Evaluation of Probiotic Strains Isolated from Artemisia argyi Fermentation Liquor and the Antagonistic Effect of Lactiplantibacillus plantarum against Pathogens
Abstract
1. Introduction
2. Materials and Methods
2.1. Isolation and Identification of Strains from AAFL
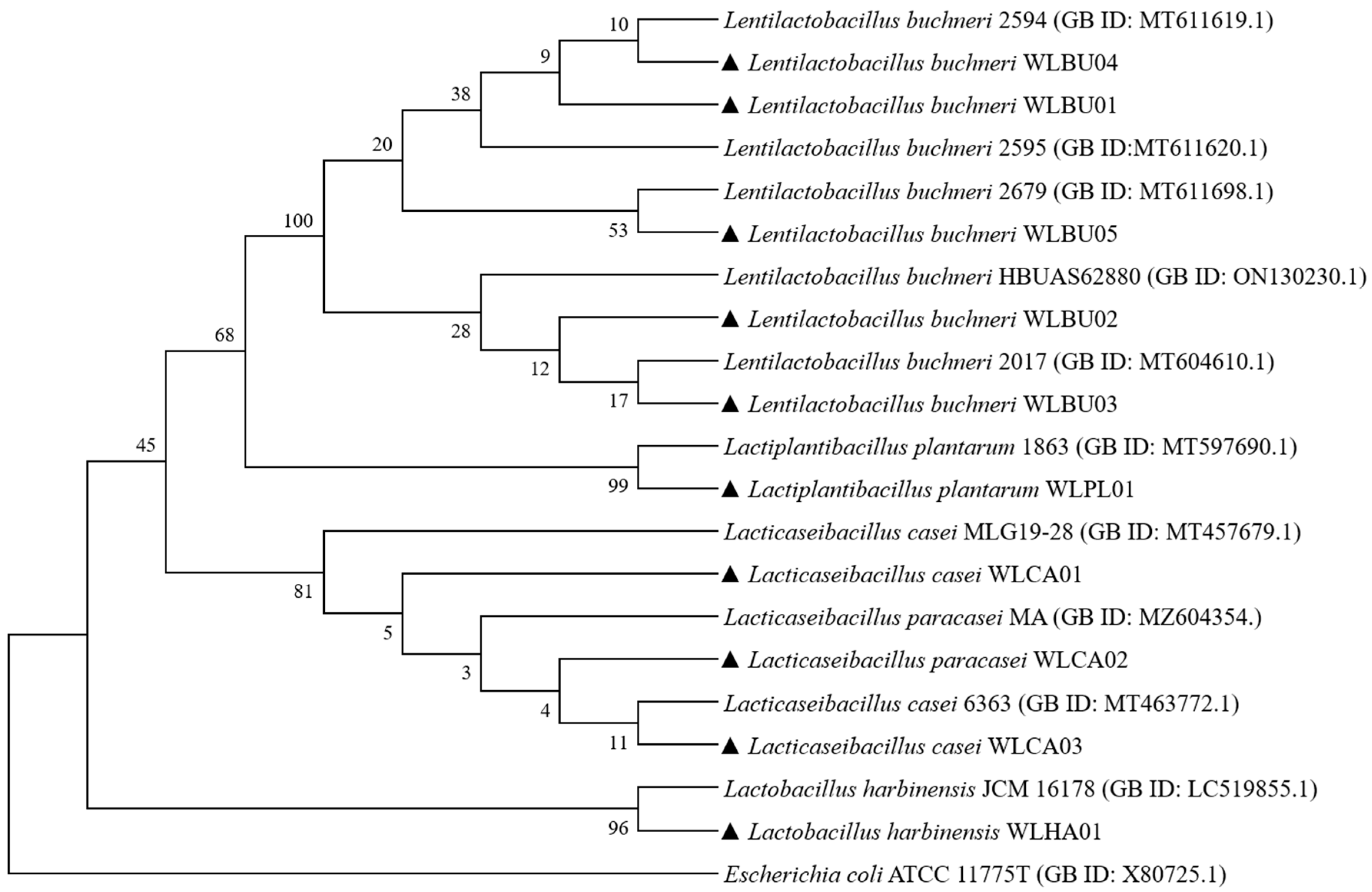
2.2. Phylogenetic Analysis
2.3. Growth Curve and Acid Production
2.4. Antimicrobial Activity Assay
2.5. Determination of Antibiotic Susceptibility
2.6. Detection of Biogenic Amine Production
2.7. Growth Characteristics of Strains in the Presence of Bile Salts and Acid
2.8. Survival of Strains under Simulated Gastric and Intestinal Fluid
2.9. Adhesive Ability of Strains to Caco-2 Cells
2.10. Preparation of AAFL-LP
2.11. Antimicrobial Activity of AAFL-LP by Oxford Cup Assay
2.12. Genomic DNA Extraction and Genome Analysis
2.13. Identification of Bacteriocin-Encoding Genes of L. plantarum WLPL01
2.14. Statistical Analysis
3. Results
3.1. Isolation and Identification of Strains from AAFL
3.2. Growth Curve and Acid Production of Strains
3.3. Antibiotic Sensitivity and Biogenic Amine Test of Strains
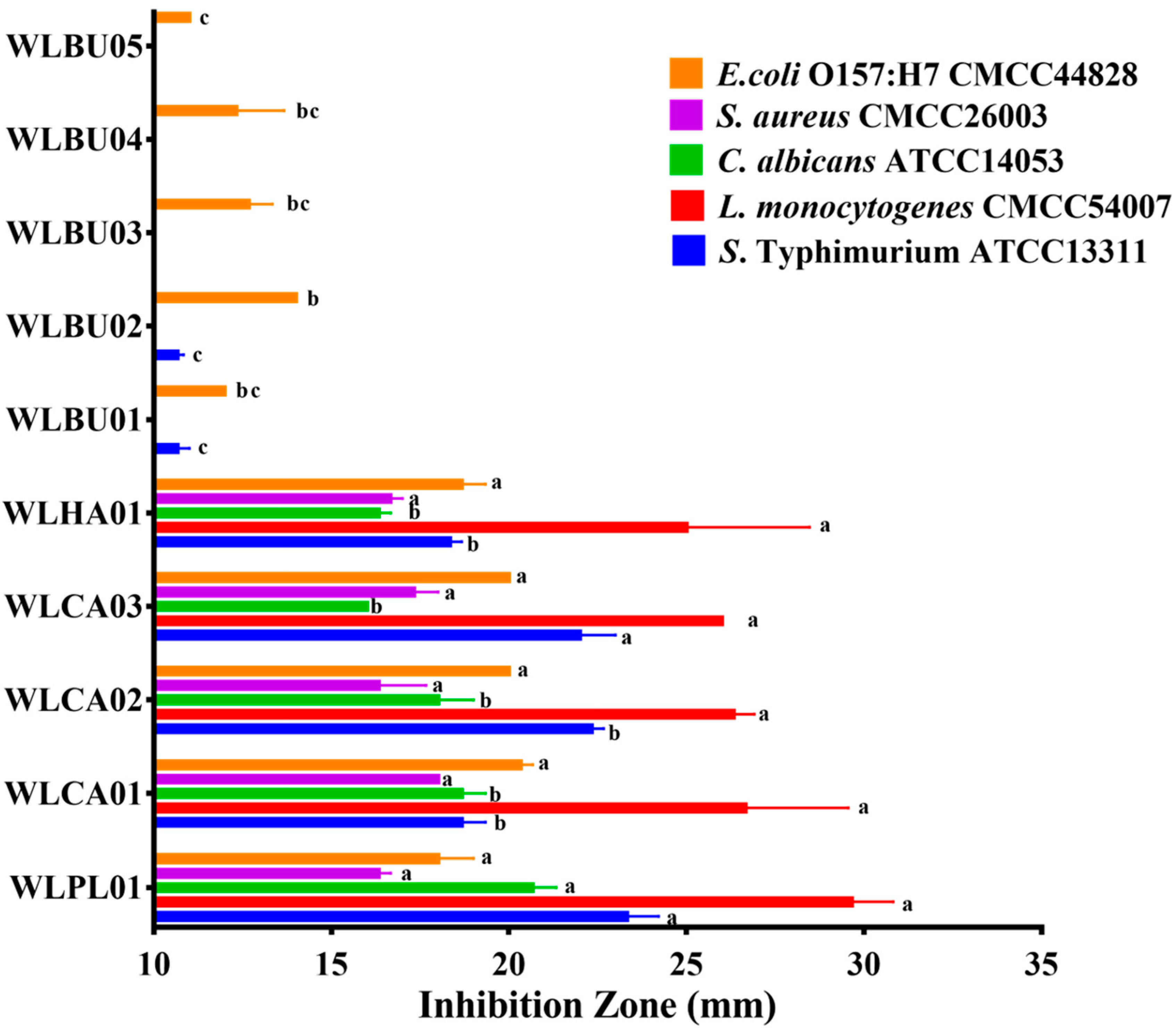
3.4. Antimicrobial Activity of Strains
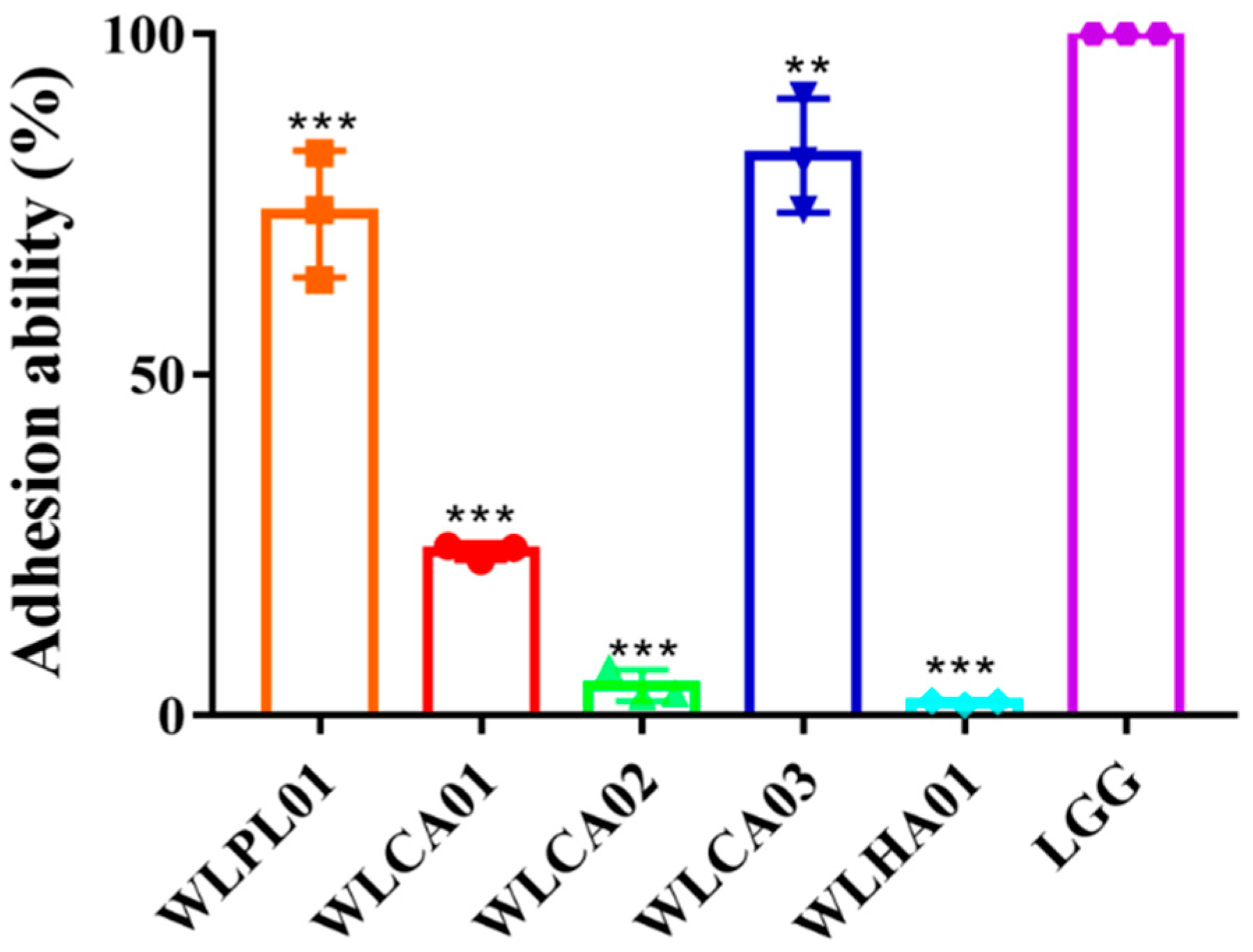
3.5. Survival of Strains in Mimic GI and Adhesion to Caco-2 Cells
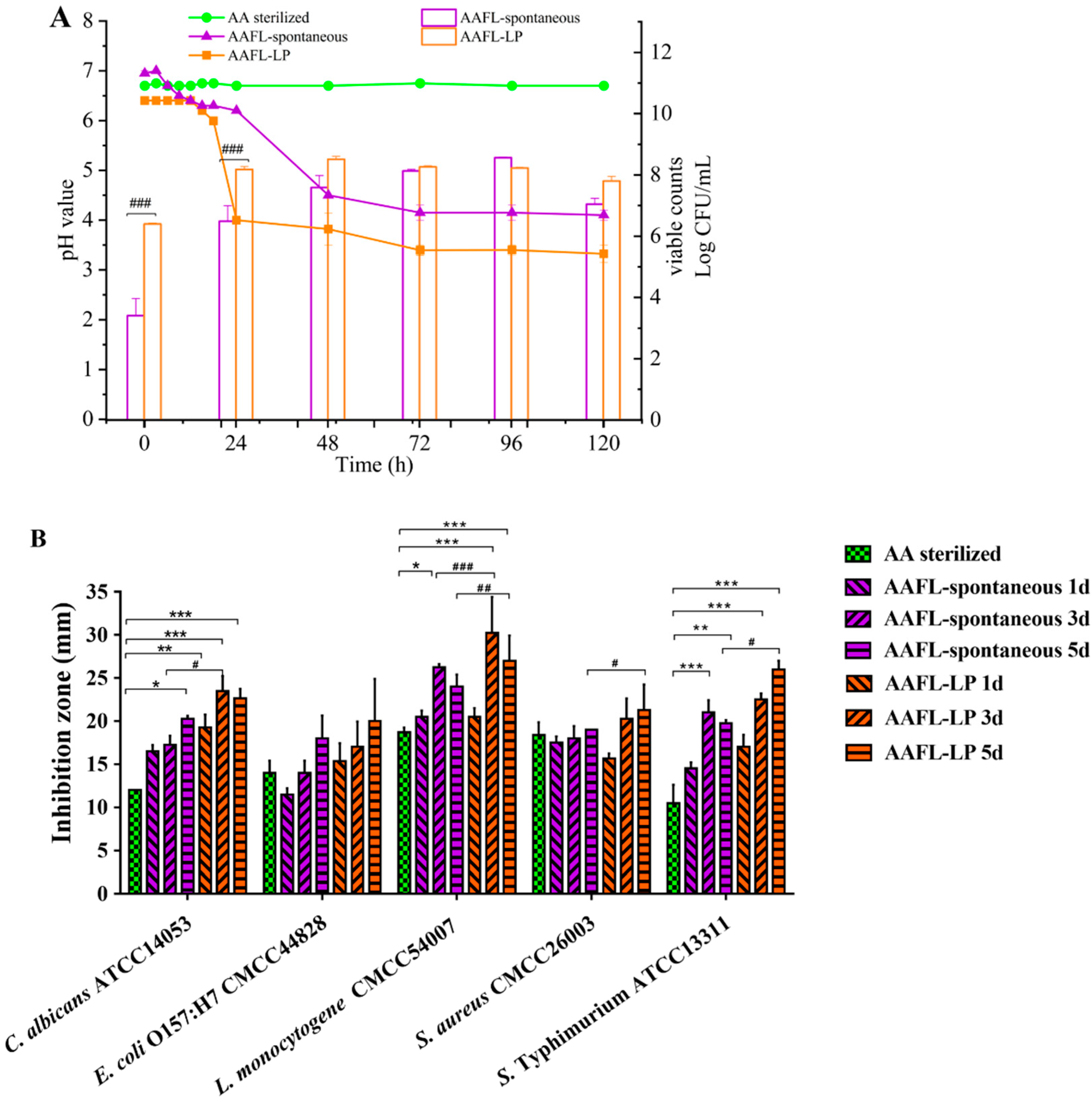
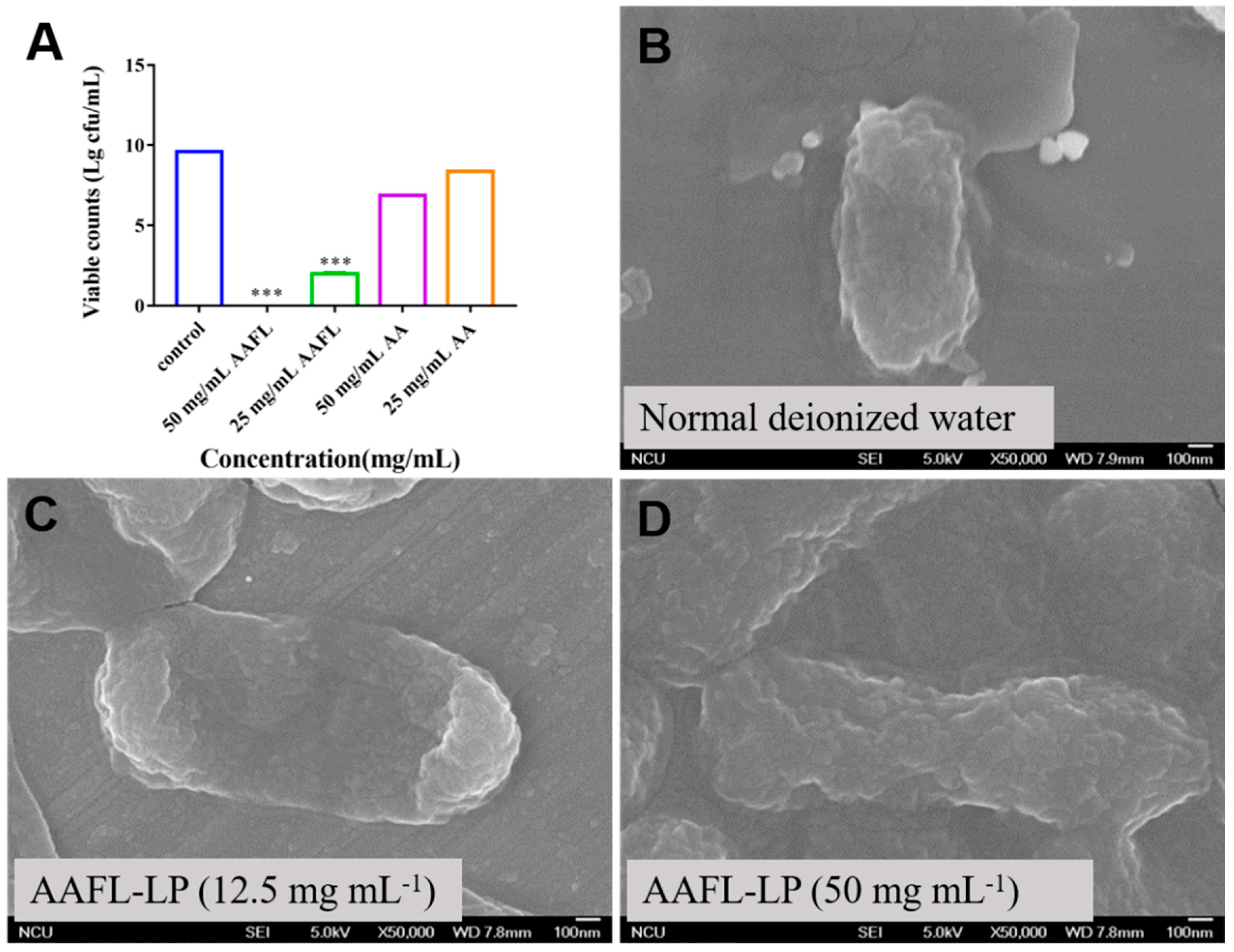
3.6. Antimicrobial Activity of L. plantarum WLPL01-Fermented AAFL (AAFL-LP) against Pathogens
3.7. Genome Analysis and Bacteriocin Identification of L. plantarum WLPL01
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Cai, Y.; Benno, Y.; Ogawa, M.; Kumai, S. Effect of Applying Lactic Acid Bacteria Isolated from Forage Crops on Fermentation Characteristics and Aerobic Deterioration of Silage. J. Dairy Sci. 1999, 82, 520–526. [Google Scholar] [CrossRef]
- Zimmermann-Klemd, A.M.; Reinhardt, J.K.; Morath, A.; Schamel, W.W.; Steinberger, P.; Leitner, J.; Huber, R.; Hamburger, M.; Gründemann, C. Immunosuppressive Activity of Artemisia argyi Extract and Isolated Compounds. Front. Pharmacol. 2020, 11, 402. [Google Scholar] [CrossRef]
- Kim, J.H.; Kim, H.-K.; Jeon, S.B.; Son, K.-H.; Kim, E.H.; Kang, S.K.; Sung, N.-D.; Kwon, B.-M. New sesquiterpene—Monoterpene lactone, artemisolide, isolated from Artemisia argyi. Tetrahedron Lett. 2002, 43, 6205–6208. [Google Scholar] [CrossRef]
- Bao, X.; Yuan, H.; Wang, C.; Liu, J.; Lan, M. Antitumor and immunomodulatory activities of a polysaccharide from Artemisia argyi. Carbohydr. Polym. 2013, 98, 1236–1243. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Li, J.; Sun, J.; Zeng, K.-W.; Cui, J.-R.; Jiang, Y.; Tu, P.-F. NO inhibitory guaianolide-derived terpenoids from Artemisia argyi. Fitoterapia 2013, 85, 169–175. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.-L.; Zhang, H.-J.; Chao, J.; Liu, J.-F. Essential oil of Artemisia argyi suppresses inflammatory responses by inhibiting JAK/STATs activation. J. Ethnopharmacol. 2017, 204, 107–117. [Google Scholar] [CrossRef]
- Kim, C.H.; Kim, G.-B.; Chang, M.B.; Bae, G.S.; Paik, I.K.; Kil, D.Y. Effect of dietary supplementation of Lactobacillus-fermented Artemisia princeps on growth performance, meat lipid peroxidation, and intestinal microflora in Hy-line Brown male chickens. Poult. Sci. 2017, 91, 2845–2851. [Google Scholar] [CrossRef]
- Wang, Z.; Tan, Z.; Wu, G.; Wang, L.; Qin, G.; Wang, Y.; Pang, H. Microbial community and fermentation characteristic of whole-crop wheat silage treated by lactic acid bacteria and Artemisia argyi during ensiling and aerobic exposure. Front. Microbiol. 2022, 13, 1004495. [Google Scholar] [CrossRef]
- Jiménez-López, J.; Ruiz-Medina, A.; Ortega-Barrales, P.; Llorent-Martínez, E. Phytochemical profile and antioxidant activity of caper berries (Capparis spinosa L.): Evaluation of the influence of the fermentation process. Food Chem. 2018, 250, 54–59. [Google Scholar] [CrossRef]
- Wang, L.; Lu, W.; Wu, Y.; Wu, Z. Impact of fermentation degree on phenolic compositions and bioactivities during the fermentation of guava leaves with Monascus anka and Bacillus sp. J. Funct. Foods 2018, 41, 183–190. [Google Scholar] [CrossRef]
- Petrova, P.M.; Petrov, K.K. Antimicrobial Activity of Starch-Degrading Lactobacillus Strains Isolated from Boza. Biotechnol. Biotechnol. Equip. 2011, 25, 114–116. [Google Scholar] [CrossRef]
- Oguntoyinbo, F.A.; Narbad, A. Multifunctional properties of Lactobacillus plantarum strains isolated from fermented cereal foods. J. Funct. Foods 2015, 17, 621–631. [Google Scholar] [CrossRef]
- Di Gioia, D.; Mazzola, G.; Nikodinoska, I.; Aloisio, I.; Langerholc, T.; Rossi, M.; Raimondi, S.; Melero, B.; Rovira, J. Lactic acid bacteria as protective cultures in fermented pork meat to prevent Clostridium spp. growth. Int. J. Food Microbiol. 2016, 235, 53–59. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.-E.; Jang, J.-Y.; Lee, J.-H.; Park, H.-W.; Choi, H.-J.; Kim, T.-W. Starter Cultures for Kimchi Fermentation. J. Microbiol. Biotechnol. 2015, 25, 559–568. [Google Scholar] [CrossRef] [PubMed]
- Saelim, K.; Jampaphaeng, K.; Maneerat, S. Functional properties of Lactiplantibacillus plantarum S0/7 isolated fermented stinky bean (Sa Taw Dong) and its use as a starter culture. J. Funct. Foods 2017, 38, 370–377. [Google Scholar] [CrossRef]
- Bae, E.-A.; Min, S.-W.; Lee, B.; Kim, N.-J.; Baek, N.-I.; Han, E.-J.; Chung, H.-G.; Kim, N.-H. Antiasthmic effect of fermented Artemisia princeps in asthmic mice induced by ovalbumin. J. Microbiol. Biotechnol. 2007, 17, 1554–1557. [Google Scholar] [PubMed]
- Jo, Y.M.; Seo, H.; Kim, G.Y.; Cheon, S.W.; Kim, S.-A.; Park, T.S.; Hurh, B.-S.; Han, N.S. Lactobacillus pentosus SMB718 as a probiotic starter producing allyl mercaptan in garlic and onion-enriched fermentation. Food Funct. 2020, 11, 10913–10924. [Google Scholar] [CrossRef]
- Zhang, F.; Jiang, M.; Wan, C.; Chen, X.; Chen, X.; Tao, X.; Shah, N.P.; Wei, H. Screening probiotic strains for safety: Evaluation of virulence and antimicrobial susceptibility of enterococci from healthy Chinese infants. J. Dairy Sci. 2016, 99, 4282–4290. [Google Scholar] [CrossRef]
- Jiang, M.; Zhang, F.; Wan, C.; Xiong, Y.; Shah, N.P.; Wei, H.; Tao, X. Evaluation of probiotic properties of Lactobacillus plantarum WLPL04 isolated from human breast milk. J. Dairy Sci. 2016, 99, 1736–1746. [Google Scholar] [CrossRef]
- Argyri, A.A.; Zoumpopoulou, G.; Karatzas, K.A.G.; Tsakalidou, E.; Nychas, G.-J.E.; Panagou, E.Z.; Tassou, C.C. Selection of potential probiotic lactic acid bacteria from fermented olives by in vitro tests. Food Microbiol. 2013, 33, 282–291. [Google Scholar] [CrossRef]
- Moreno-Arribas, M.V.; Polo, M.C.; Jorganes, F.; Muñoz, R. Screening of biogenic amine production by lactic acid bacteria isolated from grape must and wine. Int. J. Food Microbiol. 2003, 84, 117–123. [Google Scholar] [CrossRef] [PubMed]
- García-Ruiz, A.; de Llano, D.G.; Esteban-Fernández, A.; Requena, T.; Bartolomé, B.; Moreno-Arribas, M.V. Assessment of probiotic properties in lactic acid bacteria isolated from wine. Food Microbiol. 2014, 44, 220–225. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Jeong, H.; Lee, H.; Ahn, J. Assessment of cell surface properties and adhesion potential of selected probiotic strains. Lett. Appl. Microbiol. 2009, 49, 434–442. [Google Scholar] [CrossRef]
- Lindgreen, S. AdapterRemoval: Easy cleaning of next-generation sequencing reads. BMC Res. Notes 2012, 5, 337. [Google Scholar] [CrossRef] [PubMed]
- Luo, R.; Liu, B.; Xie, Y.; Li, Z.; Huang, W.; Yuan, J.; He, G.; Chen, Y.; Pan, Q.; Liu, Y.; et al. SOAPdenovo2: An empirically improved memory-efficient short-read de novo assembler. Gigascience 2012, 1, 18. [Google Scholar] [CrossRef]
- Bankevich, A.; Nurk, S.; Antipov, D.; Gurevich, A.A.; Dvorkin, M.; Kulikov, A.S.; Lesin, V.M.; Nikolenko, S.I.; Pham, S.; Prjibelski, A.D.; et al. SPAdes: A new genome assembly algorithm and its applications to single-cell sequencing. J. Comput. Biol. 2012, 19, 455–477. [Google Scholar] [CrossRef]
- Coil, D.; Jospin, G.; Darling, A.E. A5-miseq: An updated pipeline to assemble microbial genomes from Illumina MiSeq data. Bioinformatics 2014, 31, 587–589. [Google Scholar] [CrossRef]
- Koren, S.; Walenz, B.P.; Berlin, K.; Miller, J.R.; Bergman, N.H.; Phillippy, A.M. Canu: Scalable and accurate long-read assembly via adaptive k-mer weighting and repeat separation. Genome Res. 2017, 27, 722–736. [Google Scholar] [CrossRef]
- Walker, B.J.; Abeel, T.; Shea, T.; Priest, M.; Abouelliel, A.; Sakthikumar, S.; Cuomo, C.A.; Zeng, Q.; Wortman, J.; Young, S.K.; et al. Pilon: An Integrated Tool for Comprehensive Microbial Variant Detection and Genome Assembly Improvement. PLoS ONE 2014, 9, e112963. [Google Scholar] [CrossRef]
- Diep, D.B.; Straume, D.; Kjos, M.; Torres, C.; Nes, I.F. An overview of the mosaic bacteriocin pln loci from Lactobacillus plantarum. Peptides 2009, 30, 1562–1574. [Google Scholar] [CrossRef]
- Van Heel, A.J.; De Jong, A.; Montalbán-López, M.; Kok, J.; Kuipers, O.P. BAGEL3: Automated identification of genes encoding bacteriocins and (non-) bactericidal post-translationally modified peptides. Nucleic Acids Res. 2013, 41, 448–453. [Google Scholar] [CrossRef] [PubMed]
- Mundt, J.O.; Hammer, J.L. Lactobacilli on plants. Appl. Microbiol. 1968, 16, 1326–1330. [Google Scholar] [CrossRef] [PubMed]
- Casalta, E.; Montel, M.-C. Safety assessment of dairy microorganisms: The Lactococcus genus. Int. J. Food Microbiol. 2008, 126, 271–273. [Google Scholar] [CrossRef] [PubMed]
- Mathur, S.; Singh, R. Antibiotic resistance in food lactic acid bacteria—A review. Int. J. Food Microbiol. 2005, 105, 281–295. [Google Scholar] [CrossRef] [PubMed]
- Straub, B.W.; Kicherer, M.; Schilcher, S.M.; Hammes, W.P. The formation of biogenic amines by fermentation organisms. Z. Leb. Unters. Forsch. 1995, 201, 79–82. [Google Scholar] [CrossRef]
- Nishino, N.; Hattori, H.; Wada, H.; Touno, E. Biogenic amine production in grass, maize and total mixed ration silages inoculated with Lacticaseibacillus casei or Lactobacillus buchneri. J. Appl. Microbiol. 2007, 103, 325–332. [Google Scholar] [CrossRef]
- Ayad, E.; Nashat, S.; El-Sadek, N.; Metwaly, H.; El-Soda, M. Selection of wild lactic acid bacteria isolated from traditional Egyptian dairy products according to production and technological criteria. Food Microbiol. 2004, 21, 715–725. [Google Scholar] [CrossRef]
- Abad, M.J.; Bedoya, L.M.; Apaza, L.; Bermejo, P. The Artemisia L. Genus: A Review of Bioactive Essential Oils. Molecules 2012, 17, 2542–2566. [Google Scholar] [CrossRef]
- Wynne, A.G.; Mccartney, A.L.; Brostoff, J.; Hudspith, B.N.; Gibson, G.R. An in vitro assessment of the effects of broad-spectrum antibiotics on the human gut microflora and concomitant isolation of a Lactobacillus plantarum with anti-Candida activities. Anaerobe 2004, 10, 165–169. [Google Scholar] [CrossRef]
- Ennahar, S.; Aoude-Werner, D.; Sorokine, O.; Van Dorsselaer, A.; Bringel, F.; Hubert, J.-C.; Hasselmann, C. Production of pediocin AcH by Lactobacillus plantarum WHE 92 isolated from cheese. Appl. Environ. Microbiol. 1996, 62, 4381–4387. [Google Scholar] [CrossRef]
- Reenen, V.; Dicks, L.M.; Chikindas, M.L. Isolation, purification and partial characterization of plantaricin 423, a bacteriocin produced by Lactobacillus plantarum. J. Appl. Microbiol. 1998, 84, 1131–1137. [Google Scholar] [CrossRef]
- Liss, V.; Swart, A.L.; Kehl, A.; Hermanns, N.; Zhang, Y.; Chikkaballi, D.; Böhles, N.; Deiwick, J.; Hensel, M. Salmonella enterica Remodels the Host Cell Endosomal System for Efficient Intravacuolar Nutrition. Cell Host Microbe 2017, 21, 390–402. [Google Scholar] [CrossRef]
- Goel, A.; Halami, P.M.; Tamang, J.P. Genome Analysis of Lactobacillus plantarum Isolated from Some Indian Fermented Foods for Bacteriocin Production and Probiotic Marker Genes. Front. Microbiol. 2020, 11, 40. [Google Scholar] [CrossRef]
- Tai, H.F.; Foo, H.L.; Abdul Rahim, R.; Loh, T.C.; Abdullah, M.P.; Yoshinobu, K. Molecular characterization of new organisation of plnEF and plw loci of bacteriocin genes harbour concomitantly in Lactobacillus plantarum I-UL4. Microb. Cell Fact. 2015, 14, 89. [Google Scholar] [CrossRef]
- Rizzello, C.G.; Filannino, P.; Di Cagno, R.; Calasso, M.; Gobbetti, M. Quorum-Sensing Regulation of Constitutive Plantaricin by Lactobacillus plantarum Strains under a Model System for Vegetables and Fruits. Appl. Environ. Microbiol. 2014, 80, 777–787. [Google Scholar] [CrossRef]



| Strains | MICs a (μg/mL) | ||||||
|---|---|---|---|---|---|---|---|
| CHL | AMP | CIP | POLY | TET | ERY | GEN | |
| WLPL01 | 2 | 2 | 64 | 128 | 16 | ≤1 | 64 |
| WLCA01 | 2 | 2 | 64 | 512 | 1 | ≤1 | 128 R |
| WLCA02 | 2 | 2 | 128 | 512 | 1 | ≤1 | 128 R |
| WLCA03 | ≤1 | 2 | 128 | 256 | ≤1 | ≤1 | 32 |
| WLHA01 | 2 | 2 | 64 | 256 | 2 | ≤1 | 128 R |
| WLBU01 | 4 | 2 | 64 | 4 | 16 | ≤1 | ≤1 |
| WLBU02 | 8 | 2 | 128 | 4 | 32 R | ≤1 | ≤1 |
| WLBU03 | 4 | 2 | 128 | 4 | 32 R | ≤1 | ≤1 |
| WLBU04 | 4 | 2 | 128 | 8 | 32 R | ≤1 | ≤1 |
| WLBU05 | <1 | 2 | 128 | 4 | 32 R | ≤1 | ≤1 |
| Strain | Resistance to Bile Salt (%) | ||
|---|---|---|---|
| 0.15% | 0.30% | 0.45% | |
| WLPL01 | 11.90 ± 2.04 | 11.23 ± 2.15 | 12.01 ± 4.47 |
| WLCA01 | 14.43 ± 0.17 | 13.39 ± 0.42 | 12.50 ± 0.87 |
| WLCA02 | 14.50 ± 0.44 | 13.04 ± 0.42 | 11.81 ± 0.39 |
| WLCA03 | 13.35 ± 0.97 | 12.69 ± 0.62 | 11.73 ± 0.81 |
| WLHA01 | 10.34 ± 0.10 | 10.23 ± 0.17 | 9.56 ± 0.17 |
| Strain | L. plantarum WLPL01 | L. plantarum WCFS1 |
|---|---|---|
| Source | A. argyi fermentation liquor | Human saliva |
| Chromosomal (bp) | 3,142,471 | 3,308,274 |
| G + C Content (%) | 44.68% | 45.6% |
| Total number of genes | 2946 | 3174 |
| Coding genes | 2942 | 3063 |
| Total No. of RNA | 146 | 88 |
| No. of tRNA | 70 | 70 |
| No. of rRNA | 16 | 15 |
| No. of nc RNA | 60 | 3 |
| No. of CRISPER array | 0 | 0 |
| No. of prophage region | 8 | 4 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhan, H.; He, Y.; Wang, Q.; Lu, Q.; He, L.; Tao, X.; Wei, H. Evaluation of Probiotic Strains Isolated from Artemisia argyi Fermentation Liquor and the Antagonistic Effect of Lactiplantibacillus plantarum against Pathogens. Fermentation 2023, 9, 536. https://doi.org/10.3390/fermentation9060536
Zhan H, He Y, Wang Q, Lu Q, He L, Tao X, Wei H. Evaluation of Probiotic Strains Isolated from Artemisia argyi Fermentation Liquor and the Antagonistic Effect of Lactiplantibacillus plantarum against Pathogens. Fermentation. 2023; 9(6):536. https://doi.org/10.3390/fermentation9060536
Chicago/Turabian StyleZhan, Hui, Yao He, Qi’an Wang, Qingzi Lu, Lihua He, Xueying Tao, and Hua Wei. 2023. "Evaluation of Probiotic Strains Isolated from Artemisia argyi Fermentation Liquor and the Antagonistic Effect of Lactiplantibacillus plantarum against Pathogens" Fermentation 9, no. 6: 536. https://doi.org/10.3390/fermentation9060536
APA StyleZhan, H., He, Y., Wang, Q., Lu, Q., He, L., Tao, X., & Wei, H. (2023). Evaluation of Probiotic Strains Isolated from Artemisia argyi Fermentation Liquor and the Antagonistic Effect of Lactiplantibacillus plantarum against Pathogens. Fermentation, 9(6), 536. https://doi.org/10.3390/fermentation9060536





