Advancing Precision Diagnosis and Therapy with Targeted Radiopharmaceuticals in Cancer and Systemic Diseases
A special issue of Pharmaceutics (ISSN 1999-4923). This special issue belongs to the section "Drug Targeting and Design".
Deadline for manuscript submissions: 31 October 2026 | Viewed by 1824
Special Issue Editors
2. Department of Radiopharmacy and Preclinical PET Imaging, Maria Skłodowska-Curie National Research Institute of Oncology, Gliwice Branch, Wybrzeze Armii Krajowej 15, 44-101 Gliwice, Poland
Interests: immuno-PET; molecular imaging; theranostics; translational research
Special Issue Information
Dear Colleagues,
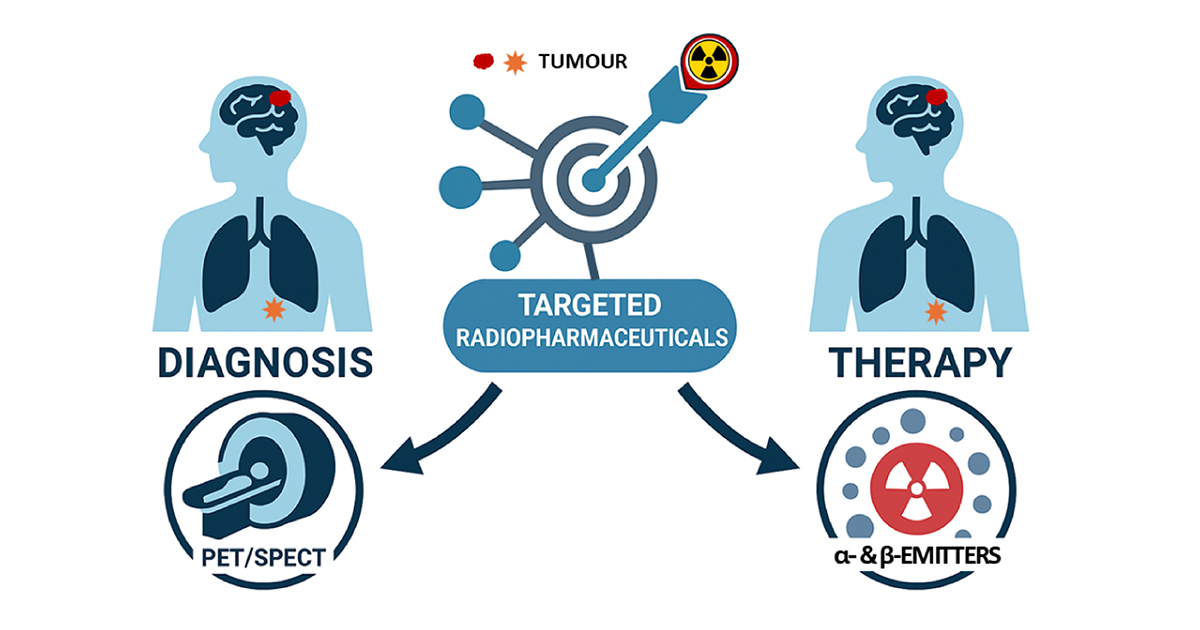
Recent advancements in radiopharmaceuticals have significantly advanced nuclear medicine, particularly in precision diagnostics and targeted therapy for cancer and neurological disorders. Among these innovations, targeted radioligand therapy (TRT) has emerged as a transformative approach that is already improving patient outcomes in prostate and neuroendocrine cancers.
TRT employs small carrier radioligands that function like precision-guided missiles. These carriers first home in on tumour-specific markers, delivering a cytotoxic payload of radioactivity directly to cancer cells while sparing most surrounding healthy tissue. Compared to external beam radiotherapy, TRTs often offer superior tumour targeting, allowing for higher radiation doses with fewer off-target effects.
Radiopharmaceuticals are now used globally on a daily basis, not only for cancer detection and staging but increasingly for therapy, particularly in oncology. They are also widely applied in the management of cardiovascular, endocrine, neurological, and psychiatric conditions. However, despite their growing clinical utility, key challenges persist in both their diagnostic and therapeutic applications.
Access to therapeutic radionuclides, particularly alpha- and beta-emitters, remains limited. The short physical half-lives of many isotopes necessitate their frequent production and rapid distribution, placing considerable logistical demands on infrastructure. The manufacturing process itself requires specialized facilities, trained personnel, and strict regulatory oversight, all of which vary widely between countries and often hinder clinical translation. In addition, radioligand therapies can be associated with off-target effects, especially in organs involved in metabolism and clearance. These challenges collectively restrict access to new agents and slow their translation from bench to bedside applications.
This Special Issue seeks to highlight recent developments in targeted radiopharmaceuticals for both imaging and therapy, with a focus on cancer and other systemic diseases. We welcome contributions on topics including novel radionuclide production and the design and preclinical/clinical evaluation of innovative radiotracers, cutting-edge TRT strategies, and technological advances that aim to streamline production and improve accessibility.
We welcome original research articles and comprehensive reviews.
Prof. Dr. Gabriela Kramer-Marek
Dr. Yingqing Lu
Guest Editors
Manuscript Submission Information
Manuscripts should be submitted online at www.mdpi.com by registering and logging in to this website. Once you are registered, click here to go to the submission form. Manuscripts can be submitted until the deadline. All submissions that pass pre-check are peer-reviewed. Accepted papers will be published continuously in the journal (as soon as accepted) and will be listed together on the special issue website. Research articles, review articles as well as short communications are invited. For planned papers, a title and short abstract (about 250 words) can be sent to the Editorial Office for assessment.
Submitted manuscripts should not have been published previously, nor be under consideration for publication elsewhere (except conference proceedings papers). All manuscripts are thoroughly refereed through a single-blind peer-review process. A guide for authors and other relevant information for submission of manuscripts is available on the Instructions for Authors page. Pharmaceutics is an international peer-reviewed open access monthly journal published by MDPI.
Please visit the Instructions for Authors page before submitting a manuscript. The Article Processing Charge (APC) for publication in this open access journal is 2900 CHF (Swiss Francs). Submitted papers should be well formatted and use good English. Authors may use MDPI's English editing service prior to publication or during author revisions.
Keywords
- radiopharmaceuticals
- diagnostics imaging
- theranostics
- radionuclides
- radioligand therapy
- cancer
- neurological disorders
- dosimetry
- preclinical studies
- clinical studies
- clinical translation
Benefits of Publishing in a Special Issue
- Ease of navigation: Grouping papers by topic helps scholars navigate broad scope journals more efficiently.
- Greater discoverability: Special Issues support the reach and impact of scientific research. Articles in Special Issues are more discoverable and cited more frequently.
- Expansion of research network: Special Issues facilitate connections among authors, fostering scientific collaborations.
- External promotion: Articles in Special Issues are often promoted through the journal's social media, increasing their visibility.
- Reprint: MDPI Books provides the opportunity to republish successful Special Issues in book format, both online and in print.
Further information on MDPI's Special Issue policies can be found here.







