Reviewing the Tradeoffs between Sunburn Mitigation and Red Color Development in Apple under a Changing Climate
Abstract
1. Introduction
2. External Quality Standards
3. Apple Sunburn
3.1. Sunburn Physiology for Fruit
3.2. Sunburn Induction Factors
| Sunburn Incidence | Cultivar | Location of Fruit Temperature Measurement | Temperature | Light Exposure | Location | Reference | |
|---|---|---|---|---|---|---|---|
| Sunburn necrosis (SN) | Presence | ‘Gala’ | Surface | 52.2 °C (10 min) | No | Washington, UT, USA | [24] |
| Sunburn browning (SB) | Presence | ‘Gala’ | Surface | 47.8 °C (60 min) | Yes | Washington, UT, USA | [24] |
| Sunburn symptoms | Presence | ‘Braeburn’ | Flesh | 40 °C | Yes | Auckland, New Zealand | [47] |
| Sunburn symptoms | 5% | ‘Mondial Gala’ | Flesh | 44 °C | Yes | Lleida, Spain | [8] |
| SN + SB | 15% | ‘Cripps’ Pink’ | Surface | 41 °C * | Yes | Stellenbosch, South Africa | [48] |
| SN + SB | 9% | ‘Cripps’ Pink’ | Surface | 33 °C * | Yes | Stellenbosch, South Africa | [49] |
| SN + SB | 14% | ‘Fuji’ | Surface | 45 °C * | Yes | Sobo-myeon, Korea | [50] |
| SN + SB | 23% | ‘Fuji’ | Surface | 48 °C * | Yes | Biobío, Chile | [9] |
| SN + SB | 5% | ‘Fuji’ | Surface | 35 °C * | Yes | Ferrara, Italy | [51] |
| SN + SB | 41% | ‘Fuji Raku Raku’ | Surface | 46 °C * | Yes | Ñuble, Chile | [52] |
| SN + SB | 19% | ‘Gala’ | Surface | 52 °C * | Yes | Biobío, Chile | [9] |
| SN + SB | 15% | ‘Gala’ | Surface | 47 °C * | Yes | Gansu, China | [53] |
| SN + SB | 39% | ‘Gala Brookfield’ | Surface | 48 °C * | Yes | Ñuble, Chile | [52] |
| SN + SB | 45% | ‘Granny Smith’ | Surface | 37 °C * | Yes | Grabouw, South Africa | [10] |
| SN + SB | 39% | ‘Granny Smith’ | Surface | 40 °C * | Yes | Grabouw, South Africa | [10] |
| SN + SB | 50% | ‘Granny Smith’ | Surface | 44 °C * | Yes | San José, Uruguay | [54] |
| SN + SB | 30% | ‘Honeycrisp’ | Surface | 41 °C * | Yes | Washington, UT, USA | [55] |
| SN + SB | 27% | ‘Honeycrisp’ | Surface | 42 °C * | Yes | New York, NY, USA | [56] |
| SN + SB | 13% | ‘Honeycrisp’ | Surface | 44 °C * | Yes | New York, NY, USA | [56] |
| SN + SB | 19% | ‘Royal Gala’ | Surface | 47 °C * | Yes | Stellenbosch, South Africa | [48] |
| SN + SB | 16% | ‘Royal Gala’ | Surface | 48 °C * | Yes | Shepparton, Australia | [11] |
| SN + SB | 17% | ‘Royal Gala’ | Surface | 53 °C * | Yes | Shepparton, Australia | [57] |
3.3. Sunburn Mitigation Strategies
4. Color Development in Apples
4.1. Improving Red Color
4.2. Tradeoffs between Sunburn Susceptibility/Mitigation and Red Color
5. The Impact of Changing Climate on Fruit Quality
6. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- IPCC. Climate Change 2022: Impacts, Adaptation, and Vulnerability. Contribution of Working Group II to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change; Pörtner, H.-O., Roberts, D.C., Tignor, M., Poloczanska, E.S., Mintenbeck, K., Alegría, A., Craig, M., Langsdorf, S., Löschke, S., Möller, V., et al., Eds.; Cambridge University Press: Cambridge, UK; New York, NY, USA, 2022; p. 3056. [Google Scholar]
- Chmielewski, F.M.; Müller, A.; Bruns, E. Climate changes and trends in phenology of fruit trees and field crops in Germany, 1961–2000. Agric. For. Meteorol. 2004, 121, 69–78. [Google Scholar] [CrossRef]
- Wolfe, D.W.; Schwartz, M.D.; Lakso, A.N.; Otsuki, Y.; Pool, R.M.; Shaulis, N.J. Climate change and shifts in spring phenology of three horticultural woody perennials in northeastern USA. Int. J. Biometeorol. 2005, 49, 303–309. [Google Scholar] [CrossRef] [PubMed]
- Brunner, J.F.; Jones, W.; Kupferman, E.; Xiao, C.L.; Faubion, D.; Tangren, G. Information from the careful evaluation of packingline culls. In Proceedings of the Washington Tree Fruit Postharvest Conference, Wenatchee, WA, USA, 2–3 December 2003; pp. 1–6. [Google Scholar]
- Musacchi, S.; Serra, S. Apple Fruit Quality: Overview on pre-harvest factors. Sci. Hortic. 2018, 234, 409–430. [Google Scholar] [CrossRef]
- Barber, H.N.; Sharpe, P.J.H. Genetics and physiology of sunscald of fruits. Agric. Meteorol. 1971, 8, 175–191. [Google Scholar] [CrossRef]
- Munné-Bosch, S.; Vincent, C. Physiological mechanisms underlying fruit sunburn. Crit. Rev. Plant Sci. 2019, 38, 140–157. [Google Scholar] [CrossRef]
- Iglesias, I.; Alegre, S.A. The effect of anti-hail nets on fruit protection, radiation, temperature, quality and probability of ‘mondial gala’ apples. J. Appl. Hortic. 2006, 8, 91–100. [Google Scholar] [CrossRef]
- Olivares-Soto, H.; Bastías, R.M. Photosynthetic efficiency of apples under protected shade nets. Chil. J. Agric. Res. 2018, 78, 126–138. [Google Scholar] [CrossRef]
- Mupambi, G.; Schmeisser, M.; Dzikiti, S.; Reynolds, S.; Steyn, W.J. Ineffectiveness of foliar S-ABA application as an apple sunburn suppressant explained through effects on peel biochemistry and leaf ecophysiology. Sci. Hortic. 2018, 232, 256–263. [Google Scholar] [CrossRef]
- Darbyshire, R.; McClymont, L.; Goodwin, I. Sun Damage Risk of royal gala apple in fruit-growing districts in Australia. N. Z. J. Crop Hortic. Sci. 2015, 43, 222–232. [Google Scholar] [CrossRef]
- Schrader, L.; Sun, J.; Zhang, J.; Felicetti, D.; Tian, J. Heat and light-induced Apple Skin Disorders: Causes and prevention. Acta Hortic. 2008, 772, 51–58. [Google Scholar] [CrossRef]
- Racsko, J.; Schrader, L.E. Sunburn of Apple Fruit: Historical Background, recent advances and future perspectives. Crit. Rev. Plant Sci. 2012, 31, 455–504. [Google Scholar] [CrossRef]
- Wagenmakers, P.S.; Callesen, O. Light distribution in apple orchard systems in relation to production and fruit quality. J. Hortic. Sci. 1995, 70, 935–948. [Google Scholar] [CrossRef]
- Racskó, J.; Nagy, J.; Szabó, Z.; Major, M.; Nyéki, J. The impact of location, row direction, plant density and rootstock on the sunburn damage of Apple cultivars. Int. J. Hortic. Sci. 2005, 11, 19–30. [Google Scholar] [CrossRef]
- Saitone, T.L.; Sexton, R.J. Impacts of minimum quality standards imposed through marketing orders or related producer organizations. Am. J. Agric. Econ. 2010, 92, 164–180. [Google Scholar] [CrossRef]
- Bockstael, N.E. The welfare implications of minimum quality standards. Am. J. Agric. Econ. 1984, 66, 466–471. [Google Scholar] [CrossRef]
- Dar, J.A.; Wani, A.A.; Ahmed, M.; Nazir, R.; Zargar, S.M.; Javaid, K. Peel colour in Apple (Malus × domestica borkh.): An economic quality parameter in fruit market. Sci. Hortic. 2019, 244, 50–60. [Google Scholar] [CrossRef]
- USDA; ARS. U.S. Standards for Grades of Apples. Available online: https://www.ams.usda.gov/grades-standards/apple-grades-standards (accessed on 15 February 2023).
- Johnson, L.K.; Dunning, R.D.; Gunter, C.C.; Dara Bloom, J.; Boyette, M.D.; Creamer, N.G. Field measurement in vegetable crops indicates need for reevaluation of on-farm food loss estimates in North America. Agric. Syst. 2018, 167, 136–142. [Google Scholar] [CrossRef]
- Baker, G.A.; Gray, L.C.; Harwood, M.J.; Osland, T.J.; Tooley, J.B. On-farm food loss in northern and Central California: Results of field survey measurements. Resour. Conserv. Recycl. 2019, 149, 541–549. [Google Scholar] [CrossRef]
- FAO. Food Loss Prevention in Perishable Crops; FAO Agricultural Service Bulletin No. 43; FAO Statistics Division: Rome, Italy, 1981. [Google Scholar]
- Gustavsson, J.; Cederberg, C.; Sonesson, U.; Emanuelsson, A. The Methodology of the FAO Study: “Global Food Losses and Food Waste—Extent, Causes and Prevention”-FAO, 2011; SIK Report No. 857; Swedish Institute for Food and Biotechnology: Goteborg, Sweden, 2013. [Google Scholar]
- Schrader, L.E.; Zhang, J.; Duplaga, W.K. Two types of sunburn in apple caused by high fruit surface (peel) temperature. Plant Health Prog. 2001, 2, 3–8. [Google Scholar] [CrossRef]
- Schrader, L.E.; Sun, J.; Felicetti, D.; Seo, J.-H.; Jedlow, L.; Zhang, J. Stress-induced disorders: Effects on apple fruit quality. In Proceedings of the Washington Tree Fruit Postharvest Conference, Wenatchee, WA, USA, 2–3 December 2003. [Google Scholar]
- Schrader, L.E.; Sun, J.; Zhang, J.; Felicetti, D. Stress management in tree fruit production. In Proceedings of the 101st Annual Meeting, Washington State Association Horticultural, Wenatchee, WA, USA, 5–7 December 2005; p. 3. [Google Scholar]
- Moore, M.H.; Rogers, W.S. Sun Scald of Fruits; Annual Report; East Malling Research Station: West Malling, UK, 1943; pp. 50–53. [Google Scholar]
- Bergh, O.; Franken, J.; van Zyl, E.J.; Kloppers, F.; Dempers, A. Sunburn on apples—Preliminary results of an investigation conducted during the 1978/79 season. Decid. Fruit Grow. 1980, 30, 8–22. [Google Scholar]
- Arseneault, M.H.; Cline, J.A. A review of Apple Preharvest Fruit Drop and practices for horticultural management. Sci. Hortic. 2016, 211, 40–52. [Google Scholar] [CrossRef]
- Felicetti, D.A.; Schrader, L.E. Photooxidative sunburn of apples: Characterization of a third type of Apple Sunburn. Int. J. Fruit Sci. 2008, 8, 160–172. [Google Scholar] [CrossRef]
- Naschitz, S.; Naor, A.; Sax, Y.; Shahak, Y.; Rabinowitch, H.D. Photo-oxidative sunscald of Apple: Effects of temperature and light on fruit peel photoinhibition, bleaching and short-term tolerance acquisition. Sci. Hortic. 2015, 197, 5–16. [Google Scholar] [CrossRef]
- McTavish, C.K.; Poirier, B.C.; Torres, C.A.; Mattheis, J.P.; Rudell, D.R. A convergence of sunlight and cold chain: The influence of sun exposure on postharvest Apple Peel Metabolism. Postharvest Biol. Technol. 2020, 164, 111164. [Google Scholar] [CrossRef]
- Morales-Quintana, L.; Waite, J.M.; Kalcsits, L.; Torres, C.A.; Ramos, P. Sun injury on apple fruit: Physiological, biochemical and molecular advances, and future challenges. Sci. Hortic. 2020, 260, 108866. [Google Scholar] [CrossRef]
- Rabinowitch, H.D.; Kedar, N.; Budowski, P. Induction of sunscald damage in tomatoes under natural and controlled conditions. Sci. Hortic. 1974, 2, 265–272. [Google Scholar] [CrossRef]
- Van Amerongen, H.; Wientjes, E. Harvesting light. In Photosynthesis in Action; Academic Press: Cambridge, MA, USA, 2022; pp. 3–16. [Google Scholar]
- Su, X.; Fan, X.; Shao, R.; Guo, J.; Wang, Y.; Yang, J.; Yang, Q.; Guo, L. Physiological and itraq-based proteomic analyses reveal that melatonin alleviates oxidative damage in maize leaves exposed to drought stress. Plant Physiol. Biochem. 2019, 142, 263–274. [Google Scholar] [CrossRef]
- Taiz, L.; Zeiger, E. Plant Physiology; W.H. Freeman: New York, NY, USA, 2002. [Google Scholar]
- Chen, L.-S.; Li, P.; Cheng, L. Effects of high temperature coupled with high light on the balance between photooxidation and photoprotection in the sun-exposed peel of Apple. Planta 2008, 228, 745–756. [Google Scholar] [CrossRef]
- Hengari, S.; Theron, K.I.; Midgley, S.J.E.; Steyn, W.J. Response of Apple (Malus domestica Borkh.) Fruit Peel photosystems to heat stress coupled with moderate photosynthetic active radiation at different fruit developmental stages. Sci. Hortic. 2014, 178, 154–162. [Google Scholar] [CrossRef]
- Makeredza, B.; Marais, H.; Schmeisser, M.; Lötze, E.; Steyn, W.J. Ripening associated red color development masks sunburn browning in Apple Peel. HortScience 2015, 50, 814–818. [Google Scholar] [CrossRef]
- Blanke, M.M.; Lenz, F. Fruit photosynthesis. Plant Cell Environ. 1989, 12, 31–46. [Google Scholar] [CrossRef]
- Merzlyak, M.N.; Solovchenko, A.E. Photostability of pigments in ripening apple fruit: A possible photoprotective role of carotenoids during plant senescence. Plant Sci. 2002, 163, 881–888. [Google Scholar] [CrossRef]
- Yuri, J.A.; Lepe, V.; Moggia, C.; Bastias, R.; Bertschinger, L. Sunburn on apple. Schweiz. Z. Obst.-Weinbau. 2004, 8, 7–10. (In German) [Google Scholar]
- Li, L.; Peters, T.; Zhang, Q.; Zhang, J.; Huang, D. Modeling Apple Surface Temperature Dynamics based on weather data. Sensors 2014, 14, 20217–20234. [Google Scholar] [CrossRef] [PubMed]
- Ranjan, R.; Khot, L.R.; Peters, R.T.; Salazar-Gutierrez, M.R.; Shi, G. In-field crop physiology sensing aided real-time apple fruit surface temperature monitoring for sunburn prediction. Comput. Electron. Agric. 2020, 175, 105558. [Google Scholar] [CrossRef]
- Lal, N.; Sahu, N. Management strategies of sunburn in Fruit Crops-a review. Int. J. Curr. Microbiol. Appl. Sci. 2017, 6, 1126–1138. [Google Scholar] [CrossRef]
- Ferguson, I.B.; Snelgar, W.; Lay-Yee, M.; Watkins, C.B.; Bowen, J.H. Expression of heat shock protein genes in apple fruit in the field. Funct. Plant Biol. 1998, 25, 155. [Google Scholar] [CrossRef]
- Gindaba, J.; Wand, S.J.E. Comparative effects of evaporative cooling, kaolin particle film, and shade net on sunburn and fruit quality in apples. HortScience 2005, 40, 592–596. [Google Scholar] [CrossRef]
- Makeredza, B.; Schmeisser, M.; Lötze, E.; Steyn, W.J. Water stress increases sunburn in ‘Cripps’ pink’ Apple. HortScience 2013, 48, 444–447. [Google Scholar] [CrossRef]
- Song, Y.Y.; Park, M.Y.; Yang, S.J.; SaGong, D.H. Influence of air temperature during midsummer on fruit sunburn occurrence in ‘fuji’/m.9 apple tree. Korean J. Agric. For. Meteorol. 2009, 11, 127–134. [Google Scholar] [CrossRef]
- Manfrini, L.; Gatti, G.; Morandi, B.; Corelli Grappadelli, L.; Bortolotti, G.; Rossi, F.; Facini, O.; Chieco, C.; Gerin, M.; Solimando, D.; et al. Microclimatic physiological and productive effect of the overcanopy irrigation in an Apple Orchard. Acta Hortic. 2020, 1281, 377–384. [Google Scholar] [CrossRef]
- Olivares-Soto, H.; Bastías, R.M.; Calderón-Orellana, A.; López, M.D. Sunburn control by nets differentially affects the antioxidant properties of fruit peel in ‘gala’ and ‘fuji’ apples. Hortic. Environ. Biotechnol. 2020, 61, 241–254. [Google Scholar] [CrossRef]
- Feng, Y.; Li, S.; Jia, R.; Yang, J.; Su, Q.; Zhao, Z. Physiological characteristics of sunburn peel after Apple Debagged. Molecules 2022, 27, 3775. [Google Scholar] [CrossRef] [PubMed]
- Severino, V.; Arias-Sibillotte, M.; Dogliotti, S.; Frins, E.; Yuri, J.A.; González-Talice, J. Pre- and postharvest management of Sunburn in ‘granny smith’ apples (Malus × domestica Borkh) under Neotropical climate conditions. Agronomy 2021, 11, 1618. [Google Scholar] [CrossRef]
- Kalcsits, L.; Musacchi, S.; Layne, D.R.; Schmidt, T.; Mupambi, G.; Serra, S.; Mendoza, M.; Asteggiano, L.; Jarolmasjed, S.; Sankaran, S.; et al. Above and below-ground environmental changes associated with the use of photoselective protective netting to reduce sunburn in apple. Agric. For. Meteorol. 2017, 237, 9–17. [Google Scholar] [CrossRef]
- Reig, G.; Donahue, D.J.; Jentsch, P. The efficacy of four sunburn mitigation strategies and their effects on yield, fruit quality, and economic performance of Honeycrisp CV. Apples under Eastern New York (USA) climatic conditions. Int. J. Fruit Sci. 2019, 20, 541–561. [Google Scholar] [CrossRef]
- McCaskill, M.R.; McClymont, L.; Goodwin, I.; Green, S.; Partington, D.L. How hail netting reduces apple fruit surface temperature: A microclimate and modelling study. Agric. For. Meteorol. 2016, 226–227, 148–160. [Google Scholar] [CrossRef]
- Evans, R.G.; Kroeger, M.W.; Mahan, M.O. Evaporative cooling of apples by Overtree sprinkling. Appl. Eng. Agric. 1995, 11, 93–99. [Google Scholar] [CrossRef]
- Unrath, C.R. The evaporative cooling effects of overtree sprinkler irrigation on ‘red delicious’ apples. J. Am. Soc. Hortic. Sci. 1972, 97, 55–58. [Google Scholar] [CrossRef]
- Parchomchuk, P.; Meheriuk, M. Orchard cooling with pulsed overtree irrigation to prevent solar injury and improve fruit quality of ‘jonagold’ apples. HortScience 1996, 31, 802–804. [Google Scholar] [CrossRef]
- Wünsche, J.N.; Greer, D.H.; Palmer, J.W.; Lang, A.; McGhie, T. Sunburn—The cost of a high light environment. Acta Hortic. 2001, 557, 349–356. [Google Scholar] [CrossRef]
- Wünsche, J.N.; Bowen, J.; Ferguson, I.; Woolf, A.; McGhie, T. Sunburn on apples—Causes and control mechanisms. Acta Hortic. 2004, 636, 631–636. [Google Scholar] [CrossRef]
- Van den Dool, K. Evaporative Cooling of Apple and Pear Orchards. Master Thesis, Stellenbosch University, Stellenbosch, South Africa, 2006. [Google Scholar]
- Glenn, D.M.; Puterka, G.J. Particle films: A new technology for Agriculture. Hortic. Rev. 2010, 31, 1–44. [Google Scholar]
- Schrader, L.E. Scientific basis of a unique formulation for reducing sunburn of fruits. HortScience 2011, 46, 6–11. [Google Scholar] [CrossRef]
- Mupambi, G.; Anthony, B.M.; Layne, D.R.; Musacchi, S.; Serra, S.; Schmidt, T.; Kalcsits, L.A. The influence of protective netting on tree physiology and fruit quality of Apple: A Review. Sci. Hortic. 2018, 236, 60–72. [Google Scholar] [CrossRef]
- Yuri, J.A.; Sepúlveda, Á.; Moya, M.; Simeone, D.; Fuentes, M. Shade Netting and Reflective Mulches Effect on Yield and Quality Variables of ‘Gala Baigent’ and ‘Fuji Raku Raku’ Apples. N. Z. J. Crop Hortic. Sci. 2022, 1–20. [Google Scholar] [CrossRef]
- Bastías, R.M.; Manfrini, L.; Grappadelli, L.C. Exploring the potential use of photo-selective nets for fruit growth regulation in apple. Chil. J. Agric. Res. 2012, 72, 224. [Google Scholar] [CrossRef]
- Mupambi, G.; Layne, D.; Kalcsits, L.; Musacchi, S.; Serra, S.; Schmidt, T.; Hanrahan, I. Use of Protective Netting in Washington State Apple Production. 2019. Available online: https://tfrec.cahnrs.wsu.edu/kalcsits/wp-content/uploads/sites/7/2019/10/Mupambi-etal-2019-TB60E-Netting.pdf (accessed on 15 March 2023).
- Gindaba, J.; Wand, S.J.E. Comparison of Climate Ameliorating Measures to Control Sunburn on ‘Fuji’ Apples. Acta Hortic. 2008, 772, 59–64. [Google Scholar] [CrossRef]
- Basile, B.; Romano, R.; Giaccone, M.; Barlotti, E.; Colonna, V.; Cirillo, C.; Shahak, Y.; Forlani, M. Use of Photo-Selective Nets for Hail Protection of Kiwifruit Vines in Southern Italy. Acta Hortic. 2008, 770, 185–192. [Google Scholar] [CrossRef]
- Castellano, S.; Russo, G.; Mugnozza, G.S. The Influence of Construction Parameters on Radiometric Performances of Agricultural Nets. Acta Hortic. 2006, 718, 283–290. [Google Scholar] [CrossRef]
- Middleton, S.G.; Mcwaters, A.D. Maximising Apple Orchard Productivity Under Hail Netting; Final Report for Project AP96014; Department of Primary Industries and Fisheries: Brisbane, Australia, 2000. [Google Scholar]
- Shahak, Y.; Gussakovsky, E.E.; Gal, E.; Ganelevin, R. Colornets: Crop Protection and Light-Quality Manipulation in One Technology. Acta Hortic. 2004, 659, 143–151. [Google Scholar] [CrossRef]
- Abdel-Ghany, A.M.; Al-Helal, I.M. Characterization of Solar Radiation Transmission through Plastic Shading Nets. Sol. Energy Mater Sol. Cells 2010, 94, 1371–1378. [Google Scholar] [CrossRef]
- Serra, S.; Borghi, S.; Mupambi, G.; Camargo-Alvarez, H.; Layne, D.; Schmidt, T.; Kalcsits, L.; Musacchi, S. Photoselective Protective Netting Improves “Honeycrisp” Fruit Quality. Plants 2020, 9, 1708. [Google Scholar] [CrossRef] [PubMed]
- Kviklys, D.; Viškelis, J.; Liaudanskas, M.; Janulis, V.; Laužikė, K.; Samuolienė, G.; Uselis, N.; Lanauskas, J. Apple fruit growth and quality depend on the position in tree canopy. Plants 2022, 11, 196. [Google Scholar] [CrossRef]
- Espley, R.V.; Hellens, R.P.; Putterill, J.; Stevenson, D.E.; Kutty-Amma, S.; Allan, A.C. Red Colouration in Apple Fruit Is Due to the Activity of the MYB Transcription Factor, MDMYB10. Plant J. 2007, 49, 414–427. [Google Scholar] [CrossRef] [PubMed]
- Siegelman, H.W.; Hendricks, S.B. Photocontrol of Anthocyanin Synthesis in Apple Skin. Plant Physiol. 1958, 33, 185–190. [Google Scholar] [CrossRef] [PubMed]
- Arakawa, O. Photoregulation of Anthocyanin Synthesis in Apple Fruit under UV-B and Red Light. Plant Cell Physiol. 1988, 29, 1385–1389. [Google Scholar]
- Saure, M.C. External Control of Anthocyanin Formation in Apple. Sci. Hortic. 1990, 42, 181–218. [Google Scholar] [CrossRef]
- Marais, E.; Jacobs, G.; Holcroft, D.M. Colour Response of ‘Cripps’ Pink’ Apples to Postharvest Irradiation Is Influenced by Maturity and Temperature. Sci. Hortic. 2001, 90, 31–41. [Google Scholar] [CrossRef]
- Gouws, A.; Steyn, W.J. The Effect of Temperature, Region and Season on Red Colour Development in Apple Peel under Constant Irradiance. Sci. Hortic. 2014, 173, 79–85. [Google Scholar] [CrossRef]
- Gonzalez, L.; Torres, E.; Àvila, G.; Carbó, J.; Bonany, J.; Alegre, S.; Asin, L. Effect of Thinning with Metamitron, Naa, BA and Naphthenic Acids on Apple (Malus Domestica) Trees. Plant Growth Regul. 2023. [Google Scholar] [CrossRef]
- Serra, S.; Leisso, R.; Giordani, L.; Kalcsits, L.; Musacchi, S. Crop Load Influences Fruit Quality, Nutritional Balance, and Return Bloom in ‘Honeycrisp’ Apple. HortScience 2016, 51, 236–244. [Google Scholar] [CrossRef]
- Ritenour, M.; Khemira, H. Red Color Development of Apple: A Literature Review. Washington State University—Tree Fruit Research and Extension Center. Postharvest Information Network. Printed 1997, Reprinted 2007. Available online: https://web.archive.org/web/20181221101058/http://postharvest.tfrec.wsu.edu:80/REP2007A.pdf (accessed on 13 April 2023).
- Weber, S.; Damerow, L.; Kunz, A.; Blanke, M. Anthocyanin Synthesis and Light Utilisation Can Be Enhanced by Reflective Mulch—Visualisation of Light Penetration into a Tree Canopy. J. Plant Physiol. 2019, 233, 52–57. [Google Scholar] [CrossRef] [PubMed]
- Mupambi, G.; Valverdi, N.A.; Camargo-Alvarez, H.; Reid, M.; Kalcsits, L.; Schmidt, T.; Castillo, F.; Toye, J. Reflective Groundcover Improves Fruit Skin Color in ‘Honeycrisp’ Apples Grown under Protective Netting. Horttechnology 2021, 31, 607–614. [Google Scholar] [CrossRef]
- Lugaresi, A.; Steffens, C.A.; Souza, M.P.; Amarante, C.V.; Brighenti, A.F.; Pasa, M.D.; Martin, M.S. Late Summer Pruning Improves the Quality and Increases the Content of Functional Compounds in Fuji Apples. Bragantia 2022, 81, 3122–3132. [Google Scholar] [CrossRef]
- Gonzalez, L.; Robinson, T.L. Effect of Different Reflective Ground Covers on the Coloring of Apples at Harvest. In Fruit Quarterly; NYS Horticultural Society: Geneva, NY, USA, 2021; pp. 15–18. [Google Scholar]
- Meinhold, T.; Damerow, L.; Blanke, M. Reflective Materials under Hailnet Improve Orchard Light Utilisation, Fruit Quality and Particularly Fruit Colouration. Sci. Hortic. 2011, 127, 447–451. [Google Scholar] [CrossRef]
- Andergassen, C.; Pichler, D. Pneumatic Defoliation of Apple Trees for Colour Improvement. Acta Hortic. 2022, 1346, 391–398. [Google Scholar] [CrossRef]
- Matsumoto, K.; Fujita, T.; Sato, S.; Chun, J.P. Comparison of the Effects of Early and Conventional Defoliation on Fruit Growth, Quality and Skin Color Development in ‘Fuji’ Apples. Hortic. Sci. Technol. 2017, 35, 410–417. [Google Scholar]
- Lee, C.H.; Seo, S.H.; Kwon, O.J.; Park, M.; Kim, W.C.; Kang, S.J. Functional Characterization of a Chemical Defoliant That Activates Fruit Cluster Leaf Defoliation in ‘Fuji’ Apple Trees. Appl. Biol. Chem. 2016, 59, 711–720. [Google Scholar] [CrossRef]
- Fanyuk, M.; Kumar Patel, M.; Ovadia, R.; Maurer, D.; Feygenberg, O.; Oren-Shamir, M.; Alkan, N. Preharvest Application of Phenylalanine Induces Red Color in Mango and Apple Fruit’s Skin. Antioxidants 2022, 11, 491. [Google Scholar] [CrossRef]
- Gonzalez, L.; Torres, E.; Àvila, G.; Bonany, J.; Alegre, S.; Carbó, J.; Martín, B.; Recasens, I.; Asin, L. Evaluation of Chemical Fruit Thinning Efficiency Using Brevis® (Metamitron) on Apple Trees (‘Gala’) under Spanish Conditions. Sci. Hortic. 2020, 261, 109003. [Google Scholar] [CrossRef]
- Hehnen, D.; Hanrahan, I.; Lewis, K.; McFerson, J.; Blanke, M. Mechanical Flower Thinning Improves Fruit Quality of Apples and Promotes Consistent Bearing. Sci. Hortic. 2012, 134, 241–244. [Google Scholar] [CrossRef]
- Bhusal, N.; Han, S.G.; Yoon, T.M. Summer Pruning and Reflective Film Enhance Fruit Quality in Excessively Tall Spindle Apple Trees. Hortic. Environ. Biotechnol. 2017, 58, 560–567. [Google Scholar] [CrossRef]
- Djordjeviä†, B.; Dejan, D.; Gordan, Z. Effects of Different Times of Summer Pruning to Productivity and Fruits Quality of Apple Cultivar Gala Brookfield. Ann. Univ. Craiova 2019, 49, 26–31. [Google Scholar]
- Morgan, D.C.; Stanley, C.J.; Volz, R.; Warrington, I.J. Summer Pruning of ‘Gala’ Apple: The Relationships between Pruning Time, Radiation Penetration, and Fruit Quality. J. Am. Soc. Hortic. Sci. 1984, 109, 637–642. [Google Scholar] [CrossRef]
- Aly, M.; El-Megeed, N.A.; Awad, R.M. Reflective Particle Films Affected on, Sunburn, Yield, Mineral Composition and Fruit Maturity of “Anna” Apple (Malus Domestica) Trees. Res. J. Agric. Sci. 2010, 6, 84–92. [Google Scholar]
- Glenn, D.M.; Prado, E.; Erez, A.; McFerson, J.; Puterka, G.J.A. Reflective, Processed-Kaolin Particle Film Affects Fruit Temperature, Radiation Reflection, and Solar Injury in Apple. J. Am. Soc. Hortic. Sci. 2002, 127, 188–193. [Google Scholar] [CrossRef]
- Sarooghinia, F.; Khadivi, A.; Abbasifar, A.; Khaleghi, A. Foliar Application of Kaolin to Reduce Sunburn in ‘Red Delicious’ Apple. Erwerbs-Obstbau 2019, 62, 83–87. [Google Scholar] [CrossRef]
- Schupp, J.R.; Fallahi, E.; Chun, I.J. Effect of Particle Film on Fruit Sunburn, Maturity and Quality of ‘Fuji’ and ‘Honeycrisp’ Apples. Horttechnology 2002, 12, 87–90. [Google Scholar] [CrossRef]
- Kalcsits, L.; Asteggiano, L.; Schmidt, T.; Musacchi, S.; Serra, S.; Layne, D.R.; Mupambi, G. Shade Netting Reduces Sunburn Damage and Soil Moisture Depletion in ‘Granny Smith’ Apples. Acta Hortic. 2018, 1228, 85–90. [Google Scholar] [CrossRef]
- IPCC. Climate Change 2007: Impacts, Adaptation and Vulnerability. Contribution of Working Group II to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change; Parry, M.L., Canziani, O.F., Palutikof, J.P., van der Linden, P.J., Hanson, C.E., Eds.; Cambridge University Press: Cambridge, UK, 2007; 976p. [Google Scholar]
- Mote, P.; Foster, J.G.; Daley-Laursen, S.B. Data mining to predict climate hotspots: An experiment in aligning federal climate enterprises in the Northwest. AGU Fall Meet. Abstr. 2014, 2014, PA41A-4024. [Google Scholar]
- Fujisawa, M.; Kobayashi, K. Climate Change Adaptation Practices of Apple Growers in Nagano, Japan. Mitig. Adapt. Strateg. Glob. Chang. 2011, 16, 865–877. [Google Scholar] [CrossRef]
- Lötze, E.; Daiber, S.H.; Midgley, S.J. Boron in combination with calcium reduces sunburn in apple fruit. J. Boron. 2017, 2, 123–127. [Google Scholar]
- Webb, L.; Darbyshire, R.; Erwin, T.; Goodwin, I. A Robust Impact Assessment That Informs Actionable Climate Change Adaptation: Future Sunburn Browning Risk in Apple. Int. J. Biometeorol. 2016, 61, 891–901. [Google Scholar] [CrossRef] [PubMed]
- Rajagopalan, K.; Chinnayakanahalli, K.J.; Stockle, C.O.; Nelson, R.L.; Kruger, C.E.; Brady, M.P.; Malek, K.; Dinesh, S.T.; Barber, M.E.; Hamlet, A.F.; et al. Impacts of near-Term Climate Change on Irrigation Demands and Crop Yields in the Columbia River Basin. Water Resour. Res. 2018, 54, 2152–2182. [Google Scholar] [CrossRef]
- Gallinat, A.S.; Primack, R.B.; Wagner, D.L. Erratum: Autumn, the Neglected Season in Climate Change Research. Trends Ecol. Evol. 2015, 30, 364. [Google Scholar] [CrossRef]
- Creasy, L.L. The role of low temperature in anthocyanin synthesis in ‘McIntosh’ apple. Proc. Am. Soc. Hortic. Sci. 1968, 93, 716–724. [Google Scholar]

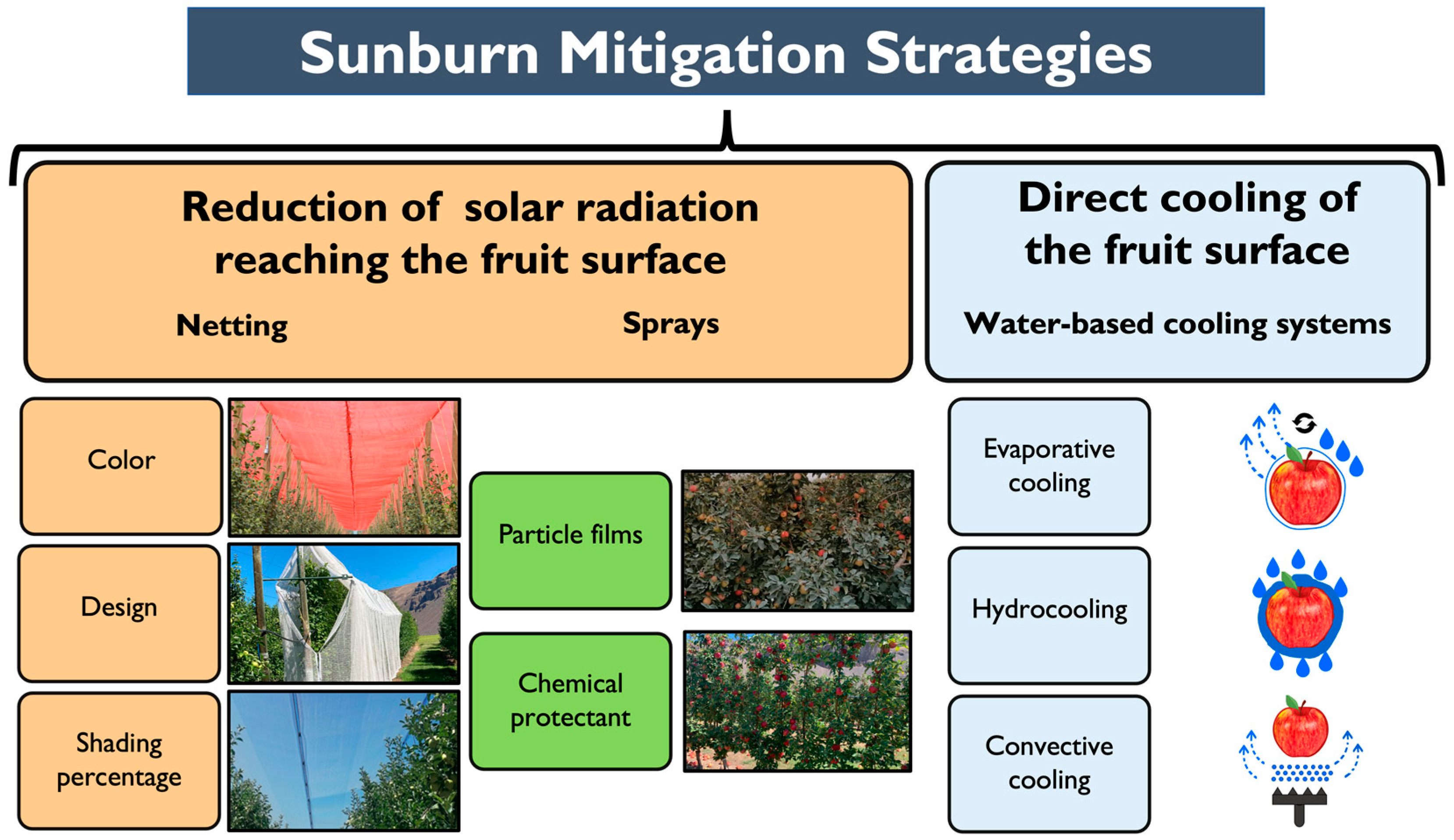
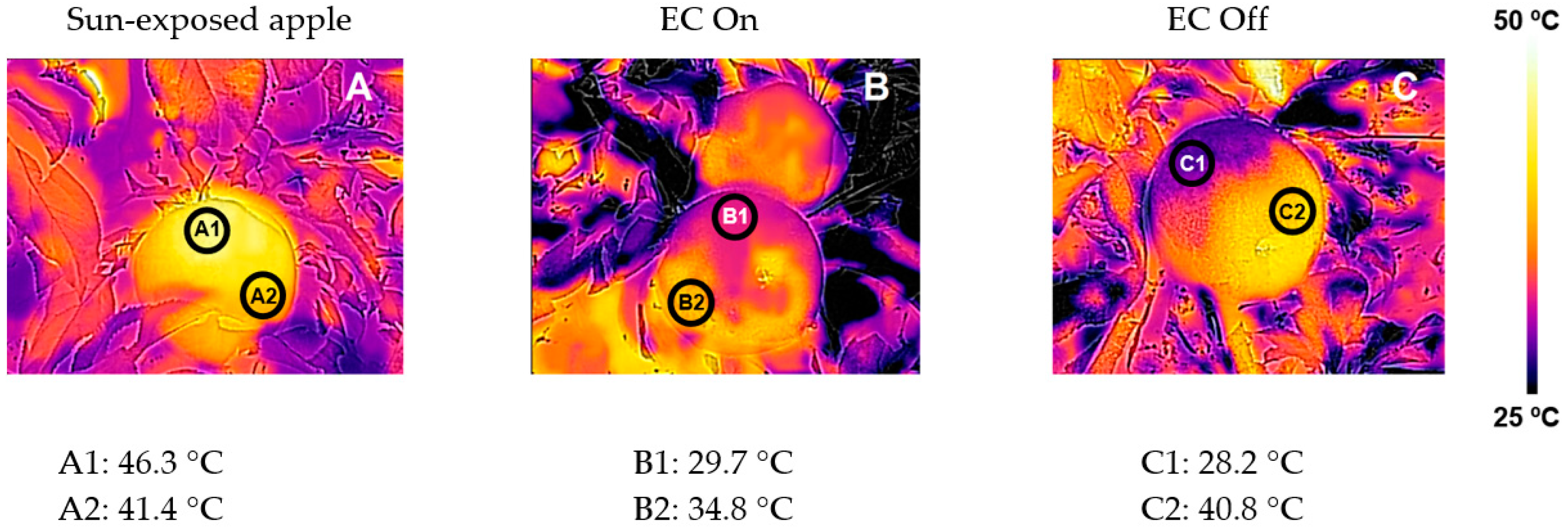
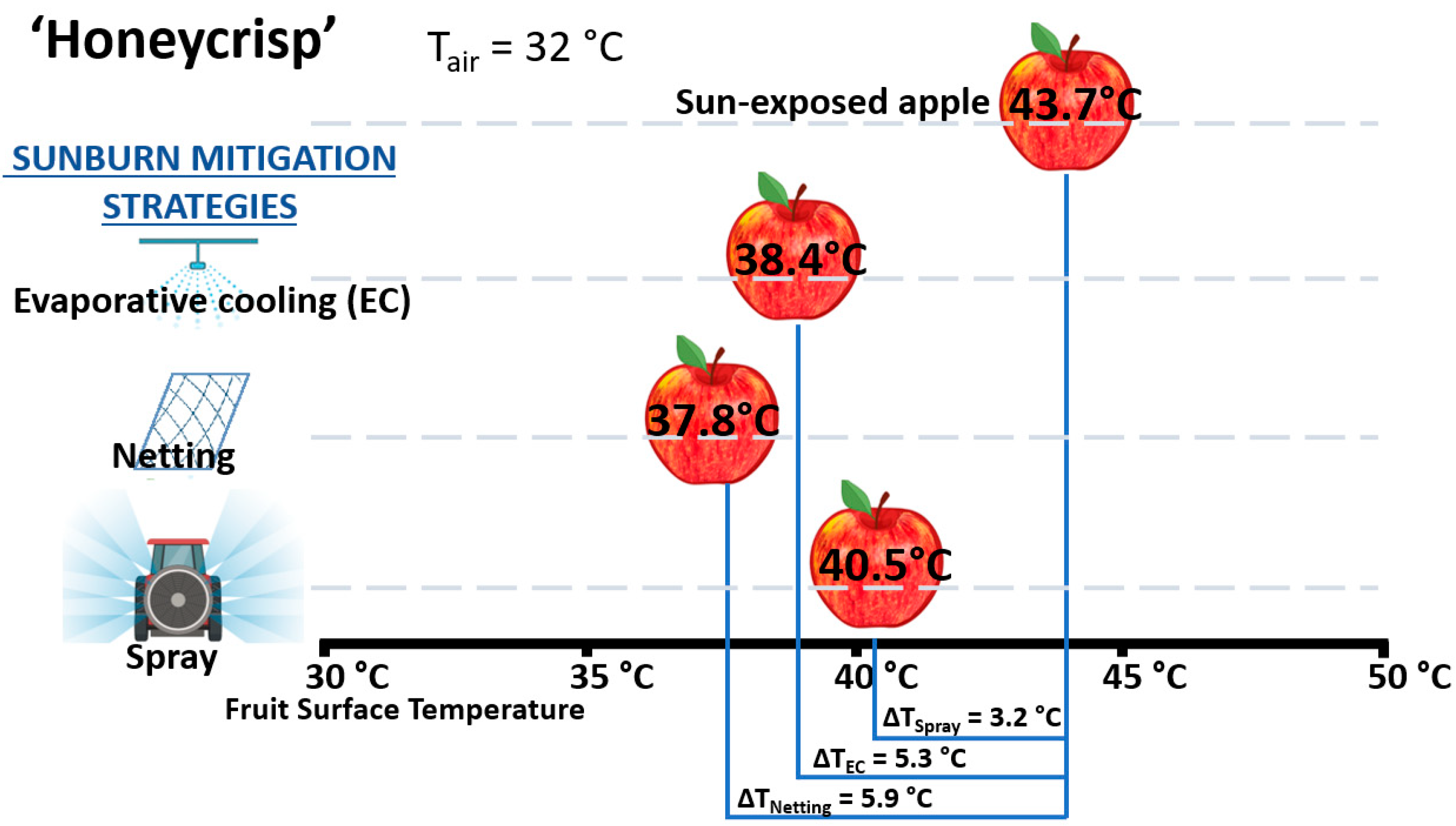
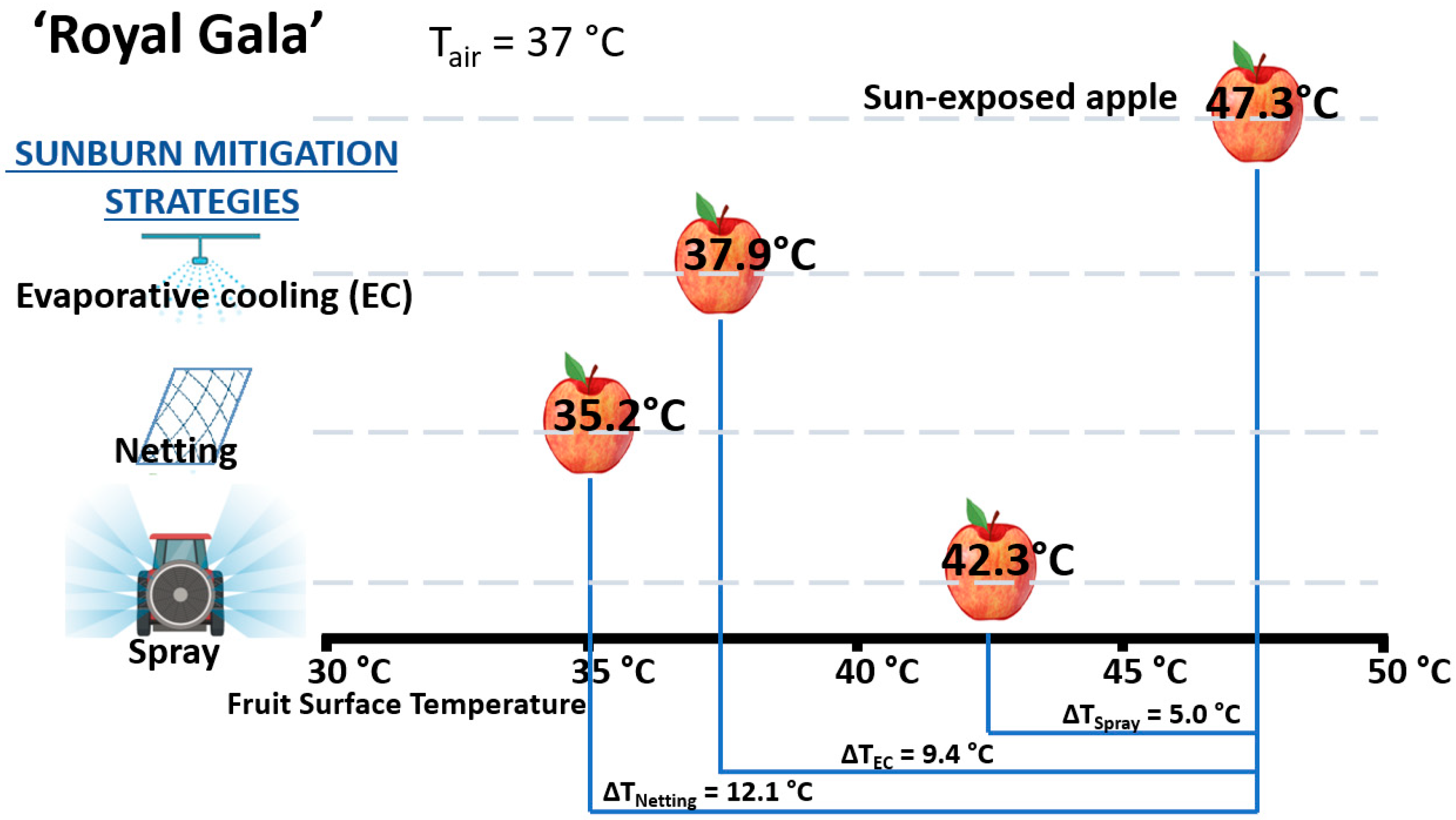
| Agronomic Strategy | Agronomic Effect | Impact on Red Color | Impact on Sunburn | Reference(s) |
|---|---|---|---|---|
| Reflective material | Red color | Increase | Increase/no change | [88,90,91] |
| Deleafing | Red color | Increase | Decrease/no change | [92,93,94] |
| Phenylalanine spray | Red color | Increase | No change | [95] |
| Fruit thinning | Red color | Increase | Decrease/no change | [83,96,97] |
| Summer pruning | Red color | Increase | Increase/no change | [89,98,99,100] |
| Protective spray | Sunburn | Decrease/no change | Decrease | [101,102,103,104] |
| Evaporative cooling | Sunburn | Increase | Decrease | [25,48,58,63] |
| Netting | Sunburn | Decrease/no change | Decrease | [55,56,66,67,68,69,88,105] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Willsea, N.; Blanco, V.; Rajagopalan, K.; Campbell, T.; Howe, O.; Kalcsits, L. Reviewing the Tradeoffs between Sunburn Mitigation and Red Color Development in Apple under a Changing Climate. Horticulturae 2023, 9, 492. https://doi.org/10.3390/horticulturae9040492
Willsea N, Blanco V, Rajagopalan K, Campbell T, Howe O, Kalcsits L. Reviewing the Tradeoffs between Sunburn Mitigation and Red Color Development in Apple under a Changing Climate. Horticulturae. 2023; 9(4):492. https://doi.org/10.3390/horticulturae9040492
Chicago/Turabian StyleWillsea, Noah, Victor Blanco, Kirti Rajagopalan, Thiago Campbell, Orlando Howe, and Lee Kalcsits. 2023. "Reviewing the Tradeoffs between Sunburn Mitigation and Red Color Development in Apple under a Changing Climate" Horticulturae 9, no. 4: 492. https://doi.org/10.3390/horticulturae9040492
APA StyleWillsea, N., Blanco, V., Rajagopalan, K., Campbell, T., Howe, O., & Kalcsits, L. (2023). Reviewing the Tradeoffs between Sunburn Mitigation and Red Color Development in Apple under a Changing Climate. Horticulturae, 9(4), 492. https://doi.org/10.3390/horticulturae9040492







