Real-Life ISO 15189 Qualification of Long-Range Drone Transportation of Medical Biological Samples: Results from a Clinical Trial
Highlights
- The drone system enabled the 80 km transportation of clinical samples without damage.
- Freezing, refrigerated, and room temperatures were maintained during flights.
- The quality of pre-analytical conditions prevented clinical impact on the interpretation of the biological results.
- Long-range transportation by drone is in accordance with the ISO 15189 standard.
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population and Biological Clinical Samples
2.2. Pre-Analytical Phase and Flight Data
2.3. Analytical Phase—Clinical Laboratory Methods
2.4. Statistical Analysis and Clinical Interpretation
2.5. Institutional Review Board Statement
3. Results
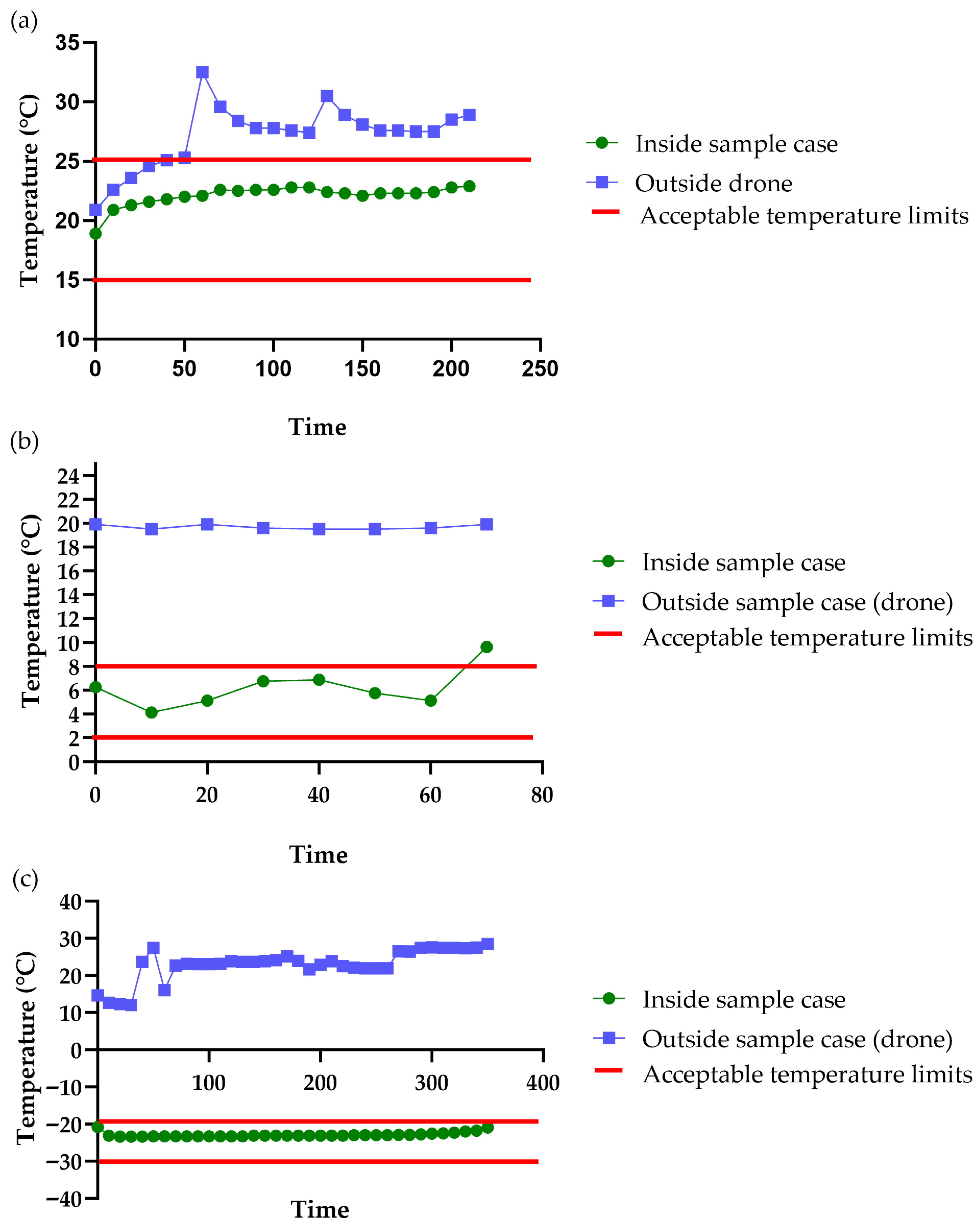
3.1. Conduction of Trial UAV Flights
3.2. TRANS-AIRGHT: Clinical Trial on Healthy Volunteers
3.3. PATH-AIRGHT: Observational Study on Pathological Samples
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ALP | Alkaline Phosphatase |
| ALT | Alanine Transaminase |
| APUHC | Amiens-Picardie University Hospital Center |
| AST | Aspartate Transferase |
| CRP | C-Reactive Protein |
| GGT | Gamma-Glutamyl Transferase |
| HBC | Human Biology Center |
| IQR | Inter-Quartile Range |
| ISO | International Organization for Standardization |
| LDH | Lactate Dehydrogenase |
| MDHC | Montreuil District Hospital Center |
| TSH | Thyroid-Stimulating Hormone |
| UAV | Unmanned Aerial Vehicles |
References
- ISO 15189:2022; Medical Laboratories—Requirements for Quality and Competence. International Organization for Standardization: Vernier, Switzerland, 2022.
- Vermeersch, P.; Frans, G.; von Meyer, A.; Costelloe, S.; Lippi, G.; Simundic, A.-M. How to Meet ISO15189:2012 Pre-Analytical Requirements in Clinical Laboratories? A Consensus Document by the EFLM WG-PRE. Clin. Chem. Lab. Med. 2021, 59, 1047–1061. [Google Scholar] [CrossRef] [PubMed]
- Bonnefont-Rousselot, D.; Delpech, M.; Chatron, P.; Gueant, J.-L.; Le Bouc, Y.; Maquart, F.-X.; Massoubre, B.; Rives, N.; Vigneron, C. Rapport 22-12. La Biologie Médicale En France: Évolutions et Enjeux. Bull. L’Acad. Natl. Méd. 2023, 207, 3–15. [Google Scholar] [CrossRef]
- Zaninotto, M.; Tasinato, A.; Vecchiato, G.; Legnaro, A.; Pinato, A.; Plebani, M. Performance Specifications in Extra-Analytical Phase of Laboratory Testing: Sample Handling and Transportation. Clin. Biochem. 2017, 50, 574–578. [Google Scholar] [CrossRef] [PubMed]
- Lippi, G.; Mattiuzzi, C. Biological Samples Transportation by Drones: Ready for Prime Time? Ann. Transl. Med. 2016, 4, 92. [Google Scholar] [CrossRef]
- De Silvestri, S.; Capasso, P.J.; Gargiulo, A.; Molinari, S.; Sanna, A. Challenges for the Routine Application of Drones in Healthcare: A Scoping Review. Drones 2023, 7, 685. [Google Scholar] [CrossRef]
- Kreier, F. Drones Bearing Parcels Deliver Big Carbon Savings. Nature 2022. [Google Scholar] [CrossRef]
- Nisingizwe, M.P.; Ndishimye, P.; Swaibu, K.; Nshimiyimana, L.; Karame, P.; Dushimiyimana, V.; Musabyimana, J.P.; Musanabaganwa, C.; Nsanzimana, S.; Law, M.R. Effect of Unmanned Aerial Vehicle (Drone) Delivery on Blood Product Delivery Time and Wastage in Rwanda: A Retrospective, Cross-Sectional Study and Time Series Analysis. Lancet Glob. Health 2022, 10, e564–e569. [Google Scholar] [CrossRef]
- Kremer, P.; Haruna, F.; Tuffour Sarpong, R.; Agamah, D.; Billy, J.; Osei-Kwakye, K.; Aidoo, P.; Dodoo, D.; Okoh-Owusu, M. An Impact Assessment of the Use of Aerial Logistics to Improve Access to Vaccines in the Western-North Region of Ghana. Vaccine 2023, 41, 5245–5252. [Google Scholar] [CrossRef]
- Kremer, P.; Haruna, F.; Briegleb, C.; Amoah, M.E.; Oteng, K.F.; Boadi, S.; Dwomoh, S.; Bentil, A.; Tinkorang, E. A Mixed Method Impact Assessment of the Use of Aerial Logistics to Improve Maternal Health and Emergencies Outcomes in the Ashanti Region of Ghana. BMC Health Serv. Res. 2025, 25, 390. [Google Scholar] [CrossRef]
- Oakey, A.; Waters, T.; Zhu, W.; Royall, P.G.; Cherrett, T.; Courtney, P.; Majoe, D.; Jelev, N.; Oakey, A.; Waters, T.; et al. Quantifying the Effects of Vibration on Medicines in Transit Caused by Fixed-Wing and Multi-Copter Drones. Drones 2021, 5, 22. [Google Scholar] [CrossRef]
- Amukele, T.K.; Sokoll, L.J.; Pepper, D.; Howard, D.P.; Street, J. Can Unmanned Aerial Systems (Drones) Be Used for the Routine Transport of Chemistry, Hematology, and Coagulation Laboratory Specimens? PLoS ONE 2015, 10, e0134020. [Google Scholar] [CrossRef] [PubMed]
- Amukele, T.K.; Street, J.; Carroll, K.; Miller, H.; Zhang, S.X. Drone Transport of Microbes in Blood and Sputum Laboratory Specimens. J. Clin. Microbiol. 2016, 54, 2622–2625. [Google Scholar] [CrossRef] [PubMed]
- Amukele, T.K.; Hernandez, J.; Snozek, C.L.; Wyatt, R.G.; Douglas, M.; Amini, R.; Street, J. Drone Transport of Chemistry and Hematology Samples Over Long Distances. Am. J. Clin. Pathol. 2017, 148, 427–435. [Google Scholar] [CrossRef] [PubMed]
- Stierlin, N.; Hemmerle, A.; Renz, H.; Risch, L.; Risch, M. Stability of Hemolytic, Lipemic, and Icteric Indices in Blood Samples Transported by Drone: A Focused Report. J. Appl. Lab. Med. 2025, 10, 704–709. [Google Scholar] [CrossRef]
- Weekx, S.; Lint, P.V.; Jacobs, S. The Effects of Drone Transportation on Routine Laboratory, Immunohematology, Flow Cytometry and Molecular Analyses. Clin. Chem. Lab. Med. 2025, 63, 311–319. [Google Scholar] [CrossRef]
- Shapira, M.; Cohen, B.; Friemann, S.; Tal, Y.; Teper, Z.; Dudkiewicz, M.; Portuguese, S.; Na’amnih, W.; Shriki, D.D. The Impact of Clinical Sample Transportation by Unmanned Aerial Systems on the Results of Laboratory Tests. Drones 2025, 9, 179. [Google Scholar] [CrossRef]
- Testa, S.; Meijer, P.; Lasne, D.; Mullier, F. Implementation of the New EUR IVD Regulation and Relation with ISO15189 Accreditation: Guidance Is Urgently Required for Haemostasis Testing. Int. J. Lab. Hematol. 2022, 44, 71–78. [Google Scholar] [CrossRef]
- Fraser, C.G.; Petersen, P.H.; Ricos, C.; Haeckel, R. Proposed Quality Specifications for the Imprecision and Inaccuracy of Analytical Systems for Clinical Chemistry. Eur. J. Clin. Chem. Clin. Biochem. 1992, 30, 311–317. [Google Scholar]
- EFLM Biological Variation. Available online: https://biologicalvariation.eu/ (accessed on 12 January 2026).
- Homier, V.; Brouard, D.; Nolan, M.; Roy, M.-A.; Pelletier, P.; McDonald, M.; de Champlain, F.; Khalil, E.; Grou-Boileau, F.; Fleet, R. Drone versus Ground Delivery of Simulated Blood Products to an Urban Trauma Center: The Montreal Medi-Drone Pilot Study. J. Trauma. Acute Care Surg. 2021, 90, 515–521. [Google Scholar] [CrossRef]
- Callewaert, N.; Pareyn, I.; Acke, T.; Desplinter, B.; Van de Pitte, K.; Van Vooren, J.; De Meyer, M.; Seeldraeyers, E.; Peeters, F.; De Meyer, S.F.; et al. Limited Impact of Drone Transport of Blood on Platelet Activation. Drones 2024, 8, 752. [Google Scholar] [CrossRef]
- Kooli, C. Ethical and Safety Implications of Medical Emergency Landing Exploitations: A Call for Policy Action. Glob. J. Qual. Saf. Healthc. 2025, 8, 89–92. [Google Scholar] [CrossRef] [PubMed]
- Johannessen, K.A.; Wear, N.K.S.; Toska, K.; Hansbo, M.; Berg, J.P.; Fosse, E. Pathologic Blood Samples Tolerate Exposure to Vibration and High Turbulence in Simulated Drone Flights, but Plasma Samples Should Be Centrifuged After Flight. IEEE J. Transl. Eng. Health Med. 2021, 9, 4000110. [Google Scholar] [CrossRef] [PubMed]
- Wiltshire, M.; Boxshall, J.; Milne, J.; Oleniacz, K.; Theobald, K.; Phillips, B. The Effects of Drone Transportation on Blood Component Quality: A Prospective Randomised Controlled Laboratory Study. Br. J. Haematol. 2024, 205, 2022–2030. [Google Scholar] [CrossRef] [PubMed]
- Kapoula, G.V.; Kontou, P.I.; Bagos, P.G. The Impact of Pneumatic Tube System on Routine Laboratory Parameters: A Systematic Review and Meta-Analysis. Clin. Chem. Lab. Med. 2017, 55, 1834–1844. [Google Scholar] [CrossRef]
- Le Quellec, S.; Paris, M.; Nougier, C.; Sobas, F.; Rugeri, L.; Girard, S.; Bordet, J.-C.; Négrier, C.; Dargaud, Y. Pre-Analytical Effects of Pneumatic Tube System Transport on Routine Haematology and Coagulation Tests, Global Coagulation Assays and Platelet Function Assays. Thromb. Res. 2017, 153, 7–13. [Google Scholar] [CrossRef]
- Cofrac. Comité Français d’Accréditation. Available online: https://tools.cofrac.fr/fr/documentation/index.php?fol_id=63 (accessed on 12 November 2025).
- Ongaro, G.; Marti, C.; Perrier, A.; Righini, M. D-dimères et suspicion d’embolie pulmonaire: Seuil adapté à l’âge. Rev. Med. Suisse 2014, 446, 1908–1912. [Google Scholar] [CrossRef]
- Heireman, L.; Van Geel, P.; Musger, L.; Heylen, E.; Uyttenbroeck, W.; Mahieu, B. Causes, Consequences and Management of Sample Hemolysis in the Clinical Laboratory. Clin. Biochem. 2017, 50, 1317–1322. [Google Scholar] [CrossRef]
- Farhana, A.; Lappin, S.L. Biochemistry, Lactate Dehydrogenase. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2025. [Google Scholar]
- Minović, I.; Dikkeschei, L.D.; Vos, M.J.; Kootstra-Ros, K.-R. Interpretation of Folate Results in Hemolytic Plasma Samples: A Practical Approach. Ann. Lab. Med. 2021, 41, 485–488. [Google Scholar] [CrossRef]
- Perlee, D.; van der Steege, K.H.; den Besten, G. The Effect of Drone Transport on the Stability of Biochemical, Coagulation and Hematological Parameters in Healthy Individuals. Clin. Chem. Lab. Med. 2021, 59, 1772–1776. [Google Scholar] [CrossRef]
- Mikesh, L.M.; Bruns, D.E. Stabilization of Glucose in Blood Specimens: Mechanism of Delay in Fluoride Inhibition of Glycolysis. Clin. Chem. 2008, 54, 930–932. [Google Scholar] [CrossRef]
- Lahmer, M.; Saddari, A.; Benaissa, E.; Ben Lahlou, Y.; Elouennass, M.; Maleb, A.; Beyyoudh, S. Non-Conformités de La Phase Préanalytique de l’examen Cytobactériologique Des Urines (ECBU) Au Laboratoire de Microbiologie Du CHU Mohammed VI d’Oujda. Tunis. Med. 2023, 101, 612–616. [Google Scholar] [PubMed]
- Fleming, C.; Russcher, H.; Lindemans, J.; de Jonge, R. Clinical Relevance and Contemporary Methods for Counting Blood Cells in Body Fluids Suspected of Inflammatory Disease. Clin. Chem. Lab. Med. 2015, 53, 1689–1706. [Google Scholar] [CrossRef] [PubMed]
- Zhu, W.; Oakey, A.; Royall, P.G.; Waters, T.P.; Cherrett, T.; Theobald, K.; Bester, A.-M.; Lucas, R. Investigating the Influence of Drone Flight on the Stability of Cancer Medicines. PLoS ONE 2023, 18, e0278873. [Google Scholar] [CrossRef] [PubMed]
- Enjoji, T.; Soyama, A.; Fukumoto, M.; Peilin, L.; Matsuguma, K.; Imamura, H.; Maruya, Y.; Hara, T.; Matsushima, H.; Kugiyama, T.; et al. Feasibility of Organ Transportation by a Drone: An Experimental Study Using a Rat Model. Transplant. Proc. 2023, 55, 2227–2231. [Google Scholar] [CrossRef]




| Car | Drone | p a | Car | Drone | p a | Car | Drone | p a | |||
|---|---|---|---|---|---|---|---|---|---|---|---|
| n = 30 | n = 30 | n = 30 | n = 30 | n = 30 | n = 30 | ||||||
| Pre-analytical delay (minutes), median [IQR] | 166.5 [135.5–183.2] | 173.0 [159.7–218.2] | 0.1 b | ||||||||
| Biochemistry markers | Blood count variables | Coagulation variables | |||||||||
| Positive plasma hemolysis index, n (%) | 0 (0%) | 0 (0%) | 1 c | White blood cells (G/L), mean (SD) | 6.7 (1.8) | 6.7 (1.8) | 0.6 | Prothrombine Time (%), mean (SD) | 97.5 (4.9) | 97.7 (4.7) | 0.5 |
| Positive serum hemolysis index, n (%) | 0 (0%) | 1 (3.33%) | 0.31 c | Neutrophils (G/L), mean (SD) | 4.0 (1.3) | 4.0 (1.3) | 0.09 | Partial Thromboplastin Time (ratio), mean (SD) | 1.03 (0.09) | 1.03 (0.07) | 0.3 |
| Glucose heparinate (mmol/L), median [IQR] | 4.00 [3.75–4.42] | 3.8 [3.57–4.42] | 0.04 | Lymphocytes (G/L), mean (SD) | 2.0 (0.6) | 1.9 (0.6) | 0.4 | Fibrinogen (g/L), mean (SD) | 3.0 (0.6) | 3.06 (0.7) | 0.2 |
| Glucose fluoride (mmol/L), mean (SD) | 4.85 (0.54) | 4.81 (0.56) | 0.09 | Monocytes (G/L), mean (SD) | 0.5 (0.1) | 0.5 (0.6) | 0.2 | Factor V (%), mean (SD) | 114.0 (17.0) | 114.6 (17.9) | 0.4 |
| Creatinine (µmol/L), mean (SD) | 72.13 (11.49) | 72.27 (11.83) | 0.7 | Eosinophils, (G/L), mean (SD) | 0.2 (0.1) | 0.2 (0.1) | 0.7 b | D-Dimers (µg/mL), mean (SD) | 0.14 (0.17) | 0.17 (0.20) | 0.1 |
| Potassium (mmol/L), mean (SD) | 3.75 (0.35) | 3.73 (0.35) | 0.3 | Red blood cells (G/L), mean (SD) | 4.7 (0.3) | 4.7 (0.3) | 0.3 | Antithrombin (%), mean (SD) | 110.7 (10.3) | 110.9 (9.5) | 0.8 |
| Sodium (mmol/L), mean (SD) | 140.8 (1.45) | 140.3 (1.54) | 0.01 | Hemoglobin (g/dL), mean (SD) | 14.2 (1.2) | 14.2 (1.2) | 0.7 | ||||
| Calcium (mmol/L), mean (SD) | 2.45 (0.06) | 2.45 (0.07) | 0.9 | Hematocrit (%), mean (SD) | 42.2 (3.1) | 42.4 (3.1) | 7 | ||||
| Phosphor (mmol/L), mean (SD) | 0.96 (0.13) | 0.95 (0.14) | 0.2 | Mean corpuscular volume (fL), mean (SD) | 90.8 (4.5) | 90.8 (4.4) | 0.8 | ||||
| Total CO2 (mmol/L), mean (SD) | 27.8 (2.0) | 28.0 (2.1) | 0.3 | Mean corpuscular hemoglobin (pg), mean (SD) | 30.5 (2.0) | 30.4 (1.9) | 0.6 | ||||
| LDH (U/L), mean (SD) | 249.9 † (49.7) | 250.3 † (34.6) | 1 | Mean corpuscular hemoglobin concentration (g/dL), mean (SD) | 33.6 (1.2) | 33.5 (1.2) | 0.4 | ||||
| Urea (mmol/L), mean (SD) | 4.77 (1.28) | 4.81 (1.35) | 0.4 | Red cell Distribution Width (%), mean (SD) | 13.2 (1.3) | 13.2 (1.3) | 0.3 | ||||
| Total bilirubin (mmol/L), mean (SD) | 10.5 (4.0) | 10.5 (4.1) | 0.7 | Platelets (G/L), mean (SD) | 255.3 (63.0) | 258.8 (62.7) | 0.03 | ||||
| ALP (U/L), mean (SD) | 74.6 (15.8) | 74.5 (15.8) | 0.9 | Mean Platelet Volume (fL), mean (SD) | 11.1 † (0.8) | 11.1 † (0.8) | 0.3 | ||||
| ALT (U/L), mean (SD) | 23.7 (10.8) | 23.4 (11.2) | 0.4 | ||||||||
| AST (U/L), mean (SD) | 22.0 (8.5) | 22.0 (8.9) | 0.9 | ||||||||
| GGT (U/L), mean (SD) | 23.9 (10.3) | 24.0 (10.5) | 0.6 | ||||||||
| Folate * (ng/mL), mean (SD) | 9.75 (4.33) | 9.03 (4.04) | 0.02 | ||||||||
| Protein (g/L), mean (SD) | 76.7 (3.54) | 76.7 (3.40) | 0.8 | ||||||||
| n = | No Transport | Drone | p a | |
|---|---|---|---|---|
| Pre-analytical delay (minutes), median [IQR] | 126 | NA | 287.0 [219.3–372.5] | |
| Biochemistry blood markers | ||||
| Positive plasma hemolysis index, n (%) | 22 | 2 (9%) | 3 (14%) | 0.6 b |
| Positive serum hemolysis index, n (%) | 24 | 1 (4%) | 0 (0%) | 0.3 b |
| Glucose heparinate (mmol/L), median [IQR] | 8 | 5.3 [3.9–5.6] | 5.0 [3.9–6.75] | 0.1 |
| Creatinine (µmol/L), median [IQR] | 20 | 86.5 [55.5–145.0] | 86 [55.5–147.0] | 0.4 |
| Potassium (mmol/L), median [IQR] | 18 | 3.77 [3.39–4.16] | 3.71 [3.32–4.14] | 0.4 |
| Sodium (mmol/L), median [IQR] | 23 | 140.0 [139.0–142.0] | 141.0 [141.0–142.0] | <0.001 |
| Calcium (mmol/L), median [IQR] | 17 | 2.36 [2.24–2.50] | 2.36 [2.26–2.50] | 0.5 |
| Phosphor (mmol/L), median [IQR] | 22 | 1.06 [0.86–1.26] | 1.03 [0.85–1.29] | 0.7 |
| Total CO2 mmol/L, median [IQR] | 23 | 27.0 [25.0–28.0] | 26.0 [25.0–28.0] | 0.2 |
| Urea (mmol/L), median [IQR] | 23 | 9.9 [4.3–11.1] | 9.7 [4.6–10.6] | 0.06 |
| Total bilirubin (mmol/L), median [IQR] | 15 | 8.0 [8.0–11.0] | 8.0 [8.0–11.0] | 1 |
| ALP (U/L), median [IQR] | 15 | 105.0 [63.0–189.0] | 103.0 [65.0–190.0] | 0.5 |
| ALT (U/L), median [IQR] | 14 | 21.0 [17.0–26.0] | 21.5 [17.0–33.2] | 0.01 |
| AST (U/L), median [IQR] | 14 | 22.0 [18.0–40.0] | 25.0 [15.5–39.0] | 0.9 |
| GGT (U/L), median [IQR] | 14 | 25.0 [18.0–76.2] | 37.0 [18.0–77.0] | 0.01 |
| Folate (ng/mL), median [IQR] | 11 | 6.2 [3.0–8.0] | 5.8 [2.2–10.3] | 0.1 |
| Protein (g/L), median [IQR] | 23 | 68.0 [61.0–71.0] | 67.0 [60.0–70.0] | 0.2 |
| Total cholesterol (mmol/L), median [IQR] | 8 | 5.53 [3.93–6.85] | 5.55 [3.97–6.92] | 0.5 |
| HDL cholesterol (mmol/L), median [IQR] | 8 | 1.17 [0.72–1.30] | 1.17 [0.72–1.29] | 0.9 |
| LDL cholesterol (mmol/L), median [IQR] | 8 | 3.69 [2.38–4.88] | 3.66 [2.45–4.87] | 0.9 |
| Triglycerides (mmol/L), median [IQR] | 8 | 1.67 [1.41–2.34] | 1.68 [1.38–2.33] | 0.2 |
| TSH (mUI/L), median [IQR] | 8 | 1.83 [1.07–3.37] | 1.83 [1.05–3.51] | 0.4 |
| Triiodothyronin (pmol/L), median [IQR] | 5 | 5.79 [4.73–7.39] | 5.60 [4.78–7.55] | 0.6 |
| Thyroxine (ng/dL), median [IQR] | 6 | 1.11 [0.98–1.27] | 1.08 [1.0.3–1.21] | 0.6 |
| Vitamin B12 (pg/mL), median [IQR] | 9 | 520.0 [383.5–579.0] | 421.0 [354.0–522.0] | 0.07 |
| 25OH Vitamin D (ngl/mL), median [IQR] | 5 | 43.4 [20.6–57.2] | 46.5 [25.1–58.9] | 0.06 |
| Iron (µmol/L), median [IQR] | 6 | 16.8 [12.7–24.3] | 18.0 [13.5–24.8] | 0.06 |
| Transferrin (µmol/L), median [IQR] | 5 | 2.30 [1.76–2.64] | 2.36 [1.79–2.68] | 0.25 |
| CRP (mg/mL), median [IQR] | 9 | 0.24 [0.00–23.0] | 1.3 [0.25–22.4] | 0.9 |
| Albumin (g/L), median [IQR] | 7 | 33.0 [26.0–41.0] | 33.0 [27.0–41.0] | 0.5 |
| Blood count variables | ||||
| White blood cells (G/L), median [IQR] | 26 | 6.9 [4.8–9.0] | 6.4 [4.9–8.6] | 0.6 |
| Neutrophils (G/L), median [IQR] | 21 | 3.8 [3.2–5.8] | 3.7 [2.8–5.0] | 0.2 |
| Lymphocytes (G/L), median [IQR] | 21 | 1.3 [0.8–2.0] | 1.4 [0.8–2.0] | 0.5 |
| Red blood cells (G/L), median [IQR] | 26 | 3.9 [3.4–4.6] | 3.9 [3.3–4.6] | 0.8 |
| Hemoglobin (g/dL), median [IQR] | 26 | 12.0 [10.2–13.2] | 12.0 [10.1–13.3] | 0.4 |
| Hematocrit (%), median [IQR] | 26 | 37.3 [31.3–40.0] | 37.3 [31.2–41.0] | 7 |
| Platelets (G/L), median [IQR] | 26 | 217.0 [181.0–303.5] | 220.5 [163.0–310.0] | 0.1 |
| Urinalysis variables | ||||
| Potassium (mmol/L), median [IQR] | 17 | 22.7 [18.8–39.1] | 23.2 [19.0–36.4] | 0.4 |
| Sodium (mmol/L), median [IQR] | 17 | 65.3 [40.0–60.6] | 65.6 [38.9–93.3] | 0.7 |
| Creatinine (µmol/L), median [IQR] | 13 | 4.3 [2.7–8.8] | 4.4 [2.7–14.5] | 0.7 |
| Urea (mmol/L), median [IQR] | 15 | 164.4 [96.9–283.8] | 174.3 [116.4–278.4] | 0.8 |
| Protein (mg/L), median [IQR] | 12 | 141.0 [109.0–310.0] | 86.0 [60.5–325.5] | 0.3 |
| Erythrocytes (103/mL), median [IQR] | 17 | 112.6 [3.15–621.8] | 13.9 [3.3–586.5] | 0.8 |
| Leucocytes (103/mL), median [IQR] | 17 | 5.8 [1.65–51.85] | 5.2 [1.3–45.6] | 0.8 |
| Hepatitis B serology variables | ||||
| Hbs antigen (index), median [IQR] (positivity threshold = 1) | 6 | 0.49 [0.46–0.50] | 0.50 [0.45–0.54] | 0.9 |
| Anti-Hbs antibodies (mUI/mL), median [IQR] | 6 | 198.3 [0.00–541.2] | 190.1 [0.00–534.4] | 0.4 |
| Anti-Hbc antibodies (index), median [IQR] (positivity threshold = 1) | 6 | 0.31 [0.15–0.43] | 0.19 [0.15–0.22] | 0.2 |
| n = | No Transport | Drone | |
|---|---|---|---|
| Plasma cytomegalovirus DNA | 4 | DNQ (20.6–34.5 UI/mL) | DNQ (20.6–34.5 UI/mL) |
| DNQ (20.6–34.5 UI/mL) | DNQ (20.6–34.5 UI/mL) | ||
| 630 UI/mL | 631 UI/mL | ||
| DNQ (20.6–34.5 UI/mL) | DNQ (20.6–34.5 UI/mL) | ||
| Cervical human papillomavirus DNA | 5 | 33.25 Ct | 33.4 Ct |
| 27.53 Ct | 29.3 Ct | ||
| 18.89 Ct | 19.65 Ct | ||
| 27.00 Ct | 27.51 Ct | ||
| 29.35 Ct | 27.53 Ct | ||
| Influenza A virus RNA (swab) | 1 | 27.89 Ct | 30.46 Ct |
| SARS-CoV-2 RNA (swab) | 3 | 18.3 Ct | 18.27 Ct |
| 33.99 Ct | 33.66 Ct | ||
| 14.65 Ct | 15.77 Ct |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Demey, B.; Bury, O.; Choquet, M.; Fontaine, J.; Dollerschell, M.; Thorel, H.; Durand-Maugard, C.; Leroy, O.; Pecquet, M.; Voyer, A.; et al. Real-Life ISO 15189 Qualification of Long-Range Drone Transportation of Medical Biological Samples: Results from a Clinical Trial. Drones 2026, 10, 71. https://doi.org/10.3390/drones10010071
Demey B, Bury O, Choquet M, Fontaine J, Dollerschell M, Thorel H, Durand-Maugard C, Leroy O, Pecquet M, Voyer A, et al. Real-Life ISO 15189 Qualification of Long-Range Drone Transportation of Medical Biological Samples: Results from a Clinical Trial. Drones. 2026; 10(1):71. https://doi.org/10.3390/drones10010071
Chicago/Turabian StyleDemey, Baptiste, Olivier Bury, Morgane Choquet, Julie Fontaine, Myriam Dollerschell, Hugo Thorel, Charlotte Durand-Maugard, Olivier Leroy, Mathieu Pecquet, Annelise Voyer, and et al. 2026. "Real-Life ISO 15189 Qualification of Long-Range Drone Transportation of Medical Biological Samples: Results from a Clinical Trial" Drones 10, no. 1: 71. https://doi.org/10.3390/drones10010071
APA StyleDemey, B., Bury, O., Choquet, M., Fontaine, J., Dollerschell, M., Thorel, H., Durand-Maugard, C., Leroy, O., Pecquet, M., Voyer, A., Dhaussy, G., & Castelain, S. (2026). Real-Life ISO 15189 Qualification of Long-Range Drone Transportation of Medical Biological Samples: Results from a Clinical Trial. Drones, 10(1), 71. https://doi.org/10.3390/drones10010071









