Dynamic Cancellation of Perceived Rotation from the Venetian Blind Effect
Abstract
1. Introduction
1.1. Venetian Blind Effect
1.2. Rationale for Current Research
2. Materials and Methods
2.1. Observers
2.2. Apparatus
2.3. Procedure
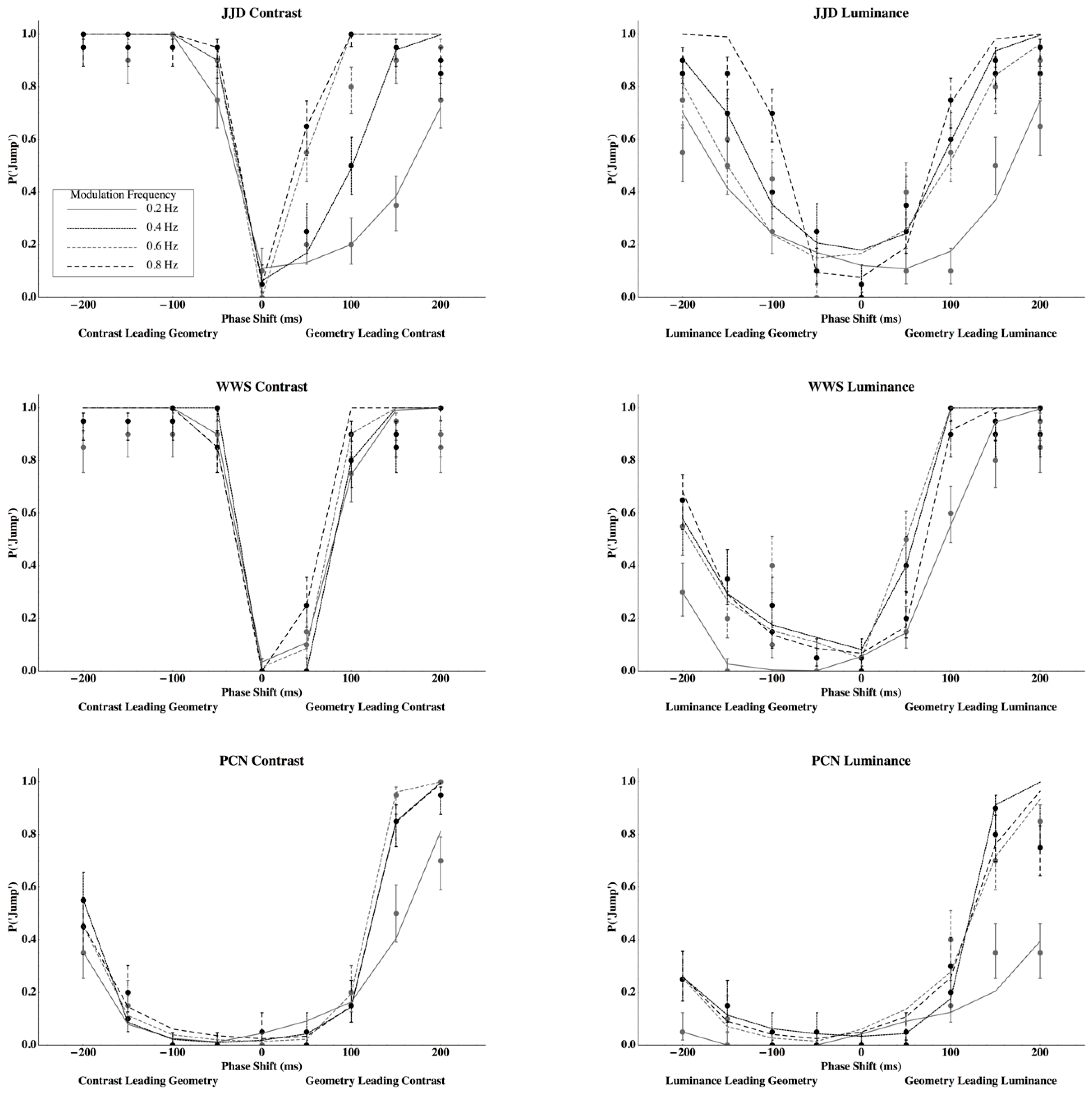
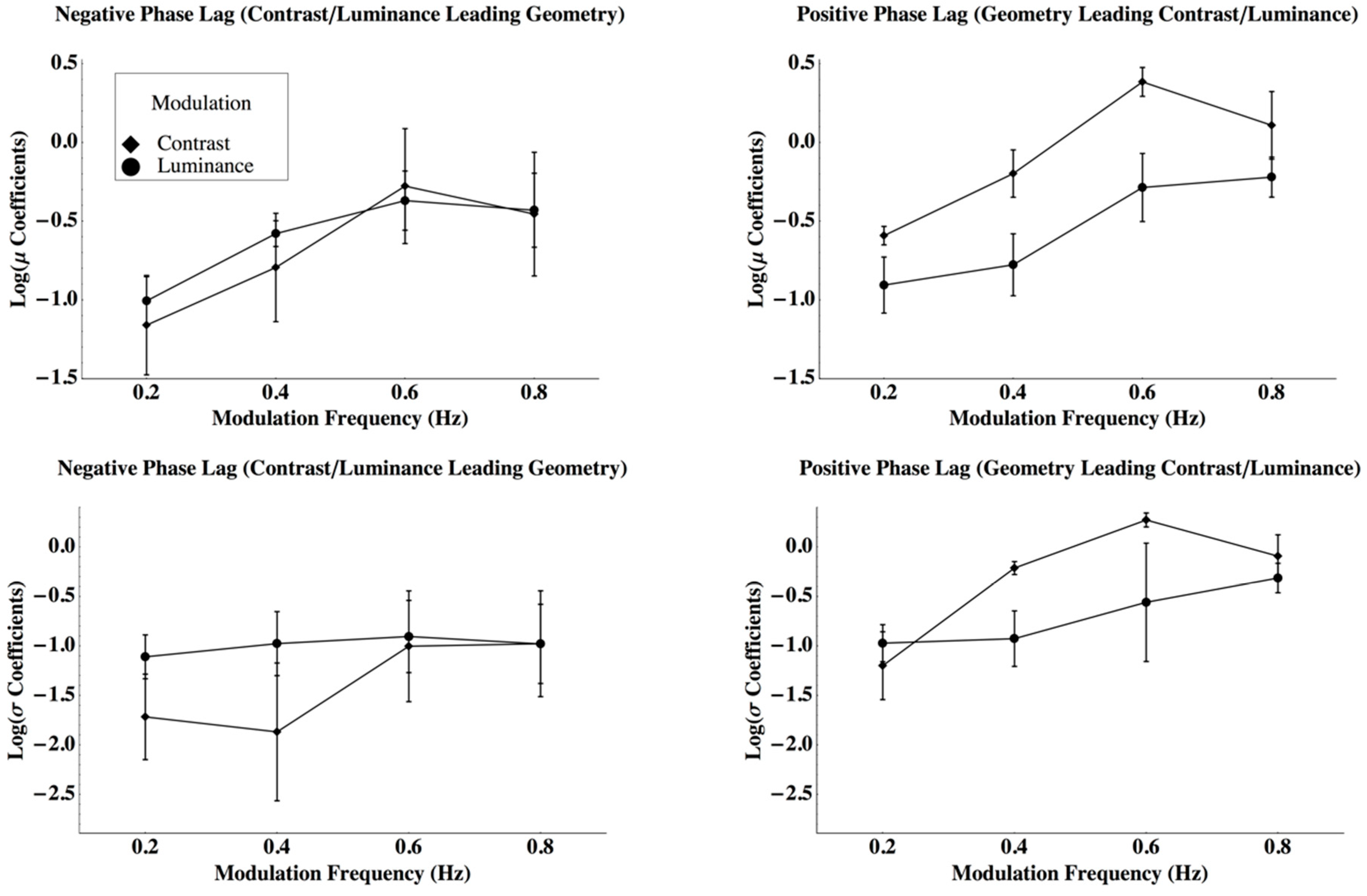
3. Results
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
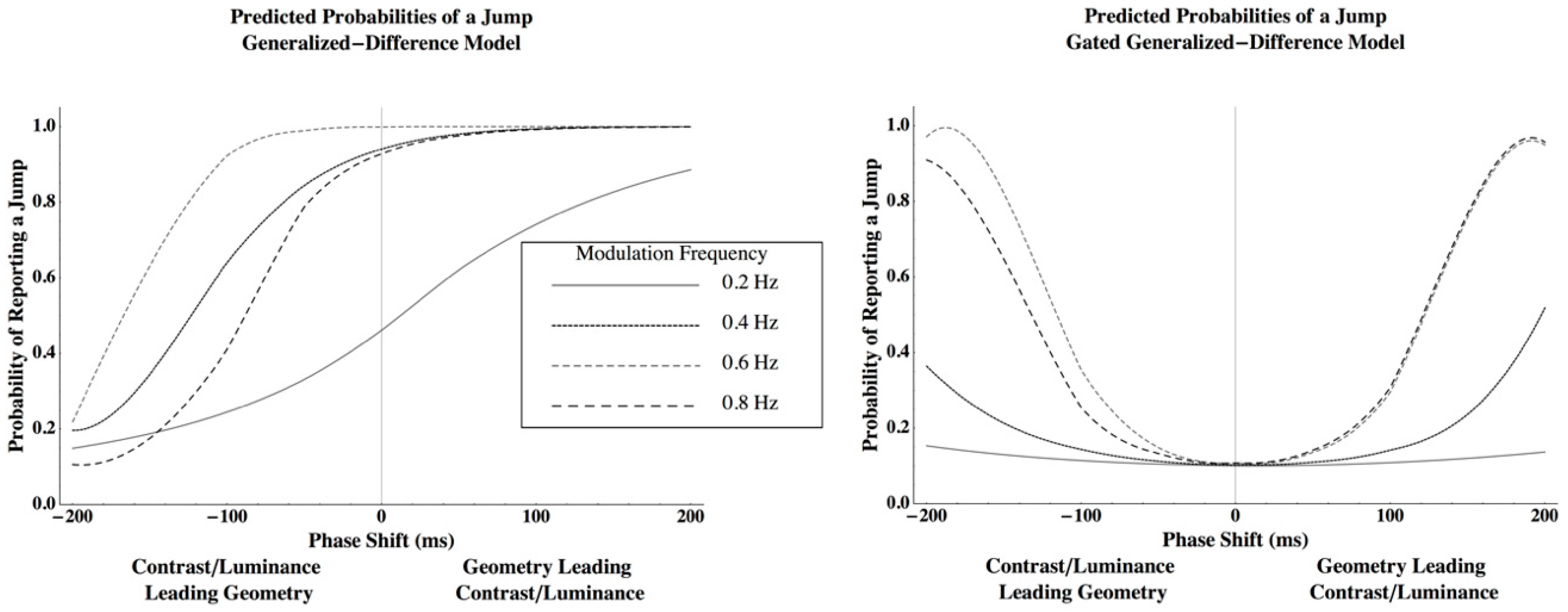
Appendix A. The Dynamic Models of the Venetian Blind Effect
Appendix A.1. Generalized Difference Model
Appendix A.2. Gated Generalized Difference Model
Appendix B. Predictions and Fits from the Dynamic Models
Appendix C. Analysis of Variance for the Laplace Parameter Estimates
References
- Michelson, A.A. Studies in Optics; 1962 Reprint; University of Chicago Press, Ltd.: Chicago, IL, USA, 1927. [Google Scholar]
- Münster, C. Ueber den Einfluss von Helligkeitsunterschieden in beiden Augen auf die stereoskipische Wahrnehmung. [The effect of interocular brightness differences on stereoscopic perception]. Zeitschrift für Sinnesphysiologie 1941, 69, 245–260. [Google Scholar]
- Larkin, E.T.; Stine, W.W. The discovery of the Venetian blind effect: A translation of Münster (1941). i-Perception 2017, 1–21. [Google Scholar] [CrossRef]
- Cibis, P.A.; Haber, H. Anisopia and perception of space. J. Opt. Soc. Am. 1951, 41, 676–683. [Google Scholar] [CrossRef] [PubMed]
- Von Békésy, G. Apparent image rotation in stereoscopic vision: The unbalance of the pupils. Percept. Psychophys. 1970, 8, 343–347. [Google Scholar] [CrossRef]
- Fiorentini, A.; Maffei, L. Binocular depth perception without geometrical cues. Vis. Res. 1971, 11, 1299–1305. [Google Scholar] [CrossRef]
- Filley, E.T.; Khutoryansky, N.; Dobias, J.J.; Stine, W.W. An investigation of the Venetian blind effect. See. Percept. 2011, 24, 241–292. [Google Scholar]
- Hetley, R.S.; Stine, W.W. Partitioning contrast or luminance disparity into perceived intensity and rotation. See. Percept. 2011, 24, 315–350. [Google Scholar] [CrossRef] [PubMed]
- Dobias, J.J.; Stine, W.W. Temporal Dynamics of the Venetian Blind Effect. Vis. Res. 2012, 60, 79–94. [Google Scholar] [CrossRef] [PubMed]
- Blake, R.; Cormack, R.H. Does contrast disparity alone generate stereopsis? Vis. Res. 1979, 19, 913–915. [Google Scholar] [CrossRef]
- Von Helmholtz, H. Helmholtz’s Treatise on Physiological Optics: Vol. II. The Sensations of Vision; Trans. from 3rd Edition; Optical Society of America: Rochester, NY, USA, 1911/1924. [Google Scholar]
- Brown, H.I. Galileo on the telescope and the eye. J. Hist. Ideas 1985, 46, 487–501. [Google Scholar] [CrossRef]
- Westheimer, G. Irradiation, border location, and the shifted-chessboard pattern. Perception 2007, 36, 483–494. [Google Scholar] [CrossRef] [PubMed]
- Bex, P.J.; Edgar, G.K. Shifts in the perceived location of a blurred edge increase with contrast. Percept. Psychophys. 1996, 58, 31–33. [Google Scholar] [CrossRef] [PubMed]
- Georgeson, M.A.; Freeman, T.C. A Perceived location of bars and edges in one dimensional images: Computational models and human vision. Vis. Res. 1997, 37, 127–142. [Google Scholar] [CrossRef]
- Mather, G.; Morgan, M.J. Irradiation: Implications for theories of edge localization. Vis. Res. 1986, 26, 1007–1015. [Google Scholar] [CrossRef]
- Morgan, M.J.; Mather, G.; Moulden, B.; Watt, R.J. Intensity response nonlinearities and the theory of edge localization. Vis. Res. 1984, 24, 713–719. [Google Scholar] [CrossRef]
- Naiman, A.C.; Makous, W. Undetected grey strips displace perceived edges nonlinearly. J. Opt. Soc. Am. A 1993, 10, 794–803. [Google Scholar] [CrossRef] [PubMed]
- Wallis, S.A.; Georgeson, M.A. Mach edges: Local features predicted by 3rd derivative spatial filtering. Vis. Res. 2009, 49, 1886–1893. [Google Scholar] [CrossRef] [PubMed]
- Gillam, B.J.; Wardle, S.G. A mid-level explanation for the venetian blind effect. Front. Psychiatry 2013, 4, 908–910. [Google Scholar] [CrossRef] [PubMed]
- Richards, W. Response functions for sine- and square-wave modulations of disparity. J. Opt. Soc. Am. 1972, 62, 907–911. [Google Scholar] [CrossRef]
- Regan, D.; Beverley, K.I. Some dynamic features of depth perception. Vis. Res. 1973, 13, 2369–2379. [Google Scholar] [CrossRef]
- Regan, D.; Beverley, K.I. The dissociation of sideways movements from movements in depth: Psychophysics. Vis. Res. 1973, 13, 2403–2415. [Google Scholar] [CrossRef]
- Beverley, K.I.; Regan, D. Evidence for the existence of neural mechanisms selectively sensitive to the direction of movement in space. J. Phys. 1973, 235, 17–29. [Google Scholar] [CrossRef]
- Beverley, K.I.; Regan, D. Selective adaptation in stereoscopic depth perception. J. Phys. 1973, 232, 40–41. [Google Scholar]
- Beverley, K.I.; Regan, D. Temporal integration of disparity information in stereoscopic perception. Exp. Brain Res. 1974, 19, 228–232. [Google Scholar] [CrossRef] [PubMed]
- Beverley, K.I.; Regan, D. Visual sensitivity to disparity pulses. Vis. Res. 1974, 14, 357–361. [Google Scholar] [CrossRef]
- Brindley, G.S. Physiology of the Retina and the Visual Pathway; Edward Arnold: London, UK, 1960. [Google Scholar]
- Teller, D.Y. Linking propositions. Vis. Res. 1984, 24, 1233–1246. [Google Scholar] [CrossRef]
- Weisstein, E.W. Fourier Series—Square Wave. MathWorld—A Wolfram Web Resource. 2012. Available online: http://mathworld.wolfram.com/FourierSeriesSquareWave.html (accessed on 15 November 2018).
- Lit, A. The magnitude of the Pulfrich stereophenomenon as a function of target velocity. J. Exp. Psychiatry 1960, 59, 165–175. [Google Scholar] [CrossRef]
- Agresti, A.; Coull, B.A. Approximate is better than “exact” for interval estimation of binomial proportions. Am. Stat. 1998, 52, 119–126. [Google Scholar]
- Wilson, E.B. Probable inference, the law of succession, and statistical inference. J. Am. Stat. Assoc. 1927, 22, 209–212. [Google Scholar] [CrossRef]
- Kirk, R.E. Experimental Design: Procedures for the Behavioral Sciences, 3rd ed.; Brooks/Cole: New York, NY, USA, 1995. [Google Scholar]
- Anderson, A.J.; Vingrys, A.J. Small Samples: Does Size Matter? Investig. Ophthalmol. Vis. Sci. 2001, 42, 1411–1413. [Google Scholar]
- Smith, P.L.; Little, D.R. Small is beautiful: In defense of the small-N design. Psychon. Bull. Rev. 2018, 25, 2083–2101. [Google Scholar] [CrossRef] [PubMed]
- Sclar, G.; Maunsell, J.H.R.; Lennie, P. Coding of image contrast in central visual pathways of the macaque monkey. Vis. Res. 1990, 30, 1–10. [Google Scholar] [CrossRef]
- Geisler, W.S.; Albrecht, D.G.; Crane, A.M. Responses of neurons in primary visual cortex to transient changes in local contrast and luminance. J. Neurosci. 2007, 27, 5063–5067. [Google Scholar] [CrossRef] [PubMed]
- Livingstone, M.; Hubel, D. Segregation of form, color, movement, and depth: Anatomy, physiology, and perception. Science 1988, 240, 740–749. [Google Scholar] [CrossRef] [PubMed]
- Sincich, L.C.; Horton, J.C. The circuitry of V1 and V2: Integration of color, form, and motion. Annu. Rev. Neurosci. 2005, 28, 303–326. [Google Scholar] [CrossRef] [PubMed]
- Yabuta, N.H.; Sawatari, A.; Callaway, E.M. Two functional channels from primary visual cortex to dorsal visual cortical areas. Science 2001, 292, 297–300. [Google Scholar] [CrossRef] [PubMed]
- Schiller, P.H.; Logothetis, N.K.; Charles, E.R. Functions of the colour-opponent and broadband channels of the visual system. Nature 1990, 343, 68–70. [Google Scholar] [CrossRef] [PubMed]
- Poggio, G.F.; Gonzalez, F.; Krause, F. Stereoscopic mechanisms in monkey visual cortex: Binocular correlation and disparity selectivity. J. Neurosci. 1988, 8, 4531–4550. [Google Scholar] [CrossRef] [PubMed]
- DeYoe, E.A.; Van Essen, D.C. Segregation of efferent connections and receptive field properties in visual area V2 of the macaque. Nature 1985, 317, 58–61. [Google Scholar] [CrossRef]
- Peterhans, E.; von der Heydt, R. Functional organization of area V2 in the alert macaque. Eur. J. Neurosci. 1993, 5, 509–524. [Google Scholar] [CrossRef] [PubMed]
- Sincich, L.C.; Horton, J.C. Divided by cytochrome oxidase: A map of the projections from V1 to V2 in macaques. Science 2002, 295, 1734–1737. [Google Scholar] [CrossRef] [PubMed]
- Cumming, B.G.; Parker, A.J. Binocular neurons in V1 of awake monkeys are selective for absolute, not relative, disparity. J. Neurosci. 1999, 19, 5602–5618. [Google Scholar] [CrossRef] [PubMed]
- Thomas, O.M.; Cumming, B.G.; Parker, A.J. A specialization for relative disparity in V2. Nat. Neurosci. 2002, 5, 472–478. [Google Scholar] [CrossRef] [PubMed]
- Parker, A.J. Binocular depth perception and the cerebral cortex. Nat. Rev. Neurosci. 2007, 8, 379–391. [Google Scholar] [CrossRef] [PubMed]
- Rockland, K.S. Elements of cortical architecture: Hierarchy revisited. In Cerebral Cortex: Extrastriate Cortex in Primates; Rockland, K.S., Kaas, J.H., Peters, A., Eds.; Plenum: New York, NY, USA, 1997; Volume 12, pp. 243–293. [Google Scholar]
- Naka, K.I.; Rushton, W.A.H. S-potentials from luminosity units in the retina of fish (Cyprinidae). J. Physiol. 1966, 185, 587–599. [Google Scholar] [CrossRef] [PubMed]
- Backus, B.T.; Banks, M.S.; van Ee, R.; Crowell, J.A. Horizontal and vertical disparity, eye position, and stereoscopic slant perception. Vis. Res. 1999, 33, 1143–1170. [Google Scholar] [CrossRef]
- Holm, S. A simple sequentially rejective multiple test procedure. Scand. J. Stat. 1979, 6, 65–70. [Google Scholar]

| Observer | Modulation Type | R2 | Adjusted R2 |
|---|---|---|---|
| JJD | Contrast | 0.995 | 0.991 |
| Luminance | 0.974 | 0.954 | |
| WWS | Contrast | 0.994 | 0.989 |
| Luminance | 0.984 | 0.973 | |
| PCN | Contrast | 0.993 | 0.987 |
| Luminance | 0.971 | 0.949 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dobias, J.J.; Stine, W.W. Dynamic Cancellation of Perceived Rotation from the Venetian Blind Effect. Vision 2019, 3, 14. https://doi.org/10.3390/vision3020014
Dobias JJ, Stine WW. Dynamic Cancellation of Perceived Rotation from the Venetian Blind Effect. Vision. 2019; 3(2):14. https://doi.org/10.3390/vision3020014
Chicago/Turabian StyleDobias, Joshua J., and Wm Wren Stine. 2019. "Dynamic Cancellation of Perceived Rotation from the Venetian Blind Effect" Vision 3, no. 2: 14. https://doi.org/10.3390/vision3020014
APA StyleDobias, J. J., & Stine, W. W. (2019). Dynamic Cancellation of Perceived Rotation from the Venetian Blind Effect. Vision, 3(2), 14. https://doi.org/10.3390/vision3020014




