Biotechnologies and Strategies for Grapevine Improvement
Abstract
1. Introduction
2. Grapevine Genetic Diversity and Molecular Markers Used in the Identification of Cultivars
2.1. Morphological Markers
2.2. Cytological Markers
2.3. Molecular Markers
3. Grapevine Plant Regeneration Methods
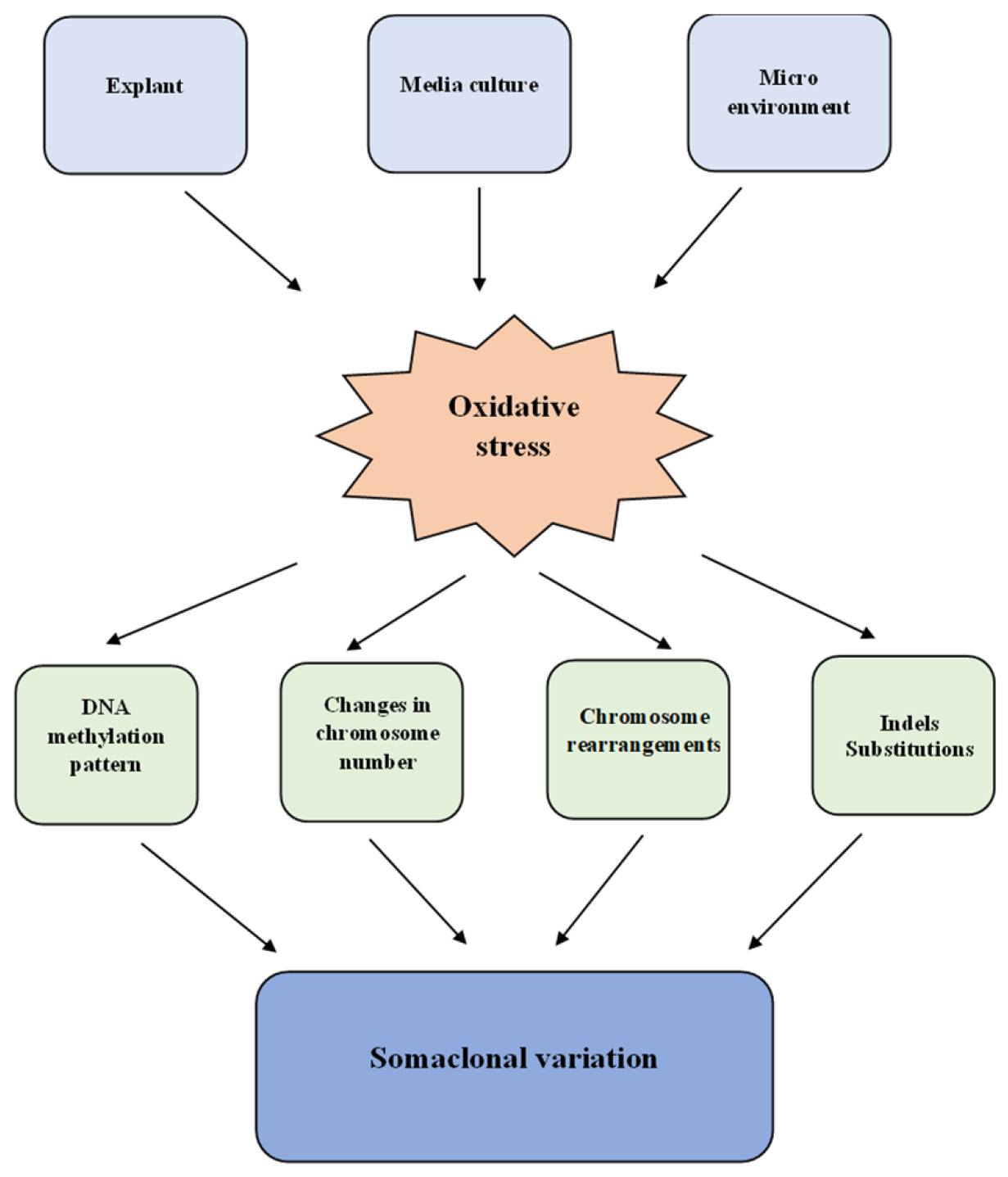
4. Somaclonal Variation
5. Genome Sequencing and Applications
6. Genetic Transformation
7. Genome Editing
8. Conclusions and Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Basso, M.F.; Fajardo, T.V.; Saldarelli, P. Grapevine virus diseases: Economic impact and current advances in viral prospection and management. Rev. Bras. Frutic. 2017, 39, e-411. [Google Scholar] [CrossRef]
- Bi, W.L.; Pan, C.; Hao, X.Y.; Cui, Z.H.; Kher, M.M.; Marković, Z.; Wang, Q.C.; Teixeira da Silva, J.A. Cryopreservation of grapevine [Vitis spp.]–A review. Vitr. Cell. Dev. Biol. Plant. 2017, 53, 449–460. [Google Scholar] [CrossRef]
- FAOSTAT. 2020. Available online: https://www.fao.org/faostat/en/#data (accessed on 5 January 2021).
- OIV 2020. State of the World Vitivinicultural Sector in 2020. Available online: https://www.oiv.int/ (accessed on 13 April 2021).
- EUROSTAT 2020. Available online: https://ec.europa.eu/eurostat/statistics-explained/index.php?title=Vineyards_in_the_EU_-_statistics&oldid=566726. (accessed on 5 March 2021).
- Reynolds, A.G. The Grapevine, Viticulture, and Winemaking: A Brief Introduction. In Grapevine Viruses: Molecular Biology, Diagnostics and Management; Meng, B., Martelli, G., Golino, D., Fuchs, M., Eds.; Springer: Berlin/Heidelberg, Germany, 2017; pp. 3–29. [Google Scholar] [CrossRef]
- Campos, G.; Chialva, C.; Miras, S.; Lijavetzky, D. New technologies and strategies for grapevine breeding through genetic transformation. Front. Plant. Sci. 2021, 25, 767522. [Google Scholar] [CrossRef] [PubMed]
- Abiri, K.; Rezaei, M.; Tahanian, H.; Heidari, P.; Khadivi, A. Morphological and pomological variability of a grape (Vitis vinifera L.) germplasm collection. Sci. Hortic. 2020, 266, 1–12. [Google Scholar] [CrossRef]
- Kambiranda, D.; Obuya, J.; Snowden, J. Grapevine Improvement through Biotechnology. In Genetic Transformation in Crops; To, K.-Y., Ed.; IntechOpen: London, UK, 2020. [Google Scholar] [CrossRef]
- Chacón-Vozmediano, J.L.; Gramaje, D.; León, M.; Armengol, J.; Moral, J.; Izquierdo-Cañas, P.M.; Martínez-Gascueña, J. Cultivar Susceptibility to Natural Infections Caused by Fungal Grapevine Trunk Pathogens in La Mancha Designation of Origin (Spain). Plants 2021, 10, 1171. [Google Scholar] [CrossRef]
- Galet, P. Apoplexie. In: Les maladies et les parasites de la vigne. Imp. Paysan du Midi Montp. 1977, 1, 409–430. [Google Scholar]
- Santos, R.B.; Figueiredo, A. Two sides of the same story in grapevine-pathogen interactions. J. Exp. Bot. 2021, 72, 3367–3380. [Google Scholar] [CrossRef]
- Cardell, M.; Amengual, A.; Romero, R. Future effects of climate change on the suitability of wine grape production across Europe. Reg. Environ. Change 2019, 19, 1–12. [Google Scholar] [CrossRef]
- Santos, J.A.; Fraga, H.; Malheiro, A.C.; Moutinho-Pereira, J.; Dinis, L.-T.; Correia, C.; Moriondo, M.; Leolini, L.; Dibari, C.; Costafreda-Aumedes, S.; et al. A Review of the Potential Climate Change Impacts and Adaptation Options for European Viticulture. Appl. Sci. 2020, 10, 3092. [Google Scholar] [CrossRef]
- Töpfer, R.; Trapp, O. A cool climate perspective on grapevine breeding: Climate change and sustainability are driving forces for changing varieties in a traditional market. Appl. Genet. 2022, 7, 1–4. [Google Scholar] [CrossRef]
- Fraga, H. Climate Change: A New Challenge for the Winemaking Sector. Agronomy 2020, 10, 1465. [Google Scholar] [CrossRef]
- Zhang, X.-M.; Wu, Y.-F.; Li, Z.; Song, C.-B.; Wang, X.-P. Advancements in plant regeneration and genetic transformation of grapevine (Vitis spp.). J. Integrat. Agric. 2021, 20, 1407–1434. [Google Scholar] [CrossRef]
- Gray, D.J.; Meredith, C.P. Biotechnology of Perennial Fruit Crops; Hammerschlag, F.A., Litz, R.E., Eds.; CAB International: Wallingford, UK, 1992; pp. 229–262. [Google Scholar]
- Vezzulli, S.; Doligez, A.; Bellin, D. Molecular Mapping of Grapevine Genes. In The Grape Genome. Compendium of Plant Genomes; Cantu, D., Walker, M., Eds.; Springer: Cham, Switzerland, 2019. [Google Scholar] [CrossRef]
- Directive 2001/18/ec of the European Parliament and of the Council of 12 March 2001 on the Deliberate Release into the Environment of Genetically Modified Organisms and Repealing Council Directive 90/220/EEC (OJ L 106, 17.4.2001, pp. 1–60). Available online: https://www.legislation.gov.uk/eudr/2001/18/contents (accessed on 21 September 2022).
- Regulation (EC) No 1829/2003 of the European Parliament and of the Council of 22 September 2003 on Genetically Modified Food and Feed (Text with EEA Relevance). Official Journal L 268, 18/10/2003 P. 0001–0023. Available online: https://eur-lex.europa.eu/legal-content/EN/ALL/?uri=CELEX%3A32003R1829 (accessed on 21 September 2022).
- Regulation (EC) No 1830/2003 of the European Parliament and of the Council of 22 September 2003 Concerning the Traceability and Labelling of Genetically Modified Organisms and the Traceability of Food and Feed Products Produced from Genetically Modified Organisms and Amending Directive 2001/18/EC. Official Journal L 268, 18/10/2003 P. 0024–0028. Available online: https://eur-lex.europa.eu/legal-content/EN/ALL/?uri=CELEX%3A32003R1829 (accessed on 21 September 2022).
- Dalla Costa, L.; Malnoy, M.; Lecourieux, D.; Deluc, L.; Ouaked-Lecourieux, F.; Deluc, L.; Ouaked-Lecourieux, F.; Thomas, M.; Torregrosa, L.J.-M. The state-of-the-art of grapevine biotechnology and new breeding technologies (NBTS). OENO One 2019, 53, 189–212. [Google Scholar] [CrossRef]
- Grassi, F.; De Lorenzis, G. Back to the origins: Background and perspectives of grapevine domestication. Int. J. Mol. Sci. 2021, 22, 4518. [Google Scholar] [CrossRef] [PubMed]
- Rivera Nuñez, D.; Walker, M.J. A review of palaeobotanical findings of early Vitis in the Mediterranean and the origins of cultivated grape-vines, with special reference to new pointers to prehistoric exploitation in the Western Mediterranean. Rev. Palaeobot. Palynol. 1989, 61, 205–237. [Google Scholar] [CrossRef]
- Grassi, F.; Labra, M.; Imazio, S.; Spada, A.; Sgorbati, S.; Scienza, A.; Sala, F. Evidence of secondary grapevine domestication centre detected by SSR analysis. Theor. App. Genet. 2003, 107, 1315–1320. [Google Scholar] [CrossRef]
- Arroyo-García, R.; Ruiz-García, L.; Bolling, L.; Ocete, R.; López, M.A.; Arnold, C.A.; Ergul, A.; Söylemezoglu, G.; Uzun, H.I.; Cabello, F.; et al. Multiple origins of cultivated grapevine (Vitis vinifera L. ssp. sativa) based on chloroplast DNA polymorphisms. Mol. Ecol. 2006, 15, 3707–3714. [Google Scholar] [CrossRef]
- Terral, J.F.; Tabard, E.; Bouby, L.; Ivorra, S.; Pastor, T.; Figueiral, I.; Picq, S.; Chevance, J.B.; Jung, C.; Fabre, L.; et al. Evolution and history of grapevine (Vitis vinifera) under domestication: New morphometric perspectives to understand seed domestication syndrome and reveal origins of ancient European cultivars. Ann. Bot. 2010, 105, 443–455. [Google Scholar] [CrossRef]
- Rusjan, D. Genetic and phenotypic diversity and relations between grapevine varieties: Slovenian germplasm. In The Mediterranean Genetic Code-Grapevine and Olive; Poljuha, D., Sladonja, B., Eds.; IntechOpen: London, UK, 2013. [Google Scholar] [CrossRef]
- Villano, C.; Aiese Cigliano, R.; Esposito, S.; D’Amelia, V.; Iovene, M.; Carputo, D.; Aversano, R. DNA-Based Technologies for grapevine biodiversity exploitation: State of the art and future perspectives. Agronomy 2022, 12, 491. [Google Scholar] [CrossRef]
- Emanuelli, F.; Lorenzi, S.; Grzeskowiak, L.; Catalano, V.; Stefanini, M.; Troggio, M.; Myles, S.; Martinez-Zapater, J.M.; Zyprian, E.; Moreira, F.M.; et al. Genetic diversity and population structure assessed by SSR and SNP markers in a large germplasm collection of grape. BMC Plant. Biol. 2013, 13, 39. [Google Scholar] [CrossRef]
- Zecca, G.; Grassi, F.; Tabidze, V.; Pipia, I.; Kotorashvili, A.; Kotaria, N.; Beridze, T. Dates and rates in grape’s plastomes: Evolution in slow motion. Curr. Genet. 2020, 66, 123–140. [Google Scholar] [CrossRef]
- Lacombe, T.; Audeguin, L.; Boselli, M.; Bucchetti, B.; Cabello, F.; Chatelet, P.; Crespan, M.; D’Onofrio, C.; Eiras Dias, J.; Ercisli, S.; et al. Grapevine European catalogue: Towards a comprehensive list. Vitis 2011, 50, 65–68. [Google Scholar] [CrossRef]
- Jackson, R.S. Grape Species and Varieties. In Food Science and Technology, Wine Science, 4th ed.; Ronald, S.J., Ed.; Academic Press: Cambridge, MA, USA, 2014; pp. 21–67. ISBN 9780123814685. [Google Scholar] [CrossRef]
- Panara, F.; Bergamini, C.; Palliotti, A.; Calderini, O. Use of Molecular Markers (Ssrs) and Public Databases in Vitis vinifera L. as the Main Case of Efficient Crop Cultivar Identification. JOJ Hortic. Arboric. 2018, 2, 555576. [Google Scholar] [CrossRef]
- Sefc, K.; Pejić, I.; Maletić, E.; Thomas, M.; Lefort, F. Microsatellite Markers for Grapevine: Tools for Cultivar Identification & Pedigree Reconstruction. In Grapevine Molecular Physiology & Biotechnology; Roubelakis-Angelakis, K.A., Ed.; Springer: Dordrecht, The Netherland, 2009. [Google Scholar] [CrossRef]
- Nasiri, A.; Taheri-Garavand, A.; Fanourakis, D.; Zhang, Y.-D.; Nikoloudakis, N. Automated Grapevine Cultivar Identification via Leaf Imaging and Deep Convolutional Neural Networks: A Proof-of-Concept Study Employing Primary Iranian Varieties. Plants 2021, 10, 1628. [Google Scholar] [CrossRef] [PubMed]
- Cho, K.H.; Noh, J.H.; Park, S.J.; Kim, S.H.; Kim, D.; Chun, J.A. Development of sequence characterized amplified region markers for the identification of grapevine cultivars. Hortic. Sci. 2015, 50, 1744–1750. [Google Scholar] [CrossRef]
- Castro, C.; Carvalho, A.; Pavia, I.; Leal, F.; Moutinho-Pereira, J.; Lima-Brito, J. Nucleolar Activity and Physical Location of Ribosomal DNA Loci in Vitis vinifera L. by Silver Staining and Sequential FISH. Sci. Hortic. 2018, 232, 57–62. [Google Scholar] [CrossRef]
- Pereira, H.S.; Barão, A.; Delgado, M.; Morais-Cecílio, L.; Viegas, W. Genomic Analysis of Grapevine Retrotransposon 1 (Gret1) in Vitis vinifera. Theor. Appl. Genet. 2005, 111, 871–878. [Google Scholar] [CrossRef]
- Falistocco, E.; Passeri, V.; Marconi, G. Investigations of 5S RDNA of Vitis vinifera L.: Sequence Analysis and physical mapping. Genome 2007, 50, 927–938. [Google Scholar] [CrossRef]
- Giannuzzi, G.; D’Addabbo, P.; Gasparro, M.; Martinelli, M.; Carelli, F.N.; Antonacci, D.; Ventura, M. Analysis of High-Identity Segmental Duplications in the Grapevine Genome. BMC Genom. 2011, 12, 436. [Google Scholar] [CrossRef]
- Pereira, H.S.; Delgado, M.; Avó, A.P.; Barão, A.; Serrano, I.; Viegas, W. Pollen grain development is highly sensitive to temperature stress in Vitis vinifera. Aust. J. Grape Wine Res. 2014, 20, 474–484. [Google Scholar] [CrossRef]
- Goswami, M.; Attri, K.; Goswami, I. Applications of Molecular Markers in Fruit Crops: A Review. IJEP 2022, 9, 121–126. Available online: http://www.pphouse.org/ijep-article-details.php?art=323 (accessed on 12 September 2022). [CrossRef]
- Bourquin, J.; Otten, L.; Walter, B. PCR-RFLP analysis of Vitis, Ampelopsis and Parthenocissus and its application to the identification of rootstocks. Vitis 1995, 34, 103–108. [Google Scholar]
- Karataş, H.; Ağaoğlu, Y.S. RAPD analysis of selected local Turkish grape cultivars (Vitis vinifera). GMR 2010, 9, 1980–1986. [Google Scholar] [CrossRef]
- Vidal, J.; Delavault, P.; Coarer, M.; Defontaine, A. Design of grapevine (Vitis vinifera L.) cultivar-specific SCAR primers for PCR fingerprinting. Appl Genet. 2000, 101, 1194–1201. [Google Scholar] [CrossRef]
- Pollastro, S.; Dongiovanni, C.; Abbatecola, A.; de Guido, M.A.; de Miccolis Angelini, R.M.; Faretra, F. Specific SCAR Primers for Fungi Associated with Wood Decay of Grapevine. Phytopathol. Mediterr. 2001, 40, S362–S368. [Google Scholar]
- Villano, C.; Carputo, D.; Frusciante, L.; Santoro, X.; Aversano, R. Use of SSR and Retrotransposon-Based Markers to Interpret the Population Structure of Native Grapevines from Southern Italy. Mol. Biotechnol. 2014, 56, 1011–1020. [Google Scholar] [CrossRef] [PubMed]
- Karimi, M.R.; Dehvari, V.; Hajiyan, M. Genetics Diversity of Some Grape Genotypes by ISSR and RAPD Markers. Eur. J. Hortic.Sci. 2011, 76, 201–207. [Google Scholar]
- Nookaraju, A.; Agrawal, D.C. Genetic homogeneity of in vitro raised plants of grapevine cv. Crimson Seedless revealed by ISSR and microsatellite markers. S. Afr. J. Bot. 2012, 78, 302–306. [Google Scholar] [CrossRef][Green Version]
- Stajner, N.; Jakse, J.; Javornik, B.; Masuelli, R.W.; Martínez, L.E. Highly variable AFLP and S-SAP markers for the identification of ‘Malbec’ and ‘Syrah’ clones. Vitis 2009, 48, 145–150. [Google Scholar]
- Péros, J.P.; Cousins, P.; Launay, A.; Cubry, P.; Walker, A.; Prado, E.; Peressotti, E.; Wiedemann-Merdinoglu, S.; Laucou, V.; Merdinoglu, D.; et al. Genetic diversity and population structure in Vitis species illustrate phylogeographic patterns in eastern North America. Mol. Ecol. 2021, 30, 2333–2348. [Google Scholar] [CrossRef]
- Peng, F.Y.; Reid, K.E.; Liao, N.; Schlosser, J.; Lijavetzky, D.; Holt, R.; Martínez Zapater, J.M.; Jones, S.; Marra, M.; Bohlmann, J.; et al. Generation of ESTs in Vitis vinifera wine grape (Cabernet Sauvignon) and table grape (Muscat Hamburg) and discovery of new candidate genes with potential roles in berry development. Gene 2007, 402, 40–50. [Google Scholar] [CrossRef]
- Ji, X.N.; Li, F.; Yang, C.J.; Li, B.; Wang, J.; Zhang, W. Expressed sequence tags (ESTs) analysis of the ripening Vitis amurensis cv. Shuang Hong berry skins. J. For. Res. 2013, 24, 495–502. [Google Scholar] [CrossRef]
- Lü, X.-L.; Zhang, G.-L.; Liao, M.-A.; Gong, R.-G.; Zeng, X.-L. Random Amplified Microsatellite Polymorphism (RAMP) Analysis of Grape Breeds Genetic Relationships. JSAU 2004, 22, 133–137. [Google Scholar] [CrossRef]
- Arroyo-García, R.; Lefort, F.; de Andrés, M.T.; Ibáñaez, J.; Borrego, J.; Jouve, N.; Cabello, F.; Martínez-Zapater, J.M. Chloroplast microsatellite polymorphisms in Vitis species. Genome 2002, 45, 1142–1149. [Google Scholar] [CrossRef]
- Veloso, M.M.; Almadanim, M.C.; Baleiras-Couto, M.; Pereira, H.S.; Carneiro, L.C.; Fevereiro, P.; Eiras-Dias, J.E. Microsatellite database of grapevine (Vitis vinifera L.) cultivars used for wine production in Portugal. Cien. Tecn. Vitivinic. 2010, 25, 53–61. [Google Scholar]
- Castro, I.; Martín, J.P.; Ortiz, J.M.; Pinto-Carnide, O. Varietal discrimination and genetic relationships of Vitis vinifera L. cultivars from two major controlled appellation (DOC) regions in Portugal. Sci. Hortic. 2011, 127, 507–514. [Google Scholar] [CrossRef]
- Castro, I.; Pinto-Carnide, O.; Ortiz, J.M.; Martin, J.P. Chloroplast genome diversity in Portuguese grapevine (Vitis vinifera L.) cultivars. Mol. Biotechnol. 2013, 54, 528–540. [Google Scholar] [CrossRef]
- Ferreira, V.; Pinto-Carnide, O.; Mota, T.; Martín, J.P.; Ortiz, J.M.; Castro, I. Identification of minority grapevine cultivars from Vinhos Verdes Portuguese DOC Region. Vitis 2015, 54, 53–58. [Google Scholar] [CrossRef]
- De Michele, R.; La Bella, F.; Gristina, A.S.; Fontana, I.; Pacifico, D.; Garfi, G.; Motisi, A.; Crucitti, D.; Abbate, L.; Carimi, F. Phylogenetic Relationship Among Wild and Cultivated Grapevine in Sicily: A Hotspot in the Middle of the Mediterranean Basin. Front. Plant. Sci. 2019, 10, 1506. [Google Scholar] [CrossRef] [PubMed]
- Bianchi, D.; Brancadoro, L.; De Lorenzis, G. Genetic Diversity and Population Structure in a Vitis spp. Core Collection Investigated by SNP Markers. Diversity 2020, 12, 103. [Google Scholar] [CrossRef]
- Flutre, T.; Le Cunff, L.; Fodor, A.; Launay, A.; Romieu, C.; Berger, G.; Bertrand, Y.; Terrier, N.; Beccavin, I.; Bouckenooghe, V.; et al. A genome-wide association and prediction study in grapevine deciphers the genetic architecture of multiple traits and identifies genes under many new QTLs. G3 Genes|Genomes|Genet. 2022, 12, jkac103. [Google Scholar] [CrossRef] [PubMed]
- Ghaffari, S.; Hasnaoui, N.; Zinelabidine, L.H.; Ferchichi, A.; Martínez-Zapater, J.M.; Ibáñez, J. Genetic diversity and parentage of Tunisian wild and cultivated grapevines (Vitis vinifera L.) as revealed by single nucleotide polymorphism (SNP) markers. Tree Genet. Genomes 2014, 10, 1103–1112. [Google Scholar] [CrossRef]
- Augusto, D.; Ibáñez, J.; Pinto-Sintra, A.L.; Falco, V.; Leal, F.; Martínez-Zapater, J.M.; Oliveira, A.A.; Castro, I. Grapevine Diversity and Genetic Relationships in Northeast Portugal Old Vineyards. Plants 2021, 10, 2755. [Google Scholar] [CrossRef] [PubMed]
- Lijavetzky, D.; Cabezas, J.A.; Ibáñez, A.; Rodríguez, V.; Martínez-Zapater, J.M. High throughput SNP discovery and genotyping in grapevine (Vitis vinifera L.) by combining a re-sequencing approach and SNPlex technology. BMC Genom. 2007, 8, 424. [Google Scholar] [CrossRef]
- Dong, X.; Chen, W.; Liang, Z.; Li, X.; Nick, P.; Chen, S.; Dong, Y.; Li, S.; Sheng, J. VitisGDB: The Multifunctional Database for Grapevine Breeding and Genetics. Mol. Plant. 2020, 13, 1098–1100. [Google Scholar] [CrossRef]
- Maul, E.; Töpfer, R. Vitis International Variety Catalogue (V IVC): A cultivar database referenced by genetic profiles and morphology. BIO Web Conf. 2015, 5, 01009. [Google Scholar] [CrossRef]
- Cabezas, J.A.; Cabezas, M.T.; Cervera, M.T.; Cervera, L.; Ruiz-García, L.; Ruiz-García, J.; Carreño, J.; Martínez-Zapater, C.J.M. A genetic analysis of seed and berry weight in grapevine. Genome 2006, 49, 1572–1585. [Google Scholar] [CrossRef]
- Maul, E.; Sudharma, K.N.; Kecke, S.; Marx, G.; Müller, C.; Audeguin, L.; Boselli, M.; Boursiquot, J.M.; Bucchetti, B.; Cabello, F.; et al. The European Vitis Database (www.Eu-Vitis.De): A Technical Innovation through An Online Uploading and Interactive Modification System. Vitis 2012, 51, 79–85. [Google Scholar]
- Lefort, F.; Roubelakis-Angelakis, K.A. The Greek Vitis Database: A MultimediaWeb-Backed Genetic Database for Germplasm Management of Vitis Resources in Greece. J. Wine Res. 2000, 11, 233–242. [Google Scholar] [CrossRef]
- Davey, J.W.; Hohenlohe, P.A.; Etter, P.D.; Boone, J.Q.; Catchen, J.M.; Blaxter, M.L. Genome-wide genetic marker discovery and genotyping using next-generation sequencing. Nat. Rev. Genet. 2011, 12, 499–510. [Google Scholar] [CrossRef]
- Marrano, A.; Birolo, G.; Prazzoli, M.L.; Lorenzi, S.; Valle, G.; Grando, M.S. SNP-Discovery by RAD-Sequencing in a Germplasm Collection of Wild and Cultivated Grapevines (V. vinifera L.). PLoS ONE 2017, 12, e0170655. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Guo, Y.; Su, K.; Liu, Z.; Ren, Z.; Li, K.; Guo, X. Construction of a highly saturated genetic map for Vitis by Next-generation restriction site-associated DNA sequencing. BMC Plant. Biol. 2018, 18, 347. [Google Scholar] [CrossRef] [PubMed]
- Gambino, G.; Dal Molin, A.; Boccacci, P.; Minio, A.; Chitarra, W.; Avanzato, C.G.; Tononi, P.; Perrone, I.; Raimondi, S.; Schneider, A. Whole-Genome Sequencing and SNV Genotyping of ‘Nebbiolo’ (Vitis vinifera L.) Clones. Sci. Rep. 2017, 7, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Carbonell-Bejerano, P.; Royo, C.; Torres-Pérez, R.; Grimplet, J.; Fernandez, L.; Franco-Zorrilla, J.M.; Lijavetzky, D.; Baroja, E.; Martínez, J.; García-Escudero, E. Catastrophic unbalanced genome rearrangements cause somatic loss of berry color in grapevine. Plant. Physiol. 2017, 175, 786–801. [Google Scholar] [CrossRef]
- Pucker, B.; Schwandner, A.; Becker, S.; Hausmann, L.; Viehöver, P.; Töpfer, R.; Weisshaar, B.; Holtgräwe, D. RNA-Seq time series of Vitis vinifera bud development reveals correlation of expression patterns with the local temperature profile. Plants 2020, 9, 1548. [Google Scholar] [CrossRef]
- Ma, Q.; Yang, J. Transcriptome profiling and identification of the functional genes involved in berry development and ripening in Vitis vinifera. Gene 2019, 680, 84–96. [Google Scholar] [CrossRef] [PubMed]
- Toffolatti, S.L.; De Lorenzis, G.; Brilli, M.; Moser, M.; Shariati, V.; Tavakol, E.; Maddalena, G.; Passera, A.; Casati, P.; Pindo, M. Novel aspects on the interaction between grapevine and Plasmopara Viticola: Dual-RNA-Seq analysis highlights gene expression dynamics in the pathogen and the plant during the battle for infection. Genes 2020, 11, 261. [Google Scholar] [CrossRef] [PubMed]
- Zou, C.; Karn, A.; Reisch, B.; Nguyen, A.; Sun, Y.; Bao, Y.; Campbell, M.S.; Church, D.; Williams, S.; Xu, X. Haplotyping the Vitis collinear core genome with rhAmpSeq improves marker transferability in a diverse genus. Nat. Commun. 2020, 11, 413. [Google Scholar] [CrossRef] [PubMed]
- Alizadeh, M.; Singh, S.K.; Jhang, T.; Sharma, T.R. Inter Simple Sequence Repeat Analysis to Confirm Genetic Stability of Micropropagated Plantlets in Three Grape (Vitis spp.) Rootstock Genotypes. J. Plant. Biochem. Biotechnol. 2008, 17, 77–80. [Google Scholar] [CrossRef]
- Hameed, U.K.A.; Abdelaziz, K.; El Sherif, N. Genetic diversity of grapevine (Vitis vinifera L.) cultivars in Al-Madinah Al-Munawara based on molecular markers and morphological traits. Bangladesh J. Plant. Taxon. 2020, 27, 113–127. [Google Scholar] [CrossRef]
- Guo, Y.; Lin, H.; Liu, Z.; Zhao, Y.; Guo, X.; Li, K. SSR and SRAP marker-based linkage map of Vitis vinifera L. Biotechnol. Biotechnol. Equip. 2014, 28, 221–229. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Gong, P.; Shi, Y.; Wang, Y.; Zhang, C. Genetic inter-relationships among Chinese wild grapes based on SRAP marker analyses. Vitis 2018, 57, 151–157. [Google Scholar] [CrossRef]
- Pelsy, F. Untranslated leader region polymorphism of Tvv1, a retrotransposon family, is a novel marker useful for analyzing genetic diversity and relatedness in the genus Vitis. Appl. Genet. 2007, 116, 15–27. [Google Scholar] [CrossRef]
- Pelsy, F.; Bevilacqua, L.; Blanc, S.; Merdinoglu, D.A. Molecular marker set combining a retrotransposon insertion and SSR polymorphisms is useful for assessing diversity in Vitis. OENO One 2021, 55, 403–414. [Google Scholar] [CrossRef]
- Roach, M.J.; Johnson, D.L.; Bohlmann, J.; van Vuuren, H.J.J.; Jones, S.J.M.; Pretorius, I.S.; Schmidt, S.A.; Borneman, A.R. Population sequencing reveals clonal diversity and ancestral inbreeding in the grapevine cultivar Chardonnay. PLoS Genet. 2018, 14, e1007807. [Google Scholar] [CrossRef] [PubMed]
- Butiuc-Keul, A.; Crăciunaş, C.; Coste, A.; Farago, M. Discrimination and genetic polymorphism in several cultivar of grapevine by RAPD markers. Rom. Biotechnol. Lett. 2010, 15, 110–115. [Google Scholar]
- Cretazzo, E.; Meneghetti, S.; De Andrés, M.T.; Gaforio, L.; Frare, E.; Cifre, J. Clone Differentiation and Varietal Identification by means of SSR, AFLP, SAMPL and M-AFLP in order to assess the clonal selection of grapevine: The case study of Manto Negro, Callet and Moll, autochthonous cultivars of Majorca. Ann. Appl. Biol. 2010, 157, 213–227. [Google Scholar] [CrossRef]
- Coste, A.; Postolache, D.; Popescu, F.; Butiuc-Keul, A. Authentication of valuable grapevine varieties from Romania, through molecular markers. Rom. Biotechnol. Lett. 2010, 15, 3–10. [Google Scholar]
- Baránková, K.; Sotolář, R.; Baránek, M. Identification of Rare Traditional Grapevine Cultivars Using SSR Markers and Their Geographical Location within the Czech Republic. Czech. J. Genet. Plant. Breed. 2020, 56, 71–78. [Google Scholar] [CrossRef]
- Jiménez-Cantizano, A.; Muñoz-Martín, A.; Amores-Arrocha, A.; Sancho-Galán, P.; Palacios, V. Identification of red grapevine cultivars (Vitis vinifera L.) preserved in ancient vineyards in Axarquia (Andalusia, Spain). Plants 2020, 9, 1572. [Google Scholar] [CrossRef]
- Ocaña, J.; Walter, B.; Schellenbaum, P. Stable MSAP markers for the distinction of Vitis vinifera cv Pinot Noir clones. Mol. Biotechnol. 2013, 55, 236–248. [Google Scholar] [CrossRef]
- Emanuelli, F.; Sordo, M.; Lorenzi, S.; Battilana, J.U.R.I.; Grando, M. Development of user-friendly functional molecular markers for VvDXS gene conferring muscat flavor in grapevine. Mol. Breed. 2014, 33, 235–241. [Google Scholar] [CrossRef] [PubMed]
- Castro, I.; D’Onofrio, C.; Martín, J.P.; Ortiz, J.M.; De Lorenzis, G.; Ferreira, V.; Pinto-Carnide, O. Effectiveness of AFLPs and retrotransposon-based markers for the identification of Portuguese grapevine cultivars and clones. Mol. Biotechnol. 2012, 52, 26–39. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Fan, X.C.; Chu, J.Q.; Liu, C.H.; Sun, X.; Fang, J.G. Identification of grapevine rootstock cultivars using expressed sequence tag-simple sequence repeats. GMR 2014, 13, 7649–7657. [Google Scholar] [CrossRef] [PubMed]
- D’Onofrio, C.; De Lorenzis, G.; Giordani, T.; Natali, L.; Cavallini, A.; Scalabrelli, G. Retrotransposon-based molecular markers for grapevine species and cultivars identification. Tree Genet. Genomes 2010, 6, 451–466. [Google Scholar] [CrossRef]
- Ulanovsky, S.; Gogorcena, Y.; Martinez de Toda, F.; Ortiz, J. Use of molecular markers in detection of synonymies and homonymies in grapevines (Vitis vinifera L.). Sci. Hortic. 2002, 92, 241–254. [Google Scholar] [CrossRef]
- Moreno-Sanz, P.; Suárez, B.; Loureiro, M.D. Identification of synonyms and homonyms in grapevine cultivars (Vitis vinifera L.) from Asturias (Spain). J. Hortic. Sci. Biotechnol. 2008, 83, 683–688. [Google Scholar] [CrossRef][Green Version]
- Castro, I.; Martín, J.P.; Ortiz, J.M.; Mota, M.T.; Pinto-Carnide, O.; Martin, J.; Ortiz, J.M.; Mota, M.T.; Pinto-Carnide, O. The Portuguese grapevine cultivar ‘Amaral’: Synonymies, homonymies and misnames. Vitis 2012, 51, 61–63. [Google Scholar] [CrossRef]
- Crespan, M.; Migliaro, D.; Larger, S.; Pindo, M.; Palmisano, M.; Manni, A.; Manni, E.; Polidori, E.; Sbaffi, F.; Silvestri, Q.; et al. Grapevine (Vitis vinifera L.) varietal assortment and evolution in the Marche region (central Italy). OENO One 2021, 55, 4628. [Google Scholar] [CrossRef]
- D’Onofrio, C.; Tumino, G.; Gardiman, M.; Crespan, M.; Bignami, C.; de Palma, L.; Barbagallo, M.G.; Muganu, M.; Morcia, C.; Novello, V.; et al. Parentage atlas of Italian grapevine varieties as inferred from SNP genotyping. Front. Plant. Sci. 2021, 11, 2265. [Google Scholar] [CrossRef]
- Nebish, A.; Tello, J.; Ferradás, Y.; Aroutiounian, R.; Martínez-Zapater, J.M.; Ibáñez, J. SSR and SNP Genetic Profiling of Armenian Grape Cultivars Gives Insights into Their Identity and Pedigree Relationships. OENO One 2021, 55, 101–114. [Google Scholar] [CrossRef]
- Botstein, D.; White, R.; Skolnick, M.; Davis, R. Construction of a genetic linkage map in man using restriction fragment length polymorphisms. Am. J. Hum. Genet. 1980, 32, 314–331. [Google Scholar] [PubMed]
- Vervalle, J.A.; Costantini, L.; Lorenzi, S.; Pindo, M.; Mora, R.; Bolognesi, G.; Marini, M.; Lashbrooke, J.G.; Tobutt, K.R.; Vivier, M.A.; et al. A high-density integrated map for grapevine based on three mapping populations genotyped by the Vitis18K SNP chip. TAG. Theor. Appl. Genet. 2022, 21, 1–20. [Google Scholar] [CrossRef]
- Duduk, B.; Botti, S.; Ivanović, M.; Krstić, B.; Dukić, N.; Bertaccini, A. Identification of Phytoplasmas associated with grapevine yellows in Serbia. J. Phytopathol. 2004, 152, 575–579. [Google Scholar] [CrossRef]
- Wang, Z.; Wang, Y.; Cao, X.; Wu, D.; Hui, M.; Han, X.; Yao, F.; Li, Y.; Li, H.; Wang, H. Screening and validation of ssr molecular markers for identification of downy mildew resistance in intraspecific hybrid F1 progeny (V. vinifera). Horticulturae 2022, 8, 706. [Google Scholar] [CrossRef]
- Goszczynski, D.E.; Jooste, A.E.C. The application of single-strand conformation polymorphism (SSCP) technique for the analysis of molecular heterogeneity of grapevine virus A. Vitis 2002, 41, 77–82. [Google Scholar]
- Aroca, A.; Raposo, R. PCR-based strategy to detect and identify species of Phaeoacremonium causing grapevine diseases. Appl. Environ. Microbiol. 2007, 73, 2911–2918. [Google Scholar] [CrossRef]
- Wang, X.C.; Guo, L.; Shangguan, L.F.; Wang, C.; Yang, G.; Qu, S.C.; Fang, J.G. Analysis of expressed sequence tags from grapevine flower and fruit and development of simple sequence repeat markers. Mol. Biol. Rep. 2012, 39, 6825–6834. [Google Scholar] [CrossRef]
- Crăciunaş, C.; Coste, A.; Farago, M.; Iliescu, M.; Iuoraş, R.; Butiuc-Keul, A. Genetic stability of several cultivars of grapevine cultivated in vitro. Acta Hortic. 2009, 812, 515–520. [Google Scholar] [CrossRef]
- Schneider, S.; Reustle, G.; Zyprian, E. Detection of somaclonal variation in grapevine regenerants from protoplasts by RAPD-PCR. Vitis 1996, 35, 99–100. [Google Scholar]
- Baránek, M.; Raddová, J.; Krizan, B.; Pidra, M. Genetic changes in grapevine genomes after stress induced by in vitro cultivation, thermotherapy and virus infection, as revealed by AFLP. Genet. Mol. Biol. 2009, 32, 834–839. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Gribaudo, I.; Torello Marinoni, D.; Gambino, G.; Mannini, F.; Akkak, A.; Botta, R. Assessment of genetic fidelity in regenerants from two Vitis vinifera cultivars. Acta Hortic. 2009, 827, 131–136. [Google Scholar] [CrossRef]
- Aljuaid, B.S.; Ismail, I.A.; Attia, A.O.; El Dessoky, S. Genetic Stability of in vitro Propagated Grapevine (Vitis vinifera L.) cv. Al-Bayadi. J. Agric. Crops 2022, 8, 12–19. [Google Scholar] [CrossRef]
- Schellenbaum, P.; Mohler, V.; Wenzel, G.; Walter, B. Variation in DNA methylation patterns of grapevine somaclones (Vitis vinifera L.). BMC Plant. Biol. 2008, 8, 78. [Google Scholar] [CrossRef] [PubMed]
- Dalbò, M.A. Genetic mapping, Qtl Analysis, and Marker-Assisted Selection for Disease Resistance Loci in Grapes. Ph.D. Thesis, Cornell University, Ithaca, NY, USA, August 1998. [Google Scholar]
- Doligez, A.; Bouquet, A.; Danglot, Y.; Lahogue, F.; Riaz, S.; Meredith, P.; Edwards, J.; This, P. Genetic mapping of grapevine (Vitis vinifera L.) applied to the detection of QTLs for seedlessness and berry weight. TAG. Theoretical and applied genetics. Theor. Und Angew. Genet. 2002, 105, 780–795. [Google Scholar] [CrossRef]
- Fanizza, G.; Lamaj, F.; Costantini, L.; Chaabane, R.; Grando, M.S. QTL analysis for fruit yield components in table grapes (Vitis vinifera). Appl Genet. 2005, 111, 658–664. [Google Scholar] [CrossRef] [PubMed]
- Costantini, L.; Moreira, F.; Zyprians, E.; Martínez-Zapater, J.; Grando, M. Molecular Maps, Qtl Mapping & Association Mapping In Grapevine Molecular Physiology & Biotechnology; Roubelakis-Angelakis, K.A., Ed.; Springer: Dordrecht, The Netherland, 2009. [Google Scholar] [CrossRef]
- Demmings, E.M.; Williams, B.R.; Lee, C.-R.; Barba, P.; Yang, S.; Hwang, C.-F.; Reisch, B.I.; Chitwood, D.H.; Londo, J.P. Quantitative trait locus analysis of leaf morphology indicates conserved shape loci in grapevine. Front. Plant. Sci. 2019, 10, 1373. [Google Scholar] [CrossRef]
- Su, K.; Guo, Y.; Zhong, W.; Lin, H.; Liu, Z.; Li, K.; Li, Y.; Guo, X. High-Density Genetic Linkage Map Construction and White Rot Resistance Quantitative Trait Loci Mapping for Genus Vitis Based on Restriction Site-Associated DNA Sequencing. Phytopathology 2021, 111, 659–670. [Google Scholar] [CrossRef]
- Su, K.; Xing, H.; Guo, Y.; Zhao, F.; Liu, Z.; Li, K.; Li, Y.; Guo, X. High-density genetic linkage map construction and cane cold hardiness QTL mapping for Vitis based on restriction site-associated DNA sequencing. BMC Genom. 2020, 21, 419. [Google Scholar] [CrossRef]
- Reshef, N.; Karn, A.; Manns, D.C.; Mansfield, A.K.; Cadle-Davidson, L.; Reisch, B.; Sacks, G.L. Stable QTL for malate levels in ripe fruit and their transferability across Vitis species. Hortic. Res. 2022, 9, uhac009. [Google Scholar] [CrossRef]
- Wang, H.; Yan, A.; Sun, L.; Zhang, G.; Wang, X.; Ren, J.; Xu, H. Novel stable QTLs identification for berry quality traits based on high-density genetic linkage map construction in table grape. BMC Plant. Biol. 2020, 20, 411. [Google Scholar] [CrossRef] [PubMed]
- Fu, P.; Wu, W.; Lai, G.; Li, R.; Peng, Y.; Yang, B.; Wang, B.; Yin, L.; Qu, J.; Song, S.; et al. Identifying Plasmopara viticola resistance loci in grapevine (Vitis amurensis) via genotyping-by-sequencing-based QTL mapping. PPB 2020, 154, 75–84. [Google Scholar] [CrossRef]
- Fehér, A. Callus, dedifferentiation, totipotency, somatic embryogenesis: What these terms mean in the era of molecular plant biology? Front. Plant. Sci. 2019, 10, 536. [Google Scholar] [CrossRef] [PubMed]
- Nuzzo, F.; Gambino, G.; Perrone, I. Unlocking grapevine in vitro regeneration: Issues and perspectives for genetic improvement and functional genomic studies. Plant. Physiol. Biochem. 2022, 193, 99–109. [Google Scholar] [CrossRef]
- Butiuc-Keul, A.; Coste, A.; Oltean, B.; Crăciunaş, C.; Halmagyi, A.; Deliu, C.; Farago, M.; Iliescu, M.; Iuoraş, R. In vitro clonal propagation of several grapevine cultivars. Acta Hortic. 2009, 843, 151–156. [Google Scholar] [CrossRef]
- Butiuc-Keul, A.; Coste, A.; Crăciunaş, C. Molecular characterization and in vitro preservation of some grapevine cultivars. Rom. Biotechnol. Lett. 2011, 16, 6226–6233. [Google Scholar]
- Krishna, H.; Alizadeh, M.; Singh, D.; Singh, U.; Chauhan, N.; Eftekhari, M.; Sadh, R.K. Somaclonal variations and their applications in horticultural crops improvement. 3 Biotech 2016, 6, 54. [Google Scholar] [CrossRef] [PubMed]
- Atak, A. New Perspectives in Grapevine (Vitis spp.) Breeding. In Plant Breeding-New Perspectives; Wang, H., Ed.; IntechOpen: London, UK, 2022; Available online: https://www.intechopen.com/online-first/82151 (accessed on 15 November 2022). [CrossRef]
- Mukherjee, P.; Husain, N.; Misra, S.C.; Rao, V.S. In vitro propagation of a grape rootstock, deGrasset Vitis champinii Planch. Effects of medium compositions and plant growth regulators. Sci. Hortic. 2010, 126, 13–19. [Google Scholar] [CrossRef]
- Yerbolova, L.S.; Ryabushkina, N.A.; Oleichenko, S.N.; Kampitova, G.A.; Galiakparov, N. The effect of growth regulators on in vitro culture of some Vitis vinifera L. cultivars. World Appl. Sci. J. 2013, 23, 76–80. [Google Scholar] [CrossRef]
- Capriotti, L.; Baraldi, E.; Mezzetti, B.; Limera, C.; Sabbadini, S. Biotechnological approaches: Gene overexpression, gene silencing, and genome editing to control fungal and oomycete diseases in grapevine. Int. J. Molec. Sci. 2020, 21, 5701. [Google Scholar] [CrossRef]
- Nicholson, K.L.; Tarlyn, N.; Armour, T.; Swanson, M.E.; Dhingra, A. Effect of phyllotactic position and cultural treatments toward successful direct shoot organogenesis in dwarf ‘Pixie’ grapevine (Vitis vinifera L.). Plant. Cell Tiss. Organ. Cult. 2012, 111, 123–129. [Google Scholar] [CrossRef]
- Zhang, P.; Yu, Z.Y.; Cheng, Z.M.; Zhang, Z.; Tao, J.M. In vitro explants regeneration of the grape ‘Wink’ (Vitis vinifera L. ‘Wink’). J. Plant Breed. Crop Sci. 2011, 3, 276–282. [Google Scholar]
- Park, H.J.; Lee, H.R.; Pyee, J.; Cha, H.C. Regeneration of grape (Vitis labruscana cv. Kyoho) by shoot-tip culture. J. Plant. Biol. 2001, 44, 185–192. [Google Scholar] [CrossRef]
- Pool, R.M. Effect of cytokinin on in vitro development of ‘concord’ flowers. Am. J. Enol. Vitic. 1975, 26, 43–46. [Google Scholar]
- Lilov, D.; Isvorska, N. Flower bud initiation from isolated meristem tissues of grapevine tendrils. Fisiol. Rast. 1978, 4, 73–78. [Google Scholar]
- Barreto, M.S. In Vitro Plant Regeneration and Transformation Studies in Grape (Vitis vinifera L.). Ph.D. Thesis, University of Pune, Pune, India.
- Torregrosa, L. Culture in vitro et Transformation Genetique de la Vigne. These, Ecole Nationale Superieure Agronomique de Montpellier, France, 1994. Available online: https://www.theses.fr/1994ENSA0024 (accessed on 12 September 2022).
- Torregrosa, L.; Bouquet, A. Agrobacterium tumefaciens and A. rhizogenes-rhizogenes cotransformation to obtain grapevine hair roots producing coat protin of grapevine chrome mosaic nepo virus. Plant. Cell Tiss. Org. Cult. 1997, 49, 59–63. [Google Scholar] [CrossRef]
- Loubser, J.T.; Meyer, A.J. Dual cultures of Meloidogyne javanica and grapevine rootstocks on artificial media. S. Afr. J. Enol. Vitic. 1990, 11, 42–45. [Google Scholar] [CrossRef][Green Version]
- Bavaresco, L.; Walker, M.A. Techniques for successfully establishing Xiphinema index in dual culture with grapes. Am. J. Enol. Vitic. 1994, 45, 273–277. [Google Scholar]
- Forneck, A.; Merkt, N.; Blaich, R. A tripartite asceptic culture system for grapes (Vitis spp.), phylloxera (Daktulosphaera vitifoliae) and mites (Tarsanemus sp.). Vitis 1998, 37, 95–96. [Google Scholar]
- Kellow, A.V. A Study of The Interaction between Susceptible and Resistant Grapevine and Phylloxera. Ph.D. Thesis, Natural and Agricultural Resource Sciences, Department of Horitculture, Vitculture and Oenology, The University of Adelaide, Adelaide, Australia, June 2000. [Google Scholar]
- Saygaç, S.; Önder, S. Effects of shoot tip size on in vitro regeneration and virus elimination of grapevine cv. Superior Seedless. Plant. Prot. Bull. 2021, 61, 5–9. [Google Scholar] [CrossRef]
- Mourad, A. Elimination of grapevine bois noir phytoplasma by tissue culture coupled or not with heat therapy or hot water treatment. Adv. Crop. Sci. Technol. 2013, 1, 1–4. [Google Scholar] [CrossRef]
- Miljanić, V.; Rusjan, D.; Škvarč, A.; Chatelet, P.; Štajner, N. Elimination of eight viruses and two viroids from preclonal candidates of six grapevine varieties (Vitis vinifera L.) through in vivo thermotherapy and in vitro meristem tip micrografting. Plants 2022, 11, 1064. [Google Scholar] [CrossRef] [PubMed]
- Malenica, N.; Jagić, M.; Pavletić, B.; Bauer, N.; Vončina, D.; Zdunić, G.; Levanić, D.L. Somatic embryogenesis as a tool for virus elimination in Croatian indigenous grapevine cultivars. Acta Bot. Croat. 2020, 79, 26–34. [Google Scholar] [CrossRef]
- Pérez-Núñez, M.T.; Chan, J.L.; Sáenz, L.; González, T.; Verdeil, J.L.; Oropeza, C. Improved somatic embryogenesis from Cocos nucifera (L.) plumule explants. Vitr. Cell. Dev. Biol. Plant. 2006, 42, 37–43. [Google Scholar] [CrossRef]
- Forleo, L.R.; D’Amico, M.; Basile, T.; Marsico, A.D.; Cardone, M.F.; Maggiolini, F.A.M.; Velasco, R.; Bergamini, C. Somatic embryogenesis in Vitis for genome editing: Optimization of protocols for recalcitrant genotypes. Horticulturae 2021, 7, 511. [Google Scholar] [CrossRef]
- Dal Santo, S.; De Paoli, E.; Pagliarani, C.; Amato, A.; Celii, M.; Boccacci, P.; Zenoni, S.; Gambino, G.; Perrone, I. Stress responses and epigenomic instability mark the loss of somatic embryogenesis competence in grapevine. Plant. Physiol. 2022, 188, 490–508. [Google Scholar] [CrossRef]
- Capriotti, L.; Limera, C.; Mezzetti, B.; Ricci, A.; Sabbadini, S. From induction to embryo proliferation: Improved somatic embryogenesis protocol in grapevine for Italian cultivars and hybrid Vitis rootstocks. Plant. Cell Tiss Organ. Cult. 2022, 151, 221–233. [Google Scholar] [CrossRef]
- Horstman, A.; Bemer, M.; Boutilier, K. A transcriptional view on somatic embryogenesis. Regeneration 2017, 4, 201–216. [Google Scholar] [CrossRef]
- Ghadirzadeh-Khorzoghi, E.; Jahanbakhshian-Davaran, Z.; Seyedi, S.M. Direct somatic embryogenesis of drought resistance pistachio (Pistacia vera L.) and expression analysis of somatic embryogenesis-related genes. S. Afr. J. Bot. 2019, 121, 558–567. [Google Scholar] [CrossRef]
- Catalano, C.; Abbate, L.; Motisi, A.; Crucitti, D.; Cangelosi, V.; Pisciotta, A.; Di Lorenzo, R.; Carimi, F.; Carra, A. Autotetraploid emergence via somatic embryogenesis in Vitis vinifera induces marked morphological changes in shoots, mature leaves, and stomata. Cells 2021, 10, 1336. [Google Scholar] [CrossRef] [PubMed]
- Corredoira, E.; Merkle, S.A.; Martínez, M.T.; Toribio, M.; Canhoto, J.M.; Correia, S.I.; Ballester, A.; Vieitez, A. Non-zygotic embryogenesis in hardwood species. Crit Rev. Plant. Sci. 2019, 38, 29–97. [Google Scholar] [CrossRef]
- Maillot, P.; Lebel, S.; Schellenbaum, P.; Jacques, A.; Walter, B. Differential regulation of SERK, LEC1-like and pathogenesis-related genes during indirect secondary somatic embryogenesis in grapevine. Plant. Physiol. Biochem. 2009, 47, 743–752. [Google Scholar] [CrossRef]
- Ricci, A.; Sabbadini, S.; Prieto, H.; Padilla, I.M.; Dardick, C.; Li, Z.; Scorza, R.; Limera, C.; Mezzetti, B.; Perez-Jimenez, M.; et al. Genetic transformation in peach (Prunus persica L.): Challenges and ways forward. Plants 2020, 9, 971. [Google Scholar] [CrossRef]
- Phillips, R.L.; Kaeppler, S.M.; Olhoft, P. Genetic instability of plant tissue cultures: Breakdown of normal controls. Proc. Natl. Acad. Sci. USA 1994, 91, 5222–5226. [Google Scholar] [CrossRef] [PubMed]
- Muler, E.; Brown, P.T.H.; Hartke, S.; Lorz, H. DNA variation in tissue-culture-derived rice plants. Theor. Appl. Genet. 1990, 80, 673–679. [Google Scholar] [CrossRef]
- Kuksova, V.B.; Piven, N.M.; Gleba, Y.Y. Somaclonal variation and in vitro induced mutagenesis in grapevine. Plant. Cell Tiss. Organ. Cult. 1997, 49, 17–27. [Google Scholar] [CrossRef]
- Palombi, M.A.; Damiano, C. Comparison between RAPD and SSR molecular markers in detecting genetic variation in kiwifruit (Actinidia deliciosa A. Chev). Plant. Cell Rep. 2002, 20, 1061–1066. [Google Scholar] [CrossRef]
- Kaeppler, S.M.; Phillips, R.L. DNA methylation and tissue culture-induced variation in plants. Vitr. Cell Dev. Biol. Plant. 1993, 29, 125–130. [Google Scholar] [CrossRef]
- Nivas, S.K.D.; Souza, L. Genetic fidelity in micropropagated plantlets of Anacardium occidentale L. (Cashew) an important fruit tree. Int. J. Sci. Res. 2014, 3, 2142–2146. [Google Scholar]
- Smulders, M.; de Klerk, G. Epigenetics in plant tissue culture. Plant. Growth Regul. 2011, 63, 137–146. [Google Scholar] [CrossRef]
- Prado, M.J.; Rodriguez, E.; Rey, L.; González, M.V.; Santos, C.; Rey, M. Detection of somaclonal variants in somatic embryogenesis-regenerated plants of Vitis vinifera by flow cytometry and microsatellite markers. Plant. Cell Tiss. Organ. Cult. 2010, 103, 49–59. [Google Scholar] [CrossRef]
- Marcotrigiano, M.; Bernatzky, R. Arrangement of cell layers in the shoot apical meristems of periclinal chimeras influences cell fate. Plant. J. 1995, 7, 193–202. [Google Scholar] [CrossRef]
- Burge, G.K.; Morgan, E.R.; Seelye, J.F. Opportunities for synthetic plant chimeral breeding: Past and future. Plant. Cell Tiss. Organ. Cult. 2002, 70, 13–21. [Google Scholar] [CrossRef]
- Franks, T.; Botta, R.; Thomas, M.R.; Franks, J. Chimerism in grapevines: Implications for cultivar identity, ancestry and genetic improvement. Theor. Appl. Genet. 2002, 104, 192–199. [Google Scholar] [CrossRef] [PubMed]
- Bertsch, C.; Kieffer, F.; Maillot, P.; Farine, S.; Butterlin, G.; Merdinoglu, D.; Walter, B. Genetic chimerism of Vitis vinifera cv. Chardonnay 96 is maintained through organogenesis but not somatic embryogenesis. BMC Plant. Biol. 2005, 5, 20. [Google Scholar] [CrossRef]
- Riaz, S.; Garrison, K.E.; Dangl, G.S.; Boursiquot, J.M.; Meredith, C.P. Genetic divergence and chimerism within ancient asexually propagated winegrape cultivars. J. Amer. Soc. Hort. Sci. 2002, 127, 508–514. [Google Scholar] [CrossRef][Green Version]
- Quackenbush, J.; Liang, F.; Holt, I.; Pertea, G.; Upton, J. The TIGR gene indices: Reconstruction and representation of expressed gene sequences. Nucleic Acids Res. 2000, 28, 141–145. [Google Scholar] [CrossRef][Green Version]
- Siemann, E.; Creasy, L. Concentration of the phytoalexin resveratrol in wine. Am. J. Enol. Viticult. 1992, 43, 49–52. [Google Scholar]
- Sotheeswaran, S.; Pasupathy, V. Distribution of resveratrol oligomers in plants. Photochemistry 1993, 32, 1083–1092. [Google Scholar] [CrossRef]
- Kikkert, J.K.; Ali, G.S.; Wallace, P.G.; Reisch, B.; Reustle, G.M. Expression of fungal chitinase in Vitis vinifera L. ‘Merlot’ and ‘Chardonnay’ plants produced by biolistic information. Acta Hortic. 2000, 528, 297–303. [Google Scholar] [CrossRef]
- Tamaoki, M.; Imai, H.; Takahashi, H.; Toda, Y.; Niwa, Y.; Nakajima, N.; Aono, M.; Kubo, A.; Saji, H. Development of visible markers for transgenic plants and their availability for environmental risk assessment. Z. Für Nat. 2006, 61, 377–386. [Google Scholar] [CrossRef] [PubMed]
- Braidot, E.; Zancani, M.; Petrussa, E.; Peresson, C.; Bertolini, A.; Patui, S.; Macri, F.; Vianello, A. Transport and accumulation of flavonoids in grapevine (Vitis vinifera L.). Plant. Signal. Behav. 2008, 3, 626–632. [Google Scholar] [CrossRef] [PubMed]
- Basha, S.M.; Musingo, M.; Colova, V.S. Compositional differences in the phenolics compounds of muscadine and bunch grape wines. Afr. J. Biotechnol. 2004, 10, 523–528. [Google Scholar] [CrossRef]
- INRA-CNRGV The French Plant Genomic Resource Center. Available online: http://cnrgv.toulouse.inra.fr (accessed on 10 October 2022).
- Huang, X.; Wei, X.; Sang, T.; Zhao, Q.; Feng, Q.; Zhao, Y.; Li, C.; Zhy, C.; Lu, T.; Zhang, Z.; et al. Genome-wide association studies of 14 agronomic traits in rice landraces. Nat. Genet. 2010, 42, 961–967. [Google Scholar] [CrossRef] [PubMed]
- Meuwissen, T.H.E.; Hayes, B.J.; Goddard, M.E. Prediction of Total Genetic Value Using Genome-Wide Dense Marker Maps. Genetics 2001, 157, 1819–1829. [Google Scholar] [CrossRef]
- Jannink, J.-L.; Lorenz, A.J.; Iwata, H. Genomic selection in plant breeding: From theory to practice. Brief. Funct. Genom. 2010, 9, 166–177. [Google Scholar] [CrossRef]
- Nakaya, A.; Isobe, S.N. Will genomic selection be a practical method for plant breeding? Ann. Bot. 2012, 110, 1303–1316. [Google Scholar] [CrossRef]
- Fodor, A.; Segura, V.; Denis, M.; Neuenschwander, S.; Fournier-Level, A.; Chatelet, P.; Homa, F.A.; Lacombe, T.; This, P.; Le Cunff, L. Genome-wide prediction methods in highly diverse and heterozygous species: Proof-of-concept through simulation in grapevine. PLoS ONE 2014, 9, e110436. [Google Scholar] [CrossRef]
- Wang, M.; Jiang, N.; Jia, T.; Leach, L.; Cockram, J.; Waugh, R.; Ramsay, L.; Thomas, B.; Luo, Z. Genome-wide association mapping of agronomic and morphologic traits in highly structured populations of barley cultivars. Theor. Appl. Genet. 2012, 124, 233–246. [Google Scholar] [CrossRef]
- Laucou, V.; Lacombe, T.; Dechesne, F.; Siret, R.; Bruno, J.-P.; Dessup, M.; Dessup, T.; Ortigosa, P.; Parra, P.; Roux, C. High throughput analysis of grape genetic diversity as a tool for germplasm collection management. Theor. Appl. Genet. 2011, 122, 1233–1245. [Google Scholar] [CrossRef] [PubMed]
- Myles, S.; Chia, J.-M.; Hurwitz, B.; Simon, C.; Zhong, G.Y.; Buckler, E.; Ware, D. Rapid Genomic Characterization of the Genus Vitis. PLoS ONE 2010, 5, e8219. [Google Scholar] [CrossRef] [PubMed]
- Bowers, J.; Boursiquot, J.M.; This, P.; Chu, K.; Johansson, H.; Meredith, C. Historical Genetics: The Parentage of Chardonnay, Gamay, and Other Wine Grapes of Northeastern France. Science 1999, 285, 1562–1565. [Google Scholar] [CrossRef]
- Boursiquot, J.-M.; Lacombe, T.; Laucou, V.; Julliard, S.; Perrin, F.-X.; Lanier, N.; Legrand, D.; Meredith, C.; This, P. Parentage of Merlot and related winegrape cultivars of southwestern France: Discovery of the missing link. Aust. J. Grape Wine Res. 2009, 15, 144–155. [Google Scholar] [CrossRef]
- Myles, S.; Boyko, A.R.; Owens, C.L.; Brown, P.J.; Grassi, F.; Aradhya, M.K.; Prins, B.; Reynolds, A.; Chia, I.-M.; Ware, D.; et al. Genetic Structure and Domestication History of the Grape. Proc. Natl. Acad. Sci. USA 2011, 108, 3530–3535. [Google Scholar] [CrossRef] [PubMed]
- Lacombe, T.; Boursiquot, J.-M.; Laucou, V.; Di Vecchi-Staraz, M.; Péros, J.-P.; This, P. Large-scale parentage analysis in an extended set of grapevine cultivars (Vitis vinifera L.). Theor. Appl. Genet. 2012, 126, 1–14. [Google Scholar] [CrossRef]
- Hannah, L.; Roehrdanz, P.R.; Ikegami, M.; Shepard, A.V.; Shaw, M.R.; Tabor, G.; Zhi, L.; Marquet, P.A.; Hijmans, R.J. Climate change, wine, and conservation. Proc. Natl. Acad. Sci. USA 2013, 110, 6907–6912. [Google Scholar] [CrossRef] [PubMed]
- Moriondo, M.; Jones, G.V.; Bois, B.; Dibari, C.; Ferrise, R.; Trombi, G.; Bindi, M. Projected shifts of wine regions in response to climate change. Clim. Change 2013, 119, 825–839. [Google Scholar] [CrossRef]
- Gray, D.J.; Jayasankar, S.; Li, Z.T. Vitaceae (Grape Vitis spp.). In Biotechnology of Fruit and Nut Crops. Biotechnology in Agriculture Series; No. 29; Litz, R.E., Ed.; CAB International: Wallingford, UK, 2005; Volume Chapter 22, pp. 672–706. [Google Scholar]
- Maraš, V.; Tello, J.; Gazivoda, A.; Mugoša, M.; Perišić, M.; Raičević, J.; Štajner, N.; Ocete, R.; Božović, V.; Popović, T.; et al. Population genetic analysis in old Montenegrin vineyards reveals ancient ways currently active to generate diversity in Vitis vinifera. Sci. Rep. 2020, 10, 15000. [Google Scholar] [CrossRef] [PubMed]
- Čuš, F.; Cesnik, H.B.; Bolta, S.V.; Gregorcic, A. Pesticide residues and microbio-logical quality of bottled wines. Food Control. 2010, 21, 150–154. [Google Scholar] [CrossRef]
- Pretorius, I.S.; Høj, P.B. Grape and wine biotechnology: Challenges, opportunities and potential benefits. Aust. J. Grape Wine Res. 2006, 11, 83–108. [Google Scholar] [CrossRef]
- Gray, D.J.; Li, Z.T.; Dhekney, S.A. Precision breeding of grapevine (Vitis vinifera L.) for improved traits. Plant. Sci. 2014, 228, 3–10. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.T.; Dhekney, S.A.; Dutt, M.; Van Aman, M.; Tattersall, J.; Kelley, K.T.; Gray, D.J. Optimizing Agrobacterium-mediated transformation of grapevine. Vitr. Cell. Dev. Biol. Plant. 2006, 42, 220–227. [Google Scholar] [CrossRef]
- Li, Z.T.; Dhekney, S.A.; Dutt, M.; Gray, D.J. An improved protocol for Agrobacterium-mediated transformation of grapevine (Vitis vinifera L.). Plant. Cell Tiss. Organ. Cult. 2008, 93, 311–321. [Google Scholar] [CrossRef]
- Torregrosa, L.; Rienth, M.; Luchaire, N.; Novelli, F.; Bigard, A.; Chatbanyong, R.; Lopez, G.; Farnos, M.; Roux, C.; Adivèze, A.; et al. The Microvine, A Biological Model, Very Versatile and Efficient to Boost Grapevine Research in Physiology and Genetics. In Proceedings of the 39th OIV Meeting, Bento Gonzalvez, Brazil, 24–28 October 2016. [Google Scholar]
- Dalla Costa, L.; Piazza, S.; Campa, M.; Flachowsky, H.; Hanke, M.V.; Malnoy, M. Efficient heat-shock removal of the selectable marker gene in genetically modified grapevine. Plant. Cell Tiss. Organ. Cult. 2016, 124, 471–481. [Google Scholar] [CrossRef]
- Muruganantham, M.; Moskovitz, Y.; Haviv, S.; Horesh, T.; Fenigstein, A.; Preez, J.D.; Stephan, D.; Burger, J.T.; Mawassi, M. Grapevine virusA-mediated gene silencing in Nicotiana benthamiana and Vitis vinifera. J. Virol. Methods 2009, 155, 167–174. [Google Scholar] [CrossRef] [PubMed]
- Kurth, E.G.; Peremyslov, V.V.; Prokhnevsky, A.I.; Kasschau, K.D.; Miller, M.; Carrington, J.C.; Dolja, V.V. Virus-derived gene expression and RNA interference vector for grapevine. J. Virol. 2012, 86, 6002–6009. [Google Scholar] [CrossRef] [PubMed]
- Meng, B.; Venkataraman, S.; Li, C.; Wang, W.; Dayan-Glick, C.; Mawassi, M. Construction and biological activities of the first infectious cDNA clones of the genus Foveavirus. Virology 2013, 435, 453–462. [Google Scholar] [CrossRef]
- Wood, K. Marker proteins for gene expression. Curr. Opin. Biotechnol. 1995, 6, 50–58. [Google Scholar] [CrossRef]
- Rosellini, D. Selectable markers and reporter genes. A well furnished tool-box for plant science and genetic engineering. Crit. Rev. Plant. Sci. 2012, 31, 401–453. [Google Scholar] [CrossRef]
- Dutt, M.; Li, Z.T.; Dhekney, S.A.; Gray, D.J. A co-transformation system to produce transgenic grapevines free of marker genes. Plant. Sci. 2008, 175, 423–430. [Google Scholar] [CrossRef]
- Li, Z.T.; Dhekney, S.A.; Gray, D.J. Use of the VvMybA1 gene for non-destructive quantification of promoter activity via color histogram analysis in grapevine (Vitis vinifera) and tobacco. Transgenic Res. 2011, 20, 1087–1097. [Google Scholar] [CrossRef]
- Dutt, M.; Li, Z.T.; Gray, D.J.; Gmitter, F.; Grosser, J.W. Purple citrus? Utilization of a Myb-related transcription factor gene for Anthocyanin production. AMER Soc. Hortic. Sci. 2013, 48, 10–11. [Google Scholar]
- Li, Z.T.; Jayasankar, S.; Gray, D.J. Expression of a bifunctional green fluorescent protein (GFP) fusion marker under the control of three constitutive promoters and enhanced derivatives in transgenic grape (Vitis vinifera). Plant. Sci. 2001, 160, 877–887. [Google Scholar] [CrossRef] [PubMed]
- Conn, S.; Zhang, W.; Franco, C. Anthocyanic vacuolar inclusions (AVIs) selectively bind acylated anthocyanins in Vitis vinifera L. (grapevine) suspension culture. Biotechnol. Lett. 2003, 25, 835–839. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.T.; Kim, K.-H.; Jasinski, J.R.; Creech, M.R.; Gray, D.J. Large-scale characterization of promoters from grapevine (Vitis spp.) using quantitative anthocyanin and GUS assay systems. Plant. Sci. 2012, 196, 132–142. [Google Scholar] [CrossRef]
- Pilati, S.; Perazzolli, M.; Malossini, A.; Cestaro, A.; Damattè, L.; Fontana, P.; Dal Ri, A.; Viola, R.; Velasco, R.; Moser, C. Genome-wide transcriptional analysis of grapevine berry ripening reveals a set of genes similarly modulated during three seasons and the occurrence of an oxidative burst at veraison. BMC Genom. 2007, 8, 428. [Google Scholar] [CrossRef]
- Mezzetti, B.; Pandolfini, T.; Navacchi, O.; Landi, L. Genetic transformation of Vitis vinifera via organogenesis. BMC Biotechnol. 2002, 2, 18. [Google Scholar] [CrossRef]
- Xie, X.; Agüero, C.B.; Wang, Y.; Andrew Walker, M. Genetic transformation of grape varieties and rootstocks via organogenesis. Plant. Cell Tiss. Organ. Cult. 2016, 126, 541–552. [Google Scholar] [CrossRef]
- Sabbadini, S.; Capriotti, L.; Molesini, B.; Pandolfini, T.; Navacchi, O.; Limera, C.; Ricci, A.; Mezzetti, B. Comparison of regeneration capacity and Agrobacterium-me.ediated cell transformation efficiency of different cultivars and rootstocks of Vitis spp. via organogenesis. Sci. Rep. 2019, 9, 582. [Google Scholar] [CrossRef]
- de Carvalho, D.C.; da Silva, A.L.L.; Schuck, M.R.; Purcino, M.; Tanno, G.N.; Biasi, L.A. Fox grape cv. Bordô (Vitis labrusca L.) and grapevine cv. Chardonnay (Vitis vinifera L.) cultivated in vitro under different carbohydrates, amino acids and 6-benzylaminopurine levels. Brazil. Arch. Biol. Technol. 2013, 56, 191–201. [Google Scholar] [CrossRef]
- Colby, S.M.; Juncosa, A.M.; Meredith, C.P. Cellular differences in Agrobacterium susceptibility and regenerative capacity restrict the development of transgenic grapevines. J. Amer. Soc. Hort. Sci. 1991, 116, 356–361. [Google Scholar] [CrossRef]
- Maillot, P.; Deglène-Benbrahim, L.; Walter, B. Efficient somatic embryogenesis from meristematic explants in grapevine (Vitis vinifera L.) cv. Chardonnay: An improved protocol. Trees 2016, 30, 1377–1387. [Google Scholar] [CrossRef]
- Zhou, Q.; Dai, L.; Cheng, S.; He, J.; Wang, D.; Zhang, J.; Wang, Y. A circulatory system useful both for long-term somatic embryogenesis and genetic transformation in Vitis vinifera L. cv. Thompson Seedless. Plant. Cell Tiss. Organ. Cult. 2014, 118, 157–168. [Google Scholar] [CrossRef]
- Saporta, R.; San Pedro, T.; Gisbert, C. Attempts at grapevine (Vitis vinifera L.) breeding through genetic transformation: The main limiting factors. Vitis 2016, 55, 173–186. [Google Scholar] [CrossRef]
- Wang, X.; Tu, M.; Wang, D.; Liu, J.; Li, Y.; Li, Z.; Wang, Y.; Wang, X. CRISPR/Cas9-mediated efficient targeted mutagenesis in grape in the first generation. Plant. Biotechnol. J. 2018, 16, 844–855. [Google Scholar] [CrossRef]
- Carra, A.; Sajeva, M.; Abbate, L.; Siragusa, M.; Pathirana, R.; Carimi, F. Factors affecting somatic embryogenesis in eight Italian grapevine cultivars and the genetic stability of embryo-derived regenerants as assessed by molecular markers. Sci. Hort. 2016, 204, 123–127. [Google Scholar] [CrossRef]
- de Carvalho, D.C.; da Silva, A.L.L.; Tanno, G.N.; Purcino, M.; Biasi, L.A. Organogenesis from leaf segments and internodes of grapevine cv. Merlot. Ciência Agrotecnol. 2011, 35, 108–114. [Google Scholar] [CrossRef]
- Perrin, M.; Martin, D.; Joly, D.; Demangeat, G.; This, P.; Masson, J.E. Medium-dependent response of grapevine somatic embryogenic cells. Plant. Sci. 2001, 161, 107–116. [Google Scholar] [CrossRef]
- Jelly, N.S.; Valat, L.; Walter, B.; Maillot, P. Transient expression assays in grapevine: A step towards genetic improvement. Plant. Biotechnol. J. 2014, 12, 1231–1245. [Google Scholar] [CrossRef]
- Torregrosa, L.; Verries, C.; Tesniere, C. Grapevine (Vitis vinifera L.) promoter analysis by biolistic-mediated transient transformation of cell suspensions. Vitis 2002, 41, 27–32. [Google Scholar] [CrossRef]
- Verries, C.; Pradal, M.; Chatelet, P.; Torregrosa, L.; Tesniere, C. Isolation and analysis of the promoter of VvAdh2, a grapevine Vitis vinifera L. ripening-related gene. Plant. Sci. 2004, 167, 1067–1074. [Google Scholar] [CrossRef]
- Harris, N.N.; Luczo, J.M.; Robinson, S.P.; Walker, A.R. Transcriptional regulation of the three grapevine chalcone synthase genes and their role in flavonoid synthesis in Shiraz. Aus. J. Grape Wine Res. 2013, 19, 221–229. [Google Scholar] [CrossRef]
- Deluc, L.; Bogs, J.; Walker, A.R.; Ferrier, T.; Decendit, A.; Merillon, J.M.; Robinson, S.P.; Barrieu, F. The transcription factor VvMYB5b contributes to the regulation of anthocyanin and proanthocyanidin biosynthesis in developing grape berries. Plant. Physiol. 2008, 147, 2041–2053. [Google Scholar] [CrossRef] [PubMed]
- Walker, A.R.; Lee, E.; Bogs, J.; McDavid, D.A.; Thomas, M.R.; Robinson, S.P. White grapes arose through the mutation of two similar and adjacent regulatory genes. Plant. J. 2007, 49, 772–785. [Google Scholar] [CrossRef]
- Czemmel, S.; Stracke, R.; Weisshaar, B.; Cordon, N.; Harris, N.N.; Walker, A.R.; Robinson, S.P.; Bogs, J. The grapevine R2R3-MYB transcription factor VvMYBF1 regulates flavonol synthesis in developing grape berries. Plant. Physiol. 2009, 151, 1513–1530. [Google Scholar] [CrossRef]
- Bogs, J.; Jaffe, F.W.; Takos, A.M.; Walker, A.R.; Robinson, S.P. The grapevine transcription factor VvMYBPA1 regulates proanthocyanidin synthesis during fruit development. Plant. Physiol. 2007, 143, 1347–1361. [Google Scholar] [CrossRef]
- Hichri, I.; Heppel, S.C.; Pillet, J.; Leon, C.; Czemmel, S.; Delrot, S.; Lauvergeat, V.; Bogs, J. The basic helix-loop-helix transcription factor MYC1 is involved in the regulation of the flavonoid biosynthesis pathway in grapevine. Mol. Plant. 2010, 3, 509–523. [Google Scholar] [CrossRef]
- Höll, J.; Vannozzi, A.; Czemmel, S.; D’Onofrio, C.; Walker, A.R.; Rausch, T.; Lucchin, M.; Boss, P.K.; Dry, I.B.; Bogs, J. The R2R3-MYB transcription factors MYB14 and MYB15 regulate stilbene biosynthesis in Vitis vinifera. Plant. Cell 2013, 25, 4135–4414. [Google Scholar] [CrossRef]
- Joubert, D.A.; de Lorenzo, G.; Vivier, M.A. Regulation of the grapevine polygalacturonase-inhibiting protein encoding gene: Expression pattern, induction profile and promoter analysis. J. Plant. Res. 2013, 126, 267–281. [Google Scholar] [CrossRef]
- Saumonneau, A.; Laloi, M.; Lallemand, M.; Rabot, A.; Atanassova, R. Dissection of the transcriptional regulation of grape ASR and response to glucose and abscisic acid. J. Exp. Bot. 2012, 63, 1495–1510. [Google Scholar] [CrossRef] [PubMed]
- Marchive, C.; Leon, C.; Kappel, C.; Coutos-Thevenot, P.; Corio-Costet, M.F.; Delrot, S.; Lauvergeat, V. Over-expression of VvWRKY1 in grapevines induces expression of jasmonic acid pathway-related genes and confers higher tolerance to the downy mildew. PLoS ONE 2013, 8, e54185. [Google Scholar] [CrossRef]
- Saumonneau, A.; Agasse, A.; Bidoyen, M.T.; Lallemand, M.; Cantereau, A.; Medici, A.; Laloi, M.; Atanassova, R. Interaction of grape ASR proteins with a DREB transcription factor in the nucleus. FEBS Lett. 2008, 582, 3281–3287. [Google Scholar] [CrossRef] [PubMed]
- Urso, S.; Zottini, M.; Ruberti, C.; Schiavo, F.L.; Stanca, A.M.; Cattivelli, L.; Vale, G. An Agrobacterium tumefaciens-mediated gene silencing system for functional analysis in grapevine. Plant. Cell Tiss. Organ. Cult. 2013, 114, 49–60. [Google Scholar] [CrossRef]
- Visser, M.; Stephan, D.; Jaynes, J.M.; Burger, J.T. A transient expression assay for the in planta efficacy screening of an antimicrobial peptide against grapevine bacterial pathogens. Lett. Appl. Microbiol. 2012, 54, 543–551. [Google Scholar] [CrossRef] [PubMed]
- Bertazzon, N.; Raiola, A.; Castiglioni, C.; Gardiman, M.; Angelini, E.; Borgo, M.; Ferrari, S. Transient silencing of the grapevine gene VvPGIP1 by agroinfiltration with a construct for RNA interference. Plant. Cell Rep. 2012, 31, 133–143. [Google Scholar] [CrossRef]
- Santos-Rosa, M.; Poutaraud, A.; Merdinoglu, D.; Mestre, P. Development of a transient expression system in grapevine via agro-infiltration. Plant. Cell Rep. 2008, 27, 1053–1063. [Google Scholar] [CrossRef]
- Guan, X.; Zhao, H.; Xu, Y.; Wang, Y. Transient expression of glyoxal oxidase from the Chinese wild grape Vitis pseudoreticulata can suppress powdery mildew in a susceptible genotype. Protoplasma 2011, 248, 415–423. [Google Scholar] [CrossRef]
- He, M.; Xu, Y.; Cao, J.; Zhu, Z.; Jiao, Y.; Wang, Y.; Guan, X.; Yang, Y.; Xu, W.; Fu, Z. Subcellular localization and functional analyses of a PR10 protein gene from Vitis pseudoreticulata in response to Plasmopara viticola infection. Protoplasma 2013, 250, 129–140. [Google Scholar] [CrossRef]
- Xu, W.; Yu, Y.; Ding, J.; Hua, Z.; Wang, Y. Characterization of a novel stilbene synthase promoter involved in pathogen- and stress-inducible expression from Chinese wild Vitis pseudoreticulata. Planta 2010, 231, 475–487. [Google Scholar] [CrossRef]
- Xu, T.F.; Zhao, X.C.; Jiao, Y.T.; Wei, J.Y.; Wang, L.; Xu, Y. A pathogenesis related protein, VpPR-10.1, from Vitis pseudoreticulata: An insight of its mode of antifungal activity. PLoS ONE 2014, 9, e95102. [Google Scholar] [CrossRef] [PubMed]
- Le Henanff, G.; Heitz, T.; Mestre, P.; Mutterer, J.; Walter, B.; Chong, J. Characterization of Vitis vinifera NPR1 homologs involved in the regulation of pathogenesis-related gene expression. BMC Plant. Biol. 2009, 9, 54. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.P.; Peremyslov, V.V.; Medina, V.; Dolja, V.V. Tandem leader proteases of grapevine leafroll-associated virus-2: Host-specific functions in the infection cycle. Virology 2009, 383, 291–299. [Google Scholar] [CrossRef] [PubMed]
- Gollop, R.; Even, S.; Colova-Tsolova, V.; Perl, A. Expression of the grape dihydroflavonol reductase gene and analysis of its promoter region. J. Exp. Bot. 2002, 53, 1397–1409. [Google Scholar] [CrossRef]
- Jelly, N.S.; Schellenbaum, P.; Walter, B.; Maillot, P. Transient expression of artificial microRNAs targeting Grapevine fanleaf virus and evidence for RNA silencing in grapevine somatic embryos. Transgenic Res. 2012, 21, 1319–1327. [Google Scholar] [CrossRef]
- Li, Z.T.; Jayasankar, S.; Gray, D.J. Bi-directional duplex promoters with duplicated enhancers significantly increase transgene expression in grape and Tobacco. Transgenic Res. 2004, 13, 143–154. [Google Scholar] [CrossRef]
- Gutoranov, G.P.; Tsvetkov, I.J.; Colova-Tsolova, V.M.; Atanassov., A.I. Genetically engineered grapevines carrying GFLV coat protein and antifreeze genes. Agricult. Conspect. Sci. 2001, 66, 71–76. [Google Scholar]
- Mulwa, R.M.S.; Norton, M.A.; Farrand, S.K.; Skirvin, R.M. Agrobacterium-mediated transformation and regeneration of transgenic ‘Chancellor’ wine grape plants expressing the tfdA gene. Vitis 2007, 46, 110–115. [Google Scholar]
- Nirala, N.K.; Das, D.K.; Srivastava, P.S.; Sopory, S.K.; Upadhyaya, K.C. Expression of a rice chitinase gene enhances antifungal potential in transgenic grapevine (Vitis vinifera L.). Vitis 2010, 49, 181–187. [Google Scholar] [CrossRef]
- Dhekney, S.A.; Li, Z.T.; Gray, D.J. Grapevines engineered to express cisgenic Vitis vinifera thaumatin-like protein exhibit fungal disease resistance. Vitr. Cell. Dev. Biol. Plant. 2011, 47, 458–466. [Google Scholar] [CrossRef]
- Nookaraju, A.; Agrawal, D.C. Enhanced tolerance of transgenic grapevines expressing chitinase and β-1,3-glucanase genes to downy mildew. Plant. Cell Tiss. Organ. Cult. 2012, 111, 15–28. [Google Scholar] [CrossRef]
- Dai, L.; Zhou, Q.; Li, R.; Du, Y.; He, J.; Wang, D.; Cheng, S.; Zhang, J.; Wang, Y. Establishment of a picloram-induced somatic embryogenesis system in Vitis vinifera cv. Chardonnay and genetic transformation of a stilbene synthase gene from wild-growing Vitis species. Plant. Cell Tiss. Organ. Cult. 2015, 121, 397–412. [Google Scholar] [CrossRef]
- Li, Z.T.; Hopkins, D.L.; Gray, D.J. Overexpression of antimicrobial lytic peptides protects grapevine from Pierce’s disease under greenhouse but not field conditions. Transgenic Res. 2015, 24, 821–830. [Google Scholar] [CrossRef] [PubMed]
- Cheng, S.; Xie, X.; Xu, Y.; Zhang, C.; Wang, X.; Zhang, J.; Wang, Y. Genetic transformation of a fruit-specific, highly expressed stilbene synthase gene from Chinese wild Vitis quinquangularis. Planta 2016, 243, 1041–1053. [Google Scholar] [CrossRef] [PubMed]
- Dai, L.; Wang, D.; Xie, X.; Zhang, C.; Wang, X.; Xu, Y.; Wang, Y.; Zhang, J. The novel gene VpPR4-1 from Vitis pseudoreticulata increases powdery mildew resistance in transgenic Vitis vinifera L. Front. Plant. Sci. 2016, 7, 695. [Google Scholar] [CrossRef] [PubMed]
- Pessina, S.; Lenzi, L.; Perazzolli, M.; Campa, M.; Dalla Costa, L.; Urso, S.; Valè, G.; Salamini, F.; Velasco, R.; Malnoy, M. Knockdown of MLO genes reduces susceptibility to powdery mildew in grapevine. Hortic. Res. 2016, 3, 16016. [Google Scholar] [CrossRef]
- He, R.; Wu, J.; Zhang, Y.; Agüero, C.B.; Li, X.; Liu, S.; Wang, C.; Andrew Walker, M.; Lu, J. Overexpression of a thaumatin-like protein gene from Vitis amurensis improves downy mildew resistance in Vitis vinifera grapevine. Protoplasma 2017, 254, 1579–1589. [Google Scholar] [CrossRef]
- Jiao, L.; Zhang, Y.; Lu, J. Overexpression of a stressresponsive U-box protein gene VaPUB affects the accumulation of resistance related proteins in Vitis vinifera ‘Thompson Seedless’. Plant. Physiol. Biochem. 2017, 112, 53–63. [Google Scholar] [CrossRef]
- Ma, H.; Xiang, G.; Li, Z.; Wang, Y.; Dou, M.; Su, L.; Yin, X.; Liu, R.; Wang, Y.; Xu, Y. Grapevine VpPR10.1 functions in resistance to Plasmopara viticola through triggering a cell death-like defense response by interacting with VpVDAC3. Plant. Biotechnol. J. 2018, 16, 1488–1501. [Google Scholar] [CrossRef]
- Su, H.; Jiao, Y.T.; Wang, F.F.; Liu, Y.E.; Niu, W.L.; Liu, G.T.; Xu, Y. Overexpression of VpPR10.1 by an efficient transformation method enhances downy mildew resistance in V. vinifera. Plant. Cell Rep. 2018, 37, 819–832. [Google Scholar] [CrossRef]
- Soliman, H.I.A. Production of genetically modified grape (Vitis vinifera L.) plants. Int. J. Hortic. Agric. Food Sci. 2018, 2, 111–120. [Google Scholar] [CrossRef]
- Jiang, J.; Xi, H.; Dai, Z.; Lecourieux, F.; Yuan, L.; Liu, X.; Patra, B.; Wei, Y.; Li, S.; Wang, L. VvWRKY8 represses stilbene synthase genes through direct interaction with VvMYB14 to control resveratrol biosynthesis in grapevine. J. Exp. Bot. 2019, 70, 715–729. [Google Scholar] [CrossRef] [PubMed]
- Xu, W.; Ma, F.; Li, R.; Zhou, Q.; Yao, W.; Jiao, Y.; Zhang, C.; Zhang, J.; Wang, X.; Xu, Y.; et al. VpSTS29/STS2 enhances fungal tolerance in grapevine through a positive feedback loop. Plant. Cell Environ. 2019, 42, 2979–2998. [Google Scholar] [CrossRef] [PubMed]
- Tu, M.; Wang, X.; Yin, W.; Wang, Y.; Li, Y.; Zhang, G.; Li, Z.; Song, J.; Wang, X. Grapevine VlbZIP30 improves drought resistance by directly activating VvNAC17 and promoting lignin biosynthesis through the regulation of three peroxidase genes. Hortic. Res. 2020, 7, 150. [Google Scholar] [CrossRef]
- Carroll, D. Genome engineering with zinc-finger nucleases. Genetics 2011, 188, 773–782. [Google Scholar] [CrossRef]
- Mahfouz, M.M.; Li, L.; Shamimuzzaman, M.; Wibowo, A.; Fang, X.; Zhu, J.-K. De novo-engineered transcription activator-like effector (TALE) hybrid nuclease with novel DNA binding specificity creates double-strand breaks. Proc. Nat. Acad. Sci. USA 2011, 108, 2623–2628. [Google Scholar] [CrossRef]
- Li, L.; Piatek, M.J.; Atef, A.; Piatek, A.; Wibowo, A.; Fang, X.; Sabir, J.S.M.; Zhu, J.-K.; Mahfouz, M.M. Rapid and highly efficient construction of TALE-based transcriptional regulators and nucleases for genome modification. Plant. Molec. Biol. 2012, 78, 407–416. [Google Scholar] [CrossRef]
- Jinek, M.; Chylinski, K.; Fonfara, I.; Hauer, M.; Doudna, J.A.; Charpentier, E. Programmable dual-RNA–guided DNA endonuclease in adaptive bacterial immunity. Science 2012, 337, 816–821. [Google Scholar] [CrossRef]
- Kaul, T.; Sony, S.K.; Verma, R.; Motelb, K.F.A.; Prakash, A.T.; Eswaran, M.; Bharti, J.; Nehra, J.; Kaul, R. Revisiting CRISPR/Cas-mediated crop improvement: Special focus on nutrition. J. Biosci. 2020, 45, 1–37. [Google Scholar] [CrossRef]
- van der Oost, J.; Westra, E.R.; Jackson, R.N.; Wiedenheft, B. Unravelling the structural and mechanistic basis of CRISPR-Cas systems. Nat. Rev. Microbiol. 2014, 12, 479–492. [Google Scholar] [CrossRef]
- Xie, K.; Zhang, J.; Yang, Y. Genome-wide prediction of highly specific guide RNA spacers for CRISPR-Cas9-mediated genome editing in model plants and major crops. Mol. Plant. 2014, 7, 923–926. [Google Scholar] [CrossRef] [PubMed]
- Bortesi, L.; Fischer, R. The CRISPR/Cas9 system for plant genome editing and beyond. Biotechnol. Adv. 2015, 33, 41–52. [Google Scholar] [CrossRef] [PubMed]
- Butiuc-Keul, A.; Farkas, A.; Carpa, R.; Dobrota, C.T.; Iordache, D. Development of smart fruit crops by genome editing. Turk. J. Agric. For. 2022, 46, 129–140. [Google Scholar] [CrossRef]
- Cong, L.; Ran, F.A.; Cox, D.; Lin, S.; Barretto, R.; Habib, N.; Hsu, P.D.; Wu, X.; Jiang, W.; Marraffini, L.A.; et al. Multiplex genome engineering using CRISPR/Cas systems. Science 2013, 339, 819–823. [Google Scholar] [CrossRef]
- Hsu, P.D.; Scott, D.A.; Weinstein, J.A.; Ran, F.A.; Konermann, S.; Agarwala, V.; Li, Y.; Fine, E.J.; Wu, X.; Shalem, O.; et al. DNA targeting specificity of RNA-guided Cas9 nucleases. Nat. Biotechnol. 2013, 31, 827–832. [Google Scholar] [CrossRef]
- Carpa, R.; Remizovschi, A.; Culda, C.A.; Butiuc-Keul, A.L. Inherent and composite hydrogels as promising materials to limit antimicrobial resistance. Gels 2022, 8, 70. [Google Scholar] [CrossRef]
- Ali, Z.; Abulfaraj, A.; Idris, A.; Ali, S.; Tashkandi, M.; Mahfouz, M.M. CRISPR/ Cas9-mediated viral interference in plants. Genome Biol. 2015, 16, 238. [Google Scholar] [CrossRef]
- Malnoy, M.; Viola, R.; Jung, M.-H.; Koo, O.-J.; Kim, J.-S.; Velasco, R.; Kanchiswamy, C.N. DNAfree genetically edited grapevine and apple protoplast using CRISPR/Cas9 ribonucleoproteins. Front. Plant. Sci. 2016, 7, 1904. [Google Scholar] [CrossRef]
- Osakabe, Y.; Liang, Z.; Ren, C.; Nishitani, C.; Osakabe, K.; Wada, M.; Komori, S.; Malnoy, M.; Velasco, R.; Poli, M.; et al. CRISPR-Cas9-mediated genome editing in apple and grapevine. Nat. Prot. 2018, 13, 2844–2863. [Google Scholar] [CrossRef]
- Li, M.Y.; Jiao, Y.T.; Wang, Y.T.; Zhang, N.; Wang, B.B.; Liu, R.Q.; Yin, X.; Xu, Y.; Liu, G.T. CRISPR/Cas9-mediated VvPR4b editing decreases downy mildew resistance in grapevine (Vitis vinifera L.). Hortic. Res. 2020, 7, 149. [Google Scholar] [CrossRef]
- Wan, D.Y.; Guo, Y.; Cheng, Y.; Hu, Y.; Xiao, S.Y.; Wang, Y.J.; Wen, Y.Q. CRISPR/Cas9-mediated mutagenesis of VvMLO3 results in enhanced resistance to powdery mildew in grapevine (Vitis vinifera). Hortic. Res. 2020, 7, 116. [Google Scholar] [CrossRef] [PubMed]
- Ren, C.; Liu, X.; Zhang, Z.; Wang, Y.; Duan, W.; Li, S.; Liang, Z. CRISPR/Cas9-mediated efficient targeted mutagenesis in Chardonnay (Vitis vinifera L.). Sci. Rep. 2016, 6, 32289. [Google Scholar] [CrossRef] [PubMed]
- Nakajima, I.; Ban, Y.; Azuma, A.; Onoue, N.; Moriguchi, T.; Yamamoto, T.; Toki, S.; Endo, M. CRISPR/Cas9-mediated targeted mutagenesis in grape. PLoS ONE 2017, 12, e0177966. [Google Scholar] [CrossRef]
- Ren, F.; Ren, C.; Zhang, Z.; Duan, W.; Lecourieux, D.; Li, S.; Liang, Z. Efficiency Optimization of CRISPR/Cas9-Mediated Targeted Mutagenesis in Grape. Front. Plant. Sci. 2019, 10, 612. [Google Scholar] [CrossRef] [PubMed]

| Application | Molecular Marker * | References |
|---|---|---|
| Genetic diversity population structure | RAPD, ISSR, SSR, retrotransposon-based markers, SRAP, SNP, RAMP, REMAP, and IRAP | [49,50,82,83,84,85,86,87,88] |
| Cultivars, rootstocks, and clone identification | RAPD, cpSSR; SCAR; RFLP, SSR, SNP, AFLP, SAMPL, M-AFLP, MSAP, CAPS, IRAP, REMAP, SSAP, EST, and retrotransposon-based markers | [31,38,45,47,89,90,91,92,93,94,95,96,97,98] |
| Synonymies and homonymies clarification | RAPD and SSR | [99,100,101] |
| Origins of cultivated grapevine/phylogeographic patterns | cpSSR, SSR, and SNP | [27,53,57,60,62,102,103,104] |
| Genetic linkage maps | RFLP, SRAP, and SNP | [105,106] |
| Disease diagnostics | RFLP, SCAR, SSR, SSCP, ITS, and RNA sequencing | [48,80,107,108,109,110] |
| Transcriptome analysis and new gene discovery | EST | [54,55,111] |
| Genetic stability and somaclonal variation | RAPD, AFLP, SSR, CDDP, ISSR, and MSAP | [112,113,114,115,116,117] |
| QTL mapping | RAPD, CAPS, AFLP, SCAR, SSR, SNP, RAD sequencing, rhAmpSeq markers, SLAF-seq, and sequencing | [70,74,75,81,118,119,120,121,122,123,124,125,126,127] |
| Method | Cultivar | Target Gene | Trait | References |
|---|---|---|---|---|
| Transient expression assays using direct transformation methods, modified from Jelly et al. [231] | ||||
| Biolistics | Cabernet Sauvignon | VvAdh1, VvAdh2 VvAdh2 | Abiotic stress | [232,233] |
| Biolistics | Chardonnay | VvMYBA1, -F1, -PA1, -PA2 and VvCHS1, VvCHS2, VvCHS3 promoters | Flavonoid synthesis | [234] |
| Biolistics | Chardonnay | VvMYB5a,-5b and VvANR, VvANS, VvCHI, VvF30 50 H, VvLAR1 promoters | Flavonoid synthesis | [235] |
| Biolistics | Chardonnay | VvMYBA1, -A2 and VvUFGT promoter Flavonoids GFP, dual Luc Walker et al. [225] VvMYBF1 and VvANR, VvCHI, VvFLS1, VvLDOX promoters | Flavonoid synthesis | [236] |
| Biolistics | Chardonnay | VvMYBF1 and VvANR, VvCHI, VvFLS1, VvLDOX promoters | Flavonoid synthesis | [237] |
| Biolistics | Chardonnay | VvMYBPA1 and VvANR, VvCHI, VvF30 50 H, VvLAR1, VvLDOX promoters | Flavonoid synthesis | [238] |
| Biolistics | Chardonnay | VvMYC1 and VvMYB5a, -5b, -A1, -A2, -PA1 and VvANR, VvCHI, VvMYC1, VvUFGT promoters | Flavonoid synthesis | [239] |
| Biolistics | Chardonnay Pinot Noir | VvMYB14, -15 and VvSTS29, -41 promoters | Stilbene synthesis | [240] |
| Biolistics | Chardonnay | VvPGIP1 promoter | Resistance to B. cinerea | [241] |
| Biolistics | Thompson Seedless | VvPGIP1 promoter | Resistance to B. cinerea | [241] |
| PEG treatment | Cabernet Sauvignon | VvMSA | Abiotic stress | [242] |
| PEG treatment | Cabernet Sauvignon | VvWRKY1 and VvJAZ1.1, VvLOX promoters | JA defence pathway | [243] |
| PEG treatment | Cabernet Sauvignon | VvMYC1 | Flavonoid synthesis | [239] |
| PEG treatment | Cabernet Sauvignon | VvMSA | Abiotic stress | [244] |
| Transient expression assays using Agrobacterium-mediated transformation, modified from Jelly et al. [220]; Zhang et al. [17] | ||||
| Agrobacterium | Superior Seedless | hpRNA against VvPDS | Gene silencing | [245] |
| Agrobacterium | Cabernet Franc, Syrah, Zinfandel | GLRaV-2 cDNA | - | [208] |
| Agrobacterium | Thompson Seedless | D4E1 (synthetic AMP) | Resistance to A. vitis, X. ampelinus | [246] |
| Agrobacterium | Cabernet Franc | hpRNA against VvPGIP1 | Resistance to B. cinerea | [247] |
| Agrobacterium | Cabernet Sauvignon, Cinsault, Muscat Ottonel, Syrah | VvVST1 | Resistance to P. viticola | [248] |
| Agrobacterium | Carignane | VpGLOX | Resistance to E. necator | [249] |
| Agrobacterium | VpPR10.2 | Resistance to P. viticola | [250] | |
| Agrobacterium | VpSTS | Resistance to E. necator | [251] | |
| Agrobacterium | VpPR10.1 | Resistance to P. viticola | [252] | |
| Agrobacterium | Syrah | VvNPR1 | Resistance to P. viticola | [253] |
| Agrobacterium | Grenache | GLRaV-2 cDNA | - | [254] |
| Agrobacterium | Prime | GVA cDNA | - | [207] |
| Agrobacterium | Prime, Thompson Seedless | GRSPaV cDNA | - | [209] |
| Agrobacterium | Gamay Red | VvDFR | Flavonoid synthesis | [255] |
| Agrobacterium | Chardonnay | amiRNAs against Grapevine fanleaf virus and GUS sensor | Resistance to GFLV | [256] |
| Agrobacterium | Thompson Seedless | CaMV35S, CsVMV, Arabidopsis ACT2 promoters | - | [257] |
| Agrobacterium | Thompson Seedless | BDDPs with CaMV35S, CsVMV promoters and enhancers | - | [213] |
| Agrobacterium | Thompson Seedless | 31 grapevine promoters (PR1, PAL, Ubiquitin etc.) | - | [217] |
| Agrobacterium | Russalka and Rupestris du lot | GFLV CP (grape fanleaf virus coat protein) and four encoding antifreeze proteins (Atf11, Atf62, Atf78, B5) for Russalka, GUS (β-glucuronidase gene) for Rupestris du Lot | Fan Leaf Virus resistance and cold resistance | [258] |
| Agrobacterium | Chancellor | tfdA gene (a 2,4-D α-ketoglutarate dioxygenase) | Tolerance to 2,4-D | [259] |
| Agrobacterium | Pusa Seedless | A rice chitinase gene | Resistance to powdery mildew | [260] |
| Agrobacterium | Thompson Seedless | vvtl–1 (a thaumatin-like protein) | Broad spectrum fungal disease resistance | [261] |
| Agrobacterium | Thompson Seedless | VvMybA1 (regulatory gene for the last metabolic step of anthocyanin biosynthesis) | Development of an anthocyanin-based quantitative reporter system | [213] |
| Agrobacterium | Crimson Seedless | Chitinase and β-1,3-glucanase genes | Tolerance to downy mildew | [262] |
| Agrobacterium | Thompson Seedless | VpPUB23 (a ubiquitin ligase gene) | Overexpression of VpPUB23 decreased powdery mildew resistance | [225] |
| Agrobacterium | Chardonnay | VpSTSgDNA2 | Developed a protocol and increased powdery mildew resistance | [263] |
| Agrobacterium | Thompson Seedless and Freedom | LIMA-A (a synthetic gene encoding lytic peptide) | Durable Pierce’s disease resistance | [264] |
| Agrobacterium | Thompson Seedless; Red Globe | VqSTS6 in Thompson Seedless; VpPR4–1 (pathogenesis-related proteins) in Red globe | Resistance to powdery mildew | [265,266] |
| Agrobacterium | Brachetto | Knockdown through RNA interference of VvMLO6, 7, 11 and 13 (mildew locus O) | Reduced susceptibility to powdery mildew | [267] |
| Agrobacterium | Thompson Seedless | Overexpression of VaTLP (thaumatin-like protein) | Resistance to downy mildew | [268] |
| Agrobacterium | Thompson Seedless | Overexpression of VaPUB (a stress-responsive U-box protein gene) | Disease resistance | [269] |
| Agrobacterium | Thompson Seedless | Overexpression of VpPR10.1 | Resistance to downy mildew | [270,271] |
| Agrobacterium | Thompson Seedless | Overexpression of AgNHX1 (Na+/H+ antiporter gene in Atriplex gmelini) | Increased salt tolerance | [272] |
| Agrobacterium | Thompson Seedless | Overexpression of VvWRKY8 | Repressed VvSTS15/21 expression and resveratrol biosynthesis | [273] |
| Agrobacterium | Thompson Seedless | Overexpression of VpSTS29/STS2 | Resistance to powdery mildew | [274] |
| Agrobacterium | Thompson Seedless | Overexpression of VlbZIP30 (a basic region/leucine zipper transcription factor) | Improved drought resistance | [275] |
| Technology | Cultivar | Target Gene | Trait | References |
|---|---|---|---|---|
| CRISPR/Cas9 | Chardonnay | MLO-7 | Resistance to powdery mildew | [289] |
| CRISPR/Cas9 | Neo Muscat | VvPDS | Albino phenotype | [294] |
| CRISPR/Cas9 | Chardonnay, 41B | VvPDS | Albino phenotype and dwarf morphology | [295] |
| CRISPR/Cas9 | Chardonnay | L-idonate dehydrogenase gene (IdnDH) | Biosynthesis of tartaric acid | [293] |
| CRISPR/Cas9 | Neo Muscat | VvPDS (phytoene desaturase gene) | CRISPR-Cas9-mediated protocol development | [294] |
| CRISPR/Cas9 | Thompson Seedless | VvWRKY52 | Resistance to B. cinerea | [227] |
| CRISPR/Cas9 | Thompson Seedless | VvPR4b | VvPR4b knockout decreased downy mildew resistance | [291] |
| Thompson Seedless | VvMLO3 and VvMLO4 | Resistance to powdery mildew | [293] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Butiuc-Keul, A.; Coste, A. Biotechnologies and Strategies for Grapevine Improvement. Horticulturae 2023, 9, 62. https://doi.org/10.3390/horticulturae9010062
Butiuc-Keul A, Coste A. Biotechnologies and Strategies for Grapevine Improvement. Horticulturae. 2023; 9(1):62. https://doi.org/10.3390/horticulturae9010062
Chicago/Turabian StyleButiuc-Keul, Anca, and Ana Coste. 2023. "Biotechnologies and Strategies for Grapevine Improvement" Horticulturae 9, no. 1: 62. https://doi.org/10.3390/horticulturae9010062
APA StyleButiuc-Keul, A., & Coste, A. (2023). Biotechnologies and Strategies for Grapevine Improvement. Horticulturae, 9(1), 62. https://doi.org/10.3390/horticulturae9010062






