Abstract
The intramolecular aminomercuration of γ-alkenylamines 1a, 1b and 4 was shown to afford the 5-endo-trig cyclized product exclusively in good yield. The utility of pyrrolidine derivatives thus obtained from D-glucose derived γ-alkenylamines 1a and 1b was demonstrated in the synthesis of 1-deoxycastanospermine (3a) and 1-deoxy-8a-epi-castanospermine (3b).
Introduction
The pyrrolidine ring skeleton constitutes an important molecular backbone for a number of heterocyclic compounds. Among the strategies available for the synthesis of pyrrolidine ring skeletons one of the most attractive is the stereoselective intramolecular amido- and/or amino-mercuration reaction of δ−alkenylamines [1]. The reaction affords 2-substituted pyrrolidine rings through preferred 5-exo-trig cyclization, in addition to the 6-endo-trig cyclized piperidine ring skeleton in minor amounts [2]. In case of γ-alkenylamines, however, only a single report is known on the mercury (II) mediated 5-endo-trig cyclization, leading to the formation of pyrrolidine ring, while the 4-exo-trig product is not obtained due to the ring strain involved in the four membered ring system [1d, 3]. Recently, we have reported the utility of D-glucose derived γ-alkenylamine 1 for the synthesis of the polyhydroxylated quinolizidine homocastanospermine alkaloids [4].
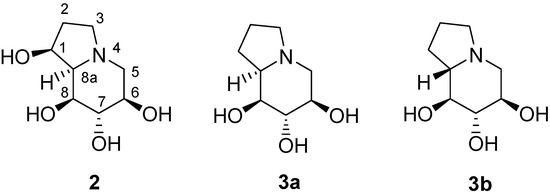
Figure 1.
This class of compounds, in particular the polyhydroxylated indolizidine alkaloid castanospermine (2), has attracted considerable attention because of their promising glycosidase inhibitory activity. In the pursuit of structure-activity relationships a number of natural and unnatural derivatives of castanospermine [5] have been synthesized and evaluated for glycosidase inhibition in the treatment of various diseases such as diabetes [6], cancer [7], and viral infections, including AIDS [8]. As a part of our continuing interest in the synthesis of azasugars [9], we thought of utilizing the intramolecular aminomercuration strategy, in a 5-endo-trig fashion, with the D-glucose derived γ-alkenylamine 1 for the construction of a pyrrolidine ring skeleton that could be elaborated to 1-deoxy castanospermine (3a) and its 8a-epi analogue 3b (Figure 1) [10]. Our results with the successful mercury (II) mediated 5-endo-trig cyclization of γ-alkenylamines towards the pyrrolidine ring skeleton are discussed.
Results and Discussion
In order to know whether the differentially substituted γ-alkenylamines obey the usual Baldwin’s ring closure rule or not [10], we have investigated the model reaction with α-phenyl-substituted-γ-alkenylamine 4. The required compound 4 was obtained by 1,3-addition of allylmagnesium bromide to the benzaldehyde-derived nitrone 5 [11], followed by reductive cleavage of the N−O bond with zinc-acetic acid (Scheme 1) [4]. The reaction of γ-alkenylamine 4 with mercury (II) acetate in (1:1) THF-water at 25 °C for 18 h, followed by reductive demercuration with sodium borohydride afforded the 5-endo-trig product 6 exclusively in 81% yield.
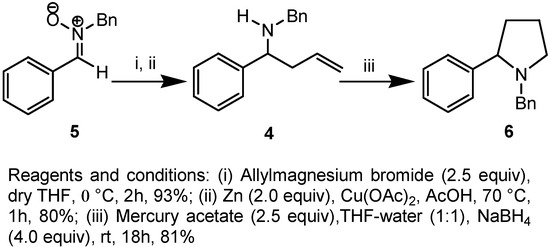
Scheme 1.
With the success in 5-endo-trig aminomercuration of γ-alkenylamine 4 in hand, we have exploited the same strategy with the D-glucose derived γ-alkenylamine 1. As reported earlier, the reaction of D-glucose nitrone 7 with allylmagnesium bromide in the presence of TMSOTf (1 equiv) at –78 °C in dry THF, afforded a diastereomeric mixture of D-gluco- and L-ido- configurated N-benzylhydroxylamines 8a and 8b in the ratio of 86:14 (Scheme 2). The treatment of 8a and 8b, individually, with zinc-acetic acid afforded the D-gluco-γ-alkenylamine 1a and L-ido-γ-alkenylamine 1b in good yield [4]. In the next step, the aminomercuration of 1a with mercury (II) acetate in THF-water (1:1) at room temperature for 18 h, followed by reductive demercuration with sodium borohydride afforded exclusively the 5-endo-trig cyclized product 9a in 78% yield. Similarly, the aminomercuration of 1b afforded the pyrrolidine derivative 9b in 76% yield. The spectroscopic and analytical data obtained for 9a and 9b were in agreement with the assigned structures.
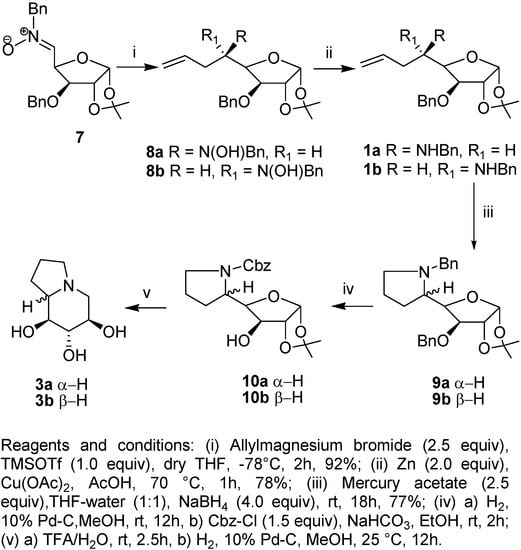
Scheme 2.
In the subsequent step, removal of N− and O−benzyl groups in 9a by hydrogenation using 10% Pd-C, followed by selective N−protection with benzyl chloroformate afforded 10a in 74% yield [12]. Deprotection of the 1,2-acetonide group in 10a with TFA-water, followed by hydrogenation afforded 1-deoxy-castanospermine (3a). The reaction sequence was repeated for L-ido-configurated-pyrrolidine derivative 9b and the corresponding C5-epimeric compound 10b was obtained in good yield. Deprotection of the 1,2-acetonide group of 10b with TFA-water, followed by hydrogenation afforded 1-deoxy-8a-epi-castanospermine (3b). The physical and the analytical data as well as the optical rotation for 3a and 3b were found to be consistent with those reported in the literature [9f].
Conclusions
We have demonstrated that the intramolecular aminomercuration of γ-alkenylamines, derived from aromatic as well as from sugar substrates, results in exclusive 5-endo-trig cyclisation leading to pyrrolidine ring skeletons. The utility of 5-endo-trig cyclized pyrrolidine derivatives 9a and 9b was demonstrated in the synthesis of 1-deoxy-castanospermine analogues 3a and 3b, respectively.
Experimental
General
Melting points were recorded with a Thomas-Hoover melting point apparatus and are uncorrected. IR spectra were recorded with a FTIR-8400, Shimadzu spectrophotometer as a thin film or in nujol mulls and are expressed in cm−1. 1H-NMR (300 MHz) and 13C-NMR (75 MHz) Mercury YH 300, Varian, spectra were recorded in CDCl3 as a solvent unless otherwise noted. NMR Chemical shifts are reported in δ ppm) downfield from TMS. Elemental analyses were carried out with Hosli elemental analyzer. Optical rotations were measured with JASCO DIP 181 polarimeter using sodium light (D line 589.3 nm) at 25 °C. TLC was performed on pre-coated plates (0.25 mm, silica gel 60 F254). Column chromatography was carried out with 100-200-mesh silica gel. The reactions were carried out in oven-dried glassware under dry N2. Allylmagnesium bromide was prepared prior to use from Mg and allyl bromide in dry ether. N-Benzylhydroxylamine hydrochloride, Cbz-Cl, and mercury acetate was purchased from Aldrich and/or Fluka. Methanol (MeOH), diethyl ether (Et2O), dichloromethane (CH2Cl2) and THF were purified and dried before use. Petroleum ether (PE) refers to a distillation fraction collected between 40-60 °C. After work up, organic layers were washed with water and brine, dried over anhydrous sodium sulfate and evaporated under reduced pressure.
Preparation of N-benzyl-N-(1-phenylbut-3-enyl)amine (4).
Allylmagnesium bromide (1M in Et2O, 1.4 mL, 6.52 mmol) was added dropwise to a stirred solution of nitrone 5 (0.725 g, 3.43 mmol) in THF at 0 °C. The mixture was stirred at 0 °C for 2h, then quenched with a saturated solution of NH4Cl (5 mL) and filtered. The filtrate was concentrated and the residue was dissolved in Et2O (30 mL). The ethereal layer was washed with brine, dried with sodium sulphate and evaporated to give oil, which was used directly in the next step. Zinc dust (0.257 g, 4.50 mmol) was added to a solution of copper (II) acetate (0.015 g) in glacial acetic acid (1 mL) under nitrogen and the mixture was stirred at room temperature for 15 min until the color disappeared. The crude oil (0.30 g, 0.75 mmol) in glacial acetic acid (0.7 mL) and water (0.3 mL) was added, the reaction mixture was heated at 70 °C for 1h and then cooled to room temperature. The sodium salt of EDTA (0.1 g) was added and the mixture was stirred for 10 min and then made alkaline to pH 10 by addition of 3N NaOH. The resulting solution was extracted with CHCl3 (10 mL x 3) the combined organic layer was washed with brine dried over Na2SO4 and evaporated to give oil. Purification by column chromatography gave 4 (0.21 g, 70%) as a thick liquid; Rf = 0.51 (EtOAc/hexane = 3:7); IR νmax (neat) = 3475, 1634 cm−1; 1H-NMR: δ 1.66-1.82 (1H, bs, exchanges with D2O), 2.32-2.51 (2H, m, H-8), 3.53 (1H, d, J = 13.2 Hz, N-CH2Ph), 3.62-3.76 (2H, m, H-7, N-CH2Ph), 4.98-5.12 (2H, m, H-9), 5.62-5.79 (1H, m, H-10), 7.18-7.42 (10H, m, Ar-H); 13C-NMR: δ 42.6, 51.1,61.5, 117.7, 126.9, 127.2, 127.3, 128.3, 128.4, 134.9; Anal. Calcd. for C17H19N: C, 86.03; H, 8.07; Found C, 86.23; H, 8.19.
Preparation of N-benzyl-2-phenyl pyrrolidine (6).
Compound 4 (0.200 g, 0.84 mmol) in THF (5 mL) was added to a stirred solution of mercury acetate (0.538 g, 1.68 mmol) in THF-water (1:1, 15 mL). The reaction mixture was stirred at rt for 18 h, quenched by sodium borohydride (0.124 g, 3.36 mmol) and then extracted into CHCl3 (10 mL x 3). The CHCl3 layer was dried and evaporated to afford a thick liquid that was purified by column chromatography to afford 6 (0.164 g, 81%) as a thick liquid; Rf = 0.55 (EtOAc/Hexane = 3:7); 1H-NMR: δ 1.70-1.98 (3H, m, H-8, H-9), 2.16-2.30 (2H, m, H-9, H-10), 3.06 (1H, d, J = 12.9 Hz, N-CH2Ph), 3.08-3.18 (1H, m, H-10), 3.97 (1H, t, J = 8.1 Hz, H-7), 3.88 (1H, d, J = 12.9 Hz, N-CH2Ph), 7.19-7.59 (10H, m, Ar-H); 13C-NMR: δ 22.4, 35.3, 53.3, 58.1, 69.6, 126.6, 126.7, 126.8, 127.1, 127.4, 127.6, 127.9, 128.1, 128.3, 128.5, 139.5, 143.6; Anal. Calcd. for C17H19N: C, 86.03; H, 8.07; Found C, 86.31; H, 8.29.
Preparation of 5,6,7,8-tetradeoxy-5,8-(N-benzylimino)-1,2-O-isopropylidene-α-D–gluco-oct-1,4- furanose (9a).
To a stirred solution of mercury acetate (0.827 g, 2.59 mmol) in THF-water (1:1, 10 mL) was added compound 1a (0.530 g, 1.29 mmol) in THF (5 mL). The reaction mixture was stirred at rt for 18 h, quenched by sodium borohydride (0.196 g, 5.18 mmol) and then extracted into CHCl3 (10mL x 3). The CHCl3 layer was dried and evaporated to afford a thick liquid that was purified by column chromatography to afford 9a (0.278 g, 78%) as a thick liquid; Rf = 0.43 (EtOAc/hexane = 3:7); [α]D = −72.8 (1.0 c, CHCl3); IR νmax (neat) = 2927, 1604 cm−1; 1H-NMR: δ 1.32 (3H, s, −CH3), 1.51 (3H, s, -CH3), 1.68-1.79 (2H, m), 1.90-2.01 (2H, m), 2.20-2.319 (1H, m), 2.85-2.89 (1H, m), 3.24 (1H, dt, J = 8.1, 4.2 Hz), 3.44 (1H, d, J = 13.2 Hz), 3.99-4.10 (3H, m), 4.40 (1H, d, J = 12.0 Hz), 4.62 (1H, d, J = 12.0 Hz), 4.63 (1H, d, J = 3.9 Hz), 5.89 (1H, d, J = 3.9 Hz), 7.18-7.38 (10H, m); 13C-NMR: δ 24.1, 26.3, 26.5, 28.6, 54.5, 60.1, 61.1, 71.2, 81.6, 82.1, 83.1, 104.5, 111.2, 126.5, 127.3, 127.6, 128.0, 128.2, 128.3, 137.2; Anal. Calcd. for C25H31NO4: C, 73.32; H, 7.63 Found C, 73.11; H, 7.45.
Preparation of 5,6,7,8-tetradeoxy-5,8-(N-benzylimino)-1,2-O-isopropylidene-β-L-ido-oct-1,4 furanose (9b).
The reaction of 1b (0.255 g, 0.62 mmol) with mercury acetate (0.397 g, 1.24 mmol) and sodium borohydride (0.092 g, 2.49 mmol) under the same reaction conditions reported for 9a, gave 9b (0.168 g, 72%) as a thick liquid; Rf = 0.40 (EtOAc/hexane = 3:7); [α]D = −61.3 (c 0.7, CHCl3); IR νmax (neat) = 2927, 1604 cm−1; 1H-NMR: δ 1.36 (3H, s), 1.54 (3H, s), 1.59-1.79 (3H, m), 1.80-1.93 (1H, m), 2.18- 2.27 (1H, m), 2.88-3.02 (2H, m), 3.32 (1H, d, J = 13 Hz), 3.88 (1H, d, J = 2.5 Hz), 4.23 (1H, bd, J = 7.0 Hz), 4.46 (1H, d, J = 11.5 Hz), 4.54 (2H, m), 4.68 (1H, d, J = 11.5 Hz), 6.01 (1H, d, J = 4.0 Hz), 7.20-7.39 (10H, m).
Preparation of 5,6,7,8-tetradeoxy-5,8-(N-benzoxycarbonylimino)-1,2-O-isopropylidene-α-D-gluco-oct-1,4-furanose (10a).
10% Pd-C (0.100 g) was added to a solution of compound 9a (0.278 g, 0.67 mmol) in MeOH (5 mL) and the solution was hydrogenated at 80 psi for 16 h. The catalyst was filtered and washed with methanol and the filtrate concentrated to give a sticky solid that was dissolved in EtOH/H2O (2 mL, 1:1) and sodium bicarbonate (0.17 g, 1.97 mmol) and then benzyloxycarbonyl chloride (0.10 g, 0.59 mmol) was added at 0 °C. The mixture was stirred at 25 °C for 2 h. Usual workup followed by extraction with CHCl3 (5 mL x 3) afford a thick liquid that was purified by column chromatography (PE/EtOAc = 92:8) to give 10a (0.13 g, 74%) as a thick liquid; Rf = 5.5 (hexane/EtOAc = 6:4); [α]D = +29.00 (c 0.56, CHCl3) [reported +28.30 (c, 1.1, CHCl3)]; IR νmax (neat) = 3366, 1675 cm−1; 1H-NMR: δ 1.31 (3H, s, CH3), 1.49 (3H, s, CH3), 1.84-2.05 (3H, m), 2.09-2.15 (1H, m), 3.38-3.45 (2H, m), 3.80 (1H, dd, J = 10.2, 1.8 Hz), 4.02 (1H, d, J = 1.8 Hz), 4.14 (1H, dd, J = 10.2, 6.2 Hz), 5.26 (1H, bs, exchanges with D2O), 4.59 (1H, d, J = 3.7 Hz), 5.14 (2H, ABq, J = 12.5 Hz), 5.90 (1H, d, J = 3.7Hz), 7.26-7.38 (5H, m); 13C-NMR: δ 23.0, 26.1, 27.1, 28.2, 48.8, 59.9, 68.1, 73.9, 81.0, 85.0, 105.2, 112.3, 128.0, 128.4, 129.1, 136.0, 157.1; Anal. Calcd. for C19H25NO6: C, 62.79; H, 6.93. Found: C, 62.68; H, 6.81.
Preparation of 5,6,7,8-Tetradeoxy-5,8-(N-benzoxycarbonylimino)-1,2-O-isopropylidene-β-L-ido-oct-1,4-furanose (10b).
The reaction of 9b (0.20 g, 0.82 mmol) with 10% Pd-C (0.06 g, 4.1 mmol) in methanol followed by reaction with benzyloxycarbonyl chloride (0.16 g, 0.94 mmol) under the same reaction conditions reported for 10a gave 10b (0.22 g, 74%) as a thick liquid; Rf = 0.42 (hexane/EtOAc = 6:4); [α]D = −72.27 (c 0.44, CHCl3); IR νmax (neat) = 3390, 1693 cm−1; 1H-NMR: δ 1.30 (3H, s), 1.48 (3H, s), 1.82-1.94 (2H, m), 1.96-2.20 (2H, m), 3.42-3.58 (2H, m), 4.0 (1H, t, J = 3.0 Hz), 4.13 (1H, d, J = 2.9 Hz), 4.26-4.34 (1H, m), 4.48 (1H, d, J = 3.6 Hz), 5.13 (2H, ABq, J = 12.3 Hz), 5.87 (1H, d, J = 3.6 Hz), 7.24-7.42 (5H, m); 13C-NMR: δ 23.6, 26.1, 26.7, 29.7, 47.2, 55.5, 67.5, 75.7, 84.0, 85.0, 104.5, 111.2, 127.8, 128.0, 128.4, 136.3, 158.7; Anal. Calcd. for C19H25NO6: C, 62.79; H, 6.93. Found: C, 62.80; H, 6.79.
Preparation of (6S,7R,8R,8aR)-6,7,8-Trihydroxy-indolizidine (3a).
A solution of 10a (0.10 g, 0.28 mmol) in 3:2 TFA/H2O (2 mL) was stirred at 25 °C for 2 h. The TFA was co-evaporated with benzene to furnish a thick liquid, which was directly used in the next reaction. To a solution of the product in MeOH (5 mL) was added 10% Pd/C (0.01 g), and the solution was hydrogenated at 80 psi for 16 h. The catalyst was filtered and washed with methanol and the filtrate concentrated to give a sticky solid which was purified by column chromatography (CHCl3/MeOH = 9:1) to give 3a (0.04 g, 90%) as a white solid; mp 176-178 °C (reported [3f] 178-181 °C); Rf = 0.45 (CHCl3/MeOH = 9/1); [α]D = +50.10 (c 0.7, MeOH) [reported +50.6 (c 0.2, MeOH)]; IR (KBr pellet) = 3380, 3270 cm−1 (broad band); 1H-NMR (pyridine-d5 + D2O) δ 1.39-1.58 (1H, m), 1.64-1.82 (2H, m), 1.93-2.06 (1H, m), 2.10-2.24 (2H, m), 2.30 (1H, t, J = 10.5 Hz), 2.94 (1H, dt, J = 8.6, 2.0 Hz), 3.43 (1H, dd, J = 5.2, 10.5 Hz), 3.86 (1H, t, J = 8.6 Hz), 3.91 (1H, t, J = 8.6 Hz), 4.33 (1H, ddd, J = 10.5, 8.6, 5.2 Hz); 13C-NMR (pyridine-d5) δ 21.9, 28.6, 53.2, 56.9, 68.3, 71.5, 75.9, 80.9; Anal. Calcd. for C8H15NO3: C, 55.47; H, 8.73. Found: C, 55.77; H, 8.98.
Preparation of (6S,7R,8R,8aS)-6,7,8-Trihydroxy-indolizidine (3b).
The reaction of 10b (0.16 g, 0.44 mmol) with TFA/H2O (2 mL, 3:2) under the same conditions reported for 3a gave the corresponding hemiacetal as a thick liquid, which on hydrogenation with 10% Pd/C (0.02 g) in methanol gave 3b as a semisolid. Column chromatography using 8:2 CHCl3/MeOH as an eluent gave (0.065 g, 87%) as a semisolid; Rf = 0.2 (CHCl3/MeOH = 1/1); [α]D = +23.0 (c 0.72, MeOH) [reported [3f] +22.5 (c 1.13, MeOH)]; IR (KBr pellet) = 3360-3250 (broad band) cm−1; 1H-NMR (pyridine-d5 + D2O) δ 1.72-2.00 (3H, m), 2.22-2.40 (1H, m), 2.84-3.00 (1H, m), 3.44 (1H, bd, J = 12.3 Hz), 3.49-3.67 (2H, m), 3.69 (1H, bd, J = 12.3 Hz), 4.42 (2H, bs), 4.55 (1H, bs); 13C-NMR (pyridine-d5) δ 21.1, 24.5, 54.7, 55.5, 64.6, 70.5, 70.7, 70.9; Anal. Calcd for C8H15NO3: C, 55.47; H, 8.73. Found: C, 55.80; H, 9.02.
Acknowledgements
We are thankful to CSIR, New Delhi for the SRF to SMJ. We are grateful to UGC, New Delhi for the financial support to procure the high field NMR (300 MHz) facility.
References and Notes
- Gasc, M. B. Tetrahedron 2002, 58, 705–711.Cardillo, G.; Orena, M. Tetrahedron 2002, 58, 3386–3395.Gasc, M. B.; Lattes, A.; Perie, J. J. Tetrahedron 1983, 39, 703.Perrie, J. J.; Laval, J. P.; Roussel, J.; Lattes, A. Tetrahedron 1971, 28, 675.
- Tokuda, M.; Yasufumi, Y.; Suginome, H. Chem. Lett. 1988, 1280–1290.Fraga, A. M.; Pencnha, E. P.; Verli, H. Tetrahedron Lett. 2002, 43, 1607–1611.Schumacher-W, M. H. G.; Peterson, S.; Peter, M. G. Liebigs Ann. Chem. 1994, 555–561.Le Moigne, F.; Tordo, P. J. Org. Chem. 1994, 59, 3365.Arnone, A.; Bravo, P.; Donadelli, A.; Resnati, G. J. Chem. Soc., Chem. Commun. 1993, 984.Stipa, P.; Finet, J. P.; Le Moigne, F.; Tordo, P. J. Org. Chem. 1993, 58, 4465.Chikkanna, D.; Han, H. Synlett 2004, 2311.Weis, C. D.; Newkome, G. R. J. Org. Chem. 1990, 55, 5801–5802.
- The 5-endo-trig radical- and/or iodo-cyclization of γ-alkenylamines are known to give pyrrolidine ring skeletons. See: Chatgilialoglu, C.; Ferreri, C.; Guerra, M.; Timokhin, V.; Froudakis, G.; Gimisis, T. J. Am. Chem. Soc. 2002, 124, 10765. [CrossRef]Knight, D. W.; Redfern, A. L.; Gilmore, J. J. Chem. Soc., Perkin Trans. 1. 2001, 2874.Lee, W. S.; Jang, K. C.; Kim, J. H.; Park, K. H. J. Chem. Soc., Chem. Commun. 1999, 251.
- Jachak, S. M.; Dhavale, D. D.; Karche, N. P.; Trombini, C. Tetrahedron 2004, 60, 3009.Jachak, S. M.; Dhavale, D. D.; Karche, N. P.; Trombini, C. Synlett 2004, 1549.
- Burgess, K.; Henderson, I. Tetrahedron 1992, 48, 4045.Izquierdo, I.; Plaza, M. T.; Robles, R.; Mota, A. J. Tetrahedron: Asymmetry 1998, 9, 1015.Jirousek, M. R.; Cheung, A. W-H.; Babine, R. E.; Sass, P. M.; Schow, S. R.; Wick, M. M. Tetrahedron Lett. 1993, 34, 3671.Marek, D.; Wadouachi, A.; Uzan, R.; Beaupere, D.; Nowogrocki, G.; Laplace, G. Tetrahedron Lett. 1996, 37, 49.Zhao, H.; Mootoo, D. R. J. Org. Chem. 1996, 61, 6762.Maggini, M.; Prato, M.; Ranelli, M.; Scorrano, G. Tetrahedron Lett. 1992, 33, 6537.Kang, S. H.; Kim, J. S. J. Chem. Soc., Chem. Commun. 1998, 1353.Kefalas, P.; Grierson, D. S. Tetrahedron Lett. 1993, 34, 3555.Ina, H.; Kibayashi, C. J. J. Org. Chem. 1993, 58, 52. [CrossRef]Furneaux, R. H.; Mason, J. M.; Tyler, P. C. Tetrahedron Lett. 1994, 35, 3143.Overkleeft, H. S.; Pandit, U. K. Tetrahedron Lett. 1996, 37, 547.Mulzer, J.; Dehmlow, H. J. J. Org. Chem. 1992, 57, 3194. [CrossRef]Burgess, K.; Chaplin, D. A.; Henderson, I.; Pan, Y. T.; Elbein, A. D. J. Org. Chem. 1992, 57, 1103. [CrossRef]Kim, N.-S.; Choi, J.-R.; Cha, J. K. J. Org. Chem. 1993, 58, 7096. [CrossRef]Furneaux, R. H.; Masson, J. M.; Tyler, P. C. Tetrahedron Lett. 1995, 36, 3055. [CrossRef]
- Truscheit, E.; Frommer, W.; Junge, B.; Muller, L.; Schmidt, D. D.; Wingender, W. Angew. Chem., Int. Ed. Engl. 1981, 20, 744. [CrossRef]Furneaux, R. H.; Gainsford, G. J.; Mason, J. M.; Tyler, P. C.; Hartley, O.; Winchester, B. G. Tetrahedron 1997, 57, 245.
- Humphries, M. J.; Matsumoto, K.; White, S. L. Cancer Res. 1986, 46, 5215.Humphries, M. J.; Matsumoto, K.; White, S. L.; Olden, K. Cancer Res. 1986, 46, 5215.
- Karples, A.; Fleet, G. W. J.; Dwek, R. A.; Petursson, S.; Namgoong, S. K.; Ramsden, N. G.; Jacob, G. S.; Rademacher, T. W. Proc. Natl. Acad. Sci. U.S.A. 1998, 85, 9229.Walker, B. D.; Kowalski, M.; Goh, W. C.; Kozarsky, K.; Krieger, M.; Rosen, C.; Rohrschneider, L.; Haseltine, W. A.; Sodroski, J. Proc. Natl. Acad. Sci. 1987, 84, 8120.Sunkara, P. S.; Bowling, T. L.; Liu, P. S.; Sjoerdsma, A. Biochem. Biophys. Res. Commun. 1987, 148, 206.
- Dhavale, D.; Desai, V.; Sindkhedkar, M.; Mali, R.; Castellari, C.; Trombini, C. Tetrahedron: Asymm. 1997, 1475.Dhavale, D.; Saha, N.; Desai, V. J. Org. Chem. 1997, 62, 7482.Dhavale, D.; Saha, N.; Desai, V. J. Org. Chem. 1999, 64, 1715.Dhavale, D.; Saha, N.; Desai, V. J. Chem. Soc., Chem. Commun. 1999, 1719.Saha, N.; Desai, V.; Dhavale, D. Tetrahedron 2001, 57, 39.Dhavale, D. D.; Patil, N. T.; Tilekar, J. N. J. Org. Chem. 2001, 66, 1065.Dhavale, D. D.; Patil, N. T.; Tilekar, J. N. Tetrahedron Lett. 2001, 42, 747.Dhavale, D.; Patil, N.; John, S.; Sabharwal, S. Bioorg. Med. Chem. 2002, 10, 2155.Dhavale, D.; Saha, N.; Desai, V.; Tilekar, J. Arkivoc 2002, (VII), 91.Patil, N.; Tilekar, J.; Jadhav, H.; Dhavale, D. Tetrahedron 2003, 59, 1873.
- In general, the intramolecular exo-trig nucleophilic addition reaction to a double bond, for rings smaller than five membered, is favored over the endo-trig one. See: Johnson, C. D. Acc. Chem. Res. 1993, 476.Baldwin, J. E.; Thomas, R. C.; Lawrence, I. K.; Lee, S. J. Org. Chem. 1997, 42, 3846.
- Franco, S.; Junquera, F.; Merchan, F.; Mercino, P.; Tejero, T. Syn. Commun. 1994, 2537.
- The observed rotation value of 10a was consistent with the reported value. The 1H- and 13C-NMR spectra were also found to be in good agreement with the structure [9f].
- Sample availability: Contact the authors.
© 2005 by MDPI (http://www.mdpi.org). Reproduction is permitted for non commercial purposes.