Assessment of the Antioxidant Activity of Lyophilized Cistus creticus Extract as a Functional Fortifier in Yogurt: A Cellular and Non-Cellular Evaluation
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Collection and Preparation of Samples
2.2.1. Collection and Characterization of Plant Material
2.2.2. Preparation of Plant Extracts
2.3. In Vitro Digestion
2.4. Biochemical Analysis
2.4.1. Determination of Total Antioxidant Capacity (TAC) Using Ferric Reducing Antioxidant Power Assay
2.4.2. Determination of Total Phenolic Content (TPC) Using Folin–Ciocalteau Method
2.5. Cellular Assays
2.5.1. Culture and Viability of the Caco-2 Cell Line
2.5.2. Cellular Antioxidant Activity (CAA) Assay
2.6. Statistical Analysis
3. Results and Discussion
3.1. Assessment of Antioxidant Activity of Samples Before and After In Vitro Digestion Using FRAP and Folin–Ciocalteau Biochemical Assays
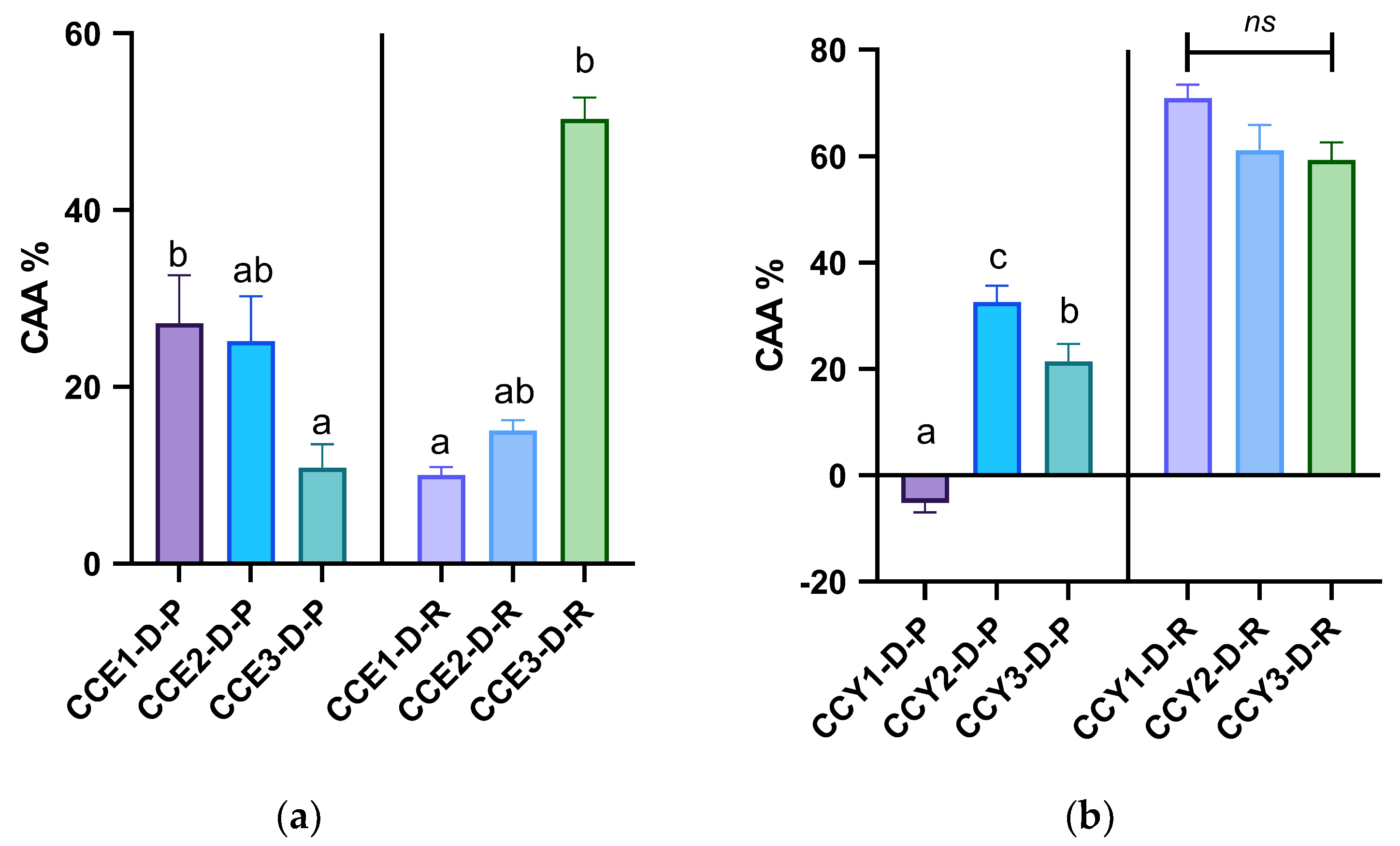
3.2. Assessment of Cellular Antioxidant Activity of Samples After In Vitro Digestion
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Civeyrel, L.; Leclercq, J.; Demoly, J.-P.; Agnan, Y.; Quèbre, N.; Pélissier, C.; Otto, T. Molecular Systematics, Character Evolution, and Pollen Morphology of Cistus and Halimium (Cistaceae). Plant Syst. Evol. 2011, 295, 23–54. [Google Scholar] [CrossRef]
- Guzmán, B.; Vargas, P. Systematics, Character Evolution, and Biogeography of Cistus L. (Cistaceae) Based on ITS, trnL-trnF, and matK Sequences. Mol. Phylogenet. Evol. 2005, 37, 644–660. [Google Scholar] [CrossRef]
- Palá-Paúl, J.; Velasco-Negueruela, A.; Pérez-Alonso, M.J.; Sanz, J. Seasonal Variation in Chemical Composition of Cistus albidus L. from Spain. J. Essent. Oil Res. 2005, 17, 19–22. [Google Scholar] [CrossRef]
- Tomou, E.-M.; Lytra, K.; Rallis, S.; Tzakos, A.G.; Skaltsa, H. An Updated Review of Genus Cistus L. since 2014: Traditional Uses, Phytochemistry, and Pharmacological Properties. Phytochem. Rev. 2022, 21, 2049–2087. [Google Scholar] [CrossRef]
- Raus de Baviera, D.; Ruiz-Canales, A.; Barrajón-Catalán, E. Cistus albidus L.—Review of a Traditional Mediterranean Medicinal Plant with Pharmacological Potential. Plants 2023, 12, 2988. [Google Scholar] [CrossRef]
- Attaguile, G.; Russo, A.; Campisi, A.; Savoca, F.; Acquaviva, R.; Ragusa, N.; Vanella, A. Antioxidant Activity and Protective Effect on DNA Cleavage of Extracts from Cistus incanus L. and Cistus monspeliensis L. Cell Biol. Toxicol. 2000, 16, 83–90. [Google Scholar] [CrossRef]
- Valenzuela, B.A.; Sanhueza, J.; Nieto, S. Natural Antioxidants in Functional Foods: From Food Safety to Health Benefits. Grasas Y Aceites 2003, 54, 295–303. [Google Scholar] [CrossRef]
- Wilson, D.W.; Nash, P.; Buttar, H.S.; Griffiths, K.; Singh, R.; De Meester, F.; Horiuchi, R.; Takahashi, T. The Role of Food Antioxidants, Benefits of Functional Foods, and Influence of Feeding Habits on the Health of the Older Person: An Overview. Antioxidants 2017, 6, 81. [Google Scholar] [CrossRef]
- Lobo, V.; Patil, A.; Phatak, A.; Chandra, N. Free Radicals, Antioxidants and Functional Foods: Impact on Human Health. Pharmacogn. Rev. 2010, 4, 118–126. [Google Scholar] [CrossRef] [PubMed]
- Shahidi, F.; Zhong, Y. Measurement of Antioxidant Activity. J. Funct. Foods 2015, 18, 757–781. [Google Scholar] [CrossRef]
- Antolovich, M.; Prenzler, P.D.; Patsalides, E.; McDonald, S.; Robards, K. Methods for Testing Antioxidant Activity. Analyst 2002, 127, 183–198. [Google Scholar] [CrossRef] [PubMed]
- Munteanu, I.G.; Apetrei, C. Analytical Methods Used in Determining Antioxidant Activity: A Review. Int. J. Mol. Sci. 2021, 22, 3380. [Google Scholar] [CrossRef]
- López-Alarcón, C.; Denicola, A. Evaluating the Antioxidant Capacity of Natural Products: A Review on Chemical and Cellular-Based Assays. Anal. Chim. Acta 2013, 763, 1–10. [Google Scholar] [CrossRef]
- Xu, B.; Dong, Q.; Yu, C.; Chen, H.; Zhao, Y.; Zhang, B.; Yu, P.; Chen, M. Advances in Research on the Activity Evaluation, Mechanism and Structure-Activity Relationships of Natural Antioxidant Peptides. Antioxidants 2024, 13, 479. [Google Scholar] [CrossRef]
- Wang, H.; Joseph, J.A. Quantifying Cellular Oxidative Stress by Dichlorofluorescein Assay Using Microplate Reader1. Free Radic. Biol. Med. 1999, 27, 612–616. [Google Scholar] [CrossRef]
- Wolfe, K.L.; Liu, R.H. Cellular Antioxidant Activity (CAA) Assay for Assessing Antioxidants, Foods, and Dietary Supplements. J. Agric. Food Chem. 2007, 55, 8896–8907. [Google Scholar] [CrossRef]
- Greene, L.A.; Tischler, A.S. Establishment of a Noradrenergic Clonal Line of Rat Adrenal Pheochromocytoma Cells Which Respond to Nerve Growth Factor. Proc. Natl. Acad. Sci. USA 1976, 73, 2424–2428. [Google Scholar] [CrossRef] [PubMed]
- Wilkening, S.; Stahl, F.; Bader, A. Comparison of Primary Human Hepatocytes and Hepatoma Cell Line Hepg2 with Regard to Their Biotransformation Properties. Drug Metab. Dispos. 2003, 31, 1035–1042. [Google Scholar] [CrossRef] [PubMed]
- Kellett, M.E.; Greenspan, P.; Pegg, R.B. Modification of the Cellular Antioxidant Activity (CAA) Assay to Study Phenolic Antioxidants in a Caco-2 Cell Line. Food Chem. 2018, 244, 359–363. [Google Scholar] [CrossRef]
- Saarela, M. (Ed.) Functional Foods; Woodhead Publishing: Sawston, UK, 2011; ISBN 978-1-84569-690-0. [Google Scholar]
- Durmus, N.; Capanoglu, E.; Kilic-Akyilmaz, M. Activity and Bioaccessibility of Antioxidants in Yoghurt Enriched with Black Mulberry as Affected by Fermentation and Stage of Fruit Addition. Int. Dairy. J. 2021, 117, 105018. [Google Scholar] [CrossRef]
- Jiménez, A.M.; Murcia, M.A.; Parras, P.; Martínez-Tomé, M. On the Importance of Adequately Choosing the Ingredients of Yoghurt and Enriched Milk for Their Antioxidant Activity. Int. J. Food Sci. Technol. 2008, 43, 1464–1473. [Google Scholar] [CrossRef]
- Guiné, R.P.F.; De Lemos, E.T. Development of New Dairy Products with Functional Ingredients. J. Culin. Sci. Technol. 2020, 18, 159–176. [Google Scholar] [CrossRef]
- Alenisan, M.A.; Alqattan, H.H.; Tolbah, L.S.; Shori, A.B. Antioxidant Properties of Dairy Products Fortified with Natural Additives: A Review. J. Assoc. Arab Univ. Basic Appl. Sci. 2017, 24, 101–106. [Google Scholar] [CrossRef]
- Thakur, N.; Dhaliwal, H.; Sharma, V. Chemical Composition, Minerals and Vitamins Analysis of Lyophilized Wheatgrass Juice Powder. Int. J. Emerg. Technol. 2019, 10, 137–144. [Google Scholar]
- ElNaker, N.A.; Daou, M.; Ochsenkühn, M.A.; Amin, S.A.; Yousef, A.F.; Yousef, L.F. A Metabolomics Approach to Evaluate the Effect of Lyophilization versus Oven Drying on the Chemical Composition of Plant Extracts. Sci. Rep. 2021, 11, 22679. [Google Scholar] [CrossRef] [PubMed]
- Sik, B.; Székelyhidi, R.; Lakatos, E.; Kapcsándi, V.; Ajtony, Z. Analytical Procedures for Determination of Phenolics Active Herbal Ingredients in Fortified Functional Foods: An Overview. Eur. Food Res. Technol. 2022, 248, 329–344. [Google Scholar] [CrossRef]
- Smith, T.J.; Karl, J.P.; Wilson, M.A.; Whitney, C.C.; Barrett, A.; Farhadi, N.F.; Chen, C.-Y.O.; Montain, S.J. Glycaemic Regulation, Appetite and Ex Vivo Oxidative Stress in Young Adults Following Consumption of High-Carbohydrate Cereal Bars Fortified with Polyphenol-Rich Berries. Br. J. Nutr. 2019, 121, 1026–1038. [Google Scholar] [CrossRef]
- Namal Senanayake, S.P.J. Green Tea Extract: Chemistry, Antioxidant Properties and Food Applications—A Review. J. Funct. Foods 2013, 5, 1529–1541. [Google Scholar] [CrossRef]
- Ahmed, M.; Ali, A.; Sarfraz, A.; Hong, Q.; Boran, H. Effect of Freeze-Drying on Apple Pomace and Pomegranate Peel Powders Used as a Source of Bioactive Ingredients for the Development of Functional Yogurt. J. Food Qual. 2022, 2022, 3327401. [Google Scholar] [CrossRef]
- Kapsokefalou, M.; Zhu, L.; Miller, D.D. Adding Iron to Green Tea May Decrease Its Antioxidant Capacity in Rats after an Oral Dose of the Mixture. Nutr. Res. 2006, 26, 480–485. [Google Scholar] [CrossRef]
- Benzie, I.F.F.; Strain, J.J. The Ferric Reducing Ability of Plasma (FRAP) as a Measure of “Antioxidant Power”: The FRAP Assay. Anal. Biochem. 1996, 239, 70–76. [Google Scholar] [CrossRef] [PubMed]
- Kupina, S.; Fields, C.; Roman, M.C.; Brunelle, S.L. Determination of Total Phenolic Content Using the Folin-C Assay: Single-Laboratory Validation, First Action 2017.13. J. AOAC Int. 2018, 101, 1466–1472. [Google Scholar] [CrossRef]
- Lind, K.F.; Hansen, E.; Østerud, B.; Eilertsen, K.-E.; Bayer, A.; Engqvist, M.; Leszczak, K.; Jørgensen, T.Ø.; Andersen, J.H. Antioxidant and Anti-Inflammatory Activities of Barettin. Mar. Drugs 2013, 11, 2655–2666. [Google Scholar] [CrossRef]
- Piccolomini, A.F.; Iskandar, M.M.; Lands, L.C.; Kubow, S. High Hydrostatic Pressure Pre-Treatment of Whey Proteins Enhances Whey Protein Hydrolysate Inhibition of Oxidative Stress and IL-8 Secretion in Intestinal Epithelial Cells. Food Nutr. Res. 2012, 56, 17549. [Google Scholar] [CrossRef]
- Feng, L.; Peng, F.; Wang, X.; Li, M.; Lei, H.; Xu, H. Identification and Characterization of Antioxidative Peptides Derived from Simulated in Vitro Gastrointestinal Digestion of Walnut Meal Proteins. Food Res. Int. 2019, 116, 518–526. [Google Scholar] [CrossRef]
- Templeton, G. A Two-Step Approach for Transforming Continuous Variables to Normal: Implications and Recommendations for IS Research. Commun. Assoc. Inf. Syst. 2011, 28, 41–58. [Google Scholar] [CrossRef]
- Cheli, F.; Baldi, A. Nutrition-Based Health: Cell-Based Bioassays for Food Antioxidant Activity Evaluation. J. Food Sci. 2011, 76, R197–R205. [Google Scholar] [CrossRef] [PubMed]
- Barrajón-Catalán, E.; Fernández-Arroyo, S.; Roldán, C.; Guillén, E.; Saura, D.; Segura-Carretero, A.; Micol, V. A Systematic Study of the Polyphenolic Composition of Aqueous Extracts Deriving from Several Cistus Genus Species: Evolutionary Relationship. Phytochem. Anal. 2011, 22, 303–312. [Google Scholar] [CrossRef] [PubMed]
- Riehle, P.; Vollmer, M.; Rohn, S. Phenolic Compounds in Cistus incanus Herbal Infusions—Antioxidant Capacity and Thermal Stability during the Brewing Process. Food Res. Int. 2013, 53, 891–899. [Google Scholar] [CrossRef]
- Kozłowska, M.; Ścibisz, I.; Przybył, J.L.; Laudy, A.E.; Majewska, E.; Tarnowska, K.; Małajowicz, J.; Ziarno, M. Antioxidant and Antibacterial Activity of Extracts from Selected Plant Material. Appl. Sci. 2022, 12, 9871. [Google Scholar] [CrossRef]
- Bernacka, K.; Bednarska, K.; Starzec, A.; Mazurek, S.; Fecka, I. Antioxidant and Antiglycation Effects of Cistus × Incanus Water Infusion, Its Phenolic Components, and Respective Metabolites. Molecules 2022, 27, 2432. [Google Scholar] [CrossRef]
- Ziarno, M.; Kozłowska, M.; Ścibisz, I.; Kowalczyk, M.; Pawelec, S.; Stochmal, A.; Szleszyński, B. The Effect of Selected Herbal Extracts on Lactic Acid Bacteria Activity. Appl. Sci. 2021, 11, 3898. [Google Scholar] [CrossRef]
- Gori, A.; Ferrini, F.; Marzano, M.C.; Tattini, M.; Centritto, M.; Baratto, M.C.; Pogni, R.; Brunetti, C. Characterisation and Antioxidant Activity of Crude Extract and Polyphenolic Rich Fractions from C. incanus Leaves. Int. J. Mol. Sci. 2016, 17, 1344. [Google Scholar] [CrossRef]
- Özkan, G.; Aras, A.; Güven, E.Ç. Investigating the Antioxidant Properties of Some Herbal Infusions During In Vitro Digestion. J. Apit. Nat. 2022, 5, 1–13. [Google Scholar] [CrossRef]
- Chen, G.-L.; Chen, S.-G.; Xie, Y.-Q.; Chen, F.; Zhao, Y.-Y.; Luo, C.-X.; Gao, Y.-Q. Total Phenolic, Flavonoid and Antioxidant Activity of 23 Edible Flowers Subjected to in Vitro Digestion. J. Funct. Foods 2015, 17, 243–259. [Google Scholar] [CrossRef]
- Costa, J.R.; Amorim, M.; Vilas-Boas, A.; Tonon, R.V.; Cabral, L.M.C.; Pastrana, L.; Pintado, M. Impact of in Vitro Gastrointestinal Digestion on the Chemical Composition, Bioactive Properties, and Cytotoxicity of Vitis vinifera L. cv. Syrah Grape Pomace Extract. Food Funct. 2019, 10, 1856–1869. [Google Scholar] [CrossRef] [PubMed]
- Tarko, T.; Duda-Chodak, A.; Zajac, N. Digestion and Absorption of Phenolic Compounds Assessed by in Vitro Simulation Methods. A Review. Rocz. Panstw. Zakl. Hig. 2013, 64, 79–84. [Google Scholar] [PubMed]
- İnan, Y.; Akyüz, S.; Kurt-Celep, I.; Celep, E.; Yesilada, E. Influence of In Vitro Human Digestion Simulation on the Phenolics Contents and Biological Activities of the Aqueous Extracts from Turkish Cistus Species. Molecules 2021, 26, 5322. [Google Scholar] [CrossRef]
- D’Archivio, M.; Filesi, C.; Di Benedetto, R.; Gargiulo, R.; Giovannini, C.; Masella, R. Polyphenols, Dietary Sources and Bioavailability. Ann. Ist. Super. Sanita 2007, 43, 348–361. [Google Scholar] [PubMed]
- Martinez-Gonzalez, A.I.; Díaz-Sánchez, Á.G.; Rosa, L.A.d.l.; Vargas-Requena, C.L.; Bustos-Jaimes, I.; Alvarez-Parrilla, A.E. Polyphenolic Compounds and Digestive Enzymes: In Vitro Non-Covalent Interactions. Molecules 2017, 22, 669. [Google Scholar] [CrossRef]
- Lavefve, L.; Howard, L.R.; Carbonero, F. Berry Polyphenols Metabolism and Impact on Human Gut Microbiota and Health. Food Funct. 2020, 11, 45–65. [Google Scholar] [CrossRef]
- Ketnawa, S.; Reginio, F.C.; Thuengtung, S.; Ogawa, Y. Changes in Bioactive Compounds and Antioxidant Activity of Plant-Based Foods by Gastrointestinal Digestion: A Review. Crit. Rev. Food Sci. Nutr. 2022, 62, 4684–4705. [Google Scholar] [CrossRef]
- Parada, J.; Aguilera, J.M. Food Microstructure Affects the Bioavailability of Several Nutrients. J. Food Sci. 2007, 72, R21–R32. [Google Scholar] [CrossRef]
- Saura-Calixto, F.; Serrano, J.; Goñi, I. Intake and Bioaccessibility of Total Polyphenols in a Whole Diet. Food Chem. 2007, 101, 492–501. [Google Scholar] [CrossRef]
- Rein, M.J.; Renouf, M.; Cruz-Hernandez, C.; Actis-Goretta, L.; Thakkar, S.K.; da Silva Pinto, M. Bioavailability of Bioactive Food Compounds: A Challenging Journey to Bioefficacy. Br. J. Clin. Pharmacol. 2013, 75, 588–602. [Google Scholar] [CrossRef] [PubMed]
- Saint-Eve, A.; Lévy, C.; Martin, N.; Souchon, I. Influence of Proteins on the Perception of Flavored Stirred Yogurts. J. Dairy Sci. 2006, 89, 922–933. [Google Scholar] [CrossRef]
- Helal, A.; Cattivelli, A.; Conte, A.; Tagliazucchi, D. In Vitro Bioaccessibility and Antioxidant Activity of Phenolic Compounds in Coffee-Fortified Yogurt. Molecules 2022, 27, 6843. [Google Scholar] [CrossRef]
- Muniandy, P.; Shori, A.B.; Hj Baba, A.S. Influence of Green, White and Black Tea Addition on the Antioxidant Activity of Probiotic Yogurt during Refrigerated Storage. Food Packag. Shelf Life 2016, 8, 1–8. [Google Scholar] [CrossRef]
- Khan, M.K.; Paniwnyk, L.; Hassan, S. Polyphenols as Natural Antioxidants: Sources, Extraction and Applications in Food, Cosmetics and Drugs. In Plant Based “Green Chemistry 2.0”: Moving from Evolutionary to Revolutionary; Li, Y., Chemat, F., Eds.; Springer: Singapore, 2019; pp. 197–235. ISBN 9789811338106. [Google Scholar]
- Meyer, A. The Problems of Using One-Dimensional Methods to Evaluate Multifunctional Food and Biological Antioxidants. J. Sci. Food Agric. 2000, 80, 1925–1941. [Google Scholar] [CrossRef]
- Haddad, M.J.; Sztupecki, W.; Delayre-Orthez, C.; Rhazi, L.; Barbezier, N.; Depeint, F.; Anton, P.M. Complexification of In Vitro Models of Intestinal Barriers, A True Challenge for a More Accurate Alternative Approach. Int. J. Mol. Sci. 2023, 24, 3595. [Google Scholar] [CrossRef]
- Karadag, A.; Ozcelik, B.; Saner, S. Review of Methods to Determine Antioxidant Capacities. Food Anal. Methods 2009, 2, 41–60. [Google Scholar] [CrossRef]
- Halliwell, B. Are Polyphenols Antioxidants or Pro-Oxidants? What Do We Learn from Cell Culture and in Vivo Studies? Arch. Biochem. Biophys. 2008, 476, 107–112. [Google Scholar] [CrossRef]
- Lambert, J.D.; Elias, R.J. The Antioxidant and Pro-Oxidant Activities of Green Tea Polyphenols: A Role in Cancer Prevention. Arch. Biochem. Biophys. 2010, 501, 65–72. [Google Scholar] [CrossRef]
- Aherne, S.A.; Kerry, J.P.; O’Brien, N.M. Effects of Plant Extracts on Antioxidant Status and Oxidant-Induced Stress in Caco-2 Cells. Br. J. Nutr. 2007, 97, 321–328. [Google Scholar] [CrossRef]
- Liu, S.; Huang, H. Assessments of Antioxidant Effect of Black Tea Extract and Its Rationals by Erythrocyte Haemolysis Assay, Plasma Oxidation Assay and Cellular Antioxidant Activity (CAA) Assay. J. Funct. Foods 2015, 18, 1095–1105. [Google Scholar] [CrossRef]
- Guo, R.; Chang, X.; Guo, X.; Brennan, C.S.; Li, T.; Fu, X.; Liu, R.H. Phenolic Compounds, Antioxidant Activity, Antiproliferative Activity and Bioaccessibility of Sea Buckthorn (Hippophaë rhamnoides L.) Berries as Affected by in Vitro Digestion. Food Funct. 2017, 8, 4229–4240. [Google Scholar] [CrossRef] [PubMed]
- Gutiérrez-Grijalva, E.P.; Antunes-Ricardo, M.; Acosta-Estrada, B.A.; Gutiérrez-Uribe, J.A.; Basilio Heredia, J. Cellular Antioxidant Activity and in Vitro Inhibition of α-Glucosidase, α-Amylase and Pancreatic Lipase of Oregano Polyphenols under Simulated Gastrointestinal Digestion. Food Res. Int. 2019, 116, 676–686. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Tao, W.; Huang, H.; Ye, X.; Sun, P. Flavonoids, Phenolic Acids, Carotenoids and Antioxidant Activity of Fresh Eating Citrus Fruits, Using the Coupled in Vitro Digestion and Human Intestinal HepG2 Cells Model. Food Chem. 2019, 279, 321–327. [Google Scholar] [CrossRef] [PubMed]
- Wolfe, K.L.; Kang, X.; He, X.; Dong, M.; Zhang, Q.; Liu, R.H. Cellular Antioxidant Activity of Common Fruits. J. Agric. Food Chem. 2008, 56, 8418–8426. [Google Scholar] [CrossRef]
- Song, W.; Derito, C.M.; Liu, M.K.; He, X.; Dong, M.; Liu, R.H. Cellular Antioxidant Activity of Common Vegetables. J. Agric. Food Chem. 2010, 58, 6621–6629. [Google Scholar] [CrossRef]
- Wang, L.; Chen, J.; Xie, H.; Ju, X.; Liu, R.H. Phytochemical Profiles and Antioxidant Activity of Adlay Varieties. J. Agric. Food Chem. 2013, 61, 5103–5113. [Google Scholar] [CrossRef]
- Corrochano, A.R.; Arranz, E.; De Noni, I.; Stuknytė, M.; Ferraretto, A.; Kelly, P.M.; Buckin, V.; Giblin, L. Intestinal Health Benefits of Bovine Whey Proteins after Simulated Gastrointestinal Digestion. J. Funct. Foods 2018, 49, 526–535. [Google Scholar] [CrossRef]
- Dalaka, E.; Politis, I.; Theodorou, G. Antioxidant Activity of Sweet Whey Derived from Bovine, Ovine and Caprine Milk Obtained from Various Small-Scale Cheese Plants in Greece before and after In Vitro Simulated Gastrointestinal Digestion. Antioxidants 2023, 12, 1676. [Google Scholar] [CrossRef]
- Mattson, M.P. Hormesis Defined. Ageing Res. Rev. 2008, 7, 1–7. [Google Scholar] [CrossRef]
- Andrés, C.M.C.; Pérez de la Lastra, J.M.; Juan, C.A.; Plou, F.J.; Pérez-Lebeña, E. Polyphenols as Antioxidant/Pro-Oxidant Compounds and Donors of Reducing Species: Relationship with Human Antioxidant Metabolism. Processes 2023, 11, 2771. [Google Scholar] [CrossRef]
- Kumar, S.; Pandey, A.K. Chemistry and Biological Activities of Flavonoids: An Overview. Sci. World J. 2013, 2013, 162750. [Google Scholar] [CrossRef]
- Aleman, R.S.; Marcia, J.; Page, R.; Kazemzadeh Pournaki, S.; Martín-Vertedor, D.; Manrique-Fernández, V.; Montero-Fernández, I.; Aryana, K. Effects of Yogurt with Carao (Cassia grandis) on Intestinal Barrier Dysfunction, α-Glycosidase Activity, Lipase Activity, Hypoglycemic Effect, and Antioxidant Activity. Fermentation 2023, 9, 566. [Google Scholar] [CrossRef]
- Aleman, R.S.; Page, R.; Cedillos, R.; Montero-Fernández, I.; Fuentes, J.A.M.; Olson, D.W.; Aryana, K. Influences of Yogurt with Functional Ingredients from Various Sources That Help Treat Leaky Gut on Intestinal Barrier Dysfunction in Caco-2 Cells. Pharmaceuticals 2023, 16, 1511. [Google Scholar] [CrossRef]
| No | Compound Name | Neutral Mass (Da) | Observed m/z (Da) | Mass Error (ppm) | Response | Formula |
|---|---|---|---|---|---|---|
| 1 | Myricitrin | 464.09548 | 463.0882 | 0.1 | 432,381 | C21H20O12 |
| 2 | Myricetin 3-galactoside | 480.09039 | 479.0833 | 0.3 | 237,079 | C21H20O13 |
| 3 | Gallocatechin | 306.07395 | 305.0664 | −0.9 | 197,753 | C15H14O7 |
| 4 | Quercitrin (Quercetin 3-O-rhamnoside) | 448.10056 | 447.093 | −0.6 | 130,032 | C21H20O11 |
| 5 | Prodelphinidin B * | 610.13226 | 609.1248 | −0.3 | 104,051 | C30H26O14 |
| 6 | Myricetin 3-arabinoside | 450.07983 | 449.0726 | 0.2 | 94,787 | C20H18O12 |
| 7 | Catechin | 290.07904 | 289.0718 | 0.0 | 57,668 | C15H14O6 |
| 8 | Quercetin 3-O-glucoside | 464.09548 | 463.088 | −0.4 | 42,322 | C21H20O12 |
| 9 | Rutin (Quercetin-3-rutinoside) | 610.15338 | 609.1463 | 0.3 | 37,512 | C27H30O16 |
| 10 | Guaijaverin (Quercetin 3-arabinoside) | 434.08491 | 433.0776 | −0.1 | 33,038 | C20H18O11 |
| 11 | Tiliroside * | 594.13734 | 593.1303 | 0.4 | 26,725 | C30H26O13 |
| 12 | Prodelphinidin B * | 610.13226 | 609.1248 | −0.4 | 13,886 | C30H26O14 |
| 13 | Tiliroside * | 594.13734 | 593.1314 | 2.2 | 13,192 | C30H26O13 |
| 14 | Quercetin 3-O-sophoroside | 626.1483 | 625.1415 | 0.7 | 12,606 | C27H30O17 |
| 15 | Prodelphinidin C * | 898.19564 | 897.1892 | 0.9 | 10,305 | C45H38O20 |
| 16 | Avicularin (Quercetin 3-arabinfuranoside) | 434.08491 | 433.078 | 0.7 | 9610 | C20H18O11 |
| 17 | Tiliroside * | 594.13734 | 593.1304 | 0.5 | 8128 | C30H26O13 |
| 18 | Prodelphinidin B * | 610.13226 | 609.1248 | −0.3 | 7044 | C30H26O14 |
| 19 | Prodelphinidin B * | 610.13226 | 609.1259 | 1.6 | 6198 | C30H26O14 |
| 20 | Luteolin-7-O-glucoside | 448.10056 | 447.0941 | 1.9 | 6055 | C21H20O11 |
| TAC (Fe2+/L) | TPC (mg GAE/g) | ||
|---|---|---|---|
| Before Digestion | CCE1 | 50.0 (8.6) a | 199.5 (11.8) a |
| CCE2 | 413.6 (13.1) b | 1278.3 (43.7) b | |
| CCE3 | 471.1 (23.7) c | 1446.2 (160.5) c | |
| After Digestion (<6 kDa) | CCE1-D-P | 46.1 (3.1) a | 58.9 (9.8) a |
| CCE2-D-P | 134.8 (26.8) b | 119.0 (8.3) b | |
| CCE3-D-P | 223.4 (36.0) c | 175.5 (9.6) c | |
| After Digestion (6–8 kDa) | CCE1-D-R | 56.5 (20.4) a | 75.4 (12.0) a |
| CCE2-D-R | 243.4 (9.1) b | 392.9 (25.9) b | |
| CCE3-D-R | 334.1 (39.6) c | 921.3 (86.9) c |
| TAC (Fe2+/L) | TPC (mg GAE/g) | ||
|---|---|---|---|
| After Digestion (<6 kDa) | CCY1-D-P | 4.5 (1.4) a | 16.9 (3.5) a |
| CCY2-D-P | 18.9 (5.7) b | 48.5 (13.4) b | |
| CCY3-D-P | 27.9 (5.2) c | 43.9 (2.4) b | |
| After Digestion (6–8 kDa) | CCY1-D-R | 9.2 (2.9) a | 34.6 (3.6) a |
| CCY2-D-R | 22.5 (8.3) b | 89.5 (4.3) b | |
| CCY3-D-R | 38.5 (3.3) c | 368.5 (34.8) c |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bousdouni, P.; Dalaka, E.; Kandyliari, A.; Gkalpinos, V.; Parisis, N.; Tzakos, A.G.; Theodorou, G.; Kapsokefalou, M.; Koutelidakis, A.E. Assessment of the Antioxidant Activity of Lyophilized Cistus creticus Extract as a Functional Fortifier in Yogurt: A Cellular and Non-Cellular Evaluation. Oxygen 2025, 5, 17. https://doi.org/10.3390/oxygen5030017
Bousdouni P, Dalaka E, Kandyliari A, Gkalpinos V, Parisis N, Tzakos AG, Theodorou G, Kapsokefalou M, Koutelidakis AE. Assessment of the Antioxidant Activity of Lyophilized Cistus creticus Extract as a Functional Fortifier in Yogurt: A Cellular and Non-Cellular Evaluation. Oxygen. 2025; 5(3):17. https://doi.org/10.3390/oxygen5030017
Chicago/Turabian StyleBousdouni, Panoraia, Eleni Dalaka, Aikaterini Kandyliari, Vasileios Gkalpinos, Nikolaos Parisis, Andreas G. Tzakos, Georgios Theodorou, Maria Kapsokefalou, and Antonios E. Koutelidakis. 2025. "Assessment of the Antioxidant Activity of Lyophilized Cistus creticus Extract as a Functional Fortifier in Yogurt: A Cellular and Non-Cellular Evaluation" Oxygen 5, no. 3: 17. https://doi.org/10.3390/oxygen5030017
APA StyleBousdouni, P., Dalaka, E., Kandyliari, A., Gkalpinos, V., Parisis, N., Tzakos, A. G., Theodorou, G., Kapsokefalou, M., & Koutelidakis, A. E. (2025). Assessment of the Antioxidant Activity of Lyophilized Cistus creticus Extract as a Functional Fortifier in Yogurt: A Cellular and Non-Cellular Evaluation. Oxygen, 5(3), 17. https://doi.org/10.3390/oxygen5030017










