Journal Description
Journal of Market Access & Health Policy
Journal of Market Access & Health Policy
(JMAHP) is an international, peer-reviewed, open access journal that covers all different subdisciplines of ‘market access’ from economic, technical, scientific, sociological, psychological and policy perspectives, published quarterly online. It is the official journal of the Market Access Society (MAS). Society members receive discounts on the APCs.
- Open Access— free for readers, with article processing charges (APC) paid by authors or their institutions.
- High Visibility: indexed within Scopus, PubMed, PMC, and other databases.
- Rapid Publication: manuscripts are peer-reviewed and a first decision is provided to authors approximately 40.8 days after submission; acceptance to publication is undertaken in 14.6 days (median values for papers published in this journal in the second half of 2025).
- Journal Rank: CiteScore - Q2 (Health Policy)
- Recognition of Reviewers: APC discount vouchers, optional signed peer review, and reviewer names published annually in the journal.
Latest Articles
Operative Costs and Anesthesia Exposure Time for Pediatric Glaucoma Surgeries
J. Mark. Access Health Policy 2026, 14(2), 34; https://doi.org/10.3390/jmahp14020034 - 3 Jun 2026
Abstract
Background: Pediatric glaucomas are sight-threatening conditions that frequently require surgical intervention and may expose vulnerable young patients to repeated episodes of general anesthesia. While the clinical efficacy of pediatric glaucoma surgeries has been described, comparative data on operative cost and anesthesia exposure
[...] Read more.
Background: Pediatric glaucomas are sight-threatening conditions that frequently require surgical intervention and may expose vulnerable young patients to repeated episodes of general anesthesia. While the clinical efficacy of pediatric glaucoma surgeries has been described, comparative data on operative cost and anesthesia exposure time are scarce, limiting evidence-based decision-making for surgeons, caregivers, and health systems. Objective: To quantify and compare the operative costs and total operating room (OR) time of common pediatric glaucoma surgeries, with particular attention to unilateral angle surgery versus immediately sequential bilateral angle surgery (ISBAS), and to single-incision ab interno trabeculotomy (SIT) versus other angle techniques. Methods: A retrospective review of patients who received glaucoma surgery by one of three experienced glaucoma surgeons between January 2012 and August 2019 at the University of Minnesota Medical Center. Each surgery was classified by type, cost, and operative time. Results: A total of 160 surgical encounters were analyzed. Average total cost was $6564 for all unilateral procedures, $6782 (±1952.03) for unilateral angle surgeries, and $11,391 (±2396) for ISBAS. Mean OR time was 121 min for all unilateral procedures, 120 min (±46.3) for unilateral angle surgeries, and 208 min (±53.8) for ISBAS—approximately the cost and time of two unilateral angle surgeries combined into a single anesthetic encounter. Compared to separate encounters for bilateral angle surgeries, ISBAS saved $2174 (p = 0.008) and trended toward saving 33 min of OR time (p = 0.068). Single-incision ab interno trabeculotomy (SIT) was $1647 more expensive than conventional incisional goniotomy or trabeculotome trabeculotomy (p < 0.0001) and reduced OR time by 23.5 min (p = 0.011). SIT was as expensive as 360-degree trabeculotomy ab externo with the iScienceTM catheter (p = 0.098) and reduced OR time by 74.4 min (p = 0.0004). There was no difference in complications between unilateral surgery and ISBAS. Conclusions: Reduced cost and a trend toward reduced anesthesia time support the use of ISBAS. SIT substantially reduced general anesthesia exposure for a neutral or slightly increased cost.
Full article
Open AccessArticle
A Decision Tree Cost Analysis of Intracranial Bleed Detection Using a Near-Infrared Device Across Various Healthcare Levels
by
Mamta Patel, Amit Kumar Mittal, Mohit Agrawal, Oshima Sachin, Kavitha Rajsekar, Bharat Choudhary, Suryanarayanan Bhaskar and Kuldeep Singh
J. Mark. Access Health Policy 2026, 14(2), 33; https://doi.org/10.3390/jmahp14020033 - 1 Jun 2026
Abstract
►▼
Show Figures
Traumatic brain injury (TBI), mainly caused by road traffic accidents, is a serious global public health concern. Computed tomography (CT) is the best way to detect intracranial haemorrhage (ICH), but it is not always feasible because it is hard to access, exposes people
[...] Read more.
Traumatic brain injury (TBI), mainly caused by road traffic accidents, is a serious global public health concern. Computed tomography (CT) is the best way to detect intracranial haemorrhage (ICH), but it is not always feasible because it is hard to access, exposes people to radiation, and is expensive, especially in low- and middle-income countries. Portable near-infrared spectroscopy (NIRS) devices offer a non-invasive, point-of-care option for early detection of ICH. The objectives of this study were to estimate the cost per case detected for patients with mild-to-moderate TBI, to estimate incremental cost and to perform a budget impact analysis to assess the financial feasibility of implementing this technology. This study employed a decision tree model from a health system perspective to calculate the cost per detected case and the incremental cost of NIRS across three tiers of care: ambulances, community health centres (CHCs), and tertiary hospitals. The cost per mild-to-moderate TBI case found was Rs. 2177.90 in ambulances, Rs. 748.09 in CHCs, and Rs. 628.14 in tertiary hospitals. The extra cost per patient was Rs. 984.15, Rs. 360.90, and Rs. 289.78, respectively. At the system level, NIRS raised the total costs for 264 ambulance patients from Rs. 37.71 lakh to Rs. 40.31 lakh and for 858 CHC patients from Rs. 115.64 lakh to Rs. 118.73 lakh. National extrapolation indicates a first-year budgetary impact of approximately Rs. 442 crores for ambulances and Rs. 187 crores for CHCs. These results support the strategic, phased implementation of NIRS to use resources better and improve early diagnosis of TBI.
Full article

Figure 1
Open AccessArticle
Use of Real-World Evidence in Health Technology Assessments of Non-Oncology Rare Disease Therapies
by
Oliver Blandy, Pierluigi Lembo, Rebecca Folorunso, Karl-Johan Myren, Helene Chevrou-Severac and Simu K. Thomas
J. Mark. Access Health Policy 2026, 14(2), 32; https://doi.org/10.3390/jmahp14020032 - 21 May 2026
Abstract
►▼
Show Figures
Background: Real-world evidence (RWE) can complement clinical trials to inform health technology assessments (HTAs). This study examined the extent to which RWE is considered in HTAs of non-oncology orphan medicinal products across six agencies globally. Methods: Published European Medicines Agency decisions were reviewed
[...] Read more.
Background: Real-world evidence (RWE) can complement clinical trials to inform health technology assessments (HTAs). This study examined the extent to which RWE is considered in HTAs of non-oncology orphan medicinal products across six agencies globally. Methods: Published European Medicines Agency decisions were reviewed to identify approved non-oncology orphan medicinal products (2018–2023) that included RWE within their submission package, which was anticipated to align with the inclusion of RWE in HTA submissions. Data were extracted from the corresponding HTA reports published by six national agencies (Australia, Canada, France, Germany, Sweden, and the UK). Results: RWE was included in 105 regulatory submissions and 52.6% of the corresponding HTA reports (range: 29.9% [Germany] to 78.8% [Canada]), nearly 90% of which received a positive decision (range: 44.4% [Australia] to 100.0% [Germany]). RWE was derived from a variety of study designs and commonly supported clinical efficacy across many therapeutic areas. Conclusions: RWE commonly supports HTAs of recently approved non-oncology orphan medicinal products, strengthening the evidence base and contributing to demonstration of product value.
Full article

Figure 1
Open AccessEditorial
EU-HTA—Why Do Words Matter?
by
Mondher Toumi, Bruno Falissard, Steven Simoens, Maarten Postma, Marta Wilk, Laurent Boyer, Renato Bernardini, Stefano Capri, Jaime Espin, Jürgen Wasem and Pascal Auquier
J. Mark. Access Health Policy 2026, 14(2), 31; https://doi.org/10.3390/jmahp14020031 - 18 May 2026
Abstract
Words matter because they have profound power to shape thoughts, emotions, actions, and social realities [...]
Full article
Open AccessPerspective
Between Rigor and Relevance: Why the EU HTA Guidelines on Indirect Comparisons Miss the Mark
by
Samuel Aballéa, Mondher Toumi, Piotr Wojciechowski, Emilie Clay, Bruno Falissard, Steven Simoens, Pascal Auquier, Stefano Capri, Renato Bernardini, Joerg Ruof, Frank-Ulrich Fricke, Oriol Sola Morales and Laurent Boyer
J. Mark. Access Health Policy 2026, 14(2), 30; https://doi.org/10.3390/jmahp14020030 - 7 May 2026
Abstract
Indirect treatment comparisons (ITCs) are essential in the context of joint clinical assessments (JCAs) under Regulation (European Union [EU]) 2021/2282, bridging evidence gaps where head-to-head data are lacking and enabling assessment across diverse national patient, intervention, comparator, and outcome (PICO) requirements. This paper
[...] Read more.
Indirect treatment comparisons (ITCs) are essential in the context of joint clinical assessments (JCAs) under Regulation (European Union [EU]) 2021/2282, bridging evidence gaps where head-to-head data are lacking and enabling assessment across diverse national patient, intervention, comparator, and outcome (PICO) requirements. This paper critically reviews the EU Health Technology Assessment Coordination Group’s (HTACG) guidelines on direct and indirect comparisons, with particular focus on ITCs. While the guidelines promote transparency and rigorous evaluation of assumptions, they adopt a restrictive stance on assumption violations, the use of unanchored comparisons, and population-adjusted methods such as matching-adjusted indirect comparisons (MAIC) and simulated treatment comparisons (STC). The guidance shows limited support for Bayesian methods and undervalues meta-regression in favor of subgroup analyses. Operational implications for health technology developers (HTDs) are substantial, including new requirements for dual systematic reviews, multiple network structures, and shifted null hypothesis testing. Moreover, the guidelines effectively dissuade the use of non-randomized comparisons in rare or rapidly evolving indications and may inadvertently hinder access to effective treatments. Emerging practices such as external control arms (ECA) or target trial emulation are underdeveloped. Notably, there is no indication that the guidelines are grounded in systematic methodological validation studies. As JCAs evolve, greater methodological flexibility, empirical grounding, and clear operational guidance will be essential. Refining the guidelines along these principles would enhance their practical utility, mitigate intrinsic assessment variability, support consistent assessments across Member States (MS), and ultimately improve patient access to innovative therapies.
Full article
(This article belongs to the Collection European Health Technology Assessment (EU HTA))
Open AccessArticle
Impact of a Digital Leakage Notification System on Healthcare Resource Utilisation and Costs for People with Stomas in the United Kingdom: Analysis of Interim Results from a Prospective Longitudinal Study
by
Alexandra Baxter, Esben Bo Boisen, Majken Linnemann Jensen and Aamir Shaikh
J. Mark. Access Health Policy 2026, 14(2), 29; https://doi.org/10.3390/jmahp14020029 - 6 May 2026
Abstract
►▼
Show Figures
Background/Objectives: Leakage of stomal effluent adversely affects the quality of life for people living with a stoma and increases healthcare resource utilisation and costs. The Heylo™ digital leakage notification system (DLNS) detects early signs of leakage under the baseplate, enabling proactive stoma
[...] Read more.
Background/Objectives: Leakage of stomal effluent adversely affects the quality of life for people living with a stoma and increases healthcare resource utilisation and costs. The Heylo™ digital leakage notification system (DLNS) detects early signs of leakage under the baseplate, enabling proactive stoma management. We evaluated the impact of the DLNS on stoma-related costs in a real-world UK setting. Methods: This costing analysis used data from the first 100 DLNS users with 6 months of follow-up in an ongoing longitudinal observational study (NCT06554015). Costs were calculated as 3-month totals before DLNS (baseline): Month 3, and Month 6 for stoma-related healthcare provider consultations, hospitalisations, and ostomy solution components (pouching systems, supporting products, and DLNS sensor layers). Least squares (LS) means were estimated using a mixed model. Results: Total LS mean costs for stoma-related consultations plus ostomy solution use decreased significantly from baseline by 21.9% at Month 3 (−£304 [95% CI: −£456, −£153]; p = 0.001) and 24.5% at Month 6 (−£340 [95% CI: −£504, −£175]; p < 0.001). Stoma-related consultation costs decreased by 50.7% at Month 3 (−£309 [95% CI: −£447, 171]; p < 0.001) and 57.9% at Month 6 (−£353 [95% CI: −£493, −£213]; p < 0.001), driven by fewer physician and stoma care nurse consultations. Mean ostomy solution costs remained similar from baseline (£784/person) to Month 6 (£782/person; p = 0.955) as DLNS sensor layer costs were offset by less use of other ostomy products. Conclusions: Initiation of the Heylo™ DLNS was associated with significant cost savings from reduced stoma-related consultations, pouching systems, and supporting product use.
Full article
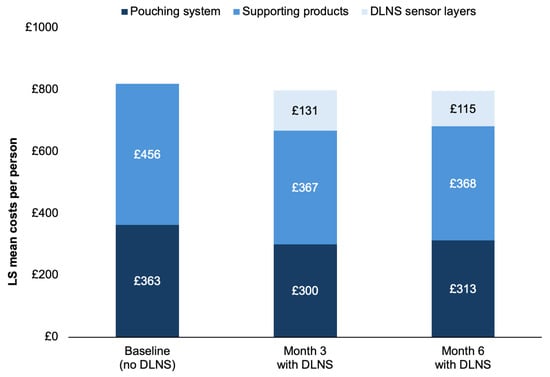
Figure 1
Open AccessArticle
Assessment and Appraisal of Drug Innovativeness in Italy: Ultimate Evidence on Key Drivers and Consistency
by
Alvise Verde, Federica Turati, Clara Trimarchi, Carlotta Galeone and Claudio Jommi
J. Mark. Access Health Policy 2026, 14(2), 28; https://doi.org/10.3390/jmahp14020028 - 2 May 2026
Abstract
►▼
Show Figures
This study aims to update and integrate empirical evidence on the key drivers and consistency of the appraisals of drug innovativeness in Italy by the Italian Medicines Agency (AIFA), and discuss if this evidence is supportive of the reform and requirements implemented in
[...] Read more.
This study aims to update and integrate empirical evidence on the key drivers and consistency of the appraisals of drug innovativeness in Italy by the Italian Medicines Agency (AIFA), and discuss if this evidence is supportive of the reform and requirements implemented in 2025. Appraisals from July 2017 to December 2024 were retrieved from the AIFA website. The association between the innovativeness appraisal, the innovativeness domains (unmet need/added therapeutic value/quality of evidence) and disease/drug/evidence-specific variables was assessed using odds ratios (ORs) from binary/multinomial logistic regression models. Innovativeness status was strongly associated with added therapeutic value (OR > 70). Medicines for rare diseases were more likely to receive conditional innovativeness (OR = 2.95). Full innovativeness was more frequently recognized for indications including paediatric patients (OR = 3.60). References to severe diseases and patient-reported outcomes (PROs) had a higher, not statistically significant, likelihood of innovativeness, whereas reference to indirect treatment comparisons had a lower likelihood (OR = 0.18). The appraisal process showed high internal consistency, but its regulation needs more specific guidance. The innovativeness regulation was reformed in July 2025, including specific recommendations on the criteria to identify the alternative treatments; the role and robustness of indirect comparisons; and the role and requirements for PROs. Our evidence provides an empirical rationale for this reform.
Full article

Figure 1
Open AccessCommentary
Primary Care or Primary Problem? Aligning Access Pathways with Patient Needs Across the Care Continuum
by
Gregory J. Privitera, James J. Gillespie and Alexa Walton
J. Mark. Access Health Policy 2026, 14(2), 27; https://doi.org/10.3390/jmahp14020027 - 1 May 2026
Abstract
In the United States, access to healthcare is shaped not only by patient need but also by payer policies that determine which providers are reimbursable, how care is sequenced, and what constitutes a legitimate entry point into the system. These gatekeeping functions, while
[...] Read more.
In the United States, access to healthcare is shaped not only by patient need but also by payer policies that determine which providers are reimbursable, how care is sequenced, and what constitutes a legitimate entry point into the system. These gatekeeping functions, while valuable for supporting clinical prioritization, risk stratification, and continuity of care, can also unintentionally reinforce structural inequities and credential hierarchies that delay or limit timely and equitable care, particularly for historically marginalized populations. While reform efforts often focus on expanding benefits or provider networks, fewer address the underlying design of access itself or the rules that govern how patients enter care. It is argued in this paper that a more equitable and efficient healthcare system requires multi-entry care models, in which nurses, behavioral health clinicians, pharmacists, and community health workers may serve as condition-appropriate, reimbursable first points of contact within coordinated care teams. Drawing on evidence from Medicare, Medicaid, the Veterans Health Administration, and commercial payers, these models may support cost containment, improve care coordination, facilitate appropriate utilization, and promote earlier patient engagement. While findings from these models are not uniform across all settings, evidence suggests that outcomes are highly dependent on implementation context, system design, and supporting infrastructure. When implemented with appropriate safeguards (such as interoperable health records, team-based care requirements, and coordinated referral tracking), multi-entry systems can preserve continuity while expanding access. Payers are uniquely positioned to lead this transformation by aligning reimbursement policy with patient needs, supporting team-based care infrastructure, and embedding accountability into access pathways, thereby creating a system that can be more responsive, inclusive, and sustainable.
Full article
Open AccessArticle
Real-World Shelf Life of Adrenaline Auto-Injectors at Pharmacies in Denmark, Finland, Sweden, and Norway
by
Jesper Nørregaard, Christoffer Mertz, Anne Danø, Jeppe Hæstrup Kamstrup and Mille Vang Lybech
J. Mark. Access Health Policy 2026, 14(2), 26; https://doi.org/10.3390/jmahp14020026 - 1 May 2026
Abstract
►▼
Show Figures
There is a well-documented gap between the prescription of adrenaline auto-injectors (AAIs) and their real-world use during anaphylaxis. Although several aspects of AAI underuse have been investigated, the potential role of shelf life in influencing patient adherence has not been quantified. This study
[...] Read more.
There is a well-documented gap between the prescription of adrenaline auto-injectors (AAIs) and their real-world use during anaphylaxis. Although several aspects of AAI underuse have been investigated, the potential role of shelf life in influencing patient adherence has not been quantified. This study assessed the real-world remaining shelf life of AAIs available at pharmacies in Denmark, Finland, Sweden, and Norway, using pharmacy-level stock data and pharmacy employee-reported perceptions. Across Denmark, Finland, and Sweden, the average remaining shelf life was 9.6 months, and in Norway it was 10.5 months at the point of dispensing. In Denmark, Finland and Sweden, 100%, 91%, and 94% of employees, respectively, considered shelf life an important or very important factor when dispensing AAIs to patients. Our findings suggest that patients and caregivers filling prescriptions for AAIs frequently receive devices with limited remaining shelf life, which may necessitate multiple renewals per year. This has potential implications in terms of adherence to clinical guidelines, dependence of expired devices during emergencies, patient cost, caregiver burden, and overall societal expenditure. These results highlight an unmet need for emergency treatment options with longer shelf life to better support continuous access to life-saving medicine during anaphylaxis.
Full article

Figure 1
Open AccessArticle
Impact of Market Access Delays on Time to Patient Access: Multi-Country Comparative Analysis Assessing the First Commercial Launch Indications for Five Oncology Medicines Across Europe and Canada
by
Barry Crean, David Parry, Alison Horsfield, James Ryan and Nektarios Oraiopoulos
J. Mark. Access Health Policy 2026, 14(2), 25; https://doi.org/10.3390/jmahp14020025 - 28 Apr 2026
Cited by 1
Abstract
►▼
Show Figures
Background: The benefit of pharmaceutical innovation manifests when patients access treatment. Following regulatory approval in Europe and Canada, reimbursement decisions depend on health technology assessments (HTAs), which can be prolonged. To quantify the impact of delays on patients, we evaluated market access timelines
[...] Read more.
Background: The benefit of pharmaceutical innovation manifests when patients access treatment. Following regulatory approval in Europe and Canada, reimbursement decisions depend on health technology assessments (HTAs), which can be prolonged. To quantify the impact of delays on patients, we evaluated market access timelines for olaparib, osimertinib, durvalumab, acalabrutinib, and trastuzumab deruxtecan across six high-income countries with established HTA systems (Canada, England, France, Germany, Italy, Spain). Methods: Time to access was from regulatory approval to reimbursement. Survival benefit was median overall survival (OS) and progression-free survival (PFS) assessed versus the comparator at approval and the latest data cut-off. The number of eligible patients per year multiplied by the years to patient access and survival benefit reflects the lost survival benefit. Results: Efficacy benefits observed at approval continued to the latest data cut-offs. The mean time to patient access was 18 months. Although this varied by country and treatment, with England and Germany typically being the fastest and France and Spain the slowest, timelines often exceeded the 180-day EU target despite identical evidence used in HTA submissions. This resulted in an estimated mean of 2836 patients being unable to access treatment and 3391 OS-derived and 2739 PFS-derived life-years lost. Conclusions: Access processes must evolve to ensure the timely realization of new medicines’ benefits.
Full article

Figure 1
Open AccessPerspective
Introducing an Expanded Value Framework in Health Technology Assessment of Vaccines
by
Farzaneh Eslami, Thi Hao Pham, Angga P. Kautsar, Cao Ba Khuong, Cornelis Boersma, Mondher Toumi, Jurjen van der Schans and Maarten J. Postma
J. Mark. Access Health Policy 2026, 14(2), 24; https://doi.org/10.3390/jmahp14020024 - 28 Apr 2026
Abstract
Health Technology Assessment (HTA) frameworks increasingly recognize the broader value elements of vaccines; however, their adoption remains inconsistent across jurisdictions and often incomplete in practice. Many HTA processes continue to prioritize narrow clinical outcomes and direct costs, leading to the underrepresentation of the
[...] Read more.
Health Technology Assessment (HTA) frameworks increasingly recognize the broader value elements of vaccines; however, their adoption remains inconsistent across jurisdictions and often incomplete in practice. Many HTA processes continue to prioritize narrow clinical outcomes and direct costs, leading to the underrepresentation of the full preventive and long-term benefits of vaccination. Building on the ISPOR “Elements of Value” framework and recent evidence, this study adapts and expands existing models specifically for vaccines to enhance HTA applicability in both high-income and resource-limited settings. We introduce an updated vaccine value framework comprising 21 distinct value elements. Notably, the original model was expanded by introducing four entirely new value drivers: (1) real-world evidence; (2) control of antimicrobial resistance; (3) health system strengthening; and (4) environmental impact. Additionally, existing elements were refined, such as broadening “fear of contagion” to “peace of mind” and expanding “productivity” to capture education and leisure gains. We map these elements to potential data sources and methodological tools to facilitate their inclusion in HTA. This study offers an operational, holistic, and context-sensitive framework that reflects current advancements in assessment. By capturing the full spectrum of vaccine value, this framework aims to support more comprehensive, transparent, and equitable HTA decision-making for global immunization programs, while considering conceptual overlap between value elements to reduce the risk of double counting.
Full article
(This article belongs to the Special Issue Health Technology Assessment of Vaccination: Strategies, Public Health and Values: 2nd Edition)
►▼
Show Figures

Figure 1
Open AccessArticle
When Do Annuity-Based Payments Help to Address the Affordability Challenge of Funding Advanced Therapies? Insights from a Budget Impact Simulation
by
Walter Van Dyck, Sissel Michelsen, David Veredas, Isabelle Huys, Jeroen Luyten and Steven Simoens
J. Mark. Access Health Policy 2026, 14(2), 23; https://doi.org/10.3390/jmahp14020023 - 20 Apr 2026
Abstract
►▼
Show Figures
Spreading payments over time by means of annuities has been proposed as a means to address the affordability challenge of funding very expensive advanced therapies, especially within managed entry agreements. This study aims to examine when annuities (in contrast with a single upfront
[...] Read more.
Spreading payments over time by means of annuities has been proposed as a means to address the affordability challenge of funding very expensive advanced therapies, especially within managed entry agreements. This study aims to examine when annuities (in contrast with a single upfront payment) offer a viable solution for both healthcare payers and manufacturers to fund one-time advanced therapies. We put forward four conditions under which annuity-based payments can be considered an acceptable payment strategy: (1) excessive budget impact, (2) cost equivalence with upfront payment, (3) compensation for financial risk and (4) a limited annuity period. We develop an exploratory model that simulates how the budget impact of annuity-based payments for advanced therapies meets these conditions across several economic and epidemiological scenarios. Given our model parameter values, results suggest that annuity-based payments are most suitable when the initial patient volume (prevalence) significantly exceeds annual new cases (incidence), and when the financial risk premium for the annuity-based payment scheme does not exceed the social discount rate. While further refinement of the model is needed, this study demonstrates that annuity-based payments can only help control the annual budget need when the focus is on a high-prevalence disease, and the therapy is financed through health impact bonds issued by a governmental payer. This arrangement ensures a low-risk premium, which is typically only available to public payers.
Full article

Figure 1
Open AccessReview
Household Out-of-Pocket Burden Costs for Pediatric Pneumonia in Low- and Middle-Income Countries: Evidence Review and Econometric Framework
by
Ioannis Smaraidos, Maria Kyrmanidou and Asterios Kampouras
J. Mark. Access Health Policy 2026, 14(2), 22; https://doi.org/10.3390/jmahp14020022 - 13 Apr 2026
Abstract
►▼
Show Figures
Pediatric pneumonia remains a major cause of morbidity and mortality in low- and middle-income countries (LMICs), imposing both health and financial burdens. While the clinical aspects of pediatric pneumonia are well-studied, less attention has been paid to its economic implications for households, particularly
[...] Read more.
Pediatric pneumonia remains a major cause of morbidity and mortality in low- and middle-income countries (LMICs), imposing both health and financial burdens. While the clinical aspects of pediatric pneumonia are well-studied, less attention has been paid to its economic implications for households, particularly regarding out-of-pocket (OOP) expenditure. This paper synthesizes current evidence from Kenya, India, Bangladesh, and Vietnam and introduces a proposed econometric framework designed to identify cost determinants and model policy interventions. The framework integrates microeconomic data, identifies cost determinants, and models the effects of clinical and policy factors (e.g., intensive care, vaccination, insurance coverage) on household expenditures. Simulated results illustrate potential findings from such an approach. Existing studies show substantial variability in hospitalization costs, with OOP payments ranging from US$30 to US$250 per episode, often exceeding 20% of monthly household income. Econometric modeling using generalized linear models (GLMs) and difference-in-differences (DiD) can disentangle the impact of hospital practices, disease severity, and policy interventions. Simulated regression results demonstrate that length of stay, intensive care admission, and absence of insurance significantly increase household costs, while pneumococcal conjugate vaccine (PCV) introduction reduces both admissions and financial burden. Hospitalization for pediatric pneumonia imposes significant OOP costs on households in LMICs. An econometric framework provides rigorous tools to estimate cost drivers, evaluate policy impacts, and guide equitable health financing reforms.
Full article

Figure 1
Open AccessArticle
Accessibility of Biologic Drugs in Morocco: Comparison with FDA and EMA Approvals (2015–2025)
by
Mounir Charrak, Yahia Cherrah and Samira Serragui
J. Mark. Access Health Policy 2026, 14(2), 21; https://doi.org/10.3390/jmahp14020021 - 9 Apr 2026
Abstract
►▼
Show Figures
This study aims to evaluate the rates and timeframes of the availability and reimbursement of biologic drugs in Morocco, after approval by the Food and Drug Administration (FDA) or the European Medicines Agency (EMA). The results will help to identify disparities in access
[...] Read more.
This study aims to evaluate the rates and timeframes of the availability and reimbursement of biologic drugs in Morocco, after approval by the Food and Drug Administration (FDA) or the European Medicines Agency (EMA). The results will help to identify disparities in access and promote rapid access to these innovative treatments. This descriptive study established an international reference list of biological medicines, based on data from the FDA and EMA for the period from 2015 to 2025. An analysis was conducted using national sources, focusing on the availability, reimbursement rates, and timeframes for each listed biological drug. Of the 233 listed biological drugs, only 13.7% (32/233) of those approved between 2015 and 2025 are available in Morocco. Of these, 87.5% (28/32) have been priced, and only 10.7% (3/28) have been approved for reimbursement. The average time between FDA/EMA approval and pricing in Morocco is 3.75 years and 3.41 years, respectively, while the average reimbursement approval time is 2.74 years. This study highlights the delay and limited access for Moroccan patients to internationally approved biologic drugs.
Full article

Figure 1
Open AccessArticle
Mapping the Use of Real-World Evidence Across the EU Health Technology Assessment Regulation: Methodological Considerations, Challenges, and Opportunities for Harmonization
by
Grammati Sarri, Bengt Liljas, Keith R. Abrams, Stephen J. Duffield and Murtuza Bharmal
J. Mark. Access Health Policy 2026, 14(2), 20; https://doi.org/10.3390/jmahp14020020 - 8 Apr 2026
Abstract
Methodological guidelines for real-world evidence (RWE) in European Union (EU) joint clinical assessments (JCA) are lacking. This manuscript explores RWE potential in EU health technology assessment (HTA) and offers recommendations for generating high-quality RWE. An environmental scan of peer-reviewed and gray literature was
[...] Read more.
Methodological guidelines for real-world evidence (RWE) in European Union (EU) joint clinical assessments (JCA) are lacking. This manuscript explores RWE potential in EU health technology assessment (HTA) and offers recommendations for generating high-quality RWE. An environmental scan of peer-reviewed and gray literature was conducted to review RWE frameworks and documents in EU regulatory and HTA decision-making. Extraction elements were standardized across key RWE themes: data quality, methodological rigor, stakeholder engagement, and applications. In JCA, RWE has multiple uses, including informing PICO simulation exercises, understanding disease landscape, identifying prognostic factors and effect modifiers, and directly or indirectly informing comparative clinical assessments. Methodological guidance from the HTA Coordination Group is limited to cases in which evidence from non-randomized studies is used as direct inputs in comparative assessments. Individual HTA bodies provide more detailed guidance, missing an opportunity to leverage RWE within JCAs that can offer insight for local Member State submissions. Generating high-quality RWE that is credible, actionable, and acceptable for JCA submissions and local HTA bodies requires careful attention to methodological considerations and early planning. Broader RWE integration that reflects patient journeys is needed. Expanding the HTA Coordination Group guidance can unlock RWE’s full potential in supporting EU JCA submissions.
Full article
(This article belongs to the Collection European Health Technology Assessment (EU HTA))
►▼
Show Figures

Figure 1
Open AccessPerspective
When Activism Becomes Survival: The Mental Health Costs of Constant Resistance in the Digital Era in the Balkans—A Health Policy Perspective
by
Aleksandar Sič, Svetozar Mijuskovic and Nebojsa Brezic
J. Mark. Access Health Policy 2026, 14(2), 19; https://doi.org/10.3390/jmahp14020019 - 2 Apr 2026
Abstract
►▼
Show Figures
Activism exposes individuals to sustained harassment, threat and psychological strain in contexts marked by discrimination and weak institutional protection. For LGBTQ communities, public engagement frequently increases vulnerability to both offline and digital harm, with cumulative consequences for mental health. Using the Balkans as
[...] Read more.
Activism exposes individuals to sustained harassment, threat and psychological strain in contexts marked by discrimination and weak institutional protection. For LGBTQ communities, public engagement frequently increases vulnerability to both offline and digital harm, with cumulative consequences for mental health. Using the Balkans as a case example, this perspective sees activist mental health through a public health and health policy lens, framing distress not as an individual coping failure but as an outcome of structural barriers and minority stress processes, including inadequate legal protection, limited access to culturally competent mental health care and insufficient accountability for platform-mediated harm. This article highlights the population-level implications of unaddressed structural stressors, like burnout, disengagement and reduced sustainability of civil society participation, by situating activist mental health within broader questions of health system performance, access to care and governance. Upstream policy responses that strengthen institutional protection, ensure equitable access to mental health services and promote safer digital environments would address these challenges, positioning activist mental health as a critical public health policy issue.
Full article

Figure 1
Open AccessArticle
Cost-Effectiveness of 3D-Printed Patient-Specific Versus Off-the-Shelf Interbody Cages in Lumbar Spinal Fusion: A Markov Model Cost-Utility Analysis
by
Jackson C. Hill, Ralph J. Mobbs, Marc Coughlan, Kevin A. Seex, Chloe A. Amaro, William R. Walsh and William C. H. Parr
J. Mark. Access Health Policy 2026, 14(2), 18; https://doi.org/10.3390/jmahp14020018 - 25 Mar 2026
Abstract
►▼
Show Figures
The aim of the present study was to compare the cost-effectiveness of 3DMorphic’s spinal 3DFusion Lumbar (3DFL) cages versus Off-The-Shelf (OTS) cages for patients undergoing lumbar interbody fusion in an Australian healthcare setting. 3DFL cages differ from generic OTS cages in that they
[...] Read more.
The aim of the present study was to compare the cost-effectiveness of 3DMorphic’s spinal 3DFusion Lumbar (3DFL) cages versus Off-The-Shelf (OTS) cages for patients undergoing lumbar interbody fusion in an Australian healthcare setting. 3DFL cages differ from generic OTS cages in that they are Patient-Specific Interbody Cages (PSICs). While several studies have discussed the clinical benefits of PSIC versus OTS cages, no studies have evaluated the cost-effectiveness of this technology. Without a direct randomised controlled trial between the two implant categories, an indirect treatment comparison was performed. The indirect comparison was informed by a clinical trial of 3DFL cages, the Australian Spine Registry and an analysis of reoperation rates for patients undergoing spinal fusion in an Australian cohort. In conclusion, the PSICs were demonstrated to be clinically superior to OTS cages as measured by Health Related Quality of Life (HRQoL) and reoperation rates. The cost-utility analysis demonstrated that 3DFL cages were cost-effective compared to OTS cages in an Australian healthcare setting.
Full article

Figure 1
Open AccessArticle
Comparative Cost Evaluation of Managed Entry Agreement Techniques Using Real-World Data from High-Cost Anticancer Drugs in Thailand
by
Piyapat Owat, Chaoncin Sooksriwong, Hataiwan Ratanabunjerdkul and Tuangrat Phodha
J. Mark. Access Health Policy 2026, 14(1), 17; https://doi.org/10.3390/jmahp14010017 - 20 Mar 2026
Abstract
High-cost innovative anticancer drugs pose challenges for health systems in balancing timely patient access with long-term financial sustainability. In Thailand, reliance on Health Technology Assessment for reimbursement decisions may delay access, highlighting the potential role of Managed Entry Agreements (MEAs) as complementary policy
[...] Read more.
High-cost innovative anticancer drugs pose challenges for health systems in balancing timely patient access with long-term financial sustainability. In Thailand, reliance on Health Technology Assessment for reimbursement decisions may delay access, highlighting the potential role of Managed Entry Agreements (MEAs) as complementary policy instruments to manage uncertainty related to price, effectiveness, and use; however, MEA application remains limited and lacks an analytical framework for technique selection. This study used real-world data from Thammasat University Hospital to examine and compare the cost-saving performance of five MEA techniques—discount, free initiation treatment, utilization cap, conditional treatment continuation, and pay-by-result—across six high-cost anticancer drugs representing dominant uncertainty characteristics. Drug procurement costs were modeled over a 24-month horizon from the payer’s perspective, and one-way sensitivity analyses were conducted using ±10% variation in median progression-free survival. Free initiation treatment generated the highest cost savings across uncertainty types, followed by conditional treatment continuation, while utilization cap and discount produced more moderate savings. Pay-by-result demonstrated the lowest cost-saving potential. Sensitivity analyses confirmed the robustness of comparative rankings. Overall, the findings indicate that MEA performance varies according to dominant sources of drug-related uncertainty and support a more structured, context-appropriate approach to MEA selection to strengthen market access and value-based pricing in Thailand.
Full article
Open AccessArticle
Validation of a De Novo Health Economic Model for Finerenone in Heart Failure with Left Ventricular Ejection Fraction ≥40%
by
Tobiasz Lemański, Kerstin Folkerts, Phil McEwan, Paul Mernagh, Mateusz Robert Żemojdzin and Michał Pochopień
J. Mark. Access Health Policy 2026, 14(1), 16; https://doi.org/10.3390/jmahp14010016 - 11 Mar 2026
Abstract
►▼
Show Figures
This study aimed to validate the health economic model for finerenone in the treatment of patients with heart failure (HF) and left ventricular ejection fraction (LVEF) ≥40% in the United Kingdom. A Markov model informed by the pivotal FINEARTS-HF trial compared finerenone +
[...] Read more.
This study aimed to validate the health economic model for finerenone in the treatment of patients with heart failure (HF) and left ventricular ejection fraction (LVEF) ≥40% in the United Kingdom. A Markov model informed by the pivotal FINEARTS-HF trial compared finerenone + standard of care (SoC) to SoC alone. Cross-validation was performed on the results (life years [LYs] and quality adjusted life years [QALYs]) for the SoC arm against three models in HF with LVEF >40%. External validation compared cardiovascular (CV) mortality and the number of total HF events (hospitalisation for heart failure [HFF] and urgent heart failure visit [UHFV]) against FINEARTS-HF. The model estimated similar discounted outcomes to other models in HF (6.47 vs. 6.63–7.91 LYs and 4.78 vs. 4.63–5.27 QALYs). CV deaths (22 vs. 27) and UHFV events (60 vs. 61) avoided with finerenone were similar between the model and FINEARTS-HF. The broad estimated range of avoided HHF events (205–303 vs. 219 in FINEARTS-HF) was largely driven by baseline patient age. This comprehensive validation exercise demonstrated that the finerenone model accurately estimated observed clinical data and was well aligned in its projections with previous models assessing similar populations.
Full article

Figure 1
Open AccessArticle
Addressing Unmet Medical Needs in Drug Development: Assessment and Implications for Regulatory and Clinical Development Strategies
by
Carla Domingo-Esteban, Inka Heikkinen and Nanco Hefting
J. Mark. Access Health Policy 2026, 14(1), 15; https://doi.org/10.3390/jmahp14010015 - 9 Mar 2026
Abstract
Unmet need is a core component of many Health Technology Assessment (HTA) processes at EU and national level. Most visibly, it is a core selection criterion for Joint Scientific Consultations (JSC) and Joint Clinical Assessment (JCA) for medical devices. This qualitative study explored
[...] Read more.
Unmet need is a core component of many Health Technology Assessment (HTA) processes at EU and national level. Most visibly, it is a core selection criterion for Joint Scientific Consultations (JSC) and Joint Clinical Assessment (JCA) for medical devices. This qualitative study explored how Unmet Medical Needs (UMNs) are understood and applied in drug development, with an emphasis on the European regulatory, HTA and access context, and examined their impact on regulatory and clinical development strategies. Twenty semi-structured interviews were conducted with representatives from regulatory authorities, HTA bodies, clinical development, industry, and patient insight roles. Data was analyzed using a thematic content approach combining deductive and inductive coding. Thematic analysis revealed general agreement on the importance of addressing UMNs, but also substantial variation in how they are defined and prioritized. Regulators often stressed disease severity and clinical evidence, while patients and clinicians emphasized quality of life. HTA representatives highlighted comparative benefit and long-term outcomes. These differing perspectives shaped how UMNs were integrated into development strategies, trial design, and regulatory planning. The findings indicate that clearer yet adaptable criteria could support earlier and more consistent alignment. Based on the analysis, a five-part roadmap to guide drug development is proposed, focusing on internal coordination, structured stakeholder engagement, collaboration between regulators and HTA bodies, adaptable definitions, and transparent decision-making. Together, these elements aim to support more systematic and predictable approaches to identifying and addressing unmet needs in drug development.
Full article
(This article belongs to the Collection European Health Technology Assessment (EU HTA))
►▼
Show Figures

Figure 1
Highly Accessed Articles
Latest Books
E-Mail Alert
News
Topics

Conferences
Special Issues
Special Issue in
JMAHP
Regenerative Medicine: From Bench to Bedside to Market
Guest Editors: Mondher Toumi, Steven SimoensDeadline: 15 December 2026
Special Issue in
JMAHP
Health Technology Assessment of Vaccination: Strategies, Public Health and Values: 2nd Edition
Guest Editors: Maarten J. Postma, Jurjen Van der Schans, Cornelis BoersmaDeadline: 28 February 2027
Topical Collections
Topical Collection in
JMAHP
European Health Technology Assessment (EU HTA)
Collection Editors: Jörg Ruof, Mondher Toumi







