Targeting Specific Sites in Biological Systems with Synchrotron X-Ray Microbeams for Radiobiological Studies at the Photon Factory
Abstract
1. Introduction
2. Early Studies of Bystander Effects using X-Ray Microbeams
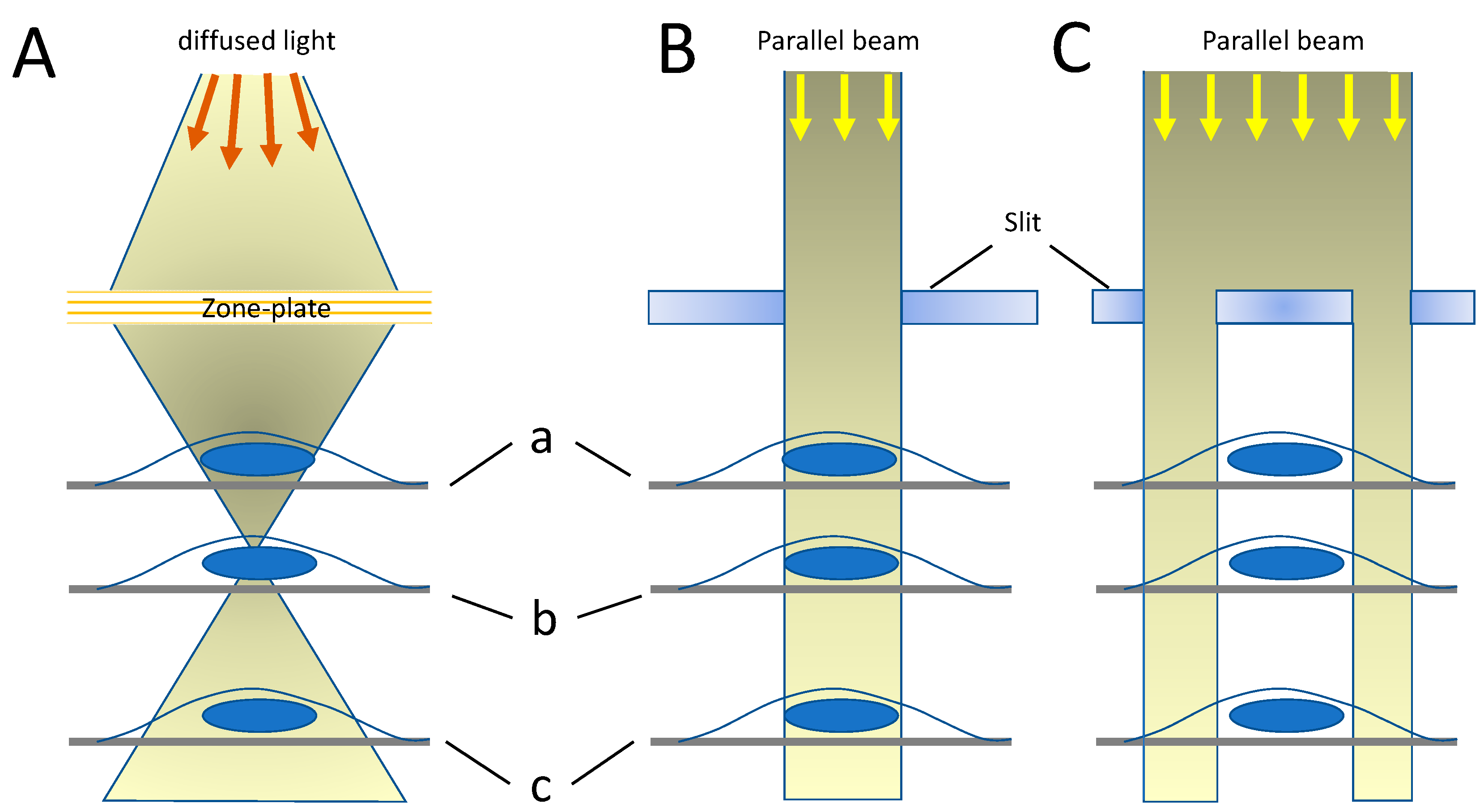
3. Synchrotron X-Ray Microbeams in Radiation Biology
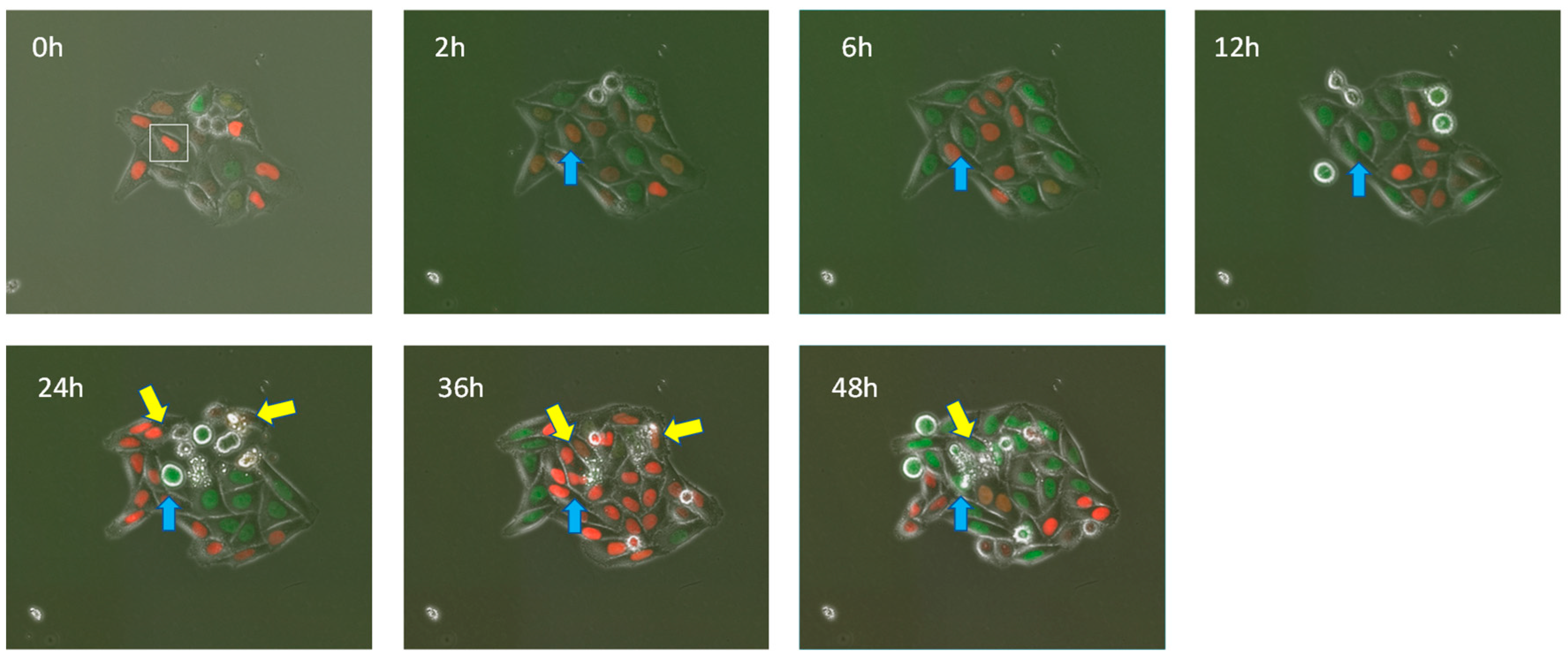
4. Cytoplasmic Irradiation with X-Ray Microbeams
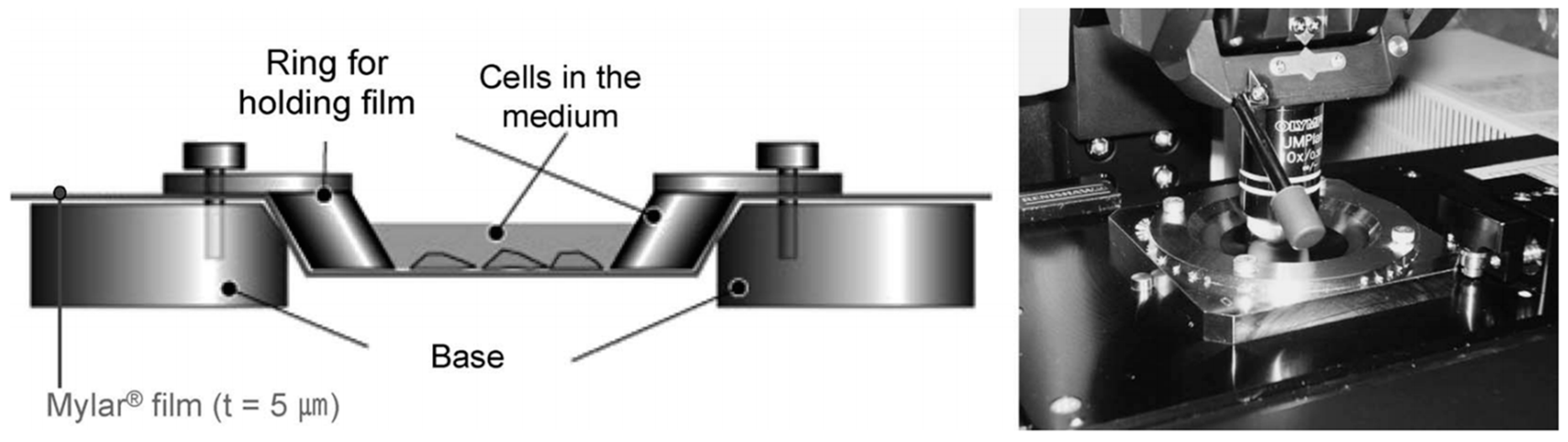
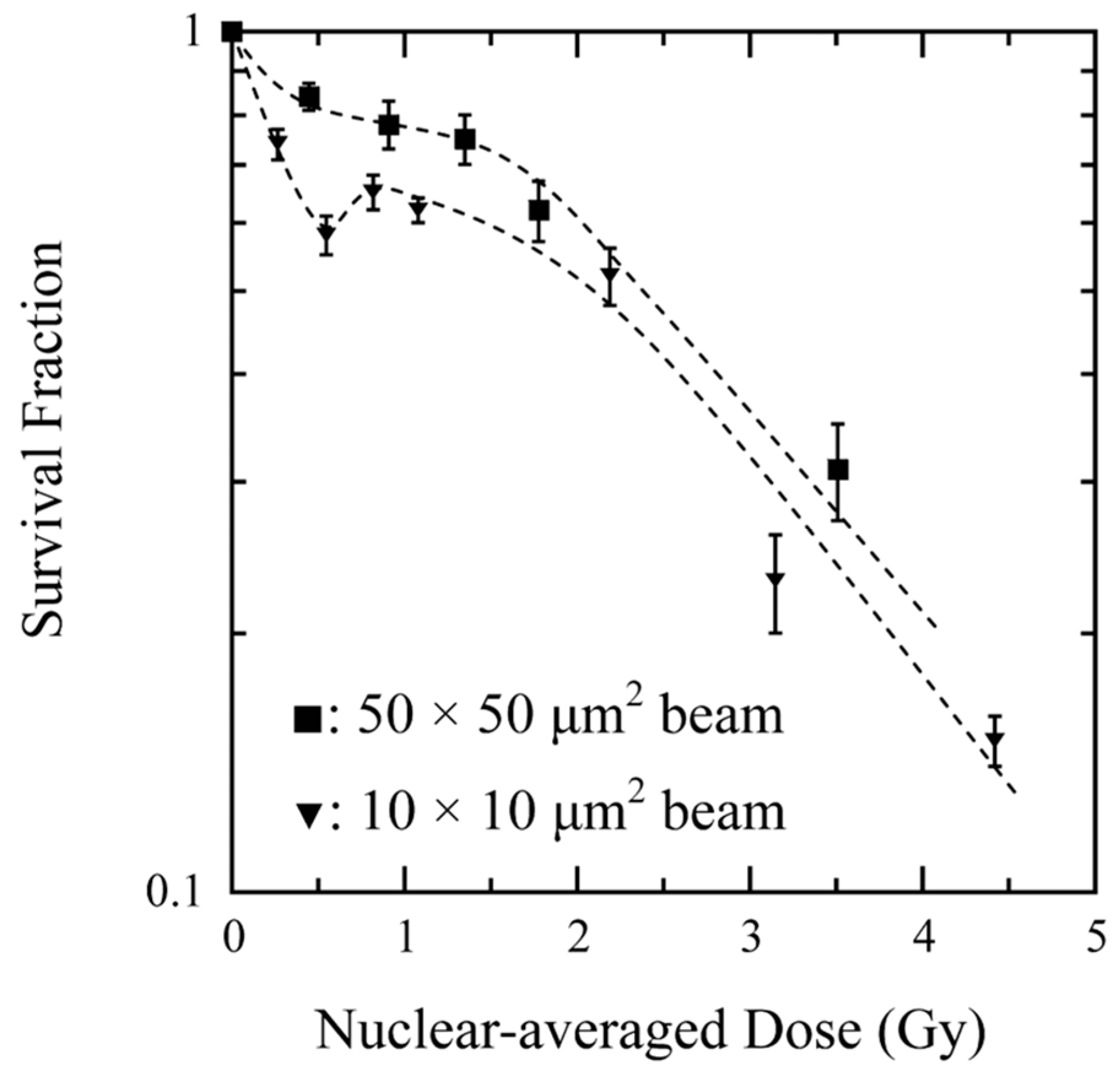
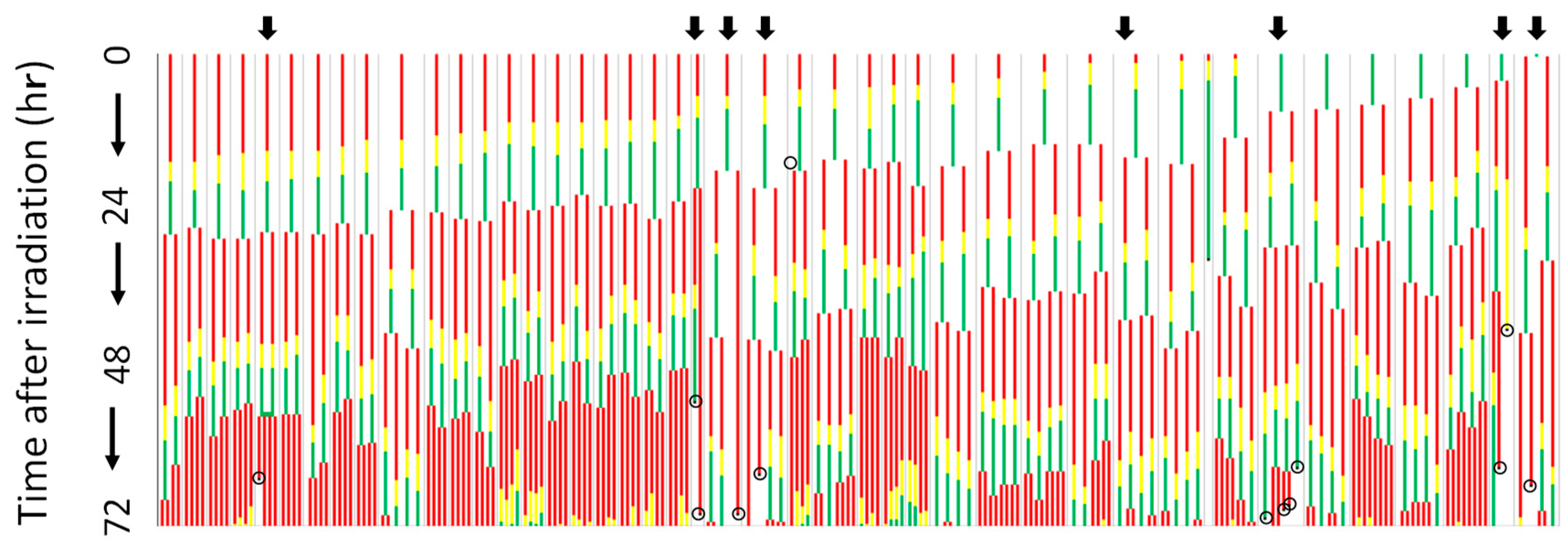
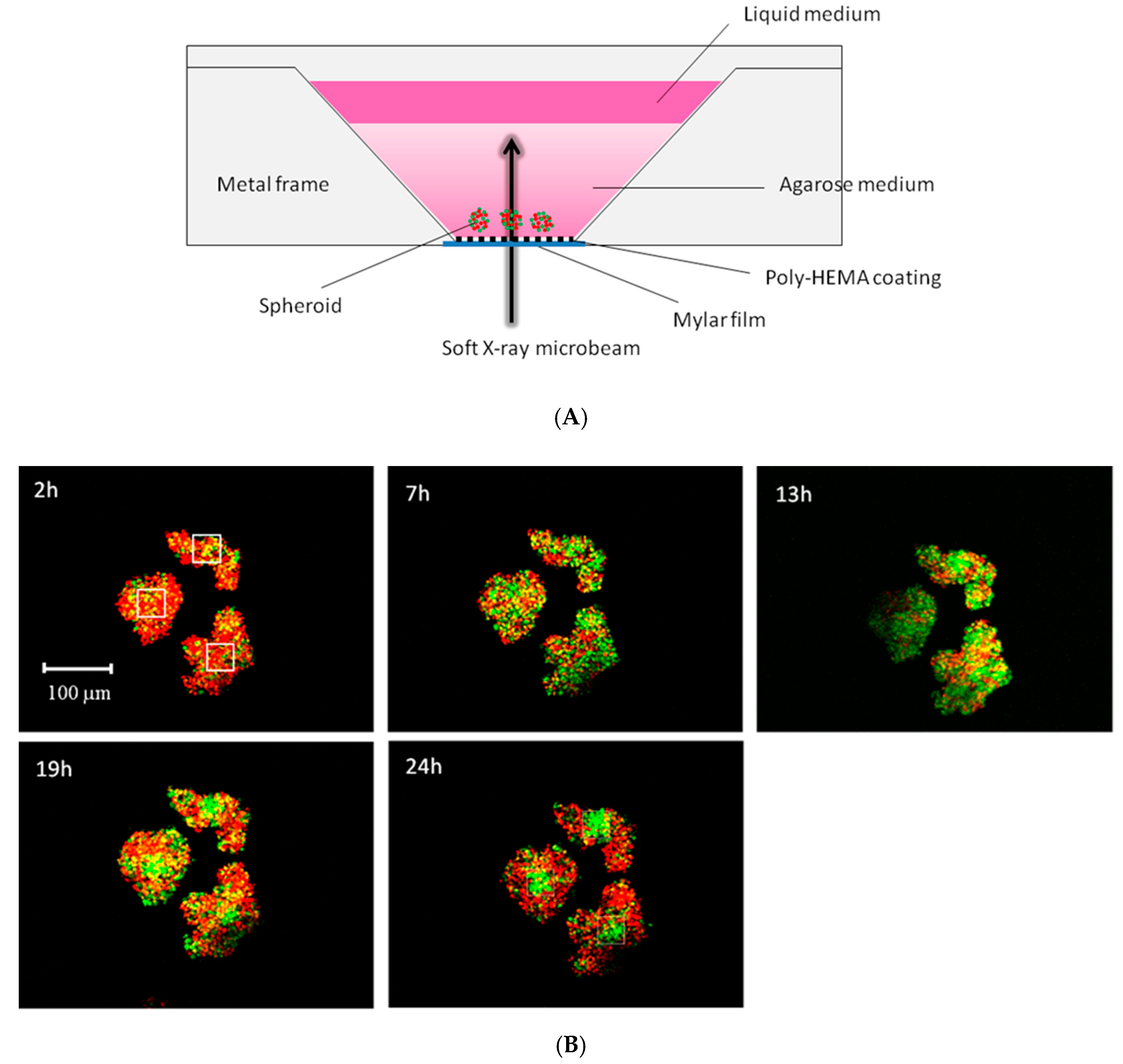
5. Microbeam Exposure to Cell Population Systems Including 3-D Cultured Cells
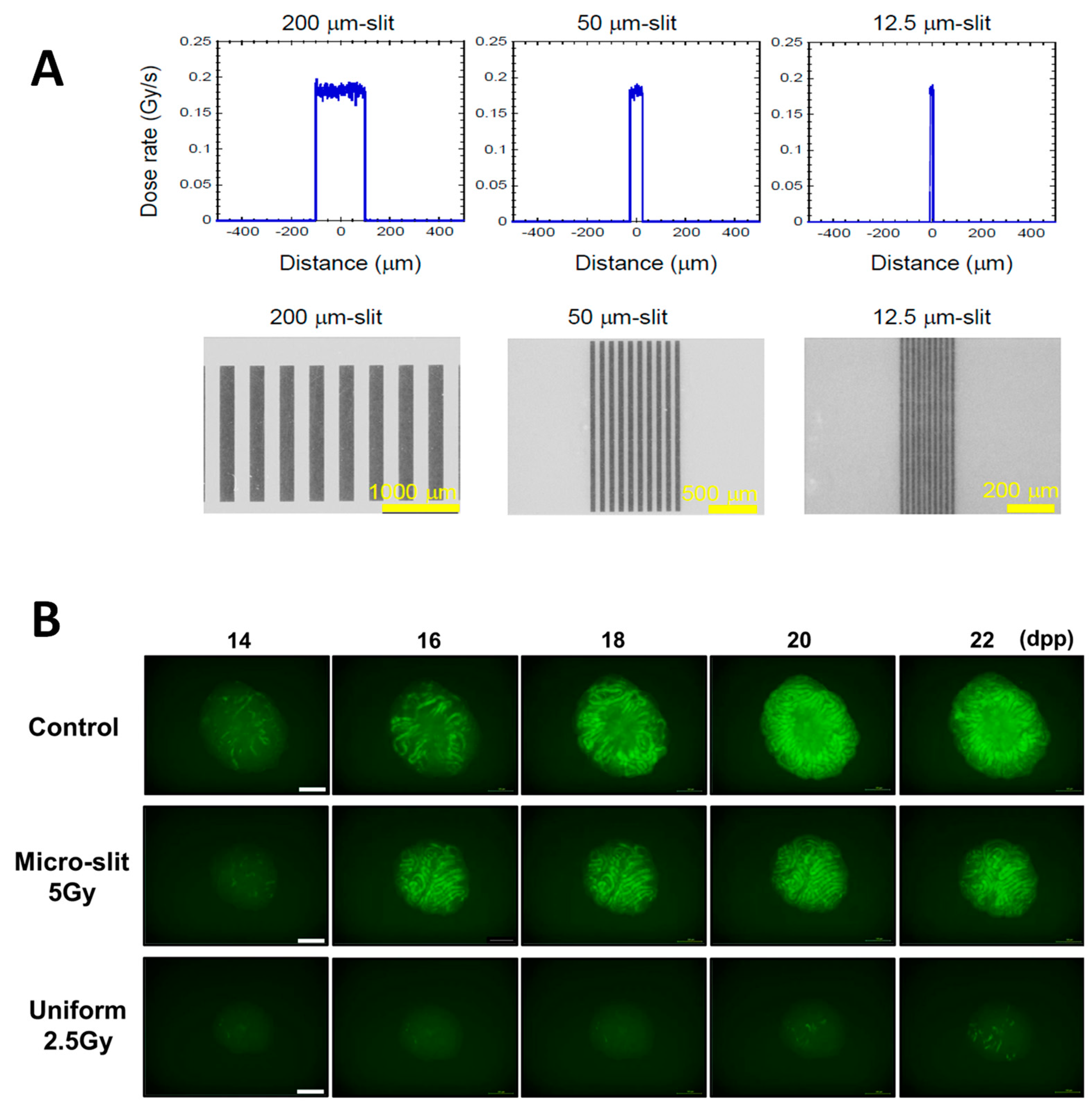
6. Clinical Appreciations of X-Ray Microbeams
7. Summary
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Drexler, G.A.; Ruiz-Gomez, M.J. Microirradiation techniques in radiobiological research. J. Biosci. 2015, 40, 629–643. [Google Scholar] [CrossRef] [PubMed]
- Prise, K.M.; Folkard, M.; Michael, B.D. A review of the bystander effect and its implications for low-dose exposure. Radiat. Prot. Dosimetry 2003, 104, 347–355. [Google Scholar] [CrossRef]
- Hall, E.J.; Hei, T.K. Genomic instability and bystander effects induced by high-LET radiation. Oncogene 2003, 22, 7034–7042. [Google Scholar] [CrossRef] [PubMed]
- Nagasawa, H.; Little, J.B. Induction of sister chromatid exchanges by extremely low doses of alpha-particles. Cancer Res. 1992, 52, 6394–6396. [Google Scholar] [PubMed]
- Deshpande, A.; Goodwin, E.H.; Bailey, S.M.; Marrone, B.L.; Lehnert, B.E. Alpha-particle-induced sister chromatid exchange in normal human lung fibroblasts: Evidence for an extranuclear target. Radiat. Res. 1996, 145, 260–267. [Google Scholar] [CrossRef]
- Lorimore, S.A.; Wright, E.G. Radiation-induced genomic instability and bystander effects: Related inflammatory-type responses to radiation-induced stress and injury? A Rev. Int. J. Radiat. Biol. 2003, 79, 15–25. [Google Scholar] [CrossRef]
- Tobias, F.; Durante, M.; Taucher-Scholz, G.; Jakob, B. Spatiotemporal analysis of DNA repair using charged particle radiation. Mutat. Res. 2010, 704, 54–60. [Google Scholar] [CrossRef]
- Barberet, P.; Seznec, H. Advances in microbeam technologies and applications to radiation biology. Radiat. Prot. Dosimetry 2015, 166, 182–187. [Google Scholar] [CrossRef]
- Kobayashi, Y.; Funayama, T.; Hamada, N.; Sakashita, T.; Konishi, T.; Imaseki, H.; Yasuda, K.; Hatashita, M.; Takagi, K.; Hatori, S.; et al. Microbeam irradiation facilities for radiobiology in Japan and China. J. Radiat. Res. 2009, 50 (Suppl. A), A29–A47. [Google Scholar] [CrossRef]
- Prise, K.M.; Folkard, M.; Michael, B.D. Bystander responses induced by low LET radiation. Oncogene 2003, 22, 7043–7049. [Google Scholar] [CrossRef]
- Schettino, G.; Folkard, M.; Prise, K.M.; Vojnovic, B.; Held, K.D.; Michael, B.D. Low-dose studies of bystander cell killing with targeted soft X rays. Radiat. Res. 2003, 160, 505–511. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, K.; Usami, N.; Hieda, K.; Takakura, K.; Maezawa, H.; Hayashi, T. Development of microbeam irradiation system for radiobiology. Nucl. Instrum. Methods Phys. Res. 2001, 467, 1329–1332. [Google Scholar] [CrossRef]
- Fukunaga, H.; Kaminaga, K.; Sato, T.; Butterworth, K.T.; Watanabe, R.; Usami, N.; Ogawa, T.; Yokoya, A.; Prise, K.M. High-precision microbeam radiotherapy reveals testicular tissue-sparing effects for male fertility preservation. Sci. Rep. 2019, 9, 12618. [Google Scholar] [CrossRef] [PubMed]
- Tomita, M.; Maeda, M.; Maezawa, H.; Usami, N.; Kobayashi, K. Bystander Cell Killing in Normal Human Fibroblasts is Induced by Synchrotron X-Ray Microbeams. Radiat. Res. 2010, 173, 380–385. [Google Scholar] [CrossRef] [PubMed]
- Maeda, M.; Usami, N.; Kobayashi, K. Low-dose hypersensitivity in nucleus-irradiated V79 cells studied with synchrotron X-ray microbeam. J. Radiat. Res. 2008, 49, 171–180. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Zhou, H.; Hong, M.; Chai, Y.; Hei, T.K. Consequences of cytoplasmic irradiation: Studies from microbeam. J. Radiat. Res. 2009, 50 (Suppl. A), A59–A65. [Google Scholar] [CrossRef]
- Ghita, M.; McMahon, S.J.; Taggart, L.E.; Butterworth, K.T.; Schettino, G.; Prise, K.M. A mechanistic study of gold nanoparticle radiosensitisation using targeted microbeam irradiation. Sci. Rep. 2017, 7, 44752. [Google Scholar] [CrossRef]
- Kaminaga, K.; Noguchi, M.; Narita, A.; Hattori, Y.; Usami, N.; Yokoya, A. Cell cycle tracking for irradiated and unirradiated bystander cells in a single colony with exposure to a soft X-ray microbeam. Int. J. Radiat. Biol. 2016, 92, 739–744. [Google Scholar] [CrossRef]
- Kaminaga, K.; Noguchi, M.; Narita, A.; Sakamoto, Y.; Kanari, Y.; Yokoya, A. Visualisation of cell cycle modifications by X-ray irradiation of single HeLa cells using fluorescent ubiquitination-based cell cycle indicators. Radiat. Prot. Dosimetry 2015, 166, 91–94. [Google Scholar] [CrossRef]
- Yokoya, A.; Kaminaga, K. Single-cell tracking and dynamical analysis of cellular population based on systems biology. Radiat. Biol. Res. Commun. 2014, 49, 418–438. (In Japanese) [Google Scholar]
- Sakamoto, Y.; Kaminaga, K.; Kanari, Y.; Usami, N.; Noguchi, M.; Yokoya, A. Live-cell imaging study of radiation effect on cell cycles of three-dimensional cultured cells exposed to synchrotron soft X-ray microbeam. Basic Investig. Breast Carcinoma 2015, 24, 21–27. [Google Scholar]
- Slatkin, D.N.; Spanne, P.; Dilmanian, F.A.; Sandborg, M. Microbeam radiation therapy. Med. Phys. 1992, 19, 1395–1400. [Google Scholar] [CrossRef]
- Grotzer, M.A.; Schultke, E.; Brauer-Krisch, E.; Laissue, J.A. Microbeam radiation therapy: Clinical perspectives. Phys. Med. 2015, 31, 564–567. [Google Scholar] [CrossRef] [PubMed]
- Laissue, J.A.; Blattmann, H.; Wagner, H.P.; Grotzer, M.A.; Slatkin, D.N. Prospects for microbeam radiation therapy of brain tumours in children to reduce neurological sequelae. Dev. Med. Child Neurol. 2007, 49, 577–581. [Google Scholar] [CrossRef] [PubMed]
- Laissue, J.A.; Bartzsch, S.; Blattmann, H.; Bräuer-Krisch, E.; Bravin, A.; Dalléry, D.; Djonov, V.; Hanson, A.L.; Hopewell, J.W.; Kaser-Hotz, B.; et al. Response of the rat spinal cord to X-ray microbeams. Radiother Oncol. 2013, 106, 106–111. [Google Scholar] [CrossRef] [PubMed]
- Dilmanian, F.A.; Qu, Y.; Feinendegen, L.E.; Peña, L.A.; Bacarian, T.; Henn, F.A.; Kalef-Ezra, J.; Liu, S.; Zhong, Z.; McDonald, J.W. Tissue-sparing effect of X-ray microplanar beams particularly in the CNS: Is a bystander effect involved? Exp. Hematol. 2007, 35 (Suppl. 1), 69–77. [Google Scholar] [CrossRef]
- Sato, T.; Katagiri, K.; Gohbara, A.; Inoue, K.; Ogonuki, N.; Ogura, A.; Kubota, Y.; Ogawa, T. In vitro production of functional sperm in cultured neonatal mouse testes. Nature 2011, 471, 504–507. [Google Scholar] [CrossRef]
- Fukunaga, H.; Kaminaga, K.; Sato, T.; Usami, N.; Watanabe, R.; Butterworth, K.T.; Ogawa, T.; Yokoya, A.; Prise, K.M. Application of an Ex Vivo Tissue Model to Investigate Radiobiological Effects on Spermatogenesis. Radiat. Res. 2018, 189, 661–667. [Google Scholar] [CrossRef]
- Sato, T.; Iwamoto, Y.; Hashimoto, S.; Ogawa, T.; Furuta, T.; Abe, S.I.; Kai, T.; Tsai, P.E.; Matsuda, N.; Iwase, H.; et al. Features of Particle and Heavy Ion Transport code System (PHITS) version 3.02. J. Nucl. Sci. Technol. 2018, 55, 684–690. [Google Scholar]

© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yokoya, A.; Usami, N. Targeting Specific Sites in Biological Systems with Synchrotron X-Ray Microbeams for Radiobiological Studies at the Photon Factory. Quantum Beam Sci. 2020, 4, 2. https://doi.org/10.3390/qubs4010002
Yokoya A, Usami N. Targeting Specific Sites in Biological Systems with Synchrotron X-Ray Microbeams for Radiobiological Studies at the Photon Factory. Quantum Beam Science. 2020; 4(1):2. https://doi.org/10.3390/qubs4010002
Chicago/Turabian StyleYokoya, Akinari, and Noriko Usami. 2020. "Targeting Specific Sites in Biological Systems with Synchrotron X-Ray Microbeams for Radiobiological Studies at the Photon Factory" Quantum Beam Science 4, no. 1: 2. https://doi.org/10.3390/qubs4010002
APA StyleYokoya, A., & Usami, N. (2020). Targeting Specific Sites in Biological Systems with Synchrotron X-Ray Microbeams for Radiobiological Studies at the Photon Factory. Quantum Beam Science, 4(1), 2. https://doi.org/10.3390/qubs4010002



