Effect of Processing Methods on the Postharvest Quality of Cape Gooseberry (Physalis peruviana L.)
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Postharvest Treatments
- -
- Coating process (CP): This methodology was based on Muley and Singhal (2020) [17]. Firstly, the whey protein (WPC) was hydrated: 30 g of protein in 188.31 mL of distilled water. Then, the pH was adjusted to 7 by adding NaOH, and the solution was heated (55 °C, 10 min) with constant stirring. After, 1.59 g of sodium carbonate was added while maintaining the agitation and temperature, and 30 g of glycerol was also added. After cooling, the solution was stored under refrigeration (5 ± 1 °C) until its application. The fruit was submerged twice for 1 min, and then the samples were drained and placed in an oven for 10 min at 30 °C to ensure the elimination of the surface liquid.
- -
- Vacuum impregnation process (VIP): This methodology was based on Ciro (2012). An impregnation solution containing 1% calcium chloride (CaCl2) and 0.05% sodium carbonate (Na2CO3) was prepared over an isotonic aqueous base (sucrose 20 °Brix). The fruit was placed in a desiccator and the solution ratio was maintained at 1:5 to ensure adequate immersion. A vacuum pump was attached to the desiccator (Welch, Gardner Denver Thomas, Inc. Sheboygan, WI USA), and the system was subjected to a vacuum pressure of 17 KPa for 5 min. Subsequently, the atmospheric pressure was restored, and the fruit remained submerged for an additional amount of time (5 min) without stirring. Finally, the impregnated samples were taken out of the desiccator, drained, and placed in an oven for 10 min at 30 °C to ensure removal of the surface liquid.
- -
- Immersion process (IP): This methodology was based on Pérez-Martínez et al. (2021). The immersion of the fruit was carried out sequentially using technological coadjutants. First, the fruit was immersed in a recipient containing a solution with 500 ppm citric acid and 500 ppm ascorbic acid for 5 min. Then, they were taken out from the recipient and immersed in a solution containing Na2CO3 0.05 M for 5 min. Finally, they were taken out and submerged in a solution containing 1% CaCl2 for 2 min. The immersed samples were drained and placed in the oven for 10 min at 30 °C to ensure the elimination of the surface liquid.
2.3. Characterization of the Fresh and Treated Fruit
2.4. Determination of Vitamin C, Carotenoids, Total Phenolic Content, and Antioxidant Capacity
- -
- Vitamin C: Vitamin C was determined following the method used by Contreras-Calderón et al. (2011) [19] based on titration of the analyzed samples with a 2,6-dichlorophenolindophenol standard solution in an acid environment. The analyses were performed in triplicate, and the results are expressed as mg/100 g.
- -
- Carotenoids: Carotenoids were determined following the methodology described by Rodriguez-Amaya and Kimura (2004) [20] using the absorption coefficient for β-carotene in petroleum ether solvent (A = 25 92). Three grams of homogenized fruit was placed in a falcon tube, and 50 mL of cold acetone was added. Then, they were homogenized in a vortex, and filtered in a Buchner funnel with filter paper. The samples were washed with small amounts of acetone until colorless. Subsequently, in a decanting funnel of 500 mL, 40 mL of petroleum ether and the acetone extract (obtained previously) was added. Distilled water (~300 mL) was slowly added to avoid the formation of an emulsion. Three or four washes were carried out to eliminate any residual acetone. Finally, the petroleum ether was collected in a 50 mL volumetric flask by passing the solution through a funnel containing anhydrous sodium sulfate to remove residual water. The separating funnel was washed with petroleum ether, collecting the washings in the volumetric balloon, gauged with petroleum ether, and the absorbance was read at 450 nm CARY 50 BIO, UV–vis (Varian Pty. Ltd., Mulgrave, Australia). The analyses were performed in triplicate, and the results are expressed as μg/100 g.
- -
- Total Phenolic Content (TPC): TPC was determined based on the method of Folin–Ciocalteu, following the steps used by Osorio-Arias et al. (2019) [21]. Firstly, the fruit was dissolved in a methanol/water (50:50) solution and an acetone-water solution (70:30) to make the extracts. Then 20 mL of extract was mixed with 1580 mL of distilled water and 100 mL of Folin–Ciocalteu reagent; after 2 min 300 mL of sodium carbonate (20 g/100 mL) was added and the final mixture was stored in the dark for 1 h. The absorbance of the solution was measured in a spectrophotometer (CARY 50 BIO, UV–vis) at 725 nm and the absorbance was compared with a calibration curve based on gallic acid. The analyses were performed in triplicate and the results were expressed as mg equivalents of gallic acid per 100 g of fresh sample (mg GAE/100 g).
- -
- Antioxidant Capacity: Two different methods were used to evaluate the antioxidant capacity of the fresh fruit. The FRAP (ferric-reducing ability of plasma) method is based on the single-electron transfer (SET) mechanism, while the ABTS (2,2′-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid)) method acts via both the SET and hydrogen atom transfer (HAT) reactions. The FRAP assay was conducted according to the methodology described by Duarte-Correa et al. (2020) [15]. For the first method, 900 μL of FRAP reagent (containing TPTZ (2,4,6-Tris(2-pyridyl)-s-triazine), FeCl3, and acetate buffer) was mixed with 90 μL of distilled water and 30 μL of the test sample and incubated at 37 °C/30 min. The absorbance was measured at 595 nm after 30 min (UV–3300 Mapada Instruments, Shanghai, China). Trolox was used for the calibration curve. The results are expressed as micromol of Trolox equivalents (TE) per gram (μmol TE/g). Regarding the second method, the ABTS assay was performed according to Osorio-Arias et al. (2019). Firstly, the radical cation solution (ABTS+) was prepared by mixing 10 mL of an ABTS stock solution (7 mM) with 10 mL of potassium persulfate (2.45 mM). The solution was let to stand overnight and, subsequently, diluted with ethanol until an absorbance reading of 0.7 was achieved (730 nm). Finally, the absorbance of the samples was determined after 30 min of mixing at 30 °C with the radical solution. The reduction in the absorbance was correlated with a Trolox calibration curve. The results are expressed as μM of Trolox equivalent (TE) per gram (μmol TE/g).
2.5. Textural Analysis of Fresh and Treated Fruit
2.6. Determination of the Weight Loss of Fresh and Treated Fruit
2.7. Sensory Analysis for Fresh and Treated Fruit
2.8. Statistical Analysis
3. Results and Discussion
3.1. Characterization of Fresh Fruit
3.2. Postharvest Treatments
3.2.1. Physicochemical and Texture Characteristics
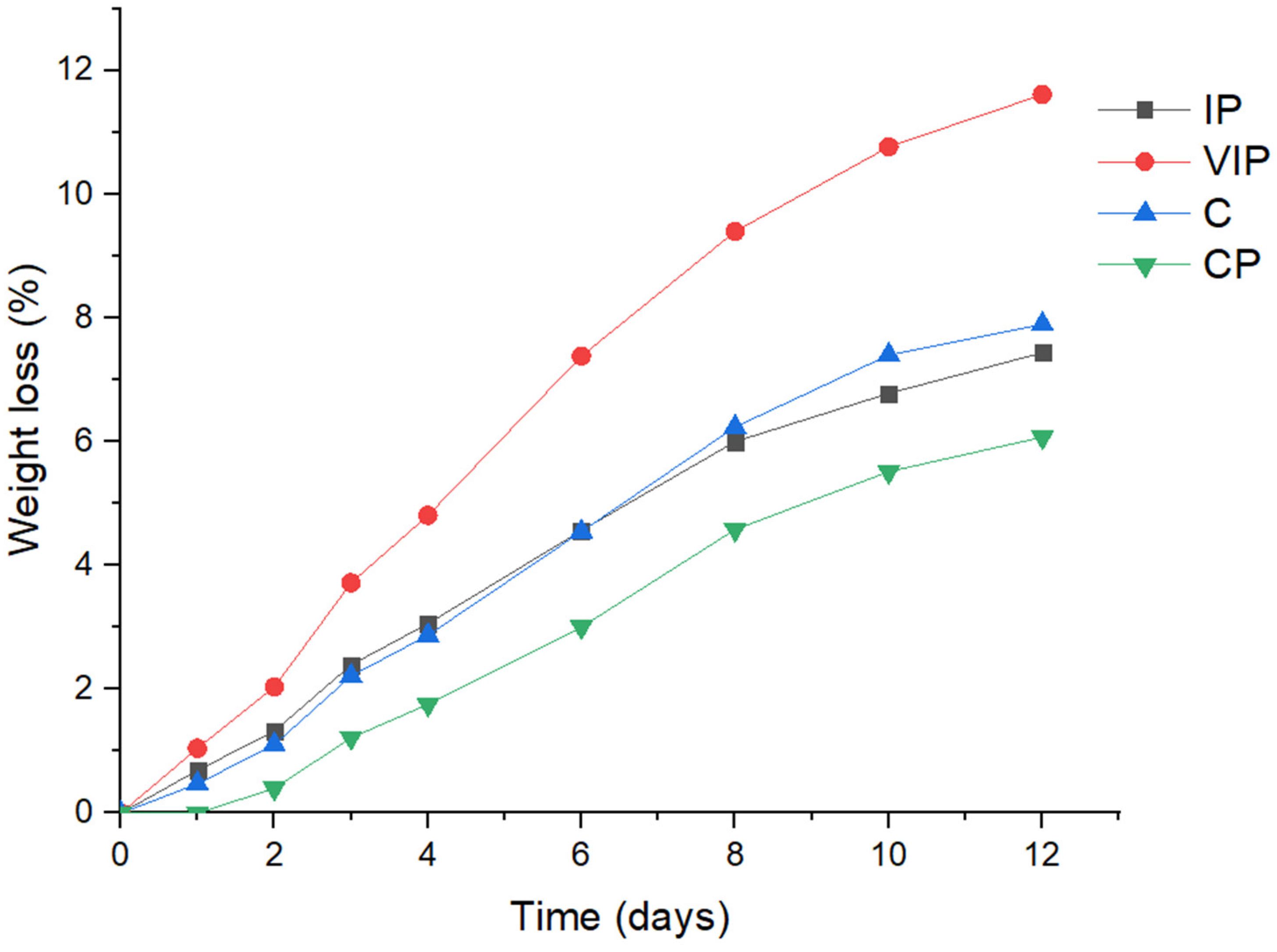
3.2.2. Weight Loss
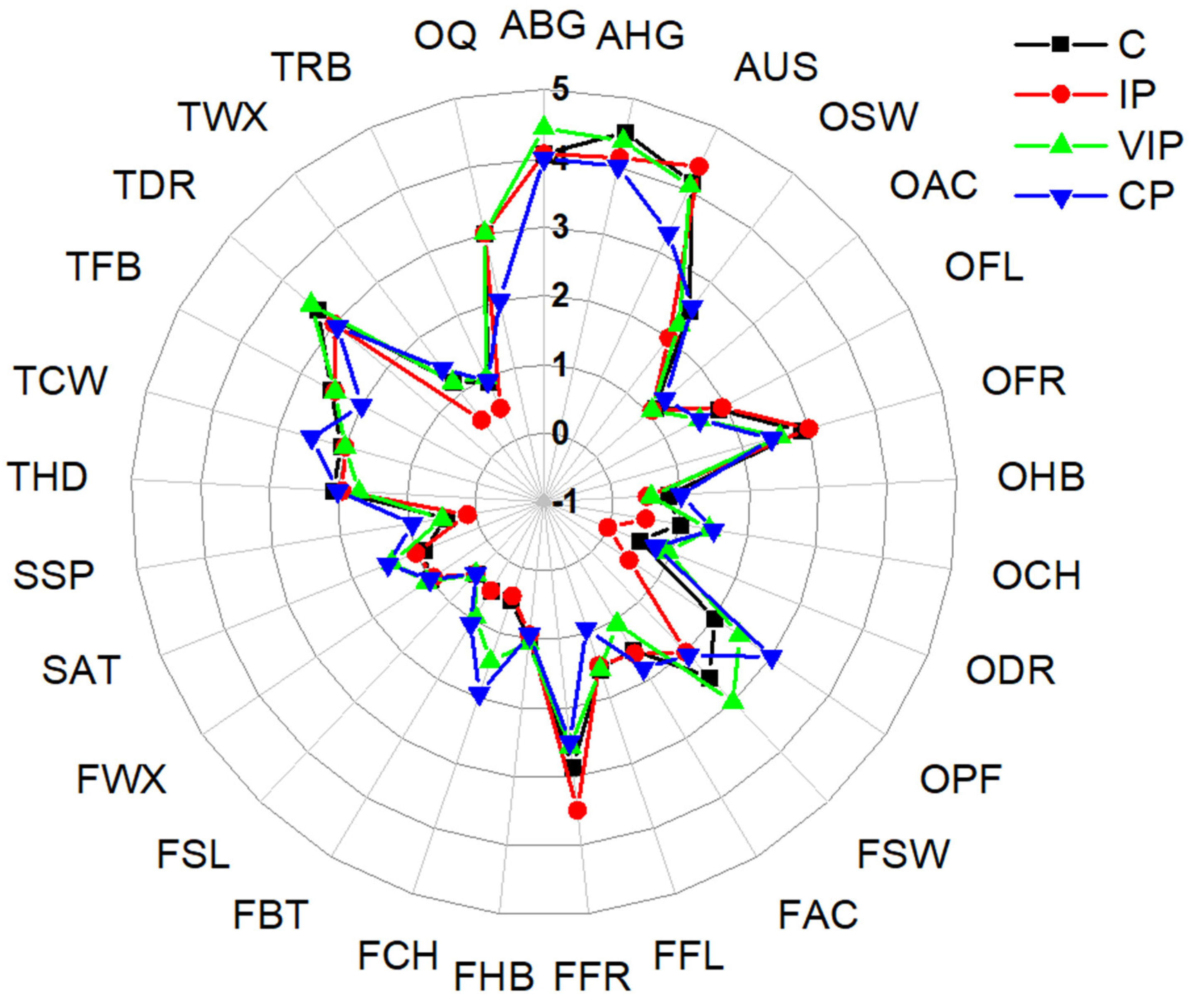
3.2.3. Sensory Analysis
3.2.4. Postharvest Treatment Selection
3.3. Immersion Process (IP) under Different Conditions
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Cardenas-Barboza, L.C.; Paredes-Cordoba, A.C.; Serna-Cock, L.; Guancha-Chalapud, M.; Torres-Leon, C. Quality of Physalis peruviana Fruits Coated with Pectin and Pectin Reinforced with Nanocellulose from P. Peruviana Calyces. Heliyon 2021, 7, e07988. [Google Scholar] [CrossRef] [PubMed]
- Akhtar, N.; Chaudhari, S.K.; Arshad, S.; Fatima, H.; Amjad, M.S. Saboon Biological Activities and Nutritional Value of Physalis peruviana L. Nat. Bio-Active Compd. Vol. 1 Prod. Appl. 2019, 587–598. [Google Scholar] [CrossRef]
- González-Locarno, M.; Pautt, Y.M.; Albis, A.; López, E.F.; Tovar, C.D.G. Assessment of Chitosan-Rue (Ruta Graveolens l.) Essential Oil-Based Coatings on Refrigerated Cape Gooseberry (Physalis peruviana L.) Quality. Appl. Sci. 2020, 10, 2684. [Google Scholar] [CrossRef]
- Etzbach, L.; Pfeiffer, A.; Weber, F.; Schieber, A. Characterization of Carotenoid Profiles in Goldenberry (Physalis peruviana L.) Fruits at Various Ripening Stages and in Different Plant Tissues by HPLC-DAD-APCI-MSn. Food Chem. 2018, 245, 508–517. [Google Scholar] [CrossRef]
- Ministerio de Agricultura y Desarrollo Rural AGRONET-Rendimiento Nacional Por Cultivo. Uchuva. Available online: https://www.agronet.gov.co/estadistica/Paginas/home.aspx?cod=1 (accessed on 31 January 2022).
- Heredia, A.M.; Quiroga, R.J.; Kirschbaum, D.S. Primer Reporte de Géneros Fúngicos Causando Decaimiento Poscosecha En Goldenberry (Physalis peruviana L.) En Argentina. Libro De Resúmenes. 4 Congr. Argent. De Fitopatol. 2017, 219, 2. [Google Scholar]
- Bisht, B.; Bhatnagar, P.; Gururani, P.; Kumar, V.; Tomar, M.S.; Sinhmar, R.; Rathi, N.; Kumar, S. Food Irradiation: Effect of Ionizing and Non-Ionizing Radiations on Preservation of Fruits and Vegetables—A Review. Trends Food Sci. Technol. 2021, 114, 372–385. [Google Scholar] [CrossRef]
- Denoya, G.I.; Colletti, A.C.; Vaudagna, S.R.; Polenta, G.A. Application of Non-Thermal Technologies as a Stress Factor to Increase the Content of Health-Promoting Compounds of Minimally Processed Fruits and Vegetables. Curr. Opin. Food Sci. 2021, 42, 224–236. [Google Scholar] [CrossRef]
- Pérez-Martínez, B.S.; Ramos-Dubón, E.J.; Ramos-Cortez, S.; Munguía, H.E. Evaluación de Dos Combinaciones de Conservantes y Su Efecto Sobre Un Producto Hortícola de IV Gama. Rev. Agrociencia-Rev. Conten. Científico La Fac. Cienc. Agronómicas La Univ. El Salvador 2021, 4, 38–49. [Google Scholar]
- Balaguera-lópez, H.E.; Martínez, C.A.; Herrera-Arévalo, A. The Role of the Calyx in the Postharvest Behavior of Cape Gooseberry Fruits Ecotype Colombia. Rev. Colomb. Cienc. Hortícolas 2014, 8, 181–191. [Google Scholar]
- Pinzón, E.H.; Reyes, A.J.; Álvarez-Herrera, J.G.; Leguizamo, M.F.; Joya, J.G. Comportamiento Del Fruto de Uchuva Physalis peruviana L., Bajo Diferentes Temperaturas de Almacenamiento. Rev. Cienc. Agrícolas 2015, 32, 26–35. [Google Scholar] [CrossRef]
- Ciro, G. Conservación de Uchuva Basada En La Impregnación a Vacío de Extractos de Plantas Con Actividad Antimicrobiana y Antioxidante. Ph.D. Thesis, Universidad de Antioquia, 2012. [Google Scholar]
- Puente, L.A.; Pinto-Muñoz, C.A.; Castro, E.S.; Cortés, M. Physalis peruviana Linnaeus, the Multiple Properties of a Highly Functional Fruit: A Review. Food Res. Int. 2011, 44, 1733–1740. [Google Scholar] [CrossRef]
- Turkkan, M.; Ozcan, M.; Erper, İ. Antifungal Effect of Carbonate and Bicarbonate Salts against Botrytis Cinerea, the Casual Agent of Grey Mould of Kiwifruit. Akad. Ziraat Derg. 2017, 6, 107–114. [Google Scholar] [CrossRef]
- Duarte-Correa, Y.; Díaz-Osorio, A.; Osorio-Arias, J.; Sobral, P.J.A.; Vega-Castro, O. Development of Fortified Low-Fat Potato Chips through Vacuum Impregnation and Microwave Vacuum Drying. Innov. Food Sci. Emerg. Technol. 2020, 64, 102437. [Google Scholar] [CrossRef]
- Instituto Colombiano de Normas Técnicas y Certificación (ICONTEC) Norma Técnica Colombiana (NTC) 4580, Frutas Frescas. Uchuva. Especificaciones; ICONTEC: Bogotá (D.C.), Colombia, 1999; Available online: https://kontii.files.wordpress.com/2012/10/ntc-4580.pdf (accessed on 31 January 2022).
- Muley, A.B.; Singhal, R.S. Extension of Postharvest Shelf Life of Strawberries (Fragaria Ananassa) Using a Coating of Chitosan-Whey Protein Isolate Conjugate. Food Chem. 2020, 329, 127213. [Google Scholar] [CrossRef]
- AOAC. Official Methods of Analysis of AOAC International, 17th ed.; Latimer, D.G.W., Ed.; Association of Offical Analytical Chemists: Gaithersburg, MD, USA, 2000; ISBN 935584870. [Google Scholar]
- Contreras-Calderón, J.; Calderón-Jaimes, L.; Guerra-Hernández, E.; García-Villanova, B. Antioxidant Capacity, Phenolic Content and Vitamin C in Pulp, Peel and Seed from 24 Exotic Fruits from Colombia. Food Res. Int. 2011, 44, 2047–2053. [Google Scholar] [CrossRef]
- Rodriguez-Amaya, D.; Kimura, M. Handbook for Carotenoid Analysis. Harvest. Tech. Monogr. 2004, 8–19. [Google Scholar]
- Osorio-Arias, J.; Delgado-Arias, S.; Cano, L.; Zapata, S.; Quintero, M.; Nuñez, H.; Ramírez, C.; Simpson, R.; Vega-Castro, O. Sustainable Management and Valorization of Spent Coffee Grounds Through the Optimization of Thin Layer Hot Air-Drying Process. Waste Biomass Valorization 2019, 11, 5015–5026. [Google Scholar] [CrossRef]
- Olivares-Tenorio, M.L.; Dekker, M.; van Boekel, M.A.J.S.; Verkerk, R. Evaluating the Effect of Storage Conditions on the Shelf Life of Cape Gooseberry (Physalis peruviana L.). Lwt 2017, 80, 523–530. [Google Scholar] [CrossRef]
- Instituto Colombiano de Normas Técnicas y Certificación (ICONTEC) Norma Tecnica Colombiana (NTC) 3932. Análisis Sensorial. In Identificación y Selección de Descriptores Para Establecer Un Perfil Sensorial Por Una Aproximación Multidimensional; Instituto Colombiano de Normas Técnicas y Certificación: Bogotá (D.C.), Colombia, 1996. [Google Scholar]
- Bravo, K.; Sepulveda-Ortega, S.; Lara-Guzman, O.; Navas-Arboleda, A.A.; Osorio, E. Influence of Cultivar and Ripening Time on Bioactive Compounds and Antioxidant Properties in Cape Gooseberry (Physalis peruviana L.). J. Sci. Food Agric. 2015, 95, 1562–1569. [Google Scholar] [CrossRef] [PubMed]
- Ramadan, M.F. Bioactive Phytochemicals, Nutritional Value, and Functional Properties of Cape Gooseberry (Physalis peruviana): An Overview. Food Res. Int. 2011, 44, 1830–1836. [Google Scholar] [CrossRef]
- Graça Dias, M.; Olmedilla-Alonso, B.; Hornero-Méndez, D.; Mercadante, A.Z.; Osorio, C.; Vargas, L.; Meléndez-Martínez, A. Tabla de Contenido de Alimentos Iberoamericanos. En Carotenoides en agroalimentación y salud (Cap. 18); Programa Iberoamericano de Ciencia y Tecnología para el Desarrollo: Madrid, Spain, 2017; pp. 354–429. [Google Scholar]
- Valente, A.; Albuquerque, T.G.; Sanches-Silva, A.; Costa, H.S. Ascorbic Acid Content in Exotic Fruits: A Contribution to Produce Quality Data for Food Composition Databases. Food Res. Int. 2011, 44, 2237–2242. [Google Scholar] [CrossRef]
- Céron, I.; Higuita, J.; Cardona, C. Capacidad Antioxidante y Contenido Fenólico Total de Tres Frutas Cultivadas En La Región Andina. Vector 2011, 18, 2–10. [Google Scholar]
- La Vega, J.C.D.; Cañarejo, M.A.; Cabascango, O.N.; Lara, M.V. Deshidratado de Physalis peruviana L. En Dos Estados de Madurez y Su Efecto Sobre El Contenido de Polifenoles Totales, Capacidad Antioxidante, Carotenos, Color y Ácido Ascórbico. Inf. Tecnológica 2019, 30, 91–100. [Google Scholar] [CrossRef]
- Duarte-Correa, Y.; Vargas-Carmona, M.I.; Vásquez-Restrepo, A.; Ruiz Rosas, I.D.; Pérez Martínez, N. Native Potato (Solanum Phureja) Powder by Refractance Window Drying: A Promising Way for Potato Processing. J. Food Process Eng. 2021, 44, e13819. [Google Scholar] [CrossRef]
- López, D.; Cuatin, L.; Andrade, J.; Osorio, O. Evaluation of an Edible Coating Based Whey Protein and Beeswax on the Physical and Chemical Quality of Gooseberry (Physalis peruviana L.). Acta Agron. 2016, 65, 326–333. [Google Scholar] [CrossRef]
- Sogvar, O.B.; Koushesh Saba, M.; Emamifar, A. Aloe Vera and Ascorbic Acid Coatings Maintain Postharvest Quality and Reduce Microbial Load of Strawberry Fruit. Postharvest Biol. Technol. 2016, 114, 29–35. [Google Scholar] [CrossRef]
- Barba, F.J.; Esteve, M.J.; Frigola, A. Physicochemical and Nutritional Characteristics of Blueberry Juice after High Pressure Processing. Food Res. Int. 2013, 50, 545–549. [Google Scholar] [CrossRef]
- Fito, P.; Andrb, A.; Chiralt, A.; Pardo, P. Coupling of Hydrodynamic Mechanism and Phenomena During Vacuum Treatments in Solid Porous Food-Liquid Systems. J. Food Eng. 1996, 21, 229–240. [Google Scholar] [CrossRef]
- Sharma, M.; Dash, K.K. Effect of Ultrasonic Vacuum Pretreatment on Mass Transfer Kinetics during Osmotic Dehydration of Black Jamun Fruit. Ultrason. Sonochem. 2019, 58, 104693. [Google Scholar] [CrossRef]
- Radziejewska-kubzdela, E.; Kido, M. Applicability of Vacuum Impregnation to Modify Physico-Chemical, Sensory and Nutritive Characteristics of Plant Origin Products—A Review. Int. J. Mol. Sci. 2014, 15, 16577–16610. [Google Scholar] [CrossRef]
- Rodríguez-ramírez, J.; Barragán-iglesias, J.; Ramírez-palma, A.J.; Méndez-lagunas, L.L. Effect of Calcium and Osmotic Pretreatments on Mass Transfer and Texture Parameters during Processing of Chilacayote (Cucurbita Ficifolia Bouché). 2023, 2023, 3873662. J. Food Process. Preserv. 2023, 2023, 3873662. [Google Scholar] [CrossRef]
- Muñoz, A.; Barbosa, A.; Bustos, D.; Ramírez, Y.; Vásquez, Y.; García, J.; Guancha, M. Conservation of Uchuva (Physalis Peruviana) by Applying a Coating Based on Chitosan and Aloe Vera, Using the Spray Method. Inf. Técnico 2017, 81, 86–94. [Google Scholar] [CrossRef]
- Duarte-Correa, Y.; Granda-Restrepo, D.; Cortés, M.; Vega-Castro, O. Potato Snacks Added with Active Components: Effects of the Vacuum Impregnation and Drying Processes. J. Food Sci. Technol. 2020, 57, 1523–1534. [Google Scholar] [CrossRef]
- Pauro-Flóres, V. Aplicación de Dos Métodos (Encerado o Inmersión En Cloruro de Calcio) Para La Conservación Poscosecha Del Aguaymanto (Physalis peruviana) Sin Cáliz; Universidad Nacional del Altiplano: Puno, Peru, 2016. [Google Scholar]
- Del Rodríguez, S.C.; Generoso, S.; Gutiérrez, D.; Questa, A. Application of Sensory Analysis in the Evaluation of Quality Fresh-cut Vegetables. Simiente 2015, 85, 1–27. [Google Scholar]
- Castagnini, J.M. Estudio Del Proceso de Obtención de Zumo de Arándanos y Su Utilización Como Ingrediente Para La Obtención de Un Alimento Funcional Por Impregnación a Vacío; Universidad Politécnica de Valencia: Valencia, Spain, 2015. [Google Scholar]
- Barrera, C.; Betoret, N.; Corell, P.; Fito, P. Effect of Osmotic Dehydration on the Stabilization of Calcium-Fortified Apple Slices (Var. Granny Smith): Influence of Operating Variables on Process Kinetics and Compositional Changes. J. Food Eng. 2009, 92, 416–424. [Google Scholar] [CrossRef]
- Martínez-Sánchez, C.; Solis-Ramos, A.; Rodríguez-Miranda, J.; Juárez-Barrientos, J.; Ramírez-Rivera, E.; Ruíz-López, I.; Gómez-Aldapa, C.; Herman-Lara, E. Evaluation of Ascorbic Acid Impregnation by Ultrasound-Assisted Osmotic Dehydration in Plantain. J. Food Process. Preserv. 2022, 46, e16839. [Google Scholar] [CrossRef]
| Parameter | Cape Gooseberry Colombia Ecotype | |
|---|---|---|
| Moisture (%) | 82.91 ± 1.01 | |
| Total soluble solids | 15.20 ± 0.26 | |
| pH | 3.34 ± 0.00 | |
| Total acidity (%) | 1.63 ± 0.02 | |
| Color | L* | 46.42 ± 0.77 |
| a* | 12.72 ± 1.69 | |
| b* | 34.00 ± 2.38 | |
| Parameter | Cape Gooseberry Colombia Ecotype |
|---|---|
| Total phenolic content (mg GAE/100 g) | 48.49 ± 10.82 |
| Carotenoids (µg/100 g) | 827.33 ± 206.70 |
| Vitamin C (mg/100 g) | 29.49 ± 1.39 |
| ABTS (µmol TE/g) | 3.86 ± 0.74 |
| FRAP (µmol TE/g) | 8.55 ± 0.07 |
| Postharvest Treatment | Moisture (%) | Total Soluble Solids | pH | Total Acidity (%) | Vitamin C (mg/100 g) | Total Phenolic Content (mg GAE/100 g) | Firmness (gf) |
|---|---|---|---|---|---|---|---|
| Control (C) | 82.91 ± 1.01 a | 15.20 ± 0.26 a | 3.34 ± 0.00 a | 1.63 ± 1.01 a | 29.49 ± 1.39 a | 48.49 ± 10.82 a | 22.20 ± 2.17 a |
| Coating process (CP) | 82.87 ± 0.77 a | 16.08 ± 0.20 b | 3.17 ± 0.01 b | 2.13 ± 0.01 b | 32.65 ± 0.60 a,b | 115.96 ± 10.82 b | 18.43 ± 1.72 a,b |
| Vacuum impregnation process (VIP) | 83.71 ± 1.57 a | 14.51 ± 0.31 c | 3.31 ± 3.31 a | 2.22 ± 0.04 c | 31.55 ± 1.38 b | 95.52 ± 14.66 b,c | 15.00 ± 4.00 b |
| Immersion process (IP) | 83.49 ± 0.76 a | 14.93 ± 0.41 a,c | 3.13 ± 0.00 b | 2.32 ± 0.03 d | 32.44 ± 1.81 b | 123.67 ± 19.90 c | 29.00 ± 4.21 c |
| Run | CaCl2 (%) | Immersion Time CaCl2 (min) | Moisture (%) | Total Soluble Solids | Firmness (gf) | Weight loss (%) |
|---|---|---|---|---|---|---|
| 1 | 3 | 2.0 | 83.12 ± 0.44 | 13.90 ± 0.10 | 29.86 ± 4.06 | 8.19 ± 1.25 |
| 2 | 3 | 3.5 | 83.41 ± 0.67 | 14.33 ± 0.31 | 25.00 ± 3.30 | 7.48 ± 1.98 |
| 3 | 1 | 2.0 | 81.78 ± 0.61 | 14.10 ± 0.26 | 28.60 ± 3.58 | 7.35 ± 3.40 |
| 4 | 1 | 3.5 | 82.73 ± 1.32 | 14.50 ± 0.10 | 30.00 ± 3.94 | 6.51 ± 1.12 |
| 5 | 3 | 5.0 | 82.39 ± 0.21 | 14.33 ± 0.12 | 26.60 ± 2.19 | 7.52 ± 1.86 |
| 6 | 1 | 5.0 | 83.11 ± 0.51 | 14.33 ± 0.12 | 25.50 ± 3.00 | 7.77 ± 3.49 |
| 7 | 3 | 2.0 | 84.45 ± 0.83 | 12.73 ± 0.38 | 28.20 ± 2.17 | 7.32 ± 1.44 |
| 8 | 3 | 3.5 | 83.61 ± 0.50 | 13.27 ± 0.12 | 28.75 ± 6.45 | 9.06 ± 2.88 |
| 9 | 1 | 2.0 | 84.29 ± 1.04 | 12.60 ± 0.10 | 34.00 ± 2.83 | 7.72 ± 1.14 |
| 10 | 1 | 5.0 | 82.37 ± 0.20 | 13.30 ± 0.36 | 27.00 ± 2.94 | 8.00 ± 1.19 |
| 11 | 1 | 3.5 | 82.77 ± 0.83 | 13.93 ± 0.12 | 28.33 ± 4.16 | 6.64 ± 1.07 |
| 12 | 3 | 5.0 | 82.08 ± 1.16 | 13.83 ± 0.06 | 22.40 ± 2.51 | 10.91 ± 4.13 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Agudelo-Sánchez, S.; Mosquera-Palacios, Y.; David-Úsuga, D.; Cartagena-Montoya, S.; Duarte-Correa, Y. Effect of Processing Methods on the Postharvest Quality of Cape Gooseberry (Physalis peruviana L.). Horticulturae 2023, 9, 1158. https://doi.org/10.3390/horticulturae9101158
Agudelo-Sánchez S, Mosquera-Palacios Y, David-Úsuga D, Cartagena-Montoya S, Duarte-Correa Y. Effect of Processing Methods on the Postharvest Quality of Cape Gooseberry (Physalis peruviana L.). Horticulturae. 2023; 9(10):1158. https://doi.org/10.3390/horticulturae9101158
Chicago/Turabian StyleAgudelo-Sánchez, Sara, Yadiela Mosquera-Palacios, Dairon David-Úsuga, Susana Cartagena-Montoya, and Yudy Duarte-Correa. 2023. "Effect of Processing Methods on the Postharvest Quality of Cape Gooseberry (Physalis peruviana L.)" Horticulturae 9, no. 10: 1158. https://doi.org/10.3390/horticulturae9101158
APA StyleAgudelo-Sánchez, S., Mosquera-Palacios, Y., David-Úsuga, D., Cartagena-Montoya, S., & Duarte-Correa, Y. (2023). Effect of Processing Methods on the Postharvest Quality of Cape Gooseberry (Physalis peruviana L.). Horticulturae, 9(10), 1158. https://doi.org/10.3390/horticulturae9101158






