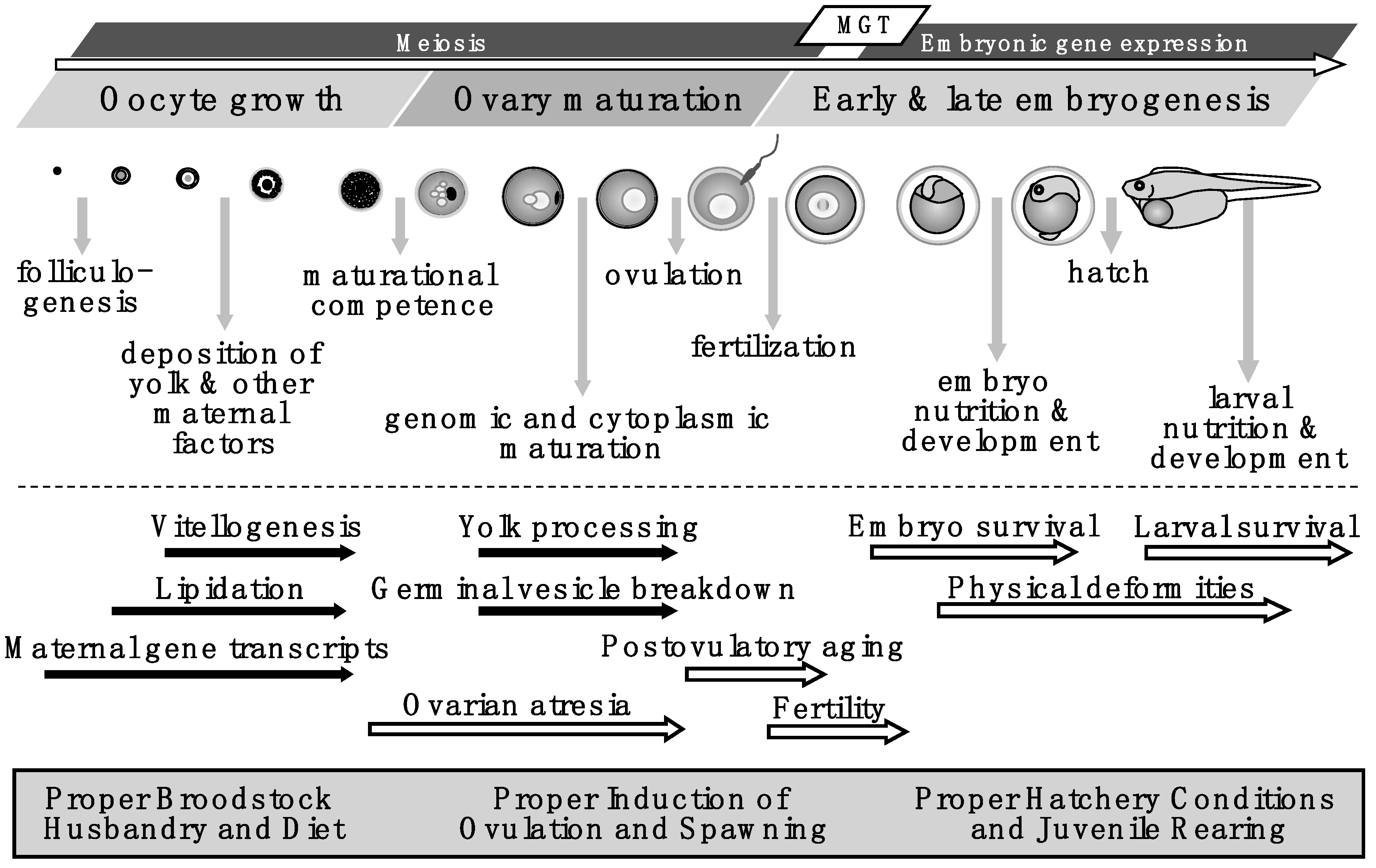
Oogenesis and Egg Quality in Finfish: Yolk Formation and Other Factors Influencing Female Fertility
Abstract
1. Introduction
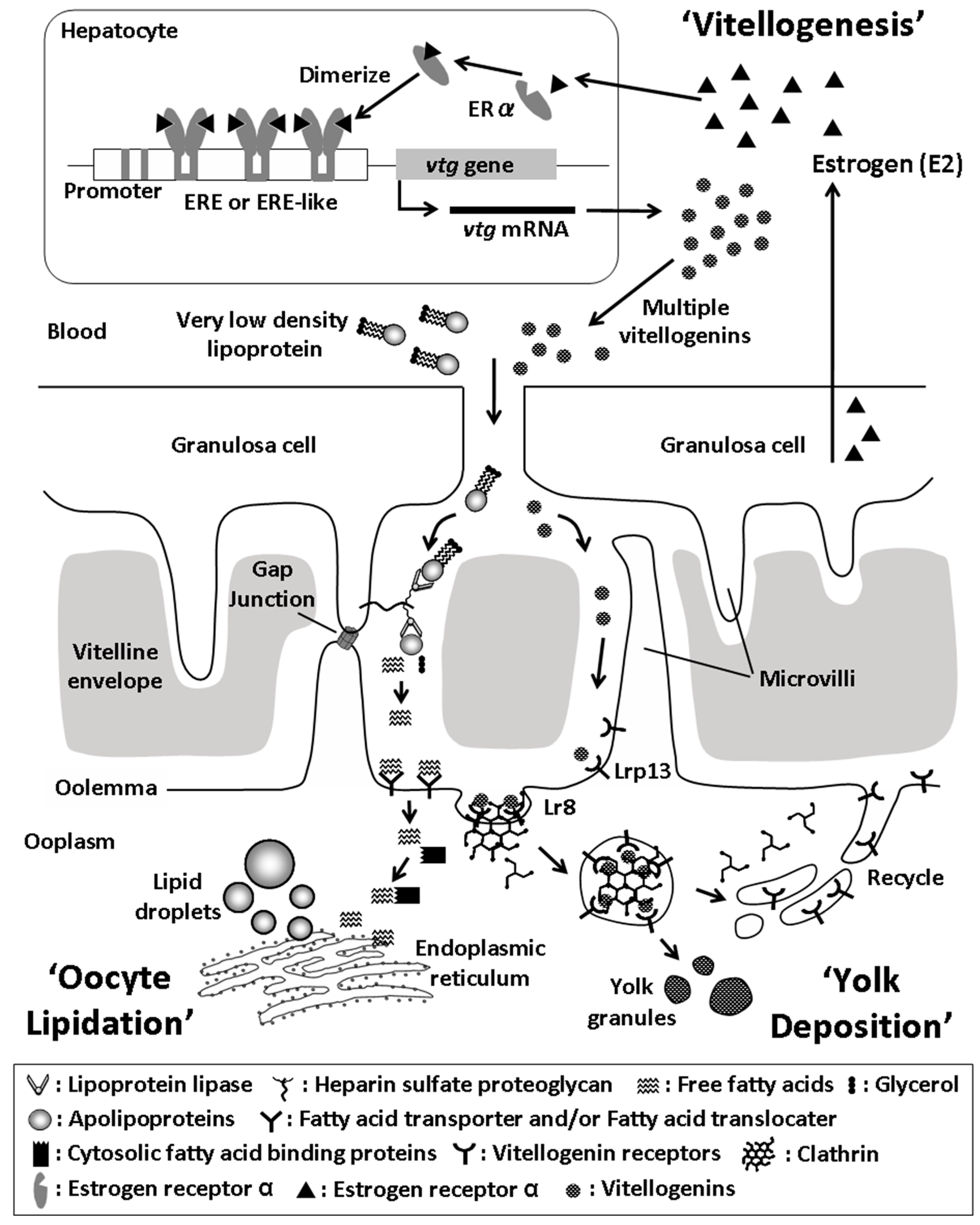
2. Ovarian Growth and Yolk Formation
3. Multiplicity of Fish Vitellogenins
4. Ovarian Maturation and Proteolysis of Yolk Proteins
5. Egg Yolk Composition and Broodstock Diet
6. Omics of Egg Quality and Maternal Gene Transcripts
7. Stress and Ovarian Atresia
8. Postovulatory Aging
9. Future Research Directions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bobe, J.; Labbé, C. Egg and sperm quality in fish. Gen. Comp. Endocrinol. 2009, 165, 535–548. [Google Scholar] [CrossRef] [PubMed]
- Chapman, R.W.; Reading, B.J.; Sullivan, C.V. Ovary transcriptome profiling via artificial intelligence reveals a transcriptomic fingerprint predicting egg quality in striped bass, Morone saxatilis. PLoS ONE 2014, 9, e96818. [Google Scholar] [CrossRef] [PubMed]
- Bobe, J. Egg quality in fish: Present and future challenges. Anim. Front. 2015, 5, 66–72. [Google Scholar] [CrossRef]
- Sullivan, C.V.; Chapman, R.W.; Reading, B.J.; Anderson, P.E. Transcriptomics of mRNA and egg quality in farmed fish: Some recent developments and future directions. Gen. Comp. Endocrinol. 2015, 221, 23–30. [Google Scholar] [CrossRef] [PubMed]
- Endo, T.; Todo, T.; Lokman, P.M.; Kudo, H.; Ijiri, S.; Adachi, S.; Yamauchi, K. Androgens and very low density lipoprotein are essential for the growth of previtellogenic oocytes from Japanese eel, Anguilla japonica, in vitro. Biol. Reprod. 2011, 84, 816–825. [Google Scholar] [CrossRef] [PubMed]
- Damsteegt, E.L.; Mizuta, H.; Hiramatsu, N.; Lokman, P.M. How does eggs get fats? Insights into ovarian fatty acid accumulation in the shortfinned eel, Anguilla australis. Gen. Comp. Endocrinol. 2015, 221, 94–100. [Google Scholar] [CrossRef] [PubMed]
- Hiramatsu, N.; Todo, T.; Sullivan, C.V.; Schilling, J.; Reading, B.J.; Matsubara, T.; Ryu, Y.W.; Mizuta, H.; Luo, W.; Nishimiya, O.; et al. Ovarian yolk formation in fishes: Molecular mechanisms underlying formation of lipid droplets and vitellogenin-derived yolk proteins. Gen. Comp. Endocrinol. 2015, 221, 9–15. [Google Scholar] [CrossRef] [PubMed]
- Lubzens, E.; Bobe, J.; Young, G.; Sullivan, C.V. Maternal investment in fish oocytes and eggs: The molecular cargo and its contributions to fertility and early development. Aquaculture 2017, 472, 107–143. [Google Scholar] [CrossRef]
- Ibáñez, A.J.; Peinado-Insurbe, J.; Sánchez, E.; Cerdá-Reverter, J.M.; Prat, F. Lipoprotein lipase (LPL) is highly expressed and active in the ovary of European sea bass (Dicentrarchus labrax L.), during gonadal development. Comp. Biochem. Physiol. A 2008, 150, 347–354. [Google Scholar] [CrossRef] [PubMed]
- Ryu, Y.-W.; Tanaka, R.; Kasahara, A.; Ito, Y.; Hiramatsu, N.; Todo, T.; Sullivan, C.V.; Hara, A. Molecular cloning and transcript expression of genes encoding two types of lipoprotein lipase in the ovary of cutthroat trout, Oncorhynchus clarkii. Zool. Sci. 2013, 30, 224–237. [Google Scholar] [CrossRef] [PubMed]
- Mushirobira, Y.; Nishimiya, O.; Nagata, J.; Todo, T.; Hara, A.; Reading, B.J.; Hiramatsu, N. Molecular cloning of vitellogenin gene promoters and in vitro and in vivo transcription profiles following estradiol-17β administration in the cutthroat trout. Gen. Comp. Endocrinol. 2018, 267, 157–166. [Google Scholar] [CrossRef] [PubMed]
- Davis, L.K.; Hiramatsu, N.; Hiramatsu, K.; Reading, B.J.; Matsubara, T.; Hara, A.; Sullivan, C.V.; Pierce, A.L.; Hirano, T.; Grau, E.G. Induction of three vitellogenins by 17β-estradiol with concurrent inhibition of the growth hormone-insulin-like growth factor 1 axis in a euryhaline teleost, the tilapia (Oreochromis mossambicus). Biol. Reprod. 2007, 77, 614–625. [Google Scholar] [CrossRef] [PubMed]
- Reading, B.J.; Hiramatsu, N.; Sullivan, C.V. Disparate binding of three types of vitellogenin to multiple forms of vitellogenin receptor in white perch. Biol. Reprod. 2011, 84, 392–399. [Google Scholar] [CrossRef] [PubMed]
- Reading, B.J.; Hiramatsu, N.; Schilling, J.; Molloy, K.T.; Glassbrook, N.; Mizuta, H.; Luo, W. Lrp13 is a novel vertebrate lipoprotein receptor that binds vitellogenins in teleost fishes. J. Lipid Res. 2014, 55, 2287–2295. [Google Scholar] [CrossRef] [PubMed]
- Reading, B.J.; Sullivan, C.V.; Schilling, J. Vitellogenesis in Fishes. In Reference Module in Life Sciences; Elsevier: Amsterdam, The Netherlands, 2017. [Google Scholar]
- Davail, B.; Pakdel, F.; Bujo, H.; Perazzolo, L.M.; Waclawek, M.; Schneider, W.J.; Le Menn, F. Evolution of oogenesis: The receptor for vitellogenin from the rainbow trout. J. Lipid Res. 1998, 39, 1929–1937. [Google Scholar] [PubMed]
- Prat, F.; Coward, K.; Sumpter, J.P.; Tyler, C.R. Molecular characterization and expression of two ovarian lipoprotein receptors in the rainbow trout, Oncorhynchus mykiss. Biol. Reprod. 1998, 58, 1146–1153. [Google Scholar] [CrossRef] [PubMed]
- Hiramatsu, N.; Chapman, R.W.; Lindzey, J.K.; Haynes, M.R.; Sullivan, C.V. Molecular characterization and expression of vitellogenin receptor from white perch (Morone americana). Biol. Reprod. 2004, 70, 1720–1730. [Google Scholar] [CrossRef] [PubMed]
- Mizuta, H.; Luo, W.; Ito, Y.; Mushirobira, Y.; Todo, T.; Hara, A.; Reading, B.J.; Sullivan, C.V.; Hiramatsu, N. Ovarian expression and localization of vitellogenin receptor with eight ligand binding repeats in the cutthroat trout (Oncorhynchus clarkii). Comp. Biochem. Physiol. Part B 2013, 166, 81–90. [Google Scholar] [CrossRef] [PubMed]
- Mushirobira, Y.; Mizuta, H.; Luo, W.; Todo, T.; Hara, A.; Reading, B.J.; Sullivan, C.V.; Hiramatsu, N. Molecular cloning and partial characterization of a low-density lipoprotein receptor-related protein 13 (Lrp13) involved in vitellogenin uptake in the cutthroat trout (Oncorhynchus clarkii). Mol. Reprod. Dev. 2015, 82, 986–1000. [Google Scholar] [CrossRef] [PubMed]
- Mizuta, H.; Mushirobira, Y.; Nagata, J.; Todo, T.; Hara, A.; Reading, B.J.; Sullivan, C.V.; Hiramatsu, N. Ovarian expression and localization of clathrin (Cltc) components in cutthroat trout, Oncorhynchus clarkii: Evidence for Cltc involvement in endocytosis of vitellogenin during oocyte growth. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 2017, 212, 24–34. [Google Scholar] [CrossRef] [PubMed]
- Hiramatsu, N.; Ichikawa, N.; Fukada, H.; Fujita, T.; Sullivan, C.V.; Hara, A. Identification and characterization of proteases involved in specific proteolysis of vitellogenin and yolk proteins in salmonids. J. Exp. Zool. 2002, 292, 11–25. [Google Scholar] [CrossRef] [PubMed]
- Carnevali, O.; Centonze, F.; Brooks, S.; Marota, I.; Sumpter, J.P. Molecular cloning and expression of ovarian cathepsin D in seabream, Sparus aurata. Biol. Reprod. 1999, 61, 785–791. [Google Scholar] [CrossRef] [PubMed]
- Finn, R.N. Vertebrate Yolk Complexes and the Functional Implications of Phosvitins and Other Subdomains in Vitellogenins. Biol. Reprod. 2007, 76, 926–935. [Google Scholar] [CrossRef] [PubMed]
- Reading, B.J.; Sullivan, C.V. Vitellogenesis in fishes. In Encyclopedia of Fish Physiology; Farrell, A.P., Cech, J.J., Richard, J.G., Stevens, E.D., Eds.; Academic Press: San Diego, CA, USA, 2011. [Google Scholar]
- Matsubara, T.; Ohkubo, N.; Andoh, T.; Sullivan, C.V.; Hara, A. Two forms of vitellogenin, yielding two distinct lipovitellins, play different roles during oocyte maturation and early development of barfin flounder, Verasper moseri, a marine teleost that spawns pelagic eggs. Dev. Biol. 1999, 213, 18–32. [Google Scholar] [CrossRef] [PubMed]
- Sawaguchi, S.; Ohkubo, N.; Amano, H.; Hiramatsu, N.; Hara, A.; Sullivan, C.V.; Matsubara, T. Controlled accumulation of multiple vitellogenins into oocytes during vitellogenesis in the barfin flounder, Varasper moseri. Cybium 2008, 32, 262. [Google Scholar]
- Williams, V.N.; Reading, B.J.; Amano, H.; Hiramatsu, N.; Schilling, J.; Salger, S.A.; Williams, T.I.; Gross, K.; Sullivan, C.V. Proportional accumulation of yolk proteins derived from multiple vitellogenins is precisely regulated during vitellogenesis in striped bass (Morone saxatilis). J. Exp. Zool. Part A 2014, 321, 301–315. [Google Scholar] [CrossRef] [PubMed]
- Schilling, J.; Loziuk, P.L.; Muddiman, D.C.; Daniels, H.V.; Reading, B.J. Mechanisms of Egg Yolk Formation and Implications on Early Life History of White Perch (Morone americana). PLoS ONE 2015, 10, e0143225. [Google Scholar] [CrossRef] [PubMed]
- Sawaguchi, S.; Koya, Y.; Yoshizaki, N.; Ohkubo, N.; Andoh, T.; Hiramatsu, N.; Sullivan, C.V.; Hara, A.; Matsubara, T. Multiple vitellogenins (Vgs) in mosquitofish (Gambusia affinis): Identification and characterization of three functional Vg genes and their circulating and yolk protein products. Biol. Reprod. 2005, 72, 1045–1060. [Google Scholar] [CrossRef] [PubMed]
- Sawaguchi, S.; Kagawa, H.; Ohkubo, N.; Hiramatsu, N.; Sullivan, C.V.; Matsubara, T. Molecular characterization of three forms of vitellogenin and their yolk protein products during oocyte growth and maturation in red seabream (Pagrus major), a marine teleost spawning pelagic eggs. Mol. Reprod. Dev. 2006, 73, 719–736. [Google Scholar] [CrossRef] [PubMed]
- Reith, M.; Munholland, J.; Kelly, J.; Finn, R.N.; Fyhn, H.J. Lipovitellins derived from two forms of vitellogenin are differentially processed during oocyte maturation in haddock (Melanogrammus aeglefinus). J. Exp. Zool. 2001, 291, 58–67. [Google Scholar] [CrossRef] [PubMed]
- Finn, R.N. The Maturational Disassembly and Differential Proteolysis of Paralogous Vitellogenins in a Marine Pelagophil Teleost: A Conserved Mechanism of Oocyte Hydration. Biol. Reprod. 2007, 76, 936–948. [Google Scholar] [CrossRef] [PubMed]
- Yilmaz, O.; Patinote, A.; Nguyen, T.; Bobe, J. Multiple vitellogenins in zebrafish (Danio rerio): Quantitative inventory of genes, transcripts and proteins, and relation to egg quality. Fish Physiol. Biochem. 2018, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Matsubara, T.; Koya, Y. Course of proteolytic cleavage in three classes of yolk proteins during oocyte maturation in barfin flounder Verasper moseri, a marine teleost spawning pelagic eggs. J. Exp. Zool. 1997, 278, 189–200. [Google Scholar] [CrossRef]
- Carnevali, O.; Cionna, C.; Tosti, L.; Cerdà, J.; Gioacchini, G. Changes in cathepsin gene expression and relative enzymatic activity during gilthead sea bream oogenesis. Mol. Reprod. Dev. 2008, 75, 97–104. [Google Scholar] [CrossRef] [PubMed]
- Carnevali, O.; Carletta, R.; Cambi, A.; Vita, A.; Bromage, N. Yolk formation and degradation during oocyte maturation in seabream Sparus aurata: Involvement of two lysosomal proteinases. Biol. Reprod. 1999, 60, 140–146. [Google Scholar] [CrossRef] [PubMed]
- Carnevali, O.; Mosconi, G.; Cardinali, M.; Meiri, I.; Polzonetti-Magni, A. Molecular components related to egg viability in the gilthead sea bream, Sparus aurata. Mol. Reprod. Dev. Incorp. Gamete Res. 2001, 58, 330–335. [Google Scholar] [CrossRef]
- Gwon, S.H.; Kim, H.K.; Baek, H.J.; Lee, Y.D.; Kwon, J.Y. Cathepsin B and D and the Survival of Early Embryos in Red Spotted Grouper, Ephinephelus akaara. Dev. Reprod. 2017, 21, 457–466. [Google Scholar] [CrossRef] [PubMed]
- Cerdà, J. Molecular pathways during marine fish egg hydration: The role of aquaporins. J. Fish Biol. 2009, 75, 2175–2196. [Google Scholar] [CrossRef] [PubMed]
- Chauvigné, C.; Zapater, C.; Cerdà, J. Role of aquaporins during teleost gametogenesis and early embryogenesis. Front. Physiol. 2011, 2, 66. [Google Scholar] [CrossRef] [PubMed]
- Żarski, D.; Nguyen, T.; Le Cam, A.; Montfort, J.; Dutto, G.; Odile Vidal, M.; Fauvel, C.; Bobe, J. Transcriptomic profiling of egg quality in sea bass (Dicentrarchus labrax) sheds light on genes involved in ubiquitination and translation. Mar. Biotechnol. 2017, 19, 102–115. [Google Scholar] [CrossRef] [PubMed]
- Aegerter, S.; Jalabert, B.; Bobe, J. Messenger RNA stockpile of cyclin B, insulinlike growth factor I, insulin-like growth factor II, insulin-like growth factor receptor Ib, and p53 in the rainbow trout oocyte in relation with developmental competence. Mol. Reprod. Dev. 2004, 67, 127–135. [Google Scholar] [CrossRef] [PubMed]
- Mathavan, S.; Serene, G.P.L.; Alicia, M.; Lance, D.M.; Karuturi, R.K.M.; Kunde, R.; Govindarajan, Y.T.; Wu, Y.L.; Lam, S.H.; Yang, H.; et al. Transcriptome analysis of zebrafish embryogenesis using microarrays. PLoS Genet. 2005, 1, e29. [Google Scholar] [CrossRef] [PubMed]
- Fernández, C.G.; Roufidou, C.; Antonopoulou, E.; Sarropoulou, E. Expression of developmental-stage-specific genes in the gilthead sea bream Sparus aurata L. Mar. Biotechnol. 2013, 15, 313–320. [Google Scholar] [CrossRef] [PubMed]
- Rise, M.L.; Nash, G.W.; Hall, J.R.; Booman, M.; Hori, T.S.; Trippel, E.A.; Gamperl, A.K. Variation in embryonic mortality and maternal transcript expression among Atlantic cod (Gadus morhua) broodstock: A functional genomics study. Mar. Genom. 2014, 18, 3–20. [Google Scholar] [CrossRef] [PubMed]
- Bonnet, E.; Fostier, A.; Bobe, J. Microarray-based analysis of fish egg quality after natural or controlled ovulation. BMC Genom. 2007, 8, 55. [Google Scholar] [CrossRef] [PubMed]
- Aegerter, S.; Jalabert, B.; Bobe, J. Large scale real-time PCR analysis of mRNA abundance in rainbow trout eggs in relationship with egg quality and post-ovulatory ageing. Mol. Reprod. Dev. Incorp. Gamete Res. 2005, 72, 377–385. [Google Scholar] [CrossRef] [PubMed]
- Mommens, M.; Fernandes, J.M.O.; Tollefsen, K.E.; Johnston, I.A.; Babiak, I. Profiling of the embryonic Atlantic halibut (Hippoglossus hippoglossus L.) transcriptome reveals maternal transcripts as potential markers of embryo quality. BMC Genom. 2014, 15, 829. [Google Scholar] [CrossRef] [PubMed]
- Lanes, C.F.C.; Fernandes, J.M.O.; Babiak, K.I. Profiling of key apoptotic, stress, and immune-related transcripts during embryonic and postembryonic development of Atlantic cod (Gadus morhua L.). Theriogenology 2012, 78, 1583–1596. [Google Scholar] [CrossRef] [PubMed]
- Mommens, M.; Fernandes, J.M.O.; Bizuayehu, T.T.; Bolla, S.L.; Johnston, I.A.; Babiak, I. Maternal gene expression in Atlantic halibut (Hippoglossus hippoglossus L.) and its relation to egg quality. BMC Res. Notes 2010, 3, 138. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, C.V.; Hiramatsu, N.; Kennedy, A.M.; Clark, R.W.; Weber, G.M.; Matsubara, T.; Hara, A. Induced maturation and spawning: Opportunities and applications for research on oogenesis. Fish Physiol. Biochem. 2003, 28, 481–486. [Google Scholar] [CrossRef]
- Patiño, R.; Sullivan, C.V. Ovarian follicle growth, maturation, and ovulation in teleost fish. Fish Physiol. Biochem. 2002, 26, 57–70. [Google Scholar] [CrossRef]
- Reading, B.J.; Hiramatsu, N.; Sawaguchi, S.; Matsubara, T.; Hara, A.; Lively, M.O.; Sullivan, C.V. Conserved and variant molecular and functional features of multiple egg yolk precursor proteins (vitellogenins) in white perch (Morone americana) and other teleosts. Mar. Biotechnol 2009, 11, 169–187. [Google Scholar] [CrossRef] [PubMed]
- Amano, H.; Fujita, T.; Hiramatsu, N.; Shimizu, M.; Sawaguchi, S.; Matsubara, T.; Kagawa, H.; Nagae, M.; Sullivan, C.V.; Hara, A. Egg yolk proteins in grey mullet (Mugil cephalus): Purification and classification of multiple lipovitellins and other vitellogenin-derived yolk proteins and molecular cloning of the parent vitellogenin genes. J. Exp. Zool. Part A 2007, 307, 324–341. [Google Scholar] [CrossRef] [PubMed]
- Patiño, R.; Thomas, P. Induction of maturation of Atlantic croaker oocytes by 17α,20β,21-trihydroxy-4-pregnen-3-one in vitro: Consideration of some biological and experimental variables. J. Exp. Zool. 1990, 255, 97–109. [Google Scholar] [CrossRef] [PubMed]
- Matsubara, T.; Sawano, K. Proteolytic cleavage of vitellogenin and yolk proteins during vitellogenin uptake and oocyte maturation in barfin flounder (Verasper moseri). J. Exp. Zool. 1995, 272, 34–45. [Google Scholar] [CrossRef]
- Zohar, Y.; Mylonas, C.C. Endocrine manipulations of spawning in cultured fish: From hormones to genes. Reprod. Biotechnol. Finfish Aquac. 2001, 197, 99–136. [Google Scholar]
- Tort, L. Impact of stress in health and reproduction. In Encyclopedia of Fish Physiology; Farrell, A.P., Cech, J.J., Richard, J.G., Stevens, E.D., Eds.; Academic Press: San Diego, CA, USA, 2011. [Google Scholar]
- Milla, S.; Wang, N.; Mandiki, S.N.M.; Kestemont, P. Corticosteroids: Friends or foes of teleost fish reproduction? Comp. Biochem. Physiol. A 2009, 153, 242–251. [Google Scholar] [CrossRef] [PubMed]
- Wendelaar Bonga, S.E. Hormonal responses to stress: Hormone response to stress. In Encyclopedia of Fish Physiology; Academic Press: San Diego, CA, USA, 2011; pp. 1515–1523. [Google Scholar]
- Gornati, R.; Papis, E.; Rimoldi, S.; Terova, G.; Saroglia, M.; Bernardini, G. Rearing density influences the expression of stress-related genes in sea bass (Dicentrarchus labrax, L.). Gene 2004, 341, 111–118. [Google Scholar] [CrossRef] [PubMed]
- Pankhurst, N.W.; Van Der Kraak, G. Evidence that acute stress inhibits ovarian steroidogenesis in rainbow trout in vivo, through the action of cortisol. Gen. Comp. Endocrinol. 2000, 117, 225–237. [Google Scholar] [CrossRef] [PubMed]
- Campbell, P.M.; Pottinger, T.G.; Sumpter, J.P. Stress reduces the quality of gametes produced by rainbow trout. Biol. Reprod. 1992, 47, 1140–1150. [Google Scholar] [CrossRef] [PubMed]
- Schreck, C.B.; Contreras-Sanchez, W.; Fitzpatrick, M.S. Effects of stress on fish reproduction, gamete quality, and progeny. Reprod. Biotechnol. Finfish Aquac. 2001, 3–24. [Google Scholar] [CrossRef]
- Schreck, C.B. Stress and fish reproduction: The roles of allostasis and hormesis. Gen. Comp. Endocrinol. 2010, 165, 549–556. [Google Scholar] [CrossRef] [PubMed]
- Baltzegar, D.A.; Reading, B.J.; Douros, J.D.; Borski, R.J. Role for leptin in promoting glucose mobilization during acute hyperosmotic stress in teleost fishes. J. Endocrinol. 2013, 220, 61–72. [Google Scholar] [CrossRef] [PubMed]
- Le Luyer, J.; Laporte, M.; Beacham, T.D.; Kaukinen, K.H.; Withler, R.E.; Leong, J.S.; Rondeau, E.B.; Koop, B.F.; Bernatchez, L. Parallel epigenetic modifications induced by hatchery rearing in a Pacific salmon. Proc. Natl. Acad. Sci. USA 2017, 114, 12964–12969. [Google Scholar] [CrossRef] [PubMed]
- Meiri, I.; Gothilf, Y.; Zohar, Y.; Elizur, A. Physiological changes in the spawning gilthead seabream, Sparus aurata, succeeding the removal of males. J. Exp. Zool. 2002, 292, 555–564. [Google Scholar] [CrossRef] [PubMed]
- Donato, D.M.; Hiramatsu, N.; Arey, K.M.; Hiramatsu, K.; Kennedy, A.M.; Morton, C.L.; Hara, A.; Sullivan, C.V. Atresia in temperate basses: Cloning of hatching enzyme (choriolysin) homologues from atretic ovaries. Fish Physiol. Biochem. 2003, 28, 329–330. [Google Scholar] [CrossRef]
- Ma, H.; Hostuttler, M.; Wei, H.; Rexroad, C.E., III; Yao, J. Characterization of the rainbow trout egg microRNA transcriptome. PLoS ONE 2012, 7, e39649. [Google Scholar] [CrossRef] [PubMed]
- Ma, H.; Weber, G.M.; Hostuttler, M.A.; Wei, H.; Wang, L.; Yao, J. MicroRNA expression profiles from eggs of different qualities associated with post-ovulatory ageing in rainbow trout (Oncorhyncchus mykiss). BMC Genom. 2015, 16, 201. [Google Scholar] [CrossRef] [PubMed]
- Craik, J.C.A.; Harvey, S.M. Egg quality in rainbow trout: The relation between egg viability, selected aspects of egg composition, and time of stripping. Aquaculture 1984, 40, 115–134. [Google Scholar] [CrossRef]
- Springate, J.R.C.; Bromage, N.R.; Elliot, J.A.K.; Hudson, D.L. The timing of ovulation and stripping and their effects on the rate of fertilization and survival to eyeing, hatch, and swim-up in the rainbow-trout (Salmo-Gairdneri R.). Aquaculture 1984, 43, 313–322. [Google Scholar] [CrossRef]
- Lahnsteiner, F. Morphological, physiological and biochemical parameters characterizing the over-ripening of rainbow trout eggs. Fish Physiol. Biochem. 2000, 23, 107–118. [Google Scholar] [CrossRef]
- Ma, H.; Weber, G.M.; Wei, H.; Yao, J. Identification of mitochondrial genome-encoded small RNAs related to egg deterioration caused by post ovulatory aging in rainbow trout. Mar. Biotechnol. 2016, 18, 584–597. [Google Scholar] [CrossRef] [PubMed]
- Mommens, M.; Storset, A.; Bibiak, I. Some quantitative indicators of postovulatory aging and its effect on larval and juvenile development of Atlantic salmon (Salmo salar). Theriogenology 2015, 84, 170–176. [Google Scholar] [CrossRef] [PubMed]
- Aegerter, S.; Jalabert, B. Effects of post-ovulatory oocyte ageing and temperature on egg quality and on the occurrence of triploid fry in rainbow trout, Oncorhynchus mykiss. Aquaculture 2004, 231, 59–71. [Google Scholar] [CrossRef]
- Rime, H.; Guitton, N.; Pineau, C.; Bonnet, E.; Bobe, J.; Jalabert, B. Post-ovulatory ageing and egg quality: A proteomic analysis of rainbow trout coelomic fluid. Reprod. Biol. Endocrinol. 2004, 2, 26–36. [Google Scholar] [CrossRef] [PubMed]
- Hajirezaee, S.; Khara, H.; Abed-Elmdoust, A.; Rafieepour, A.; Rahimi, R. Effects of egg aging on the metabolites of ovarian fluid in rainbow trout, Oncorhynchus mykiss. Aquac. Res. 2017, 49, 104–110. [Google Scholar] [CrossRef]
- Burns, K.H.; Viveiros, M.M.; Ren, Y.; Wang, P.; DeMayo, F.J.; Frail, D.E.; Eppig, J.J.; Matzuk, M.M. Roles of NPM2 in chromatin and nucleolar organization in oocytes and embryos. Science 2003, 300, 633–636. [Google Scholar] [CrossRef] [PubMed]
- Brązert, M.; Pawelczyk, L.A. Insulin-like growth factor-1 isoforms in human ovary. Preliminary report on the expression of the IGF-1 gene in PCOS patients and healthy controls. Ginekol. Pol. 2015, 86, 890–895. [Google Scholar] [CrossRef] [PubMed]
- Suen, D.F.; Norris, K.L.; Youle, R.J. Mitochondrial dynamics and apoptosis. Genes Dev. 2008, 22, 1577–1590. [Google Scholar] [CrossRef] [PubMed]
- Chinnery, P.F.; Elliott, H.R.; Hudson, G.; Samuels, D.C.; Relton, C.L. Epigenetics, epidemiology and mitochondrial DNA diseases. Int. J. Epidemiol. 2012, 41, 177–187. [Google Scholar] [CrossRef] [PubMed]
- Lord, T.; Aitken, R.J. Oxidative stress and ageing of the postovulatory oocyte. Reproduction 2013, 146, 217–227. [Google Scholar] [CrossRef] [PubMed]
- Yilmaz, O.; Patinote, A.; Nguyen, T.V.; Com, E.; Lavigne, R.; Pineau, C.; Sullivan, C.V.; Bobe, J. Scrambled eggs: Proteomic portraits and novel biomarkers of egg quality in zebrafish (Danio rerio). PLoS ONE 2017, 12, e0188084. [Google Scholar] [CrossRef] [PubMed]
- Mylonas, C.C.; Zohar, Y. Use of GnRHa-delivery systems for the control of reproduction in fish. Rev. Fish Biol. Fish. 2000, 10, 463–491. [Google Scholar] [CrossRef]
- Hodson, R.G.; Sullivan, C.V. Induced maturation and spawning of domestic and wild striped bass, Morone saxatilis (Walbaum), broodstock with implanted GnRH analogue and injected hCG. Aquac. Res. 1993, 24, 389–398. [Google Scholar] [CrossRef]
- Weber, G.M.; Sullivan, C.V. Effects of Insulin-Like Growth Factor-I on In Vitro Final Oocyte Maturation and Ovarian Steroidogenesis in Striped Bass, Morone saxatilis. Biol. Reprod. 2000, 63, 1049–1057. [Google Scholar] [CrossRef] [PubMed]
- Weber, G.M.; Sullivan, C.V. Insulin-Like Growth Factor-I Induces Oocyte Maturational Competence but Not Meiotic Resumption in White Bass (Morone chrysops) Follicles In Vitro: Evidence for Rapid Evolution of Insulin-Like Growth Factor Action. Biol. Reprod. 2005, 72, 1177–1186. [Google Scholar] [CrossRef] [PubMed]
- Schilling, J.; Nepomuceno, A.I.; Planchart, A.; Yoder, J.A.; Kelly, R.M.; Muddiman, D.C.; Daniels, H.; Hiramatsu, N.; Reading, B.J. Machine learning reveals sex-specific 17β-estradiol-responsive expression patterns in white perch (Morone americana) plasma proteins. Proteomics 2015, 15, 2678–2690. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, K.; Yamauchi, K. Sexual maturation of Japanese eel and production of eel larvae in the aquarium. Nature 1974, 251, 220–222. [Google Scholar] [CrossRef] [PubMed]
- Ganias, K. Thirty years of using the postovulatory follicles method: Overview, problems, and alternatives. Fish. Res. 2012, 117–118, 63–74. [Google Scholar] [CrossRef]
- Wallace, R.A.; Selman, K. Cellular and dynamic aspects of oocyte growth in teleosts. Am. Zool. 1981, 21, 325–343. [Google Scholar] [CrossRef]
- Bayless, J.D. Artificial Propagation and Hybridization of Striped Bass, Morone saxatilis (Walbaum); South Carolina Wildlife and Marine Resources Department: Columbia, SC, USA, 1972.
- Moser, H.G. Seasonal histological changes in the gonads of Sebastodes paucispinus, an ovoviviparious teleost (Family Scorpaenidae). J. Morphol. 1967, 123, 329–353. [Google Scholar] [CrossRef] [PubMed]
- Üçüncü, S.I.; Çakici, Ö. Atresia and apoptosis in preovulatory follicles in the ovary of Danio rerio (Zebrafish). Turk. J. Fish. Aquat. Sci. 2009, 9, 215–221. [Google Scholar] [CrossRef]
- Mylonas, C.C.; Woods, L.C., III; Zohar, Y. Cyto-histological examination of post-vitellogenesis and final oocyte maturation in captive-reared striped bass. J. Fish Biol. 1997, 50, 44–49. [Google Scholar] [CrossRef]
- Harrell, R.M.; Kerby, J.H.; Minton, R.V. (Eds.) Culture and Propagation of Striped Bass and Its Hybrids; American Fisheries Society: Bethesda, MD, USA, 1990. [Google Scholar]
- Specker, J.L.; Sullivan, C.V. Vitellogenesis in fishes: Status and perspectives. In Perspectives in Comparative Endocrinology; Davey, K.G., Peter, R.E., Tobe, S.S., Eds.; National Research Council of Canada: Ottawa, ON, Canada, 1994; pp. 304–315. [Google Scholar]
- Brooks, S.; Tyler, C.R.; Sumpter, J.P. Egg quality in fish: What makes a good egg? Rev. Fish Biol. Fish. 1997, 7, 287–416. [Google Scholar] [CrossRef]
- Al-Hafdeh, Y.S. Effects of dietary protein on growth and body composition of Nile tilapia, Oreochromis niloticus L. Aquac. Res. 1999, 30, 385–393. [Google Scholar] [CrossRef]
- Watanabe, T.; Takeuchi, T.; Saito, M.; Nishimura, K. Effect of low protein-high calorie or essential fatty acid deficiency diet on reproduction of rainbow trout. Nippon Suisan Gakkaishi 1984, 50, 1207–1215. [Google Scholar] [CrossRef]
- Trushenski, J.T. Saturated lipid sources in feeds for sunshine bass: Alterations in production performance and tissue fatty acid composition. N. Am. J. Aquac. 2009, 71, 363–373. [Google Scholar] [CrossRef]
- Fahy, E.; Subramaniam, S.; Brown, H.A.; Glass, C.K.; Merrill, A.H.; Murphy, R.C.; Raetz, C.R.H.; Russell, D.W.; Seyama, Y.; Shaw, W.; et al. A comprehensive classification system for lipids. J. Lipid Res. 2005, 46, 839–861. [Google Scholar] [CrossRef] [PubMed]
- Leray, C.; Nonnote, G.; Roubaud, D.; Leger, C. Incidence of (n-3) essential fatty acid deficiency on trout reproductive processes. Reprod. Nutr. Dev. 1985, 25, 567–581. [Google Scholar] [CrossRef] [PubMed]
- Izquierdo, M.S.; Fernández-Palacios, H.; Tacon, A.G.J. Effect of broodstock nutrition on reproductive performance of fish. Aquaculture 2001, 197, 25–42. [Google Scholar] [CrossRef]
- Tuncer, H.; Harrell, R.M. Essential fatty acid nutrition of larval striped bass (Morone saxatilis) and palmetto bass (M. saxatilis x M. chrysops). Aquaculture 1992, 101, 105–121. [Google Scholar] [CrossRef]
- Harrell, R.M.; Woods, L.C., III. Comparative fatty acid composition of eggs from domesticated and wild striped bass (Morone saxatilis). Aquaculture 1995, 133, 225–233. [Google Scholar] [CrossRef]
- Takeuchi, T.; Watanabe, T. Effects of various polyunsaturated fatty acids on growth and fatty acid compositions of rainbow trout Salmo gairdneri, coho salmon Onchorhynchus kisutch, and chum salmon Onchorhynchus keta. Bull. Jpn. Soc. Sci. Fish 1982, 48, 1745–1752. [Google Scholar] [CrossRef]
- Watanabe, T. Lipid nutrition in fish. Comp. Biochem. Physiol. B 1982, 73, 3–15. [Google Scholar] [CrossRef]
- Carrillo, M.; Zanuy, S. Manipulación de la reproducción de los teleósteos y calidad de las puestas. In Proceedings of the Actas del V Congreso Nacional de Acuicultura, Sant Carles de la Rápita, Spain, 10–13 May 1995; pp. 1–9. [Google Scholar]
- Valdebenito, I.I.; Gallegos, P.C.; Effer, B.R. Gamete quality in fish: Evaluation parameters and determining factors. Zygote 2015, 23, 177–197. [Google Scholar] [CrossRef] [PubMed]
- Fuller, A.S.; Rawles, S.D.; McEntire, M.E.; Bader, T.J.; Riche, M.; Beck, B.H.; Webster, C.D. White bass (Morone chrysops) preferentially retain n-3 PUFA in ova when fed prepared diets with varying FA content. Lipids 2017, 52, 823–836. [Google Scholar] [CrossRef] [PubMed]
- Nevenzel, J.C. Occurrence, function and biosynthesis of wax esters in marine organisms. Lipids 1970, 5, 308–319. [Google Scholar] [CrossRef] [PubMed]
- Eldridge, M.B.; Joseph, J.D.; Taberski, K.M.; Seaborn, G.T. Lipid and fatty acid composition of the endogenous energy sources of striped bass (Morone saxatilis) eggs. Lipids 1983, 18, 510–513. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, T.; Kiron, V. Broodstock management and nutritional approaches for quality offsprings in the Red Sea Bream. In Broodstock Management and Egg Larval Quality; Bromage, N.R., Roberts, R.J., Eds.; Blackwell Science: Oxford, UK, 1995. [Google Scholar]
- Rainuzzo, J.R.; Reitan, K.I.; Olsen, Y. The significance of lipids at early stages of marine fish: A review. Aquaculture 1997, 155, 105–118. [Google Scholar] [CrossRef]
- Terner, C. Studies of metabolism in embryonic development—I. The oxidative metabolism of unfertilized and embryonated trout ova. Comp. Biochem. Physiol. 1968, 24, 941–950. [Google Scholar] [CrossRef]
- Kimata, M. Changes of chemical composition during early development in the red seabream Crysophys major (Temminck et Schelegel) egg and larva. J. Fac. Mar. Sci. Technol. Tokai Univ. 1983, 16, 213–223. [Google Scholar]
- Kimata, M. Changes of chemical composition during early development of egg and larva in the half beak, Hemiramphus sajori (Temminck et Schelegel). Bull. Jpn. Soc. Sci. Fish 1982, 48, 1663–1671. [Google Scholar] [CrossRef]
- Tocher, D.R.; Fraser, A.J.; Sargent, J.R.; Gamble, J.C. Lipid class composition during embryonic and early larval development in Atlantic herring (Clupea harengus L.). Lipids 1985, 20, 84–89. [Google Scholar] [CrossRef] [PubMed]
- Palace, V.P.; Werner, J. Vitamins A and E in the maternal diet influence egg quality and early life stage development in fish: A review. Sci. Mar. 2006, 70, 41–57. [Google Scholar] [CrossRef]
- Hemre, G.I.; Mangor-Jensen, A.; Lie, O. Broodstock nutrition in turbot (Scophthalmus maximus) effect of dietary vitamin E. FiskeriDir. Skr., Ser. Ernæring. 1994, 8, 21–29. [Google Scholar]
- Craik, J.C.A. Egg quality and egg pigment content in salmonid fishes. Aquaculture 1985, 47, 61–88. [Google Scholar] [CrossRef]
- Craik, J.C.A.; Harvey, S.M. Egg Quality in Atlantic Salmon; International Council for the Exploration of the Sea: Copenhagen, Denmark, 1986; F2. [Google Scholar]
- Harris, L.E. Effects of a broodfish diet fortified with canthaxanthin on female fecundity and egg color. Aquaculture 1984, 43, 179–183. [Google Scholar] [CrossRef]
- Torrissen, O.J. Pigmentation of salmonids—Effects of carotenoids in eggs and start feeding diet on survival and growth rate. Aquaculture 1984, 43, 185–193. [Google Scholar] [CrossRef]
- Choubert, G.; Blanc, J.M. Muscle pigmentation changes during and after spanwing in male and female rainbow trout, Oncorhynchus mykiss, fed dietary carotenoids. Aquat. Living Resour. 1993, 6, 163–168. [Google Scholar] [CrossRef]
- Torrissen, O.J.; Christiansen, R. Requirements on carotenoids in fish diets. J. Appl. Ichthyol. 1995, 11, 225–230. [Google Scholar] [CrossRef]
- Takeuchi, M.; Ishii, S.; Ogiso, T. Effect of dietary vitamin E on growth, vitamin E distribution, and mortalities of the fertilized eggs and fry in ayo, Plecoglossus altivelis. Bull. Tokai Reg. Fish. Res. Lab. 1981, 104, 111–122. [Google Scholar]
- Selman, K.; Wallace, R.A. Cellular aspects of oocyte growth in teleosts. Zool. Sci. 1989, 6, 211–231. [Google Scholar]
- Kwon, J.Y.; Prat, F.; Randall, C.; Tyler, C.R. Molecular characterization of putative yolk processing enzymes and their expression during oogenesis and embryogenesis in rainbow trout (Oncorhynchus mykiss). Biol. Reprod. 2001, 65, 1701–1709. [Google Scholar] [CrossRef] [PubMed]
- Divers, S.L.; McQuillan, H.J.; Matsubara, H.; Todo, T.; Lokman, P.M. Effects of reproductive stage and 11-ketotestosterone on LPL mRNA levels in the ovary of the shortfinned eel. J. Lipid Res. 2010, 51, 3250–3258. [Google Scholar] [CrossRef] [PubMed]
- Selman, K.; Wallace, R.A.; Sarka, A.; Qi, X. Stages of oocyte development in the zebrafish, Brachydanio rerio. J. Morphol. 1993, 218, 203–224. [Google Scholar] [CrossRef] [PubMed]
- Levi, L.; Ziv, T.; Admon, A.; Levai-Sivan, B.; Lubzens, E. Insight into molecular pathways of retinal metabolism, associated with vitellogenesis in zebrafish. Am. J. Physiol. Endocrinol. Metab. 2012, 302, E626–E646. [Google Scholar] [CrossRef] [PubMed]
- Clark, R.W.; Henderson-Arzapalo, A.; Sullivan, C.V. Disparate effects of constant and annually-cycling daylength and water temperature on reproductive maturation of striped bass (Morone saxatilis). Aquaculture 2005, 249, 497–513. [Google Scholar] [CrossRef]
- Nelson, E.R.; Habibi, H.R. Estrogen receptor function and regulation in fish and other vertebrates. Gen. Comp. Endocrinol. 2013, 192, 15–24. [Google Scholar] [CrossRef] [PubMed]
- Teo, B.Y.; Tan, N.S.; Lim, E.H.; Lam, T.J.; Ding, J.L. A novel piscine vitellogenin gene: Structural and functional analyses of estrogen-inducible promoter. Mol. Cell. Endocrinol. 1998, 146, 103–120. [Google Scholar] [CrossRef]
- Teo, B.Y.; Tan, N.S.; Lam, T.J.; Ding, J.L. Synergistic effects of nuclear factors—GATA, VBP and ER in potentiating vitellogenin gene transcription. FEBS Lett. 1999, 459, 57–63. [Google Scholar] [CrossRef]
- Hawkins, M.B.; Thornton, J.W.; Crews, D.; Skipper, J.K.; Dotte, A.; Thomas, P. Identification of a third distinct estrogen receptor and reclassification of estrogen receptors in teleosts. Proc. Nat. Acad. Sci. USA 2000, 97, 10751–10756. [Google Scholar] [CrossRef] [PubMed]
- Nelson, E.R.; Habibi, H.R. Functional significance of nuclear estrogen receptor subtypes in the liver of goldfish. Endocrinology 2010, 151, 1668–1676. [Google Scholar] [CrossRef] [PubMed]
- Lu, H.; Cui, Y.; Jiang, L.; Ge, W. Functional analysis of nuclear estrogen receptors in zebrafish reproduction by genome editing approach. Endocrinology 2017, 158, 2292–2308. [Google Scholar] [CrossRef] [PubMed]
- Tohyama, S.; Ogino, Y.; Lange, A.; Myosho, T.; Kobayashi, T.; Hirano, Y.; Yamada, G.; Sato, T.; Tatarazako, N.; Tyler, C.R.; et al. Establishment of estrogen receptor 1 (ESR1)-knockout medaka: ESR1 is dispensable for sexual development and reproduction in medaka, Oryzias latipes. Dev. Growth Differ. 2017, 59, 552–561. [Google Scholar] [CrossRef] [PubMed]
- Nishimiya, O.; Katsu, Y.; Inagawa, H.; Hiramatsu, N.; Todo, T.; Hara, A. Molecular cloning and characterization of hagfish estrogen receptors. J. Steroid Biochem. Mol. Biol. 2017, 165, 190–201. [Google Scholar] [CrossRef] [PubMed]
- Nishimiya, O.; Kunihiro, Y.; Hiramatsu, N.; Inagawa, H.; Todo, T.; Matsubara, T.; Reading, B.J.; Sullivan, C.V.; Hara, A. Molecular characterization and expression analysis of estrogen receptor and vitellogenins in inshore hagfish (Eptatretus burgeri). Indian J. Sci. Technol. 2011, 4, 194–195. [Google Scholar]
- Schonbaum, C.P.; Lee, S.; Mahowald, A.P. The Drosophila yolkless gene encodes a vitellogenin receptor belonging to the low density lipoprotein receptor superfamily. Proc. Nat. Acad. Sci. USA 1995, 92, 1485–1489. [Google Scholar] [CrossRef] [PubMed]
- Milla, S.; Mandiki, S.N.M.; Hubermont, P.; Rougeot, C.; Mélard, C.; Kestemont, P. Ovarian steroidogenesis inhibition by constant photothermal conditions is caused by a lack of gonadotropin stimulation in Eurasian perch. Gen. Comp. Endocrinol. 2009, 163, 242–250. [Google Scholar] [CrossRef] [PubMed]
- Matsubara, T.; Nagae, M.; Ohkubo, N.; Andoh, T.; Sawaguchi, S.; Hiramatsu, N.; Sullivan, C.V.; Hara, A. Multiple vitellogenins and their unique roles in marine teleosts. Fish Physiol. Biochem. 2003, 28, 295–299. [Google Scholar] [CrossRef]
- Finn, R.N.; Kristoffersen, B.A. Vertebrate vitellogenin gene duplication in relation to the “3R hypothesis”: Correlation to the pelagic egg and the oceanic radiation of teleosts. PLoS ONE 2007, 2, e169. [Google Scholar] [CrossRef] [PubMed]
- Williams, V.N.; Reading, B.J.; Hiramatsu, N.; Amano, H.; Glassbrook, N.; Hara, A.; Sullivan, C.V. Multiple vitellogenins and product yolk proteins in striped bass, Morone saxatilis: Molecular characterization and processing during oocyte growth and maturation. Fish Physiol. Biochem. 2014, 40, 395–415. [Google Scholar] [CrossRef] [PubMed]
- Kolarevic, J.; Nerland, A.; Nilsen, F.; Finn, R.N. Goldsinny wrasse (Ctenolabrus rupestris) is an extreme vtgAa-type pelagophil teleost. Mol. Reprod. Dev. 2007, 75, 1011–1020. [Google Scholar] [CrossRef] [PubMed]
- Finn, R.N.; Østby, G.C.; Norberg, B.; Fyhn, H.J. In vivo oocyte hydration in Atlantic halibut (Hippoglossus hippoglossus), proteolytic liberation of free amino acids, and ion transport, are driving forces for osmotic water influx. J. Exp. Biol. 2002, 205, 211–224. [Google Scholar] [PubMed]
- Reading, B.J.; Williams, V.N.; Chapman, R.W.; Williams, T.I.; Sullivan, C.V. Dynamics of the striped bass (Morone saxatilis) ovary proteome reveal a complex network of the translasome. J. Proteome Res. 2013, 12, 1691–1699. [Google Scholar] [CrossRef] [PubMed]
- Fabra, M.; Cerdà, J. Ovarian cysteine proteinases in the teleost Fundulus heteroclitus: Molecular cloning and gene expression during vitellogenesis and oocyte maturation. Mol. Reprod. Dev. Incorp. Gamete Res. 2004, 67, 282–294. [Google Scholar] [CrossRef] [PubMed]
- Smolenaars, M.M.; Madsen, O.; Rodenburg, KW.; Van der Horst, D.J. Molecular diversity and evolution of the large lipid transfer protein superfamily. J. Lipid Res. 2007, 48, 489–502. [Google Scholar] [CrossRef] [PubMed]
- Anderson, A.J.; Arthington, A.H.; Anderson, S. Lipid Classes and Fatty Acid Composition of the Eggs of Some Australian Fish. Comp. Biochem. Physiol. 1990, 98, 267–270. [Google Scholar] [CrossRef]
- Watanabe, T.; Lee, M.; Mizutani, J.; Yamada, T.; Satoh, S.; Takeuchi, T.; Yoshida, N.; Kitada, T.; Arakawa, T. Effective components in cuttlefish meal and raw krill for improvement of quality of red sea bream (Pagrus major) eggs. Nippon Suisan Gakkaishi 1991, 57, 681–694. [Google Scholar] [CrossRef]
- Watanabe, T. Effect of broodstock diets on reproduction of fish. In Proceedings of the Advances in Tropical Aquaculture, Tahiti, French Polynesia, 20 February–4 March 1989. [Google Scholar]
- Eskelinen, P. Effects of different diets on egg production and egg quality of Atlantic salmon (Salmo salar L.). Aquaculture 1989, 79, 275–281. [Google Scholar] [CrossRef]
- Blom, J.H.; Dabrowski, K. Reproductive success of female rainbow trout (Oncorhynchus mykiss) in response to graded dietary ascorbyl monophosphate levels. Biol. Reprod. 1995, 52, 1073–1080. [Google Scholar] [CrossRef] [PubMed]
- Sandnes, K.; Ulgens, Y.; Braekkan, O.R.; Utne, F. The effect of ascorbic acid supplementation in broodstock feed on reproduction of rainbow trout (Salmo gairdneri). Aquaculture 1984, 43, 167–177. [Google Scholar] [CrossRef]
- Will, C.L.; Lührmann, R. Spliceosome structure and function. Cold Spring Harb. Perspect. Biol. 2010, 3, a003707. [Google Scholar] [CrossRef] [PubMed]
- Ramachandra, R.K.; Salem, M.; Gahr, S.; Rexroad, C.E., III; Yao, J. Cloning and characterization of microRNAs from rainbow trout (Oncorhynchus mykiss): Their expression during early embryonic development. BMC Dev. Biol. 2008, 8, 41. [Google Scholar] [CrossRef] [PubMed]
- Juanchich, A.; Le Cam, A.; Montfort, J.; Guiguen, Y.; Bobe, J. Identification of differentially expressed miRNAs and their potential targets during fish ovarian development. Biol. Reprod. 2013, 88, 128. [Google Scholar] [CrossRef] [PubMed]
- Giraldez, A.J.; Cinalli, R.M.; Glasner, M.E.; Enright, A.J.; Thomson, J.M.; Baskerville, S.; Hammond, S.M.; Bartel, D.P.; Schier, A.F. MicroRNAs regulate brain morphogenesis in zebrafish. Science 2005, 308, 833–838. [Google Scholar] [CrossRef] [PubMed]
- Jamnongjit, M.; Hammes, S.R. Ovarian steroids: The good, the bad, and the signals that raise them. Cell Cycle 2006, 5, 1178–1183. [Google Scholar] [CrossRef] [PubMed]
- Bobe, J.; Nguyen, T.; Jalabert, B. Targeted gene expression profiling in the rainbow trout (Oncorhynchus mykiss) ovary during maturational competence acquisition and oocyte maturation. Biol. Reprod. 2004, 71, 73–82. [Google Scholar] [CrossRef] [PubMed]
- Sriraman, V.; Eichenlaub-Ritter, U.; Bartsch, J.W.; Rittger, A.; Mulders, S.M.; Richards, J.S. Regulated expression of ADAM8 (a disintegrin and metalloprotease domain 8) in the mouse ovary: Evidence for a regulatory role of luteinizing hormone, progesterone receptor, and epidermal growth factor-like growth factors. Biol. Reprod. 2008, 78, 1038–1048. [Google Scholar] [CrossRef] [PubMed]
- Kanamori, A. Systematic identification of genes expressed during early oogenesis in medaka. Mol. Reprod. Dev. 2000, 55, 31–36. [Google Scholar] [CrossRef]
- Kawaguchi, M.; Yasumasu, S.; Shimizu, A.; Sano, K.; Iuchi, I.; Nishida, M. Conservation of the egg envelope digestion mechanism of hatching enzyme in euteleostean fishes. FEBS J. 2010, 277, 4973–4987. [Google Scholar] [CrossRef] [PubMed]
- Lizé, M.; Klimke, A.; Dobbelstein, M. MicroRNA-449 in cell fate determination. Cell Cycle 2011, 10, 2874–2882. [Google Scholar] [CrossRef] [PubMed]
- Sonkoly, E.; Lovén, J.; Xu, N.; Meisgen, F.; Wei, T.; Brodin, P.; Jaks, V. MicroRNA-203 functions as a tumor suppressor in basal cell carcinoma. Oncogenesis 2012, 1, e3. [Google Scholar] [CrossRef] [PubMed]
- Castets, M.D.; Schaerlinger, B.; Silvestre, F.; Gardeur, J.N.; Dieu, M.; Corbier, C.; Kestemont, P.; Fontaine, P. Combined analysis of Perca fluviatilis reproductive performance and oocyte proteomic profile. Theriogenology 2012, 78, 432–442. [Google Scholar] [CrossRef] [PubMed]
- Schilling, J.; Nepomuceno, A.; Schaff, J.E.; Muddiman, D.C.; Daniels, H.V.; Reading, B.J. Compartment proteomics analysis of white perch (Morone americana) ovary using support vector machines. J. Proteome Res. 2014, 13, 1515–1526. [Google Scholar] [CrossRef] [PubMed]
- Reading, B.J.; Chapman, R.W.; Schaff, J.E.; Scholl, E.H.; Opperman, C.H.; Sullivan, C.V. An ovary transcriptome for all maturational stages of the striped bass (Morone saxatilis), a highly advanced perciform fish. BMC Res. Notes 2012, 5, 111. [Google Scholar] [CrossRef] [PubMed]
| Species | Gene | Function | Citations |
|---|---|---|---|
| Cod | acy3 | expression in kidneys and liver | Rise et al.* [46] |
| Sea bass | alg5 | transferase activity, egg quality | Żarski et al. [42] |
| Striped bass | anapc7 | cell cycle, ubiquitin ligase | Chapman et al. [2] |
| Striped bass | anln | cell division | Chapman et al. [2] |
| Trout | apoc1 | lipoprotein metabolism | Bonnet et al.* [47] |
| Striped bass | apex1 | DNA repair | Chapman et al. [2] |
| Striped bass | arfgap2 | growth factor, signal transduction | Chapman et al. [2] |
| Striped bass | arpc5 | cell division | Chapman et al. [2] |
| Striped bass | btg1 | cell cycle | Chapman et al. [2] |
| Sea bream | Cathepsin_S | papain lysosomal endopeptidase | Fernández et al. [106] |
| Several | Cathepsin_Z | low quality eggs, cancer, tumorigenesis | Fernández et al. [106]; Aegerter et al. [122] |
| Striped bass | ccnb3 | cell cycle, reproduction | Chapman et al. [2] |
| Striped bass | ccne2 | cell cycle, reproduction | Chapman et al. [2] |
| Striped bass | cdc26 | cell cycle regulation | Chapman et al. [2] |
| Striped bass | cdc37 | cell cycle regulation, growth factor | Chapman et al. [2] |
| Striped bass | cdc42bpb | cell division | Chapman et al. [2] |
| Striped bass | cdt1 | cell division, malignant tumors | Chapman et al. [2] |
| Striped bass | cebpd | spliceosome | Chapman et al. [2] |
| Several | cenpf | centromere, cell division | Chapman et al. [2];Żarski et al. [103] |
| Striped bass | cenpk | cell division | Chapman et al. [2] |
| Striped bass | cir1 | spliceosome | Chapman et al. [2] |
| Striped bass | cnpy2 | cell cycle, ubiquitin ligase | Chapman et al. [2] |
| Striped bass | cops4 | COP9 signalosome, cell cycle | Chapman et al. [2] |
| Striped bass | cops5 | COP9 signalosome, cell cycle | Chapman et al. [2] |
| Striped bass | cops6 | COP9 signalosome, cell cycle | Chapman et al. [2] |
| Striped bass | cops8 | COP9 signalosome, cell cycle | Chapman et al. [2] |
| Striped bass | ctnnb1 | cell division, growth factor | Chapman et al. [2] |
| 44Striped bass | cul3 | cell cycle, ubiquitin ligase | Chapman et al. [2] |
| Several | cyclin-A2 | cell regulation, translation | Fernández et al. [45]; Mathavan et al. [44] |
| Trout | cycB | cell cycle, reproduction | Aegerter et al.* [43] |
| Striped bass | cyld | cell cycle regulation, apoptotic factors | Chapman et al. [2] |
| Halibut | cyp2n | cytochrome p450 | Mommens et al.* [49] |
| Cod | dcbld1 | discoidin | Rise et al.* [46] |
| Striped bass | ddb2 | cell cycle, ubiquitin ligase, DNA repair | Chapman et al. [2] |
| Cod | ddc | neurotransmitter metabolism | Rise et al.* [46] |
| Striped bass | eif1ad | cell cycle, spliceosome | Chapman et al. [2] |
| Striped bass | eif3e | cell cycle, ubiquitin ligase, spliceosome | Chapman et al. [2] |
| Striped bass | eif3k | spliceosome | Chapman et al. [2] |
| Striped bass | eif4e2 | spliceosome | Chapman et al. [2] |
| Halibut | eef1a2 bp | translation, protein synthesis | Mommens et al.* [49] |
| Striped bass | ergic2 | malignant tumors | Chapman et al. [2] |
| Sea bream | ERK-1 | mitogen activated protein kinase | Fernández et al. [45] |
| Striped bass | esr2 | cell signaling, estrogen receptor | Chapman et al. [2] |
| Striped bass | fbxo9 | cell cycle, ubiquitin ligase | Chapman et al. [2] |
| Striped bass | fibp | growth factor, cell signaling | Chapman et al. [2] |
| Sea bream | ftmt | iron storage, antimicrobial activity | Fernández et al. [45] |
| Sea bass | fucolectin 4 | immune response | Żarski et al. [42] |
| Striped bass | g2e3 | cell cycle, ubiquitin ligase, apoptosis | Chapman et al. [2] |
| Sea bass | gba | glucosylceramidase | Żarski et al. [42] |
| Sea bass | gcnl1l | glucosylceramidase, egg quality | Żarski et al. [42] |
| Striped bass | gdf9 | growth factors, cell signaling | Chapman et al. [2] |
| Striped bass | gna13 | growth factors, cell signaling | Chapman et al. [2] |
| Striped bass | gnpda1 | growth factor, cell signaling | Chapman et al. [2] |
| Cod | gsh-px | glutathione peroxidase | Lanes et al.* [50] |
| Striped bass | gtf2f1 | spliceosome | Chapman et al. [2] |
| Cod | hacd1 | cardiac development | Rise et al. * [46] |
| Trout | hck | blood formation, immunse response | Bonnet et al.* [47] |
| Striped bass | hdac1 | cell cycle, cell division | Chapman et al. [2] |
| Cod | hsp70 | stress response | Lanes et al.* [50] |
| Striped bass | hsp90ab1 | stress, spliceosome, cell signaling | Chapman et al. [2] |
| Trout | igf-1 | somatic growth, cell signaling | Aegerter et al. [43];Aegerter et al.* [48] |
| Trout | igf-2 | somatic growth, cell signaling | Aegerter et al.* [43] |
| Trout | igrf 1b | somatic growth, cell signaling | Aegerter et al.* [43] |
| Several | irf7 | immune response | Mommens et al.* [49];Żarski et al.* [42] |
| Sea bream | jnk1 | mitogen activated protein kinase | Fernández et al. [45] |
| Striped bass | jup | cell division | Chapman et al. [2] |
| Sea bass | kctd12 | G-protein receptor, cell signaling | Żarski et al. [45] |
| Striped bass | kifc1 | cell division | Chapman et al. [2] |
| Striped bass | kmt5a (setd8) | cell division | Chapman et al. [2] |
| Cod | kpna7 | nuclear protein import | Rise et al.* [46] |
| Trout | krt8 | cytoskeleton | Aegerter et al.* [48] |
| Trout | krt18 | cytoskeleton | Aegerter et al.* [48] |
| Striped bass | lmnb2 | cell division | Chapman et al. [2] |
| Striped bass | lsm1 | spliceosome | Chapman et al. [2] |
| Striped bass | lsm7 | spliceosome | Chapman et al. [2] |
| Striped bass | ltn1 | cell cycle, ubiquitin ligases | Chapman et al. [2] |
| Striped bass | map2k2 | mitogen activated protein kinase | Chapman et al. [2] |
| Striped bass | mapre1 | cell division | Chapman et al. [2] |
| Striped bass | mbip | growth factor, cell signaling, MAPK | Chapman et al. [2] |
| Striped bass | mcl1 | apoptosis, ubiquinated-proteins | Chapman et al. [2] |
| Striped bass | metap2 | spliceosome, malignant tumors | Chapman et al. [2] |
| Halibut | mhc1a | immune response | Mommens et al.* [49] |
| Halibut | mhc2a | immune response | Mommens et al.* [49] |
| Striped bass | mphosph10 | cell cycle, cell division, spliceosome | Chapman et al. [2] |
| Trout | mr-1 | hydroxyacyl glutathione hydrolase | Bonnet et al.* [47] |
| Striped bass | mrpl32 | spliceosome | Chapman et al. [2] |
| Striped bass | mrps30 | apoptosis, ubiquinated-proteins | Chapman et al. [2] |
| Halibut | ms4a8a | cell signaling | Mommens et al.* [49] |
| Striped bass | msh6 | DNA repair, malignant tumors | Chapman et al. [2] |
| Trout | myo1b | neuronal development | Bonnet et al.* [47] |
| Striped bass | ncapg2 | cell division | Chapman et al. [2] |
| Striped bass | ncl | spliceosome | Chapman et al. [2] |
| Striped bass | nop10 | spliceosome | Chapman et al. [2] |
| Trout | npm2 | histone chaperone, chromatin | Aegerter et al.* [48] |
| Trout | ntan1 | ubiquitin-dependent protein turnover | Bonnet et al.* [47] |
| Striped bass | nucb1 | growth factors, cell signaling | Chapman et al. [2] |
| Striped bass | nudt21 | spliceosome | Chapman et al. [2] |
| Sea bream | P38α | mitogen activated protein kinase | Fernández et al. [45] |
| Sea bream | P38δ | mitogen activated protein kinase | Fernández et al. [45] |
| Striped bass | pdcl | growth factors, cell signaling | Chapman et al. [2] |
| Halibut | pd-l1 | immune response, apoptosis | Mommens et al.* [49] |
| Halibut | pdpk1 | expression in testis | Mommens et al.* [51] |
| Striped bass | pfdn4 | spliceosome | Chapman et al. [2] |
| Trout | phb2 | cell signaling, estrogen receptor | Bonnet et al.* [47] |
| Sea bass | plec | cytoskeletal structure | Żarski et al. [42] |
| Sea bass | polk | DNA repair | Żarski et al. [42] |
| Striped bass | pomp | cell cycle, 26S-proteasome | Chapman et al. [2] |
| Striped bass | prpf39 | spliceosome | Chapman et al. [2] |
| Striped bass | prss27 | growth factor, cell signaling | Chapman et al. [2] |
| Striped bass | psma7 | cell cycle, 26S-proteasome | Chapman et al. [2] |
| Halibut | psmb9 | cell cycle, 26S-proteasome | Mommens et al.* [49] |
| Striped bass | psmd14 | cell cycle, 26S-proteasome | Chapman et al. [2] |
| Striped bass | ptges3 | Prostaglandin synthesis, spliceosome | Chapman et al. [2] |
| Trout | ptgs2 | Prostaglandin synthesis, cell signaling | Aegerter et al.* [48] |
| Trout | pyc | metabolism | Bonnet et al.* [47] |
| Striped bass | rad23a | cell cycle, ubiquitin ligase, DNA repair | Chapman et al. [2] |
| Striped bass | rap1b | malignant tumors, growth factor | Chapman et al. [2] |
| Striped bass | rbm22 | cell division, spliceosome | Chapman et al. [2] |
| Striped bass | rchy1 | cell cycle, ubiquitin ligase | Chapman et al. [2] |
| Several | rnf213 | ubiquitin ligase, low quality eggs | Mommens et al.* [49];Żarski et al. [42] |
| Trout | rpl24 | protein synthesis | Bonnet et al.* [47] |
| Striped bass | rps3 | DNA repair, spliceosome | Chapman et al. [2] |
| Striped bass | rps6 | protein translation, cell division | Chapman et al. [2] |
| Striped bass | smn | spliceosome | Chapman et al. [2] |
| Striped bass | snrnp27 | spliceosome | Chapman et al. [2] |
| Striped bass | snrpa | spliceosome | Chapman et al. [2] |
| Striped bass | sprk1 | spliceosome | Chapman et al. [2] |
| Striped bass | srp54 | spliceosome | Chapman et al. [2] |
| Striped bass | ssx2ip | malignant tumors | Chapman et al. [2] |
| Striped bass | tbc1d25 | growth factors, cell signaling | Chapman et al. [2] |
| Striped bass | tcirg1 | malignant tumors | Chapman et al. [2] |
| Striped bass | tlk1 | cell division | Chapman et al. [2] |
| Striped bass | tmbim6 | apoptosis, ubiquinated-proteins | Chapman et al. [2] |
| Striped bass | tollip | cell cycle, ubiquitin ligase | Chapman et al. [2] |
| Striped bass | tsen54 | spliceosome | Chapman et al. [2] |
| Sea bass | tsfm | protein synthesis, egg quality | Żarski et al. [42] |
| Striped bass | tuba3e | cell division | Chapman et al. [2] |
| Several | tub-β | cytoskeleton | Aegerter et al.* [48]; Fernández et al. [45] |
| Striped bass | ube2f | cell cycle, ubiquitin ligase | Chapman et al. [2] |
| Striped bass | ube2l3 | cell cycle, ubiquitin ligase | Chapman et al. [2] |
| Striped bass | ubl5 | cell cycle, ubiquitin ligase | Chapman et al. [2] |
| Striped bass | ubox5 | cell cycle, ubiquitin ligase | Chapman et al. [2] |
| Sea bass | usp5 | cell cycle, ubiquitin ligase | Żarski et al. [42] |
| Striped bass | usp11 | cell cycle, ubiquitin ligase | Chapman et al. [2] |
| Striped bass | usp14 | cell cycle, ubiquitin ligase | Chapman et al. [2] |
| Striped bass | utp18 | spliceosome | Chapman et al. [2] |
| Striped bass | wdr3 | cell cycle, spliceosome | Chapman et al. [2] |
| Halibut | xpo1 | nuclear export of cellular protein | Mommens et al.* [51] |
| Striped bass | znf143 | spliceosome | Chapman et al. [2] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Reading, B.J.; Andersen, L.K.; Ryu, Y.-W.; Mushirobira, Y.; Todo, T.; Hiramatsu, N. Oogenesis and Egg Quality in Finfish: Yolk Formation and Other Factors Influencing Female Fertility. Fishes 2018, 3, 45. https://doi.org/10.3390/fishes3040045
Reading BJ, Andersen LK, Ryu Y-W, Mushirobira Y, Todo T, Hiramatsu N. Oogenesis and Egg Quality in Finfish: Yolk Formation and Other Factors Influencing Female Fertility. Fishes. 2018; 3(4):45. https://doi.org/10.3390/fishes3040045
Chicago/Turabian StyleReading, Benjamin J., Linnea K. Andersen, Yong-Woon Ryu, Yuji Mushirobira, Takashi Todo, and Naoshi Hiramatsu. 2018. "Oogenesis and Egg Quality in Finfish: Yolk Formation and Other Factors Influencing Female Fertility" Fishes 3, no. 4: 45. https://doi.org/10.3390/fishes3040045
APA StyleReading, B. J., Andersen, L. K., Ryu, Y.-W., Mushirobira, Y., Todo, T., & Hiramatsu, N. (2018). Oogenesis and Egg Quality in Finfish: Yolk Formation and Other Factors Influencing Female Fertility. Fishes, 3(4), 45. https://doi.org/10.3390/fishes3040045




