1. Introduction
Frankincense essential oil (FEO) is produced from trees of the genus
Boswellia that are native to the Arabian Peninsula, northeast Africa, and India. The
Boswellia genus contains about 20 different species [
1]. Popular types include
B. carterii,
B. sacra,
B. serrata, or
B. neglecta which occur in different locations and show different compositions of oils [
2]. Some authors claim
B. carterii and
B. sacra to be the same species, but differences in chemical composition and the ratio of some enantiomers between these plants have been observed [
3].
Most of the FEOs are obtained by steam distillation of an oleo gum resin of
Boswellia species. They contain volatile compounds that cause a characteristic woody and spicy aroma [
4]. FEO often contains components such as α-pinene, α-thujene, β-pinene, limonene,
p-cymene, myrcene, or sabinene [
4,
5,
6]. The resin has a protective role in plants; thus, the essential oil also manifests many positive activities. Its good antimicrobial and anti-insect activity protects the tree from [
2]. FEO can inhibit the growth of human pathogens such as
S. aureus or
P. aeruginosa [
7,
8]. FEO has good antimicrobial properties against bacterial (
P. acnes) and fungal (
C. albicans,
Malassezia spp., and
Trichophyton spp.) pathogens that cause skin-associated infections [
9]. Moreover, FEO prevents the formation of biofilms by
Staphylococcus and
Candida microorganisms [
10]. The antiparasitic activity of FEO against
Leishmania was also reported [
11].
Frankincense has been used in traditional medicine due to its positive medicinal properties [
12]. The resin from
Boswellia has a positive impact on patients with diseases of the gastrointestinal tract [
13]. Due to the specific aroma, FEO has stress-relieving properties [
14]. FEO also shows potential for beneficial effects in neurodegenerative disorders [
15]. Moreover, the anticancer activity of FEO towards pancreatic cancer cells was reported in in vitro studies [
16]. FEO was also found to induce tumor cell-specific apoptosis in the cell lines of bladder [
17] and breast cancer [
18].
Nowadays, an increasing trend to substitute artificial substances with natural alternatives is observed. Due to the overuse of antibiotics, some microorganisms have become more resistant. Research has thus become more focused on the search for bioactive compounds that inhibit pathogenic microorganisms in medicine or the food industry. Moreover, the extended use of synthetic pesticides has led to increased concerns related to the condition of the environment and human health. This has promoted research on the anti-insect properties of natural substances.
According to previous research, FEOs have antimicrobial and anti-insect potential. On the other hand, there is not enough research that would provide information about the use of FEO as an antimicrobial agent in food preservation against pathogenic microorganisms or insects. These potential positive properties of FEO have resulted in its selection for analysis from among the plethora of readily available essential oils.
The aim of this study was to evaluate the positive properties of FEO. The antioxidant potential of this substance was determined. Antimicrobial and antibiofilm activities were tested in order to assess the potential of this oil in inhibiting pathogenic bacteria which are common risk factors in the food industry. Moreover, anti-insect activity against Oxycarenus lavaterae was tested and the chemical composition of volatile compounds of FEO was determined.
2. Materials and Methods
2.1. Essential Oil
Frankincense essential oil (FEO) was purchased from Slovak company Hanus s.r.o. The provider stated that essential oil was extracted by steam distillation from Boswellia carterii resin obtained from Somalia. The essential oil was stored in the dark at 4 °C during the analyses.
2.2. Gas Chromatography–Mass Spectrometry and Gas Chromatography Analyses
The identification of volatile compounds in the FEO sample was performed using an Agilent Technologies (Palo Alto, Santa Clara, CA, USA) 6890 N gas chromatograph equipped with a quadrupole mass spectrometer 5975 B (Agilent Technologies, Santa Clara, CA, USA). The Agilent Technologies gas chromatograph was operated by an interfaced HP Enhanced ChemStation software (Agilent Technologies, Santa Clara, CA, USA). Using an HP-5MS (30 m × 0.25 mm × 0.25 µm) capillary column separation of volatiles has been achieved. The essential oil was diluted in hexane (10% solution) prior to analysis and injected in a volume of 1 µL. With the flow rate of 1 mL/min as a carrier gas helium 5.0 was used. The temperature of the split/splitless injector was set at 280 °C, while the temperature of the MS source and MS quadruple were set at 230 °C and 150 °C, respectively. The mass scan was in the range of 35–550 amu at 70 eV. The solvent delay time was 3.00 min. The temperature program of GC and GC-MS analysis was as follows: temperature program of 60 °C to 150 °C (rate of increase 3 °C/min), and 150 °C to 280 °C (rate of increase 5 °C/min), held 4 min at 280 °C. The total run time was 60 min. The split ratio was 40.8:1.
The identification of volatile constituents was performed by the comparison of their retention indices (RI) as well as the reference spectra reported in the literature and the ones stored in the MS library (Wiley7Nist) [
19,
20]. Using the data collected with GC-FID equipped with the same HP-5MS capillary column, semiquantification of the components was performed. Only compounds in amounts higher than 0.1% were taken into consideration.
2.3. Antioxidant Activity
2.3.1. DPPH Assay
The antioxidant potential of FEO was determined using the 2,2-diphenyl-1-picrylhydrazyl (DPPH) method. An aliquot of 190 μL of DPPH radical solution in methanol (prepared so absorbance is adjusted at 0.8 at 515 nm) was mixed with 10 μL FEO in a 96-well microtiter plate. The reaction mixture was incubated at room temperature for 30 min in darkness by continuous shaking at 1000 rpm, after which the absorbance of the sample was measured spectrophotometrically at 517 nm. All measurements were performed in triplicate. Methanol was used as the control solution, whereas Trolox (1–5 mg/L in methanol) was used as the reference compound. Antioxidant activity was expressed as a percentage of DPPH
• inhibition. The calculation was performed using the equation:
where A0 was the absorbance of DPPH
• and AA was the absorbance of the sample.
The total radical scavenging capacity was expressed according to the calibration curve of Trolox (TEAC). The results were presented as mean values ± standard deviation (SD) of three independent measurements.
2.3.2. ABTS Assay
ABTS [2,20-azinobis(3-ethylbenzothiazoline-6-sulfonic acid) diammonium] radical cation was generated according to an already described procedure [
21]. The prepared radical cation was diluted prior to the analysis up to an absorbance value of 0.7 at 744 nm. An aliquot of 190 μL of this solution was mixed with 10 μL of FEO (in a 96-well microtiter plate). This was followed by a 30 min incubation at room temperature, in darkness, and with shaking at 1000 rpm. A decrease in absorbance at 744 nm was registered and the results are presented as a percentage of ABTS inhibition using the previous Equation (1). All measurements were performed in triplicate. Methanol was used as the blank sample, and Trolox was the standard reference substance. The results were expressed as the percent of inhibition as well as an equivalent of Trolox (TEAC) calculated based on the standard curve. The results were presented as mean values ± standard deviation (SD) of three independent measurements.
2.4. Antimicrobial Activity
The antimicrobial activity of FEO was tested against the following microorganisms Pseudomonas aeruginosa CCM 3955, Yersinia enterocolitica CCM 7204, Salmonella enterica subsp. enterica ser. Enteritidis CCM 4420, Bacillus subtilis CCM 1999, Staphylococcus aureus subsp. aureus CCM 2461, Enterococcus faecalis CCM 4224, Candida krusei CCM 8271, Candida albicans CCM 8261, Candida tropicalis CCM 8223, Candida glabrata CCM 8270 purchased from the Czech Collection of Microorganisms (Brno, Czech Republic).
Three microscopic filamentous fungi (Aspergillus flavus, Botrytis cinerea, and Penicillium citrinum) previously obtained from grapes and identified with MALDI-TOF MS Biotyper and 16S rRNA sequencing were used for the analyses of antifungal activity. A biofilm-forming S. enterica was obtained and cultivated from chicken meat, analyzed by 16S rRNA sequencing, and identified using MALDI-TOF MS with a score higher than 2.0.
The inocula of bacteria and microscopic filamentous fungi were incubated for 24 h. Bacterial samples were incubated in Mueller Hinton Broth (MHB, Oxoid, Basingstoke, UK) at 37 °C and microscopic filamentous fungi samples were incubated in Sabouraud dextrose broth (SDB, Oxoid, Basingstoke, UK) at 25 °C.
2.4.1. Disk Diffusion Method
The disc diffusion method was used for the analyses of the antimicrobial activity of FEO. The inoculum was diluted to 0.5 McFarland (1.5 × 108 CFU/mL) and 100 μL of it was spread onto a Petri dish with Mueller Hinton agar (MHA, Oxoid, Basingstoke, UK) for bacteria and Sabouraud dextrose agar (SDA, Oxoid, Basingstoke, UK) for microscopic filamentous fungi. Discs with a 6 mm diameter were placed in a Petri dish and 10 μL of non-diluted FEO was added. Petri dishes were incubated for 24 h at 37 °C (bacteria) or 25 °C (microscopic filamentous fungi).
The antimicrobial activity was evaluated according to the Clinical and Laboratory Standards Institute—Performance Standards for Antimicrobial Susceptibility Testing [
22]. Antibiotics (Oxoid, Basingstoke, UK) were used as a control: cefoxitin for G
− bacteria, gentamicin for G
+ bacteria, and fluconazole for microscopic filamentous fungi [
23]. Analyses were performed in triplicate.
2.4.2. Minimum Inhibitory Concentration
The Minimum Inhibitory Concentration (MIC) was evaluated by the broth microdilution method. The inoculum was adjusted to 0.5 McFarland and an aliquot of 50 μL was added to a well in a 96-well plate. FEO was distributed into the wells by two-fold serial dilution to final concentration values from 500 μL/mL to 0.2 μL/mL. Samples were incubated for 24 h at 37 °C (bacteria) and 25 °C (yeasts). As a negative control, MHB with FEO was used and MHB with inoculum served as a positive control. Absorbance was measured at 570 nm in time 0 h and after 24 h. Analyses were prepared in triplicate [
23].
The minimal inhibition concentration of FEO against fungi was measured in four concentrations (500, 250, 125, and 62.5 μL/mL) of the oil diluted in 0.1% DMSO solution. The inoculum was diluted to 0.5 McFarland (1.5 × 108 CFU/mL). A total of 100 μL of inoculum was spread to SDA. Discs with 6 mm were placed on the Petri dish and 10 μL of the appropriate concentration of FEO was added. Petri dishes were incubated for 5 days at 25 °C.
2.4.3. In Situ Inhibition of Vapor Phase FEO on Carrot
An antimicrobial analysis in situ was performed on the carrot. A 0.5 mm slice of carrot was washed in distilled water, left to dry, and subsequently transferred to a Petri dish with MHA. Bacteria and microscopic filamentous fungi were diluted to 0.5 McFarland and the carrot was inoculated with three stabs. The FEO was diluted with ethyl acetate to final concentrations of 500, 250, 125, and 62.5 μL/L. A total of 100 µL of each diluted solution was added to filter paper and ethyl acetate was allowed to evaporate. A total of 100% ethyl acetate was used as the negative control. The samples were incubated for 7 days at 37 °C (bacteria) and 25 °C (microscopic filamentous fungi). The inhibitory activity was calculated using the stereological method. The bulk density was calculated according to the formula Vv = P/p × 100 (P = stereological lattice of the colonies, p = substrate). Growth inhibition was expressed as GI = [(C − T)/C] × 100 (C = growth density of control group, T = growth density in the group with FEO) [
24]. The activity was measured in triplicate.
2.4.4. Antibiofilm Activity
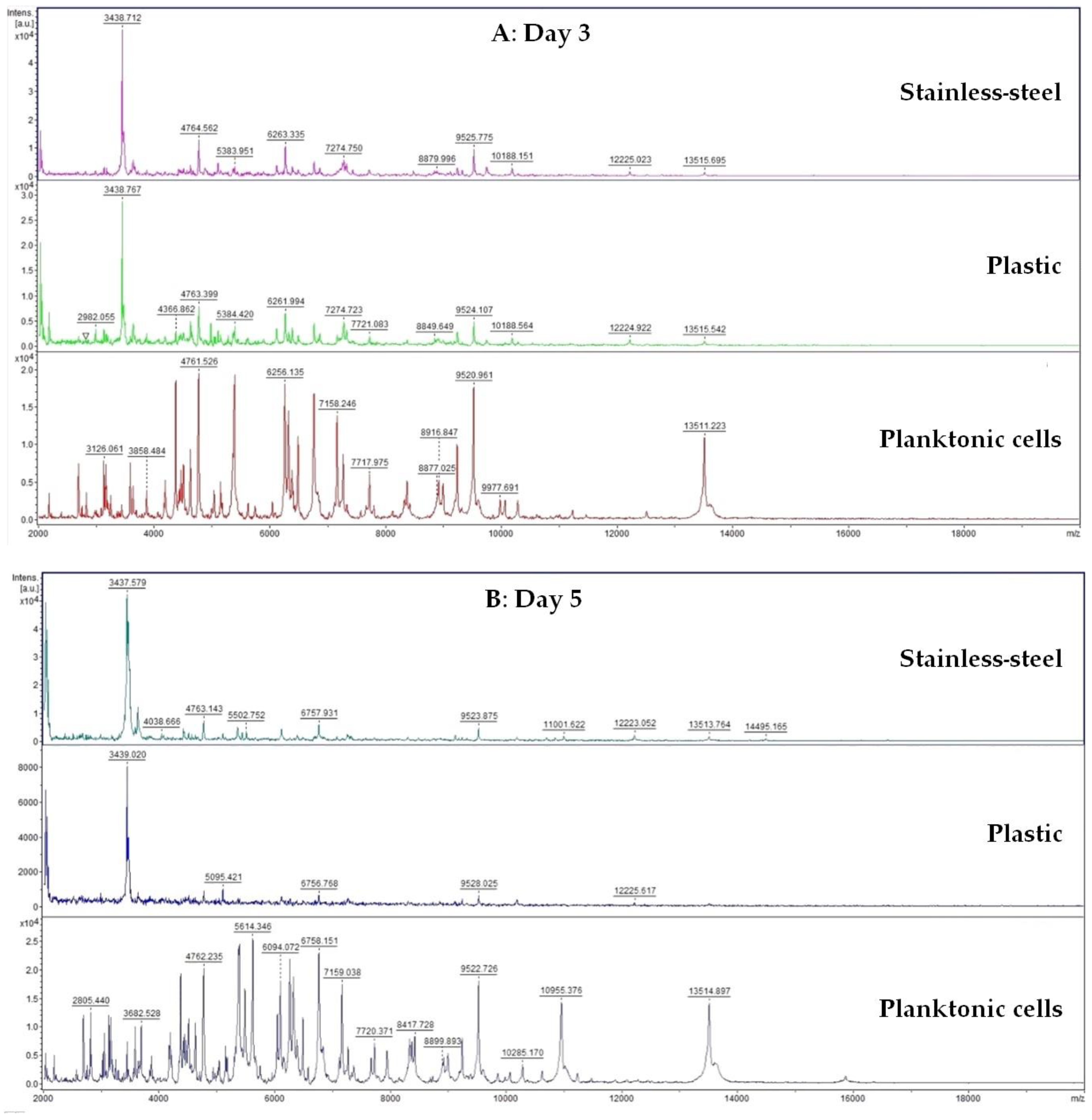
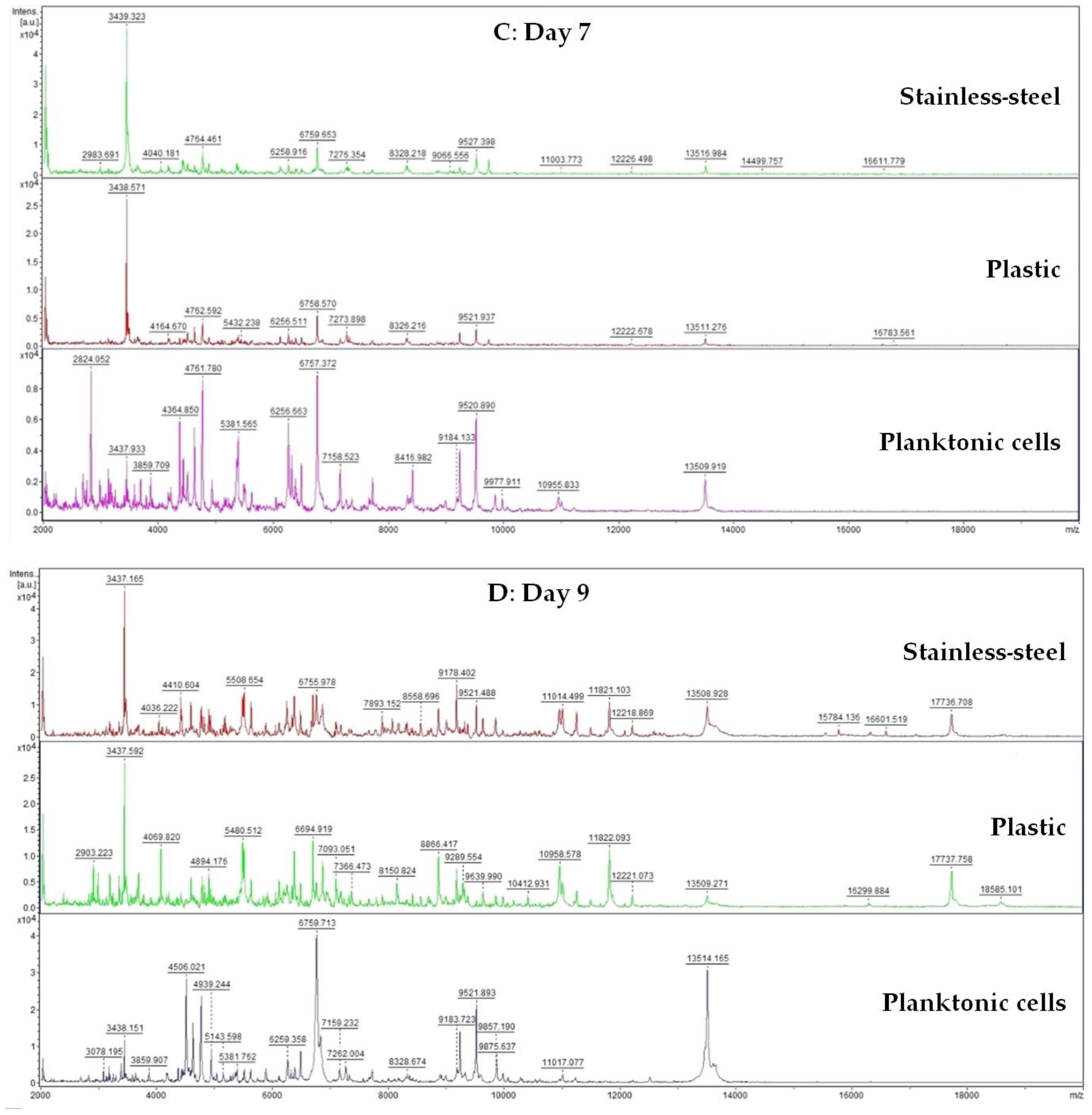
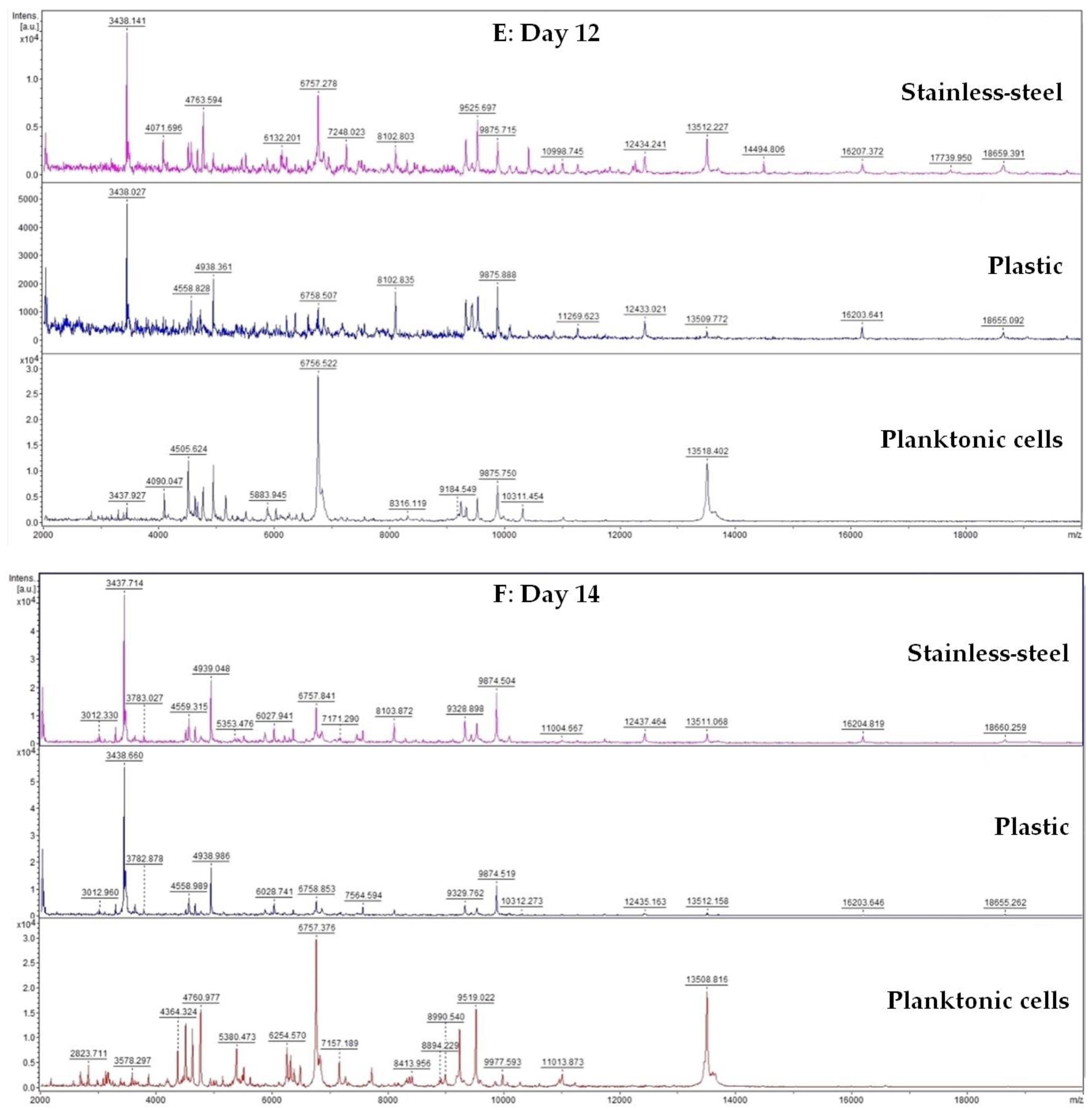
A 50 mL tube was filled with 20 mL of MHB and 100 μL of biofilm-forming S. enterica inoculum. Subsequently, a plastic and stainless-steel piece were added to the tube. FEO was added to experimental groups in the final concentration of 0.1%. Control samples were left untreated. The incubation was carried out at 37 °C with shaking at 170 rpm. Samples were analyzed on days 3, 5, 7, 9, 12, and 14.
Changes in protein spectra during biofilm development in the presence of FEO were monitored using MALDI-TOF MS Biotyper. Biofilm samples were taken from plastic and stainless-steel surfaces by a sterile cotton swab and were directly transferred to a MALDI-TOF plate. A 300 µL of culture medium was taken for analysis of planktonic cell spectra. A suspension with planktonic cells was centrifuged for 1 min at 12,000 rpm. The pellet was washed three times in ultrapure water and resuspended. A total of 1 μL was applied to a target plate.
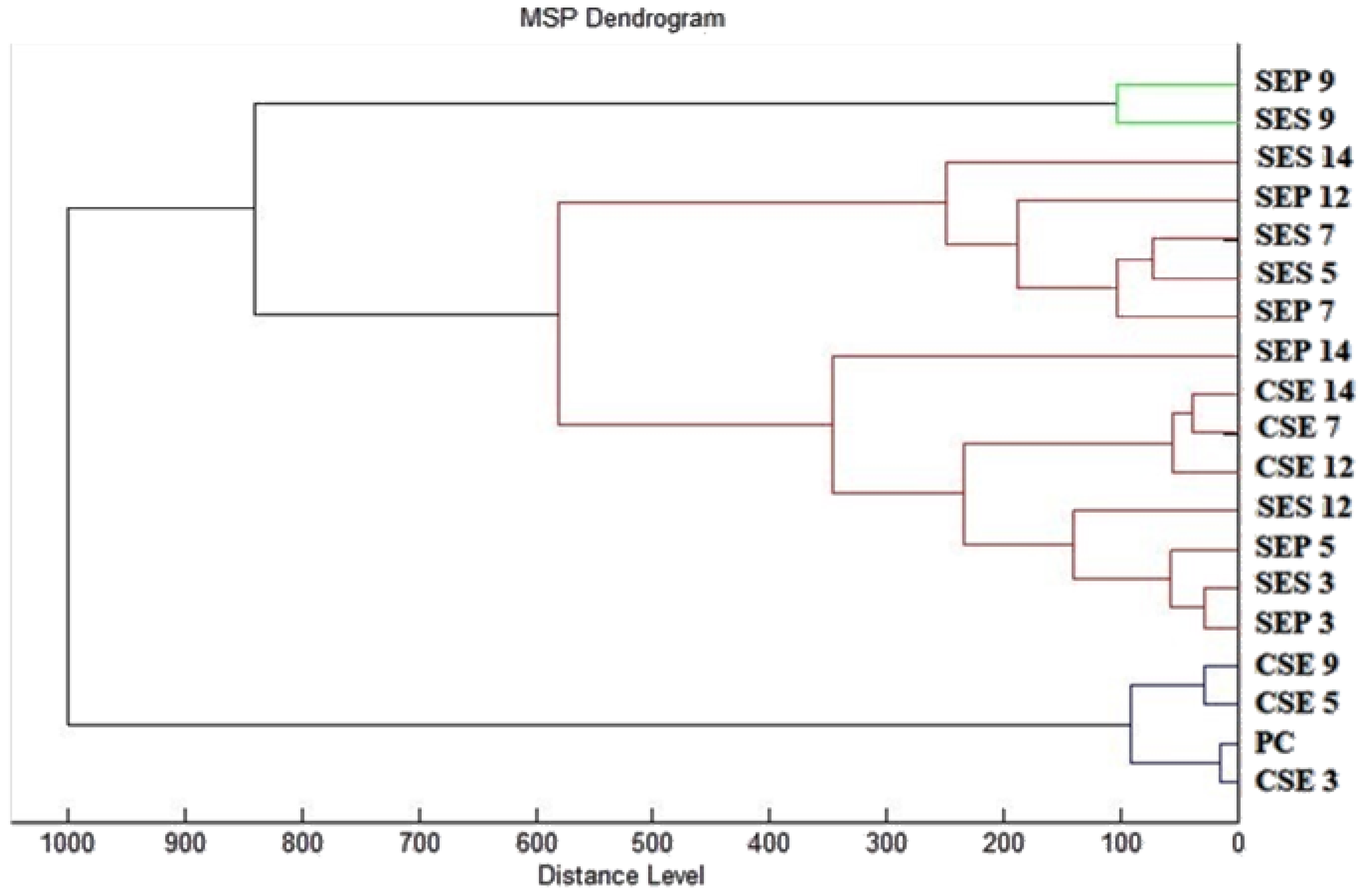
A matrix α-Cyano-4-hydroxycinnamic acid matrix (10 mg/mL) was applied to the dried target plate. MALDI-TOF MicroFlex (Bruker Daltonics) was used for the analysis of biofilm protein spectra. Spectra in the range of mass to charge ratio 200–2000 were recorded in the linear and positive mode. The protein spectra were obtained, and the similarities of the spectra were used to generate the standard global spectrum (MSP). Nineteen MSP were generated from the spectra with MALDI Biotyper 3.0 and were grouped into dendrograms using Euclidean distance [
25].
2.5. Insecticidal Activity of FEO Vapor Phase
Oxycarenus lavaterae was used for the analysis of insecticidal activity. Thirty specimens were added to Petri dishes with vented lids. Subsequently, 100 μL was added to filter paper which was placed inside the Petri. Petri plates were sealed with parafilm. Concentrations of 100%, 50%, 25%, 12.5%, 6.25%, and 3.125% of FEO were prepared in 0.1% polysorbate. A negative control sample was treated with 100 μL of 0.1% polysorbate. Specimens were exposed for 24 h at room temperature. Live and dead subjects were counted, and the percentage of insecticidal activity was calculated [
24]. The insecticidal activity was measured in triplicate.
2.6. Statistical Analysis
One-way analysis of variance (ANOVA) was performed with Prism 8.0.1 (GraphPad Software, San Diego, CA, USA) and by Tukey’s test at p < 0.05. SAS® version 8 software (SAS Institute, Cary, NC, USA) was used to process the data. MIC 50 and MIC 90 values (50% and 90% inhibition of microbial growth) were determined by logit analysis.
4. Discussion
The findings observed in the present study show that commercial FEO is characterized by a high abundance of monoterpene hydrocarbons (79.3% of the total), with α-pinene (37.0%) and α-limonene (19.8%) being the major compounds. These results are in agreement with a previously published study on the chemical composition of 21 samples of commercial FEO as well as two laboratory-distilled oils [
6]. Moreover, a report by DeCarlo et al. [
26] indicates at least three different chemotypes defined by a high abundance of α-pinene, α-thujene, and methoxydecan. According to the same report, as well as many others, the α-pinene chemotype is also rich in limonene [
4,
6,
26,
27,
28]. We can thus conclude that FEO investigated in this study belonged to the previously described α-pinene chemotype. Moreover, some previous examinations showed FEOs rich in octyl acetate, a compound not identified in the sample investigated in this study [
29,
30]. Observed differences in the chemical composition of this FEO could be explained by distinct environmental factors, such as harvesting season, geographical location, climate, as well as the part of the plant and the method used for FEO extraction, etc. Even though there are a number of previously published results on the biological activity of this species, the variations in chemical composition can seriously influence the observed results.
DPPH and ABTS assays are commonly used for the evaluation of the antioxidant potential of essential oils. Our study showed that 10 μL of FEO can neutralize 73.88 ± 0.35% of DPPH radical and 97.09 ± 0.48% of ABTS radical cation. Considering these results, we can imply a high potential of this FEO as an antioxidant. The observed differences in the results of the two performed assays can be attributed to the differences in the mechanisms involved in the reactions responsible for neutralization. It is well known that ABTS radical cation reacts much faster, including the electron transfer reaction, compared to DPPH radical, whose neutralization is dependent on the antioxidant’s ability to donate hydrogen. In previously published results, this FEO showed a weak to moderate antioxidant potential [
31,
32,
33]. Essential oils are known to be complex mixtures of compounds with different functional groups, and mainly the synergistic effect of two or more of its components is responsible for the observed effects. With that in mind, the diversity of the results obtained in various studies can be explained. Nevertheless, the results can also vary due to the assay employed in the determination of the antioxidant activity.
In our study, the antimicrobial activity of FEO was the strongest against
Candida species. Strong activity of FEO was also determined against G
+ and G
− bacteria. Abers et al. [
34] analyzed the antimicrobial activity by disc diffusion method and determined a moderate activity of FEO with inhibition zone diameters exceeding 30 mm against G
+ bacteria
S. pyogenes and
M. smegmatis and G
− bacteria
P. aeruginosa. Al-Saidi et al. [
35] determined the activity against G
− P. aeruginosa to result in diameters of 23.3–29.7 mm. Among G
+ bacteria, a comparable activity was determined for
B. subtilis and
S. aureus with ranges of inhibition zones 14.7–16.0 mm and 7.0–24.3 mm, respectively. Almutairi et al. [
36] observed the antimicrobial properties of FEO against antibiotic-resistant strains and showed mild activity against methicillin-resistant
S. aureus and multi-drug-resistant
P. aeruginosa.
Di Stefano et al. [
7] compared the growth-inhibition activity of three FEOs. They determined MIC value against bacteria
S. epidermidis,
S. hominis, and
P. acnes ranging from 0.264 to 6.16 mg/mL. MIC for
S. aureus and
P. aeruginosa ranged from 52 to 400 mg/mL, which appeared to reflect the weak activity. Notable activity was determined against
C. albicans and
M. furfur with a MIC range of 0.03–3.10 mg/mL. On the other hand, Man et al. [
37] tested six essential oils (Frankincense, myrtle, thyme, lemon, oregano, and lavender) and determined the weakest activity compared to the essential oil with MIC of 50%
v/
v or higher against G
+ and G
− bacteria. De Rapper et al. [
38] tested the activity of various
Boswellia species and found MIC values to be the lowest against fungi
C. neoformans (0.8 mg/mL) and the highest against
S. aureus (6.0 mg/mL). Van Vuuren et al. [
4] tested various FEOs and reported mean MIC values against G
+ bacteria,
S. aureus and
B. cereus, of 8.1 and 3.4 mg/mL, respectively. The reported MIC values against G
− bacteria,
E. coli and
P. vulgaris, were 6.2 and 4.0 mg /mL, respectively, and against the yeast
C. albicans, the value was 7.4 mg/mL. These values were lower compared to our study. Bogavac et al. [
39] determined the activity of FEO against a
C. albicans strain at 12.5 μL/mL which suggest a stronger activity compared to our study.
The activity of FEO together with other essential oils against fungal pathogens was evaluated as mild. Against
A. niger, an MIC value of 625 μg/mL was determined [
40]. Antifungal activity of FEO against
Aspergillus,
Penicillium,
Fusarium, or
Alternaria species was found at low concentrations, which are suitable for the inhibition of pathogenic fungi to preserve plant-based products [
32]. FEO showed promising antifungal properties against 15 different fungal species with a value of mycelial inhibition ranging from 15.9 to 56.3% at 1 µL/mL. MIC was determined in a range between 0.039 and 0.625 µL/mL [
41]. Ljaljević Grbić et al. [
42] determined MIC for
Aspergillus species in the range from 10.0 to 120.0 mg/mL and for
Penicillium species, in the range from 10 to 30 mg/mL. These results on the antifungal properties of FEO are in agreement with our study.
Many factors affect the activity of the FEOs. Generally, the changes in biological activity are a result of differences in chemical composition [
4]. Alpha-pinene, which was the most abundant molecule of our FEO, did not show any significant activity alone, but the antimicrobial activity was stronger when combined with other substances, which suggests synergism with other components in FEO [
43,
44]. On the contrary, limonene, the second most abundant compound, has shown better activity than the essential oil containing it [
45,
46]. Overall, no correlation was observed between the number of major constituents in essential oils [
47]. Therefore, the antimicrobial activity cannot be attributed only to the major constituents of essential oils, as minor compounds contribute to activity against potentially pathogenic microorganisms as well [
48].
FEO showed good inhibitory activity against G
+ and G
− bacteria on the carrot used as a model food. For microscopic filamentous fungi, determined activity was weaker compared to bacteria. Prakash et al. [
32] evaluated the potential of FEO for the preservation of
Piper nigrum L. fruits and reported that the growth of a storage mold,
A. flavus, was decreased by 65.38%. Not enough research has been focused on the application of FEO for the preservation of fruits or vegetables from potential pathogenic bacteria. On the other hand, some research has been conducted in our laboratories which aimed at the observation of the inhibition activity of essential oils against pathogenic microorganisms that grew on fruits, vegetables, and bread samples [
23,
49,
50]. In other reports the inhibitory activity of eucalyptus essential oils on sweet potatoes and bread [
51], or thyme oil against
Botrytis in grapes [
52] were investigated. Generally, essential oil components are considered to be good preservatives of food products [
53]. Various mechanisms of action have been described, including interactions of compounds with cell membranes, cell walls, intracellular proteins, enzymes, or nucleic acids [
54]. Some components of FEO have been described as sufficient inhibitors of pathogenic microorganisms. For example, α-pinene was used in the prolongation of the storage of wine [
55]. Limonene coating was found to decrease fungal growth on cucumbers [
56]. We can suggest that the chemical composition of FEO increases the potential for its use as a food preservative.
Bacteria in the form of biofilm have a better ability to survive compared to single-cell forms due to the ability to adhere and the quorum sensing mechanisms. Analyses of biofilm degradation showed the ability of FEO to inhibit growth and deteriorate the formation of biofilm. There has not been enough research on the changes in biofilm resulting from the addition of FEO. Moreover, this is the first time that biofilm degradation was analyzed on different surfaces. The antibiofilm activity of the essential oils isolated from
Boswellia against
Staphylococcus species and
Candida was reported, with FEO showing the ability to prevent the adhesion of biofilm [
10]. The properties of terpenes, which are found in essential oils, were described with regard to biofilm formation [
57]. Boudiba et al. [
58] and Soyocak et al. [
59] observed the disruption of biofilms by the inhibition of quorum sensing in bacteria after the treatment with essential oils with a similar composition.
The activity of FEO against
Oxycarenus lavaterae showed 50% mortality at the concentration of 25%. The anti-insect activity of FEO against female forms of the fly
Musca domestica was tested by Pavela et al. [
60] who rported the toxicity values of LD50 and LD90 of 72.5 and 269.3 μg/individual, respectively. The activity against the mosquito
C. quinquefasciatus larvae was found to be significant. Yang et al. [
61] observed insecticidal activity of various essential oils against the weevil
Sitophilus zeamais. Even the contact and fumigant activity of FEO was not significant, the repellent activity of FEO was moderate even after 24 h of incubation. Metayi et al. [
62] tested a nanoemulsion of FEO against the cotton bollworm
Earias insulana and found out that 80% mortality of larvae was reached at a concentration of 1800 μL. Due to the activity of FEO against pests, it can potentially serve as a repellent or pesticide. More research is necessary in order to determine the mechanisms of action.