Methods for Durability Testing and Lifetime Estimation of Thermal Interface Materials in Batteries
Abstract
:1. Introduction: Thermal Management in Electric Vehicle Batteries
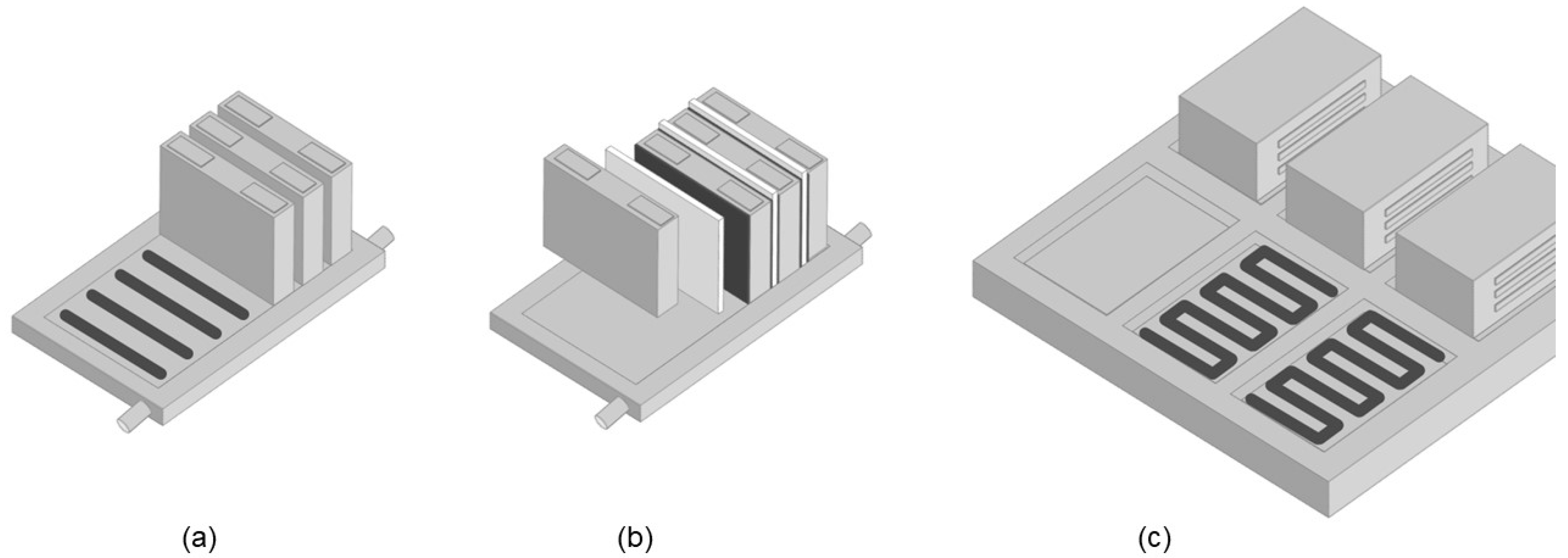
2. Thermal Interface Materials
2.1. Composition of TIMs
2.2. Types of TIMs
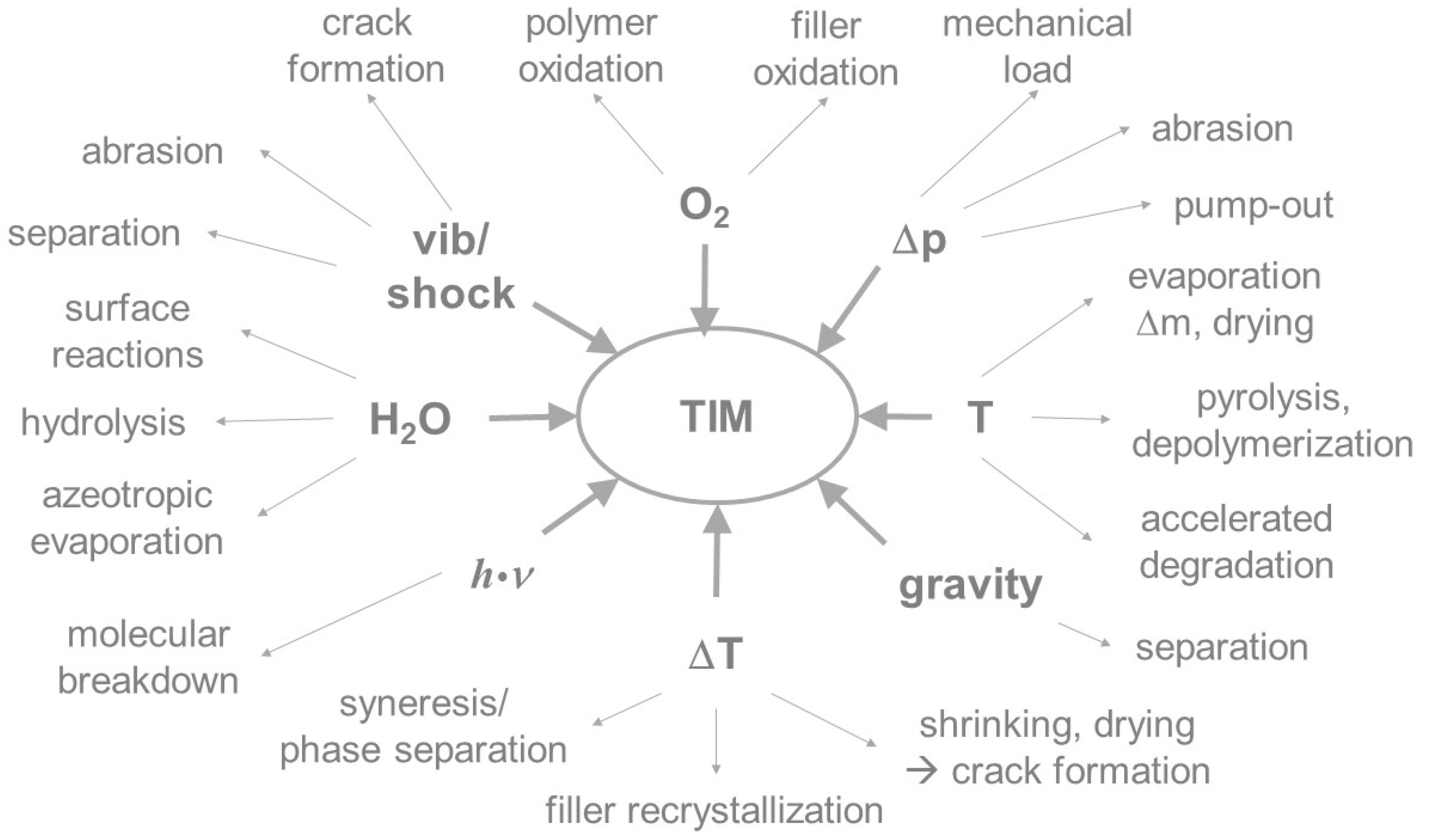
3. Degradation of Thermal Interface Materials
4. Accelerated Ageing Test Methods
4.1. Overview
4.2. High-Temperature Storage
4.3. Temperature Cycling
4.4. Climatic Storage/Alternating Climate Test
4.5. Power Cycling
4.6. Vibration Testing
4.7. Complex/Combined Testing
5. Modeling for Lifetime Estimation
5.1. General Considerations and Examples
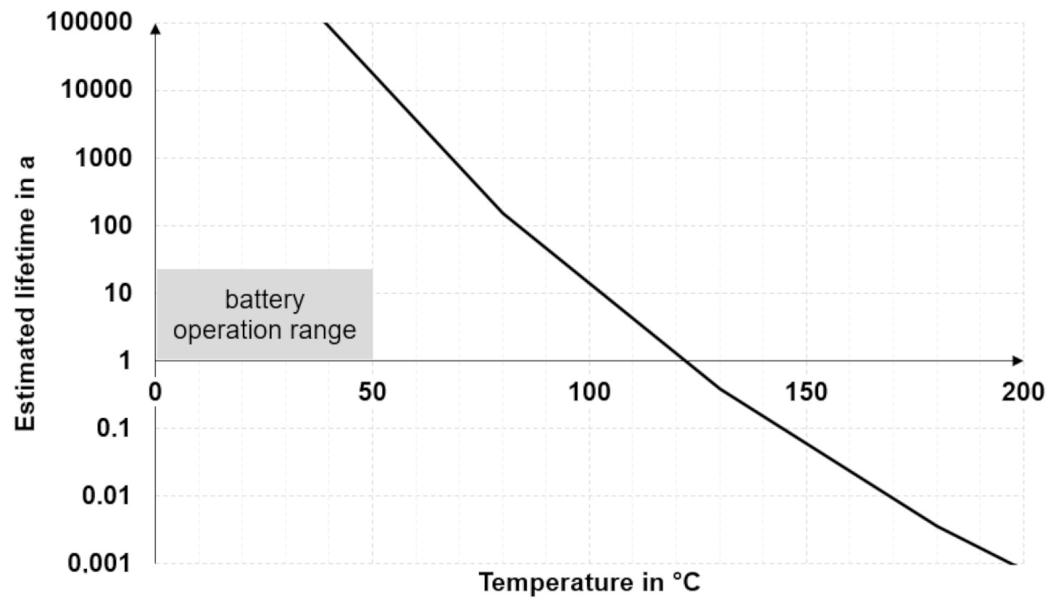
5.2. Arrhenius Model
- Monomolecular reaction
- Pre-exponential factor A ≠ f(T)
5.3. Lawson Model
5.4. Coffin-Manson Model
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Christen, R.; Rizzo, G.; Gadola, A.; Stöck, M. Test method for thermal characterization of Li-ion cells and verification of cooling concepts. Batteries 2017, 3, 3. [Google Scholar] [CrossRef]
- Thermal Challenges in Automotive High Density Lithium-Ion Battery Packs. pp. 18–20. Available online: http://www.qats.com/download/Qpedia_Dec09_Thermal_Challenges_in_Auto_High_Density_Lithium_ion_battery_packs.ashx (accessed on 12 March 2019).
- Zeyen, M.G.; Wiebelt, A. Thermisches Management der Batterie. In Handbuch Lithium-Ionen-Batterien; Korthauer, R., Ed.; Springer: Berlin/Heidelberg, Germany, 2013; pp. 165–175. ISBN 978-3-642-30653-2. [Google Scholar]
- Maurer, A. Rewards for good conduct(ivity). Eur. Coat. J. 2016, 2, 42–47. [Google Scholar]
- Gepp, M.; Filimon, R.; Koffel, S.; Lorentz, V.R.H.; März, M. Advanced thermal management for temperature homogenization in high-power lithium-ion battery systems based on prismatic cells. In Proceedings of the IEEE 24th International Symposium on Industrial Electronics (ISIE), Buzios, Brazil, 3–5 June 2015; pp. 1230–1235. [Google Scholar] [CrossRef]
- Maurer, A.; Kalka, J.; Wießler, A. Smart design of electric vehicle batteries and power electronics using thermal interface materials. In Proceedings of the EVS30 International Electric Vehicle Symposium, Stuttgart, Germany, 9–11 October 2017. [Google Scholar]
- Beetsma, J. Sedimentation during Storage. Prospector Knowledge Center, Posted on May 2016. Available online: https://knowledge.ulprospector.com/517/pc-sedimentation-during-storage (accessed on 28 December 2018).
- Beetsma, J. Coating Film Defects. Prospector Knowledge Center, Posted on April 18, 2014. Available online: https://knowledge.ulprospector.com/451/pc-coating-film-defects (accessed on 28 December 2018).
- Liebchen, R. Zuverlässigkeit in der Leistungselektronik: Möglichkeiten der thermischen Analyse und Lebensdauervorhersage. In Proceedings of the ZFW Tagung Leistungselektronik, Stuttgart, Germany, 16 June 2016. [Google Scholar]
- Schulz, M. Mythen und Legenden aus dem Reich der Wärmeleitmaterialien. Elektronik Industrie 2013, 4, 62–65. [Google Scholar]
- Sarvar, F.; Whalley, D.C.; Conway, P.P. Thermal interface materials—A review of the state of the art. In Proceedings of the 1st IEEE Electronic System Integration Technology Conference, Dresden, Germany, 5–7 September 2006; Volume 2, pp. 1292–1302. Available online: https://dspace.lboro.ac.uk/2134/4197 (accessed on 19 February 2019).
- Misra, S. Beyond thermal grease—Enhancing thermal performance and reliability. In Proceedings of the PCIM Europe 2016, Nuremberg, Germany, 10–12 May 2016. [Google Scholar]
- Eleffendi, M.A.; Yang, L.; Agyakwa, P.; Johnson, C.M. Quantification of cracked area in thermal path of high-power multi-chip modules using transient thermal impedance measurement. Microelectron. Reliab. 2016, 59, 73–83. [Google Scholar] [CrossRef]
- Pan, C.A.; Yeh, C.T.; Qiu, W.C.; Lin, R.Z.; Hung, L.Y.; Ng, K.T.; Lin, C.F.; Chung, C.K.; Jiang, D.S.; Hsiao, C.S. Assembly and reliability challenges for next generation high thermal TIM materials. In Proceedings of the IEEE 67th Electronic Components and Technology Conference (ECTC), Lake Buena Vista, FL, USA, 30 May–2 June 2017. [Google Scholar] [CrossRef]
- Chen, Y.; Feng, Y.; Zhao, J.; Shen, J.; Feng, M. Oil bleed from elastomeric thermal silicone conductive pads. Front. Chem. Sci. Eng. 2016, 10, 509–516. [Google Scholar] [CrossRef]
- Nnebe, I.M.; Feger, C. Drainage-induced dry-out of thermal greases. IEEE Trans. Adv. Packag. 2008, 31, 512–518. [Google Scholar] [CrossRef]
- Aoyagi, Y.; Chung, D.D.L. Effects of antioxidants and the solid component on the thermal stability of polyol-ester-based thermal pastes. J. Mater. Sci. 2007, 42, 2358–2375. [Google Scholar] [CrossRef]
- Cyclic aging of Lithium-ion batteries. In Proceedings of the Mat4Bat Summer School, La Rochelle, France, 2–4 June 2015; Available online: http://mat4bat.eu/wp-content/uploads/2014/03/Cycling-Aging-of-Lithium-Ion-batteries_Grolleau_EIGSI.pdf (accessed on 28 December 2018).
- SAE International Standard J2288_20080. Life Cycle Testing of Electric Vehicle Battery Modules. 2008. Available online: https://doi.org/10.4271/J2288_200806 (accessed on 12 March 2019).
- Shirk, M.; Shirk, M.; Wishart, J. Effects of Electric Vehicle Fast Charging on Battery Life and Vehicle Performance; SAE Technical Paper 2015-01-1190; SAE International: Warrendale, PA, USA, 2015; Available online: https://doi.org/10.4271/2015-01-1190 (accessed on 28 December 2018).
- VW 80000. Electric and Electronic Components in Motor Vehicles up to 3.5 t—General Requirements, Test Conditions and Test; Volkswagen Group Standard (internal document), Volkswagen AG: Wolfsburg, Germany, 2013. [Google Scholar]
- FreedomCAR Battery Test Manual for Power-Assist Hybrid Electric Vehicles. DOE/ID-11069; 2003; p. G-6. Available online: https://avt.inl.gov/sites/default/files/pdf/battery/freedomcar_manual_04_15_03.pdf (accessed on 27 December 2018).
- Eyerer, P.; Hirth, T.; Elsner, P. Polymer Engineering; Springer: Berlin/Heidelberg, Germany, 2008; p. 600ff. ISBN 978-3-540-72419-3. [Google Scholar]
- Due, J.; Robinson, A. Reliability of thermal interface materials: A review. Appl. Therm. Eng. 2013, 50, 455–463. [Google Scholar] [CrossRef]
- IEC 60216-X/DIN EN 60216-X Standard Series. Electrical Insulation Materials—Thermal Endurance Properties, 2015–2018. Available online: https://webstore.iec.ch/publication/1036 (accessed on 12 March 2019).
- Khatir, Z.; Ousten, J.-P. Study of thermal interfaces aging for power electronics applications. In Proceedings of the 14th European Conference on Power Electronics and Applications, Birmingham, UK, 30 August–1 September 2011. [Google Scholar]
- JEDEC Solid State Technology Association Standard: JESD22-A104D Temperature Cycling. 2009. Available online: https://www.jedec.org/sites/default/files/docs/22a104d.pdf (accessed on 12 March 2019).
- Roy, C.K.; Bhavnani, S.; Hamilton, M.C.; Johnson, R.W.; Knight, R.W.; Harris, D.K. Application of Low Melt Alloys as Compliant Thermal Interface Materials: A Study of Performance and Degradation under Thermal Duress. Electron. Cool. 2015, 20. Available online: https://www.electronics-cooling.com/2015/05/application-of-low-melt-alloys-as-compliant-thermal-interface-materials-a-study-of-performance-and-degradation-under-thermal-duress (accessed on 28 December 2018).
- Wikipedia: Temperature Cycling. Available online: https://en.wikipedia.org/wiki/Temperature_cycling (accessed on 27 December 2018).
- IEC 61215/DIN EN 61215, Terrestrial Photovoltaic (PV) Modules—Design Qualification and Type Approval. 2005. Available online: https://webstore.iec.ch/publication/24312 (accessed on 12 March 2019).
- JEDEC Solid State Technology Association Standard: JESD22-A118B Accelerated Moisture Resistance—Unbiased HAST. 2015. Available online: https://www.jedec.org/standards-documents/docs/jesd-22-a118 (accessed on 12 March 2019).
- Nowak, T.; Müller, M.; Walter, H.; Hölck, O.; Wüst, F.; Wittler, O.; Lang, K.D. Approach for reliability of thermal interface materials in battery cell sensors. In Proceedings of the 19th International Workshop on Thermal Investigations of ICs and Systems (THERMINIC), Berlin, Germany, 25–27 September 2013. [Google Scholar] [CrossRef]
- IEC 60068-2-30. Environmental Testing—Part 2-30: Damp Heat, Cyclic. 2005. Available online: https://webstore.iec.ch/publication/515 (accessed on 12 March 2019).
- Volkswagen AG. PV 1200: Prüfung der Klimawechselfestigkeit (Environmental Cycle); Volkswagen Group Standard (internal document): Wolfsburg, Germany, 2004. [Google Scholar]
- Schulz, M.; Pohl, W.; Allen, S. Optimizing thermal interface material. In Proceedings of the PCIM Europe 2012, Nuremberg, Germany, 8–10 May 2012. [Google Scholar]
- Nnebe, I.; Feger, C.; McGlashan-Powell, M. Power cycling effects on the structural stability of thermal pastes used in microelectronics. MRS Proc. 2006, 977, 0977-FF12-21. [Google Scholar] [CrossRef]
- Wikipedia: Vibration Testing. Available online: https://en.wikipedia.org/wiki/Vibration#Vibration_testing (accessed on 27 December 2018).
- IEC 60068-2-64. Environmental Testing—Part 2-64: Tests—Test Fh: Vibration, Broadband Random and Guidance. 2008. Available online: https://webstore.iec.ch/publication/547 (accessed on 12 March 2019).
- Volkswagen AG. VW 82161: Betriebsfestigkeit Hochvoltspeicher, Anforderungen und Prüfungen; Volkswagen Group Standard (internal document): Wolfsburg, Germany, 2012. [Google Scholar]
- Maurer, A.; Wießler, A.; Griesinger, A. Reliability and lifetime estimation of thermal interface materials for EV battery assembly. In Proceedings of the Battery Show Europe 2018, Hannover, Germany, 15–17 May 2018. [Google Scholar]
- Jakob, F.; Kimmelmann, M.; Bertsche, B. Methodik zur Auswahl von Raffungsmodellen für beschleunigte Versuche. Konstruktion 2015, 67, 86–90. [Google Scholar]
- Prasher, R. Thermal interface materials: Historical perspective, status, and future directions. Proc. IEEE 2006, 94, 1571–1586. [Google Scholar] [CrossRef]
- ASTM E 1641-04 Standard Test Method for Decomposition Kinetics by Thermogravimetry Using the Ozawa/Flynn/Wall Method; ASTM International: West Conshohocken, PA, USA, 2004.
- Goel, N.; Anoop, T.K.; Bhattacharya, A.; Cervantes, J.A.; Mongia, R.K.; Machiroutu, S.V.; Lin, H.L.; Huang, Y.C.; Fan, K.C.; Denq, B.L.; et al. Technical review of characterization methods for thermal interface materials (TIM). In Proceedings of the 11th Intersociety Conference on Thermal and Thermomechanical Phenomena in Electronic Systems, Orlando, FL, USA, 28–31 May 2008; p. 248. [Google Scholar]
- Chiu, C.P.; Chandran, B.; Mello, K.; Kelley, K. An accelerated reliability test method to predict thermal grease pump-out in flip-chip applications. In Proceedings of the 51st Electronic Components and Technology Conference, Orlando, FL, USA, 29 May–1 June 2001. [Google Scholar] [CrossRef]
- Nelson, C.; Galloway, J.; Henry, C.; Kelley, W. Thermal performance of TIMs during compressive and tensile stress states. In Proceedings of the 33rd Thermal Measurement, Modeling & Management Symposium (SEMI-THERM), San Jose, CA, USA, 13 March 2017. [Google Scholar] [CrossRef]
- Lawson, R.W. A review of the status of plastic encapsulated semiconductor components reliability. Br. Telecomm. J. 1984, 2, 95–111. [Google Scholar]
- Coffin, L.F., Jr. A study of the effects of cyclic thermal stresses on a ductile metal. Trans. ASME 1954, 76, 931–950. [Google Scholar]
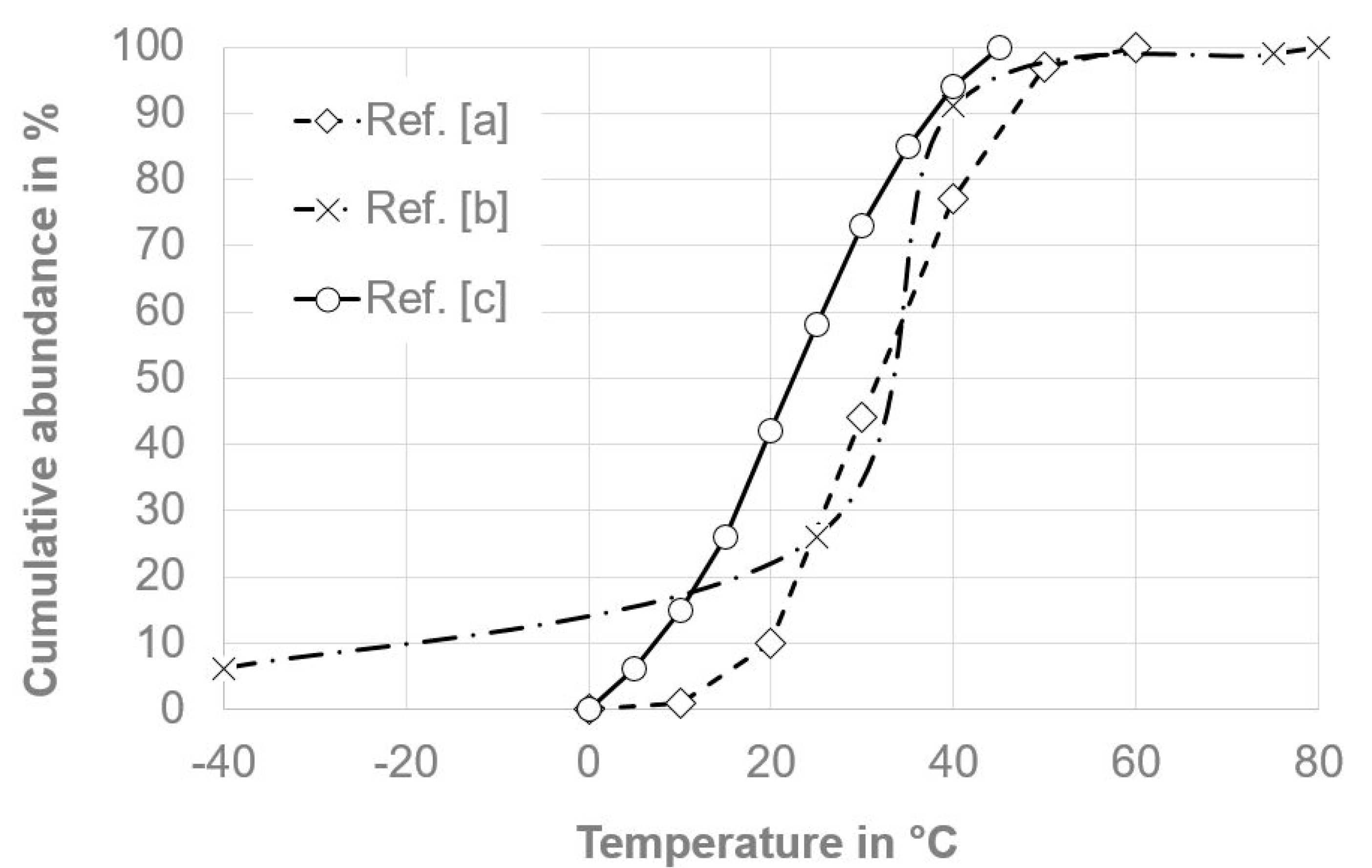
| Temperature in °C | Abundance pi in % | Acceleration Factor AT,i | Abundance/Acceleration |
|---|---|---|---|
| 0 | 0% | 93.45 | - |
| 10 | 1% | 47.57 | 0.00 |
| 20 | 9% | 25.36 | 0.02 |
| 30 | 34% | 14.09 | 0.02 |
| 40 | 33% | 8.13 | 0.04 |
| 50 | 20% | 4.85 | 0.04 |
| 60 | 3% | 2.99 | 0.01 |
| Test temperature in °C | 85 | Lifetime in h | 8000 |
| Activation energy in eV | 0.45 | Test time in h | 958 |
| Average acceleration factor | 8.4 | ||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stadler, R.; Maurer, A. Methods for Durability Testing and Lifetime Estimation of Thermal Interface Materials in Batteries. Batteries 2019, 5, 34. https://doi.org/10.3390/batteries5010034
Stadler R, Maurer A. Methods for Durability Testing and Lifetime Estimation of Thermal Interface Materials in Batteries. Batteries. 2019; 5(1):34. https://doi.org/10.3390/batteries5010034
Chicago/Turabian StyleStadler, Ralf, and Arno Maurer. 2019. "Methods for Durability Testing and Lifetime Estimation of Thermal Interface Materials in Batteries" Batteries 5, no. 1: 34. https://doi.org/10.3390/batteries5010034
APA StyleStadler, R., & Maurer, A. (2019). Methods for Durability Testing and Lifetime Estimation of Thermal Interface Materials in Batteries. Batteries, 5(1), 34. https://doi.org/10.3390/batteries5010034




