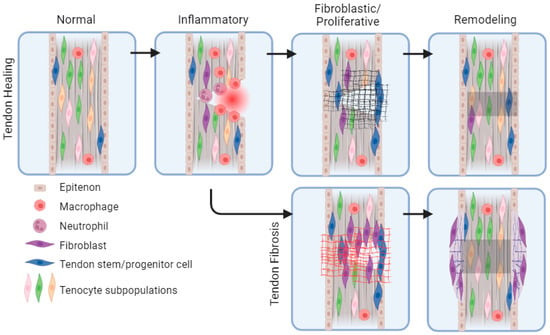
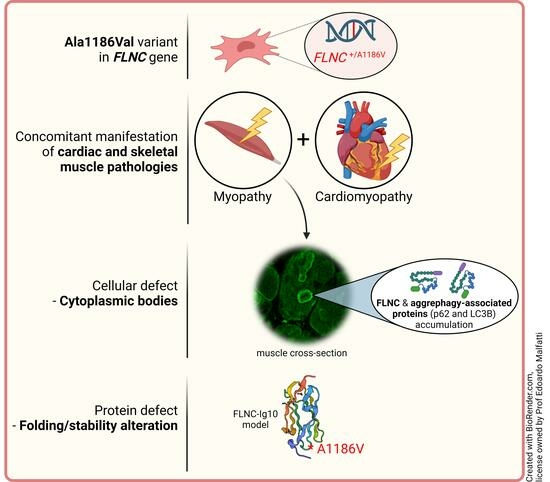
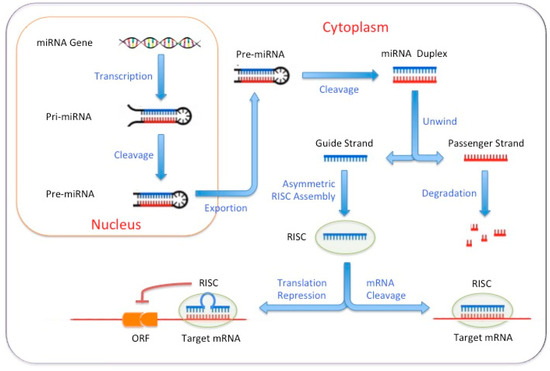
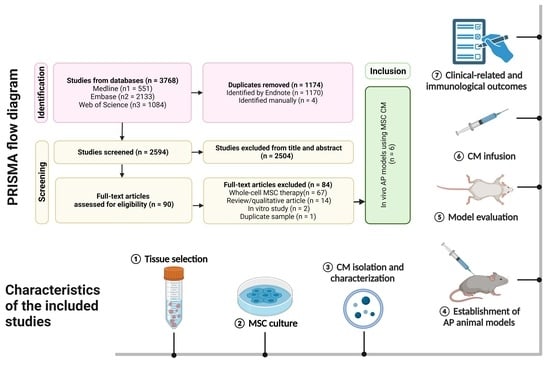
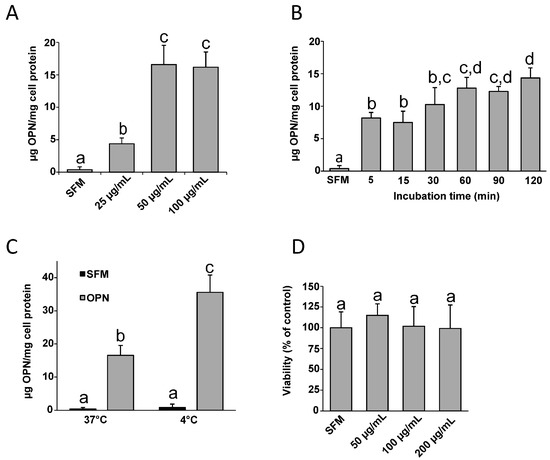
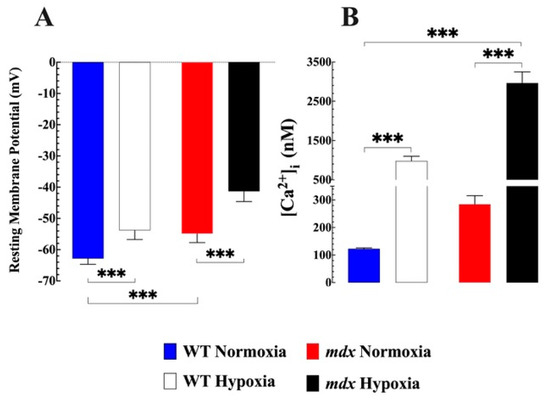
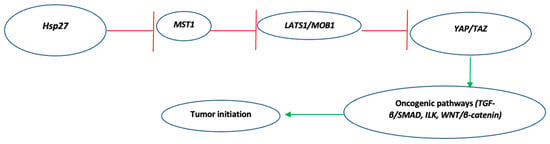
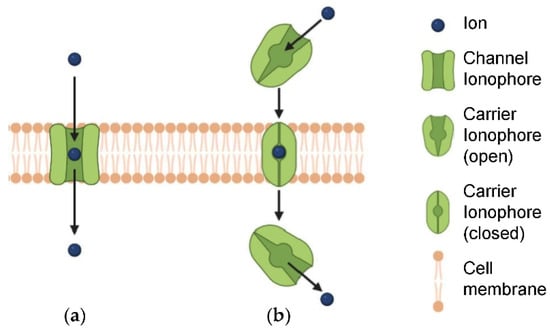
Feature Papers in Cell Biology and Pathology
A topical collection in Biomedicines (ISSN 2227-9059). This collection belongs to the section "Cell Biology and Pathology".
Viewed by 58366Editor
Interests: NMR-spectroscopy; protein structures; viral proteins; bacterial proteins; transcription; translation; allergens; nuclear magnetic resonance; crystallography; electron microscopy; fluorescence spectroscopy
Special Issues, Collections and Topics in MDPI journals
Topical Collection Information
Dear Colleagues,
The special edition “Feature Papers in Cell Biology and Pathology” will comprise important contributions by scholars in the field of cell biology and the Editorial Board members of the section Cell Biology and Pathology in Biomedicines. Their broad expertise will result in a comprehensive array of the latest findings in this field, and thus we encourage submissions of high-quality research papers or review articles.
Contributions that explore the origin of diseases at the molecular level, such as structural, biochemical, and genetic studies of receptors, oncogenes, tumor suppressor and regulatory proteins, signal pathways, and RNA/DNA, as well as articles that propose new routes for curing and preventing diseases, are of particular interest. Cutting-edge diagnostic tools ranging from PCR to magnetic resonance as applied to the detection of cellular or organismic pathological dysfunctions on all levels are also appropriate topics for this special edition. We look forward to your submissions on the above-listed research areas of cell biology and pathology.
Prof. Dr. Paul Rösch
Collection Editor
Manuscript Submission Information
Manuscripts should be submitted online at www.mdpi.com by registering and logging in to this website. Once you are registered, click here to go to the submission form. Manuscripts can be submitted until the deadline. All submissions that pass pre-check are peer-reviewed. Accepted papers will be published continuously in the journal (as soon as accepted) and will be listed together on the collection website. Research articles, review articles as well as short communications are invited. For planned papers, a title and short abstract (about 100 words) can be sent to the Editorial Office for announcement on this website.
Submitted manuscripts should not have been published previously, nor be under consideration for publication elsewhere (except conference proceedings papers). All manuscripts are thoroughly refereed through a single-blind peer-review process. A guide for authors and other relevant information for submission of manuscripts is available on the Instructions for Authors page. Biomedicines is an international peer-reviewed open access monthly journal published by MDPI.
Please visit the Instructions for Authors page before submitting a manuscript. The Article Processing Charge (APC) for publication in this open access journal is 2600 CHF (Swiss Francs). Submitted papers should be well formatted and use good English. Authors may use MDPI's English editing service prior to publication or during author revisions.