterization of multi-functional linear-dendritic block copolymer for intracellular delivery of antitumor drugs. Int. J. Pharm. 2013, 452, 363–373. [Google Scholar] [CrossRef] [PubMed]
- Sissung, T.M.; Baum, C.E.; Deeken, J.; Price, D.K.; Aragon-Ching, J.; Steinberg, S.M.; Dahut, W.; Sparreboom, A.; Figg, W.D. ABCB1 genetic variation influences the toxicity and clinical outcome of patients with androgen-independent prostate cancer treated with docetaxel. Clin. Cancer Res. 2008, 14, 4543–4549. [Google Scholar] [CrossRef] [PubMed]
- Wils, P.; Phung-Ba, V.; Warner, A.; Lechardeur, D.; Raeissi, S.; Hidalgo, I.J.; Scherman, D. Polarized transport of docetaxel and vinblastine mediated by P-glycoprotein in human intestinal epithelial cell monolayers. Biochem. Pharmacol. 1994, 48, 1528–1530. [Google Scholar] [CrossRef]
- Hu, K.; Cao, S.; Hu, F.; Feng, J. Enhanced oral bioavailability of docetaxel by lecithin nanoparticles: Preparation, in vitro, andin vivo evaluation. Int. J. Nanomed. 2012, 7, 3537–3545. [Google Scholar] [CrossRef] [PubMed]
- Dressman, J.B.; Reppas, C. Oral Drug Absorption Prediction and Assessment: Prediction and Assesment, 2nd ed.; CRC Press; Taylor & Francis Group: Boca Raton, FL, USA, 2010; Volume 193, pp. 169–200. [Google Scholar]
- Smithson, K.W.; Millar, D.B.; Jacobs, L.R.; Gray, G.M. Intestinal diffusion barrier: Unstirred water layer or membrane surface mucous coat. Science 1981, 214, 1241–1244. [Google Scholar] [CrossRef] [PubMed]
- Kaunitz, J.D.; Akiba, Y. Luminal acid elicits a protective duodenal mucosal response. Keio J. Med. 2002, 51, 29–35. [Google Scholar] [CrossRef] [PubMed]
- Cha, E.-J.; Kim, J.E.; Ahn, C. pH dependent drug release system using micelles stabilized by cationic drugs. Macromol. Res. 2010, 7, 686–689. [Google Scholar] [CrossRef]
- Anton, N.; Mojzisova, H.; Procher, E.; Benoit, J.P.; Saulnier, P. Reverse micelle-loaded lipid nano-emulsions: New technology for nano encapsulation of hydrophilic materials. Int. J. Pharm. 2010, 398, 204–209. [Google Scholar] [CrossRef] [PubMed]
- Kedar, U.; Phutane, P.; Shidhaye, S.; Kadam, V. Advances in polymeric micelles for drug delivery and tumor targeting. Nanomedicine 2010, 6, 714–729. [Google Scholar] [CrossRef] [PubMed]
- He, C.; Zhang, L.; Wang, H.; Zhang, F.; Mo, X. Physical-chemical properties and in vitro biocompatibility assessment of spider silk, collagen and polyurethane nanofiber scaffolds for vascular tissue engineering. Nano Biomed. Eng. 2009, 1, 80. [Google Scholar] [CrossRef]
- Dintaman, J.M.; Silverman, J.A. Inhibition of P-glycoprotein by d-alpha-tocopheryl polyethylene glycol 1000 succinate (TPGS). Pharm. Res. 1999, 16, 1550–1556. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.K.; Lim, S.J.; Kim, C.K. Preparation characterization and in vitro cytotoxicity of paclitaxel-loaded sterically stabilized solid lipid nanoparticles. Biomaterials 2007, 28, 2137–2146. [Google Scholar] [CrossRef] [PubMed]
- Cavalli, R.; Caputo, O.; Gasco, M.R. Preparation and characterization of solid lipid nanospheres containing paclitaxel. Eur. J. Pharm Sci. 2000, 10, 305–309. [Google Scholar] [CrossRef]
- Khalid, M.N.; Simard, P.; Hoarau, D.; Dragomir, A.; Leroux, J.C. Long circulating poly (ethylene glycol)-decorated lipid nanocapsules deliver docetaxel to solid tumors. Pharm Res. 2006, 23, 752–758. [Google Scholar] [CrossRef] [PubMed]
- Allen, C.; Mavsinger, D.; Eisenberg, A. Disablism in Housing and Comparative Community Care Discourse—Towards an Interventionist Model of Disability and Interventionist Welfare Regime Theory. Colloid Surf. B 1999, 16, 3–16. [Google Scholar] [CrossRef]
- Bunjes, H. Characterization of solid lipid nano- and microparticles. In Lipospheres in Drug Targets and Delivery; Nastruzzi, C., Ed.; CRC Press: Boca Raton, FL, USA, 2005; pp. 41–66. [Google Scholar]
- Nagayama, S.; Ogawara, K.-I.; Fukuoka, Y.; Higaki, K.; Kimura, T. Time-dependent changes in opsonin amount associated on nanoparticles alter their hepatic uptake characteristics. Int. J. Pharm. 2007, 342, 215–221. [Google Scholar] [CrossRef] [PubMed]
- Pridgen, E.M.; Alexis, F.; Farokhzad, O.C. Polymeric nanoparticle drug delivery technologies for oral delivery applications. Expert Opin. Drug Deliv. 2015, 12, 1459–1473. [Google Scholar] [CrossRef] [PubMed]
- Papahadjopoulos, D.; Kirpotin, D.B.; Park, J.W.; Hong, K.; Shao, Y.; Shalaby, R.; Colbern, G.; Benz, C.C. Targeting of drugs to solid tumors using anti-HER2 immunoliposomes. J. Liposome Res. 1998, 8, 425–442. [Google Scholar] [CrossRef]
- Muller, R.H.; Mader, K.; Gohla, S. Solid lipid nanoparticles (SLN) for controlled drug delivery—A review of the state of the art. Eur. J. Pharmacol. Biopharm. 2000, 50, 161–177. [Google Scholar] [CrossRef]
- Petersen, S.; Steiniger, F.; Fischer, D.; Fahr, A.; Bunjes, H. The physical state of lipid nanoparticles influences their effect on in vitro cell viability. Eur. J. Pharm. Biopharm. 2011, 79, 150–161. [Google Scholar] [CrossRef] [PubMed]
- Yan, F.; Zhang, C.; Zheng, Y.; Mei, L.; Tang, L.; Song, C.; Sun, H.; Huang, L. The effect of Poloxamer 188 on nanoparticles morphology, size, cancer cell uptake and cytotoxicity. Nanomedicine 2010, 6, 170–178. [Google Scholar] [CrossRef] [PubMed]
- Mu, L.; Teo, M.M.; Ning, H.Z.; Tan, C.S.; Feng, S.S. Novel powder formulations for controlled delivery of poorly soluble anticancer drug: Application and investigation of TPGS and PEG in spray-dried particulate system. J. Control. Release 2005, 103, 565–575. [Google Scholar] [CrossRef] [PubMed]
- Dahmani, F.Z.; Yang, H.; Zhou, J.; Yao, J.; Zhang, T.; Zhang, Q. Enhanced oral bioavailability of paclitaxel in pluronic/LHR mixed polymeric micelles: Preparation, in vitro and in vivo evaluation. Eur. J. Pharmcol. Sci. 2012, 47, 179–189. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Not available.


| Formulation | Content |
|---|---|
| I | DTX, Tween 80, MCT oil, water |
| II | Tween 80, MCT oil, water |
| III | DTX, Tween 20, MCT oil, water |
| IV | Tween 20, MCT oil, water |
| Formulation | Z-Average (nm) | PDI | Zeta Potential (mV) |
|---|---|---|---|
| I | 14.03 ± 1.23 | 0.132 ± 0.02 | −9.45 |
| II | 9.89 ± 2.68 | 0.125 ± 0.07 | −5.67 |
| III | 132.55 ± 12.88 | 0.256 ± 0.02 | −6.09 |
| IV | 126.2 ± 19.39 | 0.224 ± 0.06 | −2.97 |
| Condition | Time | EE % |
|---|---|---|
| A (at room temperature) | After 24 h | 99 ± 0.05 |
| B (at 2–8 °C) | After 12 months | 98.9 ± 0.1 |
| Formulation | IC50 (µM) |
|---|---|
| I | 0.044853 |
| III | 0.061314 |
| TXT | 0.093071 |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hekmat, A.; Attar, H.; Seyf Kordi, A.A.; Iman, M.; Jaafari, M.R. New Oral Formulation and in Vitro Evaluation of Docetaxel-Loaded Nanomicelles. Molecules 2016, 21, 1265. https://doi.org/10.3390/molecules21091265
Hekmat A, Attar H, Seyf Kordi AA, Iman M, Jaafari MR. New Oral Formulation and in Vitro Evaluation of Docetaxel-Loaded Nanomicelles. Molecules. 2016; 21(9):1265. https://doi.org/10.3390/molecules21091265
Chicago/Turabian StyleHekmat, Atefeh, Hossein Attar, Ali Akbar Seyf Kordi, Maryam Iman, and Mahmoud Reza Jaafari. 2016. "New Oral Formulation and in Vitro Evaluation of Docetaxel-Loaded Nanomicelles" Molecules 21, no. 9: 1265. https://doi.org/10.3390/molecules21091265
APA StyleHekmat, A., Attar, H., Seyf Kordi, A. A., Iman, M., & Jaafari, M. R. (2016). New Oral Formulation and in Vitro Evaluation of Docetaxel-Loaded Nanomicelles. Molecules, 21(9), 1265. https://doi.org/10.3390/molecules21091265






