Profiling Genetic Breeding Progress in Bagrid Catfishes
Abstract
1. Background
2. Development Status of the Aquaculture Industry for Yellow Catfish
3. Conventional Breeding Technology of Bagrid Catfishes
3.1. Selective Breeding
3.2. Genetic Engineering Applied to Breeding
3.3. Cell Engineering Applied to Breeding
3.4. Molecular Marker-Assisted Breeding
3.5. Hybrid Breeding of Bagrid Catfishes
3.5.1. Interspecific Breeding
3.5.2. Intergeneric Breeding
3.6. Summary and Comparison
4. Research Progress of Hybrid Bagrid Catfishes
4.1. Breeding Conditions
4.2. Genetic Background
4.3. Molecular Mechanism of Hybrid Vigor
4.4. Comprehensive Breeding Technology for Bagrid Catfishes
4.4.1. Cell Engineering Breeding and Molecular Marker-Assisted Breeding
Super-male and all-male P. fulvidraco

4.4.2. Selective Breeding and Hybrid Breeding
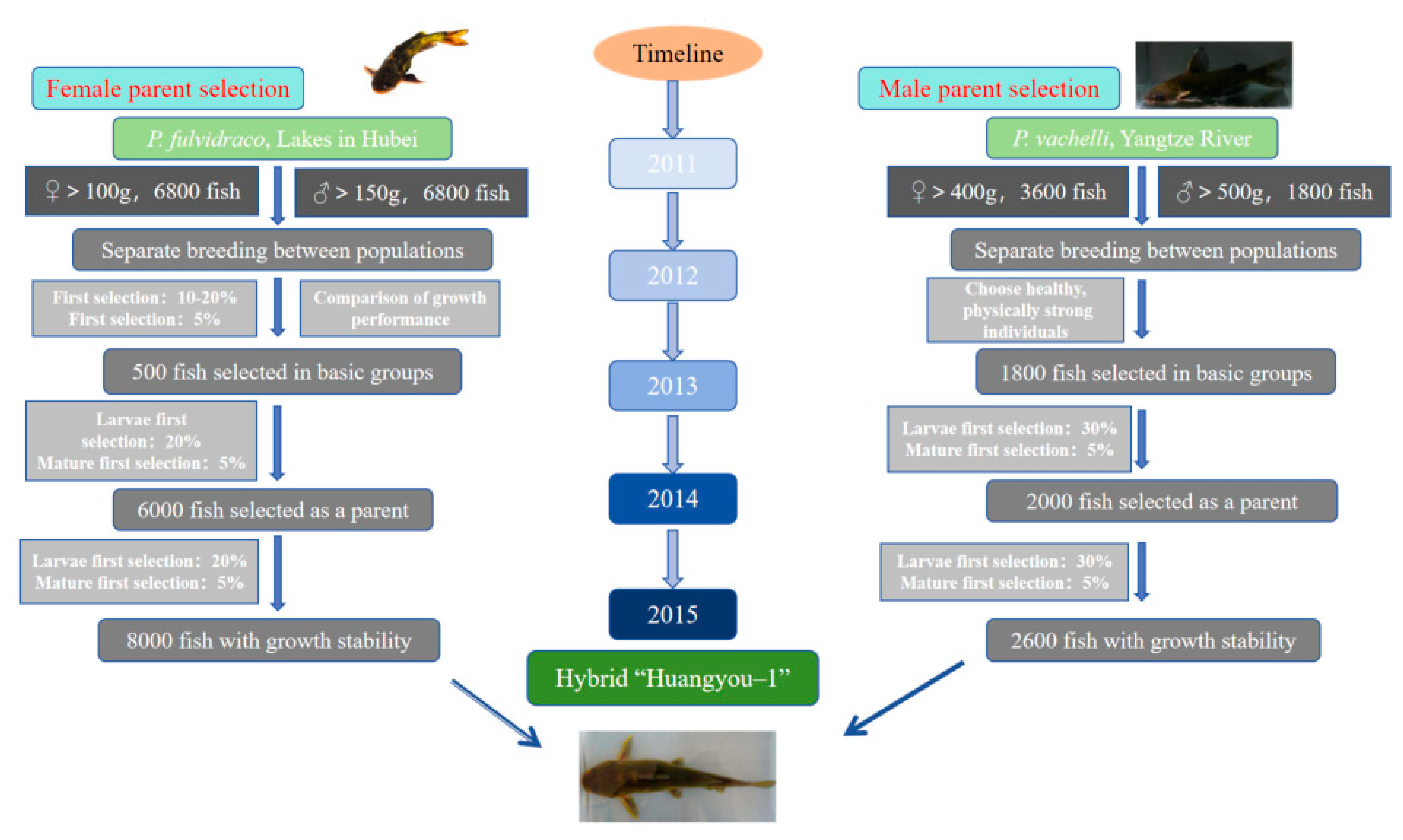
Hybrid “yellow catfish Huangyou No. 1” of P. fulvidraco ♀ × P. vachelli ♂
4.5. Stress Resistance
4.6. Metabolic Nutrition
5. Summary and Future Perspectives
5.1. Whole Genome Sequencing
5.2. Establishment of Germplasm Resource Bank
5.3. Improving Original New Varieties and Breeding Other Hybrid Bagrid Combinations
5.4. Problems and Challenges
5.4.1. Genetic Breeding
5.4.2. Broodstock Management
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Nelson, J.; Grande, T.C.; Wilson, M.V.H. Fishes of the World; John Wiley & Sons: Hoboken, NJ, USA, 2016. [Google Scholar]
- Zhang, G.; Deng, S.; Zhang, H.; Li, H.; Li, L. Distribution of different taste buds and expression of α-gustducin in the barbells of yellow catfish (Pelteobagrus fulvidraco). Fish Physiol. Biochem. 2006, 32, 55–62. [Google Scholar] [CrossRef] [PubMed]
- Qin, Q.; Chen, X.; Zhu, X.; Li, X.; Zhao, Y.; Xu, Z.; Liu, W. Insulin-like growth factor I of yellow catfish (Pelteobagrus fulvidraco): cDNA characterization, tissue distribution, and expressions in response to starvation and refeeding. Fish Physiol. Biochem. 2020, 46, 177–186. [Google Scholar] [CrossRef] [PubMed]
- Mei, J.; Gui, J.-F. Genetic basis and biotechnological manipulation of sexual dimorphism and sex determination in fish. Sci. China Life Sci. 2015, 58, 124–136. [Google Scholar] [CrossRef] [PubMed]
- Gao, D.; Zheng, M.; Lin, G.; Fang, W.; Huang, J.; Lu, J.; Sun, X. Construction of high-density genetic map and mapping of sex-related loci in the yellow catfish (Pelteobagrus fulvidraco). Mar. Biotechnol. 2020, 22, 31–40. [Google Scholar] [CrossRef]
- Tai, Z.P.; Li, D.D.; Ling, S.C.; Zhang, D.G.; Cui, H.Y.; Tan, X.Y. Identification of full-length cDNA sequences for three development-relevant genes from yellow catfish Pelteobagrus fulvidraco and their transcriptional responses to high fat diet. Comp. Biochem. Physiol. Part B 2018, 225, 67–74. [Google Scholar] [CrossRef]
- Wang, Q.I.; Shen, J.; Yan, Z.; Xiang, X.; Mu, R.; Zhu, P.; Yao, Y.; Zhu, F.; Chen, K.; Chi, S.; et al. Dietary Glycyrrhiza uralensis extracts supplementation elevated growth performance, immune responses and disease resistance against Flavobacterium columnare in yellow catfish (Pelteobagrus fulvidraco). Fish Shellfish. Immunol. 2020, 97, 153–164. [Google Scholar] [CrossRef]
- Li, D.; Xie, C.; He, X.; Tan, R.; Tian, X.; Zhang, Z.; Gao, Y. The Success of Yellow Catfish Aquaculture in China: From Rare Wild Fish to Popular Farmed Fish. In Aquaculture in China: Success Stories and Modern Trends; Wiley: Hoboken, NJ, USA, 2018; pp. 270–282. [Google Scholar]
- Jia, B.; St-Hilaire, S.; Singh, K.; Gardner, I.A. Farm-level returns and costs of yellow catfish (Pelteobagrus fulvidraco) aquaculture in Guangdong and Zhejiang provinces, China. Aquac. Rep. 2016, 4, 48–56. [Google Scholar] [CrossRef]
- Zhang, G.; Tao, P.; Chen, J.; Wang, R.; Zang, X.; Yin, S. The complete mitochondrial genome of the hybrid of Pelteobagrus fulvidraco (female symbol) × Pelteobagrus vachelli (male symbol). Mitochondrial DNA Part A 2015, 27, 4191–4192. [Google Scholar] [CrossRef]
- Liu, J.Y.; Li, A.H.; Zhou, D.R.; Wen, Z.R.; Ye, X.P. Isolation and characterization of Edwardsiella ictaluri strains as pathogens from diseased yellow catfish Pelteobagrus fulvidraco (Richardson) cultured in China. Aquac. Res. 2010, 41, 1835–1844. [Google Scholar] [CrossRef]
- Cheng, K.; Tang, Q.; Guo, X.; Karow, N.A.; Wang, C. High dose of dietary vitamin D3 modulated the yellow catfish (Pelteobagrus fulvidraco) splenic innate immune response after Edwardsiella ictaluri infection. Fish Shellfish. Immunol. 2020, 100, 41–48. [Google Scholar] [CrossRef]
- Cheng, K.; Tang, Q.; Huang, Y.; Liu, X.; Karrow, N.A.; Wang, C. Effect of vitamin D3 on the immunomodulation of head kidney after Edwardsiella ictaluri challenge in yellow catfish (Pelteobagrus fulvidraco). Fish Shellfish. Immunol. 2020, 99, 353–361. [Google Scholar] [CrossRef] [PubMed]
- Luo, Z.; Wei, C.C.; Ye, H.M.; Zhao, H.P.; Song, Y.F.; Wu, K. Effect of dietary choline levels on growth performance, lipid deposition and metabolism in juvenile yellow catfish Pelteobagrus fulvidraco. Comp. Biochem. Physiol. Part B 2016, 202, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Chiu, A.; Li, L.; Guo, S.; Bai, J.; Fedor, C.; Naylor, R.L. Feed and fishmeal use in the production of carp and tilapia in China. Aquaculture 2013, 414–415, 127–134. [Google Scholar] [CrossRef]
- Geng, Y.; Wang, K.Y.; Li, C.W.; Ren, S.Y.; Zhou, Z.Y.; Liu, X.X.; Liu, X.F.; Lai, W.M.; Huang, X.L.; Chen, D.F. Isolation and Characterization of Edwardsiella ictaluri from Southern Catfish, Silurus soldatovi meridionalis, Cultured in China. J. World Aquac. Soc. 2013, 44, 273–281. [Google Scholar] [CrossRef]
- Wang, K.L.; Ji, W.; Zhang, G.R.; Wei, K.J.; Shi, Z.C.; Zhang, X.T.; Zheng, H.; Fan, Q.X. Molecular characterization and expression analysis of three TLR genes in yellow catfish (Pelteobagrus fulvidraco): Responses to stimulation of Aeromonas hydrophila and TLR ligands. Fish Shellfish. Immunol. 2017, 66, 466–479. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Wang, J.; Ma, Z.; Li, T.; Xing, W.; Jiang, N.; Li, W.; Li, C.; Luo, L. Effects of totally replacing dietary fish oil by linseed oil or soybean oil on juvenile hybrid sturgeon, Acipenser baeri Brandt♀ × A. schrenckii Brandt♂. Aquac. Nutr. 2018, 24, 184–194. [Google Scholar] [CrossRef]
- Ostberg, C.O.; Chase, D.M.; Hauser, L. Hybridization between yellow-stone cutthroat trout and rainbow trout alters the expression of muscle growth-related genes and their relationships with growth patterns. PLoS ONE 2015, 10, e0141373. [Google Scholar] [CrossRef]
- Lhorente, J.P.; Araneda, M.; Neira, R.; Yáñez, J.M. Advances in genetic improvement for salmon and trout aquaculture: The Chilean situation and prospects. Rev. Aquac. 2019, 11, 340–353. [Google Scholar] [CrossRef]
- Andriantahina, F.; Liu, X.; Huang, H. Genetic map construction and quantitative trait locus (QTL) detection of growth-related traits in Litopenaeus vannamei for selective breeding applications. PLoS ONE 2013, 8, e75206. [Google Scholar] [CrossRef]
- Gjedrem, T.; Baranski, M. Selective Breeding in Aquaculture: An Introduction; Springer: New York, NY, USA, 2009. [Google Scholar]
- Zhou, J.; Ma, A.; Wang, X. Advances in fish breeding techniques. Fish. Inf. Strateg. 2013, 28, 199–207. (In Chinese) [Google Scholar]
- Okamoto, N.; Tayama, T.; Kawanobe, M.; Fujiki, N.; Yasuda, Y.; Sano, T. Resistance of a rainbow trout strain to infectious pancreatic necrosis. Aquaculture 1993, 117, 71–76. [Google Scholar] [CrossRef]
- Thodesen, J.; Grisdale-Helland, B.; Helland, S.J.; Gjerde, B. Feed intake, growth and feed utilization of offspring from wild and selected Atlantic salmon (Salmo salar). Aquaculture 1999, 180, 237–246. [Google Scholar] [CrossRef]
- Kause, A.; Ritola, O.; Paananen, T.; Wahlroos, H.; Mäntysaari, E.A. Genetic trends in growth, sexual maturity and skeletal deformations, and rate of inbreeding in a breeding programme for rainbow trout (Oncorhynchus mykiss). Aquaculture 2005, 247, 177–187. [Google Scholar] [CrossRef]
- Storset, A.; Strand, C.; Wetten, M.; Kjøglum, S.; Ramstad, A. Response to selection for resistance against infectious pancreatic necrosis in Atlantic salmon (Salmo salar L.). Aquaculture 2007, 272, S62–S68. [Google Scholar] [CrossRef]
- Moav, R.; Wohlfarth, G. Two-way selection for growth rate in the common carp (Cyprinus carpio L.). Genetics 1976, 82, 83–101. [Google Scholar] [CrossRef] [PubMed]
- Bondari, K. Response to bidirectional selection for body weight in channel catfish. Aquaculture 1983, 33, 73–81. [Google Scholar] [CrossRef]
- Liu, H.; Guan, B.; Xu, J.; Hou, C.; Tian, H.; Chen, H. Genetic manipulation of sex ratio for the large-scale breeding of YY super-male and XY all-male yellow catfish (Pelteobagrus fulvidraco). Mar. Biotechnol. 2013, 15, 321–328. [Google Scholar] [CrossRef]
- Liu, P.; Chen, X.; Zhong, L. Comparison of growth performance of different families of yellow catfish (Pelteobagrus fulvidraco). Nanjing Norm. Univ. Acta 2013, 36, 90–93. (In Chinese) [Google Scholar]
- Qin, Q.; Wang, M.; Chen, X. Comparative study on immune indexes of different yellow catfish family groups after injection of Aeromonas hydrophila. Freshw. Fish. 2017, 47, 40–46. (In Chinese) [Google Scholar]
- Hackett, P.B.; Alvarez, M.C. The molecular genetics of transgenic fish. Adv. Mar. Biol. 2000, 4, 77–145. [Google Scholar]
- Devlin, R.H.; Vandersteen, W.E.; Uh, M.; Stevens, E.D. Genetically modified growth affects allometry of eye and brain in salmonids. Can. J. Zool. 2012, 90, 193–202. [Google Scholar] [CrossRef]
- Devlin, R.H.; Biagi, C.A.; Yesaki, T.Y.; Smailus, D.E.; Byatt, J.C. Growth of domesticated transgenic fish. Nature 2001, 409, 781–782. [Google Scholar] [CrossRef] [PubMed]
- Rahman, M.A.; Ronyai, A.; Engidaw, B.Z.; Jauncey, K.; Hwang, G.L.; Smith, A.; Roderick, E.; Penman, D.; Varadi, L.; Maclean, N. Growth and nutritional trials on transgenic Nile tilapia containing an exogenous fish growth hormone gene. J. Fish Biol. 2005, 59, 62–78. [Google Scholar] [CrossRef]
- Lu, J.; Du, W.; Pu, W. Study on breeding technology of transgenic yellow catfish. Sci. Breed Fish 2011, 11, 10. (In Chinese) [Google Scholar]
- Liang, M.; Zhao, B.; You, Y. A preliminary study on the transformation of Clarias fuscus total DNA in catfish long-snout catfish. Southwest China J. Agric. Sci. 1994, 1, 97–101. (In Chinese) [Google Scholar]
- Ge, J.; Song, W.; Dong, Z. Establishment of the first originator of “whole fish” transgenic growth hormone gene in yellow catfish Pelteobagrus fulvidraco. J. Nanjing Univ. Nat. Sci. 2013, 49, 123–131. (In Chinese) [Google Scholar]
- Zhang, G.; Li, J.; Zhang, J.; Liang, X.; Wang, T.; Yin, S. A high-density SNP-based genetic map and several economic traits-related loci in Pelteobagrus vachelli. BMC Genom. 2020, 21, 700. [Google Scholar] [CrossRef]
- Ormandy, E.H.; Dale, J.; Griffin, G. Genetic engineering of animals: Ethical issues, including welfare concerns. Can. Vet. J. 2011, 52, 544. [Google Scholar]
- Zhou, L.; Gui, J. Natural and artificial polyploids in aquaculture. Aquac. Fish. 2017, 2, 103–111. [Google Scholar] [CrossRef]
- Yang, C.; Han, S. Triploid induction of yellow catfish Pelteobagrus fulvidraco. J. Anhui Agric. Sci. 2008, 19, 8118–8119. (In Chinese) [Google Scholar]
- Song, L.; Wang, W.; Zhou, X. Comparative study on triploid induction of yellow catfish (Pelteobagrus fulvidraco) by cold and hot shock methods. J. Fish. 2010, 34, 768–776. (In Chinese) [Google Scholar]
- Li, Y.; Dong, S.; Chen, L. Comparative study on artificial induction of triploid of yellow catfish (Pelteobagrus fulvidraco). J. Tianjin Norm. Univ. 2012, 32, 84–87. (In Chinese) [Google Scholar]
- Avise, J.C. Evolutionary perspectives on clonal reproduction in vertebrate animals. Proc. Natl. Acad. Sci. USA 2015, 112, 8867–8873. [Google Scholar] [CrossRef]
- Gui, J. Artificial propagation and seedling breeding technology of allogynogenetic Carassius auratus gibelio “Zhongke 3”. Rural. Breed. Technol. 2011, 2011, 41–42. (In Chinese) [Google Scholar]
- Sundin, K.; Brown, K.H.; Drew, R.E. Genetic analysis of a development rate QTL in backcrosses of clonal rainbow trout, Oncorhynchus mykiss. Aquaculture 2005, 247, 75–83. [Google Scholar] [CrossRef]
- Gong, C.; Yin, R.; Sun, G. Comparative study on the cultivation of large-scale seedlings of “North Flounder No. 1” and “North Flounder No. 2” and wild flounder. Sci. Breed Fish 2017, 2017, 47–49. (In Chinese) [Google Scholar]
- Xu, J.; You, F.; Wu, X. Artificial induction of diploid gynogenesis in large yellow croaker. Mar. Sci. 2006, 12, 37–42. (In Chinese) [Google Scholar]
- Wu, Q.; Chen, R.; Ye, Y. Study on artificial gynogenesis of carp and its application as a new way to establish inbred line. Genetics 1981, 1, 50–55. (In Chinese) [Google Scholar]
- Mao, H.; Zhou, L.; Gui, J. RAPD analysis of two artificial gynogenetic families of silver carp. Acta Hydrobiol. Sin. 2003, 5, 547–550. (In Chinese) [Google Scholar]
- Zhang, H. Establishment and Main Biological Characteristics of Gynogenetic Grass Carp Population. Ph.D. Thesis, Hunan Normal University, Changsha, China, 2011. (In Chinese). [Google Scholar]
- Yang, N. Studies on Distant Hybridization and Gynogeny of Tilapia (Oreochromis niloticus). Master’s Thesis, Shanghai Ocean University, Shanghai, China, 2020. (In Chinese). [Google Scholar]
- Zhang, X. Study on the Induction of Triploid and Gynogenesis and Its Main Biological Characteristics of Megalobrama amblycephala. Master’s Thesis, Huazhong Agricultural University, Wuhan, China, 2013. (In Chinese). [Google Scholar]
- Jia, F.; Wang, J.; Wu, Q. Artificial gynogenesis induced by heterologous spermatozoa in Gobio rarei. Acta Hydrobiol. Sin. 2002, 3, 246–252. (In Chinese) [Google Scholar]
- Liu, H.; Hou, J.; Chang, Y. Meiotic gynogenesis of flounder meiosis induced by red bream spermatozoa. J. Fish. 2010, 34, 508–514. (In Chinese) [Google Scholar]
- Xu, K.; Duan, W.; Xiao, J. Application and research progress of biological methods in fish genetics and breeding. Sci. China Life Sci. 2014, 44, 1272–1288. (In Chinese) [Google Scholar]
- Lincoln, R.F.; Scott, A.P. Production of all-female triploid rainbow trout. Aquaculture 1983, 30, 375–380. [Google Scholar] [CrossRef]
- Benfey, T. Use of sterile triploid Atlantic salmon (Salmo salar L.) for aquaculture in New Brunswick, Canada. ICES J. Mar. Sci. 2001, 58, 525–529. [Google Scholar] [CrossRef]
- Cherfas, N.B.; Gomelsky, B.; Ben-Dom, N.; Peretz, Y.; Hulata, G. Assessment of triploid common carp (Cyprinus carpio L.) for culture. Aquaculture 1994, 127, 11–18. [Google Scholar] [CrossRef]
- Wu, C.; Huang, X.; Chen, Q.; Hu, F.; Zhou, L.; Gong, K.; Fu, W.; Gong, D.; Zhao, R.; Zhang, C.; et al. The formation of a new type of hybrid culter derived from a hybrid lineage of Megalobrama amblycephala (♀) × Culter alburnus (♂). Aquaculture 2020, 525, 735328. [Google Scholar] [CrossRef]
- Wu, Q. Study on Artificial Induction of Polyploid of Yellow Catfish (Pelteobagrus fulvidraco). Master’s Thesis, Huazhong Agricultural University, Wuhan, China, 2010. (In Chinese). [Google Scholar]
- Tong, J.; Sun, X. Genetic and genomic analyses for economically important traits and their applications in molecular breeding of cultured fish. Sci. China Life Sci. 2015, 58, 178–186. [Google Scholar] [CrossRef]
- Eze, F. Marker-assisted selection in fish: A review. Asian J. Fish. Aquat. Res. 2019, 3, 1–11. [Google Scholar] [CrossRef]
- Wang, C.; Xie, S.; Zhu, X.; Lei, W.; Yang, Y.; Liu, J. Effects of age and dietary protein level on digestive enzyme activity and gene expression of Pelteobagrus fulvidraco larvae. Aquaculture 2006, 254, 554–562. [Google Scholar] [CrossRef]
- Wringe, B.F.; Devlin, R.H.; Ferguson, M.M.; Moghadam, H.K.; Sakhrani, D.; Danzmann, R.G. Growth-related quantitative trait loci in domestic and wild rainbow trout (Oncorhynchus mykiss). BMC Genet. 2010, 11, 63. [Google Scholar] [CrossRef]
- Liu, F.; Sun, F.; Xia, J.H.; Li, J.; Fu, G.H.; Lin, G.; Tu, R.J.; Wan, Z.Y.; Quek, D.; Yue, G.H. A genome scan revealed significant associations of growth traits with a major QTL and GHR2 in tilapia. Sci. Rep. 2014, 4, 7256. [Google Scholar] [CrossRef] [PubMed]
- Küttner, E.; Moghadam, H.K.; Skúlason, S.; Danzmann, R.G.; Ferguson, M.M. Genetic architecture of body weight, condition factor and age of sexual maturation in Icelandic Arctic charr (Salvelinus alpinus). Mol. Genet. Genom. 2011, 286, 67–79. [Google Scholar] [CrossRef] [PubMed]
- Gutierrez, A.P.; Yáñez, J.M.; Fukui, S.; Swift, B.; Davidson, W.S. Genome-wide association study (GWAS) for growth rate and age at sexual maturation in Atlantic salmon (Salmo salar). PLoS ONE 2015, 10, e0119730. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Fan, C.; Liu, Y.; Zhang, Y.; Liu, S.; Sun, D.; Deng, H.; Xu, Y.; Tian, Y.; Liao, X.; et al. A genome scan for quantitative trait loci associated with Vibrio anguillarum infection resistance in Japanese flounder (Paralichthys olivaceus) by bulked segregant analysis. Mar. Biotechnol. 2014, 16, 513–521. [Google Scholar] [CrossRef]
- Zhou, T.; Yuan, Z.; Tan, S.; Jin, Y.; Yang, Y.; Shi, H.; Wang, W.; Niu, D.; Gao, L.; Jiang, W.; et al. A review of molecular responses of catfish to bacterial diseases and abiotic stresses. Front. Physiol. 2018, 9, 1113. [Google Scholar] [CrossRef]
- Zhu, Y.; Liang, H.; Li, Z. Correlation analysis of MSTN gene polymorphism and growth traits in yellow catfish (Pelteobagrus fulvidraco). Genetics 2012, 34, 72–78. [Google Scholar]
- Zhu, C.; Liu, H.; Pan, Z. Construction of a high-density genetic linkage map and QTL mapping for growth traits in Pseudobagrus ussuriensis. Aquaculture 2019, 511, 734213. [Google Scholar] [CrossRef]
- Zhu, C.; Pan, Z.; Wang, H. A Microsatellite Marker and Specific Primers for the Identification of Female and Male Pseudobagrus ussuriensis and Its Application. Patent CN106811540B, 23 June 2020. (In Chinese). [Google Scholar]
- Ge, X.; Yin, H.; Bi, B. Genetic map construction and QTL mapping of growth-related traits in yellow catfish (Pelteobagrus fulvidraco). J. Fish. 2010, 34, 185–193. (In Chinese) [Google Scholar]
- Lu, C.; Sun, X.; Liang, L. Genetic differences between male and female of yellow catfish were analyzed by AFLP. J. Fish. 2007, 20, 24–28. [Google Scholar]
- Gui, J.; Wang, D.; Mao, H. Methods for Sex Chromosome Specific Molecular Markers and Genetic Sex Identification of Yellow Catfish (Pelteobagrus fulvidraco). Patent CN200810236650, 29 April 2009. (In Chinese). [Google Scholar]
- Wang, D.; Mao, H.; Chen, H. Isolation of Y-and X-linked SCAR markers in yellow catfish and application in the production of all-male populations. Anim. Genet. 2009, 40, 978–981. [Google Scholar] [CrossRef]
- Xin, W.; Sun, Z.; Yin, H. SRAP markers of male and female differences in yellow catfish (Pelteobagrus fulvidraco). J. Northeast. For. Univ. 2009, 37, 112–113. (In Chinese) [Google Scholar]
- Xu, H. A Comparative Study on Genetic Diversity among Different Populations of Bagrid (Pseudobagrus ussuriensis). Master’s Thesis, Soochow University, Suzhou, China, 2011. (In Chinese). [Google Scholar]
- Zhu, C.; Pan, Z.; Wang, H. A novel male-associated marker for sex identification in Pseudobagrus ussuriensis. J. Oceanol. Limnol. 2018, 036, 850–852. [Google Scholar] [CrossRef]
- Zhu, C.; Liu, H.; Cheng, L. Identification of sex-specific sequences through 2b-RAD sequencing in Pseudobagrus ussuriensis. Aquaculture 2021, 539, 736639. [Google Scholar] [CrossRef]
- Zhang, T.; Liu, S.; Lv, M. Application of related sequence amplified polymorphism (SRAP) in crop genetics and breeding. Mod. Agric. Sci. Technol. 2010, 11, 79–82. (In Chinese) [Google Scholar]
- Zhang, G.; Yin, S.; Mao, J.; Liang, F.; Zhao, C.; Li, P.; Zhou, G.; Chen, S.; Tang, Z. Integrated analysis of mRNA-seq and miRNA-seq in the liver of Pelteobagrus vachelli in response to hypoxia. Sci. Rep. 2016, 6, 22907. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Yan, A.; Zhang, Z.; Zha, J.; Bai, S.; Wu, J. Study on the hybridization of yellow catfish (Pelteobagrus fulvidraco) and yellow catfish (Pelteobagrus vagri). Freshw. Fish. 2002, 03, 3–5. (In Chinese) [Google Scholar]
- Wang, M.B.; Chen, Q.; Chen, Y.B.; Guo, C. Hybridization study of Pelteobagrus fulvidraco and Pelteobagrus vachelli. Mod. Agric. Sci. Technol. 2012, 24, 273–278. (In Chinese) [Google Scholar]
- Wang, F. Morphological difference analysis of F1 generation in Pelteobagrus fulvidraco, Pelteobagrus vagri, Leiocassis longirostris, and their hybrid. Chin. Agric. Bull. 2013, 29, 36–43. (In Chinese) [Google Scholar]
- Wang, F.; Wang, W. Early hybrid breeding of P. fulvidraco, P. vachelli and L. crassilabrus. Aquat. Sci. Technol. Inf. 2004, 1, 10–11. (In Chinese) [Google Scholar]
- Hu, F.; Fan, J.; Qin, Q.; Huo, Y.; Wang, Y.; Wu, C.; Liu, Q.; Li, W.; Chen, X.; Liu, C.; et al. The sterility of allotriploid fish and fertility of female autotriploid fish. Front. Genet. 2019, 10, 377. [Google Scholar] [CrossRef] [PubMed]
- Li, R.; Han, L.; Ai, L. Analysis on growth and morphological differences of two hybrid yellow catfish species. Aquaculture 2016, 37, 29–35. (In Chinese) [Google Scholar]
- Pei, X. Studies on Physiological Response and Gene Expression of Hybrid Yellow Catfish “Huangyou 1” to Hypoxia Stress. Master’s Thesis, Nanjing Normal University, Nanjing, China, 2020. (In Chinese). [Google Scholar]
- Sun, J.; Han, G.; Liu, Y. Comparison of growth performance and hypoxia tolerance between hybrid yellow catfish (Pelteobagrus fulvidraco × Pelteobagrus vachelli) and yellow catfish (Pelteobagrus fulvidraco). Acta Hydrobiol. Sin. 2019, 43, 1271–1279. (In Chinese) [Google Scholar]
- Xu, Z.; Zhang, H.; Xu, A. Comparison of breeding of hybrid yellow catfish and all-male yellow catfish. Sci. Fish Farming 2017, 9, 12. (In Chinese) [Google Scholar]
- Zhang, G.; Li, J.; Zhang, J.; Liang, X.; Zhang, X.; Wang, T.; Yin, S. Integrated analysis of transcriptomic, mirna and proteomic changes of a novel hybrid yellow catfish uncovers key roles for mirnas in heterosis. Mol. Cell. Proteom. 2019, 18, 1437–1453. [Google Scholar] [CrossRef]
- Zhang, G.; Liang, X.; Zhang, J.; Li, J.; Yin, S. The complete mitochondrial genome of the hybrid of Leiocassis longirostris (♂) × Pelteobagrus fulvidraco (♀). Mitochondrial DNA Part B 2017, 2, 577–578. [Google Scholar] [CrossRef]
- Zhang, J.; Li, J.; Zhang, G. Microsatellite analysis of genetic diversity of hybrid (P. fulvidraco♀ × P. vachelli♂) and its parents. Fish. Sci. 2018, 37, 612–621. (In Chinese) [Google Scholar]
- Zhou, H.; Duan, G.; Hu, Y.; Wang, H.; Jiang, H.; Ling, J.; Pan, T.; Chen, X. Morphological differences of hybrid yellow catfish with body color differentiation. Fish. Sci. 2021, 40, 1–11. (In Chinese) [Google Scholar]
- Yin, L.; Zhao, Y.; Zhou, X.Q.; Yang, C.; Feng, L.; Liu, Y.; Jiang, W.D.; Wu, P.; Zhou, J.; Zhao, J.; et al. Effect of dietary isoleucine on skin mucus barrier and epithelial physical barrier functions of hybrid bagrid catfish Pelteobagrus vachelli × Leiocassis longirostris. Fish Physiol. Biochem. 2020, 46, 1759–1774. [Google Scholar] [CrossRef]
- Zhao, Y.; Yan, M.Y.; Jiang, Q.; Yin, L.; Zhou, X.Q.; Feng, L.; Liu, Y.; Jiang, W.D.; Wu, P.; Zhao, J.; et al. Isoleucine improved growth performance, and intestinal immunological and physical barrier function of hybrid catfish Pelteobagrus vachelli × Leiocassis longirostris. Fish Shellfish. Immunol. 2021, 109, 20–33. [Google Scholar] [CrossRef]
- Pan, Z.J.; Zhu, C.K.; Wang, H.; Chang, G.L.; Ding, H.Y.; Qiang, X.G.; Yu, X.S. Induction of meiotic gynogenesis in bagrid catfish (Pseudobagrus ussuriensis) with homologous sperm and its confirmation for female homogamety. Aquac. Res. 2017, 48, 5659–5665. [Google Scholar] [CrossRef]
- Cai, Y.; Chen, Y.; Chen, X. Morphological difference of F-1 generation in Pelteobagrus vachelli and Pseudobagrus ussuriensis. Fish. Sci. 2011, 23, 264–270. (In Chinese) [Google Scholar]
- Chen, Y.; Chen, X.; Pan, Y.; Wang, M.; Qin, Q.; Deng, Y.; Cai, Y. RAPD analysis of genetic variation of hybrid catfish Pelteobagrus vachelli (♀) × Leiocassis longirostris (♂). J. Shanghai Ocean. Univ. 2010, 19, 12–18. (In Chinese) [Google Scholar]
- Dong, S.; Sun, J.; Bi, X. Changes in body length of P. fulvidraco (♀) × Pseudobagrus ussuriensis (♂) hybrid progeny during embryonic development and 30 days after embryo. Jiangsu Agric. Sci. 2016, 45, 313–315. (In Chinese) [Google Scholar]
- Shi, W.; Chen, Y.; Zhu, C. Study on hybridization effect of P. fulvidraco and P.ussuriensis. Aquac. Res. 2016, 3, 63–70. (In Chinese) [Google Scholar]
- Wei, G. Preliminary study on hybridization in Leiocassis longirostris × Pelteobagrus fulvidraco. Freshw. Fish. 1987, 6, 14–17. (In Chinese) [Google Scholar]
- He, Y.; Zeng, J.; Feng, J.; He, J. Hybrid breeding experiment of P. vachelli (♀) × Leiocassis crassirostris (♂). Aquaculture 2008, 5, 33–34. (In Chinese) [Google Scholar]
- Qiu, C. Seedling breeding and breeding test of P. fulvidraco (♀) × Pseudobagrus ussuriensis (♂). Sci. Technol. Inf. 2009, 4, 313–314. (In Chinese) [Google Scholar]
- Qin, Q.; Liang, D.; Wang, M. The study of embryonic development in Pelteobagrus vachelli and Pseudobagrus ussuriensis. J. Nanjing Norm. Univ. 2012, 35, 81–86. (In Chinese) [Google Scholar]
- Guo, S.; Luo, X.; Liang, H. Mitochondrial DNA sequence of the hybrid of Leiocassis longirostris (♀) and Pelteobagrus fulvidraco (♂). Mitochondrial DNA Part B Resour. 2016, 1, 166–167. [Google Scholar] [CrossRef][Green Version]
- Qiang, J.; Tao, F.; Bao, W.; He, J.; Liang, M.; Liang, C.; Zhu, H.; Li, X.; Chen, D.; Xu, P. miR-489-3p regulates the oxidative stress response in the liver and gill tissues of hybrid yellow catfish (Pelteobagrus fulvidraco female symbol × P. vachellimale symbol) under Cu (2+) exposure by targeting Cu/Zn-SOD. Front. Physiol. 2019, 10, 868. [Google Scholar] [CrossRef] [PubMed]
- Chu, Z.; Guo, W.; Hu, W.; Mei, J. Delayed elimination of paternal mtDNA in the interspecific hybrid of Pelteobagrus fulvidraco and Pelteobagrus vachelli during early embryogenesis. Gene 2019, 704, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Wang, W. A Preliminary Study on Hybrid Breeding of P. fulvidraco and P. vachelli. Ph.D. Thesis, Huazhong Agricultural University, Wuhan, China, 2003. (In Chinese). [Google Scholar]
- Zhao, W.; Yang, X.; Peng, Z.; Peng, Z.; Liu, Y. Molecular identification of the four species of the genus Pelteobagrus and the genetic analysis of their hybrid. Acta Hydrobiol. Sin. 2006, 30, 101–106. (In Chinese) [Google Scholar]
- Shapawi, R.; Abdullah, F.C.; Senoo, S.; Mustafa, S. Nutrition, growth and resilience of tiger grouper Epinephelus fuscoguttatus × giant Grouper Epinephelus lanceolatus hybrid—A review. Rev. Aquac. 2019, 11, 1285–1296. [Google Scholar] [CrossRef]
- Jiang, H.B.; Bao, J.; Han, Y. Mitochondrial DNA sequence of the hybrid of Takifugu flavidus (♀) × Takifugu rubripes (♂). Mitochondrial DNA Part A 2016, 27, 2117–2118. [Google Scholar] [CrossRef]
- Shao, W. A Preliminary Analysis on the Hybridization Superiority of Yellow Catfish “Huangyou No.1” by Using High-throughput Sequencing Technology. Master’s Thesis, Huazhong Agricultural University, Wuhan, China, 2018. (In Chinese). [Google Scholar]
- Bao, M.; Feng, J.; Chen, X.; Liu, Z. Artificial propagation technology of all-male yellow catfish (Pelteobagrus fulvidraco). Sci. Fish Breed 2015, 31, 8–9. (In Chinese) [Google Scholar]
- Ye, J.; Dong, T.; Dong, X. Effects of stocking density on yield and benefit of all-male and common yellow catfish (Pelteobagrus fulvidraco) culture. Aquat. Sci. Technol. Inf. 2014, 41, 61–64. (In Chinese) [Google Scholar]
- He, G.; Chen, D. Study on seedling breeding technology of all-male yellow catfish (Pelteobagrus fulvidraco). Hebei Fish. 2016, 2, 37–38. (In Chinese) [Google Scholar]
- Hu, D.; Zhou, Z.; Zhu, Q. Evaluation of resistance ability of hybrid yellow catfish and common yellow catfish to catfish Edwardsiella. Sci. Breed Fish 2016, 2016, 54–55. (In Chinese) [Google Scholar]
- Zhao, Y.; Wu, X.Y.; Xu, S.X.; Xie, J.Y.; Xiang, K.W.; Feng, L.; Liu, Y.; Jiang, W.D.; Wu, P.; Zhao, J.; et al. Dietary tryptophan affects growth performance, digestive and absorptive enzyme activities, intestinal antioxidant capacity, and appetite and GH-IGF axis-related gene expression of hybrid catfish (Pelteobagrus vachelli female symbol × Leiocassis longirostris male symbol). Fish Physiol. Biochem. 2019, 45, 1627–1647. [Google Scholar]
- Zhao, Y.; Jiang, Q.; Zhou, X.Q.; Xu, S.X.; Feng, L.; Liu, Y.; Jiang, W.D.; Wu, P.; Zhao, J.; Jiang, J. Effect of dietary threonine on growth performance and muscle growth, protein synthesis and antioxidant-related signalling pathways of hybrid catfish Pelteobagrus vachelli female symbol × Leiocassis longirostris male symbol. Br. J. Nutr. 2020, 123, 121–134. [Google Scholar] [CrossRef] [PubMed]
- Li, M.X.; Qiang, J.; Bao, J.W.; Tao, Y.F.; Zhu, H.J.; Xu, P. Growth performance, physiological parameters, and transcript levels of lipid metabolism-related genes in hybrid yellow catfish (Tachysurus fulvidraco female symbol × Pseudobagrus vachellii male symbol) fed diets containing Siberian ginseng. PLoS ONE 2021, 16, e0246417. [Google Scholar]
- Qiang, J.; Tao, F.; Bao, W.; He, J.; Li, X.; Chen, J.; Xu, P. Responses of functional miRNA-mRNA regulatory modules to a high-fat diet in the liver of hybrid yellow catfish (P. fulvidraco × P. vachelli). Genomics 2021, 113 Pt 2, 1207–1220. [Google Scholar] [CrossRef] [PubMed]
- Xu, K.; Duan, W.; Xiao, J.; Tao, M.; Zhang, C.; Liu, Y.; Liu, S. Development and application of biological technologies in fish genetic breeding. Sci. China Life Sci. 2015, 58, 187–201. [Google Scholar] [CrossRef]
- Groeneveld, L.F.; Lenstra, J.A.; Eding, H.; Toro, M.A.; Scherf, B.; Pilling, D.; Negrini, R.; Finlay, E.K.; Jianlin, H.; Groeneveld, E.J.A.G.; et al. Genetic diversity in farm animals—A review. Anim. Genet. 2010, 41, 6–31. [Google Scholar] [CrossRef]
- Liu, S.; Li, Y.; Qin, Z.; Geng, X.; Bao, L.; Kaltenboeck, L.; Kucuktas, H.; Dunham, R.; Liu, Z. High-density interspecific genetic linkage mapping provides insights into genomic incompatibility between channel catfish and blue catfish. Anim. Genet. 2016, 47, 81–90. [Google Scholar] [CrossRef]
- Mora, C.; Menozzi, D.; Kleter, G.; Aramyan, L.H.; Valeeva, N.I.; Reddy, G.P. Factors affecting the adoption of genetically modified animals in the food and pharmaceutical chains. Bio-Based Appl. Econ. 2012, 1, 313–329. [Google Scholar]
- Devlin, R.H.; D’Andrade, M.; Uh, M.; Biagi, C.A. Population effects of growth hormone transgenic coho salmon depend on food availability and genotype by environment interactions. Proc. Natl. Acad. Sci. USA 2004, 101, 9303–9308. [Google Scholar] [CrossRef]
- Myhr, A.I.; Dalmo, R.A. Introduction of genetic engineering in aquaculture: Ecological and ethical implications for science and governance. Aquaculture 2005, 250, 542–554. [Google Scholar] [CrossRef]
- Zhong, X.; Wang, X.; Zhou, T.; Jin, Y.; Tan, S.; Jiang, C.; Geng, X.; Li, N.; Shi, H.; Zeng, Q.; et al. Genome-wide association study reveals multiple novel QTL associated with low oxygen tolerance in hybrid catfish. Mar. Biotechnol. 2017, 19, 379–390. [Google Scholar] [CrossRef]
- Jin, Y.; Zhou, T.; Geng, X.; Liu, S.; Chen, A.; Yao, J.; Jiang, C.; Tan, S.; Su, B.; Liu, Z. A genome-wide association study of heat stress-associated SNP s in catfish. Anim. Genet. 2017, 48, 233–236. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, N.; Thompson, S.; Glaser, M. Global aquaculture productivity, environmental sustainability, and climate change adaptability. Environ. Manag. 2019, 63, 159–172. [Google Scholar] [CrossRef] [PubMed]






| Authors Year | Species | Polyploid | Inducing Methods | Dose or Temperature | Processing Time | Duration | Results |
|---|---|---|---|---|---|---|---|
| Wu et al., 2010 [63] | P. fulvidraco | Triploid | Cold shock | 4 °C | 3 min after fertilization | 10 min | The triploid and relative emergence rates of embryos were 58% and 53%, respectively |
| Li et al., 2012 [45] | P. fulvidraco | Triploid | Cold shock | 4 °C | 3 min after fertilization | 15 min | Triploid induction rate was 63.3% and hatching rate was 82.3% |
| Song et al., 2010 [44] | P. fulvidraco | Triploid | Cold shock | 5 °C | 2 min after fertilization | 20 min | High deformity rate and mortality for fish fry |
| Song et al., 2010 [44] | P. fulvidraco | Triploid | Hot shock | 40 °C | 2 min after fertilization | 2 min | Triploid induction rate was as high as 60% in embryo and 40~50% in juvenile |
| Wu et al., 2010 [63] | P. fulvidraco | Triploid | Hot shock | 40.5 °C | 2 min after fertilization | 2 min | The triploid rate and relative emergence rate of embryos were 59% and 36%, respectively |
| Wu et al., 2010 [63] | P. fulvidraco | Tetraploid | Hot shock | 40.4 °C | 50 min after fertilization | 1.5–1.8 min | The tetraploid rate of embryos was 10.61–62.16%, the tetraploid rate of embryos in the membrane stage was 11.11–75%, and the relative emergence rate was 9.77–36.05% |
| Li et al., 2012 [45] | P. fulvidraco | Triploid | Hot shock | 40 °C | 8 min after fertilization | 2 min | Triploid induction rate was 93.3% and hatching rate was 78.6% |
| Li et al., 2012 [45] | P. fulvidraco | Triploid | Medication | 6—DMAP, CB | - | - | High triploid deformity rate, low hatching rate, low ploidy rate, not suitable for production |
| Yang et al., 2008 [43] | P. fulvidraco | Triploid | Medication | 6—DMAP | 10 min after fertilization | 15 min | Hatching rate is 0, not suitable for production |
| Wu et al., 2010 [63] | P. fulvidraco | Triploid | Hydrostatic shock | 550 kg/cm2 pressure | 5 min after fertilization | 5 min | Triploid rate and relative emergence rate of embryos were 55% and 54%, respectively |
| Authors Year | Species | Technologies | Correlated Traits | Research Contents | Results |
|---|---|---|---|---|---|
| Xu, 2011 [81] | P. ussuriensis | SRAP | Gender | The polymorphism of male and female genomic DNA was detected, and their gender related molecular markers were screened | Twelve pairs of primer combinations with clear bands, strong reproducibility, and good polymorphism were screened out |
| Zhu et al., 2018 [82] | P. ussuriensis | PCR; Sequencing | Gender | A total of 95 female and 90 male individuals were examined | A male-associated marker pUGT54 was isolated |
| Zhu et al., 2019 [74] | P. ussuriensis | 2b-RAD | Growth | A high-resolution genetic linkage map was constructed using 2b-RAD technique | A set of QTLs related to growth, body weight, head length, and body width were identified |
| Zhu et al., 2021 [83] | P. ussuriensis | 2b-RAD | Gender | The DNA sequences associated with restrictive endoscopic sites were sequenced to identify sex-specific sequences in 5 female and 5 male fish | Three male-specific markers and seven male-specific sequences were screened |
| Zhu et al., 2020 [75] | P. ussuriensis | PCR | Growth | Selection of sex-specific microsatellite loci in the microsatellite enrichment library | A sex-specific microsatellite locus was selected |
| Zhu et al., 2012 [73] | P. fulvidraco | PCR-SSCP | Growth | SNP detection and MSTN gene were conducted and the association between MSTN gene and growth shape was analyzed | The AA genotype was the favorable genotype and the DD genotype was the unfavorable genotype for the growth |
| Ge et al., 2010 [76] | P. fulvidraco | SSR, SRAP, TRAP | Growth | A genetic linkage map was constructed for QTL mapping of five growth-related traits | All three QTLs could be used for marker-assisted breeding for growth traits |
| Lu et al., 2007 [77] | P. fulvidraco | AFLP | Gender | Genetic differences between males and females were analyzed using 20 AFLP primer combinations | A total of 6 loci showed significant amplification differences between male among 4 AFLP primer combinations |
| Gui et al., 2009 [78] | P. fulvidraco | AFLP | Gender | The PCR method of chromosome genotype identification was established, and it played an important role in production of all-male yellow catfish | Two specific AFLP fragments that could produce X or Y chromosomes were screened and transformed into SCAR markers for sequencing |
| Wang et al., 2009 [79] | P. fulvidraco | AFLP | Gender | 256 pairs of AFLP primers were used to scan the gene pool of XX, XY, and YY | Four X linkage markers and two Y linkage markers were screened out |
| Xin et al., 2009 [80] | P. fulvidraco | SRAP | Gender | A SRAP-PCR amplification reaction system was established, which can be widely used in the genetic study of yellow catfish | One SRAP marker was found to be sex-specific in males and females |
| Gao et al., 2020 [5] | P. fulvidraco | SNP | Gender | Genotyping sequencing was constructed, and 5705 SNP markers were mapped to 26 different linkage groups | 11 significant related QTLs were identified and 6 sex-related genes were identified |
| Zhang et al., 2020 [40] | P. vachelli | SNP | Anti-stress | A high-resolution linkage map was constructed using 5059 DDRAD markers | Candidate genes for 12 growth, 1 sex determination, and 1 hypoxia tolerance related traits were constructed |
| Authors Year | Hybridized Combination | Survival Rate | Fertility Rate | Emergence Rate | Characteristics |
|---|---|---|---|---|---|
| Wang et al., 2002, 2012 [86,87] | P. Fulvidraco ♀ × P. vachelli ♂ | - | 87.5% | 71.6% | High hatching rate and fertilization rate, body color and shape similar to P. fulvidraco, and growth rate is higher than parents |
| Wang et al., 2004 [89] | P. vachelli ♀ × P. fulvidraco ♂ | 79% | 53% | 6.9% | The hybrid progeny had obvious maternal benefit, low fertilization rate, and slow growth rate, which was only 0.1144 g/d and was not suitable for production |
| Wang et al., 2013 [88] | P. fulvidraco ♀ × P. vachelli ♂ | 86.5% | 70.6% | 60.7% | The hybrid combination showed high heterosis, fast growth rate, absolute weight gain rate of 0.1534 g/d, and obvious maternal effect |
| Li et al., 2016 [91] | P. fulvidraco ♀ × P. vachelli ♂ | - | - | - | Growth speed is fast, and hybrid has the maternal effect |
| Xu et al., 2017 [94] | P. fulvidraco ♀ × P. vachelli ♂ | - | - | - | The hybrid progeny was superior to all-male yellow catfish in terms of species size and survival rate |
| Zhang et al., 2017, 2018 [96,97] | P. fulvidraco ♀ × P. vachelli ♂ | - | - | - | The optimal growth temperature and density of hybrid progeny were 28.95 °C and 1.927 kg/m³. Compared with the parents, the progeny had obvious growth advantages, high quality nutritional value, higher genetic diversity, and stronger disease resistance |
| Hu et al., 2019 [90] | P. fulvidraco ♀ × P. vachelli ♂ | - | - | - | Growth performance was significantly better than P. fulvidraco, and the phenomenon of hermaphroditic growth was significantly reduced. The female was completely sterile, and the sperm quantity in the testis of the male was low and the effective vitality was low |
| Sun et al., 2019 [93] | P. fulvidraco ♀ × P. vachelli ♂ | - | - | - | The hypoxic tolerance, average body weight, and survival rate were significantly higher than the female parent, and the feed conversion ratio was lower than the female parent |
| Zhang et al., 2019 [95] | P. fulvidraco ♀ × P. vachelli ♂ | - | - | - | Heterosis in immunity, metabolism, digestion, absorption, proliferation, and development |
| Pei et al., 2020 [92] | P. fulvidraco ♀ × P. vachelli ♂ | - | - | - | Hypoxic stress induces the upregulation of oxygen-sensing protein-related genes in the progenies of the hybrids and enhances the body’s antioxidant capacity to cope with oxidative stress injury caused by reoxygenation |
| Zhang et al., 2020 [40] | P. fulvidraco ♀ × P. vachelli ♂ | - | - | - | The hybrid progeny showed heterosis in growth, antimicrobial resistance, and nutrient composition |
| Zhou et al., 2021 [98] | P. fulvidraco ♀ × P. vachelli ♂ | - | - | - | The hybrid progeny showed significant differences in spine length, caudal stalk length, and eye diameter, and the main morphological characteristics of the progeny were more similar to female parent |
| Authors Year | Hybridized Combination | Survival Rate | Fertility Rate | Emergence Rate | Characteristics |
|---|---|---|---|---|---|
| Wei et al., 1987 [106] | P. Vachelli ♀ × L. longirostris ♂ | - | - | - | The hybrid progeny is between the two parents in shape and grows faster than female parents |
| Wang et al., 2004 [89] | P. Fulvidraco ♀ × L.crassilabrus ♂ | 74% | 64.6% | 11.0% | The hybrids were not suitable for production because of their low emergence rate and morphological characteristics tending to male parents |
| He et al., 2008 [107] | P. Vachelli ♀ × L. longirostris ♂ | - | 70.0% | - | Appearance, activity distribution, and body color similar to P. vachelli |
| Qiu, 2009 [108] | P. Fulvidraco ♀ × P.ussuriensis ♂ | 87% | - | - | The weight gain of hybrid offspring is significantly higher than P. ussuriensis, and there is no significant difference with P. fulvidraco |
| Chen et al., 2010 [103] | P. Vachelli ♀ × P.ussuriensis ♂ | - | - | - | The gene heterozygosity of the first generation of hybridization was enhanced, showing some heterosis |
| Cai et al., 2011 [102] | P. Vachelli ♀ × P. ussuriensis ♂ | - | - | - | The length and body weight of the hybrid progenies were greater than that of the backcrossing and self-crossing progenies |
| Cai et al., 2011 [102] | P.ussuriensis ♀ × P. vachelli ♂ | - | - | - | The body weight of the hybrid group had the superparent advantage |
| Qin et al., 2012 [109] | P.ussuriensis ♀ × P. vachelli ♂ | - | - | - | The embryo development time was between the two parents, and the full length of the hybrid was closer to female parent |
| Wang et al., 2013 [88] | P. Vachelli ♀ × L.crassilabrus ♂ | 76.5% | 81.0% | 72.9% | High fertilization rate and seedling emergence rate, fertilized eggs and strong larvae resistant to high temperature |
| Wang et al., 2013 [88] | P. Vachelli ♀ × P.ussuriensis ♂ | 59.5% | - | - | The absolute weight gain rate and specific growth rate of hybrid progeny are higher than that of self-crossing and lower than that of backcrossing |
| Wang et al., 2013 [88] | P.ussuriensis ♀ × P. vachelli ♂ | 62.5% | - | - | The absolute weight gain rate and specific growth rate of hybrid progeny were higher than that of parental self-crossing |
| Dong et al., 2016 [104] | P. Fulvidraco ♀ × L. longirostris ♂ | - | 92.67% | 79.67% | Earlier embryo development than the female parent. No significant difference in the length of self-mating body between fry and yellow catfish. |
| Guo et al., 2016 [110] | L. Longirostris ♀ × P. fulvidraco ♂ | - | - | - | The genetic distance is closer than that of both parents |
| Dong, 2016 [104] | P. Fulvidraco ♀ × P.ussuriensis ♂ | - | 92.67% | 79.67% | The embryo development was earlier than the female parent and later than the male parent. There was no significant difference in the body length of self-crossing with the female parent |
| Shi, 2016 [105] | P. Fulvidraco ♀ × P.ussuriensis ♂ | 88% | - | - | The fertility advantage of hybrid progeny was not significant |
| Li, 2016 [91] | P. Fulvidraco ♀ × P.ussuriensis ♂ | - | - | - | The hybrid population deviated from the all-male yellow catfish population, and the absolute weight gain rate was higher than the all-male yellow catfish population |
| Shi, 2016 [104] | P.ussuriensis ♀ × P. fulvidraco ♂ | 93% | - | - | The average weight advantage decreased with the increase in time |
| Dong, 2016 [104] | P.ussuriensis ♀ × P. fulvidraco ♂ | - | 89.33% | 67.33% | The embryo development was approximately 14 h shorter than the male parent self-crossing group. The body length was significantly longer than the female parent self-crossing group |
| Cultured Yellow Catfish | Hybrid Yellow Catfish | All-Male Yellow Catfish | |
|---|---|---|---|
| Breed type | Artificial (high proportion of close relatives) | The female of hybrid fish is sterile | YY crosses with XX |
| Growth rate | The adult male is 30% larger than the female, reaching 100~150 g at the second instar | The average growth rate of yellow catfish was 29.93% faster than that of yellow catfish | 30% faster than the average growth rate of normal yellow catfish |
| Food coefficient | 1.46~1.49 | 1.1~1.3 | 1.1~1.3 |
| Tolerance to low oxygen levels | 0.255 mg/g/h, Choke point = 0.314 mg/mL | 0.206 mg/h, Choke point = 0.27 mg/mL | 0.255 mg/g/h, Choke point = 0.314 mg/mL |
| Transportation | Not tolerant to long distance transportation | Tolerance to long distance transport | Tolerance to long distance transport |
| Specification uniformity | None specification (adult male 30% larger than female) | Specification | Specification (more than 94% of the individual size reached more than 100 g) |
| Fishing net rate | Low | High | Low |
| Optimum temperature range | 22~28 °C and 23~30 °C (breeding water temperature) | 20~32 °C and 28.4 °C (Optimum growth temperature) | 22~28 °C and 23~30 °C (breeding water temperature) |
| Production cost | Low | High | High |
| Survival rate | 33~52% | 89.9~90.3% | 53.3~70.8% |
| Ovulation ratio | 73.6% | - | - |
| Fertilization rate | 72.7% | 79.8% | >80% |
| Hatching rate | 76.8% | 82.5% | 76% |
| Culture cycle | Long | Short | Short |
| Meat quality | High fat content and meat content is 66.47~68.41% | Meat content is 76.52% | High fat content and meat content is 66.47~68.41% |
| Thickness of body skin | Thin | Thick | Thick |
| Disadvantages | Long breeding cycle and low disease resistance | Higher cost of seedlings and a certain rate of malformation | Low survival rate of splashing and high seedling cost |
| Advantages | Lower production cost and lower fry price | Short breeding cycle and strong resistance to disease | Short breeding cycle |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huang, L.; Zhang, G.; Zhang, Y.; Li, X.; Luo, Z.; Liu, W.; Luo, F.; Liu, H.; Yin, S.; Jiang, J.; et al. Profiling Genetic Breeding Progress in Bagrid Catfishes. Fishes 2023, 8, 426. https://doi.org/10.3390/fishes8080426
Huang L, Zhang G, Zhang Y, Li X, Luo Z, Liu W, Luo F, Liu H, Yin S, Jiang J, et al. Profiling Genetic Breeding Progress in Bagrid Catfishes. Fishes. 2023; 8(8):426. https://doi.org/10.3390/fishes8080426
Chicago/Turabian StyleHuang, Lishi, Guosong Zhang, Yupeng Zhang, Xinlan Li, Zhong Luo, Wenyu Liu, Fu Luo, Haifeng Liu, Shaowu Yin, Jun Jiang, and et al. 2023. "Profiling Genetic Breeding Progress in Bagrid Catfishes" Fishes 8, no. 8: 426. https://doi.org/10.3390/fishes8080426
APA StyleHuang, L., Zhang, G., Zhang, Y., Li, X., Luo, Z., Liu, W., Luo, F., Liu, H., Yin, S., Jiang, J., Liang, X., & Cao, Q. (2023). Profiling Genetic Breeding Progress in Bagrid Catfishes. Fishes, 8(8), 426. https://doi.org/10.3390/fishes8080426









